Rapid-VIDITEST Calprotectin Blister
|
|
|
- Rosamund Hopkins
- 7 years ago
- Views:
Transcription
1 Rapid-VIDITEST Calprotectin Blister One Step Calprotectin Blister test. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, Vestec, Jesenice, Czech Republic, Tel.: , INTENDED USE: The Rapid-VIDITEST Calprotectin Blister test is a rapid chromatographic immunoassay (non-invasive assay) for the qualitative detection of calprotectin in human feces specimens, which might be useful for the diagnosis of inflammatory gastrointestinal disorders. INTRODUCTION: Calprotectin is a calcium-containing protein that makes up 5% of the total protein and 60% of the cytosolic protein of neutrophil. It has bacteriostatic and fungistatic properties and is found in feces at levels six times higher than that in plasma. That fecal biomarker is useful to assess the activity of inflammatory bowel disease (IBD). IBD includes Crohn's Disease (CD) and Ulcerative Colitis (UC) and are associated with elevated neutrophils. This fecal calprotectin assay is useful in differentiating organic (IBD) from functional gastrointestinal disease (IBS: Intestinal Bowel Syndrome). It is a simple, non-invasive biomarker that is especially useful in children, who may require general anesthesia for colonoscopy. And this fecal calprotectin detection can predict relapse. PRINCIPLE: The Rapid-VIDITEST Calprotectin Blister is a qualitative immunoassay for the detection of calprotectin in feces samples. The membrane is pre-coated with monoclonal antibodies against calprotectin on the test lines region. During testing, the sample reacts with the particles coated with anti-human calprotectin antibodies which were pre-dried on the test strip. The mixture moves upward on the membrane by capillary action. In the case of a positive result the specific antibodies present on the membrane will react with the mixture conjugate and generate colored line. A green colored band always appears in the control line and serves as verification that sufficient volume was added, that proper flow was obtained and as an internal control for the reagents. MATERALS PROVIDED: - Rapid-VIDITEST Calprotectin Blister tests - Instructions for use - Specimen collection vial with buffer 1
2 MATERIALS REQUIRED BUT NO PROVIDED: - Specimen collection container - Disposable gloves - Timer SPECIMEN COLLECTION AND PREPARATION: Collect sufficient quantity of faeces (1-2 g or ml for liquid sample). Stool samples should be collected in clean and dry containers (no preservatives or transport media). The samples can be stored in the refrigerator (2-4ºC/36-40ºF) for 7 days prior to testing. For longer storage the specimen must be kept frozen at 20ºC/4ºF. In this case, the sample will be totally thawed, and brought to room temperature before testing. PROCEDURES: Specimen preparation: Use a separate specimen collection vial for each sample. 1. Unscrew the cap of the vial and introduce the stick three times into the faecal specimen to pick up the sample (1). Close the vial with the buffer and stool sample. This vial with the sample can be storage during 5 days. 2. Shake the vial in order to assure good sample dispersion (2). For liquid stool samples, aspirate the faecal specimen with a dropper and add approx μl into the specimen collection vial with buffer. TEST PROCEDURE Allow the tests, stool samples and buffer to reach to room temperature (15-30ºC/59-86ºF) prior to testing. Do not open the pack until ready to perform the assay. There are two possibilities for performing the test: A) Using the blister test single pack as a Card test: 1. Cut the blister to obtain a test single pack, hold the non sealed side and open it peeling off the upper foil. Don t remove the test from the blister cavity and use it as soon as possible. 2. Shake the specimen collection tube to assure good sample dispersion. Cut the end of the top (3). 3. Place blister test single pack horizontally and identity it. Dispense 5 drops of sample+buffer on the white end of the test (4). Start the timer. Read the result at 10 minutes after dispensing the sample. 2
3 B) Using the blister test single pack as a strip test: By immersion: 1. Cut the blister to obtain a test single pack, hold the non sealed side and open it peeling off the upper foil. 2. Shake the specimen collection vial to assure good sample dispersion. Cut the end of the top (3). 3. Dispense 5 drops of sample+buffer in an identified vial and leave the test strip to stand vertically in the vial (5), taking care of not surpassing the limit of immersion indicated with the arrows. Start the timer. Read the result at 10 minutes. INTERPRETATION OF RESULTS: POSITIVE NEGATIVE INVALID POSITIVE: Two lines appears across the central zone, in the result line region a red test line marked and in the control line region a green control line marked. A calprotectin positive result could be indicative of gastrointestinal inflammatory pathology is present. NEGATIVE: Only one green band appears across the control line region marked with the letter C (control line). A negative results shows that neither active gastrointestinal inflammation is present. INVALID: A total absence of the green control colored band regardless the appearance or not of the red test line. Note: Insufficient specimen volume, incorrect procedural techniques or deterioration of the reagents are the most likely reasons for control line failure. Review the procedure and repeat the test with a new test. If the problem persists, discontinue using the test kit and contact your local distributor. 3
4 NOTES ON THE INTERPRETATION OF RESULTS: The intensity of the red coloured band in the result line region (T) will vary depending on the concentration of calprotectin in the specimen. QUALITY CONTROL: Internal procedural controls are included in the test: - A green line appearing in the control line region (C). It confirms sufficient specimen volume and correct procedural technique. - A clear background is an internal negative background control. If the test is working properly, the background in the result area should be clear and not interfere with the ability to read the result. LIMITATIONS: 1. Rapid-VIDITEST Calprotectin Blister will only indicate the presence of calprotectin in the specimen (qualitative detection) and should be used for the detection of calprotectin in feces specimens only. Neither the quantitative value nor the rate of increase in calprotectin concentration can be determined by this test. 2. An excess of sample could cause wrong results (brown bands appear). Dilute the sample with the buffer and repeat the test. 3. Some stool samples can decrease the intensity of the control line. 4. In the case of patients with active neutrophilic inflammatory bowel diseases such as Crohn s disease and Ulcerative Colitis, would be positive for fecal calprotectin. Calprotectin Strip could be used for patients with chronic diarrhea. 5. Positive results confirm the presence of calprotectin in fecal samples; nevertheless, it can be due to several causes, inflammatory bowel disease, colorectal cancer and some enteropathies). Positive results should be followed up with additional diagnostic procedures by a physician to determine the exact cause of inflammation. 6. Neonatal fecal calprotectin levels have been reported higher than normal children with a median of 167µg/g. EXPECTED VALUES: Higher levels of calprotectin in the stool are associated with an increased risk of relapse in patients with inflammatory bowel disease (IBD). Some studies established equal or higher 50µg hfcp/g faeces as cut-off value to allow detect adult patients with GI inflammatory problems. PERFORMANCE CHARACTERISTICS: Sensitivity A sample containing calprotectin at concentration equal to or higher than 50µg/g faeces produces positive results when using Rapid-VIDITEST Calprotectin Blister test. Different calprotectin dilutions were tested directly in the extraction buffer or spiked in a negative stool sample in accordance with the kit instructions to determinate the detection limit of the test. The detection of human calprotectin with Rapid-VIDITEST Calprotectin Blister test showed >94% of sensitivity correlation compared to another commercial immunoassay (Calprest Eurospital). 4
5 Specificity The detection of human calprotectin with Rapid-VIDITEST Calprotectin Blister test showed 93% of specificity correlation compared to another commercial immunoassay (Calprest Eurospital). The Rapid-VIDITEST Calprotectin Blister test is specific for human Calprotectin, showing no cross-reaction with other calprotectins. STORAGE AND STABILITY: Store as packaged in the sealed pouch either at refrigerated or room temperature (2-30ºC/36-86ºF). The test is stable through the expiration date printed on the sealed pouch. The test must remain in the sealed pouch until use. Do not freeze. PRECAUTIONS: - For professional in vitro diagnostic use only. - Do not use after expiration date. - The test should remain in the sealed pouch until use. - Do not use the test if pouch is damaged. - Follow Good Laboratory Practices, wear protective clothing, use disposal gloves, do not eat, drink or smoke in the area. - All the specimens should be considered potentially hazardous and handled in the same manner as an infectious agent. - The test should be discarded in a proper biohazard container after testing. - The test must be carried out within 2 hours of opening the sealed bag. REFERENCES: 1. VIEIRA, A. et al., Inflammatory bowel disease activity assessed by fecal calprotectin and lactoferrin: correlation with laboratory parameters, clinical, endoscopic and histological indexes, BMC Research Notes 2009, 2: HANAI, H. et al. «Relationship Between Fecal Calprotectin, Intestinal Inflammation, and Peripheral Blood Neutrophils in Patients with Active Ulcerative Colitis Digestive Diseases and Sciences, Sept. 2004, Vol 49, No 9, pp BONNIN TOMAS, A, et al. Calprotectina fecal como marcador diferencia entre patología gastrointestinal orgánica y funcional. Rev. Esp. de Enf. Dig. 2007, Vol 99, No 12, pp SYMBOLS FOR IVD COMPONENTS AND REAGENTS: In vitro diagnostic device Use by Batch code Manufacturer 5
OneStep Fecal Occult Blood RapiDip InstaTest. Cat # 13020-1
 CORTEZ DIAGNOSTICS, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 USA Tel: (818) 591-3030 Fax: (818) 591-8383 E-mail: onestep@rapidtest.com Web site: www.rapidtest.com See external label
CORTEZ DIAGNOSTICS, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 USA Tel: (818) 591-3030 Fax: (818) 591-8383 E-mail: onestep@rapidtest.com Web site: www.rapidtest.com See external label
Mouse IgM ELISA. Cat. No. KT-407 K-ASSAY. For the quantitative determination of IgM in mouse biological samples. For Research Use Only. 1 Rev.
 K-ASSAY Mouse IgM ELISA For the quantitative determination of IgM in mouse biological samples Cat. No. KT-407 For Research Use Only. 1 Rev. 072309 K-ASSAY PRODUCT INFORMATION Mouse IgM ELISA Cat. No. KT-407
K-ASSAY Mouse IgM ELISA For the quantitative determination of IgM in mouse biological samples Cat. No. KT-407 For Research Use Only. 1 Rev. 072309 K-ASSAY PRODUCT INFORMATION Mouse IgM ELISA Cat. No. KT-407
Chapter. Guaiac Screening CHAPTER 4: GUAIAC TESTING SCREENING FOR OCCULT BLOOD. Page 1 of 5 Guaiac 3-5-02.doc 6/24/2005
 Chapter 4 Guaiac Screening CHAPTER 4: GUAIAC TESTING SCREENING FOR OCCULT BLOOD Page 1 of 5 Guaiac 3-5-02.doc 6/24/2005 Procedure: Guaiac Testing Screening for Fecal Occult Blood POLICY: The stool occult
Chapter 4 Guaiac Screening CHAPTER 4: GUAIAC TESTING SCREENING FOR OCCULT BLOOD Page 1 of 5 Guaiac 3-5-02.doc 6/24/2005 Procedure: Guaiac Testing Screening for Fecal Occult Blood POLICY: The stool occult
CK-MB TEST CARD 004A302
 A rapid test for the qualitative detection of CK-MB in whole blood, serum or plasma. For professional in vitro diagnostic use only. INTENDED USE The CK-MB test card is a rapid chromatographic immunoassay
A rapid test for the qualitative detection of CK-MB in whole blood, serum or plasma. For professional in vitro diagnostic use only. INTENDED USE The CK-MB test card is a rapid chromatographic immunoassay
ELISA-VIDITEST-MICROCYSTIN LR Lot: XX
 ELISA-VIDITEST-MICROCYSTIN LR Lot: XX Instruction manual PRODUCER : VIDIA Ltd., Nad Safinou II/365, Vestec, 252 42 Jesenice u Prahy, Czech Republic, Tel.: +420261090566 1. TITLE: ELISA-VIDITEST MICROCYSTIN
ELISA-VIDITEST-MICROCYSTIN LR Lot: XX Instruction manual PRODUCER : VIDIA Ltd., Nad Safinou II/365, Vestec, 252 42 Jesenice u Prahy, Czech Republic, Tel.: +420261090566 1. TITLE: ELISA-VIDITEST MICROCYSTIN
Albumin ELISA Kit. Albumin ELISA Kit. Zur in vitro Bestimmung des Albumin in Urin und Stuhl
 ELISA Kit Super sensitiv Zur in vitro Bestimmung des in Urin und Stuhl ELISA Kit Super sensitive For the in vitro determination of in urine and stool Gültig ab / Valid from 06.03.2007 +8 C IMM-K 6330 96
ELISA Kit Super sensitiv Zur in vitro Bestimmung des in Urin und Stuhl ELISA Kit Super sensitive For the in vitro determination of in urine and stool Gültig ab / Valid from 06.03.2007 +8 C IMM-K 6330 96
Mouse Insulin ELISA. For the quantitative determination of insulin in mouse serum and plasma
 Mouse Insulin ELISA For the quantitative determination of insulin in mouse serum and plasma Please read carefully due to Critical Changes, e.g., Calculation of Results. For Research Use Only. Not For Use
Mouse Insulin ELISA For the quantitative determination of insulin in mouse serum and plasma Please read carefully due to Critical Changes, e.g., Calculation of Results. For Research Use Only. Not For Use
Anti-Zona Pellucida Antibody Latex Agglutination Test
 Instructions for use Anti-Zona Pellucida Antibody Latex Agglutination Test Cat. No.: BS-20-10 Size: 50 Determinations Storage: 2 C 8 C (36 F 46 F) Screening test for the determination of anti-zona pellucida
Instructions for use Anti-Zona Pellucida Antibody Latex Agglutination Test Cat. No.: BS-20-10 Size: 50 Determinations Storage: 2 C 8 C (36 F 46 F) Screening test for the determination of anti-zona pellucida
IgM ELISA. For the quantitative determination of IgM in human serum and plasma. For Research Use Only. Not For Use In Diagnostic Procedures.
 IgM ELISA For the quantitative determination of IgM in human serum and plasma For Research Use Only. Not For Use In Diagnostic Procedures. Please read carefully due to Critical Changes, e.g., Calibrator
IgM ELISA For the quantitative determination of IgM in human serum and plasma For Research Use Only. Not For Use In Diagnostic Procedures. Please read carefully due to Critical Changes, e.g., Calibrator
Mouse Creatine Kinase MB isoenzyme (CKMB) ELISA
 KAMIYA BIOMEDICAL COMPANY Mouse Creatine Kinase MB isoenzyme (CKMB) ELISA For the quantitative determination of mouse CKMB in serum, plasma, cell culture fluid and other biological fluids Cat. No. KT-57681
KAMIYA BIOMEDICAL COMPANY Mouse Creatine Kinase MB isoenzyme (CKMB) ELISA For the quantitative determination of mouse CKMB in serum, plasma, cell culture fluid and other biological fluids Cat. No. KT-57681
ABORhCard. ABORhCard Package Insert ABO and Rh Blood Grouping Device
 ABORhCard Package Insert ABO and Rh Blood Grouping Device ABORhCard Intended Use The ABORhCard is a qualitative in vitro test that provides a simultaneous ABO and Rh determination of an individual s ABO
ABORhCard Package Insert ABO and Rh Blood Grouping Device ABORhCard Intended Use The ABORhCard is a qualitative in vitro test that provides a simultaneous ABO and Rh determination of an individual s ABO
Inc. Wuhan. Quantity Pre-coated, ready to use 96-well strip plate 1 Plate sealer for 96 wells 4 Standard (liquid) 2
 Uscn Life Science Inc. Wuhan Website: www.uscnk.com Phone: +86 27 84259552 Fax: +86 27 84259551 E-mail: uscnk@uscnk.com ELISA Kit for Human Prostaglandin E1(PG-E1) Instruction manual Cat. No.: E0904Hu
Uscn Life Science Inc. Wuhan Website: www.uscnk.com Phone: +86 27 84259552 Fax: +86 27 84259551 E-mail: uscnk@uscnk.com ELISA Kit for Human Prostaglandin E1(PG-E1) Instruction manual Cat. No.: E0904Hu
HBV Quantitative Real Time PCR Kit
 Revision No.: ZJ0002 Issue Date: Aug 7 th, 2008 HBV Quantitative Real Time PCR Kit Cat. No.: HD-0002-01 For Use with LightCycler 1.0/LightCycler2.0/LightCycler480 (Roche) Real Time PCR Systems (Pls ignore
Revision No.: ZJ0002 Issue Date: Aug 7 th, 2008 HBV Quantitative Real Time PCR Kit Cat. No.: HD-0002-01 For Use with LightCycler 1.0/LightCycler2.0/LightCycler480 (Roche) Real Time PCR Systems (Pls ignore
Contents. Approved by: Kent Lewandrowski, M.D. 6/1/2005 Written/Updated by: Gino Pagnani Date: 4/3/09
 POCT Program Massachusetts General Hospital - Pathology Service 55 Fruit Street, Boston, MA 02114 Title: Hemoccult Sensa Fecal Occult Blood Procedure Cross References: Hemoccult Sensa Fecal Occult Blood
POCT Program Massachusetts General Hospital - Pathology Service 55 Fruit Street, Boston, MA 02114 Title: Hemoccult Sensa Fecal Occult Blood Procedure Cross References: Hemoccult Sensa Fecal Occult Blood
Direct Testing Systems and Serology
 Direct Testing Systems and Serology Rapid Manual Tests 6-2 Serology Diagnostics 6-6 BD Diagnostics Diagnostic Systems Catalog 2005/2006 6-1 Rapid Manual Tests Meningitis Test Systems 252360 Directigen
Direct Testing Systems and Serology Rapid Manual Tests 6-2 Serology Diagnostics 6-6 BD Diagnostics Diagnostic Systems Catalog 2005/2006 6-1 Rapid Manual Tests Meningitis Test Systems 252360 Directigen
Toxoplasma gondii IgM ELISA Kit Protocol
 Toxoplasma gondii IgM ELISA Kit Protocol (Cat. No.:EK-310-87) 330 Beach Road, Burlingame CA Tel: 650-558-8898 Fax: 650-558-1686 E-Mail: info@phoenixpeptide.com www.phoenixpeptide.com Toxoplasma IgM ELISA
Toxoplasma gondii IgM ELISA Kit Protocol (Cat. No.:EK-310-87) 330 Beach Road, Burlingame CA Tel: 650-558-8898 Fax: 650-558-1686 E-Mail: info@phoenixpeptide.com www.phoenixpeptide.com Toxoplasma IgM ELISA
Canine creatine kinase MB isoenzyme (CK-MB)ELISA Kit
 Canine creatine kinase MB isoenzyme (CK-MB)ELISA Kit Catalog No. CSB-E15852c (96T) This immunoassay kit allows for the in vitro quantitative determination of canine CK-MB concentrations in serum and plasma.
Canine creatine kinase MB isoenzyme (CK-MB)ELISA Kit Catalog No. CSB-E15852c (96T) This immunoassay kit allows for the in vitro quantitative determination of canine CK-MB concentrations in serum and plasma.
Immunoglobulin E (IgE) concentrations in Human. Immunoglobulin E (IgE) Human ELISA Kit
 ab108650 Immunoglobulin E (IgE) Human ELISA Kit Instructions for Use For the quantitative measurement of Immunoglobulin E (IgE) concentrations in Human serum. This product is for research use only and
ab108650 Immunoglobulin E (IgE) Human ELISA Kit Instructions for Use For the quantitative measurement of Immunoglobulin E (IgE) concentrations in Human serum. This product is for research use only and
Blood Collection and Processing SOP
 Brisbane Breast Bank Blood Collection and Processing SOP Breast Pathology Laboratory University of Queensland Centre for Clinical Research Blood Collection We collect 30ml of blood from patients who have
Brisbane Breast Bank Blood Collection and Processing SOP Breast Pathology Laboratory University of Queensland Centre for Clinical Research Blood Collection We collect 30ml of blood from patients who have
Rat Creatine Kinase MB isoenzyme,ck-mb ELISA Kit
 Rat Creatine Kinase MB isoenzyme,ck-mb ELISA Kit Catalog No: E0479r 96 Tests Operating instructions www.eiaab.com FOR RESEARCH USE ONLY; NOT FOR THERAPEUTIC OR DIAGNOSTIC APPLICATIONS! PLEASE READ THROUGH
Rat Creatine Kinase MB isoenzyme,ck-mb ELISA Kit Catalog No: E0479r 96 Tests Operating instructions www.eiaab.com FOR RESEARCH USE ONLY; NOT FOR THERAPEUTIC OR DIAGNOSTIC APPLICATIONS! PLEASE READ THROUGH
SAMPLE PROCEDURE 1029-3, 09/11
 SAMPLE PROCEDURE This Sample Procedure is not intended as a substitute for your facility s Procedure Manual or reagent labeling, but rather as a model for your use in customizing for your laboratory s
SAMPLE PROCEDURE This Sample Procedure is not intended as a substitute for your facility s Procedure Manual or reagent labeling, but rather as a model for your use in customizing for your laboratory s
IgE (Human) ELISA Kit
 IgE (Human) ELISA Kit Catalog Number KA0216 96 assays Version: 03 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of the Assay...
IgE (Human) ELISA Kit Catalog Number KA0216 96 assays Version: 03 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of the Assay...
MYCOLOGY PARASITOLOGY RHEUMATOID FACTORS VIROLOGY SAMPLE COLLECTION
 C A T A L O G U E C L I N I C A L D I A G N O S T I C S couv BACTERIOLOGYOGY MYCOLOGY PARASITOLOGY RHEUMATOID FACTORS VIROLOGY SAMPLE COLLECTION couv EXPORT DEPARTMENT Orders, technical and commercial
C A T A L O G U E C L I N I C A L D I A G N O S T I C S couv BACTERIOLOGYOGY MYCOLOGY PARASITOLOGY RHEUMATOID FACTORS VIROLOGY SAMPLE COLLECTION couv EXPORT DEPARTMENT Orders, technical and commercial
HiPer RA Test Teaching Kit
 HiPer RA Test Teaching Kit Product Code: HTI019 Number of experiments that can be performed: 20 Duration of Experiment: 1hour Storage Instructions: The kit is stable for 6 months from the date of receipt
HiPer RA Test Teaching Kit Product Code: HTI019 Number of experiments that can be performed: 20 Duration of Experiment: 1hour Storage Instructions: The kit is stable for 6 months from the date of receipt
Mouse krebs von den lungen 6 (KL-6) ELISA
 KAMIYA BIOMEDICAL COMPANY Mouse krebs von den lungen 6 (KL-6) ELISA For the quantitative determination of mouse KL-6 in serum, plasma, cell culture supernatants, body fluid and tissue homogenate Cat. No.
KAMIYA BIOMEDICAL COMPANY Mouse krebs von den lungen 6 (KL-6) ELISA For the quantitative determination of mouse KL-6 in serum, plasma, cell culture supernatants, body fluid and tissue homogenate Cat. No.
Epinephrine/Norepinephrine ELISA Kit
 Epinephrine/Norepinephrine ELISA Kit Catalog Number KA3767 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Principle of the Assay...
Epinephrine/Norepinephrine ELISA Kit Catalog Number KA3767 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Principle of the Assay...
Epi procolon The Blood Test for Colorectal Cancer Screening
 Epi procolon The Blood Test for Colorectal Cancer Screening Epi procolon is an approved blood test for colorectal cancer screening. The US Preventive Services Task Force, the American Cancer Society and
Epi procolon The Blood Test for Colorectal Cancer Screening Epi procolon is an approved blood test for colorectal cancer screening. The US Preventive Services Task Force, the American Cancer Society and
510(k) SUBSTANTIAL EQUIVALENCE DETERMINATION DECISION SUMMARY
 A. 510(k) Number: K092353 510(k) SUBSTANTIAL EQUIVALENCE DETERMINATION DECISION SUMMARY B. Purpose for Submission: This is a new 510k application for a new indication for the MONOLISA Anti-HAV IgM EIA
A. 510(k) Number: K092353 510(k) SUBSTANTIAL EQUIVALENCE DETERMINATION DECISION SUMMARY B. Purpose for Submission: This is a new 510k application for a new indication for the MONOLISA Anti-HAV IgM EIA
Please note: Contact Coppe Laboratories at 262-574-0701 if archival plasma samples need to be tested.
 Collecting a Coppe Laboratories Sample The Coppe Laboratories Sample Kit contains: Test Request Form (TRF) Heparin tube Absorbent sheet Biohazard bag Foil pouch Label for blood tube Box Seal Instructions
Collecting a Coppe Laboratories Sample The Coppe Laboratories Sample Kit contains: Test Request Form (TRF) Heparin tube Absorbent sheet Biohazard bag Foil pouch Label for blood tube Box Seal Instructions
TRIIODOTHYRONINE (T3) ELISA Kit Protocol
 TRIIODOTHYRONINE (T3) ELISA Kit Protocol (Cat. No.:EK-310-05) 330 Beach Road, Burlingame CA Tel: 650-558-8898 Fax: 650-558-1686 E-Mail: info@phoenixpeptide.com www.phoenixpeptide.com INTENDED USE For the
TRIIODOTHYRONINE (T3) ELISA Kit Protocol (Cat. No.:EK-310-05) 330 Beach Road, Burlingame CA Tel: 650-558-8898 Fax: 650-558-1686 E-Mail: info@phoenixpeptide.com www.phoenixpeptide.com INTENDED USE For the
STEP-BY-STEP INSTRUCTIONS FOR INVESTIGATIONAL USE. Rapid HCV Antibody Test FOR ORAQUICK RAPID HCV ANTIBODY TEST
 Before performing testing, all operators MUST read and become familiar with Universal Precautions for Prevention of Transmission of Human Immunodeficiency Virus, Hepatitis B Virus, and other Blood-borne
Before performing testing, all operators MUST read and become familiar with Universal Precautions for Prevention of Transmission of Human Immunodeficiency Virus, Hepatitis B Virus, and other Blood-borne
Human Free Testosterone(F-TESTO) ELISA Kit
 Human Free Testosterone(F-TESTO) ELISA Kit Catalog Number. MBS700040 For the quantitative determination of human free testosterone(f-testo) concentrations in serum, plasma. This package insert must be
Human Free Testosterone(F-TESTO) ELISA Kit Catalog Number. MBS700040 For the quantitative determination of human free testosterone(f-testo) concentrations in serum, plasma. This package insert must be
Mouse glycated hemoglobin A1c(GHbA1c) ELISA Kit
 Mouse glycated hemoglobin A1c(GHbA1c) ELISA Kit Catalog Number. CSB-E08141m For the quantitative determination of mouse glycated hemoglobin A1c(GHbA1c) concentrations in lysate for RBC. This package insert
Mouse glycated hemoglobin A1c(GHbA1c) ELISA Kit Catalog Number. CSB-E08141m For the quantitative determination of mouse glycated hemoglobin A1c(GHbA1c) concentrations in lysate for RBC. This package insert
Measles (Rubeola) IgM ELISA Catalog No. CB40-101-325099 (96 Tests)
 For Research Use Only. Not for use in Diagnostic Procedures. INTENDED USE Measles test is an enzyme linked immunosorbent assay (ELISA) for the detection of IgG class antibodies to Measles (Rubeola) in
For Research Use Only. Not for use in Diagnostic Procedures. INTENDED USE Measles test is an enzyme linked immunosorbent assay (ELISA) for the detection of IgG class antibodies to Measles (Rubeola) in
Canine Creatine Kinase MM isoenzyme(ck-mm) ELISA. kit
 BlueGene Biotech. Tel: 0086-21-61471242 Fax: 0086-21-61471242 ext 806 E-mail: sales@bluegene.cc tech@bluegene.cc www.elisakit.cc www.bluegene.cc Canine Creatine Kinase MM isoenzyme(ck-mm) ELISA kit 96
BlueGene Biotech. Tel: 0086-21-61471242 Fax: 0086-21-61471242 ext 806 E-mail: sales@bluegene.cc tech@bluegene.cc www.elisakit.cc www.bluegene.cc Canine Creatine Kinase MM isoenzyme(ck-mm) ELISA kit 96
LIA-VIDITEST anti-borrelia IgM
 LIA-VIDITEST anti-borrelia IgM ODZ - 317 Instruction manual PRODUCER: VIDIA s.r.o., Nad Safinou II 365, Vestec, 252 42 Jesenice u Prahy, Czech Republic, tel.: +420 261 090 565, www.vidia.cz 1. TITLE: LIA-VIDITEST
LIA-VIDITEST anti-borrelia IgM ODZ - 317 Instruction manual PRODUCER: VIDIA s.r.o., Nad Safinou II 365, Vestec, 252 42 Jesenice u Prahy, Czech Republic, tel.: +420 261 090 565, www.vidia.cz 1. TITLE: LIA-VIDITEST
RealLine HCV PCR Qualitative - Uni-Format
 Instructions for use PCR KIT FOR EXTRACTION OF RNA AND REAL TIME PCR DETECTION KIT FOR HEPATITIS C VIRUS RNA Research Use Only Qualitative Uni Format VBD0798 48 tests valid from: December 2013 Rev11122013
Instructions for use PCR KIT FOR EXTRACTION OF RNA AND REAL TIME PCR DETECTION KIT FOR HEPATITIS C VIRUS RNA Research Use Only Qualitative Uni Format VBD0798 48 tests valid from: December 2013 Rev11122013
Carcinoembryonic Antigen (CEA)
 Principle of the Test Carcinoembryonic Antigen (CEA) ADVIA Centaur System The ADVIA Centaur CEA assay is a two-site sandwich immunoassay using direct chemiluminometric technology, which uses constant amounts
Principle of the Test Carcinoembryonic Antigen (CEA) ADVIA Centaur System The ADVIA Centaur CEA assay is a two-site sandwich immunoassay using direct chemiluminometric technology, which uses constant amounts
POC.PC.17 Point of Care Testing AMNISURE PROCEDURE FOR RUPTURED FETAL MEMBRANES TABLE OF CONTENTS. 1.0 Principle of Procedure.. 2
 POC.PC.17 Point of Care Testing AMNISURE PROCEDURE FOR RUPTURED FETAL MEMBRANES TABLE OF CONTENTS 1.0 Principle of Procedure.. 2 2.0 Materials and Storage Requirements.. 2 3.0 Patient ID, Specimen ID and
POC.PC.17 Point of Care Testing AMNISURE PROCEDURE FOR RUPTURED FETAL MEMBRANES TABLE OF CONTENTS 1.0 Principle of Procedure.. 2 2.0 Materials and Storage Requirements.. 2 3.0 Patient ID, Specimen ID and
Human Peripheral Blood Mononuclear Cell (PBMC) Manual
 Human Peripheral Blood Mononuclear Cell (PBMC) Manual INSTRUCTION MANUAL ZBM0063.04 SHIPPING CONDITIONS Human Peripheral Blood Mononuclear Cells, cryopreserved Cryopreserved human peripheral blood mononuclear
Human Peripheral Blood Mononuclear Cell (PBMC) Manual INSTRUCTION MANUAL ZBM0063.04 SHIPPING CONDITIONS Human Peripheral Blood Mononuclear Cells, cryopreserved Cryopreserved human peripheral blood mononuclear
Malondialdehyde (MDA) ELISA
 Malondialdehyde (MDA) ELISA For the quantitative determination of MDA in serum, plasma and other biological fluids Cat. No. KT-21493 For Research Use Only. Not for use in diagnostic procedures. Page 1
Malondialdehyde (MDA) ELISA For the quantitative determination of MDA in serum, plasma and other biological fluids Cat. No. KT-21493 For Research Use Only. Not for use in diagnostic procedures. Page 1
Lead Testing and On Site Calibration for Water Testing Detection Range: 2 100ppb
 Document: AND Lead 100 7 2013 Lead Testing and On Site Calibration for Water Testing Detection Range: 2 100ppb July, 2013 Edition 1 ANDalyze, Inc., 2012. All rights reserved. Printed in USA. Table of Contents
Document: AND Lead 100 7 2013 Lead Testing and On Site Calibration for Water Testing Detection Range: 2 100ppb July, 2013 Edition 1 ANDalyze, Inc., 2012. All rights reserved. Printed in USA. Table of Contents
Dengue IgM ELISA. For the quantitative determination of IgM-class antibodies to Dengue Virus in serum.
 Dengue IgM ELISA For the quantitative determination of IgM-class antibodies to Dengue Virus in serum. For Research Use Only. Not For Use In Diagnostic Procedures. Catalog Number: 20-DEMHU-E01 Size: 96
Dengue IgM ELISA For the quantitative determination of IgM-class antibodies to Dengue Virus in serum. For Research Use Only. Not For Use In Diagnostic Procedures. Catalog Number: 20-DEMHU-E01 Size: 96
X-ray (Radiography), Lower GI Tract
 Scan for mobile link. X-ray (Radiography), Lower GI Tract What is Lower GI Tract X-ray Radiography (Barium Enema)? Lower gastrointestinal (GI) tract radiography, also called a lower GI or barium enema,
Scan for mobile link. X-ray (Radiography), Lower GI Tract What is Lower GI Tract X-ray Radiography (Barium Enema)? Lower gastrointestinal (GI) tract radiography, also called a lower GI or barium enema,
Cold Agglutination Titer detecting Cold Reacting Antibodies
 Objectives: Cold Agglutination Titer detecting Cold Reacting Antibodies 1. Perform a serial dilution to determine the amount of cold reacting antibody present in a patient specimen with the results obtained
Objectives: Cold Agglutination Titer detecting Cold Reacting Antibodies 1. Perform a serial dilution to determine the amount of cold reacting antibody present in a patient specimen with the results obtained
Uscn Life Science Inc. Wuhan
 Uscn Life Science Inc. Wuhan Website: www.uscnk.com Phone: +86 27 84259552 Fax: +86 27 84259551 E-mail: uscnk@uscnk.com ELISA Kit for Human Creatine Kinase,Mitochondrial 1A (CKMT1A) Instruction manual
Uscn Life Science Inc. Wuhan Website: www.uscnk.com Phone: +86 27 84259552 Fax: +86 27 84259551 E-mail: uscnk@uscnk.com ELISA Kit for Human Creatine Kinase,Mitochondrial 1A (CKMT1A) Instruction manual
Mouse Keyhole Limpet Hemocyanin antibody(igm) ELISA Kit
 Mouse Keyhole Limpet Hemocyanin antibody(igm) ELISA Kit Catalog No. MBS702810 (96 tests) This immunoassay kit allows for the in vitro semi-quantitative determination of mouse KLH(IgM)antibody concentrations
Mouse Keyhole Limpet Hemocyanin antibody(igm) ELISA Kit Catalog No. MBS702810 (96 tests) This immunoassay kit allows for the in vitro semi-quantitative determination of mouse KLH(IgM)antibody concentrations
INMA LABORATORY MANUAL
 INMA LABORATORY MANUAL NUTRITION BIOMARKERS Cohorte-VALENCIA This manual describes the collection, separation, storage and transport of blood samples for the INMA study. Careful collection of samples is
INMA LABORATORY MANUAL NUTRITION BIOMARKERS Cohorte-VALENCIA This manual describes the collection, separation, storage and transport of blood samples for the INMA study. Careful collection of samples is
Revised 3 Oct. 2012 rm (Vers. 16.1)
 Intended Use This kit is intended for Research Use Only. Not for use in diagnostic procedures. The DRG Dengue Virus IgM Enzyme Immunoassay Kit provides materials for the qualitative and semiquantitative
Intended Use This kit is intended for Research Use Only. Not for use in diagnostic procedures. The DRG Dengue Virus IgM Enzyme Immunoassay Kit provides materials for the qualitative and semiquantitative
LAB 14 ENZYME LINKED IMMUNOSORBENT ASSAY (ELISA)
 STUDENT GUIDE LAB 14 ENZYME LINKED IMMUNOSORBENT ASSAY (ELISA) GOAL The goal of this laboratory lesson is to explain the concepts and technique of enzyme linked immunosorbent assay (ELISA). OBJECTIVES
STUDENT GUIDE LAB 14 ENZYME LINKED IMMUNOSORBENT ASSAY (ELISA) GOAL The goal of this laboratory lesson is to explain the concepts and technique of enzyme linked immunosorbent assay (ELISA). OBJECTIVES
ab185915 Protein Sumoylation Assay Ultra Kit
 ab185915 Protein Sumoylation Assay Ultra Kit Instructions for Use For the measuring in vivo protein sumoylation in various samples This product is for research use only and is not intended for diagnostic
ab185915 Protein Sumoylation Assay Ultra Kit Instructions for Use For the measuring in vivo protein sumoylation in various samples This product is for research use only and is not intended for diagnostic
Dot Blot Analysis. Teacher s Guidebook. (Cat. # BE 502) think proteins! think G-Biosciences www.gbiosciences.com
 PR110 G-Biosciences 1-800-628-7730 1-314-991-6034 technical@gbiosciences.com A Geno Technology, Inc. (USA) brand name Dot Blot Analysis Teacher s Guidebook (Cat. # BE 502) think proteins! think G-Biosciences
PR110 G-Biosciences 1-800-628-7730 1-314-991-6034 technical@gbiosciences.com A Geno Technology, Inc. (USA) brand name Dot Blot Analysis Teacher s Guidebook (Cat. # BE 502) think proteins! think G-Biosciences
PCR and Sequencing Reaction Clean-Up Kit (Magnetic Bead System) 50 preps Product #60200
 3430 Schmon Parkway Thorold, ON, Canada L2V 4Y6 Phone: 866-667-4362 (905) 227-8848 Fax: (905) 227-1061 Email: techsupport@norgenbiotek.com PCR and Sequencing Reaction Clean-Up Kit (Magnetic Bead System)
3430 Schmon Parkway Thorold, ON, Canada L2V 4Y6 Phone: 866-667-4362 (905) 227-8848 Fax: (905) 227-1061 Email: techsupport@norgenbiotek.com PCR and Sequencing Reaction Clean-Up Kit (Magnetic Bead System)
Cancer Antigen CA125 Human ELISA Kit
 ab108653 Cancer Antigen CA125 Human ELISA Kit Instructions for Use For the quantitative measurement of Human Cancer Antigen CA125 concentrations in serum. This product is for research use only and is not
ab108653 Cancer Antigen CA125 Human ELISA Kit Instructions for Use For the quantitative measurement of Human Cancer Antigen CA125 concentrations in serum. This product is for research use only and is not
ELISA-VIDITEST anti-chlamydia trachomatis IgM
 1023 ELISA-VIDITEST anti-chlamydia trachomatis IgM Instruction manual PRODUCER: VIDIA Ltd., Nad Safinou II 365, Vestec, 252 42 Jesenice, Czech Republic, tel.: +420261 090565, www.vidia.cz 1. TITLE: ELISA-VIDITEST
1023 ELISA-VIDITEST anti-chlamydia trachomatis IgM Instruction manual PRODUCER: VIDIA Ltd., Nad Safinou II 365, Vestec, 252 42 Jesenice, Czech Republic, tel.: +420261 090565, www.vidia.cz 1. TITLE: ELISA-VIDITEST
Bovine Vitamin B12 (VB12) ELISA Kit
 Bovine Vitamin B12 (VB12) ELISA Kit Catalog Number. CSB-E14089B For the quantitative determination of endogenic bovine vitamin B12 (VB12) concentrations in serum, plasma, tissue homogenates. This package
Bovine Vitamin B12 (VB12) ELISA Kit Catalog Number. CSB-E14089B For the quantitative determination of endogenic bovine vitamin B12 (VB12) concentrations in serum, plasma, tissue homogenates. This package
Inflammatory Bowel Disease
 Inflammatory Bowel Disease Laboratory Support of Diagnosis and Management CLINICAL BACKGROUND Inflammatory bowel disease (IBD), which includes Crohn disease (CD) and ulcerative colitis (UC), is characterized
Inflammatory Bowel Disease Laboratory Support of Diagnosis and Management CLINICAL BACKGROUND Inflammatory bowel disease (IBD), which includes Crohn disease (CD) and ulcerative colitis (UC), is characterized
Collection Instructions for ARUP Drug Screen Kit #49204
 Collection Instructions for ARUP Drug Screen Kit #49204 Manufacturer Purpose of Kit / Reagent (tests) Kit / Reagent Contents (list components) Other Supplies Needed ARUP Laboratories For Point of Collection
Collection Instructions for ARUP Drug Screen Kit #49204 Manufacturer Purpose of Kit / Reagent (tests) Kit / Reagent Contents (list components) Other Supplies Needed ARUP Laboratories For Point of Collection
Rat creatine kinase MM isoenzyme (CK-MM) ELISA Kit
 Rat creatine kinase MM isoenzyme (CK-MM) ELISA Kit Catalog Number. CSB-E14405r For the quantitative determination of rat creatine kinase MM isoenzyme (CK-MM) concentrations in serum, plasma and tissue
Rat creatine kinase MM isoenzyme (CK-MM) ELISA Kit Catalog Number. CSB-E14405r For the quantitative determination of rat creatine kinase MM isoenzyme (CK-MM) concentrations in serum, plasma and tissue
Rat Fibronectin ELISA Kit
 AssayMax TM Rat Fibronectin ELISA Kit Assaypro LLC 3400 Harry S Truman Blvd St. Charles, MO 63301 T (636) 447-9175 F (636) 395-7419 www.assaypro.com For any questions regarding troubleshooting or performing
AssayMax TM Rat Fibronectin ELISA Kit Assaypro LLC 3400 Harry S Truman Blvd St. Charles, MO 63301 T (636) 447-9175 F (636) 395-7419 www.assaypro.com For any questions regarding troubleshooting or performing
Standard Mixture. TOF/TOF Calibration Mixture. Calibration Mixture 1 (Cal Mix 1, 1:10) Calibration Mixture 1 (Cal Mix 1, 1:100)
 Mass Standards Kit for Calibration of AB SCIEX TOF/TOF Instruments Protocol 1 Product Description The Mass Standards Kit includes reagents needed to test instrument function, optimize instrument parameters,
Mass Standards Kit for Calibration of AB SCIEX TOF/TOF Instruments Protocol 1 Product Description The Mass Standards Kit includes reagents needed to test instrument function, optimize instrument parameters,
This is a Waived Complexity test for Urine, and Moderately Complex for Serum.
 Sure-Vue Serum/Urine hcg-stat CLSI + More Packet Laboratory Name: Laboratory Address: Date of this packet: Sure-Vue Serum/Urine hcg-stat Laboratory Procedure This procedure is intended to provide a ready
Sure-Vue Serum/Urine hcg-stat CLSI + More Packet Laboratory Name: Laboratory Address: Date of this packet: Sure-Vue Serum/Urine hcg-stat Laboratory Procedure This procedure is intended to provide a ready
PROCEDURE NO. POC.514.01 LBH. Printed copies are for reference only. Please refer to the electronic copy for the latest version.
 Department Of Pathology POC.514.01- Blood Glucose Monitoring Accu-Chek Inform II Procedure-LBH Version# 1 Department PROCEDURE NO. PAGE NO. Point-of-Care Testing POC.514.01 LBH 1 OF 7 Printed copies are
Department Of Pathology POC.514.01- Blood Glucose Monitoring Accu-Chek Inform II Procedure-LBH Version# 1 Department PROCEDURE NO. PAGE NO. Point-of-Care Testing POC.514.01 LBH 1 OF 7 Printed copies are
Gastrointestinal Bleeding
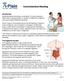 Gastrointestinal Bleeding Introduction Gastrointestinal bleeding is a symptom of many diseases rather than a disease itself. A number of different conditions can cause gastrointestinal bleeding. Some causes
Gastrointestinal Bleeding Introduction Gastrointestinal bleeding is a symptom of many diseases rather than a disease itself. A number of different conditions can cause gastrointestinal bleeding. Some causes
Aptima Specimen Transfer Kit
 Aptima Specimen Transfer Kit For Use with ThinPrep and SurePath Liquid Cytology Specimens Intended Use For in vitro diagnostic use. For US export only. The Aptima Specimen Transfer kit is only for use
Aptima Specimen Transfer Kit For Use with ThinPrep and SurePath Liquid Cytology Specimens Intended Use For in vitro diagnostic use. For US export only. The Aptima Specimen Transfer kit is only for use
EXERCISE 5: ERYTHROCYTES SEDIMENTATION RATE - ESR, SED RATE
 EXERCISE 5: ERYTHROCYTES SEDIMENTATION RATE - ESR, SED RATE Textbook: Skills: None 10 points Objectives: 1. State the principle of the Erythrocytes Sedimentation Rate - ESR. 2. List two factors which may
EXERCISE 5: ERYTHROCYTES SEDIMENTATION RATE - ESR, SED RATE Textbook: Skills: None 10 points Objectives: 1. State the principle of the Erythrocytes Sedimentation Rate - ESR. 2. List two factors which may
FBS06 P30 Antigen Test for the Presence of Seminal Fluid
 FBS06 P30 Antigen Test for the Presence of Seminal Fluid Table of Contents 1. Scope 2. Background 3. Safety 4. Materials Required 5. Standards and Controls 6. Calibration 7. Procedures 8. Sampling 9. Calculations
FBS06 P30 Antigen Test for the Presence of Seminal Fluid Table of Contents 1. Scope 2. Background 3. Safety 4. Materials Required 5. Standards and Controls 6. Calibration 7. Procedures 8. Sampling 9. Calculations
ab178663 Free Testosterone ELISA Kit
 ab178663 Free Testosterone ELISA Kit Instructions for Use A competitive immunoenzymatic assay for the quantitative measurement of Free Testosterone in serum and plasma (citrate). This product is for research
ab178663 Free Testosterone ELISA Kit Instructions for Use A competitive immunoenzymatic assay for the quantitative measurement of Free Testosterone in serum and plasma (citrate). This product is for research
ELISA-VIDITEST anti-cmv IgM capture
 1023 ELISA-VIDITEST anti-cmv IgM capture Cat. No. ODZ-162 Instruction manual Producer: VIDIA spol. s r.o.., Nad Safinou II 365, Vestec, 252 42 Jesenice, Czech Republic, Tel.: +420 261 090 565, Web: www.vidia.cz,
1023 ELISA-VIDITEST anti-cmv IgM capture Cat. No. ODZ-162 Instruction manual Producer: VIDIA spol. s r.o.., Nad Safinou II 365, Vestec, 252 42 Jesenice, Czech Republic, Tel.: +420 261 090 565, Web: www.vidia.cz,
2.3. The management in each HCF shall be responsible for ensuring good waste management practices in their premises.
 1. PURPOSE Health-care activities lead to production of medical waste that may lead to adverse health effects. Most of this waste is not more dangerous than regular household waste. However, some types
1. PURPOSE Health-care activities lead to production of medical waste that may lead to adverse health effects. Most of this waste is not more dangerous than regular household waste. However, some types
Human TNF- Product # 453310
 PRODUCT INFORMATION AND MANUAL Enzyme-linked immunosorbent assay for quantitative detection of human TNF- For Research Use Only Not for diagnostic or therapeutic procedures. 96 Wells Human TNF- Product
PRODUCT INFORMATION AND MANUAL Enzyme-linked immunosorbent assay for quantitative detection of human TNF- For Research Use Only Not for diagnostic or therapeutic procedures. 96 Wells Human TNF- Product
Bloodborne Pathogens Program Revised July, 5 2012
 Bloodborne Pathogens Program Revised July, 5 2012 Page 1 of 16 Table of Contents 1.0 INTRODUCTION...3 1.1 Purpose...3 1.2 Policy.3 2.0 EXPOSURE CONTROL METHODS 4 2.1 Universal Precautions.4 2.2 Engineering
Bloodborne Pathogens Program Revised July, 5 2012 Page 1 of 16 Table of Contents 1.0 INTRODUCTION...3 1.1 Purpose...3 1.2 Policy.3 2.0 EXPOSURE CONTROL METHODS 4 2.1 Universal Precautions.4 2.2 Engineering
Environmental Water Testing: Surface Water, Groundwater, Hard Water, Wastewater, & Seawater
 Document: AND Sol Env 08 2013 Environmental Water Testing: Surface Water, Groundwater, Hard Water, Wastewater, & Seawater Matrix specific sample preparation and testing methods for environmental waters
Document: AND Sol Env 08 2013 Environmental Water Testing: Surface Water, Groundwater, Hard Water, Wastewater, & Seawater Matrix specific sample preparation and testing methods for environmental waters
WHICH SAMPLES SHOULD BE SUBMITTED WHEN LYMPHOID NEOPLASIA IS SUSPECTED?
 WHICH SAMPLES SHOULD BE SUBMITTED WHEN LYMPHOID NEOPLASIA IS SUSPECTED? Which test should be submitted? The answer to this depends on the clinical signs, and the diagnostic question you are asking. If
WHICH SAMPLES SHOULD BE SUBMITTED WHEN LYMPHOID NEOPLASIA IS SUSPECTED? Which test should be submitted? The answer to this depends on the clinical signs, and the diagnostic question you are asking. If
Biohazardous, Medical & Biological Waste Guidance Chart
 CSULA Environmental Health and Safety Biohazardous, Medical & Biological Waste Guidance Chart The chart below provides information on how to handle most, if not all, of the items that frequently are collectively
CSULA Environmental Health and Safety Biohazardous, Medical & Biological Waste Guidance Chart The chart below provides information on how to handle most, if not all, of the items that frequently are collectively
Colorectal Cancer: Preventable, Beatable, Treatable. American Cancer Society
 Colorectal Cancer: Preventable, Beatable, Treatable American Cancer Society Reviewed January 2013 What we ll be talking about How common is colorectal cancer? What is colorectal cancer? What causes it?
Colorectal Cancer: Preventable, Beatable, Treatable American Cancer Society Reviewed January 2013 What we ll be talking about How common is colorectal cancer? What is colorectal cancer? What causes it?
Thrombin Generation Assay
 Thrombin Generation Assay Kit insert Version: February 2013 Summary Thrombin is a key enzyme of the coagulation cascade. Its measurement gives direct information about the thrombogenicity of a biomaterial
Thrombin Generation Assay Kit insert Version: February 2013 Summary Thrombin is a key enzyme of the coagulation cascade. Its measurement gives direct information about the thrombogenicity of a biomaterial
HiPer Blood Grouping Teaching Kit
 HiPer Blood Grouping Teaching Kit Product Code: HTI008 Number of experiments that can be performed: 100 Duration of Experiment: Protocol: 15 minutes Storage Instructions: The kit is stable for 6 months
HiPer Blood Grouping Teaching Kit Product Code: HTI008 Number of experiments that can be performed: 100 Duration of Experiment: Protocol: 15 minutes Storage Instructions: The kit is stable for 6 months
Package leaflet: Information for the patient. Entyvio 300 mg powder for concentrate for solution for infusion vedolizumab
 Package leaflet: Information for the patient Entyvio 300 mg powder for concentrate for solution for infusion vedolizumab This medicine is subject to additional monitoring. This will allow quick identification
Package leaflet: Information for the patient Entyvio 300 mg powder for concentrate for solution for infusion vedolizumab This medicine is subject to additional monitoring. This will allow quick identification
Content Sheet 5-1: Overview of Sample Management
 Content Sheet 5-1: Overview of Management Role in quality management system management is a part of process control, one of the essentials of a quality management system. The quality of the work a laboratory
Content Sheet 5-1: Overview of Management Role in quality management system management is a part of process control, one of the essentials of a quality management system. The quality of the work a laboratory
Antibiotic-Associated Diarrhea, Clostridium difficile- Associated Diarrhea and Colitis
 Antibiotic-Associated Diarrhea, Clostridium difficile- Associated Diarrhea and Colitis ANTIBIOTIC-ASSOCIATED DIARRHEA Disturbance of the normal colonic microflora Leading to alterations in bacterial degradation
Antibiotic-Associated Diarrhea, Clostridium difficile- Associated Diarrhea and Colitis ANTIBIOTIC-ASSOCIATED DIARRHEA Disturbance of the normal colonic microflora Leading to alterations in bacterial degradation
How To Store An Acam2000 Vaccine
 ACAM2000 Smallpox (Vaccinia) Vaccine, Live Deployment Guidance - Acambis Inc. Revision: October 2008 Information contained in this guidance will also be available through Acambis, in the event that questions,
ACAM2000 Smallpox (Vaccinia) Vaccine, Live Deployment Guidance - Acambis Inc. Revision: October 2008 Information contained in this guidance will also be available through Acambis, in the event that questions,
PCD Europe, Jan 2007. Interpretation of Hemoccult
 Interpretation of Hemoccult Is this positive? No! Yes! In the German Programme for Early Detection of Colorectal Canceronly 1 % of women and 1.5 % of men of the millions screened each year have a positive
Interpretation of Hemoccult Is this positive? No! Yes! In the German Programme for Early Detection of Colorectal Canceronly 1 % of women and 1.5 % of men of the millions screened each year have a positive
Inform II. Quick Reference Guide. for Healthcare Professionals BLOOD GLUCOSE MONITORING SYSTEM
 Inform II BLOOD GLUCOSE MONITORING SYSTEM Quick Reference Guide for Healthcare Professionals 2 Table of Contents Important... 4 General Information... 6 ACCU-CHEK Inform II Meter...10 How to Perform Patient
Inform II BLOOD GLUCOSE MONITORING SYSTEM Quick Reference Guide for Healthcare Professionals 2 Table of Contents Important... 4 General Information... 6 ACCU-CHEK Inform II Meter...10 How to Perform Patient
Cancer Antigen CA 125
 Principle of the Test Cancer Antigen CA 125 ADVIA Centaur System The ADVIA Centaur CA 125 II assay is a two-site sandwich immunoassay using direct chemiluminometric technology, which uses two monoclonal
Principle of the Test Cancer Antigen CA 125 ADVIA Centaur System The ADVIA Centaur CA 125 II assay is a two-site sandwich immunoassay using direct chemiluminometric technology, which uses two monoclonal
Rat creatine kinase MM isoenzyme (CK-MM) ELISA Kit
 Rat creatine kinase MM isoenzyme (CK-MM) ELISA Kit Catalog Number. CSB-E14405r For the quantitative determination of rat creatine kinase MM isoenzyme (CK-MM) concentrations in serum, plasma, tissue homogenates.
Rat creatine kinase MM isoenzyme (CK-MM) ELISA Kit Catalog Number. CSB-E14405r For the quantitative determination of rat creatine kinase MM isoenzyme (CK-MM) concentrations in serum, plasma, tissue homogenates.
How Does a Doctor Test for AIDS?
 Edvo-Kit #S-70 How Does a Doctor Test for AIDS? S-70 Experiment Objective: The Human Immunodefi ciency Virus (HIV) is an infectious agent that causes Acquired Immunodefi ciency Syndrome (AIDS) in humans.
Edvo-Kit #S-70 How Does a Doctor Test for AIDS? S-70 Experiment Objective: The Human Immunodefi ciency Virus (HIV) is an infectious agent that causes Acquired Immunodefi ciency Syndrome (AIDS) in humans.
What You Need to Know About Collecting QuantiFERON (QFT) TB Gold In-Tube Samples
 What You Need to Know About Collecting QuantiFERON (QFT) TB Gold In-Tube Samples I. What is QuantiFERON (QFT) TB Gold In-Tube? II. III. QFT is an interferon-gamma release assay (IGRA). QFT is a blood test.
What You Need to Know About Collecting QuantiFERON (QFT) TB Gold In-Tube Samples I. What is QuantiFERON (QFT) TB Gold In-Tube? II. III. QFT is an interferon-gamma release assay (IGRA). QFT is a blood test.
QS114. NICE quality standard for irritable bowel syndrome in adults (QS114)
 NICE quality standard for irritable bowel syndrome in adults (QS114) QS114 NICE approved the reproduction of its content for this booklet. The production of this booklet is sponsored by Thermo Fisher Scientific,
NICE quality standard for irritable bowel syndrome in adults (QS114) QS114 NICE approved the reproduction of its content for this booklet. The production of this booklet is sponsored by Thermo Fisher Scientific,
ABO-Rh Blood Typing With Synthetic Blood
 70-0101 ABO-Rh Blood Typing With Synthetic Blood Teacher s Manual World-Class Support for Science & Math The ability to type blood is an invaluable tool in the fields of medicine and criminology. Using
70-0101 ABO-Rh Blood Typing With Synthetic Blood Teacher s Manual World-Class Support for Science & Math The ability to type blood is an invaluable tool in the fields of medicine and criminology. Using
PROCEDURE COVERSHEET. Document Section: CHEMISTRY Author: JOANNE WONG Date Authored: JAN 2011. In Use Date: 2/1/2011 Discontinued Date:
 PROCEDURE COVERSHEET Procedure Title: ph (Urine, Body Fluid, Platelet Samples) Document Section: CHEMISTRY Author: JOANNE WONG Date Authored: JAN 2011 Laboratory Director: TED KURTZ, MD (PARNASSUS & CHINA
PROCEDURE COVERSHEET Procedure Title: ph (Urine, Body Fluid, Platelet Samples) Document Section: CHEMISTRY Author: JOANNE WONG Date Authored: JAN 2011 Laboratory Director: TED KURTZ, MD (PARNASSUS & CHINA
Laparoscopic Surgery of the Colon and Rectum (Large Intestine) A Simple Guide to Help Answer Your Questions
 Laparoscopic Surgery of the Colon and Rectum (Large Intestine) A Simple Guide to Help Answer Your Questions What are the Colon and Rectum? The colon and rectum together make up the large intestine. After
Laparoscopic Surgery of the Colon and Rectum (Large Intestine) A Simple Guide to Help Answer Your Questions What are the Colon and Rectum? The colon and rectum together make up the large intestine. After
Probiotics for the Treatment of Adult Gastrointestinal Disorders
 Probiotics for the Treatment of Adult Gastrointestinal Disorders Darren M. Brenner, M.D. Division of Gastroenterology Northwestern University, Feinberg School of Medicine Chicago, Illinois What are Probiotics?
Probiotics for the Treatment of Adult Gastrointestinal Disorders Darren M. Brenner, M.D. Division of Gastroenterology Northwestern University, Feinberg School of Medicine Chicago, Illinois What are Probiotics?
APPENDIX N BULK MILK TANKER SCREENING TEST FORM
 APPENDIX N BULK MILK TANKER SCREENING TEST FORM IDEXX - NEW SNAP BETA-LACTAM TEST (Raw Commingled Cow, Raw Commingled Camel, and Raw Commingled Goat Milk) IMS # 9-I1 [Unless otherwise stated all tolerances
APPENDIX N BULK MILK TANKER SCREENING TEST FORM IDEXX - NEW SNAP BETA-LACTAM TEST (Raw Commingled Cow, Raw Commingled Camel, and Raw Commingled Goat Milk) IMS # 9-I1 [Unless otherwise stated all tolerances
WHO Prequalification of Diagnostics Programme PUBLIC REPORT
 WHO Prequalification of Diagnostics Programme PUBLIC REPORT Product: ABON HIV 1/2/O Tri-Line Human Immunodeficiency Virus Rapid Test Device Number: PQDx 0141-051-00 Abstract ABON HIV 1/2/O Tri-Line Human
WHO Prequalification of Diagnostics Programme PUBLIC REPORT Product: ABON HIV 1/2/O Tri-Line Human Immunodeficiency Virus Rapid Test Device Number: PQDx 0141-051-00 Abstract ABON HIV 1/2/O Tri-Line Human
Vitamin B12. ADVIA Centaur System
 Vitamin B12 Principle of the Test ADVIA Centaur System The ADVIA Centaur VB12 assay is a competitive immunoassay using direct chemiluminescent technology in which vitamin B 12 from the patient sample competes
Vitamin B12 Principle of the Test ADVIA Centaur System The ADVIA Centaur VB12 assay is a competitive immunoassay using direct chemiluminescent technology in which vitamin B 12 from the patient sample competes
Data Sheet. PD-L1:B7-1[Biotinylated] Inhibitor Screening Assay Kit Catalog # 72026 Size: 96 reactions
![Data Sheet. PD-L1:B7-1[Biotinylated] Inhibitor Screening Assay Kit Catalog # 72026 Size: 96 reactions Data Sheet. PD-L1:B7-1[Biotinylated] Inhibitor Screening Assay Kit Catalog # 72026 Size: 96 reactions](/thumbs/40/21461210.jpg) Data Sheet PD-L1:B7-1[Biotinylated] Inhibitor Screening Assay Kit Catalog # 72026 Size: 96 reactions BACKGROUND: There have been a number of studies defining a role for PD-L1 in the regulation of immune
Data Sheet PD-L1:B7-1[Biotinylated] Inhibitor Screening Assay Kit Catalog # 72026 Size: 96 reactions BACKGROUND: There have been a number of studies defining a role for PD-L1 in the regulation of immune
WHO Prequalification of Diagnostics Programme PUBLIC REPORT. Product: Genscreen ULTRA HIV Ag-Ab Number: PQDx 0096-031-00. Abstract
 WHO Prequalification of Diagnostics Programme PUBLIC REPORT Product: Genscreen ULTRA HIV Ag-Ab Number: PQDx 0096-031-00 Abstract Genscreen ULTRA HIV Ag-Ab with product codes 72386 and 72388, manufactured
WHO Prequalification of Diagnostics Programme PUBLIC REPORT Product: Genscreen ULTRA HIV Ag-Ab Number: PQDx 0096-031-00 Abstract Genscreen ULTRA HIV Ag-Ab with product codes 72386 and 72388, manufactured
IP-10 ELISA KIT INTENDED USE
 Issue 1 15 September 2008 Page 1 /6 IP-10 ELISA KIT INTENDED USE The IP-10 ELISA is to be used for the in-vitro quantitative determination of human interferon gamma inducible 10kD protein, in cell culture
Issue 1 15 September 2008 Page 1 /6 IP-10 ELISA KIT INTENDED USE The IP-10 ELISA is to be used for the in-vitro quantitative determination of human interferon gamma inducible 10kD protein, in cell culture
The faecal occult blood (FOB) test
 The faecal occult blood (FOB) test This information is an extract from the booklet, Understanding bowel cancer screening. You may find the full booklet helpful. We can send you a copy free see page 6.
The faecal occult blood (FOB) test This information is an extract from the booklet, Understanding bowel cancer screening. You may find the full booklet helpful. We can send you a copy free see page 6.
