naproxen/esomeprazole 500mg/20mg modified release tablets (Vimovo ) SMC No. (734/11) AstraZeneca UK
|
|
|
- Delphia Pamela McKinney
- 7 years ago
- Views:
Transcription
1 naproxen/esomeprazole 500mg/20mg modified release tablets (Vimovo ) SMC No. (734/11) AstraZeneca UK 07 October 2011 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product and advises NHS Boards and Area Drug and Therapeutic Committees (ADTCs) on its use in NHS Scotland. The advice is summarised as follows: ADVICE: following a full submission naproxen 500mg/esomeprazole 20mg (Vimovo ) is not recommended for use within NHS Scotland. Indication under review: the symptomatic treatment of osteoarthritis (OA), rheumatoid arthritis (RA) and ankylosing spondylitis (AS), in patients who are at risk for developing nonsteroidal anti-inflammatory drug (NSAID)-associated gastric and/or duodenal ulcers and where treatment with lower doses of naproxen or of other NSAIDs is not considered sufficient. Studies have demonstrated that combined naproxen/esomeprazole was associated with a lower incidence of endoscopic gastric ulcers than NSAID alone and similar improvements in pain and functioning compared to a cyclo-oxygenase-2 selective inhibitor. The submitting company did not present a sufficiently robust economic analysis to gain acceptance by SMC. Overleaf is the detailed advice on this product. Chairman, Scottish Medicines Consortium Published 07 November
2 Indication Symptomatic treatment of osteoarthritis (OA), rheumatoid arthritis (RA) and ankylosing spondylitis (AS), in patients who are at risk for developing non-steroidal anti-inflammatory drug (NSAID)-associated gastric and/or duodenal ulcers and where treatment with lower doses of naproxen or of other NSAIDs is not considered sufficient. Dosing Information One tablet to be swallowed whole twice daily, taken 30 minutes before food. Product availability date 17 January 2011 Summary of evidence on comparative efficacy This is a fixed dose, modified-release combination preparation of two established medicines; the NSAID naproxen 500mg enteric-coated (gastro-resistant) and the proton pump inhibitor (PPI) esomeprazole 20mg. The preparation provides a sequential delivery of esomeprazole and naproxen,with the instant release esomeprazole rapidly absorbed and the enteric-coated naproxen released shortly afterwards. The submitting company has requested that SMC consider naproxen/esomeprazole when positioned for use in a sub-population of the licensed indication, namely for patients aged 65 years as increased age is an established risk factor for upper gastrointestinal adverse effects with NSAIDs. Two identical, randomized, double-blind, multicentre, phase III studies compared naproxen/esomeprazole with naproxen alone in 434 and 420 patients with OA, RA, AS or other medical conditions expected to require 6 months NSAID therapy. Eligible patients were aged 50 years (or 18 to 49 years with a history of uncomplicated gastric or duodenal ulcer in the previous 5 years) and were Helicobacter pylori negative. Patients were randomised to receive naproxen/esomeprazole 500mg/20mg or naproxen 500mg twice daily for 6 months with stratification for use of aspirin ( 325mg daily). The primary endpoint was the cumulative incidence of endoscopic gastric ulcers. In both studies this was significantly lower in patients who received the naproxen/esomeprazole combination than in patients who received naproxen alone: 4.1% versus 23% in one study and 7.1% versus 24% in the other. Pooled analysis also demonstrated that the incidence of endoscopic gastric ulcers was significantly lower in naproxen/esomeprazole than naproxen patients in the subgroups of patients receiving aspirin (n=201) and not receiving aspirin (n=653). The incidence of duodenal ulcers was also significantly reduced in the naproxen/esomeprazole groups in both studies. Two identical, randomised, double-blind, placebo-controlled, multicentre, phase III studies compared naproxen/esomeprazole with celecoxib in 614 and 610 patients aged 50 years with symptomatic knee OA. Patients were randomised to receive naproxen/esomeprazole 500mg/20mg twice daily, celecoxib 200mg daily or placebo for 12 weeks. These studies demonstrated the non-inferiority of naproxen/esomeprazole compared to celecoxib based on 2
3 three co-primary endpoints (mean change from baseline in Western Ontario and McMaster Osteoarthritis Index [WOMAC] pain, WOMAC function and patient global assessment [PGA] of OA). Summary of evidence on comparative safety Safety data from the four phase III studies described previously raised no new safety concerns. A single-arm, open-label, phase III study assessed the long-term safety of naproxen/esomeprazole over 12 months. This enrolled 239 patients aged 50 years (or 18 to 49 years with a history of uncomplicated gastric or duodenal ulcer in the previous 5 years) with OA, RA or other condition requiring daily NSAID treatment to receive naproxen/esomeprazole 500mg/20mg twice daily for 12 months. The results identified no new safety issues, gastrointestinal or cardiovascular. Adverse events were reported in 73% of patients and these were treatment-related in 28%, lead to discontinuation in 19% and were serious in 5.4%. The most commonly reported adverse event was dyspepsia (7.9%). Summary of clinical effectiveness issues Long-term use of NSAIDs is associated with a marked risk of upper gastrointestinal adverse effects including gastric ulcers, perforations and bleeds. Treatment guidelines from SIGN and NICE recommend that patients receiving NSAIDs, particularly those with any upper gastrointestinal risk factors, should be co-prescribed a gastroprotective agent such as a PPI. This fixed dose combination of naproxen plus esomeprazole is licensed for use in patients who are at risk for developing NSAID-associated gastric and/or duodenal ulcers and where treatment with lower doses of naproxen or of other NSAIDs is not considered sufficient. The submitting company has requested that SMC consider naproxen/esomeprazole in a sub-population of the licensed indication, namely those patients aged 65 years. Clinical efficacy data in this cohort are limited. Around one third of all patients included in the studies were 65 years but outcomes in this patient population were not reported separately. The four phase III studies described demonstrated lower rates of endoscopic gastric ulcers with the naproxen/esomeprazole combination compared with naproxen alone and non-inferiority compared with celecoxib in terms of pain and functioning. However, there are no comparative data versus likely comparators in clinical practice (i.e. NSAID plus PPI prescribed separately). The claimed advantage of this combination product is the guaranteed adherence to gastroprotection from the PPI each time the patient takes a dose of NSAID. The company suggests that this guaranteed adherence would be expected to lead to a reduction in NSAIDinduced adverse gastrointestinal events, such as hospital admissions due to gastric ulcers. Although this is plausible, the clinical studies do not provide evidence to support this potential advantage. The fixed dose combination requires patients to take a dose of esomeprazole 20mg twice daily, which is greater than the recommended dose for single-agent esomeprazole (20mg once daily) as a gastroprotectant. The potential advantages of guaranteed adherence to gastroprotection therefore needs to be balanced against the potential risks associated with patient exposure to a higher dose of PPI. 3
4 Summary of comparative health economic evidence The submitting company provided a cost-utility analysis comparing naproxen/esomeprazole with naproxen 1,000mg plus generic proton pump inhibitor (PPI), diclofenac 150mg plus PPI and ibuprofen 2,400mg plus PPI in patients with OA, RA and AS who are at risk for developing NSAID-associated gastric and/or duodenal ulcers and where treatment with lower doses of naproxen or other NSAIDs is not considered sufficient. The generic PPI component of the comparator treatments was assumed to be a weighted average of omeprazole (65%) and lansoprazole (35%) to reflect current prescribing in Scotland. The focus of the submission was in line with the company s proposed positioning for the product i.e. in patients aged 65 years on the basis that increased age is a risk factor for NSAID-associated upper GI adverse events. A lifetime Markov model was used based on the model developed by NICE for the osteoarthritis guideline. The main focus of the model was upper-gi and cardiovascular adverse events associated with long-term NSAID therapy on the assumption that naproxen/esomeprazole and the comparators were equally effective in terms of managing OA, RA and AS pain. As no comparative data were available, the adverse event rates were based on the data used in the NICE model with some adjustments made to account for dose, patient baseline risk, the addition of a PPI and the effect of sub-optimal adherence. The base case analysis assumed 73% adherence to a PPI in the comparator arms of the model with lower adherence assumed to result in an increase in upper GI adverse events. Quality of life loss and resource use associated with adverse events were also taken from the NICE model. The results of the analysis were: Naproxen/esomeprazole versus Incremental cost Incremental Quality adjusted life years (QALYs) Incremental cost per QALY Naproxen + generic PPI ,465 Diclofenac + generic PPI ,817 Ibuprofen + generic PPI ,155 The key limitations of the analysis were: No data were presented comparing naproxen/esomeprazole with any of the comparators. In addition, a formal indirect comparison was not conducted. The adverse event rates taken from the NICE model for each of the comparator treatments were not based on statistically significant differences. When the nonsignificant differences were removed the incremental cost-effectiveness ratios (ICERs) changed to 18k per QALY vs ibuprofen and 8k per QALY versus diclofenac. The patient population in which the adverse event data were obtained did not reflect the patient group being considered in the submission. As such, some adjustments had to be made to the NICE data for use in the model. The lower adherence rate with the NSAID + generic PPI treatments was based on an unpublished retrospective study with a number of limitations. The study used the percentage of NSAID-treated days covered by a PPI prescription as a proxy for adherence, the patients in the study were not confined to those aged 65 years and the 4
5 patients were not all on high-dose NSAIDs. As such, the adherence rate used in the model is uncertain. The model results were sensitive to the assumed lower adherence rate of the comparator treatments. A threshold analysis showed that for the comparison with naproxen + generic PPI the cost per QALY increased to 30k when the adherence rate was increased to 0.85 (base case rate was 0.73). In a scenario analysis where the non-significant differences were removed and a more conservative relationship between lower adherence and increase in adverse events was assumed, the ICERs were 32k, 55k and 10k per QALY vs naproxen, ibuprofen and diclofenac respectively. Due to weaknesses of the clinical data and uncertainty associated with the adherence rate used in the model, the economic case has not been demonstrated. Summary of patient and public involvement A Patient Interest Group Submission was received from Arthritis Care in Scotland. Additional information: guidelines and protocols The Scottish Intercollegiate Guidelines Network (SIGN) published guidelines, SIGN 123: Management of early rheumatoid arthritis, in February These guidelines recommend that the lowest NSAID dose compatible with symptom relief should be prescribed and gastroprotection should be introduced for patients with RA at risk of NSAID-associated gastroduodenal ulcers. The National Institute for Clinical Excellence (NICE) published national clinical guidelines for the care and management of osteoarthritis in February 2008, and for the management and treatment of rheumatoid arthritis in adults in February Both guidelines recommend that oral NSAIDs/COX-2 inhibitors be used at the lowest effective dose for the shortest possible period of time and that the lowest acquisition cost PPI be co-prescribed. Additional information: comparators NSAID and proton pump inhibitor (PPI) given separately. Those detailed within the submission are: generic naproxen 1,000 mg daily + generic PPI, generic diclofenac 150 mg daily + generic PPI, generic ibuprofen 2,400 mg daily + generic PPI. SMC has not recommended esomeprazole for the prevention of gastric and duodenal ulcers associated with NSAID therapy in patients at risk. 5
6 Cost of relevant comparators Drug Dose Regimen Cost per year ( ) Naproxen/esomeprazole (Vimovo) 500mg/20mg twice daily 182 NSAID plus PPI prescribed separately Naproxen or naproxen EC plus 500mg twice daily plus 286 to 310 esomeprazole 20mg daily Naproxen or naproxen EC plus 500mg twice daily plus 67 to 91 omeprazole 20mg daily Naproxen or naproxen EC plus 500mg twice daily plus 63 to 97 lansoprazole 15 to 30 mg daily Diclofenac plus esomeprazole 50mg three times daily plus mg daily Diclofenac plus omeprazole 50mg three times daily plus 40 20mg daily Diclofenac plus lansoprazole 50mg three times daily plus 15 to 30mg daily 36 to 46 Doses are for general comparison and do not imply therapeutic equivalence. Costs from evadis on 15 th August Additional information: budget impact The submitting company estimated that around 20,000 patients 65 years are receiving an NSAID and of these around 50% were assumed to be prescribed a PPI. Based on an estimated uptake of 0.1% in year 1 (17 patients) and 7.4% in year 5 (1,304 patients) and a discontinuation rate of 13%, the impact on the medicines budget was estimated at 839 in year 1 rising to 65k in year 5. Assuming some displacement of existing NSAIDs and generic PPIs the net medicines budget impact was estimated at 530 in year 1 and 41k in year 5. 6
7 References The undernoted references were supplied with the submission. AstraZeneca. Summary of Product Characteristics (SmPC): Vimovo tablets. Available from: Minor Jr P, Plachetka J, Orlemans E, Fort J.G, Sostek M. Clinical Trial: evaluation of gastric acid suppression with three doses of immediate-release esomeprazole in the fixed-dose combination of PN400 (naproxen/esomeprazole magnesium) compared with naproxen 500mg and enteric coated esomeprazole 20mg: a randomised, open-label, Phase I study in healthy volunteers. Aliment Pharmacol Ther 2010; 32: Goldstein J.L, Hochberg M.C, Fort J.G, Zhang Y, Hwang C, Sostek M. Clinical Trial: the incidence of NSAID-associated endoscopic gastric ulcers in patients treated with PN400 (naproxen plus esomeprazole magnesium) vs. enteric-coated naproxen alone. Aliment Pharmacol Ther 2010; 32: Hochberg M, Fort J.G, Svensson O, Hwang C, Sostek M. Fixed-dose combined of entericcoated naproxen and immediate-release esomeprazole has comparable efficacy to celecoxib for knee osteoarthritis: two randomised trials. Current Medical Research & Opinion 2011; 27: Sostek M, Fort J.G, Estborn L, Vikman K. Long term safety of naproxen and esomeprazole magnesium fixed-dose combination: phase III study in patients at risk for NSAID-associated gastric ulcers. Current Medical Research & Opinion 2011; 27: The Scottish Intercollegiate Guidelines Network (SIGN), SIGN 123, for The Management of early rheumatoid arthritis in February Available from The National Institute for Clinical Excellence (NICE). National clinical guidelines for the care and management of osteoarthritis. Available from The National Institute for Clinical Excellence (NICE). National clinical guidelines for the management and treatment of rheumatoid arthritis in adults. Available form This assessment is based on data submitted by the applicant company up to and including 16 September Drug prices are those available at the time the papers were issued to SMC for consideration. These have been confirmed from the evadis drug database. SMC is aware that for some hospital-only products national or local contracts may be in place for comparator products that can significantly reduce the acquisition cost to Health Boards. These contract prices are commercial in confidence and cannot be put in the public domain, including via the SMC Detailed Advice Document. Area Drug and Therapeutics Committees and NHS Boards are therefore asked to consider contract pricing when reviewing advice on medicines accepted by SMC. 7
8 Advice context: No part of this advice may be used without the whole of the advice being quoted in full. This advice represents the view of the Scottish Medicines Consortium and was arrived at after careful consideration and evaluation of the available evidence. It is provided to inform the considerations of Area Drug & Therapeutics Committees and NHS Boards in Scotland in determining medicines for local use or local formulary inclusion. This advice does not override the individual responsibility of health professionals to make decisions in the exercise of their clinical judgement in the circumstances of the individual patient, in consultation with the patient and/or guardian or carer. 8
A. Ketorolac*** B. Naproxen C. Ibuprofen D. Celecoxib
 1. A man, 66 years of age, with a history of knee osteoarthritis (OA) is experiencing increasing pain at rest and with physical activity. He also has a history of depression and coronary artery disease.
1. A man, 66 years of age, with a history of knee osteoarthritis (OA) is experiencing increasing pain at rest and with physical activity. He also has a history of depression and coronary artery disease.
boceprevir 200mg capsule (Victrelis ) Treatment experienced patients SMC No. (722/11) Merck, Sharpe and Dohme Ltd
 boceprevir 200mg capsule (Victrelis ) Treatment experienced patients SMC No. (722/11) Merck, Sharpe and Dohme Ltd 09 September 2011 The Scottish Medicines Consortium (SMC) has completed its assessment
boceprevir 200mg capsule (Victrelis ) Treatment experienced patients SMC No. (722/11) Merck, Sharpe and Dohme Ltd 09 September 2011 The Scottish Medicines Consortium (SMC) has completed its assessment
boceprevir 200mg capsule (Victrelis ) Treatment naïve patients SMC No. (723/11) Merck Sharpe and Dohme Ltd
 boceprevir 200mg capsule (Victrelis ) Treatment naïve patients SMC No. (723/11) Merck Sharpe and Dohme Ltd 09 September 2011 The Scottish Medicines Consortium (SMC) has completed its assessment of the
boceprevir 200mg capsule (Victrelis ) Treatment naïve patients SMC No. (723/11) Merck Sharpe and Dohme Ltd 09 September 2011 The Scottish Medicines Consortium (SMC) has completed its assessment of the
Scottish Medicines Consortium
 Scottish Medicines Consortium peginterferon alfa-2a, 135 microgram/ml and 180 microgram/ml pre-filled injections of solution for subcutaneous injection (Pegasys ) No. (561/09) Roche Products Limited 10
Scottish Medicines Consortium peginterferon alfa-2a, 135 microgram/ml and 180 microgram/ml pre-filled injections of solution for subcutaneous injection (Pegasys ) No. (561/09) Roche Products Limited 10
Ask Your Doctor if There May Be a SMARTER CHOICE
 If you have osteoarthritis, rheumatoid arthritis or ankylosing spondylitis, Could Your NSAID Pain Medicine Be Hurting Your Stomach? Ask Your Doctor if There May Be a SMARTER CHOICE 1 of 8 Making Smart
If you have osteoarthritis, rheumatoid arthritis or ankylosing spondylitis, Could Your NSAID Pain Medicine Be Hurting Your Stomach? Ask Your Doctor if There May Be a SMARTER CHOICE 1 of 8 Making Smart
Articles Presented. Journal Presentation. Dr Albert Lo. Dr Albert Lo
 * This presentation is prepared by the author in one s personal capacity for the purpose of academic exchange and does not represent the views of his/her organisations on the topic discussed. Journal Presentation
* This presentation is prepared by the author in one s personal capacity for the purpose of academic exchange and does not represent the views of his/her organisations on the topic discussed. Journal Presentation
ustekinumab 45mg solution for injection in pre-filled syringe (Stelara ) SMC No. (944/14) Janssen-Cilag Ltd
 ustekinumab 45mg solution for injection in pre-filled syringe (Stelara ) SMC No. (944/14) Janssen-Cilag Ltd 07 February 2014 The Scottish Medicines Consortium (SMC) has completed its assessment of the
ustekinumab 45mg solution for injection in pre-filled syringe (Stelara ) SMC No. (944/14) Janssen-Cilag Ltd 07 February 2014 The Scottish Medicines Consortium (SMC) has completed its assessment of the
Cost-effectiveness of dimethyl fumarate (Tecfidera ) for the treatment of adult patients with relapsing remitting multiple sclerosis
 Cost-effectiveness of dimethyl fumarate (Tecfidera ) for the treatment of adult patients with relapsing remitting multiple sclerosis The NCPE has issued a recommendation regarding the cost-effectiveness
Cost-effectiveness of dimethyl fumarate (Tecfidera ) for the treatment of adult patients with relapsing remitting multiple sclerosis The NCPE has issued a recommendation regarding the cost-effectiveness
Gloucestershire Hospitals
 TRUST GUIDELINE In the case of hard copies of this policy the content can only be assured to be accurate on the date of issue marked on the document. The Policy framework requires that the policy is fully
TRUST GUIDELINE In the case of hard copies of this policy the content can only be assured to be accurate on the date of issue marked on the document. The Policy framework requires that the policy is fully
rivaroxaban 15 and 20mg film-coated tablets (Xarelto ) SMC No. (755/12) Bayer PLC
 rivaroxaban 15 and 20mg film-coated tablets (Xarelto ) SMC No. (755/12) Bayer PLC 13 January 2012 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product and advises NHS
rivaroxaban 15 and 20mg film-coated tablets (Xarelto ) SMC No. (755/12) Bayer PLC 13 January 2012 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product and advises NHS
linagliptin, 5mg film-coated tablet (Trajenta ) SMC No. (746/11) Boehringer Ingelheim / Eli Lilly and Company Ltd
 linagliptin, 5mg film-coated tablet (Trajenta ) SMC No. (746/11) Boehringer Ingelheim / Eli Lilly and Company Ltd 09 December 2011 The Scottish Medicines Consortium (SMC) has completed its assessment of
linagliptin, 5mg film-coated tablet (Trajenta ) SMC No. (746/11) Boehringer Ingelheim / Eli Lilly and Company Ltd 09 December 2011 The Scottish Medicines Consortium (SMC) has completed its assessment of
påçííáëü=jéçáåáåéë=`çåëçêíáìã==
 påçííáëü=jéçáåáåéë=`çåëçêíáìã== adalimumab 40mg pre-filled syringe for subcutaneous injection (Humira ) No. (218/05) Abbott New indication: treatment of active and progressive psoriatic arthritis in adults
påçííáëü=jéçáåáåéë=`çåëçêíáìã== adalimumab 40mg pre-filled syringe for subcutaneous injection (Humira ) No. (218/05) Abbott New indication: treatment of active and progressive psoriatic arthritis in adults
fingolimod (as hydrochloride), 0.5mg hard capsules (Gilenya ) SMC No. (763/12) Novartis Pharmaceuticals UK Ltd
 fingolimod (as hydrochloride), 0.5mg hard capsules (Gilenya ) SMC No. (763/12) Novartis Pharmaceuticals UK Ltd 10 February 2012 The Scottish Medicines Consortium (SMC) has completed its assessment of the
fingolimod (as hydrochloride), 0.5mg hard capsules (Gilenya ) SMC No. (763/12) Novartis Pharmaceuticals UK Ltd 10 February 2012 The Scottish Medicines Consortium (SMC) has completed its assessment of the
rivaroxaban 15mg and 20mg film-coated tablets (Xarelto ) SMC No. (852/13) Bayer plc
 rivaroxaban 15mg and 20mg film-coated tablets (Xarelto ) SMC No. (852/13) Bayer plc 08 February 2013 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product and advises
rivaroxaban 15mg and 20mg film-coated tablets (Xarelto ) SMC No. (852/13) Bayer plc 08 February 2013 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product and advises
rifaximin 550mg film-coated tablets (Targaxan ) SMC No. (893/13) Norgine Pharmaceuticals Ltd
 rifaximin 550mg film-coated tablets (Targaxan ) SMC No. (893/13) Norgine Pharmaceuticals Ltd 09 August 2013 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product and
rifaximin 550mg film-coated tablets (Targaxan ) SMC No. (893/13) Norgine Pharmaceuticals Ltd 09 August 2013 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product and
Scottish Medicines Consortium
 Scottish Medicines Consortium insulin glulisine for subcutaneous injection 100 units/ml (Apidra ) No. (298/06) Sanofi Aventis 4 August 2006 The Scottish Medicines Consortium (SMC) has completed its assessment
Scottish Medicines Consortium insulin glulisine for subcutaneous injection 100 units/ml (Apidra ) No. (298/06) Sanofi Aventis 4 August 2006 The Scottish Medicines Consortium (SMC) has completed its assessment
botulinum toxin type A 50, 100, 200 Allergan units/vial (Botox ) SMC No. (916/13) Allergan Ltd
 botulinum toxin type A 50, 100, 200 Allergan units/vial (Botox ) SMC No. (916/13) Allergan Ltd 06 September 2013 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product
botulinum toxin type A 50, 100, 200 Allergan units/vial (Botox ) SMC No. (916/13) Allergan Ltd 06 September 2013 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product
Summary 1. Comparative-effectiveness
 Cost-effectiveness of Delta-9-tetrahydrocannabinol/cannabidiol (Sativex ) as add-on treatment, for symptom improvement in patients with moderate to severe spasticity due to MS who have not responded adequately
Cost-effectiveness of Delta-9-tetrahydrocannabinol/cannabidiol (Sativex ) as add-on treatment, for symptom improvement in patients with moderate to severe spasticity due to MS who have not responded adequately
Scottish Medicines Consortium
 Scottish Medicines Consortium pemetrexed 500mg infusion (Alimta ) No. (192/05) Eli Lilly 8 July 2005 The Scottish Medicines Consortium has completed its assessment of the above product and advises NHS
Scottish Medicines Consortium pemetrexed 500mg infusion (Alimta ) No. (192/05) Eli Lilly 8 July 2005 The Scottish Medicines Consortium has completed its assessment of the above product and advises NHS
Published 09 September 2013 1. 09 August 2013
 aripiprazole 5mg, 10mg, 15mg, 30mg tablets, 10mg, 15mg orodispersible tablets, 1mg/mL oral solution (Abilify ) SMC No. (891/13) Otsuka Pharmaceutical (UK) Ltd 09 August 2013 The Scottish Medicines Consortium
aripiprazole 5mg, 10mg, 15mg, 30mg tablets, 10mg, 15mg orodispersible tablets, 1mg/mL oral solution (Abilify ) SMC No. (891/13) Otsuka Pharmaceutical (UK) Ltd 09 August 2013 The Scottish Medicines Consortium
Scottish Medicines Consortium
 Scottish Medicines Consortium betamethasone valerate 2.25mg medicated plaster (Betesil ) No. (622/10) Genus Pharmaceuticals 09 July 2010 The Scottish Medicines Consortium (SMC) has completed its assessment
Scottish Medicines Consortium betamethasone valerate 2.25mg medicated plaster (Betesil ) No. (622/10) Genus Pharmaceuticals 09 July 2010 The Scottish Medicines Consortium (SMC) has completed its assessment
National Institute for Clinical Excellence
 National Institute for Clinical Excellence 11 Strand London WC2N 5HR Web: www.nice.org.uk N0016 50k 1P July 01 (ABA) Technology Appraisal Guidance - No.27 Guidance on the use of cyclo-oxygenase (Cox) II
National Institute for Clinical Excellence 11 Strand London WC2N 5HR Web: www.nice.org.uk N0016 50k 1P July 01 (ABA) Technology Appraisal Guidance - No.27 Guidance on the use of cyclo-oxygenase (Cox) II
botulinum toxin type A, 50 unit, 100 unit and 200 unit powder for solution for injection (Botox ) SMC No. (692/11) Allergan Ltd
 Resubmission botulinum toxin type A, 50 unit, 100 unit and 200 unit powder for solution for injection (Botox ) SMC No. (692/11) Allergan Ltd 08 March 2013 The Scottish Medicines Consortium (SMC) has completed
Resubmission botulinum toxin type A, 50 unit, 100 unit and 200 unit powder for solution for injection (Botox ) SMC No. (692/11) Allergan Ltd 08 March 2013 The Scottish Medicines Consortium (SMC) has completed
Indication under review: cutaneous treatment of acne vulgaris when comedones, papules and pustules are present.
 Resubmission adapalene 0.1%/benzoyl peroxide 2.5% gel (Epiduo ) SMC No. (682/11) Galderma UK Ltd 07 March 2014 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product
Resubmission adapalene 0.1%/benzoyl peroxide 2.5% gel (Epiduo ) SMC No. (682/11) Galderma UK Ltd 07 March 2014 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product
mirabegron 25mg and 50mg prolonged-release tablets (Betmiga ) SMC No. (862/13) Astellas Pharma Ltd
 mirabegron 25mg and 50mg prolonged-release tablets (Betmiga ) SMC No. (862/13) Astellas Pharma Ltd 05 April 2013 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product
mirabegron 25mg and 50mg prolonged-release tablets (Betmiga ) SMC No. (862/13) Astellas Pharma Ltd 05 April 2013 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product
Natalizumab for the treatment of adults with highly active relapsing remitting multiple sclerosis
 Natalizumab for the treatment of adults with highly active relapsing remitting multiple sclerosis Premeeting briefing This briefing presents major issues arising from the manufacturer s submission, Evidence
Natalizumab for the treatment of adults with highly active relapsing remitting multiple sclerosis Premeeting briefing This briefing presents major issues arising from the manufacturer s submission, Evidence
Survey conducted into patient experience of life with osteoarthritis. A collaboration between Pro Bono Bio, Arthritis Research UK, and LloydsPharmacy
 Survey conducted into patient experience of life with osteoarthritis. A collaboration between Pro Bono Bio, Arthritis Research UK, and LloydsPharmacy Executive summary Over 400 people with joint conditions
Survey conducted into patient experience of life with osteoarthritis. A collaboration between Pro Bono Bio, Arthritis Research UK, and LloydsPharmacy Executive summary Over 400 people with joint conditions
Scottish Medicines Consortium
 Scottish Medicines Consortium sildenafil citrate 20mg tablets (Revatio ) No. (235/06) Pfizer New indication 6 January 2006 The Scottish Medicines Consortium (SMC) has completed its assessment of the above
Scottish Medicines Consortium sildenafil citrate 20mg tablets (Revatio ) No. (235/06) Pfizer New indication 6 January 2006 The Scottish Medicines Consortium (SMC) has completed its assessment of the above
Apixaban for the prevention of stroke and systemic embolism in people with non-valvular atrial fibrillation
 Apixaban for the prevention of stroke and systemic embolism in people with non-valvular atrial fibrillation ERRATUM This report was commissioned by the NIHR HTA Programme as project number 11/49 This document
Apixaban for the prevention of stroke and systemic embolism in people with non-valvular atrial fibrillation ERRATUM This report was commissioned by the NIHR HTA Programme as project number 11/49 This document
Rivaroxaban for the prevention of stroke and systemic embolism in people with atrial fibrillation
 Rivaroxaban for the prevention of stroke and systemic embolism in people with atrial fibrillation Issued: May 2012 guidance.nice.org.uk/ta256 NICE has accredited the process used by the Centre for Health
Rivaroxaban for the prevention of stroke and systemic embolism in people with atrial fibrillation Issued: May 2012 guidance.nice.org.uk/ta256 NICE has accredited the process used by the Centre for Health
rivaroxaban 15 and 20mg film-coated tablets (Xarelto ) SMC No. (756/12) Bayer PLC
 rivaroxaban 15 and 20mg film-coated tablets (Xarelto ) SMC No. (756/12) Bayer PLC 13 January 2012 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product and advises NHS
rivaroxaban 15 and 20mg film-coated tablets (Xarelto ) SMC No. (756/12) Bayer PLC 13 January 2012 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product and advises NHS
Rivaroxaban for the treatment of deep vein thrombosis and prevention of recurrent deep vein thrombosis and pulmonary embolism
 Rivaroxaban for the treatment of deep vein thrombosis and prevention of recurrent deep vein thrombosis and pulmonary Issued: July 2012 guidance.nice.org.uk/ta NHS Evidence has accredited the process used
Rivaroxaban for the treatment of deep vein thrombosis and prevention of recurrent deep vein thrombosis and pulmonary Issued: July 2012 guidance.nice.org.uk/ta NHS Evidence has accredited the process used
Cost Effectiveness of Apixaban (Eliquis ) for the Prevention of Venous Thromboembolic Events in Adult Patients who have Undergone Elective Total Hip
 Cost Effectiveness of Apixaban (Eliquis ) for the Prevention of Venous Thromboembolic Events in Adult Patients who have Undergone Elective Total Hip Replacement or Total Knee Replacement National Centre
Cost Effectiveness of Apixaban (Eliquis ) for the Prevention of Venous Thromboembolic Events in Adult Patients who have Undergone Elective Total Hip Replacement or Total Knee Replacement National Centre
What s the Deal with NSAIDs?
 Focus on CME at the Memorial xxx University of Newfoundland What s the Deal with NSAIDs? Majed M. Khraishi, MB, BCh, FRCPC Presented at Wednesday at Noon Ask the Consultant Arthritis is one of the most
Focus on CME at the Memorial xxx University of Newfoundland What s the Deal with NSAIDs? Majed M. Khraishi, MB, BCh, FRCPC Presented at Wednesday at Noon Ask the Consultant Arthritis is one of the most
teriflunomide, 14mg, film-coated tablets (Aubagio ) SMC No. (940/14) Genzyme Ltd.
 teriflunomide, 14mg, film-coated tablets (Aubagio ) SMC No. (940/14) Genzyme Ltd. 10 January 2014 (Issued 07 February 2014) The Scottish Medicines Consortium (SMC) has completed its assessment of the above
teriflunomide, 14mg, film-coated tablets (Aubagio ) SMC No. (940/14) Genzyme Ltd. 10 January 2014 (Issued 07 February 2014) The Scottish Medicines Consortium (SMC) has completed its assessment of the above
Cost-effectiveness of teriflunomide (Aubagio ) for the treatment of adult patients with relapsing remitting multiple sclerosis
 Cost-effectiveness of teriflunomide (Aubagio ) for the treatment of adult patients with relapsing remitting multiple sclerosis The NCPE has issued a recommendation regarding the cost-effectiveness of teriflunomide
Cost-effectiveness of teriflunomide (Aubagio ) for the treatment of adult patients with relapsing remitting multiple sclerosis The NCPE has issued a recommendation regarding the cost-effectiveness of teriflunomide
Rivaroxaban for the treatment of deep vein thrombosis and prevention of recurrent deep vein thrombosis and pulmonary embolism
 Rivaroxaban for the treatment of deep vein thrombosis and prevention of recurrent deep vein thrombosis and pulmonary Issued: July 2012 guidance.nice.org.uk/ta NICE has accredited the process used by the
Rivaroxaban for the treatment of deep vein thrombosis and prevention of recurrent deep vein thrombosis and pulmonary Issued: July 2012 guidance.nice.org.uk/ta NICE has accredited the process used by the
PROTOCOL SYNOPSIS Evaluation of long-term opioid efficacy for chronic pain
 P a g e 1 PROTOCOL SYNOPSIS Evaluation of long-term opioid efficacy for chronic pain Clinical Phase 4 Study Centers Study Period 25 U.S. sites identified and reviewed by the Steering Committee and Contract
P a g e 1 PROTOCOL SYNOPSIS Evaluation of long-term opioid efficacy for chronic pain Clinical Phase 4 Study Centers Study Period 25 U.S. sites identified and reviewed by the Steering Committee and Contract
1. Comparative effectiveness of alemtuzumab
 Cost-effectiveness of alemtuzumab (Lemtrada ) for the treatment of adult patients with relapsing remitting multiple sclerosis with active disease defined by clinical or imaging features The NCPE has issued
Cost-effectiveness of alemtuzumab (Lemtrada ) for the treatment of adult patients with relapsing remitting multiple sclerosis with active disease defined by clinical or imaging features The NCPE has issued
Cyclooxygenase-2 selective non-steroidal anti-inflammatory drugs (etodolac, meloxicam, celecoxib, rofecoxib, etoricoxib, valdecoxib and
 Health Technology Assessment 2008; Vol. 12: No. 11 Cyclooxygenase-2 selective non-steroidal anti-inflammatory drugs (etodolac, meloxicam, celecoxib, rofecoxib, etoricoxib, valdecoxib and lumiracoxib) for
Health Technology Assessment 2008; Vol. 12: No. 11 Cyclooxygenase-2 selective non-steroidal anti-inflammatory drugs (etodolac, meloxicam, celecoxib, rofecoxib, etoricoxib, valdecoxib and lumiracoxib) for
NIMULID MD. 1. Introduction. 2. Nimulid MD Drug delivery system
 NIMULID MD 1. Introduction Nimulid MD is a flavoured dispersible Nimesulide tablet with fast mouth dissolving characteristics thereby providing immediate relief. Nimesulide is a non-steroidal antiinflammatory
NIMULID MD 1. Introduction Nimulid MD is a flavoured dispersible Nimesulide tablet with fast mouth dissolving characteristics thereby providing immediate relief. Nimesulide is a non-steroidal antiinflammatory
Comparative Effectiveness Review Number 4. Comparative Effectiveness and Safety of Analgesics for Osteoarthritis
 This report is based on research conducted by the Oregon Evidence-based Practice Center (EPC) under contract to the Agency for Healthcare Research and Quality (AHRQ), Rockville, MD (Contract No. 290-02-0024).
This report is based on research conducted by the Oregon Evidence-based Practice Center (EPC) under contract to the Agency for Healthcare Research and Quality (AHRQ), Rockville, MD (Contract No. 290-02-0024).
Peptic Ulcer. Anatomy The stomach is a hollow organ. It is located in the upper abdomen, under the ribs.
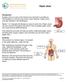 Peptic Ulcer Introduction A peptic ulcer is a sore in the lining of your stomach or duodenum. The duodenum is the first part of your small intestine. Peptic ulcers may also develop in the esophagus. Nearly
Peptic Ulcer Introduction A peptic ulcer is a sore in the lining of your stomach or duodenum. The duodenum is the first part of your small intestine. Peptic ulcers may also develop in the esophagus. Nearly
2. Background This drug had not previously been considered by the PBAC.
 PUBLIC SUMMARY DOCUMENT Product: Ambrisentan, tablets, 5 mg and 10 mg, Volibris Sponsor: GlaxoSmithKline Australia Pty Ltd Date of PBAC Consideration: July 2009 1. Purpose of Application The submission
PUBLIC SUMMARY DOCUMENT Product: Ambrisentan, tablets, 5 mg and 10 mg, Volibris Sponsor: GlaxoSmithKline Australia Pty Ltd Date of PBAC Consideration: July 2009 1. Purpose of Application The submission
What can I eat? Peptic ulcers. What are peptic ulcers? What tests are needed? Will the ulcer come back? What causes a peptic ulcer?
 In association with: INFORMATION ABOUT Peptic ulcers www.corecharity.org.uk What are peptic ulcers? What causes a peptic ulcer? How are NSAIDs and aspirin involved? How do I know if I ve got an ulcer?
In association with: INFORMATION ABOUT Peptic ulcers www.corecharity.org.uk What are peptic ulcers? What causes a peptic ulcer? How are NSAIDs and aspirin involved? How do I know if I ve got an ulcer?
NICE TA 275: Apixaban for the prevention of stroke and systemic embolism in people with non-valvular atrial fibrillation
 Service Notification in response to DHSSPS endorsed NICE Technology Appraisals NICE TA 275: Apixaban for the prevention of stroke and systemic embolism in people with non-valvular atrial fibrillation 1
Service Notification in response to DHSSPS endorsed NICE Technology Appraisals NICE TA 275: Apixaban for the prevention of stroke and systemic embolism in people with non-valvular atrial fibrillation 1
Dimethyl fumarate for treating relapsing remitting multiple sclerosis
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Final appraisal determination Dimethyl fumarate for treating relapsing remitting multiple sclerosis This guidance was developed using the single technology
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Final appraisal determination Dimethyl fumarate for treating relapsing remitting multiple sclerosis This guidance was developed using the single technology
A. Approval for the indications of osteoarthritis and rheumatoid arthritis at a dose of 10mg/day and dysmenorrhea at a dose of 20-mg bid as needed.
 Agent: Valdecoxib Indication: Analgesia, Dysmenorrhea Osteoarthritis, and Rheumatoid Arthritis Reviewer: Kent Johnson, MD Date: November 7, 2001 NDA: 21,341 EXECUTIVE SUMMARY 1-RECOMMENDATIONS A. Approval
Agent: Valdecoxib Indication: Analgesia, Dysmenorrhea Osteoarthritis, and Rheumatoid Arthritis Reviewer: Kent Johnson, MD Date: November 7, 2001 NDA: 21,341 EXECUTIVE SUMMARY 1-RECOMMENDATIONS A. Approval
rivaroxaban 2.5mg film-coated tablets (Xarelto ) SMC No. (1062/15) Bayer plc.
 rivaroxaban 2.5mg film-coated tablets (Xarelto ) SMC No. (1062/15) Bayer plc. 05 June 2015 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product and advises NHS Boards
rivaroxaban 2.5mg film-coated tablets (Xarelto ) SMC No. (1062/15) Bayer plc. 05 June 2015 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product and advises NHS Boards
fingolimod, 0.5mg, hard capsules (Gilenya ) SMC No. (992/14) Novartis Pharmaceuticals UK
 fingolimod, 0.5mg, hard capsules (Gilenya ) SMC No. (992/14) Novartis Pharmaceuticals UK 08 August 2014 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product and advises
fingolimod, 0.5mg, hard capsules (Gilenya ) SMC No. (992/14) Novartis Pharmaceuticals UK 08 August 2014 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product and advises
BEDFORDSHIRE AND LUTON JOINT PRESCRIBING COMMITTEE (JPC)
 BEDFORDSHIRE AND LUTON JOINT PRESCRIBING COMMITTEE (JPC) September 2014 Review date: September 2017 Bulletin 203: Tocilizumab (subcutaneous) in combination with methotrexate or as monotherapy for the treatment
BEDFORDSHIRE AND LUTON JOINT PRESCRIBING COMMITTEE (JPC) September 2014 Review date: September 2017 Bulletin 203: Tocilizumab (subcutaneous) in combination with methotrexate or as monotherapy for the treatment
Dabigatran etexilate for the treatment and secondary prevention of deep vein thrombosis and/or pulmonary embolism ERRATUM
 Dabigatran etexilate for the treatment and secondary prevention of deep vein thrombosis and/or pulmonary embolism ERRATUM This report was commissioned by the NIHR HTA Programme as project number 12/78
Dabigatran etexilate for the treatment and secondary prevention of deep vein thrombosis and/or pulmonary embolism ERRATUM This report was commissioned by the NIHR HTA Programme as project number 12/78
VIOXX Gastrointestinal Outcomes Research Trial (VIGOR) Bonnie Goldmann, M.D. Regulatory Affairs Merck Research Laboratories
 VIOXX Gastrointestinal Outcomes Research Trial (VIGOR) Bonnie Goldmann, M.D. Regulatory Affairs Merck Research Laboratories 1 Arachidonic Acid CO 2 H COX-1 NSAIDs COX-2 Prostanoids Prostanoids Protection
VIOXX Gastrointestinal Outcomes Research Trial (VIGOR) Bonnie Goldmann, M.D. Regulatory Affairs Merck Research Laboratories 1 Arachidonic Acid CO 2 H COX-1 NSAIDs COX-2 Prostanoids Prostanoids Protection
rituximab 1400mg solution for subcutaneous injection (Mabthera ) SMC No. (975/14) Roche Products Limited
 rituximab 1400mg solution for subcutaneous injection (Mabthera ) SMC No. (975/14) Roche Products Limited 06 June 2014 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product
rituximab 1400mg solution for subcutaneous injection (Mabthera ) SMC No. (975/14) Roche Products Limited 06 June 2014 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product
Dimethyl fumarate for treating relapsing-remitting multiple sclerosis
 Dimethyl fumarate for treating relapsing-remitting multiple Issued: August 2014 guidance.nice.org.uk/ta320 NICE has accredited the process used by the Centre for Health Technology Evaluation at NICE to
Dimethyl fumarate for treating relapsing-remitting multiple Issued: August 2014 guidance.nice.org.uk/ta320 NICE has accredited the process used by the Centre for Health Technology Evaluation at NICE to
lenalidomide, 5mg, 10mg, 15mg and 25mg hard capsules (Revlimid ) SMC No. (441/08) Celgene Limited
 Resubmission: lenalidomide, 5mg, 10mg, 15mg and 25mg hard capsules (Revlimid ) SMC No. (441/08) Celgene Limited 07 March 2014 The Scottish Medicines Consortium (SMC) has completed its assessment of the
Resubmission: lenalidomide, 5mg, 10mg, 15mg and 25mg hard capsules (Revlimid ) SMC No. (441/08) Celgene Limited 07 March 2014 The Scottish Medicines Consortium (SMC) has completed its assessment of the
Analgesics for Osteoarthritis: An Update of the
 Comparative Effectiveness Review Number 38 Analgesics for Osteoarthritis: An Update of the 2006 Comparative Effectiveness Review Comparative Effectiveness Review Number 38 Analgesics for Osteoarthritis:
Comparative Effectiveness Review Number 38 Analgesics for Osteoarthritis: An Update of the 2006 Comparative Effectiveness Review Comparative Effectiveness Review Number 38 Analgesics for Osteoarthritis:
peginterferon 63, 94 and 125 microgram solution for injection in pre-filled syringe (Plegridy ) SMC No. (1018/14) Biogen Idec Ltd.
 peginterferon 63, 94 and 125 microgram solution for injection in pre-filled syringe (Plegridy ) SMC No. (1018/14) Biogen Idec Ltd. 05 December 2014 The Scottish Medicines Consortium (SMC) has completed
peginterferon 63, 94 and 125 microgram solution for injection in pre-filled syringe (Plegridy ) SMC No. (1018/14) Biogen Idec Ltd. 05 December 2014 The Scottish Medicines Consortium (SMC) has completed
Low back pain. Quick reference guide. Issue date: May 2009. Early management of persistent non-specific low back pain
 Issue date: May 2009 Low back pain Early management of persistent non-specific low back pain Developed by the National Collaborating Centre for Primary Care About this booklet This is a quick reference
Issue date: May 2009 Low back pain Early management of persistent non-specific low back pain Developed by the National Collaborating Centre for Primary Care About this booklet This is a quick reference
Guidelines for Prevention of NSAID-Related Ulcer Complications
 728 ACG PRACTICE GUIDELINES nature publishing group Guidelines for Prevention of NSAID-Related Ulcer Complications Frank L. L anza, MD, FACG 1,2, Fr anc is K.L. C han, M D, FRCP, FACG 3, E amonn M.M. Q
728 ACG PRACTICE GUIDELINES nature publishing group Guidelines for Prevention of NSAID-Related Ulcer Complications Frank L. L anza, MD, FACG 1,2, Fr anc is K.L. C han, M D, FRCP, FACG 3, E amonn M.M. Q
NHS FORTH VALLEY Guidelines for use of high dose Intravenous Esomeprazole in Adults (Previously called the Hong Kong Protocol)
 NHS FORTH VALLEY Guidelines for use of high dose Intravenous Esomeprazole in Adults (Previously called the Hong Kong Protocol) Date of First Issue 10/05/2010 Approved 16/06/2010 Current Issue Date 18/11/2015
NHS FORTH VALLEY Guidelines for use of high dose Intravenous Esomeprazole in Adults (Previously called the Hong Kong Protocol) Date of First Issue 10/05/2010 Approved 16/06/2010 Current Issue Date 18/11/2015
Nalmefene for reducing alcohol consumption in people with alcohol dependence
 Nalmefene for reducing alcohol consumption in people with alcohol dependence Issued: November 2014 guidance.nice.org.uk/ta325 NICE has accredited the process used by the Centre for Health Technology Evaluation
Nalmefene for reducing alcohol consumption in people with alcohol dependence Issued: November 2014 guidance.nice.org.uk/ta325 NICE has accredited the process used by the Centre for Health Technology Evaluation
Tapered Withdrawal of a Proton Pump Inhibitor to Prevent Rebound Acid-Related Symptoms
 Akin Oyalowo CRC Rotation IRB Proposal August 8, 2012 Tapered Withdrawal of a Proton Pump Inhibitor to Prevent Rebound Acid-Related Symptoms A. Study Purpose and Rationale Proton pump inhibitors (PPIs)
Akin Oyalowo CRC Rotation IRB Proposal August 8, 2012 Tapered Withdrawal of a Proton Pump Inhibitor to Prevent Rebound Acid-Related Symptoms A. Study Purpose and Rationale Proton pump inhibitors (PPIs)
TA 256: Rivaroxaban for the prevention of stroke and systemic embolism in people with atrial fibrillation
 Service Notification in response to DHSSPS endorsed NICE Technology Appraisals TA 256: Rivaroxaban for the prevention of stroke and systemic embolism in people with atrial fibrillation 1 Name of Commissioning
Service Notification in response to DHSSPS endorsed NICE Technology Appraisals TA 256: Rivaroxaban for the prevention of stroke and systemic embolism in people with atrial fibrillation 1 Name of Commissioning
Using big data to identify opportunity and drive informed change in secondary care
 Using big data to identify opportunity and drive informed change in secondary care Exploring the changing patterns of Non-Steroidal Anti-Inflammatory Drug dispensing in UK hospitals ABOUT IMS HEALTH IMS
Using big data to identify opportunity and drive informed change in secondary care Exploring the changing patterns of Non-Steroidal Anti-Inflammatory Drug dispensing in UK hospitals ABOUT IMS HEALTH IMS
Rivaroxaban for treating pulmonary embolism and preventing recurrent venous thromboembolism
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Final appraisal determination Rivaroxaban for treating pulmonary embolism and preventing recurrent venous thromboembolism This guidance was developed using
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Final appraisal determination Rivaroxaban for treating pulmonary embolism and preventing recurrent venous thromboembolism This guidance was developed using
Medication Management Improvement System Protocol #4 Potentially Inappropriate Use of NSAIDs
 Medication Management Improvement System Protocol #4 Potentially Inappropriate Use of NSAIDs Problem: Use of Non-Steroidal Anti-Inflammatory Drugs (NSAIDs) in clients with any one of the following risk
Medication Management Improvement System Protocol #4 Potentially Inappropriate Use of NSAIDs Problem: Use of Non-Steroidal Anti-Inflammatory Drugs (NSAIDs) in clients with any one of the following risk
National Digestive Diseases Information Clearinghouse
 Gastritis National Digestive Diseases Information Clearinghouse U.S. Department of Health and Human Services NATIONAL INSTITUTES OF HEALTH What is gastritis? Gastritis is a condition in which the stomach
Gastritis National Digestive Diseases Information Clearinghouse U.S. Department of Health and Human Services NATIONAL INSTITUTES OF HEALTH What is gastritis? Gastritis is a condition in which the stomach
Public Assessment Report. Pharmacy to General Sales List Reclassification. Nexium Control 20mg Gastro-Resistant Tablets.
 Public Assessment Report Pharmacy to General Sales List Reclassification Nexium Control 20mg Gastro-Resistant Tablets (Esomeprazole) EMA Agency number: Pfizer Consumer Healthcare Ltd TABLE OF CONTENTS
Public Assessment Report Pharmacy to General Sales List Reclassification Nexium Control 20mg Gastro-Resistant Tablets (Esomeprazole) EMA Agency number: Pfizer Consumer Healthcare Ltd TABLE OF CONTENTS
Canadian consensus guidelines on long-term nonsteroidal anti-inflammatory drug therapy and the need for gastroprotection: benefits versus risks
 Alimentary Pharmacology & Therapeutics Canadian consensus guidelines on long-term nonsteroidal anti-inflammatory drug therapy and the need for gastroprotection: benefits versus risks A. ROSTOM*, P. MOAYYEDI
Alimentary Pharmacology & Therapeutics Canadian consensus guidelines on long-term nonsteroidal anti-inflammatory drug therapy and the need for gastroprotection: benefits versus risks A. ROSTOM*, P. MOAYYEDI
Teriflunomide for treating relapsing remitting multiple sclerosis
 Teriflunomide for treating relapsing remitting multiple Issued: January 2014 last modified: June 2014 guidance.nice.org.uk/ta NICE has accredited the process used by the Centre for Health Technology Evaluation
Teriflunomide for treating relapsing remitting multiple Issued: January 2014 last modified: June 2014 guidance.nice.org.uk/ta NICE has accredited the process used by the Centre for Health Technology Evaluation
Trastuzumab for the treatment of HER2-positive metastatic gastric cancer
 Trastuzumab for the treatment of HER2-positive metastatic gastric cancer Issued: November 2010 guidance.nice.org.uk/ta208 NICE has accredited the process used by the Centre for Health Technology Evaluation
Trastuzumab for the treatment of HER2-positive metastatic gastric cancer Issued: November 2010 guidance.nice.org.uk/ta208 NICE has accredited the process used by the Centre for Health Technology Evaluation
What Effects do Provincial Drug Plan Coverage Policies for New Drugs have on Patterns of Use and Cost?
 Enhancing the effectiveness of health care for Ontarians through research What Effects do Provincial Drug Plan Coverage Policies for New Drugs have on Patterns of Use and Cost? November 2003 What effects
Enhancing the effectiveness of health care for Ontarians through research What Effects do Provincial Drug Plan Coverage Policies for New Drugs have on Patterns of Use and Cost? November 2003 What effects
Dabigatran (Pradaxa) for stroke prevention in patients with non-valvular atrial fibrillation (da-big-a-tran)
 Dabigatran (Pradaxa) 1 Dabigatran (Pradaxa) for stroke prevention in patients with non-valvular atrial fibrillation (da-big-a-tran) Similar rate of major bleeding to warfarin KEY POINTS Dabigatran is an
Dabigatran (Pradaxa) 1 Dabigatran (Pradaxa) for stroke prevention in patients with non-valvular atrial fibrillation (da-big-a-tran) Similar rate of major bleeding to warfarin KEY POINTS Dabigatran is an
alemtuzumab, 12mg, concentrate for solution for infusion (Lemtrada ) SMC No. (959/14) Genzyme
 alemtuzumab, 12mg, concentrate for solution for infusion (Lemtrada ) SMC No. (959/14) Genzyme 04 April 2014 (Issued 06 June 2014) The Scottish Medicines Consortium (SMC) has completed its assessment of
alemtuzumab, 12mg, concentrate for solution for infusion (Lemtrada ) SMC No. (959/14) Genzyme 04 April 2014 (Issued 06 June 2014) The Scottish Medicines Consortium (SMC) has completed its assessment of
nalmefene 18mg film-coated tablets (Selincro ) SMC No. (917/13) Lundbeck Limited
 nalmefene 18mg film-coated tablets (Selincro ) SMC No. (917/13) Lundbeck Limited 06 September 2013 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product and advises
nalmefene 18mg film-coated tablets (Selincro ) SMC No. (917/13) Lundbeck Limited 06 September 2013 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product and advises
trastuzumab, 600mg/5mL solution for injection (Herceptin ) SMC No. (928/13) Roche Products Ltd
 trastuzumab, 600mg/5mL solution for injection (Herceptin ) SMC No. (928/13) Roche Products Ltd 06 December 2013 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product
trastuzumab, 600mg/5mL solution for injection (Herceptin ) SMC No. (928/13) Roche Products Ltd 06 December 2013 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product
Suffolk PCT Drug & Therapeutics Committee New Medicine Report
 Suffolk PCT Drug & Therapeutics Committee New Medicine Report This drug has been reviewed because it is a product that may be prescribed in primary care. Medicine Fesoterodine (Toviaz, Pfizer) Document
Suffolk PCT Drug & Therapeutics Committee New Medicine Report This drug has been reviewed because it is a product that may be prescribed in primary care. Medicine Fesoterodine (Toviaz, Pfizer) Document
Economic Evaluation of Natalizumab (Tysabri) for the treatment of relapsing remitting multiple sclerosis that is rapidly evolving and severe or
 Economic Evaluation of Natalizumab (Tysabri) for the treatment of relapsing remitting multiple sclerosis that is rapidly evolving and severe or sub-optimally treated Summary In January 2007 Biogen Idec
Economic Evaluation of Natalizumab (Tysabri) for the treatment of relapsing remitting multiple sclerosis that is rapidly evolving and severe or sub-optimally treated Summary In January 2007 Biogen Idec
Summary and general discussion
 Chapter 7 Summary and general discussion Summary and general discussion In this thesis, treatment of vitamin K antagonist-associated bleed with prothrombin complex concentrate was addressed. In this we
Chapter 7 Summary and general discussion Summary and general discussion In this thesis, treatment of vitamin K antagonist-associated bleed with prothrombin complex concentrate was addressed. In this we
gastrointestinal ulcers and ulcer complications 2 (COX2) enzyme might reduce this burden. 8 To date, two groups of investigators have attempted to
 Comparison of lumiracoxib with naproxen and ibuprofen in the Therapeutic Arthritis Research and Gastrointestinal Event Trial (TARGET), reduction in ulcer complications: randomised controlled trial Thomas
Comparison of lumiracoxib with naproxen and ibuprofen in the Therapeutic Arthritis Research and Gastrointestinal Event Trial (TARGET), reduction in ulcer complications: randomised controlled trial Thomas
CHAPTER 1 INTRODUCTION
 CHAPTER 1 INTRODUCTION Rheumatoid Arthritis (RA) is a chronic syndrome characterised by non-specific, usually symmetric inflammation of the peripheral joints, potentially resulting in progressive destruction
CHAPTER 1 INTRODUCTION Rheumatoid Arthritis (RA) is a chronic syndrome characterised by non-specific, usually symmetric inflammation of the peripheral joints, potentially resulting in progressive destruction
Cancer Treatments Subcommittee of PTAC Meeting held 18 September 2015. (minutes for web publishing)
 Cancer Treatments Subcommittee of PTAC Meeting held 18 September 2015 (minutes for web publishing) Cancer Treatments Subcommittee minutes are published in accordance with the Terms of Reference for the
Cancer Treatments Subcommittee of PTAC Meeting held 18 September 2015 (minutes for web publishing) Cancer Treatments Subcommittee minutes are published in accordance with the Terms of Reference for the
Over the Counter Drugs (OTCs): Considerations for Physical Therapy Practice in Canada
 Background Over the Counter Drugs (OTCs): Considerations for Physical Therapy Practice in Canada The use of medications or drugs by non-physician health professionals is evolving and is linked to collaboration
Background Over the Counter Drugs (OTCs): Considerations for Physical Therapy Practice in Canada The use of medications or drugs by non-physician health professionals is evolving and is linked to collaboration
Technology appraisal guidance Published: 22 July 2015 nice.org.uk/guidance/ta350
 Secukinumab for treating moderate to severe ere plaque psoriasis Technology appraisal guidance Published: 22 July 2015 nice.org.uk/guidance/ta350 NICE 2015. All rights reserved. Contents 1 Guidance...
Secukinumab for treating moderate to severe ere plaque psoriasis Technology appraisal guidance Published: 22 July 2015 nice.org.uk/guidance/ta350 NICE 2015. All rights reserved. Contents 1 Guidance...
Rivaroxaban for acute coronary syndromes
 Northern Treatment Advisory Group Rivaroxaban for acute coronary syndromes Lead author: Nancy Kane Regional Drug & Therapeutics Centre (Newcastle) May 2014 2014 Summary Current long-term management following
Northern Treatment Advisory Group Rivaroxaban for acute coronary syndromes Lead author: Nancy Kane Regional Drug & Therapeutics Centre (Newcastle) May 2014 2014 Summary Current long-term management following
The submission positioned dimethyl fumarate as a first-line treatment option.
 Product: Dimethyl Fumarate, capsules, 120 mg and 240 mg, Tecfidera Sponsor: Biogen Idec Australia Pty Ltd Date of PBAC Consideration: July 2013 1. Purpose of Application The major submission sought an
Product: Dimethyl Fumarate, capsules, 120 mg and 240 mg, Tecfidera Sponsor: Biogen Idec Australia Pty Ltd Date of PBAC Consideration: July 2013 1. Purpose of Application The major submission sought an
understanding GI bleeding
 understanding GI bleeding a consumer education brochure American College of Gastroenterology 4900B South 31st Street, Arlington, VA 22206 703-820-7400 www.acg.gi.org American College of Gastroenterology
understanding GI bleeding a consumer education brochure American College of Gastroenterology 4900B South 31st Street, Arlington, VA 22206 703-820-7400 www.acg.gi.org American College of Gastroenterology
London New Drugs Group APC/DTC Briefing Document Mirabegron (Betmiga TM ) for overactive bladder March 2013
 London New Drugs Group APC/DTC Briefing Document Mirabegron (Betmiga TM ) for overactive bladder March 2013 Summary Mirabegron is first of a new class of drug, beta-3-adrenoceptor agonists, for the treatment
London New Drugs Group APC/DTC Briefing Document Mirabegron (Betmiga TM ) for overactive bladder March 2013 Summary Mirabegron is first of a new class of drug, beta-3-adrenoceptor agonists, for the treatment
nilotinib 150mg hard capsules (Tasigna ) SMC No. (709/11) Novartis Pharmaceuticals UK Ltd
 nilotinib 150mg hard capsules (Tasigna ) SMC No. (709/11) Novartis Pharmaceuticals UK Ltd 08 July 2011 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product and advises
nilotinib 150mg hard capsules (Tasigna ) SMC No. (709/11) Novartis Pharmaceuticals UK Ltd 08 July 2011 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product and advises
Drug Class Review Proton Pump Inhibitors
 Drug Class Review Proton Pump Inhibitors Final Report Update 5 May 2009 Update 4: May 2006 Update 3: May 2005 Update 2: April 2004 Update 1: April 2003 Original Report: November 2002 The literature on
Drug Class Review Proton Pump Inhibitors Final Report Update 5 May 2009 Update 4: May 2006 Update 3: May 2005 Update 2: April 2004 Update 1: April 2003 Original Report: November 2002 The literature on
U.S. Scientific Update Aricept 23 mg Tablets. Dr. Lynn Kramer President NeuroScience Product Creation Unit Eisai Inc.
 U.S. Scientific Update Aricept 23 mg Tablets Dr. Lynn Kramer President NeuroScience Product Creation Unit Eisai Inc. Unmet Need in Moderate to Severe Alzheimer s Disease (AD) Ongoing clinical deterioration
U.S. Scientific Update Aricept 23 mg Tablets Dr. Lynn Kramer President NeuroScience Product Creation Unit Eisai Inc. Unmet Need in Moderate to Severe Alzheimer s Disease (AD) Ongoing clinical deterioration
Alimentary Pharmacology & Therapeutics
 Alimentary Pharmacology & Therapeutics Changing perceptions and practices regarding aspirin, nonsteroidal anti-inflammatory drugs, and cyclooxygenase-2 selective nonsteroidal anti-inflammatory drugs among
Alimentary Pharmacology & Therapeutics Changing perceptions and practices regarding aspirin, nonsteroidal anti-inflammatory drugs, and cyclooxygenase-2 selective nonsteroidal anti-inflammatory drugs among
How To Weigh Data From A Study
 PRINCIPLES ON RESPONSIBLE SHARING OF TRUTHFUL AND NON-MISLEADING INFORMATION ABOUT MEDICINES WITH HEALTH CARE PROFESSIONALS AND PAYERS INTRODUCTION In the era of data-driven medicine, where all parties
PRINCIPLES ON RESPONSIBLE SHARING OF TRUTHFUL AND NON-MISLEADING INFORMATION ABOUT MEDICINES WITH HEALTH CARE PROFESSIONALS AND PAYERS INTRODUCTION In the era of data-driven medicine, where all parties
Choosing Pain Medicine for Osteoarthritis. A Guide for Consumers
 Choosing Pain Medicine for Osteoarthritis A Guide for Consumers Fast Facts on Pain Relievers Acetaminophen (Tylenol ) works on mild pain and has fewer risks than other pain pills. Prescription (Rx) pain
Choosing Pain Medicine for Osteoarthritis A Guide for Consumers Fast Facts on Pain Relievers Acetaminophen (Tylenol ) works on mild pain and has fewer risks than other pain pills. Prescription (Rx) pain
NSAIDs and Peptic Ulcers
 NSAIDs and Peptic Ulcers National Digestive Diseases Information Clearinghouse U.S. Department of Health and Human Services NATIONAL INSTITUTES OF HEALTH What is a peptic ulcer? A peptic ulcer is a sore
NSAIDs and Peptic Ulcers National Digestive Diseases Information Clearinghouse U.S. Department of Health and Human Services NATIONAL INSTITUTES OF HEALTH What is a peptic ulcer? A peptic ulcer is a sore
CDEC RECORD OF ADVICE
 CDEC RECORD OF ADVICE RIVAROXABAN (Xarelto Bayer Inc.) Indication: Deep Vein Thrombosis Without Symptomatic Pulmonary Embolism This document summarizes the Canadian Drug Expert Committee (CDEC) response
CDEC RECORD OF ADVICE RIVAROXABAN (Xarelto Bayer Inc.) Indication: Deep Vein Thrombosis Without Symptomatic Pulmonary Embolism This document summarizes the Canadian Drug Expert Committee (CDEC) response
Pharmacists improving care in care homes
 The Royal Pharmaceutical Society believes that better utilisation of pharmacists skills in care homes will bring significant benefits to care home residents, care homes providers and the NHS. Introduction
The Royal Pharmaceutical Society believes that better utilisation of pharmacists skills in care homes will bring significant benefits to care home residents, care homes providers and the NHS. Introduction
New Evidence reports on presentations given at EULAR 2012. Rituximab for the Treatment of Rheumatoid Arthritis
 New Evidence reports on presentations given at EULAR 2012 Rituximab for the Treatment of Rheumatoid Arthritis Report on EULAR 2012 presentations Long-term safety of rituximab: 10-year follow-up in the
New Evidence reports on presentations given at EULAR 2012 Rituximab for the Treatment of Rheumatoid Arthritis Report on EULAR 2012 presentations Long-term safety of rituximab: 10-year follow-up in the
Product: Tazarotene, cream, 500 micrograms per g (0.05%) and 1.0 mg per g (0.1%), 30 g, Zorac
 PUBLIC SUMMARY DOCUMENT Product: Tazarotene, cream, 500 micrograms per g (0.05%) and 1.0 mg per g (0.1%), 30 g, Zorac Sponsor: Genepharm Australasia Ltd Date of PBAC Consideration: July 2007 1. Purpose
PUBLIC SUMMARY DOCUMENT Product: Tazarotene, cream, 500 micrograms per g (0.05%) and 1.0 mg per g (0.1%), 30 g, Zorac Sponsor: Genepharm Australasia Ltd Date of PBAC Consideration: July 2007 1. Purpose
