NEW ZEALAND DATA SHEET. BETA Cream: A white, soft, homogeneous cream containing 0.1% w/w betamethasone as the valerate ester.
|
|
|
- Nickolas Stevenson
- 7 years ago
- Views:
Transcription
1 NEW ZEALAND DATA SHEET BETA Betamethasone Valerate Cream 0.1% w/w Betamethasone Valerate Ointment 0.1% w/w Presentation BETA Cream: A white, soft, homogeneous cream containing 0.1% w/w betamethasone as the valerate ester. BETA Ointment: A soft, smooth, translucent ointment containing 0.1% w/w betamethasone as the valerate ester. Indications BETA preparations are indicated for the treatment of: eczema, including atopic, infantile and discoid eczemas; prurigo nodularis; psoriasis (excluding widespread plaque psoriasis); neurodermatoses, including lichen simplex, lichen planus; seborrhoeic dermatitis; contact sensitivity reactions; discoid lupus erythematosus; insect bite reactions; prickly heat; and they may be used as an adjunct to systemic steroid therapy in generalised erythroderma. Dosage and Administration Adults, Elderly and Children over 1 year Apply thinly and gently rub in using only enough to cover the entire affected area once or twice daily for up to 4 weeks until improvement occurs, then reduce the frequency of the application or change the treatment to a less potent preparation. Allow adequate time for absorption after each application before applying an emollient. BETA Cream is especially appropriate for moist or weeping surfaces and BETA Ointment for dry, lichenified or scaly lesions, but this is not invariably so. In the more resistant lesions, such as the thickened plaques of psoriasis on the elbows and knees, the effect of BETA can be enhanced, if necessary, by occluding the treatment area with polythene film. Overnight occlusion only is usually adequate to bring about a satisfactory response in such lesions; thereafter, improvement can usually be maintained by regular application without occlusion. If the condition worsens or does not improve within 2 to 4 weeks, treatment and diagnosis should be reevaluated. Atopic dermatitis (eczema) Therapy with BETA preparations should be gradually discontinued once control is achieved and an emollient continued as maintenance therapy. Rebound of pre-existing dermatoses can occur with abrupt discontinuation of BETA preparations. Page 1 of 6
2 Recalcitrant dermatoses Patients who frequently relapse Once an acute episode has been treated effectively with a continuous course of topical corticosteroid, intermittent dosing (once daily, twice weekly, without occlusion) may be considered. This has been shown to be helpful in reducing the frequency of relapse. Application should be continued to all previously affected sites or to known sites of potential relapse. This regime should be combined with routine daily use of emollients. The condition and the benefits and risks of continued treatment must be re-evaluated on a regular basis. Children aged 1 year and over BETA preparations are contraindicated in children under one year of age (see Contraindications). Children are more likely to develop local and systemic side effects of topical corticosteroids and, in general, require shorter courses and less potent agents than adults. Care should be taken when using BETA preparations to ensure the amount applied is the minimum that provides therapeutic benefit. Elderly Clinical studies have not identified differences in responses between the elderly and younger patients. The greater frequency of decreased hepatic or renal function in the elderly may delay elimination if systemic absorption occurs. Therefore the minimum quantity should be used for the shortest duration to achieve the desired clinical benefit. Renal/Hepatic Impairment In case of systemic absorption (when application is over a large surface area for a prolonged period) metabolism and elimination may be delayed therefore increasing the risk of systemic toxicity. Therefore, the minimum quantity should be used for the shortest duration to achieve the desired clinical benefit. Contraindications The following conditions should not be treated with BETA preparations: Untreated cutaneous infections Rosacea Acne vulgaris Pruritus without inflammation Perianal and genital pruritus Hypersensitivity to any component of BETA preparations Perioral dermatitis. BETA preparations are contraindicated in dermatoses in infants under one year of age, including dermatitis. Warnings and Precautions Hypersensitivity BETA preparations should not be used in patients with a history of local hypersensitivity to corticosteroids or to any of the excipients in the preparation. Local hypersensitivity reactions (see Adverse Effects) may resemble symptoms of the condition under treatment. Reversible hypothalamic-pituitary-adrenal (HPA) axis suppression Manifestations of hypercortisolism (Cushing s syndrome) and reversible hypothalamic-pituitary-adrenal (HPA) axis suppression, leading to glucocorticosteroid insufficiency, can occur in some individuals as a result of increased systemic absorption of topical steroids. If either of the above are observed, withdraw the drug Page 2 of 6
3 gradually by reducing the frequency of application, or by substituting a less potent corticosteroid. Abrupt withdrawal of treatment may result in glucocorticosteroid insufficiency (see Adverse Effects). Risk factors for increased systemic effects are: Potency and formulation of topical steroid Duration of exposure Application to a large surface area Use on occluded areas of skin e.g. on intertriginous areas or under occlusive dressings (in infants the nappy may act as an occlusive dressing) Increasing hydration of the stratum corneum Use on thin skin areas such as the face Use on broken skin or other conditions where the skin barrier may be impaired In comparison with adults, children may absorb proportionally larger amounts of topical corticosteroids and thus be more susceptible to systemic adverse effects. This is because children have an immature skin barrier and a greater surface area to body weight ratio compared with adults. Use in children In infants and children under 12 years of age, long-term continuous topical corticosteroid therapy should be avoided where possible, as adrenal suppression can occur. Infection risk with occlusion Bacterial infection is encouraged by the warm, moist conditions within skin folds or caused by occlusive dressings. When using occlusive dressings, the skin should be cleansed before a fresh dressing is applied. Use in Psoriasis Topical corticosteroids should be used with caution in psoriasis as rebound relapses, development of tolerances, risk of generalised pustular psoriasis and development of local or systemic toxicity due to impaired barrier function of the skin have been reported in some cases. If used in psoriasis careful patient supervision is important. Application to the face Prolonged application to the face is undesirable as this area is more susceptible to atrophic changes. Application to the eyelids If applied to the eyelids, care is needed to ensure that the preparation does not enter the eye, as cataract and glaucoma might result from repeated exposure. Concomitant infection Appropriate antimicrobial therapy should be used whenever treating inflammatory lesions which have become infected. Any spread of infection requires withdrawal of topical corticosteroid therapy and administration of appropriate antimicrobial therapy. Chronic leg ulcers Topical corticosteroids are sometimes used to treat the dermatitis around chronic leg ulcers. However, this use may be associated with a higher occurrence of local hypersensitivity reactions and an increased risk of local infection. Fertility There are no data in humans to evaluate the effect of topical corticosteroids on fertility. Use in pregnancy There are limited data from the use of betamethasone valerate in pregnant women. Topical administration of corticosteroids to pregnant animals can cause abnormalities of foetal development. Page 3 of 6
4 The relevance of this finding to humans has not been established; however, administration of BETA preparations during pregnancy should only be considered if the expected benefit to the mother outweighs the risk to the foetus. The minimum quantity should be used for the minimum duration. Use in lactation The safe use of topical corticosteroids during lactation has not been established. It is not known whether topical administration of corticosteroids could result in sufficient systemic absorption to produce detectable amounts in breast milk. Administration of BETA preparations during lactation should only be considered if the expected benefit to the mother outweighs the risk to the infant. If used during lactation BETA preparations should not be applied to the breasts to avoid accidental ingestion by the infant. Effects on ability to drive and use machines There have been no studies to investigate the effect of betamethasone valerate on driving performance or the ability to operate machinery. A detrimental effect on such activities would not be anticipated from the adverse effects profile of topical betamethasone valerate. Adverse Effects Adverse events are listed below by MedDRA system organ class and frequency. Frequencies are defined as: very common ( 1/10), common ( 1/100 and <1/10), uncommon ( 1/1000 and <1/100), rare ( 1/10,000 and <1/1000) and very rare (<1/10,000) including isolated reports. Post-marketing data Infections and infestations Opportunistic infection. Immune system disorders Local hypersensitivity. If signs of hypersensitivity appear, application should stop immediately. Endocrine disorders Hypothalamic-pituitary-adrenal (HPA) axis suppression. Cushingoid features (e.g. moon face, central obesity), delayed weight gain/growth retardation in children, osteoporosis, glaucoma, hyperglycaemia/glucosuria, cataract, hypertension, increased weight/obesity, decreased endogenous cortisol levels, alopecia, trichorrhexis. Skin and subcutaneous tissue disorders Common: Pruritus, local skin burning / skin pain. Allergic contact dermatitis/dermatitis, erythema, rash, urticaria, pustular psoriasis, skin thinning*/ skin atrophy*, skin wrinkling*, skin dryness*, striae*, telangiectasias*, pigmentation changes*, hypertrichosis, exacerbation of underlying symptoms. General disorders and administration site conditions Application site irritation / pain. * Skin features secondary to local and / or systemic effects of HPA axis suppression. Interactions Co-administered medicines that can inhibit CYP3A4 (e.g. ritonavir, itraconazole) have been shown to inhibit the metabolism of corticosteroids leading to increased systemic exposure. The extent to which this interaction Page 4 of 6
5 is clinically relevant depends on the dose and route of administration of the corticosteroids and the potency of the CYP3A4 inhibitor. Overdosage Symptoms and signs Topically applied betamethasone valerate may be absorbed in sufficient amounts to produce systemic effects. Acute overdosage is unlikely to occur, however, in the case of chronic overdosage or misuse, the features of hypercortisolism may occur (see Adverse Effects). Treatment In the event of overdose, BETA preparations should be withdrawn gradually by reducing the frequency of application, or by substituting a less potent corticosteroid because of the risk of glucocorticosteroid insufficiency. Further management should be as clinically indicated or as recommended by the National Poisons Centre (telephone 0800 POISON or ). Pharmaceutical Precautions Store below 25 C. Medicine Classification Prescription Medicine. Package Quantities BETA Cream and BETA Ointment: Tubes of 30g, 50g or 100g. Not all pack sizes may be marketed. Further Information The least potent corticosteroid which will control the disease should be selected. Actions Betamethasone valerate is an active topical corticosteroid which produces a rapid response in those inflammatory dermatoses that are normally responsive to topical corticosteroid therapy, and is often effective in the less responsive conditions such as psoriasis. Pharmacokinetics As with other topical corticosteroids, sufficient betamethasone valerate may be absorbed to give systemic effects if applied under an occlusive dressing or when the skin is broken. Active Ingredient The active ingredient in BETA Cream and BETA Ointment is betamethasone valerate. Page 5 of 6
6 Chemical structure: Excipients Excipients in BETA Cream: white soft paraffin, cetostearyl alcohol, liquid paraffin, ceteareth 20, monobasic sodium phosphate, sodium hydroxide, purified water, and chlorocresol as preservative. Excipients in BETA Ointment: white soft paraffin, white beeswax, propylene glycol and stearyl alcohol. BETA Ointment and BETA Cream do not contain lanolin or parabens. Name and Address Mylan New Zealand Ltd PO Box Ellerslie AUCKLAND Telephone: Date of Preparation 24 April 2014 Page 6 of 6
NEW ZEALAND DATA SHEET BETNOVATE -C CREAM and OINTMENT
 NEW ZEALAND DATA SHEET BETNOVATE -C CREAM and OINTMENT Name of each active ingredient Betamethasone valerate 0.1% w/w and clioquinol 3% w/w Presentations BETNOVATE-C Cream is a smooth, straw-coloured water-miscible
NEW ZEALAND DATA SHEET BETNOVATE -C CREAM and OINTMENT Name of each active ingredient Betamethasone valerate 0.1% w/w and clioquinol 3% w/w Presentations BETNOVATE-C Cream is a smooth, straw-coloured water-miscible
ZOVIRAX Cold Sore Cream
 Data Sheet ZOVIRAX Cold Sore Cream Aciclovir 5% w/w Presentation Topical cream Indications ZOVIRAX Cold Sore Cream is indicated for the treatment of Herpes simplex virus infections of the lips and face
Data Sheet ZOVIRAX Cold Sore Cream Aciclovir 5% w/w Presentation Topical cream Indications ZOVIRAX Cold Sore Cream is indicated for the treatment of Herpes simplex virus infections of the lips and face
1g cream or ointment contains 1 mg methylprednisolone aceponate.
 CONSUMER MEDICINE INFORMATION ADVANTAN 1g cream or ointment contains 1 mg methylprednisolone aceponate. What is in this leaflet Please read this leaflet carefully before you start using ADVANTAN. It will
CONSUMER MEDICINE INFORMATION ADVANTAN 1g cream or ointment contains 1 mg methylprednisolone aceponate. What is in this leaflet Please read this leaflet carefully before you start using ADVANTAN. It will
Betnovate -C 0.1% / 3% w/w Cream Betamethasone (as valerate) and Clioquinol
 [GSK Logo] Package Leaflet: Information for the User Betnovate -C 0.1% / 3% w/w Cream Betamethasone (as valerate) and Clioquinol Read all of this leaflet carefully before you start using this medicine
[GSK Logo] Package Leaflet: Information for the User Betnovate -C 0.1% / 3% w/w Cream Betamethasone (as valerate) and Clioquinol Read all of this leaflet carefully before you start using this medicine
Topical Tacrolimus or Pimecrolimus for the treatment of mild, moderate or severe atopic eczema. Effective Shared Care Agreement
 Topical Tacrolimus or Pimecrolimus for the treatment of mild, moderate or severe atopic eczema. Effective Shared Care Agreement A Copy of this page signed by all three parties should be retained in the
Topical Tacrolimus or Pimecrolimus for the treatment of mild, moderate or severe atopic eczema. Effective Shared Care Agreement A Copy of this page signed by all three parties should be retained in the
NEW ZEALAND DATA SHEET NAPHCON-A Naphazoline hydrochloride and pheniramine maleate.
 NEW ZEALAND DATA SHEET NAPHCON-A Naphazoline hydrochloride and pheniramine maleate. PRESENTATION Eye Drops: NAPHCON-A Eye Drops are a combination of an antihistamine (pheniramine maleate) and a decongestant
NEW ZEALAND DATA SHEET NAPHCON-A Naphazoline hydrochloride and pheniramine maleate. PRESENTATION Eye Drops: NAPHCON-A Eye Drops are a combination of an antihistamine (pheniramine maleate) and a decongestant
PACKAGE LEAFLET: INFORMATION FOR THE USER. Elidel 10 mg/g Cream. pimecrolimus
 PACKAGE LEAFLET: INFORMATION FOR THE USER Elidel 10 mg/g Cream pimecrolimus Read all of this leaflet carefully before you start using Elidel cream Keep this leaflet. You may need to read it again. If you
PACKAGE LEAFLET: INFORMATION FOR THE USER Elidel 10 mg/g Cream pimecrolimus Read all of this leaflet carefully before you start using Elidel cream Keep this leaflet. You may need to read it again. If you
For Mild to Moderate Plaque Psoriasis and Moderate to Severe Atopic Dermatitis
 For Mild to Moderate Plaque Psoriasis and Moderate to Severe Atopic Dermatitis OLUX-E (clobetasol propionate) Foam, 0.05% Please see Important Safety Information on back page and accompanying Full Prescribing
For Mild to Moderate Plaque Psoriasis and Moderate to Severe Atopic Dermatitis OLUX-E (clobetasol propionate) Foam, 0.05% Please see Important Safety Information on back page and accompanying Full Prescribing
Public Assessment Report. Pharmacy to General Sales List Reclassification. Pirinase Hayfever Relief for Adults 0.05% Nasal Spray.
 Public Assessment Report Pharmacy to General Sales List Reclassification Pirinase Hayfever Relief for Adults 0.05% Nasal Spray (Fluticasone) PL 00079/0688 Glaxo Wellcome UK Limited TABLE OF CONTENTS Introduction
Public Assessment Report Pharmacy to General Sales List Reclassification Pirinase Hayfever Relief for Adults 0.05% Nasal Spray (Fluticasone) PL 00079/0688 Glaxo Wellcome UK Limited TABLE OF CONTENTS Introduction
Hydrozole Cream Hydrocortisone (microfine) 1% w/w and Clotrimazole 1% w/w
 CONSUMER MEDICINE INFORMATION What is in this leaflet? This leaflet answers some common questions about Hydrozole It does not contain all the available information. It does not take the place of talking
CONSUMER MEDICINE INFORMATION What is in this leaflet? This leaflet answers some common questions about Hydrozole It does not contain all the available information. It does not take the place of talking
NERIDERM Cream, Ointment, Fatty Ointment
 PRESCRIBING INFORMATION NERIDERM Cream, Ointment, Fatty Ointment Qualitative and Quantitive Composition 1 g Neriderm contains 1 mg (0.1%) diflucortolone valerate. Pharmaceutical Form Cream, Ointment, Fatty
PRESCRIBING INFORMATION NERIDERM Cream, Ointment, Fatty Ointment Qualitative and Quantitive Composition 1 g Neriderm contains 1 mg (0.1%) diflucortolone valerate. Pharmaceutical Form Cream, Ointment, Fatty
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE VETERINARY MEDICINAL PRODUCT Prednicare Tablets 5mg 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains: Active Substance(s) Prednisolone
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE VETERINARY MEDICINAL PRODUCT Prednicare Tablets 5mg 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains: Active Substance(s) Prednisolone
TOPICAL THERAPY. ADVANTAGES - increased dose of medication to affected area. - reduced systemic side effects and toxicity
 TOPICAL THERAPY ADVANTAGES - increased dose of medication to affected area. - reduced systemic side effects and toxicity DISADVANTAGES - takes time - may be greasy or messy - may have different preparations
TOPICAL THERAPY ADVANTAGES - increased dose of medication to affected area. - reduced systemic side effects and toxicity DISADVANTAGES - takes time - may be greasy or messy - may have different preparations
PHOSPHATE-SANDOZ Tablets (High dose phosphate supplement)
 1 PHOSPHATE-SANDOZ Tablets (High dose phosphate supplement) PHOSPHATE-SANDOZ PHOSPHATE-SANDOZ Tablets are a high dose phosphate supplement containing sodium phosphate monobasic. The CAS registry number
1 PHOSPHATE-SANDOZ Tablets (High dose phosphate supplement) PHOSPHATE-SANDOZ PHOSPHATE-SANDOZ Tablets are a high dose phosphate supplement containing sodium phosphate monobasic. The CAS registry number
PRODUCT MONOGRAPH. HydroVal. (Hydrocortisone Valerate USP) Cream & Ointment 0.2 % Topical Corticosteroid
 PRODUCT MONOGRAPH HydroVal (Hydrocortisone Valerate USP) Cream & Ointment 0.2 % Topical Corticosteroid TaroPharma Preparation Date: A Division of Taro Pharmaceuticals Inc. September 02, 2003 130 East Drive
PRODUCT MONOGRAPH HydroVal (Hydrocortisone Valerate USP) Cream & Ointment 0.2 % Topical Corticosteroid TaroPharma Preparation Date: A Division of Taro Pharmaceuticals Inc. September 02, 2003 130 East Drive
It can therefore be bought without a prescription but should be used correctly to ensure its efficacy and to reduce side effects.
 140 mg Medicated plaster Diclofenac Sodium BEFORE USE, CAREFULLY READ ALL THE INFORMATION GIVEN ON THE INFORMATIVE LEAFLET This medicinal product is intended for SELF-MEDICATION. It can be used to treat
140 mg Medicated plaster Diclofenac Sodium BEFORE USE, CAREFULLY READ ALL THE INFORMATION GIVEN ON THE INFORMATIVE LEAFLET This medicinal product is intended for SELF-MEDICATION. It can be used to treat
FastTest. You ve read the book... ... now test yourself
 FastTest You ve read the book...... now test yourself To ensure you have learned the key points that will improve your patient care, read the authors questions below. Please refer back to relevant sections
FastTest You ve read the book...... now test yourself To ensure you have learned the key points that will improve your patient care, read the authors questions below. Please refer back to relevant sections
The clinical studies have been performed in children, adolescents and adults, from 4 years up to 55 years of age.
 NAME OF THE MEDICINE TdaP-Booster. Diphtheria, tetanus and pertussis (acellular mono-component) vaccine (adsorbed, reduced antigen content). DESCRIPTION TdaP-Booster is a suspension for injection in pre-filled
NAME OF THE MEDICINE TdaP-Booster. Diphtheria, tetanus and pertussis (acellular mono-component) vaccine (adsorbed, reduced antigen content). DESCRIPTION TdaP-Booster is a suspension for injection in pre-filled
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Aknemycin Solution 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 10 g of solution contains 0.2 g of erythromycin. Structural formula of
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Aknemycin Solution 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 10 g of solution contains 0.2 g of erythromycin. Structural formula of
VISTARIL (hydroxyzine pamoate) Capsules and Oral Suspension
 VISTARIL (hydroxyzine pamoate) Capsules and Oral Suspension DESCRIPTION Hydroxyzine pamoate is designated chemically as 1-(p-chlorobenzhydryl) 4- [2-(2-hydroxyethoxy) ethyl] diethylenediamine salt of 1,1
VISTARIL (hydroxyzine pamoate) Capsules and Oral Suspension DESCRIPTION Hydroxyzine pamoate is designated chemically as 1-(p-chlorobenzhydryl) 4- [2-(2-hydroxyethoxy) ethyl] diethylenediamine salt of 1,1
PACKAGE LEAFLET: INFORMATION FOR THE USER. VITAMINE B12 STEROP 1mg/1ml Solution for injection / oral solution. Cyanocobalamin
 PACKAGE LEAFLET: INFORMATION FOR THE USER VITAMINE B12 STEROP 1mg/1ml Solution for injection / oral solution Cyanocobalamin Read all of this leaflet carefully before you start using this medicine because
PACKAGE LEAFLET: INFORMATION FOR THE USER VITAMINE B12 STEROP 1mg/1ml Solution for injection / oral solution Cyanocobalamin Read all of this leaflet carefully before you start using this medicine because
SUMMARY OF THE RISK MANAGEMENT PLAN (by medicinal product)
 PART VI SUMMARY OF THE RISK MANAGEMENT PLAN (by medicinal product) Format and content of the summary of the RMP The summary of the RMP part VI contains information based on RMP modules SI, SVIII and RMP
PART VI SUMMARY OF THE RISK MANAGEMENT PLAN (by medicinal product) Format and content of the summary of the RMP The summary of the RMP part VI contains information based on RMP modules SI, SVIII and RMP
Summary of Product Characteristics
 Summary of Product Characteristics 1 NAME OF THE MEDICINAL PRODUCT Isotrexin 2% + 0.05% w/w Gel 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Erythromycin Isotretinoin 2.00% w/w 0.05% w/w Excipients: Contains
Summary of Product Characteristics 1 NAME OF THE MEDICINAL PRODUCT Isotrexin 2% + 0.05% w/w Gel 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Erythromycin Isotretinoin 2.00% w/w 0.05% w/w Excipients: Contains
3% Sodium Chloride Injection, USP 5% Sodium Chloride Injection, USP
 PRESCRIBING INFORMATION 3% Sodium Chloride Injection, USP 5% Sodium Chloride Injection, USP IV Fluid and Electrolyte Replenisher Baxter Corporation Mississauga, Ontario L5N 0C2 Canada Date of Revision:
PRESCRIBING INFORMATION 3% Sodium Chloride Injection, USP 5% Sodium Chloride Injection, USP IV Fluid and Electrolyte Replenisher Baxter Corporation Mississauga, Ontario L5N 0C2 Canada Date of Revision:
DATA SHEET. Fluticasone propionate Inhaler (CFC-Free) (50, 125 or 250 micrograms per actuation). Qualitative and quantitative composition
 DATA SHEET FLIXOTIDE Inhaler Fluticasone propionate Inhaler (CFC-Free) (50, 125 or 250 micrograms per actuation). Qualitative and quantitative composition FLIXOTIDE 50 Inhaler is a pressurised metered-dose
DATA SHEET FLIXOTIDE Inhaler Fluticasone propionate Inhaler (CFC-Free) (50, 125 or 250 micrograms per actuation). Qualitative and quantitative composition FLIXOTIDE 50 Inhaler is a pressurised metered-dose
Ibuprofen 10% w/w gel Nurofen Maximum Strength 10 % Gel
 Ibuprofen 10% w/w gel Nurofen Maximum Strength 10 % Gel PL 10972/0089 UKPAR TABLE OF CONTENTS Lay Summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 11 Summary of Product Characteristics
Ibuprofen 10% w/w gel Nurofen Maximum Strength 10 % Gel PL 10972/0089 UKPAR TABLE OF CONTENTS Lay Summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 11 Summary of Product Characteristics
Adjunctive psychosocial intervention. Conditions requiring dose reduction. Immediate, peak plasma concentration is reached within 1 hour.
 Shared Care Guideline for Prescription and monitoring of Naltrexone Hydrochloride in alcohol dependence Author(s)/Originator(s): (please state author name and department) Dr Daly - Consultant Psychiatrist,
Shared Care Guideline for Prescription and monitoring of Naltrexone Hydrochloride in alcohol dependence Author(s)/Originator(s): (please state author name and department) Dr Daly - Consultant Psychiatrist,
White, circular, biconvex, uncoated tablets with a score line on one side, plain on the other.
 Nausicalm Cyclizine hydrochloride Ph. Eur. 50 mg Presentation White, circular, biconvex, uncoated tablets with a score line on one side, plain on the other. Uses Actions The active ingredient-cyclizine
Nausicalm Cyclizine hydrochloride Ph. Eur. 50 mg Presentation White, circular, biconvex, uncoated tablets with a score line on one side, plain on the other. Uses Actions The active ingredient-cyclizine
PRODUCT INFORMATION. Silver sulfadiazine is a sulfonamide and has broad antimicrobial activity against both Grampositive and Gram-negative organisms.
 PRODUCT INFORMATION Name of the Medicine: FLAMAZINE CREAM 1.0% w/w Silver sulfadiazine 1% w/w Composition: Active ingredient. Silver sulfadiazine. Excipients. Polysorbate 60 Ph. Eur, Polysorbate 80 Ph.
PRODUCT INFORMATION Name of the Medicine: FLAMAZINE CREAM 1.0% w/w Silver sulfadiazine 1% w/w Composition: Active ingredient. Silver sulfadiazine. Excipients. Polysorbate 60 Ph. Eur, Polysorbate 80 Ph.
PACKAGE LEAFLET: INFORMATION FOR THE USER. Alvesco 40, 80 and 160 mcg Inhaler Ciclesonide
 PACKAGE LEAFLET PACKAGE LEAFLET: INFORMATION FOR THE USER Alvesco 40, 80 and 160 mcg Inhaler Ciclesonide Read all of this leaflet carefully before you start using this medicine because it contains important
PACKAGE LEAFLET PACKAGE LEAFLET: INFORMATION FOR THE USER Alvesco 40, 80 and 160 mcg Inhaler Ciclesonide Read all of this leaflet carefully before you start using this medicine because it contains important
I B2.4. Design of the patient information leaflet for VariQuin
 (English translation of official Dutch version) I B2.4. Design of the patient information leaflet for VariQuin Information for the Patient: Read this package leaflet carefully when you have some time to
(English translation of official Dutch version) I B2.4. Design of the patient information leaflet for VariQuin Information for the Patient: Read this package leaflet carefully when you have some time to
What is Psoriasis? Common Areas Affected. Type Who Does it Affect Characteristics
 What is? is a term derived from the Greek word psōra which means itch and is a common, long lasting, inflammatory skin condition which affects 1-3% of the UK population and about 80 million people worldwide.
What is? is a term derived from the Greek word psōra which means itch and is a common, long lasting, inflammatory skin condition which affects 1-3% of the UK population and about 80 million people worldwide.
Maintenance of abstinence in alcohol dependence
 Shared Care Guideline for Prescription and monitoring of Acamprosate Calcium Author(s)/Originator(s): (please state author name and department) Dr Daly - Consultant Psychiatrist, Alcohol Services Dr Donnelly
Shared Care Guideline for Prescription and monitoring of Acamprosate Calcium Author(s)/Originator(s): (please state author name and department) Dr Daly - Consultant Psychiatrist, Alcohol Services Dr Donnelly
Leukocytoclastic Vasculitis and Stasis Dermatitis With Id Reaction
 Id Reaction December 01, 2007 By David L. Kaplan, MD [1] A Photo Quiz to Hone Dermatologic Skills Case 1: A slightly pruritic eruption developed on the lower legs of a 39-year-old woman after she had an
Id Reaction December 01, 2007 By David L. Kaplan, MD [1] A Photo Quiz to Hone Dermatologic Skills Case 1: A slightly pruritic eruption developed on the lower legs of a 39-year-old woman after she had an
Hypromellose Eye Drops BP 0.3% w/v PL 23097/0006
 Hypromellose Eye Drops BP 0.3% w/v PL 23097/0006 UKPAR TABLE OF CONTENTS Lay summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 9 Summary of product characteristics Page 10 Patient
Hypromellose Eye Drops BP 0.3% w/v PL 23097/0006 UKPAR TABLE OF CONTENTS Lay summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 9 Summary of product characteristics Page 10 Patient
Summary of the risk management plan (RMP) for Otezla (apremilast)
 EMA/741412/2014 Summary of the risk management plan (RMP) for Otezla (apremilast) This is a summary of the risk management plan (RMP) for Otezla, which details the measures to be taken in order to ensure
EMA/741412/2014 Summary of the risk management plan (RMP) for Otezla (apremilast) This is a summary of the risk management plan (RMP) for Otezla, which details the measures to be taken in order to ensure
COMMITTEE ON HERBAL MEDICINAL PRODUCTS (HMPC) FINAL COMMUNITY HERBAL MONOGRAPH ON HYPERICUM PERFORATUM L., HERBA (TRADITIONAL USE)
 European Medicines Agency Evaluation of Medicines for Human Use London, 12 November 2009 Doc. Ref.: EMEA/HMPC/745582/2009 COMMITTEE ON HERBAL MEDICINAL PRODUCTS (HMPC) FINAL COMMUNITY HERBAL MONOGRAPH
European Medicines Agency Evaluation of Medicines for Human Use London, 12 November 2009 Doc. Ref.: EMEA/HMPC/745582/2009 COMMITTEE ON HERBAL MEDICINAL PRODUCTS (HMPC) FINAL COMMUNITY HERBAL MONOGRAPH
FLAMAZINE CREAM MSDS No. 185
 SECTION 1 IDENTIFICATION OF THE MATERIAL AND SUPPLIER Product Product Name FLAMAZINE CREAM Other Names - Manufacturer s Product Code 66800531, 66800532, 66150153, 66150155 Use Topical antibacterial cream,
SECTION 1 IDENTIFICATION OF THE MATERIAL AND SUPPLIER Product Product Name FLAMAZINE CREAM Other Names - Manufacturer s Product Code 66800531, 66800532, 66150153, 66150155 Use Topical antibacterial cream,
Hull & East Riding Prescribing Committee
 Hull & East Riding Prescribing Committee Guideline on Prescribing of Gonadorelin (GnRH) Analogues and Progesterone Receptor Modulators in treatment of Endometriosis or prior to Endometrial Ablation, or
Hull & East Riding Prescribing Committee Guideline on Prescribing of Gonadorelin (GnRH) Analogues and Progesterone Receptor Modulators in treatment of Endometriosis or prior to Endometrial Ablation, or
See 17 for PATIENT COUNSELING INFORMATION. Revised: 3/2016 FULL PRESCRIBING INFORMATION: CONTENTS* WARNING: ADRENAL CRISIS IN THE SETTING OF SHOCK OR
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use LYSODREN safely and effectively. See full prescribing information for LYSODREN. LYSODREN (mitotane)
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use LYSODREN safely and effectively. See full prescribing information for LYSODREN. LYSODREN (mitotane)
TOPICAL TREATMENTS FOR PSORIASIS
 TOPICAL TREATMENTS FOR PSORIASIS What are the aims of this leaflet? Patients with psoriasis are usually treated with preparations that are applied to the skin. This leaflet has been written to help you
TOPICAL TREATMENTS FOR PSORIASIS What are the aims of this leaflet? Patients with psoriasis are usually treated with preparations that are applied to the skin. This leaflet has been written to help you
Stepping toward a different treatment option LEARN WHAT ACTHAR CAN DO FOR YOU
 FOR MS RELAPSES Stepping toward a different treatment option LEARN WHAT ACTHAR CAN DO FOR YOU As a person with multiple sclerosis (MS), you know firsthand the profound impact MS relapses can have on your
FOR MS RELAPSES Stepping toward a different treatment option LEARN WHAT ACTHAR CAN DO FOR YOU As a person with multiple sclerosis (MS), you know firsthand the profound impact MS relapses can have on your
The format of this leaflet was determined by the Ministry of Health and its content was checked and approved in May 2013.
 The format of this leaflet was determined by the Ministry of Health and its content was checked and approved in May 2013 Havrix 720 Junior 1 NAME OF THE MEDICINAL PRODUCT Havrix 720 Junior 2 QUALITATIVE
The format of this leaflet was determined by the Ministry of Health and its content was checked and approved in May 2013 Havrix 720 Junior 1 NAME OF THE MEDICINAL PRODUCT Havrix 720 Junior 2 QUALITATIVE
Learn More About Product Labeling
 Learn More About Product Labeling Product label The product label is developed during the formal process of review and approval by regulatory agencies of any medicine or medical product. There are specific
Learn More About Product Labeling Product label The product label is developed during the formal process of review and approval by regulatory agencies of any medicine or medical product. There are specific
NEW ZEALAND DATA SHEET
 NEW ZEALAND DATA SHEET BUTACORT Budesonide (50 µg & 100 µg per actuation) Aqueous Nasal Suspension Presentation Budesonide aqueous spray 50µg and 100µg per metered actuation. 50 µg/actuation: An almost
NEW ZEALAND DATA SHEET BUTACORT Budesonide (50 µg & 100 µg per actuation) Aqueous Nasal Suspension Presentation Budesonide aqueous spray 50µg and 100µg per metered actuation. 50 µg/actuation: An almost
EBMT Education Day for Nurses and AHPs April 2012 Skin care: not every rash is GVHD
 EBMT Education Day for Nurses and AHPs April 2012 Skin care: not every rash is GVHD Eileen Parry Consultant Dermatologist Tameside Hospital Foundation Trust Overview How to assess a patient with a rash
EBMT Education Day for Nurses and AHPs April 2012 Skin care: not every rash is GVHD Eileen Parry Consultant Dermatologist Tameside Hospital Foundation Trust Overview How to assess a patient with a rash
NEUROTONE THR 00904/0005 UKPAR
 NEUROTONE THR 00904/0005 UKPAR TABLE OF CONTENTS Lay summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 10 Summary of Product Characteristics Page 11 Product Information Leaflet
NEUROTONE THR 00904/0005 UKPAR TABLE OF CONTENTS Lay summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 10 Summary of Product Characteristics Page 11 Product Information Leaflet
CONTRAINDICATIONS. None (4).
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use Taclonex Ointment safely and effectively. See Full Prescribing Information for Taclonex Ointment.
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use Taclonex Ointment safely and effectively. See Full Prescribing Information for Taclonex Ointment.
Thioctacid 600 T Solution for Injection contains 600 mg alpha-lipoic acid
 Package Leaflet: Information for the User Thioctacid 600 T Solution for Injection contains 600 mg alpha-lipoic acid For use in adults Active substance: Alpha-lipoic acid, Trometamol salt (1:1) Read all
Package Leaflet: Information for the User Thioctacid 600 T Solution for Injection contains 600 mg alpha-lipoic acid For use in adults Active substance: Alpha-lipoic acid, Trometamol salt (1:1) Read all
X-Plain Psoriasis Reference Summary
 X-Plain Psoriasis Reference Summary Introduction Psoriasis is a long-lasting skin disease that causes the skin to become inflamed. Patches of thick, red skin are covered with silvery scales. It affects
X-Plain Psoriasis Reference Summary Introduction Psoriasis is a long-lasting skin disease that causes the skin to become inflamed. Patches of thick, red skin are covered with silvery scales. It affects
D.RIGOPOULOS, D.IOANNIDES,* D.KALOGEROMITROS, S.GREGORIOU AND A.KATSAMBAS
 British Journal of Dermatology 24; 151: 171 175. DOI: 1.1111/j.1365-2133.24.628.x Therapeutics Pimecrolimus cream 1% vs. betamethasone 17-valerate Æ1% cream in the treatment of seborrhoeic dermatitis.
British Journal of Dermatology 24; 151: 171 175. DOI: 1.1111/j.1365-2133.24.628.x Therapeutics Pimecrolimus cream 1% vs. betamethasone 17-valerate Æ1% cream in the treatment of seborrhoeic dermatitis.
Department of Dermatology, Churchill Hospital PUVA Treatment
 Oxford University Hospitals NHS Trust Department of Dermatology, Churchill Hospital PUVA Treatment information for patients CONTENTS What is PUVA 3 What conditions are treated with PUVA? 3 How is PUVA
Oxford University Hospitals NHS Trust Department of Dermatology, Churchill Hospital PUVA Treatment information for patients CONTENTS What is PUVA 3 What conditions are treated with PUVA? 3 How is PUVA
LEFLUNOMIDE (Adults)
 Shared Care Guideline DRUG: Introduction: LEFLUNOMIDE (Adults) Indication: Disease modifying drug for rheumatoid arthritis and psoriatic arthritis Licensing Information: Disease modifying drug for active
Shared Care Guideline DRUG: Introduction: LEFLUNOMIDE (Adults) Indication: Disease modifying drug for rheumatoid arthritis and psoriatic arthritis Licensing Information: Disease modifying drug for active
MEDICATION GUIDE. PROTOPIC [pro-top-ik] (tacrolimus) Ointment 0.03% Ointment 0.1%
![MEDICATION GUIDE. PROTOPIC [pro-top-ik] (tacrolimus) Ointment 0.03% Ointment 0.1% MEDICATION GUIDE. PROTOPIC [pro-top-ik] (tacrolimus) Ointment 0.03% Ointment 0.1%](/thumbs/27/10084771.jpg) MEDICATION GUIDE PROTOPIC [pro-top-ik] (tacrolimus) Ointment 0.03% Ointment 0.1% Read the Medication Guide every time you or a family member gets PROTOPIC Ointment. There may be new information. This Medication
MEDICATION GUIDE PROTOPIC [pro-top-ik] (tacrolimus) Ointment 0.03% Ointment 0.1% Read the Medication Guide every time you or a family member gets PROTOPIC Ointment. There may be new information. This Medication
PACKAGE LEAFLET: INFORMATION FOR THE USER. ADRENALINE (TARTRATE) STEROP 1 mg/1 ml Solution for injection. Adrenaline (Levorenine, Epinephrine)
 PACKAGE LEAFLET: INFORMATION FOR THE USER ADRENALINE (TARTRATE) STEROP 1 mg/1 ml Solution for injection Adrenaline (Levorenine, Epinephrine) Read all of this leaflet carefully before you start using this
PACKAGE LEAFLET: INFORMATION FOR THE USER ADRENALINE (TARTRATE) STEROP 1 mg/1 ml Solution for injection Adrenaline (Levorenine, Epinephrine) Read all of this leaflet carefully before you start using this
READ THIS FOR SAFE AND EFFECTIVE USE OF YOUR MEDICINE PATIENT MEDICATION INFORMATION. [new-ka la]
![READ THIS FOR SAFE AND EFFECTIVE USE OF YOUR MEDICINE PATIENT MEDICATION INFORMATION. [new-ka la] READ THIS FOR SAFE AND EFFECTIVE USE OF YOUR MEDICINE PATIENT MEDICATION INFORMATION. [new-ka la]](/thumbs/35/17235501.jpg) READ THIS FOR SAFE AND EFFECTIVE USE OF YOUR MEDICINE PATIENT MEDICATION INFORMATION Pr NUCALA [new-ka la] mepolizumab lyophilized powder for subcutaneous injection Read this carefully before you start
READ THIS FOR SAFE AND EFFECTIVE USE OF YOUR MEDICINE PATIENT MEDICATION INFORMATION Pr NUCALA [new-ka la] mepolizumab lyophilized powder for subcutaneous injection Read this carefully before you start
0.9% Sodium Chloride Injection, USP In VIAFLEX Plastic Container
 Page 1 of 8 PRESCRIBING INFORMATION 0.9% Sodium Chloride Injection, USP In VIAFLEX Plastic Container IV Fluid and Electrolyte Replenisher Baxter Corporation Mississauga, Ontario L5N 0C2 Canada Date of
Page 1 of 8 PRESCRIBING INFORMATION 0.9% Sodium Chloride Injection, USP In VIAFLEX Plastic Container IV Fluid and Electrolyte Replenisher Baxter Corporation Mississauga, Ontario L5N 0C2 Canada Date of
RETIRIDES - Information for the user and usage instructions Translated from Spanish by Vaughter Wellness Ltd.
 RETIRIDES - Information for the user and usage instructions Translated from Spanish by Vaughter Wellness Ltd. Table of Contents 1. Name of the product...2 2. Qualitative and quantitative composition...2
RETIRIDES - Information for the user and usage instructions Translated from Spanish by Vaughter Wellness Ltd. Table of Contents 1. Name of the product...2 2. Qualitative and quantitative composition...2
Felimazole 5 mg Coated Tablet
 .. CARTON TEXT - FRONT PANEL PRESCRIPTION ANIMAL REMEDY READ SAFETY DIRECTIONS BEFORE OPENING OR USING KEEP OUT OF REACH OF CHILDREN FOR ANIMAL TREATMENT ONLY RLP Approved Felimazole 5 mg Coated Tablet
.. CARTON TEXT - FRONT PANEL PRESCRIPTION ANIMAL REMEDY READ SAFETY DIRECTIONS BEFORE OPENING OR USING KEEP OUT OF REACH OF CHILDREN FOR ANIMAL TREATMENT ONLY RLP Approved Felimazole 5 mg Coated Tablet
Naltrexone Shared Care Guideline for the treatment of alcohol dependence and opioid dependance
 Naltrexone Shared Care Guideline for the treatment of alcohol dependence and opioid dependance Introduction Indication/Licensing information: Naltrexone is licensed for use as an additional therapy, within
Naltrexone Shared Care Guideline for the treatment of alcohol dependence and opioid dependance Introduction Indication/Licensing information: Naltrexone is licensed for use as an additional therapy, within
Dechra Veterinary Products Limited (A business unit of Dechra Pharmaceuticals PLC) Sansaw Business Park Hadnall, Shrewsbury Shropshire SY4 4AS
 Dechra Veterinary Products Limited (A business unit of Dechra Pharmaceuticals PLC) Sansaw Business Park Hadnall, Shrewsbury Shropshire SY4 4AS Tel: 01939 211200 Fax: 01939 211201 Email: info.uk@dechra.com
Dechra Veterinary Products Limited (A business unit of Dechra Pharmaceuticals PLC) Sansaw Business Park Hadnall, Shrewsbury Shropshire SY4 4AS Tel: 01939 211200 Fax: 01939 211201 Email: info.uk@dechra.com
P AC K AG E L E AF L E T: INFORMAT I ON FO R THE USER. 500 mg, film-coated tablet Active substance: metformin hydrochloride
 P AC K AG E L E AF L E T: INFORMAT I ON FO R THE USER Siofor 500 500 mg, film-coated tablet Active substance: metformin hydrochloride For use in children above 10 years and adults Read all of this leaflet
P AC K AG E L E AF L E T: INFORMAT I ON FO R THE USER Siofor 500 500 mg, film-coated tablet Active substance: metformin hydrochloride For use in children above 10 years and adults Read all of this leaflet
There is a risk of renal impairment in dehydrated children and adolescents.
 PACKAGE LEAFLET: INFORMATION FOR THE USER MELFEN 200mg FILM-COATED TABLETS MELFEN 400mg FILM-COATED TABLETS Ibuprofen Read all of this leaflet carefully before you start taking this medicine because it
PACKAGE LEAFLET: INFORMATION FOR THE USER MELFEN 200mg FILM-COATED TABLETS MELFEN 400mg FILM-COATED TABLETS Ibuprofen Read all of this leaflet carefully before you start taking this medicine because it
PRODUCT INFORMATION Stieva-A Creams 0.025% w/w, 0.05% w/w and 0.1% w/w
 NAME OF THE MEDICINE Stieva-A Cream PRODUCT INFORMATION Stieva-A Creams 0.025% w/w, 0.05% w/w and 0.1% w/w DESCRIPTION Chemical names: 3, 7-dimethyl-9-(2,6,6-trimethyl-1-cyclohexene-1-yl)-2,4,6,8-non-tetraenoic
NAME OF THE MEDICINE Stieva-A Cream PRODUCT INFORMATION Stieva-A Creams 0.025% w/w, 0.05% w/w and 0.1% w/w DESCRIPTION Chemical names: 3, 7-dimethyl-9-(2,6,6-trimethyl-1-cyclohexene-1-yl)-2,4,6,8-non-tetraenoic
VOLTAREN OPHTHA EYE DROPS
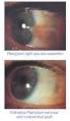 פורמט עלון זה נקבע ע"ימשרד הבריאות ותוכנו נבדק ואושר על ידו באפריל 2008 PRESCRIBING INFORMATION VOLTAREN OPHTHA EYE DROPS 1 Name of the medicinal product Voltaren Ophtha Eye Drops. 2 Qualitative and quantitative
פורמט עלון זה נקבע ע"ימשרד הבריאות ותוכנו נבדק ואושר על ידו באפריל 2008 PRESCRIBING INFORMATION VOLTAREN OPHTHA EYE DROPS 1 Name of the medicinal product Voltaren Ophtha Eye Drops. 2 Qualitative and quantitative
INITIATING ORAL AUBAGIO (teriflunomide) THERAPY
 FOR YOUR PATIENTS WITH RELAPSING FORMS OF MS INITIATING ORAL AUBAGIO (teriflunomide) THERAPY WARNING: HEPATOTOXICITY AND RISK OF TERATOGENICITY Severe liver injury including fatal liver failure has been
FOR YOUR PATIENTS WITH RELAPSING FORMS OF MS INITIATING ORAL AUBAGIO (teriflunomide) THERAPY WARNING: HEPATOTOXICITY AND RISK OF TERATOGENICITY Severe liver injury including fatal liver failure has been
Summary of the risk management plan (RMP) for Ionsys (fentanyl)
 EMA/764409/2015 Summary of the risk management plan (RMP) for Ionsys (fentanyl) This is a summary of the risk management plan (RMP) for Ionsys, which details the measures to be taken in order to ensure
EMA/764409/2015 Summary of the risk management plan (RMP) for Ionsys (fentanyl) This is a summary of the risk management plan (RMP) for Ionsys, which details the measures to be taken in order to ensure
SYNACTHEN DEPOT i.m. (tetracosactide hexaacetate) 1 mg/ml Suspension for injection
 SYNACTHEN DEPOT i.m. (tetracosactide hexaacetate) 1 mg/ml Suspension for injection Consumer Medicine Information What is in this leaflet Read all of this leaflet carefully before you are given SYNACTHEN
SYNACTHEN DEPOT i.m. (tetracosactide hexaacetate) 1 mg/ml Suspension for injection Consumer Medicine Information What is in this leaflet Read all of this leaflet carefully before you are given SYNACTHEN
SYNACTHEN i.m./i.v. tetracosactide hexaacetate
 New Zealand Consumer Medicine Information SYNACTHEN i.m./i.v. tetracosactide hexaacetate 250 micrograms/ml solution for injection or infusion What is in this leaflet This leaflet answers some common questions
New Zealand Consumer Medicine Information SYNACTHEN i.m./i.v. tetracosactide hexaacetate 250 micrograms/ml solution for injection or infusion What is in this leaflet This leaflet answers some common questions
Professor Andrew Wright,
 Professor Andrew Wright, Systemic treatments are drugs taken as tablets or injections that travel through the bloodstream, dampening down the immune system to reach and treat eczema all over the body.
Professor Andrew Wright, Systemic treatments are drugs taken as tablets or injections that travel through the bloodstream, dampening down the immune system to reach and treat eczema all over the body.
COMMITTEE ON HERBAL MEDICINAL PRODUCTS (HMPC) FINAL COMMUNITY HERBAL MONOGRAPH ON AVENA SATIVA L., FRUCTUS
 European Medicines Agency Evaluation of Medicines for Human Use London, 4 September 2008 Doc. Ref. EMEA/HMPC/368600/2007 COMMITTEE ON HERBAL MEDICINAL PRODUCTS (HMPC) FINAL COMMUNITY HERBAL MONOGRAPH ON
European Medicines Agency Evaluation of Medicines for Human Use London, 4 September 2008 Doc. Ref. EMEA/HMPC/368600/2007 COMMITTEE ON HERBAL MEDICINAL PRODUCTS (HMPC) FINAL COMMUNITY HERBAL MONOGRAPH ON
Treatment of diseases affecting the kidney using steroids
 Treatment of diseases affecting the kidney using steroids Hope Building Renal 0161 206 5223 All Rights Reserved 2015. Document for issue as handout. This information has been written by the medical and
Treatment of diseases affecting the kidney using steroids Hope Building Renal 0161 206 5223 All Rights Reserved 2015. Document for issue as handout. This information has been written by the medical and
FLAMAZINE CREAM 1.0% w/w
 PRODUCT INFORMATION NAME OF THE MEDICINE: FLAMAZINE CREAM 1.0% w/w Silver sulfadiazine 1% w/w Composition: Active ingredient. Silver sulfadiazine. Excipients. Polysorbate 60 Ph. Eur, Polysorbate 80 Ph.
PRODUCT INFORMATION NAME OF THE MEDICINE: FLAMAZINE CREAM 1.0% w/w Silver sulfadiazine 1% w/w Composition: Active ingredient. Silver sulfadiazine. Excipients. Polysorbate 60 Ph. Eur, Polysorbate 80 Ph.
Scottish Medicines Consortium
 Scottish Medicines Consortium betamethasone valerate 2.25mg medicated plaster (Betesil ) No. (622/10) Genus Pharmaceuticals 09 July 2010 The Scottish Medicines Consortium (SMC) has completed its assessment
Scottish Medicines Consortium betamethasone valerate 2.25mg medicated plaster (Betesil ) No. (622/10) Genus Pharmaceuticals 09 July 2010 The Scottish Medicines Consortium (SMC) has completed its assessment
Efficacy and Safety of Calcipotriol Ointment in Psoriasis Vulgaris - Experiences in Hong Kong
 ORIGINAL ARTICLES Efficacy and Safety of Calcipotriol Ointment in Psoriasis Vulgaris - Experiences in Hong Kong Drs. C. W. Fung, L.Y. Chong, C.Y. Leung, C. N. Look, K.K. Lo, K. M. Ho Social Hygiene Service
ORIGINAL ARTICLES Efficacy and Safety of Calcipotriol Ointment in Psoriasis Vulgaris - Experiences in Hong Kong Drs. C. W. Fung, L.Y. Chong, C.Y. Leung, C. N. Look, K.K. Lo, K. M. Ho Social Hygiene Service
160S01105, Page 1 of 7. Human Hepatitis B Immunoglobulin, solution for intramuscular injection.
 160S01105, Page 1 of 7 New Zealand Data Sheet Hepatitis B Immunoglobulin-VF NAME OF THE MEDICINE Human Hepatitis B Immunoglobulin, solution for intramuscular injection. DESCRIPTION Hepatitis B Immunoglobulin-VF
160S01105, Page 1 of 7 New Zealand Data Sheet Hepatitis B Immunoglobulin-VF NAME OF THE MEDICINE Human Hepatitis B Immunoglobulin, solution for intramuscular injection. DESCRIPTION Hepatitis B Immunoglobulin-VF
Guidelines of care for the management of psoriasis and psoriatic arthritis
 FROM THE ACADEMY Guidelines of care for the management of psoriasis and psoriatic arthritis Section 3. Guidelines of care for the management and treatment of psoriasis with topical therapies Alan Menter,
FROM THE ACADEMY Guidelines of care for the management of psoriasis and psoriatic arthritis Section 3. Guidelines of care for the management and treatment of psoriasis with topical therapies Alan Menter,
EFFIMET 1000 XR Metformin Hydrochloride extended release tablet
 BRAND NAME: Effimet XR. THERAPEUTIC CATEGORY: Anti-Diabetic PHARMACOLOGIC CLASS: Biguanides EFFIMET 1000 XR Metformin Hydrochloride extended release tablet COMPOSITION AND PRESENTATION Composition Each
BRAND NAME: Effimet XR. THERAPEUTIC CATEGORY: Anti-Diabetic PHARMACOLOGIC CLASS: Biguanides EFFIMET 1000 XR Metformin Hydrochloride extended release tablet COMPOSITION AND PRESENTATION Composition Each
Mucodyne-Clear 250 mg/5 ml Syrup PL 04425/0665
 Mucodyne-Clear 250 mg/5 ml Syrup PL 04425/0665 UKPAR TABLE OF CONTENTS Lay summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 9 Summary of product characteristics Page 10 Patient
Mucodyne-Clear 250 mg/5 ml Syrup PL 04425/0665 UKPAR TABLE OF CONTENTS Lay summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 9 Summary of product characteristics Page 10 Patient
Nursing Process Focus: Patients Receiving LINDANE (Kwell)
 Nursing Process Focus: Patients Receiving LINDANE (Kwell) ASSESSMENT Prior to administration: Obtain complete health history including allergies, drug history, and possible drug interactions. Assess vital
Nursing Process Focus: Patients Receiving LINDANE (Kwell) ASSESSMENT Prior to administration: Obtain complete health history including allergies, drug history, and possible drug interactions. Assess vital
Welcome to Dr Shreya s Homeopathy! We provide expert homeopathic prescription to you!
 Hello! Lovers of Homeopathy and Natural Health! Hello! Welcome to Dr Shreya s Homeopathy! We provide expert homeopathic prescription to you! Dr Shreya s Homeopathy is an online homeopathic consultancy
Hello! Lovers of Homeopathy and Natural Health! Hello! Welcome to Dr Shreya s Homeopathy! We provide expert homeopathic prescription to you! Dr Shreya s Homeopathy is an online homeopathic consultancy
5.07.09. Aubagio. Aubagio (teriflunomide) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.07.09 Subject: Aubagio Page: 1 of 6 Last Review Date: December 5, 2014 Aubagio Description Aubagio (teriflunomide)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.07.09 Subject: Aubagio Page: 1 of 6 Last Review Date: December 5, 2014 Aubagio Description Aubagio (teriflunomide)
PHARMACOVIGILANCE GUIDELINES
 PHARMACOVIGILANCE GUIDELINES What is Pharmacovigilance? Pharmacovigilance is defined as the science and activities concerned with the detection, assessment, understanding and prevention of adverse reactions
PHARMACOVIGILANCE GUIDELINES What is Pharmacovigilance? Pharmacovigilance is defined as the science and activities concerned with the detection, assessment, understanding and prevention of adverse reactions
DO YOU HAVE ITCHY ALLERGY EYES? Find out about lasting relief
 DO YOU HAVE ITCHY ALLERGY EYES? Find out about lasting relief Common causes of itching due to eye allergies include: Pollen from trees, grasses, and ragweed Dust mites Cat dander Approximately 10 million
DO YOU HAVE ITCHY ALLERGY EYES? Find out about lasting relief Common causes of itching due to eye allergies include: Pollen from trees, grasses, and ragweed Dust mites Cat dander Approximately 10 million
DERBYSHIRE JOINT AREA PRESCRIBING COMMITTEE (JAPC) SHARED CARE AGREEMENT FRAMEWORK
 DERBYSHIRE JOINT AREA PRESCRIBING COMMITTEE (JAPC) SHARED CARE AGREEMENT FRAMEWORK ACAMPROSATE CALCIUM (Campral EC) for alcohol abstinence ESCA: Adjunct in the treatment of chronic alcohol dependence (under
DERBYSHIRE JOINT AREA PRESCRIBING COMMITTEE (JAPC) SHARED CARE AGREEMENT FRAMEWORK ACAMPROSATE CALCIUM (Campral EC) for alcohol abstinence ESCA: Adjunct in the treatment of chronic alcohol dependence (under
IMPORTANT: PLEASE READ
 PART III: CONSUMER INFORMATION combined hepatitis A (inactivated) and hepatitis B (recombinant) vaccine This leaflet is part III of a three-part "Product Monograph" published when was approved for sale
PART III: CONSUMER INFORMATION combined hepatitis A (inactivated) and hepatitis B (recombinant) vaccine This leaflet is part III of a three-part "Product Monograph" published when was approved for sale
RADIOPHARMACEUTICALS BASED ON MONOCLONAL ANTIBODIES
 RADIOPHARMACEUTICALS BASED ON MONOCLONAL ANTIBODIES Guideline Title Radiopharmaceuticals based on Monoclonal Antibodies Legislative basis Directives 65/65/EEC, 75/318/EEC as amended, Directive 89/343/EEC
RADIOPHARMACEUTICALS BASED ON MONOCLONAL ANTIBODIES Guideline Title Radiopharmaceuticals based on Monoclonal Antibodies Legislative basis Directives 65/65/EEC, 75/318/EEC as amended, Directive 89/343/EEC
IMPORTANT DRUG WARNING Regarding Mycophenolate-Containing Products
 Dear Healthcare Provider: Mycophenolate REMS (Risk Evaluation and Mitigation Strategy) has been mandated by the FDA (Food and Drug Administration) due to postmarketing reports showing that exposure to
Dear Healthcare Provider: Mycophenolate REMS (Risk Evaluation and Mitigation Strategy) has been mandated by the FDA (Food and Drug Administration) due to postmarketing reports showing that exposure to
Sponsor Novartis. Generic Drug Name Secukinumab. Therapeutic Area of Trial Psoriasis. Approved Indication investigational
 Clinical Trial Results Database Page 2 Sponsor Novartis Generic Drug Name Secukinumab Therapeutic Area of Trial Psoriasis Approved Indication investigational Clinical Trial Results Database Page 3 Study
Clinical Trial Results Database Page 2 Sponsor Novartis Generic Drug Name Secukinumab Therapeutic Area of Trial Psoriasis Approved Indication investigational Clinical Trial Results Database Page 3 Study
Intravenous Methyl Prednisolone in Multiple Sclerosis
 Intravenous Methyl Prednisolone in Multiple Sclerosis Exceptional healthcare, personally delivered Relapse management in multiple sclerosis Relapses in multiple sclerosis (MS) are common and caused by
Intravenous Methyl Prednisolone in Multiple Sclerosis Exceptional healthcare, personally delivered Relapse management in multiple sclerosis Relapses in multiple sclerosis (MS) are common and caused by
Treatments for allergy are usually straightforward, safe and effective. Common treatments include:
 Allergy Medications The treatments prescribed for allergy control the symptoms and reactions; they do not cure the condition. However, using treatments as prescribed can show a huge change in a patient
Allergy Medications The treatments prescribed for allergy control the symptoms and reactions; they do not cure the condition. However, using treatments as prescribed can show a huge change in a patient
PARACETAMOL REXIDOL. 600 mg Tablet. Analgesic-Antipyretic. Paracetamol 600 mg
 (Insert Text) UL Consumer Health PARACETAMOL REXIDOL 600 mg Tablet Analgesic-Antipyretic FORMULATION Each tablet contains: Paracetamol 600 mg PRODUCT DESCRIPTION Rexidol is a round, yellow, flat, bevel-edged
(Insert Text) UL Consumer Health PARACETAMOL REXIDOL 600 mg Tablet Analgesic-Antipyretic FORMULATION Each tablet contains: Paracetamol 600 mg PRODUCT DESCRIPTION Rexidol is a round, yellow, flat, bevel-edged
Guideline for the use of subcutaneous hydration in palliative care (hypodermoclysis)
 Guideline for the use of subcutaneous hydration in palliative care (hypodermoclysis) Date Approved by Network Governance September 2012 Date for Review September 2015 Page 1 of 7 1 Scope of Guideline 1.1
Guideline for the use of subcutaneous hydration in palliative care (hypodermoclysis) Date Approved by Network Governance September 2012 Date for Review September 2015 Page 1 of 7 1 Scope of Guideline 1.1
Cutaneous Lymphoma FAST FACTS
 Cutaneous Lymphoma FAST FACTS What is Cutaneous Lymphoma? Cutaneous lymphomas are types of non-hodgkin s lymphomas (NHL) that originate in the lymphocytes (white blood cells). Unlike most other types of
Cutaneous Lymphoma FAST FACTS What is Cutaneous Lymphoma? Cutaneous lymphomas are types of non-hodgkin s lymphomas (NHL) that originate in the lymphocytes (white blood cells). Unlike most other types of
A normal eye is protected by a layer of natural tears.
 A normal eye is protected by a layer of natural tears. If you need to use eye drops several times a day to make your eyes comfortably moist, you should consult your eye doctor. Helps increase tear production
A normal eye is protected by a layer of natural tears. If you need to use eye drops several times a day to make your eyes comfortably moist, you should consult your eye doctor. Helps increase tear production
Treatment options a simple guide
 Guide Treatment options a simple guide To decide which treatment is right for you, a good starting point is to know what options you have and to understand the pros and cons of each one. People respond
Guide Treatment options a simple guide To decide which treatment is right for you, a good starting point is to know what options you have and to understand the pros and cons of each one. People respond
ALLERGENIC EXTRACT. Prescription Set of Serial Dilutions (or Maintenance Vial (s)) INSTRUCTIONS FOR USE. U.S. Government License No.
 ALLERGENIC EXTRACT Prescription Set of Serial Dilutions (or Maintenance Vial (s)) INSTRUCTIONS FOR USE U.S. Government License No. 308 Revised 07/04 PO Box 800 Lenoir, NC 28645 USA DESCRIPTION This set
ALLERGENIC EXTRACT Prescription Set of Serial Dilutions (or Maintenance Vial (s)) INSTRUCTIONS FOR USE U.S. Government License No. 308 Revised 07/04 PO Box 800 Lenoir, NC 28645 USA DESCRIPTION This set
An introduction to modern MS treatments
 BETAFERON is a Prescription Medicine. Use strictly as directed. Consult your pharmacist or other health professional in case of side effects. BETAFERON is reimbursed for some patients. See your neurologist
BETAFERON is a Prescription Medicine. Use strictly as directed. Consult your pharmacist or other health professional in case of side effects. BETAFERON is reimbursed for some patients. See your neurologist
Trileptal (Oxcarbazepine)
 Brand and Generic Names: Trileptal Tablets: 150mg, 300mg, 600mg Liquid Suspension: 300mg/5mL Generic name: oxcarbazepine What is Trileptal and what does it treat? Trileptal (Oxcarbazepine) Oxcarbazepine
Brand and Generic Names: Trileptal Tablets: 150mg, 300mg, 600mg Liquid Suspension: 300mg/5mL Generic name: oxcarbazepine What is Trileptal and what does it treat? Trileptal (Oxcarbazepine) Oxcarbazepine
Community herbal monograph on Commiphora molmol Engler, gummi-resina
 12 July 2011 EMA/HMPC/96911/2010 Committee on Herbal Medicinal Products (HMPC) Community herbal monograph on Commiphora molmol Engler, gummi-resina Final Discussion in Working Party on Community monographs
12 July 2011 EMA/HMPC/96911/2010 Committee on Herbal Medicinal Products (HMPC) Community herbal monograph on Commiphora molmol Engler, gummi-resina Final Discussion in Working Party on Community monographs
