Is the 2 nd law of thermodynamics just a consequence of the 1 st law for an ideal gas?
|
|
|
- Shanna Douglas
- 7 years ago
- Views:
Transcription
1 Is the 2 nd law of thermodynamics just a consequence of the 1 st law for an ideal gas? The question arises from the following reasoning, which apparently brings from A (1 st law) to B (2 nd law). Let us postulate the following hypothesis: 1 st law : ΔU = Q W, d U =Q W (1), U being a state function (independent on the past history) (assumed that heat is positive when received by a system, wor is positive when done by the system on the outside) reversible transformations (any state during a transformation is an equilibrium state, all heat and wor exchanges are reversed if the transformation is accomplished bacwards along the same path) only ideal gases involved, i.e. pv =n RT (2) applies and specific heats = 1 n [ Q d T ]V ; c p = 1 n [ Q d T ] p (3) are constant and see what chain of reasoning may be developed. 1. Mayer's relation c p = R (4), proved as follows. Let us consider two states 1 and 2 of an ideal gas, represented by the two points (p 1,) and (p 2,) in Clapeyron's plane. Being U a state variable, its variation when passing from state 1 to state 2 will be independent on the transformation path; therefore, with reference to the figure next, U 132 =U 142 =U 12, paths and consisting both only of isobaric and isochoric transformations. Based on 1 st law : U 132 =32 W 132 =nc p T 3 T 1 nc V T 2 T 3 p 1 V (5') U 142 =42 W 142 =n C v T 4 T 1 nc p T 2 T 4 p 2 V (5 ). For an ideal gas nt = pv/r, by which we can eliminate the temperatures in the (5) and rewrite them completely in terms of pressures and volumes, i.e. U 132 = c p R 1 p 1 V R p (6') ; U 142 = c p R 1 p 2 V R p (6 ) wherein V = ; p= p 2 p 1. Now, by equating U 132 =U 142 we finally come to the relation: R pv = c p R 1 pv (7), which must be valid for any transformation starting form point 1 and ending in point 2, and therefore for transformations for which we may consider p 0 ; V 0 ; therefore it must be R = c p R 1, from which Mayer's relation follows: =c p R.
2 2. Internal energy and temperature In an ideal gas the internal energy U depends only on the temperature. Proof is that by combining Mayer's relation and the equation of ideal gases with e.g. the (6') we obtain: U 12 = U 132 = c p (8) R 1 p 1V R p= R p 1V p= R n R T U 12=n T 3. Wor in an isothermic process Such wor may be found by integrating the ideal gas equation p= n R T p dv =n R T ln (9) V W = and is equal to the heat exchange Q, since along an isotherm it must be ΔU=0 (point 2. above, relation (8). 3. Adiabatic slope In a reversible cycle undergone by an ideal gas, based on 1 st law: Q=dW U = pdv n dt = pdv d pv R ; hence Q= 1 R c p p dv V (10) A system will receive heat in a process if δq > 0 ; the process will be adiabatic if δq = 0. from the (10): Q 0 c p p dv V 0 dv c p p V = p V (11) (with = c p ) The adiabatic slope will be the limit condition of the (10), i.e. dv = p V V p dv =0 d p V =0 pv =constant (12) (equation of adiabats). The heat exchanged during a process depends on the slope of the corresponding pv curve: during an expansion Q 0 dv [ (11'), with dv [ ]ad dv ]ad= p V, i.e. he heat exchange is positive if the slope of the expansion curve is (a number) greater than the slope of the adiabat in the same point; during a compression, the opposite is true, i.e. Q 0 dv [ (11 ). dv ]ad which is summarised in the figure next.
3 4. Carnot's theorem The efficiency of this cycle is : =1 Q n R T L ln V 3 2 V =1 4 n R ln (12) Along isotherms it will be also : p 1 = p 2 ; p 3 V 3 = p 4 p 4 = V 3 ; = p 3 p 2 and considering the equations of adiabatics: p 1 = p 4 ; p 2 = p 3 V 3 p 1 p 2 Let us consider a power reversible cycle (A 1 B 2 A), as in the figure next, where exchanges of heat are indicated, together with the two extreme tangent adiabats. The shaded portion between the points A and B, delimited by the tangent adiabats, is the portion of the cycle where Q>0, the remaining portion is where Q<0; the upper portion between the points 1 and 2, delimited by tangent isochorics is the expansion phase (W>0), the lower portion between points 1 and 2 is the compression phase (W<0). In Carnot's cycle heat is exchanged only at constant temperature and with only two heat sources, i.e. the cycle comprises two isotherms and two adiabats, as in the figure below. p 1 = p 4 p 3 V 3 then, from the (12') and the (12 ) we come to the we finally find Carnot's efficiency: (12'), (12 ) = V 3 (12'''), and from the (12) = Car (14) It can now be proved that if we consider a power reversible cycle represented by a closed curve in pv plane, for which a single heat input phase and a single heat output phase may be identified between two points A and B, its efficiency is lower or equal to Carnot efficiency between the highest and the lowest reached temperatures. Given such a cycle, let us consider the circumscribed Carnot's cycle, I.e. with isotherms and adiabats tangent to it (see figure next). Four
4 additional power cycles may be defined with processes from Carnot's cycle and from the given cycle, i.e.: Cycle 1A (1 mid of 1-2 A 1); Cycle 1B (2 B mid of 1-2 2); Cycle 2A (A mid of A); Cycle 2B (B 3 mid of 3-4 B), producing each a wor corresponding to the respective enclosed area W 1A, W 1B, W 2A, W 2B.. In cycles 1A and 1B the system receives respective heats Q' 1A and Q' 1B, whose sum is the heat absorbed by a system in Carnot's cycle along isotherm 1-2, and outputs respective heats A and B, whose sum is the heat received by the system in the heat input phase of the given cycle (due to reversibility of all processes, the process B A delivers exactly the same heat that is received in the process A B), i.e. Q ' 1 =Q ' 1A Q ' 1B ; =A B. Similarly, for cycles 2A and 2B it can be observed that in such cycles altogether the system receives the heat output during the given cycle in the heat output phase, and that they output altogether the heat output by the system in Carnot's isotherm 3-4 : Q ' 2 =Q ' 2A Q ' 2B ; Q 2 =Q 2A Q 2B. Since in a reversible cycle it is ΔU = 0, for the 1 st law of thermodynamics we will have also: Q ' 1A =A W 1A ; Q ' 1B =B W 1B ; Q 2A =Q ' 2A W 2A ; Q 2B =Q ' 2B W 2B and, in particular Q ' 1 ; Q 2 Q ' 2 (6) From the (6) it follows that Q 2 Q ' 2 Q ' 1 1 Q 2 1 Q ' 2 Q ' 1, I.e. Car (15) (the equal relation being valid only if W 1A=W 1B=W 2A=W 2B=0, I.e. if the cycles coincide). If we now consider a general reversible power cycle, by drawing tangent adiabats and identifying their contact points (points A, B, C, D, E in the figure next) with the cycle, we can split the power cycle into partial cycles having each a single heat input phase and a single output phase defined by two points (e.g. cycle C D B A C in the next figure, having heat input from C to D, and cycle E A B E having heat input from E to A), to which the previous result applies, i.e. for the -th of these cycles it will be = W (16), Q i the subdivision borders between the partial cycles being adiabats, without heat exchange. Furthermore the total wor accomplished by the cycle will be the total enclosed area, i.e. W = Q i = W Q i, and the total input heat will be. If we consider the highest and lower temperatures and T L reached in the cycle, it will be: T L T L ;, ; hence 1 T L,. W Q i Q Finally we have i 1 T L = W Q i = Q i = Q i Q i 1 T L, i.e. the total efficiency of any generic cycle is not greater than Carnot efficiency between the highest and the lowest reached temperatures. (Carnot's theorem)
5 5. The 2 nd law Having proved Carnot's theorem, the 2 nd law becomes just a direct consequence. E.g. let us consider a Carnot power engine between sources and T L, as in the figure next. If heat transfer was possible from source T L to source without spending any wor, we could combine a machine that performs such heat transfer with a Carnot power engine, to bring bac a part Q'2 of Carnot's output heat to the hottest source. For such a resulting machine the actual input heat would be Q i = Q ' 1, and therefore = W Q i W, contrary to Carnot's theorem. Therefore it must be impossible to transfer heat from a lower temperature source to a higher temperature one without spending wor. In the end, for reversible processes involving an ideal gas, apparently we have come to the 2 nd law in Clausius' formulation, starting only from the 1 st law (it would be easy to come similarly from Carnot's theorem to other formulations of the 2 nd law).
Answer, Key Homework 6 David McIntyre 1
 Answer, Key Homework 6 David McIntyre 1 This print-out should have 0 questions, check that it is complete. Multiple-choice questions may continue on the next column or page: find all choices before making
Answer, Key Homework 6 David McIntyre 1 This print-out should have 0 questions, check that it is complete. Multiple-choice questions may continue on the next column or page: find all choices before making
The First Law of Thermodynamics
 Thermodynamics The First Law of Thermodynamics Thermodynamic Processes (isobaric, isochoric, isothermal, adiabatic) Reversible and Irreversible Processes Heat Engines Refrigerators and Heat Pumps The Carnot
Thermodynamics The First Law of Thermodynamics Thermodynamic Processes (isobaric, isochoric, isothermal, adiabatic) Reversible and Irreversible Processes Heat Engines Refrigerators and Heat Pumps The Carnot
We will try to get familiar with a heat pump, and try to determine its performance coefficient under different circumstances.
 C4. Heat Pump I. OBJECTIVE OF THE EXPERIMENT We will try to get familiar with a heat pump, and try to determine its performance coefficient under different circumstances. II. INTRODUCTION II.1. Thermodynamic
C4. Heat Pump I. OBJECTIVE OF THE EXPERIMENT We will try to get familiar with a heat pump, and try to determine its performance coefficient under different circumstances. II. INTRODUCTION II.1. Thermodynamic
The Second Law of Thermodynamics
 The Second aw of Thermodynamics The second law of thermodynamics asserts that processes occur in a certain direction and that the energy has quality as well as quantity. The first law places no restriction
The Second aw of Thermodynamics The second law of thermodynamics asserts that processes occur in a certain direction and that the energy has quality as well as quantity. The first law places no restriction
FUNDAMENTALS OF ENGINEERING THERMODYNAMICS
 FUNDAMENTALS OF ENGINEERING THERMODYNAMICS System: Quantity of matter (constant mass) or region in space (constant volume) chosen for study. Closed system: Can exchange energy but not mass; mass is constant
FUNDAMENTALS OF ENGINEERING THERMODYNAMICS System: Quantity of matter (constant mass) or region in space (constant volume) chosen for study. Closed system: Can exchange energy but not mass; mass is constant
Heat and Work. First Law of Thermodynamics 9.1. Heat is a form of energy. Calorimetry. Work. First Law of Thermodynamics.
 Heat and First Law of Thermodynamics 9. Heat Heat and Thermodynamic rocesses Thermodynamics is the science of heat and work Heat is a form of energy Calorimetry Mechanical equivalent of heat Mechanical
Heat and First Law of Thermodynamics 9. Heat Heat and Thermodynamic rocesses Thermodynamics is the science of heat and work Heat is a form of energy Calorimetry Mechanical equivalent of heat Mechanical
Supplementary Notes on Entropy and the Second Law of Thermodynamics
 ME 4- hermodynamics I Supplementary Notes on Entropy and the Second aw of hermodynamics Reversible Process A reversible process is one which, having taken place, can be reversed without leaving a change
ME 4- hermodynamics I Supplementary Notes on Entropy and the Second aw of hermodynamics Reversible Process A reversible process is one which, having taken place, can be reversed without leaving a change
THE KINETIC THEORY OF GASES
 Chapter 19: THE KINETIC THEORY OF GASES 1. Evidence that a gas consists mostly of empty space is the fact that: A. the density of a gas becomes much greater when it is liquefied B. gases exert pressure
Chapter 19: THE KINETIC THEORY OF GASES 1. Evidence that a gas consists mostly of empty space is the fact that: A. the density of a gas becomes much greater when it is liquefied B. gases exert pressure
Exergy: the quality of energy N. Woudstra
 Exergy: the quality of energy N. Woudstra Introduction Characteristic for our society is a massive consumption of goods and energy. Continuation of this way of life in the long term is only possible if
Exergy: the quality of energy N. Woudstra Introduction Characteristic for our society is a massive consumption of goods and energy. Continuation of this way of life in the long term is only possible if
Problem Set 4 Solutions
 Chemistry 360 Dr Jean M Standard Problem Set 4 Solutions 1 Two moles of an ideal gas are compressed isothermally and reversibly at 98 K from 1 atm to 00 atm Calculate q, w, ΔU, and ΔH For an isothermal
Chemistry 360 Dr Jean M Standard Problem Set 4 Solutions 1 Two moles of an ideal gas are compressed isothermally and reversibly at 98 K from 1 atm to 00 atm Calculate q, w, ΔU, and ΔH For an isothermal
Chapter 18 Temperature, Heat, and the First Law of Thermodynamics. Problems: 8, 11, 13, 17, 21, 27, 29, 37, 39, 41, 47, 51, 57
 Chapter 18 Temperature, Heat, and the First Law of Thermodynamics Problems: 8, 11, 13, 17, 21, 27, 29, 37, 39, 41, 47, 51, 57 Thermodynamics study and application of thermal energy temperature quantity
Chapter 18 Temperature, Heat, and the First Law of Thermodynamics Problems: 8, 11, 13, 17, 21, 27, 29, 37, 39, 41, 47, 51, 57 Thermodynamics study and application of thermal energy temperature quantity
a) Use the following equation from the lecture notes: = ( 8.314 J K 1 mol 1) ( ) 10 L
 hermodynamics: Examples for chapter 4. 1. One mole of nitrogen gas is allowed to expand from 0.5 to 10 L reversible and isothermal process at 300 K. Calculate the change in molar entropy using a the ideal
hermodynamics: Examples for chapter 4. 1. One mole of nitrogen gas is allowed to expand from 0.5 to 10 L reversible and isothermal process at 300 K. Calculate the change in molar entropy using a the ideal
The final numerical answer given is correct but the math shown does not give that answer.
 Note added to Homework set 7: The solution to Problem 16 has an error in it. The specific heat of water is listed as c 1 J/g K but should be c 4.186 J/g K The final numerical answer given is correct but
Note added to Homework set 7: The solution to Problem 16 has an error in it. The specific heat of water is listed as c 1 J/g K but should be c 4.186 J/g K The final numerical answer given is correct but
Problem Set 3 Solutions
 Chemistry 360 Dr Jean M Standard Problem Set 3 Solutions 1 (a) One mole of an ideal gas at 98 K is expanded reversibly and isothermally from 10 L to 10 L Determine the amount of work in Joules We start
Chemistry 360 Dr Jean M Standard Problem Set 3 Solutions 1 (a) One mole of an ideal gas at 98 K is expanded reversibly and isothermally from 10 L to 10 L Determine the amount of work in Joules We start
Chapter 2 Classical Thermodynamics: The Second Law
 Chapter 2 Classical hermodynamics: he Second Law 2.1 Heat engines and refrigerators 2.2 he second law of thermodynamics 2.3 Carnot cycles and Carnot engines 2.4* he thermodynamic temperature scale 2.5
Chapter 2 Classical hermodynamics: he Second Law 2.1 Heat engines and refrigerators 2.2 he second law of thermodynamics 2.3 Carnot cycles and Carnot engines 2.4* he thermodynamic temperature scale 2.5
Chapter 8 Maxwell relations and measurable properties
 Chapter 8 Maxwell relations and measurable properties 8.1 Maxwell relations Other thermodynamic potentials emerging from Legendre transforms allow us to switch independent variables and give rise to alternate
Chapter 8 Maxwell relations and measurable properties 8.1 Maxwell relations Other thermodynamic potentials emerging from Legendre transforms allow us to switch independent variables and give rise to alternate
Define the notations you are using properly. Present your arguments in details. Good luck!
 Umeå Universitet, Fysik Vitaly Bychkov Prov i fysik, Thermodynamics, 0-0-4, kl 9.00-5.00 jälpmedel: Students may use any book(s) including the textbook Thermal physics. Minor notes in the books are also
Umeå Universitet, Fysik Vitaly Bychkov Prov i fysik, Thermodynamics, 0-0-4, kl 9.00-5.00 jälpmedel: Students may use any book(s) including the textbook Thermal physics. Minor notes in the books are also
Physics 5D - Nov 18, 2013
 Physics 5D - Nov 18, 2013 30 Midterm Scores B } Number of Scores 25 20 15 10 5 F D C } A- A A + 0 0-59.9 60-64.9 65-69.9 70-74.9 75-79.9 80-84.9 Percent Range (%) The two problems with the fewest correct
Physics 5D - Nov 18, 2013 30 Midterm Scores B } Number of Scores 25 20 15 10 5 F D C } A- A A + 0 0-59.9 60-64.9 65-69.9 70-74.9 75-79.9 80-84.9 Percent Range (%) The two problems with the fewest correct
Thermodynamics - Example Problems Problems and Solutions
 Thermodynamics - Example Problems Problems and Solutions 1 Examining a Power Plant Consider a power plant. At point 1 the working gas has a temperature of T = 25 C. The pressure is 1bar and the mass flow
Thermodynamics - Example Problems Problems and Solutions 1 Examining a Power Plant Consider a power plant. At point 1 the working gas has a temperature of T = 25 C. The pressure is 1bar and the mass flow
Partial Derivatives. @x f (x; y) = @ x f (x; y) @x x2 y + @ @x y2 and then we evaluate the derivative as if y is a constant.
 Partial Derivatives Partial Derivatives Just as derivatives can be used to eplore the properties of functions of 1 variable, so also derivatives can be used to eplore functions of 2 variables. In this
Partial Derivatives Partial Derivatives Just as derivatives can be used to eplore the properties of functions of 1 variable, so also derivatives can be used to eplore functions of 2 variables. In this
Phys222 W11 Quiz 1: Chapters 19-21 Keys. Name:
 Name:. In order for two objects to have the same temperature, they must a. be in thermal equilibrium.
Name:. In order for two objects to have the same temperature, they must a. be in thermal equilibrium.
HEAT UNIT 1.1 KINETIC THEORY OF GASES. 1.1.1 Introduction. 1.1.2 Postulates of Kinetic Theory of Gases
 UNIT HEAT. KINETIC THEORY OF GASES.. Introduction Molecules have a diameter of the order of Å and the distance between them in a gas is 0 Å while the interaction distance in solids is very small. R. Clausius
UNIT HEAT. KINETIC THEORY OF GASES.. Introduction Molecules have a diameter of the order of Å and the distance between them in a gas is 0 Å while the interaction distance in solids is very small. R. Clausius
Problem Set 1 3.20 MIT Professor Gerbrand Ceder Fall 2003
 LEVEL 1 PROBLEMS Problem Set 1 3.0 MIT Professor Gerbrand Ceder Fall 003 Problem 1.1 The internal energy per kg for a certain gas is given by U = 0. 17 T + C where U is in kj/kg, T is in Kelvin, and C
LEVEL 1 PROBLEMS Problem Set 1 3.0 MIT Professor Gerbrand Ceder Fall 003 Problem 1.1 The internal energy per kg for a certain gas is given by U = 0. 17 T + C where U is in kj/kg, T is in Kelvin, and C
The Second Law of Thermodynamics
 Objectives MAE 320 - Chapter 6 The Second Law of Thermodynamics The content and the pictures are from the text book: Çengel, Y. A. and Boles, M. A., Thermodynamics: An Engineering Approach, McGraw-Hill,
Objectives MAE 320 - Chapter 6 The Second Law of Thermodynamics The content and the pictures are from the text book: Çengel, Y. A. and Boles, M. A., Thermodynamics: An Engineering Approach, McGraw-Hill,
Stirling heat engine Internal combustion engine (Otto cycle) Diesel engine Steam engine (Rankine cycle) Kitchen Refrigerator
 Lecture. Real eat Engines and refrigerators (Ch. ) Stirling heat engine Internal combustion engine (Otto cycle) Diesel engine Steam engine (Rankine cycle) Kitchen Refrigerator Carnot Cycle - is not very
Lecture. Real eat Engines and refrigerators (Ch. ) Stirling heat engine Internal combustion engine (Otto cycle) Diesel engine Steam engine (Rankine cycle) Kitchen Refrigerator Carnot Cycle - is not very
Isentropic flow. Wikepedia
 Isentropic flow Wikepedia In thermodynamics, an isentropic process or isoentropic process (ισον = "equal" (Greek); εντροπία entropy = "disorder"(greek)) is one in which for purposes of engineering analysis
Isentropic flow Wikepedia In thermodynamics, an isentropic process or isoentropic process (ισον = "equal" (Greek); εντροπία entropy = "disorder"(greek)) is one in which for purposes of engineering analysis
Chapter 1 Classical Thermodynamics: The First Law. 1.2 The first law of thermodynamics. 1.3 Real and ideal gases: a review
 Chapter 1 Classical Thermodynamics: The First Law 1.1 Introduction 1.2 The first law of thermodynamics 1.3 Real and ideal gases: a review 1.4 First law for cycles 1.5 Reversible processes 1.6 Work 1.7
Chapter 1 Classical Thermodynamics: The First Law 1.1 Introduction 1.2 The first law of thermodynamics 1.3 Real and ideal gases: a review 1.4 First law for cycles 1.5 Reversible processes 1.6 Work 1.7
) and mass of each particle is m. We make an extremely small
 Umeå Universitet, Fysik Vitaly Bychkov Prov i fysik, Thermodynamics, --6, kl 9.-5. Hjälpmedel: Students may use any book including the textbook Thermal physics. Present your solutions in details: it will
Umeå Universitet, Fysik Vitaly Bychkov Prov i fysik, Thermodynamics, --6, kl 9.-5. Hjälpmedel: Students may use any book including the textbook Thermal physics. Present your solutions in details: it will
Thermodynamics AP Physics B. Multiple Choice Questions
 Thermodynamics AP Physics B Name Multiple Choice Questions 1. What is the name of the following statement: When two systems are in thermal equilibrium with a third system, then they are in thermal equilibrium
Thermodynamics AP Physics B Name Multiple Choice Questions 1. What is the name of the following statement: When two systems are in thermal equilibrium with a third system, then they are in thermal equilibrium
KINETIC THEORY OF GASES AND THERMODYNAMICS SECTION I Kinetic theory of gases
 KINETIC THEORY OF GASES AND THERMODYNAMICS SECTION I Kinetic theory of gases Some important terms in kinetic theory of gases Macroscopic quantities: Physical quantities like pressure, temperature, volume,
KINETIC THEORY OF GASES AND THERMODYNAMICS SECTION I Kinetic theory of gases Some important terms in kinetic theory of gases Macroscopic quantities: Physical quantities like pressure, temperature, volume,
Gibbs Free Energy and Chemical Potential. NC State University
 Chemistry 433 Lecture 14 Gibbs Free Energy and Chemical Potential NC State University The internal energy expressed in terms of its natural variables We can use the combination of the first and second
Chemistry 433 Lecture 14 Gibbs Free Energy and Chemical Potential NC State University The internal energy expressed in terms of its natural variables We can use the combination of the first and second
ME 201 Thermodynamics
 ME 0 Thermodynamics Second Law Practice Problems. Ideally, which fluid can do more work: air at 600 psia and 600 F or steam at 600 psia and 600 F The maximum work a substance can do is given by its availablity.
ME 0 Thermodynamics Second Law Practice Problems. Ideally, which fluid can do more work: air at 600 psia and 600 F or steam at 600 psia and 600 F The maximum work a substance can do is given by its availablity.
vap H = RT 1T 2 = 30.850 kj mol 1 100 kpa = 341 K
 Thermodynamics: Examples for chapter 6. 1. The boiling point of hexane at 1 atm is 68.7 C. What is the boiling point at 1 bar? The vapor pressure of hexane at 49.6 C is 53.32 kpa. Assume that the vapor
Thermodynamics: Examples for chapter 6. 1. The boiling point of hexane at 1 atm is 68.7 C. What is the boiling point at 1 bar? The vapor pressure of hexane at 49.6 C is 53.32 kpa. Assume that the vapor
Technical Thermodynamics
 Technical Thermodynamics Chapter 2: Basic ideas and some definitions Prof. Dr.-Ing. habil. Egon Hassel University of Rostock, Germany Faculty of Mechanical Engineering and Ship Building Institute of Technical
Technical Thermodynamics Chapter 2: Basic ideas and some definitions Prof. Dr.-Ing. habil. Egon Hassel University of Rostock, Germany Faculty of Mechanical Engineering and Ship Building Institute of Technical
Electrochemical Kinetics ( Ref. :Bard and Faulkner, Oldham and Myland, Liebhafsky and Cairns) R f = k f * C A (2) R b = k b * C B (3)
 Electrochemical Kinetics ( Ref. :Bard and Faulkner, Oldham and Myland, Liebhafsky and Cairns) 1. Background Consider the reaction given below: A B (1) If k f and k b are the rate constants of the forward
Electrochemical Kinetics ( Ref. :Bard and Faulkner, Oldham and Myland, Liebhafsky and Cairns) 1. Background Consider the reaction given below: A B (1) If k f and k b are the rate constants of the forward
Compressible Fluids. Faith A. Morrison Associate Professor of Chemical Engineering Michigan Technological University November 4, 2004
 94 c 2004 Faith A. Morrison, all rights reserved. Compressible Fluids Faith A. Morrison Associate Professor of Chemical Engineering Michigan Technological University November 4, 2004 Chemical engineering
94 c 2004 Faith A. Morrison, all rights reserved. Compressible Fluids Faith A. Morrison Associate Professor of Chemical Engineering Michigan Technological University November 4, 2004 Chemical engineering
Thermodynamics of Mixing
 Thermodynamics of Mixing Dependence of Gibbs energy on mixture composition is G = n A µ A + n B µ B and at constant T and p, systems tend towards a lower Gibbs energy The simplest example of mixing: What
Thermodynamics of Mixing Dependence of Gibbs energy on mixture composition is G = n A µ A + n B µ B and at constant T and p, systems tend towards a lower Gibbs energy The simplest example of mixing: What
Statistical Mechanics, Kinetic Theory Ideal Gas. 8.01t Nov 22, 2004
 Statistical Mechanics, Kinetic Theory Ideal Gas 8.01t Nov 22, 2004 Statistical Mechanics and Thermodynamics Thermodynamics Old & Fundamental Understanding of Heat (I.e. Steam) Engines Part of Physics Einstein
Statistical Mechanics, Kinetic Theory Ideal Gas 8.01t Nov 22, 2004 Statistical Mechanics and Thermodynamics Thermodynamics Old & Fundamental Understanding of Heat (I.e. Steam) Engines Part of Physics Einstein
Chapter 10 Temperature and Heat
 Chapter 10 Temperature and Heat What are temperature and heat? Are they the same? What causes heat? What Is Temperature? How do we measure temperature? What are we actually measuring? Temperature and Its
Chapter 10 Temperature and Heat What are temperature and heat? Are they the same? What causes heat? What Is Temperature? How do we measure temperature? What are we actually measuring? Temperature and Its
39th International Physics Olympiad - Hanoi - Vietnam - 2008. Theoretical Problem No. 3
 CHANGE OF AIR TEMPERATURE WITH ALTITUDE, ATMOSPHERIC STABILITY AND AIR POLLUTION Vertical motion of air governs many atmospheric processes, such as the formation of clouds and precipitation and the dispersal
CHANGE OF AIR TEMPERATURE WITH ALTITUDE, ATMOSPHERIC STABILITY AND AIR POLLUTION Vertical motion of air governs many atmospheric processes, such as the formation of clouds and precipitation and the dispersal
CHAPTER 7 THE SECOND LAW OF THERMODYNAMICS. Blank
 CHAPTER 7 THE SECOND LAW OF THERMODYNAMICS Blank SONNTAG/BORGNAKKE STUDY PROBLEM 7-1 7.1 A car engine and its fuel consumption A car engine produces 136 hp on the output shaft with a thermal efficiency
CHAPTER 7 THE SECOND LAW OF THERMODYNAMICS Blank SONNTAG/BORGNAKKE STUDY PROBLEM 7-1 7.1 A car engine and its fuel consumption A car engine produces 136 hp on the output shaft with a thermal efficiency
15 THERMODYNAMICS. Learning Objectives
 CHAPTER 15 THERMODYNAMICS 505 15 THERMODYNAMICS Figure 15.1 A steam engine uses heat transfer to do work. Tourists regularly ride this narrow-gauge steam engine train near the San Juan Skyway in Durango,
CHAPTER 15 THERMODYNAMICS 505 15 THERMODYNAMICS Figure 15.1 A steam engine uses heat transfer to do work. Tourists regularly ride this narrow-gauge steam engine train near the San Juan Skyway in Durango,
State Newton's second law of motion for a particle, defining carefully each term used.
 5 Question 1. [Marks 20] An unmarked police car P is, travelling at the legal speed limit, v P, on a straight section of highway. At time t = 0, the police car is overtaken by a car C, which is speeding
5 Question 1. [Marks 20] An unmarked police car P is, travelling at the legal speed limit, v P, on a straight section of highway. At time t = 0, the police car is overtaken by a car C, which is speeding
OPTIMAL DESIGN AND OPERATION OF HELIUM REFRIGERATION SYSTEMS *
 OPTIMAL DESIGN AND OPERATION OF HELIUM REFRIGERATION SYSTEMS * Abstract Helium refrigerators are of keen interest to present and future particle physics programs utilizing superconducting magnet or radio
OPTIMAL DESIGN AND OPERATION OF HELIUM REFRIGERATION SYSTEMS * Abstract Helium refrigerators are of keen interest to present and future particle physics programs utilizing superconducting magnet or radio
Thermodynamics for Materials and Metallurgical Engineers
 Thermodynamics for Materials and Metallurgical Engineers Stanley M. Howard, PhD South Dakota School of Mines and Technology Department of Materials and Metallurgical Engineering Rapid City, SD 57701 USA
Thermodynamics for Materials and Metallurgical Engineers Stanley M. Howard, PhD South Dakota School of Mines and Technology Department of Materials and Metallurgical Engineering Rapid City, SD 57701 USA
APPLIED THERMODYNAMICS TUTORIAL 1 REVISION OF ISENTROPIC EFFICIENCY ADVANCED STEAM CYCLES
 APPLIED THERMODYNAMICS TUTORIAL 1 REVISION OF ISENTROPIC EFFICIENCY ADVANCED STEAM CYCLES INTRODUCTION This tutorial is designed for students wishing to extend their knowledge of thermodynamics to a more
APPLIED THERMODYNAMICS TUTORIAL 1 REVISION OF ISENTROPIC EFFICIENCY ADVANCED STEAM CYCLES INTRODUCTION This tutorial is designed for students wishing to extend their knowledge of thermodynamics to a more
OPTIMIZATION OF DIAMETER RATIO FOR ALPHA-TYPE STIRLING ENGINES
 OPTIMIZATION OF DIAMETER RATIO FOR ALPHA-TYPE STIRLING ENGINES VLAD MARIO HOMUTESCU* DAN-TEODOR BĂLĂNESCU* * Gheorghe Asachi Technical University of Iassy Department of of ermotechnics ermal Engines and
OPTIMIZATION OF DIAMETER RATIO FOR ALPHA-TYPE STIRLING ENGINES VLAD MARIO HOMUTESCU* DAN-TEODOR BĂLĂNESCU* * Gheorghe Asachi Technical University of Iassy Department of of ermotechnics ermal Engines and
Thermodynamics. Chapter 13 Phase Diagrams. NC State University
 Thermodynamics Chapter 13 Phase Diagrams NC State University Pressure (atm) Definition of a phase diagram A phase diagram is a representation of the states of matter, solid, liquid, or gas as a function
Thermodynamics Chapter 13 Phase Diagrams NC State University Pressure (atm) Definition of a phase diagram A phase diagram is a representation of the states of matter, solid, liquid, or gas as a function
14.01 Principles of Microeconomics, Fall 2007 Chia-Hui Chen October 15, 2007. Lecture 13. Cost Function
 Short-Run Cost Function. Principles of Microeconomics, Fall Chia-Hui Chen October, ecture Cost Functions Outline. Chap : Short-Run Cost Function. Chap : ong-run Cost Function Cost Function et w be the
Short-Run Cost Function. Principles of Microeconomics, Fall Chia-Hui Chen October, ecture Cost Functions Outline. Chap : Short-Run Cost Function. Chap : ong-run Cost Function Cost Function et w be the
Introduction to the Ideal Gas Law
 Course PHYSICS260 Assignment 5 Consider ten grams of nitrogen gas at an initial pressure of 6.0 atm and at room temperature. It undergoes an isobaric expansion resulting in a quadrupling of its volume.
Course PHYSICS260 Assignment 5 Consider ten grams of nitrogen gas at an initial pressure of 6.0 atm and at room temperature. It undergoes an isobaric expansion resulting in a quadrupling of its volume.
Condensers & Evaporator Chapter 5
 Condensers & Evaporator Chapter 5 This raises the condenser temperature and the corresponding pressure thereby reducing the COP. Page 134 of 263 Condensers & Evaporator Chapter 5 OBJECTIVE QUESTIONS (GATE,
Condensers & Evaporator Chapter 5 This raises the condenser temperature and the corresponding pressure thereby reducing the COP. Page 134 of 263 Condensers & Evaporator Chapter 5 OBJECTIVE QUESTIONS (GATE,
Physics 176 Topics to Review For the Final Exam
 Physics 176 Topics to Review For the Final Exam Professor Henry Greenside May, 011 Thermodynamic Concepts and Facts 1. Practical criteria for identifying when a macroscopic system is in thermodynamic equilibrium:
Physics 176 Topics to Review For the Final Exam Professor Henry Greenside May, 011 Thermodynamic Concepts and Facts 1. Practical criteria for identifying when a macroscopic system is in thermodynamic equilibrium:
Chapter 6 The first law and reversibility
 Chapter 6 The first law and reversibility 6.1 The first law for processes in closed systems We have discussed the properties of equilibrium states and the relationship between the thermodynamic parameters
Chapter 6 The first law and reversibility 6.1 The first law for processes in closed systems We have discussed the properties of equilibrium states and the relationship between the thermodynamic parameters
Thermodynamics: Lecture 2
 Thermodynamics: Lecture 2 Chris Glosser February 11, 2001 1 OUTLINE I. Heat and Work. (A) Work, Heat and Energy: U = Q + W. (B) Methods of Heat Transport. (C) Infintesimal Work: Exact vs Inexact Differentials
Thermodynamics: Lecture 2 Chris Glosser February 11, 2001 1 OUTLINE I. Heat and Work. (A) Work, Heat and Energy: U = Q + W. (B) Methods of Heat Transport. (C) Infintesimal Work: Exact vs Inexact Differentials
Availability. Second Law Analysis of Systems. Reading Problems 10.1 10.4 10.59, 10.65, 10.66, 10.67 10.69, 10.75, 10.81, 10.
 Availability Readg Problems 10.1 10.4 10.59, 10.65, 10.66, 10.67 10.69, 10.75, 10.81, 10.88 Second Law Analysis of Systems AVAILABILITY: the theoretical maximum amount of reversible work that can be obtaed
Availability Readg Problems 10.1 10.4 10.59, 10.65, 10.66, 10.67 10.69, 10.75, 10.81, 10.88 Second Law Analysis of Systems AVAILABILITY: the theoretical maximum amount of reversible work that can be obtaed
Give all answers in MKS units: energy in Joules, pressure in Pascals, volume in m 3, etc. Only work the number of problems required. Chose wisely.
 Chemistry 45/456 0 July, 007 Midterm Examination Professor G. Drobny Universal gas constant=r=8.3j/mole-k=0.08l-atm/mole-k Joule=J= Nt-m=kg-m /s 0J= L-atm. Pa=J/m 3 =N/m. atm=.0x0 5 Pa=.0 bar L=0-3 m 3.
Chemistry 45/456 0 July, 007 Midterm Examination Professor G. Drobny Universal gas constant=r=8.3j/mole-k=0.08l-atm/mole-k Joule=J= Nt-m=kg-m /s 0J= L-atm. Pa=J/m 3 =N/m. atm=.0x0 5 Pa=.0 bar L=0-3 m 3.
Contents. 2 Lines and Circles 3 2.1 Cartesian Coordinates... 3 2.2 Distance and Midpoint Formulas... 3 2.3 Lines... 3 2.4 Circles...
 Contents Lines and Circles 3.1 Cartesian Coordinates.......................... 3. Distance and Midpoint Formulas.................... 3.3 Lines.................................. 3.4 Circles..................................
Contents Lines and Circles 3.1 Cartesian Coordinates.......................... 3. Distance and Midpoint Formulas.................... 3.3 Lines.................................. 3.4 Circles..................................
Lecture 1: Physical Equilibria The Temperature Dependence of Vapor Pressure
 Lecture 1: Physical Equilibria The Temperature Dependence of Vapor Pressure Our first foray into equilibria is to examine phenomena associated with two phases of matter achieving equilibrium in which the
Lecture 1: Physical Equilibria The Temperature Dependence of Vapor Pressure Our first foray into equilibria is to examine phenomena associated with two phases of matter achieving equilibrium in which the
The Mean Value Theorem
 The Mean Value Theorem THEOREM (The Extreme Value Theorem): If f is continuous on a closed interval [a, b], then f attains an absolute maximum value f(c) and an absolute minimum value f(d) at some numbers
The Mean Value Theorem THEOREM (The Extreme Value Theorem): If f is continuous on a closed interval [a, b], then f attains an absolute maximum value f(c) and an absolute minimum value f(d) at some numbers
The First Law of Thermodynamics
 The First aw of Thermodynamics Q and W are process (path)-dependent. (Q W) = E int is independent of the process. E int = E int,f E int,i = Q W (first law) Q: + heat into the system; heat lost from the
The First aw of Thermodynamics Q and W are process (path)-dependent. (Q W) = E int is independent of the process. E int = E int,f E int,i = Q W (first law) Q: + heat into the system; heat lost from the
Thermodynamics 2nd year physics A. M. Steane 2000, revised 2004, 2006
 Thermodynamics 2nd year physics A. M. Steane 2000, revised 2004, 2006 We will base our tutorials around Adkins, Equilibrium Thermodynamics, 2nd ed (McGraw-Hill). Zemansky, Heat and Thermodynamics is good
Thermodynamics 2nd year physics A. M. Steane 2000, revised 2004, 2006 We will base our tutorials around Adkins, Equilibrium Thermodynamics, 2nd ed (McGraw-Hill). Zemansky, Heat and Thermodynamics is good
COMPREHENSIVE INSTRUCTIONAL PROGRAM REVIEW. Physics and Astronomy Moreno Valley College. Submitted by: Dipen Bhattacharya, PhD Associate Professor
 COMPREHENSIVE INSTRUCTIONAL PROGRAM REVIEW Physics and Astronomy Moreno Valley College Submitted by: Dipen Bhattacharya, PhD Associate Professor Round Three 2012-2016 Office of Educational Services 1 Comprehensive
COMPREHENSIVE INSTRUCTIONAL PROGRAM REVIEW Physics and Astronomy Moreno Valley College Submitted by: Dipen Bhattacharya, PhD Associate Professor Round Three 2012-2016 Office of Educational Services 1 Comprehensive
explain your reasoning
 I. A mechanical device shakes a ball-spring system vertically at its natural frequency. The ball is attached to a string, sending a harmonic wave in the positive x-direction. +x a) The ball, of mass M,
I. A mechanical device shakes a ball-spring system vertically at its natural frequency. The ball is attached to a string, sending a harmonic wave in the positive x-direction. +x a) The ball, of mass M,
UNIT 2 REFRIGERATION CYCLE
 UNIT 2 REFRIGERATION CYCLE Refrigeration Cycle Structure 2. Introduction Objectives 2.2 Vapour Compression Cycle 2.2. Simple Vapour Compression Refrigeration Cycle 2.2.2 Theoretical Vapour Compression
UNIT 2 REFRIGERATION CYCLE Refrigeration Cycle Structure 2. Introduction Objectives 2.2 Vapour Compression Cycle 2.2. Simple Vapour Compression Refrigeration Cycle 2.2.2 Theoretical Vapour Compression
The first law: transformation of energy into heat and work. Chemical reactions can be used to provide heat and for doing work.
 The first law: transformation of energy into heat and work Chemical reactions can be used to provide heat and for doing work. Compare fuel value of different compounds. What drives these reactions to proceed
The first law: transformation of energy into heat and work Chemical reactions can be used to provide heat and for doing work. Compare fuel value of different compounds. What drives these reactions to proceed
Lecture 14 Chapter 19 Ideal Gas Law and Kinetic Theory of Gases. Chapter 20 Entropy and the Second Law of Thermodynamics
 Lecture 14 Chapter 19 Ideal Gas Law and Kinetic Theory of Gases Now we to look at temperature, pressure, and internal energy in terms of the motion of molecules and atoms? Relate to the 1st Law of Thermodynamics
Lecture 14 Chapter 19 Ideal Gas Law and Kinetic Theory of Gases Now we to look at temperature, pressure, and internal energy in terms of the motion of molecules and atoms? Relate to the 1st Law of Thermodynamics
Module P7.3 Internal energy, heat and energy transfer
 F L E X I B L E L E A R N I N G A P P R O A C H T O P H Y S I C S Module P7.3 Internal energy, heat and energy transfer 1 Opening items 1.1 Module introduction 1.2 Fast track questions 1.3 Ready to study?
F L E X I B L E L E A R N I N G A P P R O A C H T O P H Y S I C S Module P7.3 Internal energy, heat and energy transfer 1 Opening items 1.1 Module introduction 1.2 Fast track questions 1.3 Ready to study?
Differential Relations for Fluid Flow. Acceleration field of a fluid. The differential equation of mass conservation
 Differential Relations for Fluid Flow In this approach, we apply our four basic conservation laws to an infinitesimally small control volume. The differential approach provides point by point details of
Differential Relations for Fluid Flow In this approach, we apply our four basic conservation laws to an infinitesimally small control volume. The differential approach provides point by point details of
Chapter 17: Change of Phase
 Chapter 17: Change of Phase Conceptual Physics, 10e (Hewitt) 3) Evaporation is a cooling process and condensation is A) a warming process. B) a cooling process also. C) neither a warming nor cooling process.
Chapter 17: Change of Phase Conceptual Physics, 10e (Hewitt) 3) Evaporation is a cooling process and condensation is A) a warming process. B) a cooling process also. C) neither a warming nor cooling process.
ES-7A Thermodynamics HW 1: 2-30, 32, 52, 75, 121, 125; 3-18, 24, 29, 88 Spring 2003 Page 1 of 6
 Spring 2003 Page 1 of 6 2-30 Steam Tables Given: Property table for H 2 O Find: Complete the table. T ( C) P (kpa) h (kj/kg) x phase description a) 120.23 200 2046.03 0.7 saturated mixture b) 140 361.3
Spring 2003 Page 1 of 6 2-30 Steam Tables Given: Property table for H 2 O Find: Complete the table. T ( C) P (kpa) h (kj/kg) x phase description a) 120.23 200 2046.03 0.7 saturated mixture b) 140 361.3
Note on growth and growth accounting
 CHAPTER 0 Note on growth and growth accounting 1. Growth and the growth rate In this section aspects of the mathematical concept of the rate of growth used in growth models and in the empirical analysis
CHAPTER 0 Note on growth and growth accounting 1. Growth and the growth rate In this section aspects of the mathematical concept of the rate of growth used in growth models and in the empirical analysis
Applications of Fermat s Little Theorem and Congruences
 Applications of Fermat s Little Theorem and Congruences Definition: Let m be a positive integer. Then integers a and b are congruent modulo m, denoted by a b mod m, if m (a b). Example: 3 1 mod 2, 6 4
Applications of Fermat s Little Theorem and Congruences Definition: Let m be a positive integer. Then integers a and b are congruent modulo m, denoted by a b mod m, if m (a b). Example: 3 1 mod 2, 6 4
DRAFT. Appendix C.2 - Air Conditioning Thermodynamics 1
 Appendix C.2 - Air Conditioning Thermodynamics 1 To aid in discussing the alternative technologies, it is helpful to have a basic description of how air conditioning systems work. Heat normally flows from
Appendix C.2 - Air Conditioning Thermodynamics 1 To aid in discussing the alternative technologies, it is helpful to have a basic description of how air conditioning systems work. Heat normally flows from
APPLIED THERMODYNAMICS. TUTORIAL No.3 GAS TURBINE POWER CYCLES. Revise gas expansions in turbines. Study the Joule cycle with friction.
 APPLIED HERMODYNAMICS UORIAL No. GAS URBINE POWER CYCLES In this tutorial you will do the following. Revise gas expansions in turbines. Revise the Joule cycle. Study the Joule cycle with friction. Extend
APPLIED HERMODYNAMICS UORIAL No. GAS URBINE POWER CYCLES In this tutorial you will do the following. Revise gas expansions in turbines. Revise the Joule cycle. Study the Joule cycle with friction. Extend
TOPIC 4: DERIVATIVES
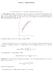 TOPIC 4: DERIVATIVES 1. The derivative of a function. Differentiation rules 1.1. The slope of a curve. The slope of a curve at a point P is a measure of the steepness of the curve. If Q is a point on the
TOPIC 4: DERIVATIVES 1. The derivative of a function. Differentiation rules 1.1. The slope of a curve. The slope of a curve at a point P is a measure of the steepness of the curve. If Q is a point on the
State Newton's second law of motion for a particle, defining carefully each term used.
 5 Question 1. [Marks 28] An unmarked police car P is, travelling at the legal speed limit, v P, on a straight section of highway. At time t = 0, the police car is overtaken by a car C, which is speeding
5 Question 1. [Marks 28] An unmarked police car P is, travelling at the legal speed limit, v P, on a straight section of highway. At time t = 0, the police car is overtaken by a car C, which is speeding
7.6 Approximation Errors and Simpson's Rule
 WileyPLUS: Home Help Contact us Logout Hughes-Hallett, Calculus: Single and Multivariable, 4/e Calculus I, II, and Vector Calculus Reading content Integration 7.1. Integration by Substitution 7.2. Integration
WileyPLUS: Home Help Contact us Logout Hughes-Hallett, Calculus: Single and Multivariable, 4/e Calculus I, II, and Vector Calculus Reading content Integration 7.1. Integration by Substitution 7.2. Integration
ENERGY CONSERVATION The First Law of Thermodynamics and the Work/Kinetic-Energy Theorem
 PH-211 A. La Rosa ENERGY CONSERVATION The irst Law of Thermodynamics and the Work/Kinetic-Energy Theorem ENERGY TRANSER of ENERGY Heat-transfer Q Macroscopic external Work W done on a system ENERGY CONSERVATION
PH-211 A. La Rosa ENERGY CONSERVATION The irst Law of Thermodynamics and the Work/Kinetic-Energy Theorem ENERGY TRANSER of ENERGY Heat-transfer Q Macroscopic external Work W done on a system ENERGY CONSERVATION
Topic 3b: Kinetic Theory
 Topic 3b: Kinetic Theory What is temperature? We have developed some statistical language to simplify describing measurements on physical systems. When we measure the temperature of a system, what underlying
Topic 3b: Kinetic Theory What is temperature? We have developed some statistical language to simplify describing measurements on physical systems. When we measure the temperature of a system, what underlying
L 2 : x = s + 1, y = s, z = 4s + 4. 3. Suppose that C has coordinates (x, y, z). Then from the vector equality AC = BD, one has
 The line L through the points A and B is parallel to the vector AB = 3, 2, and has parametric equations x = 3t + 2, y = 2t +, z = t Therefore, the intersection point of the line with the plane should satisfy:
The line L through the points A and B is parallel to the vector AB = 3, 2, and has parametric equations x = 3t + 2, y = 2t +, z = t Therefore, the intersection point of the line with the plane should satisfy:
( 1)2 + 2 2 + 2 2 = 9 = 3 We would like to make the length 6. The only vectors in the same direction as v are those
 1.(6pts) Which of the following vectors has the same direction as v 1,, but has length 6? (a), 4, 4 (b),, (c) 4,, 4 (d), 4, 4 (e) 0, 6, 0 The length of v is given by ( 1) + + 9 3 We would like to make
1.(6pts) Which of the following vectors has the same direction as v 1,, but has length 6? (a), 4, 4 (b),, (c) 4,, 4 (d), 4, 4 (e) 0, 6, 0 The length of v is given by ( 1) + + 9 3 We would like to make
(a) We have x = 3 + 2t, y = 2 t, z = 6 so solving for t we get the symmetric equations. x 3 2. = 2 y, z = 6. t 2 2t + 1 = 0,
 Name: Solutions to Practice Final. Consider the line r(t) = 3 + t, t, 6. (a) Find symmetric equations for this line. (b) Find the point where the first line r(t) intersects the surface z = x + y. (a) We
Name: Solutions to Practice Final. Consider the line r(t) = 3 + t, t, 6. (a) Find symmetric equations for this line. (b) Find the point where the first line r(t) intersects the surface z = x + y. (a) We
THERMODYNAMICS: COURSE INTRODUCTION
 UNIFIED ENGINEERING 2000 Lecture Outlines Ian A. Waitz THERMODYNAMICS: COURSE INTRODUCTION Course Learning Objectives: To be able to use the First Law of Thermodynamics to estimate the potential for thermomechanical
UNIFIED ENGINEERING 2000 Lecture Outlines Ian A. Waitz THERMODYNAMICS: COURSE INTRODUCTION Course Learning Objectives: To be able to use the First Law of Thermodynamics to estimate the potential for thermomechanical
Equilibrium. Ron Robertson
 Equilibrium Ron Robertson Basic Ideas A. Extent of Reaction Many reactions do not go to completion. Those that do not are reversible with a forward reaction and reverse reaction. To be really correct we
Equilibrium Ron Robertson Basic Ideas A. Extent of Reaction Many reactions do not go to completion. Those that do not are reversible with a forward reaction and reverse reaction. To be really correct we
Section 12.6: Directional Derivatives and the Gradient Vector
 Section 26: Directional Derivatives and the Gradient Vector Recall that if f is a differentiable function of x and y and z = f(x, y), then the partial derivatives f x (x, y) and f y (x, y) give the rate
Section 26: Directional Derivatives and the Gradient Vector Recall that if f is a differentiable function of x and y and z = f(x, y), then the partial derivatives f x (x, y) and f y (x, y) give the rate
How To Calculate The Performance Of A Refrigerator And Heat Pump
 THERMODYNAMICS TUTORIAL 5 HEAT PUMPS AND REFRIGERATION On completion of this tutorial you should be able to do the following. Discuss the merits of different refrigerants. Use thermodynamic tables for
THERMODYNAMICS TUTORIAL 5 HEAT PUMPS AND REFRIGERATION On completion of this tutorial you should be able to do the following. Discuss the merits of different refrigerants. Use thermodynamic tables for
Entropy and the Kinetic Theory: the Molecular Picture
 previous index next Entropy and the Kinetic Theory: the Molecular Picture Michael Fowler 7/15/08 Searching for a Molecular Description of Entropy Clausius introduced entropy as a new thermodynamic variable
previous index next Entropy and the Kinetic Theory: the Molecular Picture Michael Fowler 7/15/08 Searching for a Molecular Description of Entropy Clausius introduced entropy as a new thermodynamic variable
88 CHAPTER 2. VECTOR FUNCTIONS. . First, we need to compute T (s). a By definition, r (s) T (s) = 1 a sin s a. sin s a, cos s a
 88 CHAPTER. VECTOR FUNCTIONS.4 Curvature.4.1 Definitions and Examples The notion of curvature measures how sharply a curve bends. We would expect the curvature to be 0 for a straight line, to be very small
88 CHAPTER. VECTOR FUNCTIONS.4 Curvature.4.1 Definitions and Examples The notion of curvature measures how sharply a curve bends. We would expect the curvature to be 0 for a straight line, to be very small
Some Comments on the Derivative of a Vector with applications to angular momentum and curvature. E. L. Lady (October 18, 2000)
 Some Comments on the Derivative of a Vector with applications to angular momentum and curvature E. L. Lady (October 18, 2000) Finding the formula in polar coordinates for the angular momentum of a moving
Some Comments on the Derivative of a Vector with applications to angular momentum and curvature E. L. Lady (October 18, 2000) Finding the formula in polar coordinates for the angular momentum of a moving
Increasing for all. Convex for all. ( ) Increasing for all (remember that the log function is only defined for ). ( ) Concave for all.
 1. Differentiation The first derivative of a function measures by how much changes in reaction to an infinitesimal shift in its argument. The largest the derivative (in absolute value), the faster is evolving.
1. Differentiation The first derivative of a function measures by how much changes in reaction to an infinitesimal shift in its argument. The largest the derivative (in absolute value), the faster is evolving.
Math 2443, Section 16.3
 Math 44, Section 6. Review These notes will supplement not replace) the lectures based on Section 6. Section 6. i) ouble integrals over general regions: We defined double integrals over rectangles in the
Math 44, Section 6. Review These notes will supplement not replace) the lectures based on Section 6. Section 6. i) ouble integrals over general regions: We defined double integrals over rectangles in the
AP Physics B 2008 Scoring Guidelines
 P Physics B 008 Scoring Guidelines The College Board: Connecting Students to College Success The College Board is a not-for-profit membership association whose mission is to connect students to college
P Physics B 008 Scoring Guidelines The College Board: Connecting Students to College Success The College Board is a not-for-profit membership association whose mission is to connect students to college
3 Contour integrals and Cauchy s Theorem
 3 ontour integrals and auchy s Theorem 3. Line integrals of complex functions Our goal here will be to discuss integration of complex functions = u + iv, with particular regard to analytic functions. Of
3 ontour integrals and auchy s Theorem 3. Line integrals of complex functions Our goal here will be to discuss integration of complex functions = u + iv, with particular regard to analytic functions. Of
AP Calculus AB 2005 Free-Response Questions
 AP Calculus AB 25 Free-Response Questions The College Board: Connecting Students to College Success The College Board is a not-for-profit membership association whose mission is to connect students to
AP Calculus AB 25 Free-Response Questions The College Board: Connecting Students to College Success The College Board is a not-for-profit membership association whose mission is to connect students to
15.1. Exact Differential Equations. Exact First-Order Equations. Exact Differential Equations Integrating Factors
 SECTION 5. Eact First-Order Equations 09 SECTION 5. Eact First-Order Equations Eact Differential Equations Integrating Factors Eact Differential Equations In Section 5.6, ou studied applications of differential
SECTION 5. Eact First-Order Equations 09 SECTION 5. Eact First-Order Equations Eact Differential Equations Integrating Factors Eact Differential Equations In Section 5.6, ou studied applications of differential
SOLUTIONS. f x = 6x 2 6xy 24x, f y = 3x 2 6y. To find the critical points, we solve
 SOLUTIONS Problem. Find the critical points of the function f(x, y = 2x 3 3x 2 y 2x 2 3y 2 and determine their type i.e. local min/local max/saddle point. Are there any global min/max? Partial derivatives
SOLUTIONS Problem. Find the critical points of the function f(x, y = 2x 3 3x 2 y 2x 2 3y 2 and determine their type i.e. local min/local max/saddle point. Are there any global min/max? Partial derivatives
Problem Set 12: Kinetic Theory; Mechanical Equivalent of Heat Solutions
 MASSACHUSETTS INSTITUTE OF TECHNOLOGY Department of Physics Physics 8.01T Fall Term 2004 Problem Set 12: Kinetic Theory; Mechanical Equivalent of Heat Solutions Problem 1: Isothermal Ideal Gas Atmosphere
MASSACHUSETTS INSTITUTE OF TECHNOLOGY Department of Physics Physics 8.01T Fall Term 2004 Problem Set 12: Kinetic Theory; Mechanical Equivalent of Heat Solutions Problem 1: Isothermal Ideal Gas Atmosphere
Chem 338 Homework Set #5 solutions October 10, 2001 From Atkins: 5.2, 5.9, 5.12, 5.13, 5.15, 5.17, 5.21
 Chem 8 Homework Set #5 solutions October 10, 2001 From Atkins: 5.2, 5.9, 5.12, 5.1, 5.15, 5.17, 5.21 5.2) The density of rhombic sulfur is 2.070 g cm - and that of monoclinic sulfur is 1.957 g cm -. Can
Chem 8 Homework Set #5 solutions October 10, 2001 From Atkins: 5.2, 5.9, 5.12, 5.1, 5.15, 5.17, 5.21 5.2) The density of rhombic sulfur is 2.070 g cm - and that of monoclinic sulfur is 1.957 g cm -. Can
39 kg of water at 10 C is mixed with 360 kg of ice at -7 C.
 39 kg of water at 10 C is mixed with 360 kg of ice at -7 C. (The heat capacity of water is 4190 J/(kg C), that of ice is 2090 J/(kg C), and the heat of fusion of water is 3.34x10 5 J/kg. A. 320 J/K B.
39 kg of water at 10 C is mixed with 360 kg of ice at -7 C. (The heat capacity of water is 4190 J/(kg C), that of ice is 2090 J/(kg C), and the heat of fusion of water is 3.34x10 5 J/kg. A. 320 J/K B.
k 2f, k 2r C 2 H 5 + H C 2 H 6
 hemical Engineering HE 33 F pplied Reaction Kinetics Fall 04 Problem Set 4 Solution Problem. The following elementary steps are proposed for a gas phase reaction: Elementary Steps Rate constants H H f,
hemical Engineering HE 33 F pplied Reaction Kinetics Fall 04 Problem Set 4 Solution Problem. The following elementary steps are proposed for a gas phase reaction: Elementary Steps Rate constants H H f,
