Present: Deputy Director of Pharmacy, WUTH (Chair) Lead Pharmacist Medicines Management, WUTH (Secretary)
|
|
|
- Hector Walsh
- 7 years ago
- Views:
Transcription
1 Meeting Notes Meeting Name: Wirral Drug & Therapeutics Panel Date: Wednesday 20 th May 2015 Location: Maternity Conference Room, APH Present: Mrs S Robinson (SRo) Mrs M Charlton (MC) Dr M George (MG) Dr F Newton (FN) Mrs R Pugh (RP) Mrs H Hurley (HH) Mr S Riley (SR) Dr Saket Jalan (SJ) Mrs S Hulse (SH) Lisa Knight (LK) Dianne Atherton (DA) Deputy Director of Pharmacy, WUTH (Chair) Lead Pharmacist Medicines Management, WUTH (Secretary) Consultant Rheumatologist, WUTH GP Partner, Wirral LMC Prescribing Advisor, NHS Wirral Finance Dept, WUTH Senior Prescribing Advisor, NHS Wirral Prescribing Lead, NHS Wirral Team Leader, Pharmacy Clinical Support Services, WUTH Pharmacist, Wirral Community NHS Trust GP NHS Wirral Attending: Mrs Penny Robinson Secretary, WUTH Dr Guy Pritchard (GP) for item 39/15.01 Dr R Dasgupta (RD) for items 39/15.02/03/04 Agenda Item 33/15 Apologies Apologies received: Dr Adrian Hughes, Dr B Hammer, Mrs Pippa Roberts, Dr J Cunniffe, Mrs Naomi Holder, Dr E Sim, Ms Louise Morris, Dr M Van Miert, Ms A Rowlands. Not present no apology sent: Ms M Carroll, Mr S Mwenechanya, Introductions were made around the table and Sue Robinson explained that she was chairing the meeting in the absence of Dr Van Miert due to illness, and as Dr Jalan, the incoming deputy chair, was attending for the first time. Agenda Item 34/15 Conflicts of Interest None Agenda Item 35/15 Minutes of Previous Meeting These were passed as an accurate record. HH noted two people have the same initials; a further initial will be added to differentiate between them. Agenda Item 36/15 Matters arising 36/15.01 WDTP Annual Report The Panel reviewed the WDTP Annual Report for 2014 and was approved. Page 1 of 6
2 It was agreed that the WDTP Gap analysis action plan should be added as an agenda item until all the actions have been completed. 36/15.02 WDTP ToR update ToR were approved but was noted that they may need to be reviewed again once the CCG Governance structure has been reviewed. As the acting chair was absent from the meeting, it was agreed to discuss the re-election of the Chair at the next meeting. On membership, Consultant Anaesthetist, Surgery and Anaesthetics Directorate to be amended to Consultant, WUTH and not include a speciality/directorate or division. Action: remove clinical speciality for Chair of D&T 36/15.03 WDTP appeals process SH presented a short paper outlining the appeals process for Wirral Drug & Therapeutics Panel. The WDTP were asked to approve the process. It was highlighted that it needed to specify who was on the panel and that it should include someone independent from the WDTP. The composition of the appeals panel is to be discussed and agreed at the next meeting. Action: Next meeting of Wirral Drug and Therapeutics to approve appeals process and clarify the appeals panel composition. 36/15.04 WDTP ethical framework The Panel has developed this Ethical Framework to enable it to make fair and consistent decisions which treat patients equally. The purpose of the ethical framework is to support and underpin the decision making processes of the Wirral Drugs and Therapeutics Panel. The WDTP were asked to approve this framework. It was agreed that it was important that confirmation was received from all Panel members that they had read the framework. Action: LM to send out to membership to confirm all not in attendance today have read the Ethical Framework. 36/15.05 WDTP Application form MC has updated the WDTP application form. The guide to completing the form has been moved to the back page and back to top links have been inserted to enable the applicant to move around the document much easier. The WDTP were asked to approve the new template application form. 36/15.06 Management of HIV medicines that are not currently on WUTH formulary Author(s) Marianne Charlton Lead Pharmacist Medicines Mgmt, WUTH There are a number of medicines used to treat HIV that are not currently used at WUTH that have been approved clinically and financially by NHS ENGLAND (NHSE) and are part of the North West HIV Guidelines or as agreed in the HIV Specialist Multi-Disciplinary Team MDT. Any funds spent on these medicines at WUTH are cross charged to NHSE. Options for approving the above HIV medicines onto WUTH formulary: Option 1 Continue with the current D&T application process for HIV medicines that have been evaluated by and are being funded externally through NHSE. Option 2 A report detailing these medicines, their indications and the level of evidence associated with each medicine to be presented to WDTP for consideration and approval. This process would be repeated periodically when new HIV medicines are added to the North West HIV Guidelines. Page 2 of 6
3 Option 3 For these medicines to be placed onto the formulary automatically in a similar way to those medicines that approved by NICE and Cancer Drug Fund medicines. WDTP to be informed of these updates via a report at each meeting. There is a neutral financial impact. There is a neutral governance impact as these medicines are clinically evaluated by NHSE as part of the decision as to whether to fund. The Panel were asked to consider these options. After some discussion, the Panel agreed Option 3. Agenda Item 37/15 Medicines Management Policies & Procedures 37/ Self-Medication Policy (update) Author(s) - Pippa Roberts, Director Medicines Management, Clinical Director Pharmacy Services & Fiona McFarlane, Lead Pharmacist for Education & Training These are the main changes to this policy: Use of patient medicines information card optional Cytotoxics medicines can be self-administered Emphasise that Patients on Parkinson s Disease drugs should self-medicate these medicines if at all possible The Panel were asked to approve these changes. The Panel approved the changes. 37/ Controlled Drugs Policy (update) Author(s) - Amy Janvier, Lead Clinical Dispensary Pharmacist These are the main changes to this policy: Tramadol legally re-classified as a schedule 3 CD Cerner has replaced PCIS as Trust s electronic prescribing system Job title of Accountable Officer has changed 1 References to the Controlled Drugs (supervision of Management and Use ) Regulations 2013 added 5.2 Pharmacy led CD top-up of wards and departments occurs on a Tuesday 5.5 Change to PCA contents 5.6 Addition of information about epidurals 7.6 Addition of PODs should be stored on a separate shelf 8.1 Midazolam buccal liquid to be treated as a full CD 9.10 Removed as laboratory no longer holds a Home Office licence to hold schedule 1 CDs 10 Addition of information about witnessing a continuous CD infusion. Addition of trained CSWs can witness administration of schedule 3 CDs 11.2 Addition of pharmacy technicians can remove CDs for destruction from a ward /department 14.2 Changed requirements for a pharmacist to be involved for the transfer of CDs in a ward move The Panel were asked to approve these changes. The Panel approved these changes Agenda Item 38/15 Reports back None Agenda Item 39/15 New Medicine requests 39/15.01 Infliximab, adalimumab and golimumab for treating moderately to severely active ulcerative colitis after the failure of conventional therapy (NICE TA329) Author: Dr Guy Pritchard, Consultant Gastroenterologist, WUTH Dr Guy Pritchard presented his case for infliximab, adalimumab and golimumab, which are recommended, within their marketing authorisations, as options for treating moderately to severely active ulcerative colitis Page 3 of 6
4 in adults whose disease has responded inadequately to conventional therapy including corticosteroids and mercaptopurine or azathioprine, or who cannot tolerate, or have medical contraindications for, such therapies as per NICE TA 329. GP was asked how they would choose which agent the patients will receive. He commented that there are no head to head studies between the agents and the individual trial outcomes are comparable. Patients who had already had infliximab for rescue therapy would go on to receive infliximab again. If not they would choose between adalimumab or golimumab as more cost effective than infliximab. GP commented that he favours golimumab as a monthly injection (vs twice monthly with adalimumab) and there is anecdotal evidence that patients find the injection less painful than adalimumab. When asked what they do when patient doesn t respond to one anti-tnf,gp confirmed that they would swap patient on to an alternative anti-tnf. However, this may change when the NICE TA for vedolizumab is published. GP also said that he would consider using biosimilars (if available and licensed for this indication) at a later date if they were to be introduced into the Trust RAG status:red 39/15.02 Dabigatran for the treatment and secondary prevention of VTE (NICE TA327) Author: Dr Dasgupta, Consultant Haematologist, WUTH Dabigatran etexilate is recommended as a possible treatment for adults with deep vein thrombosis or pulmonary embolism as per NICE TA 327. Primary care and WUTH dabigitran checklists have been amended to include this indication. RD commented that he will write some guidance on the advantages and disadvantages of the different NOACS to help clinicians decide which agent to select but is waiting until the NICE TA for apixiban for the treatment and secondary prevention of VTE is published.. Updated checklists approved Rag status: Amber 39/15.03 Rituximab Auto-immune haemolytic anaemia Author: Dr Dasgupta, Consultant Haematologist, WUTH Proposed position in therapy: 1 st Line treatment: Corticosteroids 2 nd Line treatment: Splenectomy or Rituximab (if unsuitable for splenectomy) 3 rd Line treatment: Rituximab if patients refractory to prior splenectomy RD stated that 30-40% of patients don t respond to steroids and some of these patients are not suitable for splenectomy so rituximab would be an option. When asked what other options there are, RD stated that IVIGs don t work for this condition and other immunosuppressants e.g azathioprine take a long time to work and are only effective in 30-40% of patients. RD stated that the response may last for a year and if they relapse then they would consider repeating the course. If a patient has not had a response after 4 doses they would not repeat the course. They have used rituximab in 2 patients so far and have had good responses. Other CCG commissioning bodies include rituximab for this indication.. Rag status: Red 39/15.04 Darbopoetin for treatment of anaemia in patients with cancer having chemotherapy (NICE TA323) Author: Dr Dasgupta. Consultant Haematologist, WUTH Haematology would use in patients with non-myeloid malignancy undergoing chemotherapy as per the NICE TA. RD presented his application to the Panel and stated that historically darbepoetin has been used as it is the easiest option for the patient, requiring one injection per week initially, reducing to once every 2 or 3 weeks and it is used for other indications within haematology. There are cheaper options, however, these are given three times a week so need to consider nursing administration costs on top of drug costs. SR asked Page 4 of 6
5 how patients self-administer vs nurse administration. RD did not have exact figures but speculated that about 50% of patients will self-administer.. The Panel noted that NICE TA 323 states that if different erythropoiesisstimulating agents are equally suitable, the product with the lowest acquisition cost should be used. The panel requested that the haematologists review the suitability of the different erythropoiesis-stimulating agents (including biosimilars) for this indication and the other indications that they use these agents for within haematology to ensure that darbopoetin is currently the most cost-effective preparation available for these indications and report back the review within 6 months. Rag status: Red Agenda Item 40/15 Chairs Actions The Panel noted and approved the list of Chairs actions. MC added one further drug, Cobicistat for HIV for an individual patient, from Dr Gokhale which is not NHSE commissioned but has been recommended by the Liverpool HIV MDT. The panel approved this drug for this patient. Agenda Item 41/15 Health Treatment Panel applications None Agenda Item 42/15 Unlicensed Medicine updates MC explained to the Panel the current supply problems with Ethanolamine used infrequently by gastroenterologists for bleeding oesophageal varices. The stock WUTH holds expired in March 2015, on a 5 year lifespan, and further stock, from a new supplier, is not expected until July. There is an unlicensed version available from America, however the cost is substantially higher and the Trust would be left with unlicensed stock when the licensed stock becomes available again. No other trusts have stock of the licensed or unlicensed version. There are 3 options with how the supply problem can be managed:- Use the out of date stock until new stock arrives Don t stock it until licensed comes back in Use unlicensed product from America The panel discussed the risks of each option but felt that it should be escalated higher up in the Trust for a decision as Panel members felt they didn t have the knowledge on the legal aspects of the different options. Action: SR to escalate Agenda Item 43/15 NICE Guidance New NICE TAs published since last WDTP meeting: TA 335 Rivaroxaban for acute management of ACS TA 336 Empagliflozin for type 2 diabetes TA 337 Rifamixin for hepatic encephalopathy These were approved to be added to the formulary restricted to relevant consultants until application forms have been submitted to WDTP for positioning. MC discussed TA 338 Pomalidomide for multiple myeloma which was a negative TA. However, it is used in the Trust for this indication as is currently funded by CDF and Trust will continue to use unless CDF withdraw funding. Page 5 of 6
6 Agenda Item 45/15 Cancer Drug Fund Medicines A list of all the current Cancer drugs in use at WUTH was distributed with the papers for information. Agenda Item 46/15 Clinical Trials A list of all the current clinical trials was distributed with the papers for information. Agenda Item 47/15 Minutes from other Committees for noting 47/ Medicine Clinical Guidelines Team 5 th December 2014 & 13 th February / Wirral Wide Wound Formulary Group None 47/ Chemotherapy Steering Group 5 th February 2015 & 9 th April / Medical Gas Team 14 th January / Non Medical Prescribing Team 6th November 2014 & 22nd January /15.06 Antimicrobial Stewardship Team - None Agenda Item 48/15 any other business There was no other business. Future Meeting Dates 49/15 Next meeting 15 th July pm in Boardroom Education Centre, APH (papers for submission needed by 12 th June 2015) Page 6 of 6
HERTFORDSHIRE MEDICINES MANAGEMENT COMMITTEE (HMMC) RIVAROXABAN RECOMMENDED see specific recommendations for licensed indications below
 Name: generic (trade) Rivaroxaban (Xarelto ) HERTFORDSHIRE MEDICINES MANAGEMENT COMMITTEE (HMMC) RIVAROXABAN RECOMMENDED see specific recommendations for licensed indications below What it is Indications
Name: generic (trade) Rivaroxaban (Xarelto ) HERTFORDSHIRE MEDICINES MANAGEMENT COMMITTEE (HMMC) RIVAROXABAN RECOMMENDED see specific recommendations for licensed indications below What it is Indications
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE. Multiple Technology Appraisal
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Multiple Technology Appraisal Infliximab, adalimumab and golimumab for treating moderately to severely active ulcerative colitis after the failure of conventional
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Multiple Technology Appraisal Infliximab, adalimumab and golimumab for treating moderately to severely active ulcerative colitis after the failure of conventional
Rivaroxaban: Prescribing Guidance for the treatment of provoked venous thromboembolism (VTE)
 Rivaroxaban: Prescribing Guidance for the treatment of provoked venous thromboembolism (VTE) Amber Drug Level 2 Leeds We have started your patient on rivaroxaban for the treatment of provoked VTE (deep
Rivaroxaban: Prescribing Guidance for the treatment of provoked venous thromboembolism (VTE) Amber Drug Level 2 Leeds We have started your patient on rivaroxaban for the treatment of provoked VTE (deep
New Oral Anticoagulants. How safe are they outside the trials?
 New Oral Anticoagulants How safe are they outside the trials? Objectives The need for anticoagulant therapy Indications for anticoagulation Traditional anticoagulant therapies Properties of new oral anticoagulants
New Oral Anticoagulants How safe are they outside the trials? Objectives The need for anticoagulant therapy Indications for anticoagulation Traditional anticoagulant therapies Properties of new oral anticoagulants
Service Specification Template Department of Health, updated June 2015
 Service Specification Template Department of Health, updated June 2015 Service Specification No. : 2 Service: Commissioner Lead: Provider Lead: Period: Anti-coagulation monitoring Date of Review: 31 st
Service Specification Template Department of Health, updated June 2015 Service Specification No. : 2 Service: Commissioner Lead: Provider Lead: Period: Anti-coagulation monitoring Date of Review: 31 st
Joint Formulary Management Group (FMG) Terms of Reference Version.2
 Joint Formulary Management Group (FMG) Terms of Reference Version.2 Approved by the DTC and APC Date of approval: September 203 Review date: September 205 BACKGROUND The Formulary Management Group (FMG)
Joint Formulary Management Group (FMG) Terms of Reference Version.2 Approved by the DTC and APC Date of approval: September 203 Review date: September 205 BACKGROUND The Formulary Management Group (FMG)
Indication: Indication: Protaxos is indicated in adults.
 Maklumat tambahan indikasi untuk upload pada laman web Year 2013 Products Approved For Additional Indication (DCA 262 28 Mac 2013) NO PRODUCT (ACTIVE INGREDIENT) 1. 1.1 EVISTA TABLET 60MG [ Raloxifene
Maklumat tambahan indikasi untuk upload pada laman web Year 2013 Products Approved For Additional Indication (DCA 262 28 Mac 2013) NO PRODUCT (ACTIVE INGREDIENT) 1. 1.1 EVISTA TABLET 60MG [ Raloxifene
Dabigatran etexilate for the treatment and secondary prevention of deep vein thrombosis and/or pulmonary embolism ERRATUM
 Dabigatran etexilate for the treatment and secondary prevention of deep vein thrombosis and/or pulmonary embolism ERRATUM This report was commissioned by the NIHR HTA Programme as project number 12/78
Dabigatran etexilate for the treatment and secondary prevention of deep vein thrombosis and/or pulmonary embolism ERRATUM This report was commissioned by the NIHR HTA Programme as project number 12/78
Technology Appraisal Committee Meeting (Committee A)
 Technology Appraisal Committee Meeting (Committee A) Minutes: Date and Time: Venue: Confirmed Thursday 28 th May 2015, 10.00am 5.00pm Prospero House 241 Borough High Street London SE1 1GA Present: Dr Jane
Technology Appraisal Committee Meeting (Committee A) Minutes: Date and Time: Venue: Confirmed Thursday 28 th May 2015, 10.00am 5.00pm Prospero House 241 Borough High Street London SE1 1GA Present: Dr Jane
Rivaroxaban to prevent blood clots for patients who have a lower limb plaster cast. Information for patients Pharmacy
 Rivaroxaban to prevent blood clots for patients who have a lower limb plaster cast Information for patients Pharmacy Your doctor has prescribed a tablet called rivaroxaban. This leaflet tells you about
Rivaroxaban to prevent blood clots for patients who have a lower limb plaster cast Information for patients Pharmacy Your doctor has prescribed a tablet called rivaroxaban. This leaflet tells you about
Minutes. August 19 th 2014, 12:30-2:30 pm Pharmacy Dept. CMFT
 Minutes August 19 th 2014, 12:30-2:30 pm Pharmacy Dept. CMFT Present: Elizabeth Arkell (EA), Associate Director of Pharmacy, UHSM Foundation Trust Jennifer Bartlett (JB) Senior Medicines Management Pharmacist
Minutes August 19 th 2014, 12:30-2:30 pm Pharmacy Dept. CMFT Present: Elizabeth Arkell (EA), Associate Director of Pharmacy, UHSM Foundation Trust Jennifer Bartlett (JB) Senior Medicines Management Pharmacist
Executive Summary. Motive for the request for advice
 Executive Summary Motive for the request for advice Currently nearly 400,000 people in the Netherlands are being treated with anticoagulants of a type Vitamin K antagonists (VKAs). Although VKAs are very
Executive Summary Motive for the request for advice Currently nearly 400,000 people in the Netherlands are being treated with anticoagulants of a type Vitamin K antagonists (VKAs). Although VKAs are very
Enoxaparin for long term anticoagulation in patients unsuitable for oral anticoagulants
 Enoxaparin for long term anticoagulation in patients unsuitable for oral anticoagulants Traffic light classification- Amber 2 Information sheet for Primary Care Prescribers Relevant Licensed Indications
Enoxaparin for long term anticoagulation in patients unsuitable for oral anticoagulants Traffic light classification- Amber 2 Information sheet for Primary Care Prescribers Relevant Licensed Indications
Anticoagulation at the end of life. Rhona Maclean Rhona.maclean@sth.nhs.uk
 Anticoagulation at the end of life Rhona Maclean Rhona.maclean@sth.nhs.uk Content Anticoagulant Therapies Indications for anticoagulation Venous thromboembolism (VTE) Atrial Fibrillation Mechnical Heart
Anticoagulation at the end of life Rhona Maclean Rhona.maclean@sth.nhs.uk Content Anticoagulant Therapies Indications for anticoagulation Venous thromboembolism (VTE) Atrial Fibrillation Mechnical Heart
Title Use of rivaroxaban in suspected DVT in the Emergency Department Standard Operating Procedure. Author s job title. Pharmacist.
 Document Control Title Use of rivaroxaban in suspected DVT in the Emergency Department Author Pharmacist Directorate Clinical Support Services Version Date Issued Status 0.1 Oct Draft 2015 1.0 Dec Final
Document Control Title Use of rivaroxaban in suspected DVT in the Emergency Department Author Pharmacist Directorate Clinical Support Services Version Date Issued Status 0.1 Oct Draft 2015 1.0 Dec Final
CDEC RECORD OF ADVICE
 CDEC RECORD OF ADVICE RIVAROXABAN (Xarelto Bayer Inc.) Indication: Deep Vein Thrombosis Without Symptomatic Pulmonary Embolism This document summarizes the Canadian Drug Expert Committee (CDEC) response
CDEC RECORD OF ADVICE RIVAROXABAN (Xarelto Bayer Inc.) Indication: Deep Vein Thrombosis Without Symptomatic Pulmonary Embolism This document summarizes the Canadian Drug Expert Committee (CDEC) response
Patient Group Direction Hospital: Bristol Royal Infirmary Department: UHBristol Thrombosis Service University Hospitals Bristol NHS Foundation Trust.
 Patient Group Direction Hospital: Bristol Royal Infirmary Department: UHBristol Thrombosis Service University Hospitals Bristol NHS Foundation Trust. This Patient Group Direction (PGD) has been written
Patient Group Direction Hospital: Bristol Royal Infirmary Department: UHBristol Thrombosis Service University Hospitals Bristol NHS Foundation Trust. This Patient Group Direction (PGD) has been written
Shared care protocol for the management of patients with Rheumatoid Arthritis treated with disease modifying antirheumatic drugs (DMARDs)
 Tameside Hospital NHS Foundation Trust and NHS Tameside and Glossop Shared care protocol for the management of patients with Rheumatoid Arthritis treated with disease modifying antirheumatic drugs (DMARDs)
Tameside Hospital NHS Foundation Trust and NHS Tameside and Glossop Shared care protocol for the management of patients with Rheumatoid Arthritis treated with disease modifying antirheumatic drugs (DMARDs)
National Guidance and New Protocols
 National Guidance and New Protocols Dr Jane Strong Consultant Haematologist DVT clinical lead Acute VTE chair DAWN AC Twentieth User Group Meeting 8 th October 2012 DVT patient pathway Assessment Diagnosis
National Guidance and New Protocols Dr Jane Strong Consultant Haematologist DVT clinical lead Acute VTE chair DAWN AC Twentieth User Group Meeting 8 th October 2012 DVT patient pathway Assessment Diagnosis
Committee Approval Date: September 12, 2014 Next Review Date: September 2015
 Medication Policy Manual Policy No: dru361 Topic: Pradaxa, dabigatran Date of Origin: September 12, 2014 Committee Approval Date: September 12, 2014 Next Review Date: September 2015 Effective Date: November
Medication Policy Manual Policy No: dru361 Topic: Pradaxa, dabigatran Date of Origin: September 12, 2014 Committee Approval Date: September 12, 2014 Next Review Date: September 2015 Effective Date: November
GENERAL PRACTICE BASED PHARMACIST
 GENERAL PRACTICE BASED PHARMACIST JOB PURPOSE Provide expertise in clinical medicines review 1 and address public health and social needs of patients in GP practices Reduce inappropriate poly-pharmacy
GENERAL PRACTICE BASED PHARMACIST JOB PURPOSE Provide expertise in clinical medicines review 1 and address public health and social needs of patients in GP practices Reduce inappropriate poly-pharmacy
Trust Guideline for Thromboprophylaxis in Trauma and Orthopaedic Inpatients
 A clinical guideline recommended for use In: By: For: Key words: Department of Orthopaedics, NNUHT Medical staff Trauma & Orthopaedic Inpatients Deep vein thrombosis, Thromboprophylaxis, Orthopaedic Surgery
A clinical guideline recommended for use In: By: For: Key words: Department of Orthopaedics, NNUHT Medical staff Trauma & Orthopaedic Inpatients Deep vein thrombosis, Thromboprophylaxis, Orthopaedic Surgery
Pharmacy Department. Rotational Pharmacy Technician. Information Pack
 Pharmacy Department Rotational Pharmacy Technician Information Pack 1 Contents 1. Chelsea and Westminster Healthcare NHS Foundation Trust 3 2. The Pharmacy 4 3. Services 4 3.1. Clinical Services 4 3.2.
Pharmacy Department Rotational Pharmacy Technician Information Pack 1 Contents 1. Chelsea and Westminster Healthcare NHS Foundation Trust 3 2. The Pharmacy 4 3. Services 4 3.1. Clinical Services 4 3.2.
NICE TA 275: Apixaban for the prevention of stroke and systemic embolism in people with non-valvular atrial fibrillation
 Service Notification in response to DHSSPS endorsed NICE Technology Appraisals NICE TA 275: Apixaban for the prevention of stroke and systemic embolism in people with non-valvular atrial fibrillation 1
Service Notification in response to DHSSPS endorsed NICE Technology Appraisals NICE TA 275: Apixaban for the prevention of stroke and systemic embolism in people with non-valvular atrial fibrillation 1
Inpatient Anticoagulation Safety. To provide safe and effective anticoagulation therapy through a collaborative approach.
 Inpatient Anticoagulation Safety Purpose: Policy: To provide safe and effective anticoagulation therapy through a collaborative approach. Upon the written order of a physician, Heparin, Low Molecular Weight
Inpatient Anticoagulation Safety Purpose: Policy: To provide safe and effective anticoagulation therapy through a collaborative approach. Upon the written order of a physician, Heparin, Low Molecular Weight
Breakfast symposium: From hospital to home - the focus on the patient
 Breakfast symposium: From hospital to home - the focus on the patient Nadya Hamedi DARZI Fellow UCLPartners and Barts Health NHS Trust in collaboration with North Central London Local Pharmaceutical Committee
Breakfast symposium: From hospital to home - the focus on the patient Nadya Hamedi DARZI Fellow UCLPartners and Barts Health NHS Trust in collaboration with North Central London Local Pharmaceutical Committee
GLOUCESTERSHIRE HOSPITALS NHS FOUNDATION TRUST
 GLOUCESTERSHIRE HOSPITALS NHS FOUNDATION TRUST MEDICINES MANAGEMENT STRATEGY 2006/07 ANNUAL REPORT 1. Aim An annual report on the Trust s Medicines Management Strategy is part of the requirements for Standards
GLOUCESTERSHIRE HOSPITALS NHS FOUNDATION TRUST MEDICINES MANAGEMENT STRATEGY 2006/07 ANNUAL REPORT 1. Aim An annual report on the Trust s Medicines Management Strategy is part of the requirements for Standards
Drug name: RIVAROXABAN (Xarelto ) For the treatment of acute venous thromboembolism in adult patients.
 Drug name: RIVAROXABAN (Xarelto ) For the treatment of acute venous thromboembolism in adult patients. DOCUMENT TO BE SCANNED INTO ELECTRONIC RECORDS AS AND FILED IN NOTES Patient Name : Date of Birth:
Drug name: RIVAROXABAN (Xarelto ) For the treatment of acute venous thromboembolism in adult patients. DOCUMENT TO BE SCANNED INTO ELECTRONIC RECORDS AS AND FILED IN NOTES Patient Name : Date of Birth:
NHS FORTH VALLEY RIVAROXABAN AS TREATMENT FOR DEEP VEIN THROMBOSIS AND PULMONARY EMBOLISM IN ADULTS
 NHS FORTH VALLEY RIVAROXABAN AS TREATMENT FOR DEEP VEIN THROMBOSIS AND PULMONARY EMBOLISM IN ADULTS Date of First Issue 01/12/ 2012 Approved 15/11/2012 Current Issue Date 29/10/2014 Review Date 29/10/2016
NHS FORTH VALLEY RIVAROXABAN AS TREATMENT FOR DEEP VEIN THROMBOSIS AND PULMONARY EMBOLISM IN ADULTS Date of First Issue 01/12/ 2012 Approved 15/11/2012 Current Issue Date 29/10/2014 Review Date 29/10/2016
Medical Insurance Long Term (chronic) Conditions Explained
 Medical Insurance Long Term (chronic) Conditions Explained October 2013 FS 28452 wpa.org.uk EMS 505226 IS 553152 Introduction This leaflet explains how we manage claims for our policyholders whose medical
Medical Insurance Long Term (chronic) Conditions Explained October 2013 FS 28452 wpa.org.uk EMS 505226 IS 553152 Introduction This leaflet explains how we manage claims for our policyholders whose medical
National Guidance and New Protocols
 National Guidance and New Protocols Dr Jane Strong Consultant Haematologist DVT clinical lead Acute VTE chair DAWN AC Twentieth User Group Meeting 8 th October 2012 Autumn dawn Restless geese take flight
National Guidance and New Protocols Dr Jane Strong Consultant Haematologist DVT clinical lead Acute VTE chair DAWN AC Twentieth User Group Meeting 8 th October 2012 Autumn dawn Restless geese take flight
How To Be A Senior Pharmacy Technician
 JOB DESCRIPTION JOB TITLE : Senior Pharmacy Technician DEPARTMENT : Pharmacy Heartlands, Solihull & Good Hope Hospitals GRADE : Band 5 HOURS OF DUTY : 37.5 hours per week. The Trust operates a 7 day working
JOB DESCRIPTION JOB TITLE : Senior Pharmacy Technician DEPARTMENT : Pharmacy Heartlands, Solihull & Good Hope Hospitals GRADE : Band 5 HOURS OF DUTY : 37.5 hours per week. The Trust operates a 7 day working
East & South East England Specialist Pharmacy Services Medicines Use and Safety Division Community Health Services Transcribing
 East & outh East England pecialist Pharmacy ervices East of England, London, outh Central & outh East Coast Medicines Use and afety Division Community Health ervices Transcribing Guidance to support the
East & outh East England pecialist Pharmacy ervices East of England, London, outh Central & outh East Coast Medicines Use and afety Division Community Health ervices Transcribing Guidance to support the
PRESCRIBING OF NHS MEDICATION RECOMMENDED DURING OR AFTER A PRIVATE EPISODE OF CARE
 East Surrey CCG Guildford & Waverley CCG North West Surrey CCG Surrey Downs CCG Surrey Heath CCG PRESCRIBING OF NHS MEDICATION RECOMMENDED DURING OR AFTER A PRIVATE EPISODE OF CARE Version: 2.2 Name of
East Surrey CCG Guildford & Waverley CCG North West Surrey CCG Surrey Downs CCG Surrey Heath CCG PRESCRIBING OF NHS MEDICATION RECOMMENDED DURING OR AFTER A PRIVATE EPISODE OF CARE Version: 2.2 Name of
NWMIC Medicines FAQ. New oral anticoagulants (NOACs) and management of dental patients - Dabigatran, rivaroxaban and apixaban.
 NWMIC Medicines FAQ New oral anticoagulants (NOACs) and management of dental patients - Date prepared: May 2013, updated November 2013 Summary In primary care; Consider liaising with the local hospital
NWMIC Medicines FAQ New oral anticoagulants (NOACs) and management of dental patients - Date prepared: May 2013, updated November 2013 Summary In primary care; Consider liaising with the local hospital
UCB. Certolizumab pegol (CIMZIA ) for the treatment of Rheumatoid Arthritis PATIENT ACCESS SCHEME (PAS) SUBMISSION TO NICE
 UCB Certolizumab pegol (CIMZIA ) for the treatment of Rheumatoid Arthritis PATIENT ACCESS SCHEME (PAS) SUBMISSION TO NICE July 23 d 2009 1 Executive summary UCB have proposed a patient access scheme (PAS)
UCB Certolizumab pegol (CIMZIA ) for the treatment of Rheumatoid Arthritis PATIENT ACCESS SCHEME (PAS) SUBMISSION TO NICE July 23 d 2009 1 Executive summary UCB have proposed a patient access scheme (PAS)
TA 256: Rivaroxaban for the prevention of stroke and systemic embolism in people with atrial fibrillation
 Service Notification in response to DHSSPS endorsed NICE Technology Appraisals TA 256: Rivaroxaban for the prevention of stroke and systemic embolism in people with atrial fibrillation 1 Name of Commissioning
Service Notification in response to DHSSPS endorsed NICE Technology Appraisals TA 256: Rivaroxaban for the prevention of stroke and systemic embolism in people with atrial fibrillation 1 Name of Commissioning
Introduction. Background to this event. Raising awareness 09/11/2015
 Introduction Primary Care Medicines Governance HSCB Background to this event New class of medicines Availability of training Increasing volume of prescriptions Reports of medication incidents Raising awareness
Introduction Primary Care Medicines Governance HSCB Background to this event New class of medicines Availability of training Increasing volume of prescriptions Reports of medication incidents Raising awareness
adult services between 1 January and 31 December 2013 had an operation where the
 Organisational audit: round 4 Adult services audit tool Section 1: Demographics DEM 1.1 How many IBD patients does your service manage? DEM 1.2 Is this figure: an estimate (enter e ) or from a database/register
Organisational audit: round 4 Adult services audit tool Section 1: Demographics DEM 1.1 How many IBD patients does your service manage? DEM 1.2 Is this figure: an estimate (enter e ) or from a database/register
The Oxford IBD Service
 Gastroenterology Unit, John Radcliffe Hospital The Oxford IBD Service Information for patients Information for patients 1 The Oxford Inflammatory Bowel Disease (IBD) Service Consultant Gastroenterologists
Gastroenterology Unit, John Radcliffe Hospital The Oxford IBD Service Information for patients Information for patients 1 The Oxford Inflammatory Bowel Disease (IBD) Service Consultant Gastroenterologists
Uncontrolled when printed. Version 1.1. Acute Sector. Lead Author/Co-ordinator: Mr Simon Barker Consultant Orthopaedic Surgeon Julie Fraser
 Acute Sector NHS Grampian Staff Local Treatment Protocol For Venous Thromoboembolic Prophylaxis Using Rivaroxaban 10mg Tablets In Adult Patients Undergoing Elective Hip Or Knee Replacement Surgery. Lead
Acute Sector NHS Grampian Staff Local Treatment Protocol For Venous Thromoboembolic Prophylaxis Using Rivaroxaban 10mg Tablets In Adult Patients Undergoing Elective Hip Or Knee Replacement Surgery. Lead
Oxford Anticoagulation & Thrombosis Service Deep Vein Thrombosis (DVT) and Pulmonary Embolism (PE)
 Oxford University Hospitals NHS Trust Oxford Anticoagulation & Thrombosis Service Deep Vein Thrombosis (DVT) and Pulmonary Embolism (PE) Information for people with a blood clot (thrombus) What is this
Oxford University Hospitals NHS Trust Oxford Anticoagulation & Thrombosis Service Deep Vein Thrombosis (DVT) and Pulmonary Embolism (PE) Information for people with a blood clot (thrombus) What is this
Summary of the role and operation of NHS Research Management Offices in England
 Summary of the role and operation of NHS Research Management Offices in England The purpose of this document is to clearly explain, at the operational level, the activities undertaken by NHS R&D Offices
Summary of the role and operation of NHS Research Management Offices in England The purpose of this document is to clearly explain, at the operational level, the activities undertaken by NHS R&D Offices
How To Prepare A Meeting For A Health Care Conference
 THORACIC ONCOLOGY MULTIDISCIPLINARY TEAM MEETINGS: OPERATIONAL POLICY EXECUTIVE SUMMARY 1. All patients in Lothian with thoracic malignancies should be discussed at designated times in pathway (see App
THORACIC ONCOLOGY MULTIDISCIPLINARY TEAM MEETINGS: OPERATIONAL POLICY EXECUTIVE SUMMARY 1. All patients in Lothian with thoracic malignancies should be discussed at designated times in pathway (see App
NATIONAL PROFILES FOR PHARMACY CONTENTS
 NATIONAL PROFILES FOR PHARMACY CONTENTS Profile Title AfC Banding Page Pharmacy Support Worker 2 2 Pharmacy Support Worker Higher Level Pharmacy Technician 4 4 Pharmacy Technician Higher Level (Pharmacy
NATIONAL PROFILES FOR PHARMACY CONTENTS Profile Title AfC Banding Page Pharmacy Support Worker 2 2 Pharmacy Support Worker Higher Level Pharmacy Technician 4 4 Pharmacy Technician Higher Level (Pharmacy
Updates to the Alberta Human Services Drug Benefit Supplement
 Updates to the Alberta Human Services Drug Benefit Supplement Effective January 1, 2016 Inquiries should be directed to: Pharmacy Services Alberta Blue Cross 10009 108 Street NW Edmonton AB T5J 3C5 Telephone
Updates to the Alberta Human Services Drug Benefit Supplement Effective January 1, 2016 Inquiries should be directed to: Pharmacy Services Alberta Blue Cross 10009 108 Street NW Edmonton AB T5J 3C5 Telephone
Title of Guideline. Thrombosis Pharmacist)
 Title of Guideline Contact Name and Job Title (author) Guideline for patients receiving Rivaroxaban (Xarelto ) requiring Emergency Surgery or treatment for Haemorrhage Julian Holmes (Haemostasis and Thrombosis
Title of Guideline Contact Name and Job Title (author) Guideline for patients receiving Rivaroxaban (Xarelto ) requiring Emergency Surgery or treatment for Haemorrhage Julian Holmes (Haemostasis and Thrombosis
Pathway for the management of DVT in primary Care
 Pathway for the management of DVT in primary Care Final Version: Approved by NHS DGS CCG: June 2015 This document aims to support practices in DGS CCG in the Management & Treatment of patients with suspected
Pathway for the management of DVT in primary Care Final Version: Approved by NHS DGS CCG: June 2015 This document aims to support practices in DGS CCG in the Management & Treatment of patients with suspected
Updates to the Alberta Drug Benefit List. Effective January 1, 2016
 Updates to the Alberta Drug Benefit List Effective January 1, 2016 Inquiries should be directed to: Pharmacy Services Alberta Blue Cross 10009 108 Street NW Edmonton AB T5J 3C5 Telephone Number: (780)
Updates to the Alberta Drug Benefit List Effective January 1, 2016 Inquiries should be directed to: Pharmacy Services Alberta Blue Cross 10009 108 Street NW Edmonton AB T5J 3C5 Telephone Number: (780)
Current Rheumatoid Arthritis Treatment Options: Update for Managed Care and Specialty Pharmacists
 Current Rheumatoid Arthritis Treatment Options: Update for Managed Care and Specialty Pharmacists 1. Which of the following matches of biologic targets that contribute to rheumatoid arthritis (RA) and
Current Rheumatoid Arthritis Treatment Options: Update for Managed Care and Specialty Pharmacists 1. Which of the following matches of biologic targets that contribute to rheumatoid arthritis (RA) and
New Anticoagulants and GI bleeding
 New Anticoagulants and GI bleeding DR DANNY MYERS MD FRCP(C) CLINICAL ASSISTANT PROFESSOR OF MEDICINE, UBC Conflicts of Interest None I am unbiased in the use of NOAC s vs Warfarin based on risk benefit
New Anticoagulants and GI bleeding DR DANNY MYERS MD FRCP(C) CLINICAL ASSISTANT PROFESSOR OF MEDICINE, UBC Conflicts of Interest None I am unbiased in the use of NOAC s vs Warfarin based on risk benefit
Job Description. Professionally accountable to the Medical Director with respect to Trust-wide Medicines Optimisation.
 Job Description JOB DETAILS Job Title: Chief of Pharmacy Band: 9 Hours: 37.5 Department / Ward: Directorate: Pharmacy Cross Site Central Clinical Services ORGANISATIONAL ARRANGEMENTS Operationally accountable
Job Description JOB DETAILS Job Title: Chief of Pharmacy Band: 9 Hours: 37.5 Department / Ward: Directorate: Pharmacy Cross Site Central Clinical Services ORGANISATIONAL ARRANGEMENTS Operationally accountable
Rivaroxaban (Xarelto) for preventing blood clots after hip or knee replacement surgery
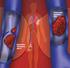 medicineupdate Asking the right questions about new medicines Rivaroxaban (Xarelto) for preventing blood clots after hip or knee replacement surgery This Medicine Update is for people who have been prescribed
medicineupdate Asking the right questions about new medicines Rivaroxaban (Xarelto) for preventing blood clots after hip or knee replacement surgery This Medicine Update is for people who have been prescribed
Humulin R (U500) insulin: Prescribing Guidance
 Leeds Humulin R (U500) insulin: Prescribing Guidance Amber Drug Level 2 We have started your patient on Humulin R (U500) insulin for the treatment of diabetic patients with marked insulin resistance requiring
Leeds Humulin R (U500) insulin: Prescribing Guidance Amber Drug Level 2 We have started your patient on Humulin R (U500) insulin for the treatment of diabetic patients with marked insulin resistance requiring
Appendix 3 INDIVIDUAL PATIENT DRUG TREATMENT. POLICY AND PROCESS FOR DECISION MAKING September 2007
 Appendix 3 INDIVIDUAL PATIENT DRUG TREATMENT POLICY AND PROCESS FOR DECISION MAKING September 2007 Approved by Board: 6 th September 2007 Date Implemented: 1 st October 2007 Review Date: September 2008
Appendix 3 INDIVIDUAL PATIENT DRUG TREATMENT POLICY AND PROCESS FOR DECISION MAKING September 2007 Approved by Board: 6 th September 2007 Date Implemented: 1 st October 2007 Review Date: September 2008
Formulary notes for the List of Approved Medicines
 Formulary notes for the List of Approved Medicines Last updated May 2016 Contents Introduction... 2 Formulary notes... 2 Generic listing of medicines... 2 PBS items... 2 Restrictions... 3 Applications
Formulary notes for the List of Approved Medicines Last updated May 2016 Contents Introduction... 2 Formulary notes... 2 Generic listing of medicines... 2 PBS items... 2 Restrictions... 3 Applications
Preventing Blood Clots in Adult Patients. Information For Patients
 Preventing Blood Clots in Adult Patients Information For Patients 1 This leaflet will give you information on how to reduce the risk of developing blood clots during and after your stay in hospital. If
Preventing Blood Clots in Adult Patients Information For Patients 1 This leaflet will give you information on how to reduce the risk of developing blood clots during and after your stay in hospital. If
NSQHS Standard 1 Governance
 NSQHS Standard 1 Governance Definitions sheet Governance Audit Tools Definitions Contents 1. Open Disclosure Program Page 1 2. ACUTE Clinical Record Audit Tools Page 2 -----------------------------------------------------------------------------------
NSQHS Standard 1 Governance Definitions sheet Governance Audit Tools Definitions Contents 1. Open Disclosure Program Page 1 2. ACUTE Clinical Record Audit Tools Page 2 -----------------------------------------------------------------------------------
CDEC FINAL RECOMMENDATION
 CDEC FINAL RECOMMENDATION RIVAROXABAN (Xarelto Bayer Inc.) New Indication: Pulmonary Embolism Note: The Canadian Drug Expert Committee (CDEC) previously reviewed rivaroxaban for the treatment of deep vein
CDEC FINAL RECOMMENDATION RIVAROXABAN (Xarelto Bayer Inc.) New Indication: Pulmonary Embolism Note: The Canadian Drug Expert Committee (CDEC) previously reviewed rivaroxaban for the treatment of deep vein
HARROGATE AND RURAL DISTRICT CLINICAL COMMISSIONING GROUP. Minutes of the Meeting of the Harrogate and Rural District Clinical Commissioning Group
 HARROGATE AND RURAL DISTRICT CLINICAL COMMISSIONING GROUP Minutes of the Meeting of the Harrogate and Rural District Clinical Commissioning Group Quality and Clinical Governance Committee 11 March 2014
HARROGATE AND RURAL DISTRICT CLINICAL COMMISSIONING GROUP Minutes of the Meeting of the Harrogate and Rural District Clinical Commissioning Group Quality and Clinical Governance Committee 11 March 2014
OVERVIEW OF IPTR AND NON-FORMULARY PROCESS IN THE ACUTE SECTOR
 5.2: POLICY FOR THE MANAGEMENT OF INDIVIDUAL PATIENT TREATMENT REQUESTS IMPORTANT NOTE: This policy document is subject to review pending the introduction of the Peer Approval Clinical System which will
5.2: POLICY FOR THE MANAGEMENT OF INDIVIDUAL PATIENT TREATMENT REQUESTS IMPORTANT NOTE: This policy document is subject to review pending the introduction of the Peer Approval Clinical System which will
East Kent Prescribing Group
 East Kent Prescribing Group Rivaroxaban (Xarelto ) Safety Information Approved by the East Kent Prescribing Group. Approved by: East Kent Prescribing Group (Representing Ashford CCG, Canterbury and Coastal
East Kent Prescribing Group Rivaroxaban (Xarelto ) Safety Information Approved by the East Kent Prescribing Group. Approved by: East Kent Prescribing Group (Representing Ashford CCG, Canterbury and Coastal
Birmingham, Sandwell and Solihull Cardiac and Stroke Network. Rivaroxaban or warfarin for treatment of Atrial Fibrillation: Position statement
 Birmingham, Sandwell and Solihull Cardiac and Stroke Network Rivaroxaban or warfarin for treatment of Atrial Fibrillation: Position statement Introduction This guidance informs prescribers and commissioners
Birmingham, Sandwell and Solihull Cardiac and Stroke Network Rivaroxaban or warfarin for treatment of Atrial Fibrillation: Position statement Introduction This guidance informs prescribers and commissioners
Naltrexone Shared Care Guideline for the treatment of alcohol dependence and opioid dependance
 Naltrexone Shared Care Guideline for the treatment of alcohol dependence and opioid dependance Introduction Indication/Licensing information: Naltrexone is licensed for use as an additional therapy, within
Naltrexone Shared Care Guideline for the treatment of alcohol dependence and opioid dependance Introduction Indication/Licensing information: Naltrexone is licensed for use as an additional therapy, within
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE. Health Technology Appraisal
 NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE Health Technology Appraisal Adalimumab, etanercept, infliximab, rituximab and abatacept for the treatment of rheumatoid arthritis after the failure
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE Health Technology Appraisal Adalimumab, etanercept, infliximab, rituximab and abatacept for the treatment of rheumatoid arthritis after the failure
Patient Information Leaflet: Part 1 select-d
 Patient Information Leaflet: Part 1 select-d Anticoagulation Therapy in SELECTeD Cancer Patients at Risk of Recurrence of Venous Thromboembolism Introduction This
Patient Information Leaflet: Part 1 select-d Anticoagulation Therapy in SELECTeD Cancer Patients at Risk of Recurrence of Venous Thromboembolism Introduction This
PRESCRIBING GUIDELINES FOR THE MANAGEMENT OF PATIENTS ANTICOAGULANT THERAPY
 PRESCRIBING GUIDELINES FOR THE MANAGEMENT OF PATIENTS ON ANTICOAGULANT THERAPY Prepared by: NPSA Anticoagulation Steering Group Approved by: Wirral Drug & Therapeutics Committee 14 th May 2008 Review:
PRESCRIBING GUIDELINES FOR THE MANAGEMENT OF PATIENTS ON ANTICOAGULANT THERAPY Prepared by: NPSA Anticoagulation Steering Group Approved by: Wirral Drug & Therapeutics Committee 14 th May 2008 Review:
Quality Standards Rheumatoid Arthritis
 Quality Standards Rheumatoid Arthritis Minutes of the TEG 2 meeting held on 5 th November at the NICE offices in Manchester Attendees Topic Expert Group Members Robert Moots (RM), Patrick Kiely (PK), Diana
Quality Standards Rheumatoid Arthritis Minutes of the TEG 2 meeting held on 5 th November at the NICE offices in Manchester Attendees Topic Expert Group Members Robert Moots (RM), Patrick Kiely (PK), Diana
Minutes of Medicines Commissioning Committee Meeting Wednesday 16 September 2015 Severus Room, West Offices, York
 1. Apologies / Attendance Minutes of Medicines Commissioning Committee Meeting Wednesday 16 September 2015 Severus Room, West Offices, York DEC JAN FEB MAR APR MAY JUN AUG SEP Strategic Lead Pharmacist-
1. Apologies / Attendance Minutes of Medicines Commissioning Committee Meeting Wednesday 16 September 2015 Severus Room, West Offices, York DEC JAN FEB MAR APR MAY JUN AUG SEP Strategic Lead Pharmacist-
Rivaroxaban for the treatment of deep vein thrombosis (DVT) and pulmonary embolism (PE)
 Rivaroxaban for the treatment of deep vein thrombosis (DVT) and pulmonary embolism (PE) This leaflet aims to answer your questions about rivaroxaban that may be prescribed for you when you are diagnosed
Rivaroxaban for the treatment of deep vein thrombosis (DVT) and pulmonary embolism (PE) This leaflet aims to answer your questions about rivaroxaban that may be prescribed for you when you are diagnosed
How To Take Xarelto
 A patient's guide Your clinic's contact details are: Name: Contact number: Contents 2 Why have I been prescribed Xarelto? 2 What is Xarelto? 3 How do I take Xarelto? 3 What should I do if I miss a dose
A patient's guide Your clinic's contact details are: Name: Contact number: Contents 2 Why have I been prescribed Xarelto? 2 What is Xarelto? 3 How do I take Xarelto? 3 What should I do if I miss a dose
A competency framework for all prescribers updated draft for consultation
 A competency framework for all prescribers updated draft for consultation Consultation closes 15 April 2016 Contents 1 Introduction... 3 2 Uses of the framework... 4 3 Scope of the competency framework...
A competency framework for all prescribers updated draft for consultation Consultation closes 15 April 2016 Contents 1 Introduction... 3 2 Uses of the framework... 4 3 Scope of the competency framework...
Rivaroxaban shared care guidelines for the prevention of stroke and embolism in adult patients with nonvalvular atrial fibrillation.
 South West Essex Rivaroxaban Shared Care Guideline (SCG) Rivaroxaban shared care guidelines for the prevention of stroke and embolism in adult patients with nonvalvular atrial fibrillation. Introduction
South West Essex Rivaroxaban Shared Care Guideline (SCG) Rivaroxaban shared care guidelines for the prevention of stroke and embolism in adult patients with nonvalvular atrial fibrillation. Introduction
Dorset Cardiac Centre
 P a g e 1 Dorset Cardiac Centre Patients with Atrial Fibrillation/Flutter undergoing DC Cardioversion or Ablation procedures- Guidelines for Novel Oral Anti-coagulants (NOACS) licensed for this use February
P a g e 1 Dorset Cardiac Centre Patients with Atrial Fibrillation/Flutter undergoing DC Cardioversion or Ablation procedures- Guidelines for Novel Oral Anti-coagulants (NOACS) licensed for this use February
BEDFORDSHIRE AND LUTON JOINT PRESCRIBING COMMITTEE (JPC)
 BEDFORDSHIRE AND LUTON JOINT PRESCRIBING COMMITTEE (JPC) September 2014 Review date: September 2017 Bulletin 203: Tocilizumab (subcutaneous) in combination with methotrexate or as monotherapy for the treatment
BEDFORDSHIRE AND LUTON JOINT PRESCRIBING COMMITTEE (JPC) September 2014 Review date: September 2017 Bulletin 203: Tocilizumab (subcutaneous) in combination with methotrexate or as monotherapy for the treatment
GUIDELINES FOR THE USE OF PALIPERIDONE PALMITATE (Xeplion ) Version: 2
 SH CP 29 GUIDELINES FOR THE USE OF PALIPERIDONE PALMITATE (Xeplion ) Summary: Guidelines for the use of paliperidone palmitate (Xeplion ) Keywords (minimum of 5): (To assist policy search engine) Target
SH CP 29 GUIDELINES FOR THE USE OF PALIPERIDONE PALMITATE (Xeplion ) Summary: Guidelines for the use of paliperidone palmitate (Xeplion ) Keywords (minimum of 5): (To assist policy search engine) Target
Guidelines for Cancer Pain Management in Substance Misusers Dr Jane Neerkin, Dr Chi-Chi Cheung and Dr Caroline Stirling
 Guidelines for Cancer Pain Management in Substance Misusers Dr Jane Neerkin, Dr Chi-Chi Cheung and Dr Caroline Stirling Patients with a substance misuse history are at increased risk of receiving inadequate
Guidelines for Cancer Pain Management in Substance Misusers Dr Jane Neerkin, Dr Chi-Chi Cheung and Dr Caroline Stirling Patients with a substance misuse history are at increased risk of receiving inadequate
NHS DORSET CLINICAL COMMISSIONING GROUP POSITION STATEMENT ON ORAL ANTICOAGULANTS IN ATRIAL FIBRILLATION
 Version 3 August 2014 NHS DORSET CLINICAL COMMISSIONING GROUP POSITION STATEMENT ON ORAL ANTICOAGULANTS IN ATRIAL FIBRILLATION Dorset CCG commissions the use of newer oral anti-coagulants in accordance
Version 3 August 2014 NHS DORSET CLINICAL COMMISSIONING GROUP POSITION STATEMENT ON ORAL ANTICOAGULANTS IN ATRIAL FIBRILLATION Dorset CCG commissions the use of newer oral anti-coagulants in accordance
Anticoagulant therapy
 Anticoagulation: The risks Anticoagulant therapy 1990 2002: 600 incidents reported 120 resulted in death of patient 92 deaths related to warfarin usage 28 reports related to heparin usage Incidents in
Anticoagulation: The risks Anticoagulant therapy 1990 2002: 600 incidents reported 120 resulted in death of patient 92 deaths related to warfarin usage 28 reports related to heparin usage Incidents in
STROKE PREVENTION IN ATRIAL FIBRILLATION
 STROKE PREVENTION IN ATRIAL FIBRILLATION OBJECTIVE: To guide clinicians in the selection of antithrombotic therapy for the secondary prevention of ischemic stroke and arterial thromboembolism in patients
STROKE PREVENTION IN ATRIAL FIBRILLATION OBJECTIVE: To guide clinicians in the selection of antithrombotic therapy for the secondary prevention of ischemic stroke and arterial thromboembolism in patients
Anticoagulants for stroke prevention in atrial fibrillation Patient frequently asked questions
 Anticoagulants for stroke prevention in atrial fibrillation Patient frequently asked questions What is atrial fibrillation?...2 What are dabigatran, rivaroxaban and apixaban and what are they used for?...2
Anticoagulants for stroke prevention in atrial fibrillation Patient frequently asked questions What is atrial fibrillation?...2 What are dabigatran, rivaroxaban and apixaban and what are they used for?...2
Arthroscopic rotator cuff repair
 Arthroscopic rotator cuff repair The aim of this leaflet is to help answer some of the questions you may have about having an arthroscopic rotator cuff repair. It explains the benefits, risks and alternatives
Arthroscopic rotator cuff repair The aim of this leaflet is to help answer some of the questions you may have about having an arthroscopic rotator cuff repair. It explains the benefits, risks and alternatives
Rivaroxaban for the treatment of deep vein thrombosis and prevention of recurrent deep vein thrombosis and pulmonary embolism
 Implementation of NICE TA 261 Rivaroxaban for the treatment of deep vein thrombosis and prevention of recurrent deep vein thrombosis and pulmonary embolism Contents 1. Executive summary 2. Introduction
Implementation of NICE TA 261 Rivaroxaban for the treatment of deep vein thrombosis and prevention of recurrent deep vein thrombosis and pulmonary embolism Contents 1. Executive summary 2. Introduction
2. Background This indication of rivaroxaban had not previously been considered by the PBAC.
 PUBLIC SUMMARY DOCUMENT Product: Rivaroxaban, tablets, 15mg and 20mg, Xarelto Sponsor: Bayer Australia Ltd Date of PBAC Consideration: March 2013 1. Purpose of Application The application requested the
PUBLIC SUMMARY DOCUMENT Product: Rivaroxaban, tablets, 15mg and 20mg, Xarelto Sponsor: Bayer Australia Ltd Date of PBAC Consideration: March 2013 1. Purpose of Application The application requested the
Careers in Haematology
 Careers in Haematology A Guide for Medical Students and Junior Doctors Haematology is the medical speciality concerned with blood disorders. Your non-medical friends however will always think that you
Careers in Haematology A Guide for Medical Students and Junior Doctors Haematology is the medical speciality concerned with blood disorders. Your non-medical friends however will always think that you
Rivaroxaban for the treatment of deep vein thrombosis and prevention of recurrent deep vein thrombosis and pulmonary embolism
 Rivaroxaban for the treatment of deep vein thrombosis and prevention of recurrent deep vein thrombosis and pulmonary Issued: July 2012 guidance.nice.org.uk/ta NHS Evidence has accredited the process used
Rivaroxaban for the treatment of deep vein thrombosis and prevention of recurrent deep vein thrombosis and pulmonary Issued: July 2012 guidance.nice.org.uk/ta NHS Evidence has accredited the process used
MEDICINE SPECIALTIES JOB PLANNING GUIDANCE AND MODEL JOB PLAN EXAMPLE
 MEDICINE SPECIALTIES JOB PLANNING GUIDANCE AND MODEL JOB PLAN EXAMPLE INTRODUCTION Since 1991 it has been a contractual requirement for all consultants to have a job plan, which is agreed and reviewed
MEDICINE SPECIALTIES JOB PLANNING GUIDANCE AND MODEL JOB PLAN EXAMPLE INTRODUCTION Since 1991 it has been a contractual requirement for all consultants to have a job plan, which is agreed and reviewed
Formulary Management
 Formulary Management Formulary management is an integrated patient care process which enables physicians, pharmacists and other health care professionals to work together to promote clinically sound, cost-effective
Formulary Management Formulary management is an integrated patient care process which enables physicians, pharmacists and other health care professionals to work together to promote clinically sound, cost-effective
Health Care Needs Assessment of Services for Adults with Rheumatoid Arthritis
 Health Care Needs Assessment of Services for Adults with Rheumatoid Arthritis PART D: Cost Implications for NHS Rheumatoid Arthritis Services in Scotland Scottish Public Health Network August 2013 1 Table
Health Care Needs Assessment of Services for Adults with Rheumatoid Arthritis PART D: Cost Implications for NHS Rheumatoid Arthritis Services in Scotland Scottish Public Health Network August 2013 1 Table
Patient Information Leaflet
 Patient Information Leaflet METHOTREXATE We hope this fact sheet will provide you with some information about Methotrexate and answer some of the questions you may have. Methotrexate is available in tablet
Patient Information Leaflet METHOTREXATE We hope this fact sheet will provide you with some information about Methotrexate and answer some of the questions you may have. Methotrexate is available in tablet
Prescribing Framework for Donepezil in the Treatment and Management of Dementia
 Hull & East Riding Prescribing Committee Prescribing Framework for Donepezil in the Treatment and Management of Dementia Patients Name:.. NHS Number: Patients Address:... (Use addressograph sticker) GP
Hull & East Riding Prescribing Committee Prescribing Framework for Donepezil in the Treatment and Management of Dementia Patients Name:.. NHS Number: Patients Address:... (Use addressograph sticker) GP
Spinal cord stimulation
 Spinal cord stimulation This leaflet aims to answer your questions about having spinal cord stimulation. It explains the benefits, risks and alternatives, as well as what you can expect when you come to
Spinal cord stimulation This leaflet aims to answer your questions about having spinal cord stimulation. It explains the benefits, risks and alternatives, as well as what you can expect when you come to
MEDICAL ASSISTANCE HANDBOOK PRIOR AUTHORIZATION OF PHARMACEUTICAL SERVICES. A. Prescriptions That Require Prior Authorization
 MEDICAL ASSISTANCE HBOOK PRI AUTHIZATION OF PHARMACEUTICAL SERVICES I. Requirements for Prior Authorization of Anticoagulants A. Prescriptions That Require Prior Authorization Prescriptions for Anticoagulants
MEDICAL ASSISTANCE HBOOK PRI AUTHIZATION OF PHARMACEUTICAL SERVICES I. Requirements for Prior Authorization of Anticoagulants A. Prescriptions That Require Prior Authorization Prescriptions for Anticoagulants
HOSPITAL PHARMACY PRACTICE IN THE UK AND THE RESPONSIBLE PHARMACIST REQUIREMENTS
 HOSPITAL PHARMACY PRACTICE IN THE UK AND THE RESPONSIBLE PHARMACIST REQUIREMENTS ROYAL PHARMACEUTICAL SOCIETY OF GREAT BRITAIN AND THE PHARMACEUTICAL SOCIETY OF NORTHERN IRELAND STATUS OF THIS DOCUMENT
HOSPITAL PHARMACY PRACTICE IN THE UK AND THE RESPONSIBLE PHARMACIST REQUIREMENTS ROYAL PHARMACEUTICAL SOCIETY OF GREAT BRITAIN AND THE PHARMACEUTICAL SOCIETY OF NORTHERN IRELAND STATUS OF THIS DOCUMENT
Doncaster & Bassetlaw Medicines Formulary
 Doncaster & Bassetlaw Medicines Formulary Section 3.2: Corticosteroids Beclometasone 50, 100 and 250micrograms/dose Clickhaler Clenil Modulite (Beclometasone CFC free) 50, 100, and 250micrograms/dose MDI
Doncaster & Bassetlaw Medicines Formulary Section 3.2: Corticosteroids Beclometasone 50, 100 and 250micrograms/dose Clickhaler Clenil Modulite (Beclometasone CFC free) 50, 100, and 250micrograms/dose MDI
How To Get A Dose Of Bayer Healthcare'S Oral Anticoagulant, Xarelto
 News Release FOR UK HEALTHCARE MEDIA ONLY Bayer HealthCare Bayer plc Bayer House Strawberry Hill Newbury Berkshire, RG14 1JA www.bayer.co.uk Bayer s Xarelto (rivaroxaban) Recommended by CHMP for EU Approval
News Release FOR UK HEALTHCARE MEDIA ONLY Bayer HealthCare Bayer plc Bayer House Strawberry Hill Newbury Berkshire, RG14 1JA www.bayer.co.uk Bayer s Xarelto (rivaroxaban) Recommended by CHMP for EU Approval
Service delivery interventions
 Service delivery interventions S A S H A S H E P P E R D D E P A R T M E N T O F P U B L I C H E A L T H, U N I V E R S I T Y O F O X F O R D CO- C O O R D I N A T I N G E D I T O R C O C H R A N E E P
Service delivery interventions S A S H A S H E P P E R D D E P A R T M E N T O F P U B L I C H E A L T H, U N I V E R S I T Y O F O X F O R D CO- C O O R D I N A T I N G E D I T O R C O C H R A N E E P
Medication Management Guidelines for Nurses and Midwives
 Medication Management Guidelines for Nurses and Midwives 1. Introduction As the statutory body responsible for the regulation of nursing and midwifery practice in Western Australia (WA), the Nurses & Midwives
Medication Management Guidelines for Nurses and Midwives 1. Introduction As the statutory body responsible for the regulation of nursing and midwifery practice in Western Australia (WA), the Nurses & Midwives
