Chapter 13 Carbohydrates
|
|
|
- Wendy Lloyd
- 7 years ago
- Views:
Transcription
1 Chapter 13 Carbohydrates 1
2 Carbohydrates Carbohydrates are a major source of energy from our diet made from elements carbon, hydrogen, and oxygen also called saccharides, which means sugars Carbohydrates are produced by photosynthesis in plants such as glucose are synthesized in plants from C 2, 2, and energy from the sun are oxidized in living cells to produce C 2, 2, and energy 2
3 Types of Carbohydrates The types of carbohydrates are monosaccharides, the simplest carbohydrates disaccharides, which consist of two monosaccharides polysaccharides, which contain many monosaccharides 3
4 Monosaccharides Monosaccharides consist of three to eight carbon chains with one carbon in a carbonyl group are further classified as an aldose, which contains an aldehyde group, or a ketose, which contains a ketone carbonyl group have several hydroxyl groups 4
5 Types of Monosaccharides Monosaccharides are also classified by the number of carbon atoms present. triose (3 C atoms) tetrose (4 C atoms) pentose (5 C atoms) hexose (6 C atoms) An aldopentose indicates a five-carbon saccharide with an aldehyde group. 5
6 Aldoses Aldoses are monosaccharides with an aldehyde group many hydroxyl ( ) groups C C C aldose C 2 Erythose, an aldotetrose (4 carbons, aldehyde group) 6
7 Ketoses Ketoses are monosaccharides with a ketone group many hydroxyl ( ) groups Fructose, a ketohexose (6 carbons, ketone group) C 2 C= C C C C 2 ketose
8 Fischer Projections A Fischer projection is used to represent carbohydrates places the most oxidized group at the top shows chiral carbons as the intersection of vertical and horizontal lines 8
9 D and L Notations In a Fischer projection, the group on the chiral carbon farthest from the carbonyl group determines an L or D isomer. left is assigned the letter L for the L form right is assigned the letter D for the D form 9
10 Examples of D and L Isomers of Monosaccharides 10
11 Learning Check Identify each as the D or L isomer. A. B. C. C C 2 C C 2 -Ribose -Threose -Fructose C 2 C 2 11
12 Solution Identify each as the D or L isomer. A. B. C. C C 2 C C 2 C 2 C 2 L-Ribose L-Threose D-Fructose 12
13 D-glucose is Glucose and Fructose found in fruits, corn syrup, and honey an aldohexose with the formula C known as dextrose and blood sugar in the body D-fructose is obtained from lactose an aldohexose with the formula C important in cell membranes of the brain and nerve cells The sweet taste of honey comes from the monosaccharides D-glucose and D-fructose.
14 Blood Glucose Level In the body, glucose has a normal blood level of mg/dl a glucose tolerance test measures blood glucose for several hours after ingesting glucose
15 D-Galactose D-Galactose is an aldohexose, C is not found free in nature is obtained from lactose, a disaccharide has a similar structure to glucose except for the on carbon 4 In a condition called galactosemia, an enzyme needed to convert galactose to glucose is missing. C C 2 D-Galactose 15
16 Cyclic Structures Cyclic structures are the prevalent form of monosaccharides with five or six carbon atoms form when the hydroxyl group on carbon 5 reacts with the aldehyde group or ketone group
17 Guide for Drawing ayworth Structures 17
18 Drawing the Cyclic Structure for Glucose Step 1 Number the carbon chain and turn clockwise to form a linear open chain. 18
19 Drawing the Cyclic Structure for Glucose Step 2 Fold clockwise to make a hexagon. Bond the carbon 5 to carbon 1. Place the carbon 6 group above the ring. Write the groups on carbon 2 and 4 below the ring. Write the group on carbon 3 above the ring. Write a new on carbon 1. 19
20 Drawing the Cyclic Structure for Glucose Step 3 Write the new on carbon 1 down for the form and up for the form. 20
21 Mutarotation of -D-Glucose and β-d-glucose in Solution When placed in solution, cyclic structures open and close -D-glucose converts to β-d-glucose and vice versa at any time, only a small amount of open chain forms C 2 C 2 C C 2 -D-Glucose D-Glucose (open) β-d-glucose (36%) (trace) (64%)
22 Cyclic Structure of Fructose Fructose is a ketohexose forms a cyclic structure reacts the on carbon 5 with the C= on carbon 2 C 2 C C 2 C 2 C 2 C C C 2 C C 2 D-Fructose α-d-fructose -D-Fructose 22
23 Learning Check Write the cyclic forms of D-galactose. C C 2 D-Galactose 23
24 Solution Step 1 Number the carbon chain and turn clockwise to form a linear open chain C C 2 D-Galactose 2 C C
25 Solution Step 2 Fold clockwise to make a hexagon. Bond the carbon 5 to carbon 1 and change the = to on carbon 1. Place the carbon 6 group above the ring. Write the groups on carbon 2 and carbon 4 below the ring and the group on carbon 3 above the ring. 4 6 C C 1 25
26 Solution Step 3 Write the new on carbon 1 down for the form or up for the form. 2 C 2 C -D-Galactose -D-Galactose
27 Reducing Sugars Reducing sugars are monosaccharides that can be oxidized to give a carboxylic acid contain an aldehyde group in the open chain form undergo reaction in the Benedict s test include the monosaccharides glucose, galactose, and fructose 27
28 xidation of D-Glucose 28
29 Conversion of Fructose to Glucose Fructose, a ketohexose, is also a reducing sugar. In Benedict s solution, which is basic, a rearrangement occurs between the ketone group on carbon 2 and the hydroxyl group on carbon 1. This produces an aldehyde group with an adjacent hydroxyl, thereby converting fructose to glucose. 29
30 Reduction of Monosaccharides The reduction of monosaccharides produces sugar alcohols, or alditols involves the reduction of the carbonyl group to an alcohol such as D-glucose gives D-glucitol, also called sorbitol 30
31 Learning Check Write the products of the oxidation and reduction of D-mannose. C C 2 D-Mannose 31
32 Solution Write the products of the oxidation and reduction of D-mannose. C 2 Reduction C 2 C C 2 xidation C C 2 D-Mannitol D-Mannose D-Mannonic acid 32
33 Important Disaccharides A disaccharide consists of two monosaccharides. Monosaccharides glucose + glucose glucose + galactose glucose + fructose Disaccharide maltose + 2 lactose + 2 sucrose
34 Maltose Maltose is a disaccharide also known as malt sugar composed of two D-glucose molecules obtained from the hydrolysis of starch used in cereals, candies, and brewing found in both the and β forms Maltose is linked by an -1,4-glycosidic bond formed from the on carbon 1 of the first glucose and on carbon 4 of the second glucose. 34
35 Lactose Lactose is found in milk and milk products is a disaccharide of β-d-galactose and α- or β-d-glucose contains a β-1,4-glycosidic bond 35
36 Sucrose Sucrose, or table sugar, is obtained from sugar cane and sugar beets consists of α-d-glucose and β-d-fructose has an α,β-1,2- glycosidic bond 36
37 Learning Check For the following disaccharide, list the two monosaccharide units and the type of glycosidic bond present. 37
38 Solution For the following disaccharide, provide the reaction for two monosaccharide units and the type of glycosidic bond present. galactose + glucose melibiose + 2 The sugar melibiose is formed from two monosaccharides, galactose and glucose, which are joined by a -1,6-glycosidic bond. 38
39 Sweetness of Sweeteners Sugars and artificial sweeteners differ in sweetness are compared to sucrose (table sugar), which is assigned a value of
40 Artificial Sweeteners - Sucralose Sucrolose is made from sucrose by replacing some of the hydroxyl groups with chlorine atoms. Sucralose is the sweetening agent in Splenda.
41 Artificial Sweeteners - Aspartame Aspartame is marketed as Nutra-Sweet a noncarbohydrate sweetener made from aspartic acid and a methyl ester of the amino acid phenylalanine 41
42 Polysaccharides Polysaccharides are polymers of D-glucose include amylose and amylopectin, starches made of α-d-glucose include glycogen (animal starch in muscle), which is made of α-d-glucose include cellulose (plants and wood), which is made of β-d-glucose C 2 α-d-glucose 42
43 Polysaccharide: Amylose Amylose is a straight-chain polysaccharide made of glucose molecules linked by -1,4 glycosidic bonds
44 Polysaccharide: Amylopectin Amylopectin, found in plants, is a polymer of α-d-glucose molecules is a branched-chain polysaccharide has α-1,4-glycosidic bonds between the glucose units has α-1,6 bonds to branches 44
45 Glycogen Glycogen, or animal starch, is the polysaccharide that stores α-d-glucose in muscle is similar to amylopectin, but is more highly branched the glucose units are joined by α-1,4-glycosidic bonds, and branches occurring about every glucose units are attached by α-1,6-glycosidic bonds 45
46 Cellulose Cellulose, the major structural unit of wood, is a polysaccharide of glucose units in unbranched chains has β-1,4-glycosidic bonds cannot be digested by humans because humans cannot break down β-1,4-glycosidic bonds 46
47 47 Concept Map for Carbohydrates
3) How many monosaccharides are connected to each other in a disaccharide? A) 1 B) 2 C) 3 D) 4
 General, Organic, and Biochemistry, 2e (Frost) HOMEWORK Chapter 6 Carbohydrates Life s Sweet Molecules 6.1 Multiple-Choice 1) Which of the following is a polysaccharide? Glucose Sucrose C) Starch D) Maltose
General, Organic, and Biochemistry, 2e (Frost) HOMEWORK Chapter 6 Carbohydrates Life s Sweet Molecules 6.1 Multiple-Choice 1) Which of the following is a polysaccharide? Glucose Sucrose C) Starch D) Maltose
CHEM 121. Chapter 18. Name: Date: 1. Which of the following compounds is both an aldose and a hexose? A) Page 1
 CEM 121. Chapter 18. Name: Date: 1. Which of the following compounds is both an aldose and a hexose? A) B) C) D) Page 1 2. Which of the following structures is that of an L-monosaccharide? A) B) C) D)
CEM 121. Chapter 18. Name: Date: 1. Which of the following compounds is both an aldose and a hexose? A) B) C) D) Page 1 2. Which of the following structures is that of an L-monosaccharide? A) B) C) D)
I. Chapter 5 Summary. II. Nucleotides & Nucleic Acids. III. Lipids
 I. Chapter 5 Summary A. Simple Sugars (CH 2 O) n : 1. One C contains a carbonyl (C=O) rest contain - 2. Classification by functional group: aldoses & ketoses 3. Classification by number of C's: trioses,
I. Chapter 5 Summary A. Simple Sugars (CH 2 O) n : 1. One C contains a carbonyl (C=O) rest contain - 2. Classification by functional group: aldoses & ketoses 3. Classification by number of C's: trioses,
Chapter 3 Molecules of Cells
 Bio 100 Molecules of cells 1 Chapter 3 Molecules of Cells Compounds containing carbon are called organic compounds Molecules such as methane that are only composed of carbon and hydrogen are called hydrocarbons
Bio 100 Molecules of cells 1 Chapter 3 Molecules of Cells Compounds containing carbon are called organic compounds Molecules such as methane that are only composed of carbon and hydrogen are called hydrocarbons
Solid Phase Peptide Synthesis
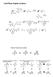 Solid Phase Peptide Synthesis Fischer Projections revisited C C C Remember that group rotations are always allowed C C 80 rotations of entire molecule are allowed C 80 C 90 rotations of entire molecule
Solid Phase Peptide Synthesis Fischer Projections revisited C C C Remember that group rotations are always allowed C C 80 rotations of entire molecule are allowed C 80 C 90 rotations of entire molecule
CHEMISTRY AND BIOLOGICAL ROLE OF CARBOHYDRATES IN THE BODY-1
 CHEMISTRY AND BIOLOGICAL ROLE OF CARBOHYDRATES IN THE BODY-1 Chiral centers: Asymmetric carbons, i.e carbon atom with four different substituents Enantiomers : Mirror images Stereoisomers MONOSACCHARIDE
CHEMISTRY AND BIOLOGICAL ROLE OF CARBOHYDRATES IN THE BODY-1 Chiral centers: Asymmetric carbons, i.e carbon atom with four different substituents Enantiomers : Mirror images Stereoisomers MONOSACCHARIDE
The Chemistry of Carbohydrates
 The Chemistry of Carbohydrates Experiment #5 Objective: To determine the carbohydrate class of an unknown by carrying out a series of chemical reactions with the unknown and known compounds in each class
The Chemistry of Carbohydrates Experiment #5 Objective: To determine the carbohydrate class of an unknown by carrying out a series of chemical reactions with the unknown and known compounds in each class
Elements in Biological Molecules
 Chapter 3: Biological Molecules 1. Carbohydrates 2. Lipids 3. Proteins 4. Nucleic Acids Elements in Biological Molecules Biological macromolecules are made almost entirely of just 6 elements: Carbon (C)
Chapter 3: Biological Molecules 1. Carbohydrates 2. Lipids 3. Proteins 4. Nucleic Acids Elements in Biological Molecules Biological macromolecules are made almost entirely of just 6 elements: Carbon (C)
Biochemistry of Cells
 Biochemistry of Cells 1 Carbon-based Molecules Although a cell is mostly water, the rest of the cell consists mostly of carbon-based molecules Organic chemistry is the study of carbon compounds Carbon
Biochemistry of Cells 1 Carbon-based Molecules Although a cell is mostly water, the rest of the cell consists mostly of carbon-based molecules Organic chemistry is the study of carbon compounds Carbon
The Structure and Function of Macromolecules: Carbohydrates, Lipids & Phospholipids
 The Structure and Function of Macromolecules: Carbohydrates, Lipids & Phospholipids The FOUR Classes of Large Biomolecules All living things are made up of four classes of large biological molecules: Carbohydrates
The Structure and Function of Macromolecules: Carbohydrates, Lipids & Phospholipids The FOUR Classes of Large Biomolecules All living things are made up of four classes of large biological molecules: Carbohydrates
Chapter 5: The Structure and Function of Large Biological Molecules
 Name Period Concept 5.1 Macromolecules are polymers, built from monomers 1. The large molecules of all living things fall into just four main classes. Name them. 2. Circle the three classes that are called
Name Period Concept 5.1 Macromolecules are polymers, built from monomers 1. The large molecules of all living things fall into just four main classes. Name them. 2. Circle the three classes that are called
Disaccharides consist of two monosaccharide monomers covalently linked by a glycosidic bond. They function in sugar transport.
 1. The fundamental life processes of plants and animals depend on a variety of chemical reactions that occur in specialized areas of the organism s cells. As a basis for understanding this concept: 1.
1. The fundamental life processes of plants and animals depend on a variety of chemical reactions that occur in specialized areas of the organism s cells. As a basis for understanding this concept: 1.
BIOLOGICAL MOLECULES OF LIFE
 BIOLOGICAL MOLECULES OF LIFE C A R B O H Y D R A T E S, L I P I D S, P R O T E I N S, A N D N U C L E I C A C I D S The Academic Support Center @ Daytona State College (Science 115, Page 1 of 29) Carbon
BIOLOGICAL MOLECULES OF LIFE C A R B O H Y D R A T E S, L I P I D S, P R O T E I N S, A N D N U C L E I C A C I D S The Academic Support Center @ Daytona State College (Science 115, Page 1 of 29) Carbon
Chapter 3: Biological Molecules. 1. Carbohydrates 2. Lipids 3. Proteins 4. Nucleic Acids
 Chapter 3: Biological Molecules 1. Carbohydrates 2. Lipids 3. Proteins 4. Nucleic Acids Elements in Biological Molecules Biological macromolecules are made almost entirely of just 6 elements: Carbon (C)
Chapter 3: Biological Molecules 1. Carbohydrates 2. Lipids 3. Proteins 4. Nucleic Acids Elements in Biological Molecules Biological macromolecules are made almost entirely of just 6 elements: Carbon (C)
Chapter 5. The Structure and Function of Macromolecule s
 Chapter 5 The Structure and Function of Macromolecule s Most Macromolecules are polymers: Polymer: (poly: many; mer: part) Large molecules consisting of many identical or similar subunits connected together.
Chapter 5 The Structure and Function of Macromolecule s Most Macromolecules are polymers: Polymer: (poly: many; mer: part) Large molecules consisting of many identical or similar subunits connected together.
Macromolecules 1 Carbohydrates, Lipids & Nucleic Acids
 VEA Bringing Learning to Life Program Support Notes Macromolecules 1 Carbohydrates, Lipids & Nucleic Acids Grades 10 - College 25mins Teacher Notes by Sue Wright, B. Sc., Dip. Ed. Produced by VEA Pty Ltd
VEA Bringing Learning to Life Program Support Notes Macromolecules 1 Carbohydrates, Lipids & Nucleic Acids Grades 10 - College 25mins Teacher Notes by Sue Wright, B. Sc., Dip. Ed. Produced by VEA Pty Ltd
Sugars, Starches, and Fibers Are All Carbohydrates
 Sugars, Starches, and Fibers Are All Carbohydrates What are carbohydrates? Today's food advertisements call them carbs, but they are not all the same. They are a group of compounds that have some similarities
Sugars, Starches, and Fibers Are All Carbohydrates What are carbohydrates? Today's food advertisements call them carbs, but they are not all the same. They are a group of compounds that have some similarities
A disaccharide is formed when a dehydration reaction joins two monosaccharides. This covalent bond is called a glycosidic linkage.
 CH 5 Structure & Function of Large Molecules: Macromolecules Molecules of Life All living things are made up of four classes of large biological molecules: carbohydrates, lipids, proteins, and nucleic
CH 5 Structure & Function of Large Molecules: Macromolecules Molecules of Life All living things are made up of four classes of large biological molecules: carbohydrates, lipids, proteins, and nucleic
Catalysis by Enzymes. Enzyme A protein that acts as a catalyst for a biochemical reaction.
 Catalysis by Enzymes Enzyme A protein that acts as a catalyst for a biochemical reaction. Enzymatic Reaction Specificity Enzyme Cofactors Many enzymes are conjugated proteins that require nonprotein portions
Catalysis by Enzymes Enzyme A protein that acts as a catalyst for a biochemical reaction. Enzymatic Reaction Specificity Enzyme Cofactors Many enzymes are conjugated proteins that require nonprotein portions
1. The diagram below represents a biological process
 1. The diagram below represents a biological process 5. The chart below indicates the elements contained in four different molecules and the number of atoms of each element in those molecules. Which set
1. The diagram below represents a biological process 5. The chart below indicates the elements contained in four different molecules and the number of atoms of each element in those molecules. Which set
Chapter 5: The Structure and Function of Large Biological Molecules
 Name Period Chapter 5: The Structure and Function of Large Biological Molecules Concept 5.1 Macromolecules are polymers, built from monomers 1. The large molecules of all living things fall into just four
Name Period Chapter 5: The Structure and Function of Large Biological Molecules Concept 5.1 Macromolecules are polymers, built from monomers 1. The large molecules of all living things fall into just four
Biological molecules:
 Biological molecules: All are organic (based on carbon). Monomers vs. polymers: Monomers refer to the subunits that, when polymerized, make up a larger polymer. Monomers may function on their own in some
Biological molecules: All are organic (based on carbon). Monomers vs. polymers: Monomers refer to the subunits that, when polymerized, make up a larger polymer. Monomers may function on their own in some
Carbohydrates, proteins and lipids
 Carbohydrates, proteins and lipids Chapter 3 MACROMOLECULES Macromolecules: polymers with molecular weights >1,000 Functional groups THE FOUR MACROMOLECULES IN LIFE Molecules in living organisms: proteins,
Carbohydrates, proteins and lipids Chapter 3 MACROMOLECULES Macromolecules: polymers with molecular weights >1,000 Functional groups THE FOUR MACROMOLECULES IN LIFE Molecules in living organisms: proteins,
The Molecules of Cells
 The Molecules of Cells I. Introduction A. Most of the world s population cannot digest milk-based foods. 1. These people are lactose intolerant because they lack the enzyme lactase. 2. This illustrates
The Molecules of Cells I. Introduction A. Most of the world s population cannot digest milk-based foods. 1. These people are lactose intolerant because they lack the enzyme lactase. 2. This illustrates
How To Understand The Chemistry Of Organic Molecules
 CHAPTER 3 THE CHEMISTRY OF ORGANIC MOLECULES 3.1 Organic Molecules The chemistry of carbon accounts for the diversity of organic molecules found in living things. Carbon has six electrons, four of which
CHAPTER 3 THE CHEMISTRY OF ORGANIC MOLECULES 3.1 Organic Molecules The chemistry of carbon accounts for the diversity of organic molecules found in living things. Carbon has six electrons, four of which
Organic Molecules of Life - Exercise 2
 Organic Molecules of Life - Exercise 2 Objectives -Know the difference between a reducing sugar and a non-reducing sugar. -Distinguish Monosaccharides from Disaccharides and Polysaccharides -Understand
Organic Molecules of Life - Exercise 2 Objectives -Know the difference between a reducing sugar and a non-reducing sugar. -Distinguish Monosaccharides from Disaccharides and Polysaccharides -Understand
Organic Compounds. Essential Questions: What is Organic? What are the 4 major Organic Compounds? How are they made? What are they used for?
 Organic Compounds Essential Questions: What is Organic? What are the 4 major Organic Compounds? How are they made? What are they used for? Aristotle: Francesco Redi: What do we already know? Spontaneous
Organic Compounds Essential Questions: What is Organic? What are the 4 major Organic Compounds? How are they made? What are they used for? Aristotle: Francesco Redi: What do we already know? Spontaneous
4. Which carbohydrate would you find as part of a molecule of RNA? a. Galactose b. Deoxyribose c. Ribose d. Glucose
 1. How is a polymer formed from multiple monomers? a. From the growth of the chain of carbon atoms b. By the removal of an OH group and a hydrogen atom c. By the addition of an OH group and a hydrogen
1. How is a polymer formed from multiple monomers? a. From the growth of the chain of carbon atoms b. By the removal of an OH group and a hydrogen atom c. By the addition of an OH group and a hydrogen
Lecture Overview. Hydrogen Bonds. Special Properties of Water Molecules. Universal Solvent. ph Scale Illustrated. special properties of water
 Lecture Overview special properties of water > water as a solvent > ph molecules of the cell > properties of carbon > carbohydrates > lipids > proteins > nucleic acids Hydrogen Bonds polarity of water
Lecture Overview special properties of water > water as a solvent > ph molecules of the cell > properties of carbon > carbohydrates > lipids > proteins > nucleic acids Hydrogen Bonds polarity of water
Carbon-organic Compounds
 Elements in Cells The living substance of cells is made up of cytoplasm and the structures within it. About 96% of cytoplasm and its included structures are composed of the elements carbon, hydrogen, oxygen,
Elements in Cells The living substance of cells is made up of cytoplasm and the structures within it. About 96% of cytoplasm and its included structures are composed of the elements carbon, hydrogen, oxygen,
Qualitative Testing for Carbohydrates
 R E A 446 modular laboratory program in chemistry program editor:. A. Neidig Qualitative Testing for arbohydrates prepared by James. Schreck, University of Northern olorado, and William M. Loffredo, East
R E A 446 modular laboratory program in chemistry program editor:. A. Neidig Qualitative Testing for arbohydrates prepared by James. Schreck, University of Northern olorado, and William M. Loffredo, East
CHEM 2353 Fundamentals of Organic Chemistry. Carbohydrates. Organic and Biochemistry for Today (4 th ed.) Spencer L. Seager / Michael R.
 hapter 7 Notes EM 2353 Fundamentals of rganic hemistry hapter 7 arbohydrates rganic and Biochemistry for Today (4 th ed.) Spencer L. Seager / Michael R. Slabaugh Mr. Kevin A. Boudreaux Angelo State University
hapter 7 Notes EM 2353 Fundamentals of rganic hemistry hapter 7 arbohydrates rganic and Biochemistry for Today (4 th ed.) Spencer L. Seager / Michael R. Slabaugh Mr. Kevin A. Boudreaux Angelo State University
WATER CHAPTER 3 - BIOCHEMISTRY "THE CHEMISTRY OF LIFE" POLARITY HYDROGEN BONDING
 CHAPTER 3 - BIOCHEMISTRY "THE CHEMISTRY OF LIFE" WATER Compare the body of the jellyfish with our own bodies. The jellyfish will die if it is removed from its water environment, yet we can live in the
CHAPTER 3 - BIOCHEMISTRY "THE CHEMISTRY OF LIFE" WATER Compare the body of the jellyfish with our own bodies. The jellyfish will die if it is removed from its water environment, yet we can live in the
Recognizing Organic Molecules: Carbohydrates, Lipids and Proteins
 Recognizing Organic Molecules: Carbohydrates, Lipids and Proteins Oct 15 8:05 PM What is an Organic Molecule? An Organic Molecule is a molecule that contains carbon and hydrogen and oxygen Carbon is found
Recognizing Organic Molecules: Carbohydrates, Lipids and Proteins Oct 15 8:05 PM What is an Organic Molecule? An Organic Molecule is a molecule that contains carbon and hydrogen and oxygen Carbon is found
Worksheet 13.1. Chapter 13: Human biochemistry glossary
 Worksheet 13.1 Chapter 13: Human biochemistry glossary α-helix Refers to a secondary structure of a protein where the chain is twisted to form a regular helix, held by hydrogen bonds between peptide bonds
Worksheet 13.1 Chapter 13: Human biochemistry glossary α-helix Refers to a secondary structure of a protein where the chain is twisted to form a regular helix, held by hydrogen bonds between peptide bonds
Name: Hour: Elements & Macromolecules in Organisms
 Name: Hour: Elements & Macromolecules in Organisms Most common elements in living things are carbon, hydrogen, nitrogen, and oxygen. These four elements constitute about 95% of your body weight. All compounds
Name: Hour: Elements & Macromolecules in Organisms Most common elements in living things are carbon, hydrogen, nitrogen, and oxygen. These four elements constitute about 95% of your body weight. All compounds
10.1 The function of Digestion pg. 402
 10.1 The function of Digestion pg. 402 Macromolecules and Living Systems The body is made up of more than 60 % water. The water is found in the cells cytoplasm, the interstitial fluid and the blood (5
10.1 The function of Digestion pg. 402 Macromolecules and Living Systems The body is made up of more than 60 % water. The water is found in the cells cytoplasm, the interstitial fluid and the blood (5
Chapter 13 Organic Chemistry
 Chapter 13 Organic Chemistry 13-1. Carbon Bonds 13-2. Alkanes 13-3. Petroleum Products 13-4. Structural Formulas 13-5. Isomers 13-6. Unsaturated Hydrocarbons 13-7. Benzene 13-8. Hydrocarbon Groups 13-9.
Chapter 13 Organic Chemistry 13-1. Carbon Bonds 13-2. Alkanes 13-3. Petroleum Products 13-4. Structural Formulas 13-5. Isomers 13-6. Unsaturated Hydrocarbons 13-7. Benzene 13-8. Hydrocarbon Groups 13-9.
MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question.
 Ch23_PT MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) All of the following statements concerning digestion are correct except A) The major physical
Ch23_PT MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) All of the following statements concerning digestion are correct except A) The major physical
Elements & Macromolecules in Organisms
 Name: Date: Per: Table # Elements & Macromolecules in rganisms Most common elements in living things are carbon, hydrogen, nitrogen, and oxygen. These four elements constitute about 95% of your body weight.
Name: Date: Per: Table # Elements & Macromolecules in rganisms Most common elements in living things are carbon, hydrogen, nitrogen, and oxygen. These four elements constitute about 95% of your body weight.
Water. Definition: A mole (or mol ) Water can IONIZE transiently. NONpolar covalent molecules do not dissolve in water + + + + + + + + + + + + + + + +
 Today s Topics Polar Covalent Bonds ydrogen bonding Properties of water p Water C bonds are Nonpolar Will these molecules dissolve in water? Start Macromolecules Carbohydrates & Lipids Sept 4, 05 Why are
Today s Topics Polar Covalent Bonds ydrogen bonding Properties of water p Water C bonds are Nonpolar Will these molecules dissolve in water? Start Macromolecules Carbohydrates & Lipids Sept 4, 05 Why are
Carbohydrates Lipids Proteins Nucleic Acids
 Carbohydrates Lipids Proteins Nucleic Acids Carbon The element of life! All living things contain the element carbon. Organic means it contains carbon The reason for this is because of carbon s ability
Carbohydrates Lipids Proteins Nucleic Acids Carbon The element of life! All living things contain the element carbon. Organic means it contains carbon The reason for this is because of carbon s ability
lt\ Nutrition a-glncose www.cambridge.org in this web service Cambridge University Press Glucose+ fructose Glucose+ galactose
 P. T. Marshall and G. M. ughes 1 Nutrition 1.1 The basic biochemistry of mammalian metabolites The mammals are heterotrophic, that is to say they derive their essential nutrients, via food chains, from
P. T. Marshall and G. M. ughes 1 Nutrition 1.1 The basic biochemistry of mammalian metabolites The mammals are heterotrophic, that is to say they derive their essential nutrients, via food chains, from
Lab 3 Organic Molecules of Biological Importance
 Name Biology 3 ID Number Lab 3 Organic Molecules of Biological Importance Section 1 - Organic Molecules Section 2 - Functional Groups Section 3 - From Building Blocks to Macromolecules Section 4 - Carbohydrates
Name Biology 3 ID Number Lab 3 Organic Molecules of Biological Importance Section 1 - Organic Molecules Section 2 - Functional Groups Section 3 - From Building Blocks to Macromolecules Section 4 - Carbohydrates
II. Chemistry of Sugars
 II. Chemistry of Sugars Sugars-or saccharides- are the most abundant bio-molecule on the planet. They are important in a number of biological roles. Most obviously you will know them as a major component
II. Chemistry of Sugars Sugars-or saccharides- are the most abundant bio-molecule on the planet. They are important in a number of biological roles. Most obviously you will know them as a major component
Macromolecules in my food!!
 Macromolecules in my food!! Name Notes/Background Information Food is fuel: All living things need to obtain fuel from something. Whether it is self- made through the process of photosynthesis, or by ingesting
Macromolecules in my food!! Name Notes/Background Information Food is fuel: All living things need to obtain fuel from something. Whether it is self- made through the process of photosynthesis, or by ingesting
Conduct A Qualitative Test For Starch, Fat, A Reducing Sugar, A Protein
 Conduct A Qualitative Test For Starch, Fat, A Reducing Sugar, A Protein Biology Leaving Cert Experiments Materials/Equipment Starch solution (1%) Iodine Solution Glucose Solution (1%) 100 C) Benedict s
Conduct A Qualitative Test For Starch, Fat, A Reducing Sugar, A Protein Biology Leaving Cert Experiments Materials/Equipment Starch solution (1%) Iodine Solution Glucose Solution (1%) 100 C) Benedict s
The Molecules of Life - Overview. The Molecules of Life. The Molecules of Life. The Molecules of Life
 The Molecules of Life - Overview The Molecules of Life The Importance of Carbon Organic Polymers / Monomers Functions of Organic Molecules Origin of Organic Molecules The Molecules of Life Water is the
The Molecules of Life - Overview The Molecules of Life The Importance of Carbon Organic Polymers / Monomers Functions of Organic Molecules Origin of Organic Molecules The Molecules of Life Water is the
Cellular Respiration: Practice Questions #1
 Cellular Respiration: Practice Questions #1 1. Which statement best describes one of the events taking place in the chemical reaction? A. Energy is being stored as a result of aerobic respiration. B. Fermentation
Cellular Respiration: Practice Questions #1 1. Which statement best describes one of the events taking place in the chemical reaction? A. Energy is being stored as a result of aerobic respiration. B. Fermentation
BIOMOLECULES. reflect
 reflect A child s building blocks are relatively simple structures. When they come together, however, they can form magnifi cent structures. The elaborate city scene to the right is made of small, simple
reflect A child s building blocks are relatively simple structures. When they come together, however, they can form magnifi cent structures. The elaborate city scene to the right is made of small, simple
Lab 2 Biochemistry. Learning Objectives. Introduction. Lipid Structure and Role in Food. The lab has the following learning objectives.
 1 Lab 2 Biochemistry Learning Objectives The lab has the following learning objectives. Investigate the role of double bonding in fatty acids, through models. Developing a calibration curve for a Benedict
1 Lab 2 Biochemistry Learning Objectives The lab has the following learning objectives. Investigate the role of double bonding in fatty acids, through models. Developing a calibration curve for a Benedict
Nutrients: Carbohydrates, Proteins, and Fats. Chapter 5 Lesson 2
 Nutrients: Carbohydrates, Proteins, and Fats Chapter 5 Lesson 2 Carbohydrates Definition- the starches and sugars found in foods. Carbohydrates are the body s preferred source of energy providing four
Nutrients: Carbohydrates, Proteins, and Fats Chapter 5 Lesson 2 Carbohydrates Definition- the starches and sugars found in foods. Carbohydrates are the body s preferred source of energy providing four
Human Physiology Lab (Biol 236L) Digestive Physiology: Amylase hydrolysis of starch
 Human Physiology Lab (Biol 236L) Digestive Physiology: Amylase hydrolysis of starch Introduction Enzymes are proteins composed of amino acid building blocks. Enzymes catalyze or increase the rate of metabolic
Human Physiology Lab (Biol 236L) Digestive Physiology: Amylase hydrolysis of starch Introduction Enzymes are proteins composed of amino acid building blocks. Enzymes catalyze or increase the rate of metabolic
3120-1 - Page 1. Name:
 Name: 1) Which series is arranged in correct order according to decreasing size of structures? A) DNA, nucleus, chromosome, nucleotide, nitrogenous base B) chromosome, nucleus, nitrogenous base, nucleotide,
Name: 1) Which series is arranged in correct order according to decreasing size of structures? A) DNA, nucleus, chromosome, nucleotide, nitrogenous base B) chromosome, nucleus, nitrogenous base, nucleotide,
Digestive System Functions
 Digestive System Functions A. Gastrointestinal Processes 1. Ingestion: placing food in mouth (voluntary) 2. Propulsion: moving food through GI tract a. Peristalsis: alternating waves of contraction and
Digestive System Functions A. Gastrointestinal Processes 1. Ingestion: placing food in mouth (voluntary) 2. Propulsion: moving food through GI tract a. Peristalsis: alternating waves of contraction and
chapter 5 Carbohydrates
 part two The Energy-Yielding Nutrients and Alcohol chapter 5 Carbohydrates Chapter utline Carbohydrates An Introduction Structures and Functions of Simple Carbohydrates Monosaccharides: Glucose, Fructose,
part two The Energy-Yielding Nutrients and Alcohol chapter 5 Carbohydrates Chapter utline Carbohydrates An Introduction Structures and Functions of Simple Carbohydrates Monosaccharides: Glucose, Fructose,
T F T F in childr T F
 T F T F in childr T F 108 Plant-derived energy nutrients 3 mon. 123 124. 110 Glucose is the preferred source of energy for the brain. What As noted earlier (in Chapter 1), carbohydrates are one of the
T F T F in childr T F 108 Plant-derived energy nutrients 3 mon. 123 124. 110 Glucose is the preferred source of energy for the brain. What As noted earlier (in Chapter 1), carbohydrates are one of the
Alcohols An alcohol contains a hydroxyl group ( OH) attached to a carbon chain. A phenol contains a hydroxyl group ( OH) attached to a benzene ring.
 Chapter : rganic Compounds with xygen Alcohols, Ethers Alcohols An alcohol contains a hydroxyl group ( H) attached to a carbon chain. A phenol contains a hydroxyl group ( H) attached to a benzene ring.
Chapter : rganic Compounds with xygen Alcohols, Ethers Alcohols An alcohol contains a hydroxyl group ( H) attached to a carbon chain. A phenol contains a hydroxyl group ( H) attached to a benzene ring.
Copyright 2010 Pearson Education, Inc. Chapter Fourteen 1
 An alcohol has an OH bonded to an alkyl group; a phenol has an OH bonded directly to an aromatic ring; and an ether has an O bonded to two organic groups. Chapter Fourteen 1 Ethyl alcohol, dimethyl ether,
An alcohol has an OH bonded to an alkyl group; a phenol has an OH bonded directly to an aromatic ring; and an ether has an O bonded to two organic groups. Chapter Fourteen 1 Ethyl alcohol, dimethyl ether,
Chapter 4. Carbohydrates: Bountiful Sources of Energy and Nutrients
 hapter 4 arbohydrates: Bountiful Sources of Energy and Nutrients hapter Objectives After reading this chapter, you will be able to: 1. Describe the difference between simple and complex carbohydrates,
hapter 4 arbohydrates: Bountiful Sources of Energy and Nutrients hapter Objectives After reading this chapter, you will be able to: 1. Describe the difference between simple and complex carbohydrates,
Chemical Basis of Life Module A Anchor 2
 Chemical Basis of Life Module A Anchor 2 Key Concepts: - Water is a polar molecule. Therefore, it is able to form multiple hydrogen bonds, which account for many of its special properties. - Water s polarity
Chemical Basis of Life Module A Anchor 2 Key Concepts: - Water is a polar molecule. Therefore, it is able to form multiple hydrogen bonds, which account for many of its special properties. - Water s polarity
Photo Cell Resp Practice. A. ATP B. oxygen C. DNA D. water. The following equation represents the process of photosynthesis in green plants.
 Name: ate: 1. Which molecule supplies the energy for cellular functions?. TP. oxygen. N. water 2. Photosynthesis The following equation represents the process of photosynthesis in green plants. What happens
Name: ate: 1. Which molecule supplies the energy for cellular functions?. TP. oxygen. N. water 2. Photosynthesis The following equation represents the process of photosynthesis in green plants. What happens
Chapter 14 Glycolysis. Glucose. 2 Pyruvate 2 Lactate (sent to liver to be converted back to glucose) TCA Cycle
 Chapter 14 Glycolysis Requires mitochondria and O 2 Glucose glycolysis anaerobic respiration 2 Pyruvate 2 Lactate (sent to liver to be converted back to glucose) pyruvate dehydrogenase acetyl-coa TCA Cycle
Chapter 14 Glycolysis Requires mitochondria and O 2 Glucose glycolysis anaerobic respiration 2 Pyruvate 2 Lactate (sent to liver to be converted back to glucose) pyruvate dehydrogenase acetyl-coa TCA Cycle
Chapter 2 Chemical Principles
 Chapter 2 Chemical Principles I. Chemistry. [Students should read this section on their own]. a. Chemistry is the study of the interactions between atoms and molecules. b. The atom is the smallest unit
Chapter 2 Chemical Principles I. Chemistry. [Students should read this section on their own]. a. Chemistry is the study of the interactions between atoms and molecules. b. The atom is the smallest unit
Plants: The Ultimate Green Machines Science, Grade Level 7
 DRAFT 1 Lesson Title: Plants: The Ultimate Green Machines Grade Level: 7 Subject Area: Science Setting: Garden, Classroom, Laboratory Instructional Time: 1-2 class periods Plants: The Ultimate Green Machines
DRAFT 1 Lesson Title: Plants: The Ultimate Green Machines Grade Level: 7 Subject Area: Science Setting: Garden, Classroom, Laboratory Instructional Time: 1-2 class periods Plants: The Ultimate Green Machines
Copyright 2010 Pearson Education, Inc. Chapter Twenty Three 1
 23.2 Glucose Metabolism: An Overview When glucose enters a cell from the bloodstream, it is immediately converted to glucose 6- phosphate. Once this phosphate is formed, glucose is trapped within the cell
23.2 Glucose Metabolism: An Overview When glucose enters a cell from the bloodstream, it is immediately converted to glucose 6- phosphate. Once this phosphate is formed, glucose is trapped within the cell
Chapter 5 Classification of Organic Compounds by Solubility
 Chapter 5 Classification of Organic Compounds by Solubility Deductions based upon interpretation of simple solubility tests can be extremely useful in organic structure determination. Both solubility and
Chapter 5 Classification of Organic Compounds by Solubility Deductions based upon interpretation of simple solubility tests can be extremely useful in organic structure determination. Both solubility and
Photosynthesis and Sucrose Production
 Photosynthesis and Sucrose Production 2 Starch and sucrose, key substrates for the development of dental caries, are exclusively synthesized by plants. They are made in plant leaves by a process called
Photosynthesis and Sucrose Production 2 Starch and sucrose, key substrates for the development of dental caries, are exclusively synthesized by plants. They are made in plant leaves by a process called
ASPARTAME The Study of a Peptide Bond
 ASPARTAME The Study of a Peptide Bond INTRODUCTION Aspartame, commonly known as NutraSweet or Equal, is the most popular artificial, low-calorie sweetener available to consumers today. Chemically, aspartame
ASPARTAME The Study of a Peptide Bond INTRODUCTION Aspartame, commonly known as NutraSweet or Equal, is the most popular artificial, low-calorie sweetener available to consumers today. Chemically, aspartame
Cellular Energy. 1. Photosynthesis is carried out by which of the following?
 Cellular Energy 1. Photosynthesis is carried out by which of the following? A. plants, but not animals B. animals, but not plants C. bacteria, but neither animals nor plants D. all living organisms 2.
Cellular Energy 1. Photosynthesis is carried out by which of the following? A. plants, but not animals B. animals, but not plants C. bacteria, but neither animals nor plants D. all living organisms 2.
Bulletin 887B. HPLC Carbohydrate Column Selection Guide
 9 North Harrison Road Bellefonte, PA -00 USA Telephone 00-- -9- Fax 00--0-9-0 email: supelco@sial.com sigma-aldrich.com/supelco HPLC Carbohydrate Column Selection Guide Because carbohydrates exhibit a
9 North Harrison Road Bellefonte, PA -00 USA Telephone 00-- -9- Fax 00--0-9-0 email: supelco@sial.com sigma-aldrich.com/supelco HPLC Carbohydrate Column Selection Guide Because carbohydrates exhibit a
pathway that involves taking in heat from the environment at each step. C.
 Study Island Cell Energy Keystone Review 1. Cells obtain energy by either capturing light energy through photosynthesis or by breaking down carbohydrates through cellular respiration. In both photosynthesis
Study Island Cell Energy Keystone Review 1. Cells obtain energy by either capturing light energy through photosynthesis or by breaking down carbohydrates through cellular respiration. In both photosynthesis
Chapter 2. The Chemistry of Life Worksheets
 Chapter 2 The Chemistry of Life Worksheets (Opening image courtesy of David Iberri, http://en.wikipedia.org/wiki/file:camkii.png, and under the Creative Commons license CC-BY-SA 3.0.) Lesson 2.1: Matter
Chapter 2 The Chemistry of Life Worksheets (Opening image courtesy of David Iberri, http://en.wikipedia.org/wiki/file:camkii.png, and under the Creative Commons license CC-BY-SA 3.0.) Lesson 2.1: Matter
THE HISTORY OF CELL BIOLOGY
 SECTION 4-1 REVIEW THE HISTORY OF CELL BIOLOGY Define the following terms. 1. cell 2. cell theory Write the correct letter in the blank. 1. One early piece of evidence supporting the cell theory was the
SECTION 4-1 REVIEW THE HISTORY OF CELL BIOLOGY Define the following terms. 1. cell 2. cell theory Write the correct letter in the blank. 1. One early piece of evidence supporting the cell theory was the
Chapter 12 Organic Compounds with Oxygen and Sulfur
 Chapter 12 Organic Compounds with Oxygen and Sulfur 1 Alcohols An alcohol contains a hydroxyl group ( OH) that replaces a hydrogen atom in a hydrocarbon. A phenol contains a hydroxyl group ( OH) attached
Chapter 12 Organic Compounds with Oxygen and Sulfur 1 Alcohols An alcohol contains a hydroxyl group ( OH) that replaces a hydrogen atom in a hydrocarbon. A phenol contains a hydroxyl group ( OH) attached
2. Which type of macromolecule contains high-energy bonds and is used for long-term energy storage?
 Energy Transport Study Island 1. During the process of photosynthesis, plants use energy from the Sun to convert carbon dioxide and water into glucose and oxygen. These products are, in turn, used by the
Energy Transport Study Island 1. During the process of photosynthesis, plants use energy from the Sun to convert carbon dioxide and water into glucose and oxygen. These products are, in turn, used by the
Anatomy and Physiology Placement Exam 2 Practice with Answers at End!
 Anatomy and Physiology Placement Exam 2 Practice with Answers at End! General Chemical Principles 1. bonds are characterized by the sharing of electrons between the participating atoms. a. hydrogen b.
Anatomy and Physiology Placement Exam 2 Practice with Answers at End! General Chemical Principles 1. bonds are characterized by the sharing of electrons between the participating atoms. a. hydrogen b.
How To Eat Healthily
 Pierce College Putman/NUTR& 101 Unit 04 Practice Exam: Carbohydrates 1. Which is not a monosaccharide? a. lactose b. glucose c. fructose d. galactose 2. Used for immediate energy in the body: a. carbohydrates
Pierce College Putman/NUTR& 101 Unit 04 Practice Exam: Carbohydrates 1. Which is not a monosaccharide? a. lactose b. glucose c. fructose d. galactose 2. Used for immediate energy in the body: a. carbohydrates
Digestive System Module 7: Chemical Digestion and Absorption: A Closer Look
 OpenStax-CNX module: m49457 1 Digestive System Module 7: Chemical Digestion and Absorption: A Closer Look Donna Browne Based on Chemical Digestion and Absorption: A Closer Look by OpenStax This work is
OpenStax-CNX module: m49457 1 Digestive System Module 7: Chemical Digestion and Absorption: A Closer Look Donna Browne Based on Chemical Digestion and Absorption: A Closer Look by OpenStax This work is
McMush. Testing for the Presence of Biomolecules
 Biology McMush Testing for the Presence of Biomolecules MATERIALS AND RESOURCES EACH GROUP aprons beaker, 250 ml 2 clamps, test tube goggles graduated cylinder, 50 ml paper towels test tube brush test
Biology McMush Testing for the Presence of Biomolecules MATERIALS AND RESOURCES EACH GROUP aprons beaker, 250 ml 2 clamps, test tube goggles graduated cylinder, 50 ml paper towels test tube brush test
What impacts blood glucose levels?
 What you eat and how much you eat has an impact on your blood glucose levels. Your blood glucose level reflects how well your diabetes is controlled. There are many aspects to eating for target BG (Blood
What you eat and how much you eat has an impact on your blood glucose levels. Your blood glucose level reflects how well your diabetes is controlled. There are many aspects to eating for target BG (Blood
Reactions of Fats and Fatty Acids
 Reactions of Fats and Fatty Acids Outline Fats and Oils Fatty Acid Biosynthesis Biodiesel Homework We hear quite a lot about the place of fats and oils in human nutrition. Foods high in fat are at the
Reactions of Fats and Fatty Acids Outline Fats and Oils Fatty Acid Biosynthesis Biodiesel Homework We hear quite a lot about the place of fats and oils in human nutrition. Foods high in fat are at the
Lipids. Classes of Lipids. Types of Lipids. Saturated and Unsaturated Fatty Acids. Fatty Acids. 15.1 Lipids 15.2 Fatty Acids
 hapter 15 15.1 15.2 Fatty Acids are biomolecules that contain fatty acids or a steroid nucleus. soluble in organic solvents, but not in water. named for the Greek word lipos, which means fat. extracted
hapter 15 15.1 15.2 Fatty Acids are biomolecules that contain fatty acids or a steroid nucleus. soluble in organic solvents, but not in water. named for the Greek word lipos, which means fat. extracted
PHOTOSYNTHESIS AND CELLULAR RESPIRATION
 reflect Wind turbines shown in the photo on the right are large structures with blades that move in response to air movement. When the wind blows, the blades rotate. This motion generates energy that is
reflect Wind turbines shown in the photo on the right are large structures with blades that move in response to air movement. When the wind blows, the blades rotate. This motion generates energy that is
The molecules of life. The molecules that make up living things are really big They are called macromolecules
 Food Labels All living things use materials and energy Our food comes from living things The food labels we see show us what our food is made of The stuff we are studying today can be found on food labels
Food Labels All living things use materials and energy Our food comes from living things The food labels we see show us what our food is made of The stuff we are studying today can be found on food labels
What happens to the food we eat? It gets broken down!
 Enzymes Essential Questions: What is an enzyme? How do enzymes work? What are the properties of enzymes? How do they maintain homeostasis for the body? What happens to the food we eat? It gets broken down!
Enzymes Essential Questions: What is an enzyme? How do enzymes work? What are the properties of enzymes? How do they maintain homeostasis for the body? What happens to the food we eat? It gets broken down!
Bioenergetics Module A Anchor 3
 Bioenergetics Module A Anchor 3 Key Concepts: - ATP can easily release and store energy by breaking and re-forming the bonds between its phosphate groups. This characteristic of ATP makes it exceptionally
Bioenergetics Module A Anchor 3 Key Concepts: - ATP can easily release and store energy by breaking and re-forming the bonds between its phosphate groups. This characteristic of ATP makes it exceptionally
Introduction to Biodiesel Chemistry Terms and Background Information
 Introduction to Biodiesel Chemistry Terms and Background Information Basic rganic Chemistry rganic chemistry is the branch of chemistry that deals with organic compounds. rganic compounds are compounds
Introduction to Biodiesel Chemistry Terms and Background Information Basic rganic Chemistry rganic chemistry is the branch of chemistry that deals with organic compounds. rganic compounds are compounds
Unit Vocabulary: o Organic Acid o Alcohol. o Ester o Ether. o Amine o Aldehyde
 Unit Vocabulary: Addition rxn Esterification Polymer Alcohol Ether Polymerization Aldehyde Fermentation Primary Alkane Functional group Saponification Alkene Halide (halocarbon) Saturated hydrocarbon Alkyne
Unit Vocabulary: Addition rxn Esterification Polymer Alcohol Ether Polymerization Aldehyde Fermentation Primary Alkane Functional group Saponification Alkene Halide (halocarbon) Saturated hydrocarbon Alkyne
Proteins and Nucleic Acids
 Proteins and Nucleic Acids Chapter 5 Macromolecules: Proteins Proteins Most structurally & functionally diverse group of biomolecules. : o Involved in almost everything o Enzymes o Structure (keratin,
Proteins and Nucleic Acids Chapter 5 Macromolecules: Proteins Proteins Most structurally & functionally diverse group of biomolecules. : o Involved in almost everything o Enzymes o Structure (keratin,
Chapter 49 - Nutrients and the Digestive System I. Nutrients (chemical substances necessary for organisms to grow and function properly)
 Chapter 49 - Nutrients and the Digestive System I. Nutrients (chemical substances necessary for organisms to grow and function properly) 6 basic nutrients - 4 food groups (milk, meat, fruit and vegetable,
Chapter 49 - Nutrients and the Digestive System I. Nutrients (chemical substances necessary for organisms to grow and function properly) 6 basic nutrients - 4 food groups (milk, meat, fruit and vegetable,
The correct answer is d C. Answer c is incorrect. Reliance on the energy produced by others is a characteristic of heterotrophs.
 1. An autotroph is an organism that a. extracts energy from organic sources b. converts energy from sunlight into chemical energy c. relies on the energy produced by other organisms as an energy source
1. An autotroph is an organism that a. extracts energy from organic sources b. converts energy from sunlight into chemical energy c. relies on the energy produced by other organisms as an energy source
USING THE FOOD LABEL TO FIND ITEMS THAT MEET THE EAT SMART IN PARKS GUIDELINES
 USING THE FOOD LABEL TO FIND ITEMS THAT MEET THE EAT SMART IN PARKS GUIDELINES FOOD LABELS Food Nacho chips label Although one serving of chips (1 ounce) meets the calorie guideline, 3 ounces of chips
USING THE FOOD LABEL TO FIND ITEMS THAT MEET THE EAT SMART IN PARKS GUIDELINES FOOD LABELS Food Nacho chips label Although one serving of chips (1 ounce) meets the calorie guideline, 3 ounces of chips
1. Essay: The Digestive and Absorption Processes of Macronutrients
 Jenny Kim Professor Rosario Nutrition: Macronutrients Project June 26, 2014 1. Essay: The Digestive and Absorption Processes of Macronutrients Whenever we eat, the foods we ingest in our bodies undergo
Jenny Kim Professor Rosario Nutrition: Macronutrients Project June 26, 2014 1. Essay: The Digestive and Absorption Processes of Macronutrients Whenever we eat, the foods we ingest in our bodies undergo
Alcohols. Copyright 2009 by Pearson Education, Inc. Copyright 2009 Pearson Education, Inc. CH 3 CH 2 CH 2 OH 1-propanol OH
 Chapter 12 rganic Compounds with xygen and Sulfur 12.1 Alcohols, Thiols, and Ethers Alcohols An alcohol contains a hydroxyl group ( ) attached to a carbon chain. A phenol contains a hydroxyl group ( )
Chapter 12 rganic Compounds with xygen and Sulfur 12.1 Alcohols, Thiols, and Ethers Alcohols An alcohol contains a hydroxyl group ( ) attached to a carbon chain. A phenol contains a hydroxyl group ( )
2. PHOTOSYNTHESIS. The general equation describing photosynthesis is light + 6 H 2 O + 6 CO 2 C 6 H 12 O 6 + 6 O 2
 2. PHOTOSYNTHESIS Photosynthesis is the process by which light energy is converted to chemical energy whereby carbon dioxide and water are converted into organic molecules. The process occurs in most algae,
2. PHOTOSYNTHESIS Photosynthesis is the process by which light energy is converted to chemical energy whereby carbon dioxide and water are converted into organic molecules. The process occurs in most algae,
Unit 5 Photosynthesis and Cellular Respiration
 Unit 5 Photosynthesis and Cellular Respiration Advanced Concepts What is the abbreviated name of this molecule? What is its purpose? What are the three parts of this molecule? Label each part with the
Unit 5 Photosynthesis and Cellular Respiration Advanced Concepts What is the abbreviated name of this molecule? What is its purpose? What are the three parts of this molecule? Label each part with the
Keystone Review Practice Test Module A Cells and Cell Processes. 1. Which characteristic is shared by all prokaryotes and eukaryotes?
 Keystone Review Practice Test Module A Cells and Cell Processes 1. Which characteristic is shared by all prokaryotes and eukaryotes? a. Ability to store hereditary information b. Use of organelles to control
Keystone Review Practice Test Module A Cells and Cell Processes 1. Which characteristic is shared by all prokaryotes and eukaryotes? a. Ability to store hereditary information b. Use of organelles to control
LAB 3: DIGESTION OF ORGANIC MACROMOLECULES
 LAB 3: DIGESTION OF ORGANIC MACROMOLECULES INTRODUCTION Enzymes are a special class of proteins that lower the activation energy of biological reactions. These biological catalysts change the rate of chemical
LAB 3: DIGESTION OF ORGANIC MACROMOLECULES INTRODUCTION Enzymes are a special class of proteins that lower the activation energy of biological reactions. These biological catalysts change the rate of chemical
