Experiment #12 Carbohydrates
|
|
|
- Marylou Hardy
- 7 years ago
- Views:
Transcription
1 Experiment #12 arbohydrates Introduction The name carbohydrate suggests that this class of compounds somehow consists of hydrated carbon. This idea gains some credence from the facts that (1) the molecular formulas of many carbohydrates are of the form x ( 2 ) y and (2) when certain dehydrating agents are added to certain carbohydrates (concentrated sulfuric acid to sucrose, for example) a black carbonaceous mass is formed. In fact, carbohydrates are polyhydroxy aldehydes or ketones, or compounds that can be hydrolyzed to give polyhydroxy aldehydes or ketones. The simplest carbohydrates are called monosaccharides; they are simple sugars and, as the name suggests, contain one sugar unit. When two sugar units are covalently bonded together, the carbohydrate is a disaccharide. If a few sugar units are joined together, the compound is called an oligosaccharide, and if many sugar units are joined together, the carbohydrate is called a polysaccharide. Glucose (grape or blood sugar) and fructose (found in many fruits) are monosaccharides; sucrose (table sugar) is a disaccharide; starch and cellulose are polysaccharides. Monosaccharides that are aldehydes can be called aldoses and those that are ketones can be called ketoses. Alcohols can react with aldehydes and unhindered ketones to produce compounds known as s. (The functional group has a carbon attached to an - and an -R group; it is an alcohol and an ether.) In aqueous solution this is a reversible reaction; an equilibrium is established in which molecules of the alcohol and carbonyl components react to form molecules of the at the same time that other molecules of the are breaking apart to form molecules of alcohol and carbonyl compound. R + R' R'' alcohol aldehyde or ketone R' R'' R In the above example the hydroxy group was part of one molecule (the alcohol) and the carbonyl was part of another (aldehyde or ketone). In the case of monosaccharides the monosaccharide molecule contains both the hydroxy group and the carbonyl group. ence, these two groups can react with each other within the same molecule. As a result of the new bond that is formed between the oxygen of the hydroxy group and the carbonyl carbon, the monosaccharide becomes a cyclic. This is shown below for the monosaccharide glucose. It is the group in bold face that reacts with the aldehyde group, shown in italics. The new bond that is formed between the hydroxy oxygen and carbonyl carbon to make the ring is shown bold.
2 Experiment #12 arbohydrates Page 2 [We should mention, in passing, that glucose, like most carbohydrates, is a chiral molecule. The enantiomer shown is the D-enantiomer, and it is this enantiomer that almost always shows up in nature, in free or combined form. The other enantiomer, found only rarely in nature, is the L-enantiomer and it is, of course, the mirror image of D-glucose.] 2 D-glucose Glucose in its open chain (polyhydroxyaldehyde) form. xygen on carbon-5 bonds to carbonyl carbon forming a ring. The carbonyl carbon becomes a new chiral center, leading to two diastereomers. 2 + β-d-glucose 2 Glucose in its (pyranose) forms. Two diastereomers of the (or pyranose) form of D-glucose are formed in the reaction as the result of the fact that the reaction generates a new chiral center in the molecule. When a monosaccharide contains a six-member ring including one oxygen, it can be called a pyranose, being named after pyran, the six-member cyclic ether that contains one oxygen. Glucose is a reducing sugar. No, you won t lose weight on a glucose diet, but it will reduce even weak oxidizing agents like Fehling s solution (u +2 u +1 ) or Tollen s reagent (Ag +1 Pyran, a cyclic ether. Monosaccharides incorporating this structure are called pyranoses. Ag 0 ) This is because, owing to the equilibrium shown above, at least some of the glucose is in the polyhydroxyaldehyde form and aldehydes are very easily oxidized. Almost always, sugars that have a ring structure are in equilibrium with the open-chain aldose or ketose form. As a result, these are reducing sugars. This is reasonable for aldoses, because aldehydes are easily oxidized, but what about the ketones, which are not usually easily oxidized? Well, it turns out that in the basic test solutions (Fehling s or Tollen s) ketoses can isomerize to aldoses. onsequently, ketoses that form s are reducing sugars (or, more exactly, are isomerized in the test solution to aldoses which are reducing sugars). In any event, the ketoses are called reducing sugars because the test reagent gives a positive test. Let s consider the generic that was formed from an aldehyde or ketone and an alcohol, above. As shown below it can react with a second molecule of alcohol to form an acetal. The acetal is stable in neutral aqueous solutions; it will not revert to and alcohol unless a fairly strong acid catalyst is added. This is different from the which usually produces at least some alcohol and aldehyde or ketone when dissolved in neutral aqueous solutions.
3 Experiment #12 arbohydrates Page 3 R''' + R' R'' alcohol R 3 + R''' R' R'' R acetal + 2 This same reaction can occur between the group of one monosaccharide molecule and a hydroxy group of another monosaccharide molecule. This leads to a disaccharide. For example, if the alcohol - on carbon-4 of an α-dglucose molecule reacts with the - of a second molecule the disaccharide maltose is formed maltose Locked ring: acetal penable ring: 2 Maltose is a reducing sugar. The ring on the left contains the acetal group and will not open to give an aldehyde in neutral or basic aqueous solutions. owever, the containing ring on the right will open in neutral or basic aqueous solutions to form an aldehyde. Therefore, maltose is a reducing sugar. Not all disaccharides are Sucrose - reducing sugars; not all a non-reducing disaccharides have a ring disaccharide: that contains a both rings group. have acetal Sucrose (table sugar), for groups; there example, is composed of are no 2 the monosaccharides α- s D-glucose and β-dfructose. In this case allow ring- that would both rings contain the opening to acetal group and neither an aldehyde ring will open to give an or hydroxyketone. aldehyde or α- hydroxyketone in neutral or basic solution. Therefore, sucrose is not a reducing sugar. 2 Locked rings: acetals 2 β-d-fructose
4 Experiment #12 arbohydrates Page 4 Starch is composed of two polysaccharides, amylose (ca. 20%) and amylopectin (ca. 80%). Both are polymers of α- D-glucose in its pyranose form. Amylose is a linear polymer, while amylopectin is branched. In other words, aside from branching which may occur, starch is the polysaccharide version of the disaccharide maltose. In each starch molecule, the pyranose glucose unit repeats itself hundreds or amylose 2 2 n Locked ring: acetal penable ring: 2 thousands of times. Each of these glucose units is held to the next through an acetal ether linkage. The last unit in the chain contains the group. owever, since there is only one unit out of hundreds or thousands of acetal units, starch does not give a positive test with Tollen s or Fehling s test solutions. If non-reducing disaccharides or the components of starch are hydrolyzed, the acetal functional groups will be converted to s. ydrolysis is catalyzed by strong acids and certain enzymes. For example, when sucrose, a non-reducing sugar, is hydrolyzed the reducing sugars glucose and fructose are formed. Likewise, when starch is hydrolyzed, the reducing sugar glucose is produced. Fehling s solution contains copper(ii) sulfate, sodium tartrate, and sodium hydroxide. The tartrate forms a blue complex with u +2, which would otherwise be precipitated as u() 2 in the basic solution. In the presence of a reducing sugar the u +2 is reduced to u +1. u +1 is not complexed by tartrate; it precipitates from solution as brick-red copper (I) oxide, u 2. The appearance of the brick-red color is a positive test as is a yellow-green color, which results from seeing the red precipitate through a blue solution. When iodine is added to starch an intense dark blue color results. This is the result of the I 2 molecules being lined up head to tail surrounded by a helix of starch. Because of this color, aqueous starch can be used to detect iodine and aqueous iodine can be used to detect starch. bjectives of the Experiment 1. To examine the reducing and non-reducing properties of several carbohydrates. 2. To hydrolyze acetal groups in sucrose and starch and thereby form products that have the group. 3. To dehydrate a carbohydrate with sulfuric acid.
5 Experiment #12 arbohydrates Page 5 Procedure aution: Aqueous sodium hydroxide and sulfuric acid are caustic. Wear goggles and in case of contact, wash with lots of water. Set up a steam bath and a hot water bath that is large enough to hold two medium size test tubes and which is maintained at 45 o. 1. A reducing carbohydrate or not? Label 5 medium size test tubes as follows: glucose, fructose, sucrose, lactose, starch. Place 20 drops of Fehling s A solution and 20 drops of Fehling s B solution in each of the test tubes. Add 10 drops of the corresponding 2% aqueous carbohydrate solution to each of the test tubes. Place the test tubes into the steam bath and steam them for about five minutes. Record the results on the report sheet. 2. ydrolysis of sucrose (acid versus base catalysis). ere you will determine if sucrose is hydrolyzed under acidic conditions, basic conditions, or both. Label two medium size test tubes #1 and #2. To each of the test tubes add 3 ml of 2% aqueous sucrose. To test tube #1 add 3 ml of deionized water and 3 drops of 3M sulfuric acid. To test tube #2 add 3 ml of deionized water and 3 drops of 3M aqueous sodium hydroxide. Steam both test tubes in the steam bath for 5 minutes. Allow the tubes to cool to room temperature. Add 3M aqueous sodium hydroxide to tube #1 dropwise, with stirring, until the contents turn red litmus paper blue. You will now test the contents of each tube with Fehling s solution (as you did above in part 1) as follows Label medium size test tubes #1a and #2a. Place 20 drops of Fehling s A solution and 20 drops of Fehling s B solution in each of the test tubes. Add 20 drops of the liquid in test tube #1 to test tube #1a and 20 drops of the liquid in test tube #2 to test tube #2a. Place test tubes #1a and #2a into the steam bath and steam them for about five minutes. Record the results on the report sheet. 3. ydrolysis of starch (acid versus enzyme catalysis). Label two medium size test tubes #1 and #2. To tube #1 add 2 ml (40 drops) of 3M sulfuric acid. To tube #2 add about 2 ml of your saliva. Try not to hack up a lungie here; we re looking for saliva, produced by the salivary glands in your mouth, which allegedly contains an enzyme that hydrolyzes starch. Add 2 ml of 2% aqueous starch solution to each of the test tubes. Stir the contents and place the tubes in the 45 o water bath for 30 minutes.
6 Experiment #12 arbohydrates Page 6 Label two small test tubes #1 and #2. Using two clean Pasteur pipettes transfer a few drops of liquid from each tube you have been heating into the correspondingly numbered small test tube. To each small test tube add 2 drops of 0.01M aqueous iodine solution. Record the colors of the materials in each test tube, comparing them with each other. 4. Kinetics of the acid catalyzed hydrolysis of starch. Place 5 ml of starch solution in a medium size test tube and add 1 ml (20 drops) of 3M sulfuric acid. Stir the mixture and heat the tube in the steam bath for 5 minutes. Using a clean Pasteur pipette, transfer 3 or 4 drops from the heating test tube to a clean small test tube. Add 2 drops of 0.01M aqueous iodine solution to the small test tube. bserve the contents of the small test tube. If starch is still present, you will see a blue color. If you do see a blue color, continue heating the starch-acid mixture and reperform the iodine test using a clean Pasteur pipette and small test tube. ontinue in this manner until you get a negative test for starch (no blue color). Record how long this took. 5. Dehydration of sucrose using concentrated sulfuric acid. Warning! oncentrated sulfuric acid is very caustic be careful and, as usual, wear goggles. If you get any on you wash it off immediately with lots of water. Since oxides of sulfur will be released into the air in this experiment, you should conduct it under a fume hood. Place about 40 grams of sucrose in a 150 ml beaker. Place the beaker in a fume hood. While you are stirring the sucrose with a glass stirring rod add concentrated sulfuric acid at a moderate rate until the sucrose is thoroughly wetted with the acid but is not awash in it. When you are finished adding the sulfuric acid, there should be a thin layer of acid above the sucrose-acid mixture. Wait a few minutes. Record your observations. When the contents of the beaker have cooled, hold the beaker under running water. After the solid contents of the beaker have been thoroughly washed with water you may deposit solids in a waste basket. lean the beaker with soap and water. sulfuric acid sucrose + sulfuric acid BK 11/03, Rev. 12/03, 4/04
The Chemistry of Carbohydrates
 The Chemistry of Carbohydrates Experiment #5 Objective: To determine the carbohydrate class of an unknown by carrying out a series of chemical reactions with the unknown and known compounds in each class
The Chemistry of Carbohydrates Experiment #5 Objective: To determine the carbohydrate class of an unknown by carrying out a series of chemical reactions with the unknown and known compounds in each class
3) How many monosaccharides are connected to each other in a disaccharide? A) 1 B) 2 C) 3 D) 4
 General, Organic, and Biochemistry, 2e (Frost) HOMEWORK Chapter 6 Carbohydrates Life s Sweet Molecules 6.1 Multiple-Choice 1) Which of the following is a polysaccharide? Glucose Sucrose C) Starch D) Maltose
General, Organic, and Biochemistry, 2e (Frost) HOMEWORK Chapter 6 Carbohydrates Life s Sweet Molecules 6.1 Multiple-Choice 1) Which of the following is a polysaccharide? Glucose Sucrose C) Starch D) Maltose
Conduct A Qualitative Test For Starch, Fat, A Reducing Sugar, A Protein
 Conduct A Qualitative Test For Starch, Fat, A Reducing Sugar, A Protein Biology Leaving Cert Experiments Materials/Equipment Starch solution (1%) Iodine Solution Glucose Solution (1%) 100 C) Benedict s
Conduct A Qualitative Test For Starch, Fat, A Reducing Sugar, A Protein Biology Leaving Cert Experiments Materials/Equipment Starch solution (1%) Iodine Solution Glucose Solution (1%) 100 C) Benedict s
CHEM 121. Chapter 18. Name: Date: 1. Which of the following compounds is both an aldose and a hexose? A) Page 1
 CEM 121. Chapter 18. Name: Date: 1. Which of the following compounds is both an aldose and a hexose? A) B) C) D) Page 1 2. Which of the following structures is that of an L-monosaccharide? A) B) C) D)
CEM 121. Chapter 18. Name: Date: 1. Which of the following compounds is both an aldose and a hexose? A) B) C) D) Page 1 2. Which of the following structures is that of an L-monosaccharide? A) B) C) D)
Qualitative Testing for Carbohydrates
 R E A 446 modular laboratory program in chemistry program editor:. A. Neidig Qualitative Testing for arbohydrates prepared by James. Schreck, University of Northern olorado, and William M. Loffredo, East
R E A 446 modular laboratory program in chemistry program editor:. A. Neidig Qualitative Testing for arbohydrates prepared by James. Schreck, University of Northern olorado, and William M. Loffredo, East
Organic Molecules of Life - Exercise 2
 Organic Molecules of Life - Exercise 2 Objectives -Know the difference between a reducing sugar and a non-reducing sugar. -Distinguish Monosaccharides from Disaccharides and Polysaccharides -Understand
Organic Molecules of Life - Exercise 2 Objectives -Know the difference between a reducing sugar and a non-reducing sugar. -Distinguish Monosaccharides from Disaccharides and Polysaccharides -Understand
Experiment 10 Enzymes
 Experiment 10 Enzymes Enzymes are proteins that act as catalysts for biological reactions. Enzymes, like all catalysts, speed up reactions without being used up themselves. They do this by lowering the
Experiment 10 Enzymes Enzymes are proteins that act as catalysts for biological reactions. Enzymes, like all catalysts, speed up reactions without being used up themselves. They do this by lowering the
Lab 2 Biochemistry. Learning Objectives. Introduction. Lipid Structure and Role in Food. The lab has the following learning objectives.
 1 Lab 2 Biochemistry Learning Objectives The lab has the following learning objectives. Investigate the role of double bonding in fatty acids, through models. Developing a calibration curve for a Benedict
1 Lab 2 Biochemistry Learning Objectives The lab has the following learning objectives. Investigate the role of double bonding in fatty acids, through models. Developing a calibration curve for a Benedict
LAB 3: DIGESTION OF ORGANIC MACROMOLECULES
 LAB 3: DIGESTION OF ORGANIC MACROMOLECULES INTRODUCTION Enzymes are a special class of proteins that lower the activation energy of biological reactions. These biological catalysts change the rate of chemical
LAB 3: DIGESTION OF ORGANIC MACROMOLECULES INTRODUCTION Enzymes are a special class of proteins that lower the activation energy of biological reactions. These biological catalysts change the rate of chemical
Experiment #8 properties of Alcohols and Phenols
 Introduction Experiment #8 properties of Alcohols and Phenols As has been mentioned before, over 20 million organic compounds have been identified. If each substance had to be studied as an entity completely
Introduction Experiment #8 properties of Alcohols and Phenols As has been mentioned before, over 20 million organic compounds have been identified. If each substance had to be studied as an entity completely
Chapter 3 Molecules of Cells
 Bio 100 Molecules of cells 1 Chapter 3 Molecules of Cells Compounds containing carbon are called organic compounds Molecules such as methane that are only composed of carbon and hydrogen are called hydrocarbons
Bio 100 Molecules of cells 1 Chapter 3 Molecules of Cells Compounds containing carbon are called organic compounds Molecules such as methane that are only composed of carbon and hydrogen are called hydrocarbons
Human Physiology Lab (Biol 236L) Digestive Physiology: Amylase hydrolysis of starch
 Human Physiology Lab (Biol 236L) Digestive Physiology: Amylase hydrolysis of starch Introduction Enzymes are proteins composed of amino acid building blocks. Enzymes catalyze or increase the rate of metabolic
Human Physiology Lab (Biol 236L) Digestive Physiology: Amylase hydrolysis of starch Introduction Enzymes are proteins composed of amino acid building blocks. Enzymes catalyze or increase the rate of metabolic
Chapter 5 Classification of Organic Compounds by Solubility
 Chapter 5 Classification of Organic Compounds by Solubility Deductions based upon interpretation of simple solubility tests can be extremely useful in organic structure determination. Both solubility and
Chapter 5 Classification of Organic Compounds by Solubility Deductions based upon interpretation of simple solubility tests can be extremely useful in organic structure determination. Both solubility and
The Structure and Function of Macromolecules: Carbohydrates, Lipids & Phospholipids
 The Structure and Function of Macromolecules: Carbohydrates, Lipids & Phospholipids The FOUR Classes of Large Biomolecules All living things are made up of four classes of large biological molecules: Carbohydrates
The Structure and Function of Macromolecules: Carbohydrates, Lipids & Phospholipids The FOUR Classes of Large Biomolecules All living things are made up of four classes of large biological molecules: Carbohydrates
EXPERIMENT 6 (Organic Chemistry II) Identification of Ketones and Aldehydes
 EXPERIMENT 6 (rganic hemistry II) Identification of Ketones and Aldehydes Pahlavan/herif hemicals 2,4-Dinitrophenylhydrazine(DNP) 0.10 M AgN 3 2-propanol Benzaldehyde 10% Na 2-butanol Methyl ethyl Ketone
EXPERIMENT 6 (rganic hemistry II) Identification of Ketones and Aldehydes Pahlavan/herif hemicals 2,4-Dinitrophenylhydrazine(DNP) 0.10 M AgN 3 2-propanol Benzaldehyde 10% Na 2-butanol Methyl ethyl Ketone
Recovery of Elemental Copper from Copper (II) Nitrate
 Recovery of Elemental Copper from Copper (II) Nitrate Objectives: Challenge: Students should be able to - recognize evidence(s) of a chemical change - convert word equations into formula equations - perform
Recovery of Elemental Copper from Copper (II) Nitrate Objectives: Challenge: Students should be able to - recognize evidence(s) of a chemical change - convert word equations into formula equations - perform
Biochemistry of Cells
 Biochemistry of Cells 1 Carbon-based Molecules Although a cell is mostly water, the rest of the cell consists mostly of carbon-based molecules Organic chemistry is the study of carbon compounds Carbon
Biochemistry of Cells 1 Carbon-based Molecules Although a cell is mostly water, the rest of the cell consists mostly of carbon-based molecules Organic chemistry is the study of carbon compounds Carbon
Catalysis by Enzymes. Enzyme A protein that acts as a catalyst for a biochemical reaction.
 Catalysis by Enzymes Enzyme A protein that acts as a catalyst for a biochemical reaction. Enzymatic Reaction Specificity Enzyme Cofactors Many enzymes are conjugated proteins that require nonprotein portions
Catalysis by Enzymes Enzyme A protein that acts as a catalyst for a biochemical reaction. Enzymatic Reaction Specificity Enzyme Cofactors Many enzymes are conjugated proteins that require nonprotein portions
A disaccharide is formed when a dehydration reaction joins two monosaccharides. This covalent bond is called a glycosidic linkage.
 CH 5 Structure & Function of Large Molecules: Macromolecules Molecules of Life All living things are made up of four classes of large biological molecules: carbohydrates, lipids, proteins, and nucleic
CH 5 Structure & Function of Large Molecules: Macromolecules Molecules of Life All living things are made up of four classes of large biological molecules: carbohydrates, lipids, proteins, and nucleic
EXPERIMENT 9 (Organic Chemistry II) Pahlavan - Cherif Synthesis of Aspirin - Esterification
 EXPERIMENT 9 (rganic hemistry II) Pahlavan - herif Materials Hot plate 125-mL Erlenmeyer flask Melting point capillaries Melting point apparatus Büchner funnel 400-mL beaker Stirring rod hemicals Salicylic
EXPERIMENT 9 (rganic hemistry II) Pahlavan - herif Materials Hot plate 125-mL Erlenmeyer flask Melting point capillaries Melting point apparatus Büchner funnel 400-mL beaker Stirring rod hemicals Salicylic
Lecture Overview. Hydrogen Bonds. Special Properties of Water Molecules. Universal Solvent. ph Scale Illustrated. special properties of water
 Lecture Overview special properties of water > water as a solvent > ph molecules of the cell > properties of carbon > carbohydrates > lipids > proteins > nucleic acids Hydrogen Bonds polarity of water
Lecture Overview special properties of water > water as a solvent > ph molecules of the cell > properties of carbon > carbohydrates > lipids > proteins > nucleic acids Hydrogen Bonds polarity of water
experiment5 Understanding and applying the concept of limiting reagents. Learning how to perform a vacuum filtration.
 81 experiment5 LECTURE AND LAB SKILLS EMPHASIZED Synthesizing an organic substance. Understanding and applying the concept of limiting reagents. Determining percent yield. Learning how to perform a vacuum
81 experiment5 LECTURE AND LAB SKILLS EMPHASIZED Synthesizing an organic substance. Understanding and applying the concept of limiting reagents. Determining percent yield. Learning how to perform a vacuum
Chapter 5: The Structure and Function of Large Biological Molecules
 Name Period Concept 5.1 Macromolecules are polymers, built from monomers 1. The large molecules of all living things fall into just four main classes. Name them. 2. Circle the three classes that are called
Name Period Concept 5.1 Macromolecules are polymers, built from monomers 1. The large molecules of all living things fall into just four main classes. Name them. 2. Circle the three classes that are called
Lab 3 Organic Molecules of Biological Importance
 Name Biology 3 ID Number Lab 3 Organic Molecules of Biological Importance Section 1 - Organic Molecules Section 2 - Functional Groups Section 3 - From Building Blocks to Macromolecules Section 4 - Carbohydrates
Name Biology 3 ID Number Lab 3 Organic Molecules of Biological Importance Section 1 - Organic Molecules Section 2 - Functional Groups Section 3 - From Building Blocks to Macromolecules Section 4 - Carbohydrates
Amino Acids, Peptides, and Proteins
 1 Amino Acids, Peptides, and Proteins Introduction Amino Acids Amino acids are the building blocks of proteins. In class you learned the structures of the 20 common amino acids that make up proteins. All
1 Amino Acids, Peptides, and Proteins Introduction Amino Acids Amino acids are the building blocks of proteins. In class you learned the structures of the 20 common amino acids that make up proteins. All
The Huntington Library, Art Collections, and Botanical Gardens. How Sweet It Is: Enzyme Action in Seed Germination
 The Huntington Library, Art Collections, and Botanical Gardens How Sweet It Is: Enzyme Action in Seed Germination Overview This experiment is intended to familiarize students with the macromolecule starch,
The Huntington Library, Art Collections, and Botanical Gardens How Sweet It Is: Enzyme Action in Seed Germination Overview This experiment is intended to familiarize students with the macromolecule starch,
Experiment 8 - Double Displacement Reactions
 Experiment 8 - Double Displacement Reactions A double displacement reaction involves two ionic compounds that are dissolved in water. In a double displacement reaction, it appears as though the ions are
Experiment 8 - Double Displacement Reactions A double displacement reaction involves two ionic compounds that are dissolved in water. In a double displacement reaction, it appears as though the ions are
Synthesis of Aspirin and Oil of Wintergreen
 Austin Peay State University Department of hemistry hem 1121 autions Purpose Introduction Acetic Anhydride corrosive and a lachrymator all transfers should be done in the vented fume hood Methanol, Ethanol
Austin Peay State University Department of hemistry hem 1121 autions Purpose Introduction Acetic Anhydride corrosive and a lachrymator all transfers should be done in the vented fume hood Methanol, Ethanol
I. Chapter 5 Summary. II. Nucleotides & Nucleic Acids. III. Lipids
 I. Chapter 5 Summary A. Simple Sugars (CH 2 O) n : 1. One C contains a carbonyl (C=O) rest contain - 2. Classification by functional group: aldoses & ketoses 3. Classification by number of C's: trioses,
I. Chapter 5 Summary A. Simple Sugars (CH 2 O) n : 1. One C contains a carbonyl (C=O) rest contain - 2. Classification by functional group: aldoses & ketoses 3. Classification by number of C's: trioses,
Aldehydes can react with alcohols to form hemiacetals. 340 14. Nucleophilic substitution at C=O with loss of carbonyl oxygen
 340 14. Nucleophilic substitution at C= with loss of carbonyl oxygen Ph In Chapter 13 we saw this way of making a reaction go faster by raising the energy of the starting material. We also saw that the
340 14. Nucleophilic substitution at C= with loss of carbonyl oxygen Ph In Chapter 13 we saw this way of making a reaction go faster by raising the energy of the starting material. We also saw that the
Elements & Macromolecules in Organisms
 Name: Date: Per: Table # Elements & Macromolecules in rganisms Most common elements in living things are carbon, hydrogen, nitrogen, and oxygen. These four elements constitute about 95% of your body weight.
Name: Date: Per: Table # Elements & Macromolecules in rganisms Most common elements in living things are carbon, hydrogen, nitrogen, and oxygen. These four elements constitute about 95% of your body weight.
Biology 13A Lab #13: Nutrition and Digestion
 Biology 13A Lab #13: Nutrition and Digestion Lab #13 Table of Contents: Expected Learning Outcomes.... 102 Introduction...... 103 Food Chemistry & Nutrition.... 104 Activity 1: Testing for the Presence
Biology 13A Lab #13: Nutrition and Digestion Lab #13 Table of Contents: Expected Learning Outcomes.... 102 Introduction...... 103 Food Chemistry & Nutrition.... 104 Activity 1: Testing for the Presence
Carbon-organic Compounds
 Elements in Cells The living substance of cells is made up of cytoplasm and the structures within it. About 96% of cytoplasm and its included structures are composed of the elements carbon, hydrogen, oxygen,
Elements in Cells The living substance of cells is made up of cytoplasm and the structures within it. About 96% of cytoplasm and its included structures are composed of the elements carbon, hydrogen, oxygen,
McMush. Testing for the Presence of Biomolecules
 Biology McMush Testing for the Presence of Biomolecules MATERIALS AND RESOURCES EACH GROUP aprons beaker, 250 ml 2 clamps, test tube goggles graduated cylinder, 50 ml paper towels test tube brush test
Biology McMush Testing for the Presence of Biomolecules MATERIALS AND RESOURCES EACH GROUP aprons beaker, 250 ml 2 clamps, test tube goggles graduated cylinder, 50 ml paper towels test tube brush test
Name: Hour: Elements & Macromolecules in Organisms
 Name: Hour: Elements & Macromolecules in Organisms Most common elements in living things are carbon, hydrogen, nitrogen, and oxygen. These four elements constitute about 95% of your body weight. All compounds
Name: Hour: Elements & Macromolecules in Organisms Most common elements in living things are carbon, hydrogen, nitrogen, and oxygen. These four elements constitute about 95% of your body weight. All compounds
Disaccharides consist of two monosaccharide monomers covalently linked by a glycosidic bond. They function in sugar transport.
 1. The fundamental life processes of plants and animals depend on a variety of chemical reactions that occur in specialized areas of the organism s cells. As a basis for understanding this concept: 1.
1. The fundamental life processes of plants and animals depend on a variety of chemical reactions that occur in specialized areas of the organism s cells. As a basis for understanding this concept: 1.
Macromolecules in my food!!
 Macromolecules in my food!! Name Notes/Background Information Food is fuel: All living things need to obtain fuel from something. Whether it is self- made through the process of photosynthesis, or by ingesting
Macromolecules in my food!! Name Notes/Background Information Food is fuel: All living things need to obtain fuel from something. Whether it is self- made through the process of photosynthesis, or by ingesting
CHEMICAL DETERMINATION OF EVERYDAY HOUSEHOLD CHEMICALS
 CHEMICAL DETERMINATION OF EVERYDAY HOUSEHOLD CHEMICALS Purpose: It is important for chemists to be able to determine the composition of unknown chemicals. This can often be done by way of chemical tests.
CHEMICAL DETERMINATION OF EVERYDAY HOUSEHOLD CHEMICALS Purpose: It is important for chemists to be able to determine the composition of unknown chemicals. This can often be done by way of chemical tests.
General Chemistry I (FC, 09-10) Lab #3: The Empirical Formula of a Compound. Introduction
 General Chemistry I (FC, 09-10) Introduction A look at the mass relationships in chemistry reveals little order or sense. The ratio of the masses of the elements in a compound, while constant, does not
General Chemistry I (FC, 09-10) Introduction A look at the mass relationships in chemistry reveals little order or sense. The ratio of the masses of the elements in a compound, while constant, does not
10-ml Graduated cylinder 40 ml 3% Hydrogen peroxide solution (found in stores) Straight-edged razor blade Scissors and Forceps (tweezers)
 Name: Class: Date: Objectives * Measure the effects of changes in temperature, ph, and enzyme concentration on reaction rates of an enzyme catalyzed reaction in a controlled experiment. * Explain how environmental
Name: Class: Date: Objectives * Measure the effects of changes in temperature, ph, and enzyme concentration on reaction rates of an enzyme catalyzed reaction in a controlled experiment. * Explain how environmental
Laboratory 22: Properties of Alcohols
 Introduction Alcohols represent and important class of organic molecules. In this experiment you will study the physical and chemical properties of alcohols. Solubility in water, and organic solvents,
Introduction Alcohols represent and important class of organic molecules. In this experiment you will study the physical and chemical properties of alcohols. Solubility in water, and organic solvents,
EXPERIMENT 7 Reaction Stoichiometry and Percent Yield
 EXPERIMENT 7 Reaction Stoichiometry and Percent Yield INTRODUCTION Stoichiometry calculations are about calculating the amounts of substances that react and form in a chemical reaction. The word stoichiometry
EXPERIMENT 7 Reaction Stoichiometry and Percent Yield INTRODUCTION Stoichiometry calculations are about calculating the amounts of substances that react and form in a chemical reaction. The word stoichiometry
Absorption and Transport of Nutrients
 Page1 Digestion Food travels from mouth esophagus stomach small intestine colon rectum anus. Food mixes with digestive juices, moving it through the digestive tract Large molecules of food are broken into
Page1 Digestion Food travels from mouth esophagus stomach small intestine colon rectum anus. Food mixes with digestive juices, moving it through the digestive tract Large molecules of food are broken into
LABORATORY 5 DETECTION OF FUNCTIONAL GROUPS IN ORGANIC COMPOUNDS
 LABATY 5 DETETIN F FUNTINAL GUPS IN GANI MPUNDS I. haracteristic reactions differentiating saturated aliphatic hydrocarbons from unsaturated aliphatic hydrocarbons 1. The test of bromine addition. Unsaturated
LABATY 5 DETETIN F FUNTINAL GUPS IN GANI MPUNDS I. haracteristic reactions differentiating saturated aliphatic hydrocarbons from unsaturated aliphatic hydrocarbons 1. The test of bromine addition. Unsaturated
Experiment 5 Preparation of Cyclohexene
 Experiment 5 Preparation of yclohexene In this experiment we will prepare cyclohexene from cyclohexanol using an acid catalyzed dehydration reaction. We will use the cyclohexanol that we purified in our
Experiment 5 Preparation of yclohexene In this experiment we will prepare cyclohexene from cyclohexanol using an acid catalyzed dehydration reaction. We will use the cyclohexanol that we purified in our
Properties of Acids and Bases
 Lab 22 Properties of Acids and Bases TN Standard 4.2: The student will investigate the characteristics of acids and bases. Have you ever brushed your teeth and then drank a glass of orange juice? What
Lab 22 Properties of Acids and Bases TN Standard 4.2: The student will investigate the characteristics of acids and bases. Have you ever brushed your teeth and then drank a glass of orange juice? What
The Empirical Formula of a Compound
 The Empirical Formula of a Compound Lab #5 Introduction A look at the mass relationships in chemistry reveals little order or sense. The ratio of the masses of the elements in a compound, while constant,
The Empirical Formula of a Compound Lab #5 Introduction A look at the mass relationships in chemistry reveals little order or sense. The ratio of the masses of the elements in a compound, while constant,
Organic Compounds. Essential Questions: What is Organic? What are the 4 major Organic Compounds? How are they made? What are they used for?
 Organic Compounds Essential Questions: What is Organic? What are the 4 major Organic Compounds? How are they made? What are they used for? Aristotle: Francesco Redi: What do we already know? Spontaneous
Organic Compounds Essential Questions: What is Organic? What are the 4 major Organic Compounds? How are they made? What are they used for? Aristotle: Francesco Redi: What do we already know? Spontaneous
Chapter 5. The Structure and Function of Macromolecule s
 Chapter 5 The Structure and Function of Macromolecule s Most Macromolecules are polymers: Polymer: (poly: many; mer: part) Large molecules consisting of many identical or similar subunits connected together.
Chapter 5 The Structure and Function of Macromolecule s Most Macromolecules are polymers: Polymer: (poly: many; mer: part) Large molecules consisting of many identical or similar subunits connected together.
Elements in Biological Molecules
 Chapter 3: Biological Molecules 1. Carbohydrates 2. Lipids 3. Proteins 4. Nucleic Acids Elements in Biological Molecules Biological macromolecules are made almost entirely of just 6 elements: Carbon (C)
Chapter 3: Biological Molecules 1. Carbohydrates 2. Lipids 3. Proteins 4. Nucleic Acids Elements in Biological Molecules Biological macromolecules are made almost entirely of just 6 elements: Carbon (C)
Experiment 6 Qualitative Tests for Alcohols, Alcohol Unknown, IR of Unknown
 Experiment 6 Qualitative Tests for Alcohols, Alcohol Unknown, I of Unknown In this experiment you are going to do a series of tests in order to determine whether or not an alcohol is a primary (1 ), secondary
Experiment 6 Qualitative Tests for Alcohols, Alcohol Unknown, I of Unknown In this experiment you are going to do a series of tests in order to determine whether or not an alcohol is a primary (1 ), secondary
Chapter 3: Biological Molecules. 1. Carbohydrates 2. Lipids 3. Proteins 4. Nucleic Acids
 Chapter 3: Biological Molecules 1. Carbohydrates 2. Lipids 3. Proteins 4. Nucleic Acids Elements in Biological Molecules Biological macromolecules are made almost entirely of just 6 elements: Carbon (C)
Chapter 3: Biological Molecules 1. Carbohydrates 2. Lipids 3. Proteins 4. Nucleic Acids Elements in Biological Molecules Biological macromolecules are made almost entirely of just 6 elements: Carbon (C)
CHEMISTRY AND BIOLOGICAL ROLE OF CARBOHYDRATES IN THE BODY-1
 CHEMISTRY AND BIOLOGICAL ROLE OF CARBOHYDRATES IN THE BODY-1 Chiral centers: Asymmetric carbons, i.e carbon atom with four different substituents Enantiomers : Mirror images Stereoisomers MONOSACCHARIDE
CHEMISTRY AND BIOLOGICAL ROLE OF CARBOHYDRATES IN THE BODY-1 Chiral centers: Asymmetric carbons, i.e carbon atom with four different substituents Enantiomers : Mirror images Stereoisomers MONOSACCHARIDE
ISOLATION OF CAFFEINE FROM TEA
 ISLATIN F CAFFEINE FRM TEA Introduction In this experiment, caffeine is isolated from tealeaves. The chief problem with the isolation is that caffeine does not exist alone in the tealeaves, but other natural
ISLATIN F CAFFEINE FRM TEA Introduction In this experiment, caffeine is isolated from tealeaves. The chief problem with the isolation is that caffeine does not exist alone in the tealeaves, but other natural
Water. Definition: A mole (or mol ) Water can IONIZE transiently. NONpolar covalent molecules do not dissolve in water + + + + + + + + + + + + + + + +
 Today s Topics Polar Covalent Bonds ydrogen bonding Properties of water p Water C bonds are Nonpolar Will these molecules dissolve in water? Start Macromolecules Carbohydrates & Lipids Sept 4, 05 Why are
Today s Topics Polar Covalent Bonds ydrogen bonding Properties of water p Water C bonds are Nonpolar Will these molecules dissolve in water? Start Macromolecules Carbohydrates & Lipids Sept 4, 05 Why are
EXPERIMENT 10 Chemistry 110. Solutions Part 2 ACIDS, BASES, AND ELECTROLYTES
 EXPERIMENT 10 Chemistry 110 Solutions Part 2 ACIDS, BASES, AND ELECTROLYTES PURPOSE: The purpose of this experiment is to determine the properties of solutions of acids, bases and electrolytes. Students
EXPERIMENT 10 Chemistry 110 Solutions Part 2 ACIDS, BASES, AND ELECTROLYTES PURPOSE: The purpose of this experiment is to determine the properties of solutions of acids, bases and electrolytes. Students
Carbohydrates, proteins and lipids
 Carbohydrates, proteins and lipids Chapter 3 MACROMOLECULES Macromolecules: polymers with molecular weights >1,000 Functional groups THE FOUR MACROMOLECULES IN LIFE Molecules in living organisms: proteins,
Carbohydrates, proteins and lipids Chapter 3 MACROMOLECULES Macromolecules: polymers with molecular weights >1,000 Functional groups THE FOUR MACROMOLECULES IN LIFE Molecules in living organisms: proteins,
CHM220 Addition lab. Experiment: Reactions of alkanes, alkenes, and cycloalkenes*
 CM220 Addition lab Experiment: Reactions of alkanes, alkenes, and cycloalkenes* Purpose: To investigate the physical properties, solubility, and density of some hydrocarbon. To compare the chemical reactivity
CM220 Addition lab Experiment: Reactions of alkanes, alkenes, and cycloalkenes* Purpose: To investigate the physical properties, solubility, and density of some hydrocarbon. To compare the chemical reactivity
Making Biodiesel from Virgin Vegetable Oil: Teacher Manual
 Making Biodiesel from Virgin Vegetable Oil: Teacher Manual Learning Goals: Students will understand how to produce biodiesel from virgin vegetable oil. Students will understand the effect of an exothermic
Making Biodiesel from Virgin Vegetable Oil: Teacher Manual Learning Goals: Students will understand how to produce biodiesel from virgin vegetable oil. Students will understand the effect of an exothermic
The Molecules of Cells
 The Molecules of Cells I. Introduction A. Most of the world s population cannot digest milk-based foods. 1. These people are lactose intolerant because they lack the enzyme lactase. 2. This illustrates
The Molecules of Cells I. Introduction A. Most of the world s population cannot digest milk-based foods. 1. These people are lactose intolerant because they lack the enzyme lactase. 2. This illustrates
CHM 130LL: ph, Buffers, and Indicators
 CHM 130LL: ph, Buffers, and Indicators Many substances can be classified as acidic or basic. Acidic substances contain hydrogen ions, H +, while basic substances contain hydroxide ions, OH. The relative
CHM 130LL: ph, Buffers, and Indicators Many substances can be classified as acidic or basic. Acidic substances contain hydrogen ions, H +, while basic substances contain hydroxide ions, OH. The relative
Table 1. Common esters used for flavors and fragrances
 ESTERS An Introduction to rganic hemistry Reactions 2012, 2006, 1990, 1982 by David A. Katz. All rights reserved. Reproduction permitted for educationa use provided original copyright is included. In contrast
ESTERS An Introduction to rganic hemistry Reactions 2012, 2006, 1990, 1982 by David A. Katz. All rights reserved. Reproduction permitted for educationa use provided original copyright is included. In contrast
Chemical Changes. Measuring a Chemical Reaction. Name(s)
 Chemical Changes Name(s) In the particle model of matter, individual atoms can be bound tightly to other atoms to form molecules. For example, water molecules are made up of two hydrogen atoms bound to
Chemical Changes Name(s) In the particle model of matter, individual atoms can be bound tightly to other atoms to form molecules. For example, water molecules are made up of two hydrogen atoms bound to
Properties of Alcohols and Phenols Experiment #3
 Properties of Alcohols and Phenols Experiment #3 Objectives: To observe the solubility of alcohols relative to their chemical structure, to perform chemical tests to distinguish primary, secondary and
Properties of Alcohols and Phenols Experiment #3 Objectives: To observe the solubility of alcohols relative to their chemical structure, to perform chemical tests to distinguish primary, secondary and
The Properties of Water (Instruction Sheet)
 The Properties of Water (Instruction Sheet) Property : High Polarity Activity #1 Surface Tension: PILE IT ON. Materials: 1 DRY penny, 1 eye dropper, water. 1. Make sure the penny is dry. 2. Begin by estimating
The Properties of Water (Instruction Sheet) Property : High Polarity Activity #1 Surface Tension: PILE IT ON. Materials: 1 DRY penny, 1 eye dropper, water. 1. Make sure the penny is dry. 2. Begin by estimating
Chemical Basis of Life Module A Anchor 2
 Chemical Basis of Life Module A Anchor 2 Key Concepts: - Water is a polar molecule. Therefore, it is able to form multiple hydrogen bonds, which account for many of its special properties. - Water s polarity
Chemical Basis of Life Module A Anchor 2 Key Concepts: - Water is a polar molecule. Therefore, it is able to form multiple hydrogen bonds, which account for many of its special properties. - Water s polarity
Chapter 16: Tests for ions and gases
 The position of hydrogen in the reactivity series Hydrogen, although not a metal, is included in the reactivity series because it, like metals, can be displaced from aqueous solution, only this time the
The position of hydrogen in the reactivity series Hydrogen, although not a metal, is included in the reactivity series because it, like metals, can be displaced from aqueous solution, only this time the
Determination of Specific Nutrients in Various Foods. Abstract. Humans need to consume food compounds such as carbohydrates, proteins, fats,
 Determination of Specific Nutrients in Various Foods Abstract Humans need to consume food compounds such as carbohydrates, proteins, fats, and vitamins to meet their energy requirements. In this lab, reagents
Determination of Specific Nutrients in Various Foods Abstract Humans need to consume food compounds such as carbohydrates, proteins, fats, and vitamins to meet their energy requirements. In this lab, reagents
1. The diagram below represents a biological process
 1. The diagram below represents a biological process 5. The chart below indicates the elements contained in four different molecules and the number of atoms of each element in those molecules. Which set
1. The diagram below represents a biological process 5. The chart below indicates the elements contained in four different molecules and the number of atoms of each element in those molecules. Which set
IB Chemistry 1 Mole. One atom of C-12 has a mass of 12 amu. One mole of C-12 has a mass of 12 g. Grams we can use more easily.
 The Mole Atomic mass units and atoms are not convenient units to work with. The concept of the mole was invented. This was the number of atoms of carbon-12 that were needed to make 12 g of carbon. 1 mole
The Mole Atomic mass units and atoms are not convenient units to work with. The concept of the mole was invented. This was the number of atoms of carbon-12 that were needed to make 12 g of carbon. 1 mole
Solid Phase Peptide Synthesis
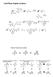 Solid Phase Peptide Synthesis Fischer Projections revisited C C C Remember that group rotations are always allowed C C 80 rotations of entire molecule are allowed C 80 C 90 rotations of entire molecule
Solid Phase Peptide Synthesis Fischer Projections revisited C C C Remember that group rotations are always allowed C C 80 rotations of entire molecule are allowed C 80 C 90 rotations of entire molecule
Determination of a Chemical Formula
 1 Determination of a Chemical Formula Introduction Molar Ratios Elements combine in fixed ratios to form compounds. For example, consider the compound TiCl 4 (titanium chloride). Each molecule of TiCl
1 Determination of a Chemical Formula Introduction Molar Ratios Elements combine in fixed ratios to form compounds. For example, consider the compound TiCl 4 (titanium chloride). Each molecule of TiCl
Chemical Processes of Digestion
 Chemical Processes of Digestion Objective: To explain in short essays or diagrams how carbohydrates, proteins, and fats are digested into end products that can be absorbed into the blood, at the level
Chemical Processes of Digestion Objective: To explain in short essays or diagrams how carbohydrates, proteins, and fats are digested into end products that can be absorbed into the blood, at the level
Experiment 8 Preparation of Cyclohexanone by Hypochlorite Oxidation
 Experiment 8 Preparation of Cyclohexanone by ypochlorite xidation In this experiment we will prepare cyclohexanone from cyclohexanol using hypochlorite oxidation. We will use common household bleach that
Experiment 8 Preparation of Cyclohexanone by ypochlorite xidation In this experiment we will prepare cyclohexanone from cyclohexanol using hypochlorite oxidation. We will use common household bleach that
Metabolism: Cellular Respiration, Fermentation and Photosynthesis
 Metabolism: Cellular Respiration, Fermentation and Photosynthesis Introduction: All organisms require a supply of energy and matter to build themselves and to continue to function. To get that supply of
Metabolism: Cellular Respiration, Fermentation and Photosynthesis Introduction: All organisms require a supply of energy and matter to build themselves and to continue to function. To get that supply of
Enzyme Pre-Lab. Using the Enzyme worksheet and Enzyme lab handout answer the Pre-Lab questions the pre-lab must be complete before beginning the lab.
 Enzyme Pre-Lab Using the Enzyme worksheet and Enzyme lab handout answer the Pre-Lab questions the pre-lab must be complete before beginning the lab. Background: In this investigation, you will study several
Enzyme Pre-Lab Using the Enzyme worksheet and Enzyme lab handout answer the Pre-Lab questions the pre-lab must be complete before beginning the lab. Background: In this investigation, you will study several
SYNTHESIS AND ANALYSIS OF A COORDINATION COMPOUND OF COPPER
 Chemistry 111 Lab: Synthesis of a Copper Complex Page H-1 SYNTHESIS AND ANALYSIS OF A COORDINATION COMPOUND OF COPPER In this experiment you will synthesize a compound by adding NH 3 to a concentrated
Chemistry 111 Lab: Synthesis of a Copper Complex Page H-1 SYNTHESIS AND ANALYSIS OF A COORDINATION COMPOUND OF COPPER In this experiment you will synthesize a compound by adding NH 3 to a concentrated
Recognizing Organic Molecules: Carbohydrates, Lipids and Proteins
 Recognizing Organic Molecules: Carbohydrates, Lipids and Proteins Oct 15 8:05 PM What is an Organic Molecule? An Organic Molecule is a molecule that contains carbon and hydrogen and oxygen Carbon is found
Recognizing Organic Molecules: Carbohydrates, Lipids and Proteins Oct 15 8:05 PM What is an Organic Molecule? An Organic Molecule is a molecule that contains carbon and hydrogen and oxygen Carbon is found
Macromolecules 1 Carbohydrates, Lipids & Nucleic Acids
 VEA Bringing Learning to Life Program Support Notes Macromolecules 1 Carbohydrates, Lipids & Nucleic Acids Grades 10 - College 25mins Teacher Notes by Sue Wright, B. Sc., Dip. Ed. Produced by VEA Pty Ltd
VEA Bringing Learning to Life Program Support Notes Macromolecules 1 Carbohydrates, Lipids & Nucleic Acids Grades 10 - College 25mins Teacher Notes by Sue Wright, B. Sc., Dip. Ed. Produced by VEA Pty Ltd
Chapter 5: The Structure and Function of Large Biological Molecules
 Name Period Chapter 5: The Structure and Function of Large Biological Molecules Concept 5.1 Macromolecules are polymers, built from monomers 1. The large molecules of all living things fall into just four
Name Period Chapter 5: The Structure and Function of Large Biological Molecules Concept 5.1 Macromolecules are polymers, built from monomers 1. The large molecules of all living things fall into just four
Analysis of Vitamin C Using Iodine. Introduction
 Analysis of Vitamin C Using Iodine Introduction Vitamin C (ascorbic acid) is oxidized to dehydroascorbic acid using a mild oxidizing agent such as iodine. The oxidation is a two- electron process, following
Analysis of Vitamin C Using Iodine Introduction Vitamin C (ascorbic acid) is oxidized to dehydroascorbic acid using a mild oxidizing agent such as iodine. The oxidation is a two- electron process, following
BIOLOGICAL MOLECULES OF LIFE
 BIOLOGICAL MOLECULES OF LIFE C A R B O H Y D R A T E S, L I P I D S, P R O T E I N S, A N D N U C L E I C A C I D S The Academic Support Center @ Daytona State College (Science 115, Page 1 of 29) Carbon
BIOLOGICAL MOLECULES OF LIFE C A R B O H Y D R A T E S, L I P I D S, P R O T E I N S, A N D N U C L E I C A C I D S The Academic Support Center @ Daytona State College (Science 115, Page 1 of 29) Carbon
Sugar or Salt? Ionic and Covalent Bonds
 Lab 11 Sugar or Salt? Ionic and Covalent Bonds TN Standard 2.1: The student will investigate chemical bonding. Have you ever accidentally used salt instead of sugar? D rinking tea that has been sweetened
Lab 11 Sugar or Salt? Ionic and Covalent Bonds TN Standard 2.1: The student will investigate chemical bonding. Have you ever accidentally used salt instead of sugar? D rinking tea that has been sweetened
10.1 The function of Digestion pg. 402
 10.1 The function of Digestion pg. 402 Macromolecules and Living Systems The body is made up of more than 60 % water. The water is found in the cells cytoplasm, the interstitial fluid and the blood (5
10.1 The function of Digestion pg. 402 Macromolecules and Living Systems The body is made up of more than 60 % water. The water is found in the cells cytoplasm, the interstitial fluid and the blood (5
Santa Monica College Chemistry 11
 Types of Reactions Objectives The objectives of this laboratory are as follows: To perform and observe the results of a variety of chemical reactions. To become familiar with the observable signs of chemical
Types of Reactions Objectives The objectives of this laboratory are as follows: To perform and observe the results of a variety of chemical reactions. To become familiar with the observable signs of chemical
Cambridge International Examinations Cambridge International General Certificate of Secondary Education
 Cambridge International Examinations Cambridge International General Certificate of Secondary Education *0123456789* CHEMISTRY 0620/03 Paper 3 Theory (Core) For Examination from 2016 SPECIMEN PAPER 1 hour
Cambridge International Examinations Cambridge International General Certificate of Secondary Education *0123456789* CHEMISTRY 0620/03 Paper 3 Theory (Core) For Examination from 2016 SPECIMEN PAPER 1 hour
IDENTIFICATION OF ALCOHOLS
 IDENTIFICATION OF ALCOHOLS Alcohols are organic compounds that which considered as derivatives of water. One of the hydrogen atoms of water molecule (H-O-H) has been replaced by an alkyl or substituted
IDENTIFICATION OF ALCOHOLS Alcohols are organic compounds that which considered as derivatives of water. One of the hydrogen atoms of water molecule (H-O-H) has been replaced by an alkyl or substituted
AN EXPERIMENT IN ALCHEMY: COPPER TO SILVER TO GOLD 2005, 2000, 1996 by David A. Katz. All rights reserved
 AN EXPERIMENT IN ALCHEMY: COPPER TO SILVER TO GOLD 2005, 2000, 1996 by David A. Katz. All rights reserved INTRODUCTION One of the goals of the ancient alchemists was to convert base metals into gold. Although
AN EXPERIMENT IN ALCHEMY: COPPER TO SILVER TO GOLD 2005, 2000, 1996 by David A. Katz. All rights reserved INTRODUCTION One of the goals of the ancient alchemists was to convert base metals into gold. Although
# 12 Condensation Polymerization: Preparation of Two Types of Polyesters
 # 12 Condensation Polymerization: Preparation of Two Types of Polyesters Submitted by: Arturo Contreras, Visiting Scholar, Center for Chemical Education, Miami University, Middletown, OH; 1996 1997. I.
# 12 Condensation Polymerization: Preparation of Two Types of Polyesters Submitted by: Arturo Contreras, Visiting Scholar, Center for Chemical Education, Miami University, Middletown, OH; 1996 1997. I.
Oxidation States of Copper Two forms of copper oxide are found in nature, copper(i) oxide and copper(ii) oxide.
 The Empirical Formula of a Copper Oxide Reading assignment: Chang, Chemistry 10 th edition, pp. 55-58. Goals The reaction of hydrogen gas with a copper oxide compound will be studied quantitatively. By
The Empirical Formula of a Copper Oxide Reading assignment: Chang, Chemistry 10 th edition, pp. 55-58. Goals The reaction of hydrogen gas with a copper oxide compound will be studied quantitatively. By
DYES AND DYEING 2003 by David A. Katz. All rights reserved. Permission for classroom use provided original copyright is included.
 DYES AND DYEING 2003 by David A. Katz. All rights reserved. Permission for classroom use provided original copyright is included. Dyeing of textiles has been practiced for thousands of years with the first
DYES AND DYEING 2003 by David A. Katz. All rights reserved. Permission for classroom use provided original copyright is included. Dyeing of textiles has been practiced for thousands of years with the first
Physical Changes and Chemical Reactions
 Physical Changes and Chemical Reactions Gezahegn Chaka, Ph.D., and Sudha Madhugiri, Ph.D., Collin College Department of Chemistry Objectives Introduction To observe physical and chemical changes. To identify
Physical Changes and Chemical Reactions Gezahegn Chaka, Ph.D., and Sudha Madhugiri, Ph.D., Collin College Department of Chemistry Objectives Introduction To observe physical and chemical changes. To identify
Vitamin C Content of Fruit Juice
 1 Vitamin C Content of Fruit Juice Introduction Vitamin C Vitamins are organic compounds that have important biological functions. For instance, in humans they enable a variety of enzymes in the body to
1 Vitamin C Content of Fruit Juice Introduction Vitamin C Vitamins are organic compounds that have important biological functions. For instance, in humans they enable a variety of enzymes in the body to
Effect of temperature and ph on the enzymatic activity of salivary amylase
 Effect of temperature and ph on the enzymatic activity of salivary amylase Gae Khalil Rodillas, Nonia Carla Ysabel Samson, Raphael Jaime Santos* and Brylle Tabora Department of Biological Sciences, College
Effect of temperature and ph on the enzymatic activity of salivary amylase Gae Khalil Rodillas, Nonia Carla Ysabel Samson, Raphael Jaime Santos* and Brylle Tabora Department of Biological Sciences, College
EXPERIMENT 12: Empirical Formula of a Compound
 EXPERIMENT 12: Empirical Formula of a Compound INTRODUCTION Chemical formulas indicate the composition of compounds. A formula that gives only the simplest ratio of the relative number of atoms in a compound
EXPERIMENT 12: Empirical Formula of a Compound INTRODUCTION Chemical formulas indicate the composition of compounds. A formula that gives only the simplest ratio of the relative number of atoms in a compound
Biology for Science Majors
 Biology for Science Majors Lab 10 AP BIOLOGY Concepts covered Respirometers Metabolism Glycolysis Respiration Anaerobic vs. aerobic respiration Fermentation Lab 5: Cellular Respiration ATP is the energy
Biology for Science Majors Lab 10 AP BIOLOGY Concepts covered Respirometers Metabolism Glycolysis Respiration Anaerobic vs. aerobic respiration Fermentation Lab 5: Cellular Respiration ATP is the energy
Assessment Schedule 2013 Chemistry: Demonstrate understanding of the properties of organic compounds (91391)
 NCEA Level 3 Chemistry (91391) 2013 page 1 of 8 Assessment Schedule 2013 Chemistry: Demonstrate understanding of the properties of organic compounds (91391) Evidence Statement Q Evidence Achievement Achievement
NCEA Level 3 Chemistry (91391) 2013 page 1 of 8 Assessment Schedule 2013 Chemistry: Demonstrate understanding of the properties of organic compounds (91391) Evidence Statement Q Evidence Achievement Achievement
Organic Chemistry Lab Experiment 4 Preparation and Properties of Soap
 Organic Chemistry Lab Experiment 4 Preparation and Properties of Soap Introduction A soap is the sodium or potassium salt of a long-chain fatty acid. The fatty acid usually contains 12 to 18 carbon atoms.
Organic Chemistry Lab Experiment 4 Preparation and Properties of Soap Introduction A soap is the sodium or potassium salt of a long-chain fatty acid. The fatty acid usually contains 12 to 18 carbon atoms.
Biological molecules:
 Biological molecules: All are organic (based on carbon). Monomers vs. polymers: Monomers refer to the subunits that, when polymerized, make up a larger polymer. Monomers may function on their own in some
Biological molecules: All are organic (based on carbon). Monomers vs. polymers: Monomers refer to the subunits that, when polymerized, make up a larger polymer. Monomers may function on their own in some
Name Lab #3: Solubility of Organic Compounds Objectives: Introduction: soluble insoluble partially soluble miscible immiscible
 Lab #3: Solubility of rganic Compounds bjectives: - Understanding the relative solubility of organic compounds in various solvents. - Exploration of the effect of polar groups on a nonpolar hydrocarbon
Lab #3: Solubility of rganic Compounds bjectives: - Understanding the relative solubility of organic compounds in various solvents. - Exploration of the effect of polar groups on a nonpolar hydrocarbon
