Prescribing Information. EUMOVATE Cream (clobetasone butyrate, 0.05%) EUMOVATE Ointment (clobetasone butyrate, 0.05%)
|
|
|
- Valentine Webster
- 7 years ago
- Views:
Transcription
1 Prescribing Information EUMOVATE Cream EUMOVATE Ointment Topical Corticosteroid
2 Prescribing Information EUMOVATE Cream EUMOVATE Ointment THERAPEUTIC CLASSIFICATION Topical corticosteroid ACTIONS AND CLINICAL PHARMACOLOGY The corticosteroids are a class of compounds comprising steroid hormones secreted by the adrenal cortex and their synthetic analogs. In pharmacologic doses, corticosteroids are used primarily for their anti-inflammatory and/or immunosuppressive effects. Topical corticosteroids such as clobetasone 17-butyrate are effective in the treatment of corticosteroid-responsive dermatoses primarily because of their anti-inflammatory, anti-pruritic, and vasoconstrictive actions. However, while the physiologic, pharmacologic and clinical effects of the corticosteroids are well known, the exact mechanisms of their actions in each disease are uncertain. Clobetasone butyrate has been shown to have topical and systemic pharmacologic and metabolic effects characteristic of the corticosteroid class of drugs.
3 Indications and Clinical Use EUMOVATE (clobetasone butyrate) Cream and Ointment are indicated in the treatment of milder forms of eczema, seborrheic dermatitis, and other corticosteroid-responsive skin conditions, which do not require the use of a more potent topical corticosteroid. Contraindications Clobetasone butyrate cream and ointment are not indicated for the treatment of primarily infected skin lesions caused by infection with fungi or bacteria if no anti-infective agent is used simultaneously, primary cutaneous viral infections (i.e., herpes simplex, vaccinia and varicella) or tuberculous skin lesions. Clobetasone butyrate cream and ointment are also contraindicated in patients with a hypersensitivity to any of the components of the preparation. Warnings EUMOVATE (clobetasone butyrate) Cream and Ointment should not be used in the eye and should be used with caution in lesions close to the eye as glaucoma may result. Posterior subcapsular cataracts have been reported following systemic use of corticosteroids. If clobetasone butyrate is used under occlusive dressing over extensive areas for prolonged periods, it is possible that sufficient absorption may take place to give rise to transient adrenal suppression. Long-term continuous topical corticosteroid therapy should be avoided where possible as adrenal suppression can occur even without occlusion. Patients should be advised to inform subsequent physicians of the prior use of corticosteroids.
4 Precautions Significant systemic absorption may result when corticosteroids are applied over large areas of the body. To minimise this possibility, treatment should be interrupted periodically or one area of the body should be treated at a time when long-term therapy is anticipated. In infants, the diaper may act as an occlusive dressing and increase absorption. As with all corticosteroids, prolonged application of EUMOVATE (clobetasone butyrate) Cream and Ointment to the face is undesirable. Prolonged or extensive use of topycal corticosteroid products may product atrophy of the skin and subcutaneous tissue, particularly on flexor surfaces and on the face. If this is noted, the use of EUMOVATE should be discontinued. EUMOVATE should be used with caution in patients with stasis dermatitis and other skin diseases associated with impaired circulation. If a symptomatic response is not noted within a few days to a week, the local application of EUMOVATE should be discontinued and the patient re-evaluated. In case of bacterial infections of the skin, appropriate anti-bacterial agents should be used as primary therapy. If it is considered necessary, the topical corticosteroid may be used as an adjunct to control inflammation, erythema and itching. If a symptomatic response is not noted within a few days to a week, the local application of corticosteroid should be discontinued until the infection is brought under control. During the use of topical corticosteroids, secondary infections may occur. Appropriate antimicrobial therapy should be used whenever treating inflammatory lesions which have become infected. Any spread of infection requires withdrawal of topical corticosteroid therapy,
5 and systemic administration of antimicrobial agents. Bacterial infection is encouraged by the warm, moist conditions induced by occlusive dressings, and so the skin should be cleansed before a fresh dressing is applied. The safety and effectiveness of EUMOVATE when used under occlusive dressings has not been determined. Although hypersensitivity reactions are rare with topically applied corticosteroids, the drug should be discontinued and appropriate therapy initiated if there are signs of hypersensitivity. Topical administration of corticosteroids to pregnant animals can cause abnormalities of fetal development. The relevance of this finding to human beings has not been established. However, the administration of clobetasone butyrate topical preparations during pregnancy and lactation should only be considered if the expected benefit to the mother is greater than any possible risk to the fetus. Drugs of this class should not be used extensively in pregnant patients in large amounts or for prolonged periods of time. Adverse Reactions When large areas of the body are being treated with EUMOVATE (clobetasone butyrate) Cream or Ointment it is possible that some patients will absorb sufficient steroid to cause transient adrenal suppression despite the low degree of systemic activity associated with clobetasone butyrate. Local burning, irritation, itching, skin atrophy, dryness of the skin, atrophy of subcutaneous tissues, telangiectasia, striae, change in pigmentation, secondary infection and hypertrichosis have been observed following topical corticosteroid therapy. Exacerbation of symptoms may occur.
6 Local atrophic changes could possibly occur in situations where moisture increases absorption of clobetasone butyrate, but only after prolonged use. In the unlikely event of signs of hypersensitivity appearing, application should stop immediately. Symptoms and Treatment of Overdosage Acute overdosage is very unlikely to occur. However, in the case of chronic overdosage or misuse, the features of hypercorticism may appear. As with any corticosteroid, treatment should be discontinued if the symptoms of hypercorticism appear. Dosage and Administration EUMOVATE (clobetasone butyrate) Cream. 0.05% and Ointment, 0.05% should be applied thinly to cover the affected area, and gently rubbed into the skin. Frequency of application is two to three times daily according to the severity of the condition. The total dose of EUMOVATE applied should not exceed 100 grams per week in adults.
7 Pharmaceutical Information Drug Substance Proper Name: Chemical Name: clobetasone butyrate (BANM, USAN, rinnm) 17 butyryloxy-21-chloro-9α-fluoro-16β-methyl-pregna-1,4-diene-3,11,20-trione Structural Formula: Molecular Formula: C 26 H 32 CIFO 5 Molecular Weight: 479 Description: Solubility: Melting Point: Composition: white to cream colored crystalline powder Clobetasone butyrate is insoluble in water, slightly soluble in ethanol, methanol and diethyl ether, soluble in dioxan and very soluble in toluene, chloroform, ethyl acetate, dimethysulphoxide and dimethylformamide 178ΕC Each gram of EUMOVATE Cream contains 0.05% w/w clobetasone butyrate in a cream base. Each gram of EUMOVATE Ointment contains 0.05% w/w clobetasone butyrate in an ointment base. Storage Conditions: Store below 30ΕC. Availability of Dosage Forms EUMOVATE (clobetasone butyrate) Cream, 0.05% and Ointment, 0.05% are available in 15 gram and 30 gram tubes.
Topical Tacrolimus or Pimecrolimus for the treatment of mild, moderate or severe atopic eczema. Effective Shared Care Agreement
 Topical Tacrolimus or Pimecrolimus for the treatment of mild, moderate or severe atopic eczema. Effective Shared Care Agreement A Copy of this page signed by all three parties should be retained in the
Topical Tacrolimus or Pimecrolimus for the treatment of mild, moderate or severe atopic eczema. Effective Shared Care Agreement A Copy of this page signed by all three parties should be retained in the
ZOVIRAX Cold Sore Cream
 Data Sheet ZOVIRAX Cold Sore Cream Aciclovir 5% w/w Presentation Topical cream Indications ZOVIRAX Cold Sore Cream is indicated for the treatment of Herpes simplex virus infections of the lips and face
Data Sheet ZOVIRAX Cold Sore Cream Aciclovir 5% w/w Presentation Topical cream Indications ZOVIRAX Cold Sore Cream is indicated for the treatment of Herpes simplex virus infections of the lips and face
NEW ZEALAND DATA SHEET BETNOVATE -C CREAM and OINTMENT
 NEW ZEALAND DATA SHEET BETNOVATE -C CREAM and OINTMENT Name of each active ingredient Betamethasone valerate 0.1% w/w and clioquinol 3% w/w Presentations BETNOVATE-C Cream is a smooth, straw-coloured water-miscible
NEW ZEALAND DATA SHEET BETNOVATE -C CREAM and OINTMENT Name of each active ingredient Betamethasone valerate 0.1% w/w and clioquinol 3% w/w Presentations BETNOVATE-C Cream is a smooth, straw-coloured water-miscible
1g cream or ointment contains 1 mg methylprednisolone aceponate.
 CONSUMER MEDICINE INFORMATION ADVANTAN 1g cream or ointment contains 1 mg methylprednisolone aceponate. What is in this leaflet Please read this leaflet carefully before you start using ADVANTAN. It will
CONSUMER MEDICINE INFORMATION ADVANTAN 1g cream or ointment contains 1 mg methylprednisolone aceponate. What is in this leaflet Please read this leaflet carefully before you start using ADVANTAN. It will
PRODUCT MONOGRAPH. HydroVal. (Hydrocortisone Valerate USP) Cream & Ointment 0.2 % Topical Corticosteroid
 PRODUCT MONOGRAPH HydroVal (Hydrocortisone Valerate USP) Cream & Ointment 0.2 % Topical Corticosteroid TaroPharma Preparation Date: A Division of Taro Pharmaceuticals Inc. September 02, 2003 130 East Drive
PRODUCT MONOGRAPH HydroVal (Hydrocortisone Valerate USP) Cream & Ointment 0.2 % Topical Corticosteroid TaroPharma Preparation Date: A Division of Taro Pharmaceuticals Inc. September 02, 2003 130 East Drive
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Aknemycin Solution 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 10 g of solution contains 0.2 g of erythromycin. Structural formula of
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Aknemycin Solution 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 10 g of solution contains 0.2 g of erythromycin. Structural formula of
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE VETERINARY MEDICINAL PRODUCT Prednicare Tablets 5mg 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains: Active Substance(s) Prednisolone
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE VETERINARY MEDICINAL PRODUCT Prednicare Tablets 5mg 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains: Active Substance(s) Prednisolone
PRODUCT INFORMATION. Silver sulfadiazine is a sulfonamide and has broad antimicrobial activity against both Grampositive and Gram-negative organisms.
 PRODUCT INFORMATION Name of the Medicine: FLAMAZINE CREAM 1.0% w/w Silver sulfadiazine 1% w/w Composition: Active ingredient. Silver sulfadiazine. Excipients. Polysorbate 60 Ph. Eur, Polysorbate 80 Ph.
PRODUCT INFORMATION Name of the Medicine: FLAMAZINE CREAM 1.0% w/w Silver sulfadiazine 1% w/w Composition: Active ingredient. Silver sulfadiazine. Excipients. Polysorbate 60 Ph. Eur, Polysorbate 80 Ph.
TOPICAL THERAPY. ADVANTAGES - increased dose of medication to affected area. - reduced systemic side effects and toxicity
 TOPICAL THERAPY ADVANTAGES - increased dose of medication to affected area. - reduced systemic side effects and toxicity DISADVANTAGES - takes time - may be greasy or messy - may have different preparations
TOPICAL THERAPY ADVANTAGES - increased dose of medication to affected area. - reduced systemic side effects and toxicity DISADVANTAGES - takes time - may be greasy or messy - may have different preparations
NERIDERM Cream, Ointment, Fatty Ointment
 PRESCRIBING INFORMATION NERIDERM Cream, Ointment, Fatty Ointment Qualitative and Quantitive Composition 1 g Neriderm contains 1 mg (0.1%) diflucortolone valerate. Pharmaceutical Form Cream, Ointment, Fatty
PRESCRIBING INFORMATION NERIDERM Cream, Ointment, Fatty Ointment Qualitative and Quantitive Composition 1 g Neriderm contains 1 mg (0.1%) diflucortolone valerate. Pharmaceutical Form Cream, Ointment, Fatty
For Mild to Moderate Plaque Psoriasis and Moderate to Severe Atopic Dermatitis
 For Mild to Moderate Plaque Psoriasis and Moderate to Severe Atopic Dermatitis OLUX-E (clobetasol propionate) Foam, 0.05% Please see Important Safety Information on back page and accompanying Full Prescribing
For Mild to Moderate Plaque Psoriasis and Moderate to Severe Atopic Dermatitis OLUX-E (clobetasol propionate) Foam, 0.05% Please see Important Safety Information on back page and accompanying Full Prescribing
Public Assessment Report. Pharmacy to General Sales List Reclassification. Pirinase Hayfever Relief for Adults 0.05% Nasal Spray.
 Public Assessment Report Pharmacy to General Sales List Reclassification Pirinase Hayfever Relief for Adults 0.05% Nasal Spray (Fluticasone) PL 00079/0688 Glaxo Wellcome UK Limited TABLE OF CONTENTS Introduction
Public Assessment Report Pharmacy to General Sales List Reclassification Pirinase Hayfever Relief for Adults 0.05% Nasal Spray (Fluticasone) PL 00079/0688 Glaxo Wellcome UK Limited TABLE OF CONTENTS Introduction
FLAMAZINE CREAM 1.0% w/w
 PRODUCT INFORMATION NAME OF THE MEDICINE: FLAMAZINE CREAM 1.0% w/w Silver sulfadiazine 1% w/w Composition: Active ingredient. Silver sulfadiazine. Excipients. Polysorbate 60 Ph. Eur, Polysorbate 80 Ph.
PRODUCT INFORMATION NAME OF THE MEDICINE: FLAMAZINE CREAM 1.0% w/w Silver sulfadiazine 1% w/w Composition: Active ingredient. Silver sulfadiazine. Excipients. Polysorbate 60 Ph. Eur, Polysorbate 80 Ph.
SUMMARY OF THE RISK MANAGEMENT PLAN (by medicinal product)
 PART VI SUMMARY OF THE RISK MANAGEMENT PLAN (by medicinal product) Format and content of the summary of the RMP The summary of the RMP part VI contains information based on RMP modules SI, SVIII and RMP
PART VI SUMMARY OF THE RISK MANAGEMENT PLAN (by medicinal product) Format and content of the summary of the RMP The summary of the RMP part VI contains information based on RMP modules SI, SVIII and RMP
VISTARIL (hydroxyzine pamoate) Capsules and Oral Suspension
 VISTARIL (hydroxyzine pamoate) Capsules and Oral Suspension DESCRIPTION Hydroxyzine pamoate is designated chemically as 1-(p-chlorobenzhydryl) 4- [2-(2-hydroxyethoxy) ethyl] diethylenediamine salt of 1,1
VISTARIL (hydroxyzine pamoate) Capsules and Oral Suspension DESCRIPTION Hydroxyzine pamoate is designated chemically as 1-(p-chlorobenzhydryl) 4- [2-(2-hydroxyethoxy) ethyl] diethylenediamine salt of 1,1
See 17 for PATIENT COUNSELING INFORMATION. Revised: 3/2016 FULL PRESCRIBING INFORMATION: CONTENTS* WARNING: ADRENAL CRISIS IN THE SETTING OF SHOCK OR
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use LYSODREN safely and effectively. See full prescribing information for LYSODREN. LYSODREN (mitotane)
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use LYSODREN safely and effectively. See full prescribing information for LYSODREN. LYSODREN (mitotane)
Foam: A Unique Delivery Vehicle for Topically Applied Formulations
 : A Unique Delivery Vehicle for Topically Applied Formulations Dov Tamarkin, PhD ix Ltd. Key Words:, Emulsion, Lipophilic Emulsion, Nanoemulsion, Aqueous, Hydroethanolic, Potent-Solvent, Suspension, Ointment,
: A Unique Delivery Vehicle for Topically Applied Formulations Dov Tamarkin, PhD ix Ltd. Key Words:, Emulsion, Lipophilic Emulsion, Nanoemulsion, Aqueous, Hydroethanolic, Potent-Solvent, Suspension, Ointment,
EFFIMET 1000 XR Metformin Hydrochloride extended release tablet
 BRAND NAME: Effimet XR. THERAPEUTIC CATEGORY: Anti-Diabetic PHARMACOLOGIC CLASS: Biguanides EFFIMET 1000 XR Metformin Hydrochloride extended release tablet COMPOSITION AND PRESENTATION Composition Each
BRAND NAME: Effimet XR. THERAPEUTIC CATEGORY: Anti-Diabetic PHARMACOLOGIC CLASS: Biguanides EFFIMET 1000 XR Metformin Hydrochloride extended release tablet COMPOSITION AND PRESENTATION Composition Each
Conjunctivitis - Pink Eye
 Conjunctivitis - Pink Eye Introduction Conjunctivitis is often called pink eye. It causes inflammation of the thin, clear lining inside the eyelid and on the white of the eye. The inflammation gives the
Conjunctivitis - Pink Eye Introduction Conjunctivitis is often called pink eye. It causes inflammation of the thin, clear lining inside the eyelid and on the white of the eye. The inflammation gives the
PACKAGE LEAFLET: INFORMATION FOR THE USER. Elidel 10 mg/g Cream. pimecrolimus
 PACKAGE LEAFLET: INFORMATION FOR THE USER Elidel 10 mg/g Cream pimecrolimus Read all of this leaflet carefully before you start using Elidel cream Keep this leaflet. You may need to read it again. If you
PACKAGE LEAFLET: INFORMATION FOR THE USER Elidel 10 mg/g Cream pimecrolimus Read all of this leaflet carefully before you start using Elidel cream Keep this leaflet. You may need to read it again. If you
D.RIGOPOULOS, D.IOANNIDES,* D.KALOGEROMITROS, S.GREGORIOU AND A.KATSAMBAS
 British Journal of Dermatology 24; 151: 171 175. DOI: 1.1111/j.1365-2133.24.628.x Therapeutics Pimecrolimus cream 1% vs. betamethasone 17-valerate Æ1% cream in the treatment of seborrhoeic dermatitis.
British Journal of Dermatology 24; 151: 171 175. DOI: 1.1111/j.1365-2133.24.628.x Therapeutics Pimecrolimus cream 1% vs. betamethasone 17-valerate Æ1% cream in the treatment of seborrhoeic dermatitis.
PHOSPHATE-SANDOZ Tablets (High dose phosphate supplement)
 1 PHOSPHATE-SANDOZ Tablets (High dose phosphate supplement) PHOSPHATE-SANDOZ PHOSPHATE-SANDOZ Tablets are a high dose phosphate supplement containing sodium phosphate monobasic. The CAS registry number
1 PHOSPHATE-SANDOZ Tablets (High dose phosphate supplement) PHOSPHATE-SANDOZ PHOSPHATE-SANDOZ Tablets are a high dose phosphate supplement containing sodium phosphate monobasic. The CAS registry number
COMMITTEE ON HERBAL MEDICINAL PRODUCTS (HMPC) FINAL COMMUNITY HERBAL MONOGRAPH ON HYPERICUM PERFORATUM L., HERBA (TRADITIONAL USE)
 European Medicines Agency Evaluation of Medicines for Human Use London, 12 November 2009 Doc. Ref.: EMEA/HMPC/745582/2009 COMMITTEE ON HERBAL MEDICINAL PRODUCTS (HMPC) FINAL COMMUNITY HERBAL MONOGRAPH
European Medicines Agency Evaluation of Medicines for Human Use London, 12 November 2009 Doc. Ref.: EMEA/HMPC/745582/2009 COMMITTEE ON HERBAL MEDICINAL PRODUCTS (HMPC) FINAL COMMUNITY HERBAL MONOGRAPH
3% Sodium Chloride Injection, USP 5% Sodium Chloride Injection, USP
 PRESCRIBING INFORMATION 3% Sodium Chloride Injection, USP 5% Sodium Chloride Injection, USP IV Fluid and Electrolyte Replenisher Baxter Corporation Mississauga, Ontario L5N 0C2 Canada Date of Revision:
PRESCRIBING INFORMATION 3% Sodium Chloride Injection, USP 5% Sodium Chloride Injection, USP IV Fluid and Electrolyte Replenisher Baxter Corporation Mississauga, Ontario L5N 0C2 Canada Date of Revision:
Acne (Acne Vulgaris) A common type of bacteria that lives on the skin, known as Propionibacterium acnes, sometimes
 Acne (Acne Vulgaris) Acne, clinically known as acne vulgaris, is the most common skin disease. It affects 85% of teenagers, some as young as 12, and often continues into adulthood. It is also called pimples,
Acne (Acne Vulgaris) Acne, clinically known as acne vulgaris, is the most common skin disease. It affects 85% of teenagers, some as young as 12, and often continues into adulthood. It is also called pimples,
ALLERGENIC EXTRACT. Prescription Set of Serial Dilutions (or Maintenance Vial (s)) INSTRUCTIONS FOR USE. U.S. Government License No.
 ALLERGENIC EXTRACT Prescription Set of Serial Dilutions (or Maintenance Vial (s)) INSTRUCTIONS FOR USE U.S. Government License No. 308 Revised 07/04 PO Box 800 Lenoir, NC 28645 USA DESCRIPTION This set
ALLERGENIC EXTRACT Prescription Set of Serial Dilutions (or Maintenance Vial (s)) INSTRUCTIONS FOR USE U.S. Government License No. 308 Revised 07/04 PO Box 800 Lenoir, NC 28645 USA DESCRIPTION This set
PHENYLEPHRINE HYDROCHLORIDE INJECTION USP
 PRESCRIBING INFORMATION PHENYLEPHRINE HYDROCHLORIDE INJECTION USP 10 mg/ml Sandoz Canada Inc. Date of Preparation: September 1992 145 Jules-Léger Date of Revision : January 13, 2011 Boucherville, QC, Canada
PRESCRIBING INFORMATION PHENYLEPHRINE HYDROCHLORIDE INJECTION USP 10 mg/ml Sandoz Canada Inc. Date of Preparation: September 1992 145 Jules-Léger Date of Revision : January 13, 2011 Boucherville, QC, Canada
What Alcohol Does to the Body. Chapter 25 Lesson 2
 What Alcohol Does to the Body Chapter 25 Lesson 2 Short-Term Effects of Drinking The short-term term effects of alcohol on the body depend on several factors including: amount of alcohol consumed, gender,
What Alcohol Does to the Body Chapter 25 Lesson 2 Short-Term Effects of Drinking The short-term term effects of alcohol on the body depend on several factors including: amount of alcohol consumed, gender,
Betnovate -C 0.1% / 3% w/w Cream Betamethasone (as valerate) and Clioquinol
 [GSK Logo] Package Leaflet: Information for the User Betnovate -C 0.1% / 3% w/w Cream Betamethasone (as valerate) and Clioquinol Read all of this leaflet carefully before you start using this medicine
[GSK Logo] Package Leaflet: Information for the User Betnovate -C 0.1% / 3% w/w Cream Betamethasone (as valerate) and Clioquinol Read all of this leaflet carefully before you start using this medicine
COMMITTEE ON HERBAL MEDICINAL PRODUCTS (HMPC) FINAL COMMUNITY HERBAL MONOGRAPH ON AVENA SATIVA L., FRUCTUS
 European Medicines Agency Evaluation of Medicines for Human Use London, 4 September 2008 Doc. Ref. EMEA/HMPC/368600/2007 COMMITTEE ON HERBAL MEDICINAL PRODUCTS (HMPC) FINAL COMMUNITY HERBAL MONOGRAPH ON
European Medicines Agency Evaluation of Medicines for Human Use London, 4 September 2008 Doc. Ref. EMEA/HMPC/368600/2007 COMMITTEE ON HERBAL MEDICINAL PRODUCTS (HMPC) FINAL COMMUNITY HERBAL MONOGRAPH ON
Revised: 9/2014. LUMIGAN (bimatoprost ophthalmic solution) 0.01% for topical ophthalmic use. Initial U.S. Approval: 2001
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use LUMIGAN (bimatoprost ophthalmic solution) 0.01% safely and effectively. See full prescribing information
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use LUMIGAN (bimatoprost ophthalmic solution) 0.01% safely and effectively. See full prescribing information
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Aknemycin Plus 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 100 g of solution contains: Erythromycin 4.0 g, Tretinoin 0.025 g. For a full
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Aknemycin Plus 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 100 g of solution contains: Erythromycin 4.0 g, Tretinoin 0.025 g. For a full
FLAMAZINE CREAM MSDS No. 185
 SECTION 1 IDENTIFICATION OF THE MATERIAL AND SUPPLIER Product Product Name FLAMAZINE CREAM Other Names - Manufacturer s Product Code 66800531, 66800532, 66150153, 66150155 Use Topical antibacterial cream,
SECTION 1 IDENTIFICATION OF THE MATERIAL AND SUPPLIER Product Product Name FLAMAZINE CREAM Other Names - Manufacturer s Product Code 66800531, 66800532, 66150153, 66150155 Use Topical antibacterial cream,
CHLORHEXIDINE ACETATE WITH CETRIMIDE (%) ANTISEPTIC SOLUTION
 CHLORHEXIDINE ACETATE WITH CETRIMIDE (%) ANTISEPTIC SOLUTION NAME OF THE MEDICINE Proprietary name: Non-proprietary name: Chemical Name: Molecular formula: Baxter Chlorhexidine Cetrimide Antiseptic Solution
CHLORHEXIDINE ACETATE WITH CETRIMIDE (%) ANTISEPTIC SOLUTION NAME OF THE MEDICINE Proprietary name: Non-proprietary name: Chemical Name: Molecular formula: Baxter Chlorhexidine Cetrimide Antiseptic Solution
It can therefore be bought without a prescription but should be used correctly to ensure its efficacy and to reduce side effects.
 140 mg Medicated plaster Diclofenac Sodium BEFORE USE, CAREFULLY READ ALL THE INFORMATION GIVEN ON THE INFORMATIVE LEAFLET This medicinal product is intended for SELF-MEDICATION. It can be used to treat
140 mg Medicated plaster Diclofenac Sodium BEFORE USE, CAREFULLY READ ALL THE INFORMATION GIVEN ON THE INFORMATIVE LEAFLET This medicinal product is intended for SELF-MEDICATION. It can be used to treat
Professor Andrew Wright,
 Professor Andrew Wright, Systemic treatments are drugs taken as tablets or injections that travel through the bloodstream, dampening down the immune system to reach and treat eczema all over the body.
Professor Andrew Wright, Systemic treatments are drugs taken as tablets or injections that travel through the bloodstream, dampening down the immune system to reach and treat eczema all over the body.
NEW ZEALAND DATA SHEET NAPHCON-A Naphazoline hydrochloride and pheniramine maleate.
 NEW ZEALAND DATA SHEET NAPHCON-A Naphazoline hydrochloride and pheniramine maleate. PRESENTATION Eye Drops: NAPHCON-A Eye Drops are a combination of an antihistamine (pheniramine maleate) and a decongestant
NEW ZEALAND DATA SHEET NAPHCON-A Naphazoline hydrochloride and pheniramine maleate. PRESENTATION Eye Drops: NAPHCON-A Eye Drops are a combination of an antihistamine (pheniramine maleate) and a decongestant
Facial Use of Low Potency Steroids With Anti-microbials and Retinoids
 Facial Use of Low Potency Steroids With Anti-microbials and Retinoids Steven L. Harlan, MD Medical Director, Dermatology Center, Des Moines, IA Introduction: There have been many reports of topical corticosteroids
Facial Use of Low Potency Steroids With Anti-microbials and Retinoids Steven L. Harlan, MD Medical Director, Dermatology Center, Des Moines, IA Introduction: There have been many reports of topical corticosteroids
Glossary. amenorrhea, primary - from the beginning and lifelong; menstruation never begins at puberty.
 Glossary amenorrhea - absence or cessation of menstrual periods. amenorrhea, primary - from the beginning and lifelong; menstruation never begins at puberty. A amenorrhea, secondary - due to some physical
Glossary amenorrhea - absence or cessation of menstrual periods. amenorrhea, primary - from the beginning and lifelong; menstruation never begins at puberty. A amenorrhea, secondary - due to some physical
Results of the first clinical study on a new treatment for Psori asis: the T.M.S.T. method
 MINERAL MOR LTD Results of the first clinical study on a new treatment for Psori asis: the TMST method Marco Harari MD 12122012 Conclusions: 1 No side effects were recorded during the 5week study period
MINERAL MOR LTD Results of the first clinical study on a new treatment for Psori asis: the TMST method Marco Harari MD 12122012 Conclusions: 1 No side effects were recorded during the 5week study period
Summary of Product Characteristics
 Summary of Product Characteristics 1 NAME OF THE MEDICINAL PRODUCT Isotrexin 2% + 0.05% w/w Gel 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Erythromycin Isotretinoin 2.00% w/w 0.05% w/w Excipients: Contains
Summary of Product Characteristics 1 NAME OF THE MEDICINAL PRODUCT Isotrexin 2% + 0.05% w/w Gel 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Erythromycin Isotretinoin 2.00% w/w 0.05% w/w Excipients: Contains
A normal eye is protected by a layer of natural tears.
 A normal eye is protected by a layer of natural tears. If you need to use eye drops several times a day to make your eyes comfortably moist, you should consult your eye doctor. Helps increase tear production
A normal eye is protected by a layer of natural tears. If you need to use eye drops several times a day to make your eyes comfortably moist, you should consult your eye doctor. Helps increase tear production
A: Nursing Knowledge. Alberta Licensed Practical Nurses Competency Profile 1
 A: Nursing Knowledge Alberta Licensed Practical Nurses Competency Profile 1 Competency: A-1 Anatomy and Physiology A-1-1 A-1-2 A-1-3 A-1-4 A-1-5 A-1-6 A-1-7 A-1-8 Identify the normal structures and functions
A: Nursing Knowledge Alberta Licensed Practical Nurses Competency Profile 1 Competency: A-1 Anatomy and Physiology A-1-1 A-1-2 A-1-3 A-1-4 A-1-5 A-1-6 A-1-7 A-1-8 Identify the normal structures and functions
INFUSE Bone Graft. Patient Information Brochure
 INFUSE Bone Graft Patient Information Brochure This Patient Guide is designed to help you decide whether or not to have surgery using INFUSE Bone Graft to treat your broken tibia (lower leg). There are
INFUSE Bone Graft Patient Information Brochure This Patient Guide is designed to help you decide whether or not to have surgery using INFUSE Bone Graft to treat your broken tibia (lower leg). There are
INITIATING ORAL AUBAGIO (teriflunomide) THERAPY
 FOR YOUR PATIENTS WITH RELAPSING FORMS OF MS INITIATING ORAL AUBAGIO (teriflunomide) THERAPY WARNING: HEPATOTOXICITY AND RISK OF TERATOGENICITY Severe liver injury including fatal liver failure has been
FOR YOUR PATIENTS WITH RELAPSING FORMS OF MS INITIATING ORAL AUBAGIO (teriflunomide) THERAPY WARNING: HEPATOTOXICITY AND RISK OF TERATOGENICITY Severe liver injury including fatal liver failure has been
Leukocytoclastic Vasculitis and Stasis Dermatitis With Id Reaction
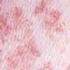 Id Reaction December 01, 2007 By David L. Kaplan, MD [1] A Photo Quiz to Hone Dermatologic Skills Case 1: A slightly pruritic eruption developed on the lower legs of a 39-year-old woman after she had an
Id Reaction December 01, 2007 By David L. Kaplan, MD [1] A Photo Quiz to Hone Dermatologic Skills Case 1: A slightly pruritic eruption developed on the lower legs of a 39-year-old woman after she had an
X-Plain Psoriasis Reference Summary
 X-Plain Psoriasis Reference Summary Introduction Psoriasis is a long-lasting skin disease that causes the skin to become inflamed. Patches of thick, red skin are covered with silvery scales. It affects
X-Plain Psoriasis Reference Summary Introduction Psoriasis is a long-lasting skin disease that causes the skin to become inflamed. Patches of thick, red skin are covered with silvery scales. It affects
The clinical studies have been performed in children, adolescents and adults, from 4 years up to 55 years of age.
 NAME OF THE MEDICINE TdaP-Booster. Diphtheria, tetanus and pertussis (acellular mono-component) vaccine (adsorbed, reduced antigen content). DESCRIPTION TdaP-Booster is a suspension for injection in pre-filled
NAME OF THE MEDICINE TdaP-Booster. Diphtheria, tetanus and pertussis (acellular mono-component) vaccine (adsorbed, reduced antigen content). DESCRIPTION TdaP-Booster is a suspension for injection in pre-filled
Stepping toward a different treatment option LEARN WHAT ACTHAR CAN DO FOR YOU
 FOR MS RELAPSES Stepping toward a different treatment option LEARN WHAT ACTHAR CAN DO FOR YOU As a person with multiple sclerosis (MS), you know firsthand the profound impact MS relapses can have on your
FOR MS RELAPSES Stepping toward a different treatment option LEARN WHAT ACTHAR CAN DO FOR YOU As a person with multiple sclerosis (MS), you know firsthand the profound impact MS relapses can have on your
FOR ORAL OR RECTAL ADMINISTRATION FOR THE PREVENTION AND TREATMENT OF PORTAL-SYSTEMIC ENCEPHALOPATHY
 FORM NO. 1360 VC3074 Rev.2/08 LACTULOSE SOLUTION, USP 10 g/15 ml FOR ORAL OR RECTAL ADMINISTRATION FOR THE PREVENTION AND TREATMENT OF PORTAL-SYSTEMIC ENCEPHALOPATHY Rx Only DESCRIPTION Lactulose is a
FORM NO. 1360 VC3074 Rev.2/08 LACTULOSE SOLUTION, USP 10 g/15 ml FOR ORAL OR RECTAL ADMINISTRATION FOR THE PREVENTION AND TREATMENT OF PORTAL-SYSTEMIC ENCEPHALOPATHY Rx Only DESCRIPTION Lactulose is a
MicroSilver BG TM. The innovative agent for beautiful, healthy skin.
 The innovative agent for beautiful, healthy skin. Inhalt Why MicroSilver BG TM? 3 What is MicroSilver BG TM? 3 How does MicroSilver BG TM work? 3 Products and usage 4 MicroSilver BG TM still used today
The innovative agent for beautiful, healthy skin. Inhalt Why MicroSilver BG TM? 3 What is MicroSilver BG TM? 3 How does MicroSilver BG TM work? 3 Products and usage 4 MicroSilver BG TM still used today
Learn More About Product Labeling
 Learn More About Product Labeling Product label The product label is developed during the formal process of review and approval by regulatory agencies of any medicine or medical product. There are specific
Learn More About Product Labeling Product label The product label is developed during the formal process of review and approval by regulatory agencies of any medicine or medical product. There are specific
0.9% Sodium Chloride Injection, USP In VIAFLEX Plastic Container
 Page 1 of 8 PRESCRIBING INFORMATION 0.9% Sodium Chloride Injection, USP In VIAFLEX Plastic Container IV Fluid and Electrolyte Replenisher Baxter Corporation Mississauga, Ontario L5N 0C2 Canada Date of
Page 1 of 8 PRESCRIBING INFORMATION 0.9% Sodium Chloride Injection, USP In VIAFLEX Plastic Container IV Fluid and Electrolyte Replenisher Baxter Corporation Mississauga, Ontario L5N 0C2 Canada Date of
Treatments for allergy are usually straightforward, safe and effective. Common treatments include:
 Allergy Medications The treatments prescribed for allergy control the symptoms and reactions; they do not cure the condition. However, using treatments as prescribed can show a huge change in a patient
Allergy Medications The treatments prescribed for allergy control the symptoms and reactions; they do not cure the condition. However, using treatments as prescribed can show a huge change in a patient
Mucodyne-Clear 250 mg/5 ml Syrup PL 04425/0665
 Mucodyne-Clear 250 mg/5 ml Syrup PL 04425/0665 UKPAR TABLE OF CONTENTS Lay summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 9 Summary of product characteristics Page 10 Patient
Mucodyne-Clear 250 mg/5 ml Syrup PL 04425/0665 UKPAR TABLE OF CONTENTS Lay summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 9 Summary of product characteristics Page 10 Patient
IMPORTANT DRUG WARNING Regarding Mycophenolate-Containing Products
 Dear Healthcare Provider: Mycophenolate REMS (Risk Evaluation and Mitigation Strategy) has been mandated by the FDA (Food and Drug Administration) due to postmarketing reports showing that exposure to
Dear Healthcare Provider: Mycophenolate REMS (Risk Evaluation and Mitigation Strategy) has been mandated by the FDA (Food and Drug Administration) due to postmarketing reports showing that exposure to
The Annual Direct Care of Asthma
 The Annual Direct Care of Asthma The annual direct health care cost of asthma in the United States is approximately $11.5 billion; indirect costs (e.g. lost productivity) add another $4.6 billion for a
The Annual Direct Care of Asthma The annual direct health care cost of asthma in the United States is approximately $11.5 billion; indirect costs (e.g. lost productivity) add another $4.6 billion for a
PHARMACOVIGILANCE GUIDELINES
 PHARMACOVIGILANCE GUIDELINES What is Pharmacovigilance? Pharmacovigilance is defined as the science and activities concerned with the detection, assessment, understanding and prevention of adverse reactions
PHARMACOVIGILANCE GUIDELINES What is Pharmacovigilance? Pharmacovigilance is defined as the science and activities concerned with the detection, assessment, understanding and prevention of adverse reactions
Chapter 4 Physiological Therapeutics. 1 Cryotherapy
 Chapter 4 Physiological Therapeutics 1 Cryotherapy CRYOTHERAPY PHYSIOLOGIC EFFECTS OF ICE APPLICATION 1. Decreased circulation 5. Increased tissue stiffness 2. Local vasoconstriction 6. Decreased muscle
Chapter 4 Physiological Therapeutics 1 Cryotherapy CRYOTHERAPY PHYSIOLOGIC EFFECTS OF ICE APPLICATION 1. Decreased circulation 5. Increased tissue stiffness 2. Local vasoconstriction 6. Decreased muscle
Hand Dermatitis in Health Care Workers
 Hand in Health Care Workers Safety and Health Assessment and Research for Prevention (SHARP) Program Washington Department of Labor and Industries PO Box 44330 Olympia, WA 98504-4330 1-888-66-SHARP www.lni.wa.gov/sharp/derm
Hand in Health Care Workers Safety and Health Assessment and Research for Prevention (SHARP) Program Washington Department of Labor and Industries PO Box 44330 Olympia, WA 98504-4330 1-888-66-SHARP www.lni.wa.gov/sharp/derm
EBMT Education Day for Nurses and AHPs April 2012 Skin care: not every rash is GVHD
 EBMT Education Day for Nurses and AHPs April 2012 Skin care: not every rash is GVHD Eileen Parry Consultant Dermatologist Tameside Hospital Foundation Trust Overview How to assess a patient with a rash
EBMT Education Day for Nurses and AHPs April 2012 Skin care: not every rash is GVHD Eileen Parry Consultant Dermatologist Tameside Hospital Foundation Trust Overview How to assess a patient with a rash
Tungsten Heavy Alloy. Densimet, Mallory Metal, Densalloy, High Density Tungsten Alloy
 Safety Data Sheet SECTION 1: IDENTIFICATION Chemical Name: Synonyms: CAS #: Tungsten Heavy Alloy Densimet, Mallory Metal, Densalloy, High Density Tungsten Alloy 7440-33-7 (Tungsten), 7440-02-0 (Nickel),
Safety Data Sheet SECTION 1: IDENTIFICATION Chemical Name: Synonyms: CAS #: Tungsten Heavy Alloy Densimet, Mallory Metal, Densalloy, High Density Tungsten Alloy 7440-33-7 (Tungsten), 7440-02-0 (Nickel),
I B2.4. Design of the patient information leaflet for VariQuin
 (English translation of official Dutch version) I B2.4. Design of the patient information leaflet for VariQuin Information for the Patient: Read this package leaflet carefully when you have some time to
(English translation of official Dutch version) I B2.4. Design of the patient information leaflet for VariQuin Information for the Patient: Read this package leaflet carefully when you have some time to
HYDROCORTISONE 10 MG TABLETS
 HYDROCORTISONE 10 MG TABLETS (Hydrocortisone) PL 20072/0238 UKPAR TABLE OF CONTENTS Lay Summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 12 Steps taken after authorisation summary
HYDROCORTISONE 10 MG TABLETS (Hydrocortisone) PL 20072/0238 UKPAR TABLE OF CONTENTS Lay Summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 12 Steps taken after authorisation summary
Hydrozole Cream Hydrocortisone (microfine) 1% w/w and Clotrimazole 1% w/w
 CONSUMER MEDICINE INFORMATION What is in this leaflet? This leaflet answers some common questions about Hydrozole It does not contain all the available information. It does not take the place of talking
CONSUMER MEDICINE INFORMATION What is in this leaflet? This leaflet answers some common questions about Hydrozole It does not contain all the available information. It does not take the place of talking
Presenting Genital Symptoms and Physical Signs of Vaginal Atrophy
 Sexual Health in the Menopause Presenting Genital Symptoms and Physical Signs of Vaginal Atrophy Symptoms Dryness Itching Burning Dyspareunia Signs on Physical Exam Pale, smooth, or shiny vaginal epithelium
Sexual Health in the Menopause Presenting Genital Symptoms and Physical Signs of Vaginal Atrophy Symptoms Dryness Itching Burning Dyspareunia Signs on Physical Exam Pale, smooth, or shiny vaginal epithelium
PACKAGE LEAFLET: INFORMATION FOR THE USER. Alvesco 40, 80 and 160 mcg Inhaler Ciclesonide
 PACKAGE LEAFLET PACKAGE LEAFLET: INFORMATION FOR THE USER Alvesco 40, 80 and 160 mcg Inhaler Ciclesonide Read all of this leaflet carefully before you start using this medicine because it contains important
PACKAGE LEAFLET PACKAGE LEAFLET: INFORMATION FOR THE USER Alvesco 40, 80 and 160 mcg Inhaler Ciclesonide Read all of this leaflet carefully before you start using this medicine because it contains important
Probiotics for the Treatment of Adult Gastrointestinal Disorders
 Probiotics for the Treatment of Adult Gastrointestinal Disorders Darren M. Brenner, M.D. Division of Gastroenterology Northwestern University, Feinberg School of Medicine Chicago, Illinois What are Probiotics?
Probiotics for the Treatment of Adult Gastrointestinal Disorders Darren M. Brenner, M.D. Division of Gastroenterology Northwestern University, Feinberg School of Medicine Chicago, Illinois What are Probiotics?
SHINGLES (Herpes zoster infection)
 SHINGLES (Herpes zoster infection) What are the aims of this leaflet? This leaflet has been written to help you understand more about shingles. It will tell you what it is, what causes it, what can be
SHINGLES (Herpes zoster infection) What are the aims of this leaflet? This leaflet has been written to help you understand more about shingles. It will tell you what it is, what causes it, what can be
NIZORAL (KETOCONAZOLE) 2% SHAMPOO DESCRIPTION
 NIZORAL (KETOCONAZOLE) 2% SHAMPOO DESCRIPTION NIZORAL (ketoconazole) 2% Shampoo is a red-orange liquid for topical application, containing the broad spectrum synthetic antifungal agent ketoconazole in
NIZORAL (KETOCONAZOLE) 2% SHAMPOO DESCRIPTION NIZORAL (ketoconazole) 2% Shampoo is a red-orange liquid for topical application, containing the broad spectrum synthetic antifungal agent ketoconazole in
MATERIAL SAFETY DATA SHEET
 Page 1 of 5 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND THE COMPANY/UNDERTAKING Pfizer Inc Pfizer Pharmaceuticals Group 235 East 42nd Street New York, New York 10017 1-212-573-2222 Emergency telephone
Page 1 of 5 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND THE COMPANY/UNDERTAKING Pfizer Inc Pfizer Pharmaceuticals Group 235 East 42nd Street New York, New York 10017 1-212-573-2222 Emergency telephone
PHYSICIAN OFFICE BILLING INFORMATION SHEET FOR IMLYGIC (talimogene laherparepvec)
 PHYSICIAN OFFICE BILLING INFORMATION SHEET FOR IMLYGIC (talimogene laherparepvec) INDICATION IMLYGIC is a genetically modified oncolytic viral therapy indicated for the local treatment of unresectable
PHYSICIAN OFFICE BILLING INFORMATION SHEET FOR IMLYGIC (talimogene laherparepvec) INDICATION IMLYGIC is a genetically modified oncolytic viral therapy indicated for the local treatment of unresectable
Intravenous Methyl Prednisolone in Multiple Sclerosis
 Intravenous Methyl Prednisolone in Multiple Sclerosis Exceptional healthcare, personally delivered Relapse management in multiple sclerosis Relapses in multiple sclerosis (MS) are common and caused by
Intravenous Methyl Prednisolone in Multiple Sclerosis Exceptional healthcare, personally delivered Relapse management in multiple sclerosis Relapses in multiple sclerosis (MS) are common and caused by
Material Safety Data Sheet
 Material Safety Data Sheet Silver sulfate MSDS 3 He a lt h Fire Re a c t iv it y Pe rs o n a l Pro t e c t io n 3 E Section 1: Chemical Product and Company Identification Product Name: Silver sulfate Catalog
Material Safety Data Sheet Silver sulfate MSDS 3 He a lt h Fire Re a c t iv it y Pe rs o n a l Pro t e c t io n 3 E Section 1: Chemical Product and Company Identification Product Name: Silver sulfate Catalog
NEHSNORTH EASTERN HEALTH SPECIALISTS
 COSMETIC DERMATOLOGY NEHSNORTH EASTERN HEALTH SPECIALISTS nehs.com.au CONSENT FORM ACNE Treatment I, DOB:, of authorize of North Eastern Health Specialist to perform hair removal with the BBL / Nd-Yag
COSMETIC DERMATOLOGY NEHSNORTH EASTERN HEALTH SPECIALISTS nehs.com.au CONSENT FORM ACNE Treatment I, DOB:, of authorize of North Eastern Health Specialist to perform hair removal with the BBL / Nd-Yag
Informed Consent For Laser Hair Removal
 Informed Consent For Laser Hair Removal INSTRUCTIONS This informed-consent document has been prepared to help inform you about laser procedures, its risks, as well as alternative treatment(s). It is important
Informed Consent For Laser Hair Removal INSTRUCTIONS This informed-consent document has been prepared to help inform you about laser procedures, its risks, as well as alternative treatment(s). It is important
READ THIS FOR SAFE AND EFFECTIVE USE OF YOUR MEDICINE PATIENT MEDICATION INFORMATION. [new-ka la]
![READ THIS FOR SAFE AND EFFECTIVE USE OF YOUR MEDICINE PATIENT MEDICATION INFORMATION. [new-ka la] READ THIS FOR SAFE AND EFFECTIVE USE OF YOUR MEDICINE PATIENT MEDICATION INFORMATION. [new-ka la]](/thumbs/35/17235501.jpg) READ THIS FOR SAFE AND EFFECTIVE USE OF YOUR MEDICINE PATIENT MEDICATION INFORMATION Pr NUCALA [new-ka la] mepolizumab lyophilized powder for subcutaneous injection Read this carefully before you start
READ THIS FOR SAFE AND EFFECTIVE USE OF YOUR MEDICINE PATIENT MEDICATION INFORMATION Pr NUCALA [new-ka la] mepolizumab lyophilized powder for subcutaneous injection Read this carefully before you start
Beaumont Hospital Department of Nephrology and Renal Nursing. Guideline for administering Ferinject
 Beaumont Hospital Department of Nephrology and Renal Nursing Guideline Name: Guideline for administering Ferinject Guideline Number: 18 Guideline Version: a Developed By: Louise Kelly CNM 1 Renal Day Care
Beaumont Hospital Department of Nephrology and Renal Nursing Guideline Name: Guideline for administering Ferinject Guideline Number: 18 Guideline Version: a Developed By: Louise Kelly CNM 1 Renal Day Care
Short Synacthen Test for the Investigation of Adrenal Insufficiency
 Pathology at the Royal Derby Hospital Short Synacthen Test Standard Clinical Guidelines Chemical Pathology Department Valid Until 31 st March 2015 Document Code: CHISCG1 Short Synacthen Test for the Investigation
Pathology at the Royal Derby Hospital Short Synacthen Test Standard Clinical Guidelines Chemical Pathology Department Valid Until 31 st March 2015 Document Code: CHISCG1 Short Synacthen Test for the Investigation
Nursing Process Focus: Patients Receiving LINDANE (Kwell)
 Nursing Process Focus: Patients Receiving LINDANE (Kwell) ASSESSMENT Prior to administration: Obtain complete health history including allergies, drug history, and possible drug interactions. Assess vital
Nursing Process Focus: Patients Receiving LINDANE (Kwell) ASSESSMENT Prior to administration: Obtain complete health history including allergies, drug history, and possible drug interactions. Assess vital
CONTRAINDICATIONS. None (4).
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use Taclonex Ointment safely and effectively. See Full Prescribing Information for Taclonex Ointment.
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use Taclonex Ointment safely and effectively. See Full Prescribing Information for Taclonex Ointment.
Teriflunomide is the active metabolite of Leflunomide, a drug employed since 1994 for the treatment of rheumatoid arthritis (Baselt, 2011).
 Page 1 of 10 ANALYTE NAME AND STRUCTURE TERIFLUNOMIDE Teriflunomide TRADE NAME Aubagio CATEGORY Antimetabolite TEST CODE PURPOSE Therapeutic Drug Monitoring GENERAL RELEVANCY BACKGROUND sclerosis. The
Page 1 of 10 ANALYTE NAME AND STRUCTURE TERIFLUNOMIDE Teriflunomide TRADE NAME Aubagio CATEGORY Antimetabolite TEST CODE PURPOSE Therapeutic Drug Monitoring GENERAL RELEVANCY BACKGROUND sclerosis. The
An introduction to modern MS treatments
 BETAFERON is a Prescription Medicine. Use strictly as directed. Consult your pharmacist or other health professional in case of side effects. BETAFERON is reimbursed for some patients. See your neurologist
BETAFERON is a Prescription Medicine. Use strictly as directed. Consult your pharmacist or other health professional in case of side effects. BETAFERON is reimbursed for some patients. See your neurologist
Influence of ph Most local anesthetics are weak bases.
 Local anesthetics The agent must depress nerve conduction. The agent must have both lipophilic and hydrophilic properties to be effective by parenteral injection. Structure-activity relationships The typical
Local anesthetics The agent must depress nerve conduction. The agent must have both lipophilic and hydrophilic properties to be effective by parenteral injection. Structure-activity relationships The typical
Treatment of diseases affecting the kidney using steroids
 Treatment of diseases affecting the kidney using steroids Hope Building Renal 0161 206 5223 All Rights Reserved 2015. Document for issue as handout. This information has been written by the medical and
Treatment of diseases affecting the kidney using steroids Hope Building Renal 0161 206 5223 All Rights Reserved 2015. Document for issue as handout. This information has been written by the medical and
STANDARDS AND GUIDELINES TITLE: CIRCULATION DATE: March June 2013 REVISED: June 2013 APPROVAL DATE: July 29, 2013
 College of Homeopaths of Ontario 163 Queen Street East, 4 th Floor, Toronto, Ontario, M5A 1S1 TEL 416-862-4780 OR 1-844-862-4780 FAX 416-874-4077 www.collegeofhomeopaths.on.ca STANDARDS AND GUIDELINES
College of Homeopaths of Ontario 163 Queen Street East, 4 th Floor, Toronto, Ontario, M5A 1S1 TEL 416-862-4780 OR 1-844-862-4780 FAX 416-874-4077 www.collegeofhomeopaths.on.ca STANDARDS AND GUIDELINES
MATERIAL SAFETY DATA SHEET
 MATERIAL SAFETY DATA SHEET 1. Product and Supplier Identification Product Name: Product: Chemical Name: Product Use: Glass Fiber Woven Cloth Orca Composites Woven Cloth (All) E-Glass FRP Manufacturing
MATERIAL SAFETY DATA SHEET 1. Product and Supplier Identification Product Name: Product: Chemical Name: Product Use: Glass Fiber Woven Cloth Orca Composites Woven Cloth (All) E-Glass FRP Manufacturing
Section II When you are finished with this section, you will be able to: Define medication (p 2) Describe how medications work (p 3)
 Section II When you are finished with this section, you will be able to: Define medication (p 2) Describe how medications work (p 3) List the different medication effects (p5) List the ways that medications
Section II When you are finished with this section, you will be able to: Define medication (p 2) Describe how medications work (p 3) List the different medication effects (p5) List the ways that medications
FUCIBET LIPID CREAM (FUSIDIC ACID 2%, BETAMETHASONE 0.1%) PL 00043/0218 UKPAR TABLE OF CONTENTS
 FUCIBET LIPID CREAM (FUSIDIC ACID 2%, BETAMETHASONE 0.1%) PL 00043/0218 UKPAR TABLE OF CONTENTS Lay Summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 23 Summary of Product Characteristics
FUCIBET LIPID CREAM (FUSIDIC ACID 2%, BETAMETHASONE 0.1%) PL 00043/0218 UKPAR TABLE OF CONTENTS Lay Summary Page 2 Scientific discussion Page 3 Steps taken for assessment Page 23 Summary of Product Characteristics
P AC K AG E L E AF L E T: INFORMAT I ON FO R THE USER. 500 mg, film-coated tablet Active substance: metformin hydrochloride
 P AC K AG E L E AF L E T: INFORMAT I ON FO R THE USER Siofor 500 500 mg, film-coated tablet Active substance: metformin hydrochloride For use in children above 10 years and adults Read all of this leaflet
P AC K AG E L E AF L E T: INFORMAT I ON FO R THE USER Siofor 500 500 mg, film-coated tablet Active substance: metformin hydrochloride For use in children above 10 years and adults Read all of this leaflet
PRODUCT INFORMATION OINTMENT, CREAM & LOTION
 PRDUCT INFRMATIN ADVANTAN INTMENT, FATTY INTMENT, CREAM & LTIN NAME F THE DRUG The chemical name is 21-acetoxy-11β-hydroxy-6α-methyl-17-propionyloxy-1, 4- pregnadiene-3, 20-dione. The chemical structure
PRDUCT INFRMATIN ADVANTAN INTMENT, FATTY INTMENT, CREAM & LTIN NAME F THE DRUG The chemical name is 21-acetoxy-11β-hydroxy-6α-methyl-17-propionyloxy-1, 4- pregnadiene-3, 20-dione. The chemical structure
COMMITTEE ON HERBAL MEDICINAL PRODUCTS (HMPC) FINAL COMMUNITY HERBAL MONOGRAPH ON HYPERICUM PERFORATUM L., HERBA (WELL-ESTABLISHED MEDICINAL USE)
 European Medicines Agency Evaluation of Medicines for Human Use London, 12 November 2009 Doc. Ref.: EMA/HMPC/101304/2008 COMMITTEE ON HERBAL MEDICINAL PRODUCTS (HMPC) FINAL COMMUNITY HERBAL MONOGRAPH ON
European Medicines Agency Evaluation of Medicines for Human Use London, 12 November 2009 Doc. Ref.: EMA/HMPC/101304/2008 COMMITTEE ON HERBAL MEDICINAL PRODUCTS (HMPC) FINAL COMMUNITY HERBAL MONOGRAPH ON
Guidelines for the Use of Controlled Substances in the Treatment of Pain Adopted by the New Hampshire Medical Society, July 1998
 Guidelines for the Use of Controlled Substances in the Treatment of Pain Adopted by the New Hampshire Medical Society, July 1998 Section I: Preamble The New Hampshire Medical Society believes that principles
Guidelines for the Use of Controlled Substances in the Treatment of Pain Adopted by the New Hampshire Medical Society, July 1998 Section I: Preamble The New Hampshire Medical Society believes that principles
Leader's Resource. Note: Both men and women can have an STD without physical symptoms.
 Leader's Resource Information on Sexually Transmitted Diseases (STDs) Signs and Symptoms of STDs Note: Both men and women can have an STD without physical symptoms. Any of the following can indicate to
Leader's Resource Information on Sexually Transmitted Diseases (STDs) Signs and Symptoms of STDs Note: Both men and women can have an STD without physical symptoms. Any of the following can indicate to
BECONASE AQ (beclomethasone dipropionate, monohydrate) Nasal Spray, 42 mcg
 BECONASE AQ (beclomethasone dipropionate, monohydrate) Nasal Spray, 42 mcg For Intranasal Use Only. PRESCRIBING INFORMATION SHAKE WELL BEFORE USE. DESCRIPTION Beclomethasone dipropionate, monohydrate,
BECONASE AQ (beclomethasone dipropionate, monohydrate) Nasal Spray, 42 mcg For Intranasal Use Only. PRESCRIBING INFORMATION SHAKE WELL BEFORE USE. DESCRIPTION Beclomethasone dipropionate, monohydrate,
MATERIAL SAFETY DATA SHEET
 MATERIAL SAFETY DATA SHEET PRODUCT NAME: Butler Neomycin and Polymyxin B Sulfates and Dexamethasone Ophthalmic AS SOLD BY BUTLER SCHEIN ANIMAL HEALTH A Service of MATERIAL SAFETY DATA SHEET Issued: 09/14/95
MATERIAL SAFETY DATA SHEET PRODUCT NAME: Butler Neomycin and Polymyxin B Sulfates and Dexamethasone Ophthalmic AS SOLD BY BUTLER SCHEIN ANIMAL HEALTH A Service of MATERIAL SAFETY DATA SHEET Issued: 09/14/95
MATERIAL SAFETY DATA SHEET. CAPTURE Carpet Dry Cleaner - Advanced Formula
 Page 1 of 5 MATERIAL SAFETY DATA SHEET CAPTURE Carpet Dry Cleaner - Advanced Formula 1. PRODUCT AND COMPANY IDENTIFICATION Product Identification Product Name: CAPTURE Carpet Dry Cleaner - Advanced Formula
Page 1 of 5 MATERIAL SAFETY DATA SHEET CAPTURE Carpet Dry Cleaner - Advanced Formula 1. PRODUCT AND COMPANY IDENTIFICATION Product Identification Product Name: CAPTURE Carpet Dry Cleaner - Advanced Formula
What are spider veins? What is the best treatment for spider veins?
 Laser Spider/Leg Vein Q & A What are spider veins? Millions of women and men are bothered by unsightly spider veins on their faces and legs. Spider veins or telangiectasias are those small red, blue and
Laser Spider/Leg Vein Q & A What are spider veins? Millions of women and men are bothered by unsightly spider veins on their faces and legs. Spider veins or telangiectasias are those small red, blue and
Michigan Guidelines for the Use of Controlled Substances for the Treatment of Pain
 Michigan Guidelines for the Use of Controlled Substances for the Treatment of Pain Section I: Preamble The Michigan Boards of Medicine and Osteopathic Medicine & Surgery recognize that principles of quality
Michigan Guidelines for the Use of Controlled Substances for the Treatment of Pain Section I: Preamble The Michigan Boards of Medicine and Osteopathic Medicine & Surgery recognize that principles of quality
ACNE. Nisakorn Saewan, Ph.D.
 ACNE, Ph.D. Contents Definition Cause of acne Type of acne Acne grades Acne treatments Objectives To explain the definition of acne To elucidate cause of acne To identify type and grade of acne To explain
ACNE, Ph.D. Contents Definition Cause of acne Type of acne Acne grades Acne treatments Objectives To explain the definition of acne To elucidate cause of acne To identify type and grade of acne To explain
