Rigid Container Testing of Steam Sterilizers
|
|
|
- Nancy York
- 7 years ago
- Views:
Transcription
1 Rigid Container Testing of Steam Sterilizers Copyright 2015, Sterile Processing University, LLC, All Rights Reserved. This material may not be copied or used without permission of the author. Nancy Chobin, RN, AAS, ACSP, CSPDM **This in-service has been Approved by the CBSPD, Inc. for 1 CEU. Objectives: To review the AAMI recommended protocol for rigid container testing To describe the process of rigid container testing Testing of Steam Sterilizers - AAMI ST-79 recommends routine biological monitoring of steam sterilizers weekly, preferably daily. Biological monitoring with a process challenge device (PCD) containing live bacterial spores of geo bacillus stearothermophilus is recommended because this spore is the most resistant to steam cycles. Biological Testing - The use of biological indicators (BIs) provides evidence of efficacy by challenging the sterilizer with a large number of highly resistant bacterial spores. Biological monitoring provides the only direct measure of the lethality of a sterilization cycle. (AAMI ST-79, 2013: Section ). Sterilizer manufacturers validate their sterilization cycles using BIs; therefore, routine sterilizer efficacy monitoring in health care facilities should also be conducted using BIs. Conventional PCDs are based on linen test packs, not rigid containers. Therefore, AAMI recommends periodic testing of rigid container systems. Rigid sterilization container systems vary widely in design, mechanics, and materials of construction. Work practices, sterilizer performance characteristics, and the facility utilities supplying the sterilizer can affect the dynamics of the sterilization process. These factors can impact the specific performance characteristics of rigid sterilization container systems and their suitability for specific sterilization methods and cycles. Health care personnel bear the ultimate responsibility for ensuring that any packaging method or material, including a rigid sterilization container system, is suitable for use in sterilization processing and sterility maintenance. (AAMI, ST-79) User Responsibilities - Before purchasing any packaging system, collect all the information including the container manufacturer s instructions for use (IFU). Perform testing (pre-purchase testing) to ensure that items to be packaged can be sterilized by the specific sterilizers and/or sterilization methods to be used within the facility. Understand that the interaction of the container system, medical device, and sterilizer technologies is complex. copied or used without permission of the author. Page 1
2 Containers that will be used for steam sterilization need to permit complete air removal, adequate steam penetration, and drying. The design of the rigid container system needs to be compatible with the design and performance characteristics of the sterilizer(s) in which it is used. Pre-purchase evaluation testing - assures the particular container system being considered will be acceptable to the users that it will perform properly in the health care facility s sterilizing equipment. This testing is not a substitute for the more extensive validation testing conducted by manufacturers to qualify their products. Why Do We Have to Test? - Manufacturers of container systems can only test using properly designed and operating sterilization equipment. Various sizes of sterilizers having the same sterilization cycle could have different air-removal efficiencies. Manufacturers cannot possibly test all combinations of sterilizer sizes, cycles, and process efficiencies. Users need to perform testing to verify that there are no problems or to identify technical problems to be resolved in consultation with the container system manufacturer, the sterilizer manufacturer, and consultants. Before a rigid sterilization container system is purchased, you need to determine whether your facility can verify the manufacturer's test results. If not, you should seek advice from the container manufacturer concerning instructions and guidelines for use of the system. Testing should be conducted in the health care facility to 1) ensure that the conditions essential to sterilization can be achieved and 2) that the specific configuration of the container contents is acceptable for the sterilization process and for the requirements at the point of use. Sterilization process conditions such as exposure time should be evaluated by physical, biological, and chemical monitoring. In each rigid sterilization container system to be tested, BIs and CIs should be placed strategically alongside each other at locations that present the greatest challenge to air evacuation and sterilant penetration. In gravity-displacement steam sterilizers, the corners of the container system and the underside of the lid, away from the filters, are the likeliest locations for air pockets. Consult with the container manufacturer regarding their recommended location for placement of CIs and BIs. The location should represent the areas of greatest challenge to steam penetration and air removal. Understand that the location can vary by container manufacturer, so if your facility is using more than one manufacturer s container, you may have different locations for the BIs and CIs. Dynamic Air Removal Testing - The purpose of this testing is: to assess four (4) fundamentally essential aspects of the use of rigid sterilization container systems in dynamic-air-removal steam sterilization: 1) Will the container system design permit adequate air removal from the container system when the sterilizer chamber has reached the point of maximum air removal? 2) Does the container system design allow adequate steam penetration to reach equilibrium between the sterilizer chamber and the interior and contents of the container? 3) Will the combination of sterilizer and container system design achieve sterilization conditions? copied or used without permission of the author. Page 2
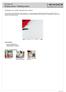
3 4) Will the combination of sterilizer and container system design permit adequate drying and thus help promote sterility maintenance? How to Perform the Test - Sterile processing needs to identify each size container (from each container manufacturer if more than one manufacturer). When measuring containers, understand that containers have three (3) dimensions; height, length and width. Photo Above Rigid Containers with Different Dimensions If any one of the three dimensions is different, it is considered a different size container. The container system should represent the sizes that are available and being considered for purchase/use or are already being used at your facility for routine processing. According to AAMI ST-79, The rationale for this is that the sterilant enters through discrete portals in a container system. The sterilant must then diffuse throughout the inside of the container system and then to the items being sterilized. Two barriers need to be overcome before the inside of the container system reaches equilibrium with the sterilizer chamber. Container systems are very different from flexible wraps in which the entire barrier is permeable. Then, assemble five (5) BI vials (use control vials) to place inside each container. You will need one (1) additional vial to act as the control. Ensure all 5 BI vials for the container and the control vial are from the same lot #. Once you have identified the number of different size containers, prepare the containers as follows: Unless otherwise directed by the container manufacturer: Assemble set that is usually contained inside the container prepared with the largest instrument sets (including any optional absorbent material) recommended by the container system manufacturer. Assemble BI vials (you can use string attached to vials to keep them in place). copied or used without permission of the author. Page 3
4 Place one BI vial in each corner of the inside basket. Attach the string to the corner of the basket with autoclave tape to keep in place. Place a CI in the same location as the BI. Affix a 5 th BI vial under the lid the BI should suspend into container when lid applied. Check with the container manufacturer for their recommended location of BIs on the lid. Photo Above BI vial attached to corner Of inside basket, CI in corner Photo Above Rigid container with set inside and BI and CI in 4 corners copied or used without permission of the author. Page 4
5 To know where the BI vials were located inside the set, you can label the vials with different letters (e.g. A,B,C,D) to identify their location. Use a photo or grid to label/document where the vials were placed. Document where the containers were placed on the sterilizer cart; i.e. top, middle, lower shelf. Close Container. Apply locks. Affix load card; label as TEST. Prepare for sterilization. Tests Recommended AAMI recommends that a maximum-load test and a small-load test be performed for representative sterilizers. This means if all your sterilizers are from the same manufacturer, same age and same size, all the testing can be performed on one of the sterilizers. For the remaining sterilizers, it is necessary only to run the small-load test. For the maximum-load test, place the container systems on the bottom shelf over the drain and two test container systems on each of the other sterilizer shelves (if space permits). To test a sterilizer having three shelves, six containers will be needed; for 2 shelves, 4 containers will be needed. The chamber is otherwise fully loaded with conventionally packaged items. A sterilization cycle is run with the exposure time recommended by the container system manufacturer. Photo Above - Maximum Load Test Loading of the Sterilizer. Note the 2 containers on each shelf that contain the BIs. AAMI explains the need for the Maximum Load Test is to ensure that the large volume of air in this type of load is removed adequately and that the steam supply is sufficient to achieve sterilization in a load in copied or used without permission of the author. Page 5
6 which the considerable mass results in significant condensation. Maximum-load testing also permits the user to determine if additional steps are necessary to achieve adequate drying. Small Load Testing With this test, only one test container is placed on the bottom shelf over the drain in an otherwise empty chamber. A sterilization cycle is run with the exposure time recommended by the container system manufacturer. AAMI explains the need for the Small Load Test is to identify any problems associated with the smallload effect, a phenomenon in sterilizers having dynamic air removal in which residual air in the chamber can become entrained in packaged items as steam enters the chamber. At the end of the sterilization cycle and cooling, the containers should be removed. List the containers by size/set on documentation form. Open the containers one at a time so the test results do not get mixed up. Record the results of CIs on the log form per location in set. Activate and incubate BIs/control vial. Record BI results on documentation form, by location inside container. Interpretation of Test Results - To qualify the container system sterilizer combination, all BIs should be negative and all CIs should show complete endpoint responses (negative BIs and pass on the CIs). In the maximum-load test, positive BIs or incompletely responding CIs suggest that 1. the sterilization process is inadequate 2. there could be a problem with the sterilizer itself 3. a problem with the container system 4. or the sterilizer-container system combination In the small-load test, failures indicate: 1. insufficient steam penetration 2. Insufficient air removal Any of these failures could be caused by the sterilizer, the container system, or the sterilizer/container system combination. Test Failures - The first step is to check the physical monitors to ensure that the cycle parameters were correct. If the cycle parameters were correct, then the sterilizer should be evaluated with BI test packs and Bowie-Dick test packs to identify any equipment malfunction. Container systems should not be used in this evaluation. The sterilizer manufacturer should be consulted if the performance of the sterilizer is questionable. If the sterilizer appears to be functioning properly, the container system manufacturer should be consulted for assistance in resolving the problem. copied or used without permission of the author. Page 6
7 Gravity Displacement Cycles If rigid containers are used in these cycles, there is a special protocol for rigid container testing of gravity cycles. Refer to AAMI ST-79 for specific information. Extensive testing of all gravity-displacement steam sterilizers is recommended because of the potential problem with air removal from containers in gravity-displacement steam sterilization. Summary - Rigid container testing is an important part of quality assurance testing. Containers should be tested pre-purchase and routinely thereafter (e.g. annually). This testing does NOT replace routine BI testing and does NOT have to be performed the first load of the day. If you have more than one manufacturers rigid container, each manufacturers containers must be tested separately. Quality Assurance is required to assure the adequacy of the sterilization process. QA testing is ongoing and must be performed as recommended. QUIZ Rigid Container Testing Please click on the link below to take the quiz. Good Luck! copied or used without permission of the author. Page 7
Best Practices for Loaner Surgical Instruments
 Best Practices for Loaner Surgical Instruments Copyright 2015, Sterile Processing University, LLC, All Rights Reserved. This material may not be copied or used without permission of the author. Nancy Chobin,
Best Practices for Loaner Surgical Instruments Copyright 2015, Sterile Processing University, LLC, All Rights Reserved. This material may not be copied or used without permission of the author. Nancy Chobin,
Heat Transfer. Condensation. Revaporization
 Objectives Wrapping a Towel Around the Problem of Wet Packs Identify causes of wet packs in steam sterilization Review suggested documentation that can assist in solving wet pack problems Why are Wet Packs
Objectives Wrapping a Towel Around the Problem of Wet Packs Identify causes of wet packs in steam sterilization Review suggested documentation that can assist in solving wet pack problems Why are Wet Packs
STANDARD OPERATING PROCEDURES DIVISION OF COMPARATIVE MEDICINE UNIVERSITY OF SOUTH FLORIDA
 STANDARD OPERATING PROCEDURES DIVISION OF COMPARATIVE MEDICINE UNIVERSITY OF SOUTH FLORIDA SOP#: 1002.9 Date Issued: 10/98 Date Revised: 5/16 Page 1 of 5 TITLE: SCOPE: RESPONSIBILITY: PURPOSE: Animal Care
STANDARD OPERATING PROCEDURES DIVISION OF COMPARATIVE MEDICINE UNIVERSITY OF SOUTH FLORIDA SOP#: 1002.9 Date Issued: 10/98 Date Revised: 5/16 Page 1 of 5 TITLE: SCOPE: RESPONSIBILITY: PURPOSE: Animal Care
Biological Indicators and
 & and BIs play an important role in the monitoring of sterilizers, along with physical and chemical indicators. However, only BIs contain live spores that directly measure the lethality of a sterilization
& and BIs play an important role in the monitoring of sterilizers, along with physical and chemical indicators. However, only BIs contain live spores that directly measure the lethality of a sterilization
3039878000or8009926372
 3039878000or8009926372 bi s a l e s @me s a l a bs. c om Regulatory officials and sterilization experts have voiced concerns regarding the appropriateness of using a Biological Indicator (BI) Ampoule interchangeably
3039878000or8009926372 bi s a l e s @me s a l a bs. c om Regulatory officials and sterilization experts have voiced concerns regarding the appropriateness of using a Biological Indicator (BI) Ampoule interchangeably
Air Removal from the Sterilization Chamber
 TM Volume 5, Number 2 March 2008 Garrett Krushefski Scientific & Technical Services Manager Air Removal from the Sterilization Chamber I recently received a question from a customer asking why we state
TM Volume 5, Number 2 March 2008 Garrett Krushefski Scientific & Technical Services Manager Air Removal from the Sterilization Chamber I recently received a question from a customer asking why we state
THE BASICS OF PACKAGING
 THE BASICS OF PACKAGING Objectives: Review the regulatory requirements for packaging Present the purpose, function and essential characteristics of packaging Describe packaging options, their use, and
THE BASICS OF PACKAGING Objectives: Review the regulatory requirements for packaging Present the purpose, function and essential characteristics of packaging Describe packaging options, their use, and
Steam Sterilization and the 2007 Revision of PDA Technical Report 1
 Steam Sterilization and the 2007 Revision of PDA Technical Report 1 14 November 2007 Presented By: Mike Finger (Tunnell Consulting) Don Drew (Abbott Bioresearch Center) Agenda: Introduction Sterilization
Steam Sterilization and the 2007 Revision of PDA Technical Report 1 14 November 2007 Presented By: Mike Finger (Tunnell Consulting) Don Drew (Abbott Bioresearch Center) Agenda: Introduction Sterilization
1/22/2012. When is it a good time to learn about sterile processing in your facility? Continuing Education Contact Hours. Learning Objectives
 Continuing Education Contact Hours Participants must complete the entire presentation/seminar to achieve successful completion and receive contact hour credit. Partial credit will not be given. All of
Continuing Education Contact Hours Participants must complete the entire presentation/seminar to achieve successful completion and receive contact hour credit. Partial credit will not be given. All of
Sterilization of Surgical Implants: Did You Know
 Sterilization of Surgical Implants: Did You Know by Rose Seavey, RN, MBA, CNOR, ACSP Many thanks to the team at 3M Health Care for working with Managing Infection Control to provide the following accre
Sterilization of Surgical Implants: Did You Know by Rose Seavey, RN, MBA, CNOR, ACSP Many thanks to the team at 3M Health Care for working with Managing Infection Control to provide the following accre
Steam Sterilization Cycles for Lab Applications
 Steam Sterilization Cycles for Lab Applications Presented by Gary Butler STERIS Life Sciences August 2009 Early Steam Sterilizers Koch Upright Sterilizer First Pressurized Sterilizer First built by Pasteur-Chamberlain
Steam Sterilization Cycles for Lab Applications Presented by Gary Butler STERIS Life Sciences August 2009 Early Steam Sterilizers Koch Upright Sterilizer First Pressurized Sterilizer First built by Pasteur-Chamberlain
3 Attest Biological Monitoring System
 3 Attest Biological Monitoring System 1 Technical Product Profile A Technical Service Publication 2 Table of Contents Introduction...5 Product Description...5 Indicators... 5 Test Frequency... 8 Usage
3 Attest Biological Monitoring System 1 Technical Product Profile A Technical Service Publication 2 Table of Contents Introduction...5 Product Description...5 Indicators... 5 Test Frequency... 8 Usage
AST Standards of Practice for Packaging Material and Preparing Items for Sterilization
 AST Standards of Practice for Packaging Material and Preparing Items for Sterilization Introduction The following Standards of Practice were researched and authored by the AST Education and Professional
AST Standards of Practice for Packaging Material and Preparing Items for Sterilization Introduction The following Standards of Practice were researched and authored by the AST Education and Professional
THE BASICS OF STERILIZATION
 THE BASICS OF STERILIZATION Objectives: State the importance of sterilization to patient care Review three key essentials of the sterilization process Describe sterilization methods used, application,
THE BASICS OF STERILIZATION Objectives: State the importance of sterilization to patient care Review three key essentials of the sterilization process Describe sterilization methods used, application,
Infection Control in the Dental Practice through Proper Sterilization
 Midmark Corporation 60 Vista Drive P.O. Box 286 Versailles, Ohio 45380-0286 937-526-3662 midmark.com Infection Control in the Dental Practice through Proper Sterilization In today's world, the need for
Midmark Corporation 60 Vista Drive P.O. Box 286 Versailles, Ohio 45380-0286 937-526-3662 midmark.com Infection Control in the Dental Practice through Proper Sterilization In today's world, the need for
Autoclave Safety. Autoclaves are sterilizers using high pressure and high temperature steam. The potential safety risks for the operators are:
 Autoclave Safety Purpose: Sterilization refers to the complete killing of all living organisms, including spores. Common sterilization techniques include the application of wet heat, dry heat, chemicals,
Autoclave Safety Purpose: Sterilization refers to the complete killing of all living organisms, including spores. Common sterilization techniques include the application of wet heat, dry heat, chemicals,
Fundamental Autoclave Techniques
 Fundamental Autoclave Techniques Version 3, September 2010 Veronika Tatarinoff GSBmE University of NSW Brandy Nelson University of Kentucky The following information is directed at aseptic/sterile surgical,
Fundamental Autoclave Techniques Version 3, September 2010 Veronika Tatarinoff GSBmE University of NSW Brandy Nelson University of Kentucky The following information is directed at aseptic/sterile surgical,
A GUIDELINE FOR THE SAFE USE OF AUTOCLAVES
 A GUIDELINE FOR THE SAFE USE OF AUTOCLAVES Table of Contents 1.0 PURPOSE... 4 2.0 INTRODUCTION... 4 3.0 PERSONAL PROTECTIVE EQUIPMENT... 4 4.0 TRAINING... 5 5.0 ITEMS FOR AUTOCLAVING (THE CAN AND CAN NOTS)...
A GUIDELINE FOR THE SAFE USE OF AUTOCLAVES Table of Contents 1.0 PURPOSE... 4 2.0 INTRODUCTION... 4 3.0 PERSONAL PROTECTIVE EQUIPMENT... 4 4.0 TRAINING... 5 5.0 ITEMS FOR AUTOCLAVING (THE CAN AND CAN NOTS)...
#002. Flash Sterilization. S t u d y G u i d e. The Hot Issues of. A Continuing Education Study Guide presented by
 #002 S t u d y G u i d e The Hot Issues of Flash Sterilization A Continuing Education Study Guide presented by For more information, visit the education page of our website www.steris.com The Hot Issue
#002 S t u d y G u i d e The Hot Issues of Flash Sterilization A Continuing Education Study Guide presented by For more information, visit the education page of our website www.steris.com The Hot Issue
Top 10 considerations when validating an autoclave
 Top 10 considerations when validating an autoclave Validating an autoclave is a daunting and time-consuming task. This white paper details the tricks, tips and traps to such a validation project from how
Top 10 considerations when validating an autoclave Validating an autoclave is a daunting and time-consuming task. This white paper details the tricks, tips and traps to such a validation project from how
Packaging for Sterilization
 Packaging for Sterilization IT S NOT JUST A BOX, A BAG OR GIFT WRAPPING! by Rose Seavey, RN, BS, MBA, CNOR, ACSP Objectives After completion of this self-study activity, the learner will be able to: 1.
Packaging for Sterilization IT S NOT JUST A BOX, A BAG OR GIFT WRAPPING! by Rose Seavey, RN, BS, MBA, CNOR, ACSP Objectives After completion of this self-study activity, the learner will be able to: 1.
FACT SHEET : Using Autoclaves Safely
 CSULA Environmental Health and Safety Biosafety Office FACT SHEET : Using Autoclaves Safely Most science research laboratories on campus require the use of autoclaves. The primary purpose of the autoclave
CSULA Environmental Health and Safety Biosafety Office FACT SHEET : Using Autoclaves Safely Most science research laboratories on campus require the use of autoclaves. The primary purpose of the autoclave
Comparison Study of the Growth Promotion Capabilities of Self Contained Biological Indicator Culture Medium in Extended Steam Sterilization Cycles
 Comparison Study of the Growth Promotion Capabilities of Self Contained Biological Indicator Culture Medium in Extended Steam Sterilization Cycles Charles Hughes, Gary Socola, Donald Tumminelli, and Mike
Comparison Study of the Growth Promotion Capabilities of Self Contained Biological Indicator Culture Medium in Extended Steam Sterilization Cycles Charles Hughes, Gary Socola, Donald Tumminelli, and Mike
Sterile Cleanroom Management
 Sterile Cleanroom Management Lynn Stanard, Sr. Quality Manager, Berkshire Corporation When manufacturing in an aseptic environment, it is critical to ensure that the various cleanroom consumables, such
Sterile Cleanroom Management Lynn Stanard, Sr. Quality Manager, Berkshire Corporation When manufacturing in an aseptic environment, it is critical to ensure that the various cleanroom consumables, such
01.05 Cleaning, Sterilization, High-Level Disinfection and Storage of Patient Care Devices and Other Items
 01.05 Cleaning, Sterilization, High-Level Disinfection and Storage of Patient Care Devices and Other Items Page 1 of 36 INDEX Page Purpose, Audience, Policy, General Recommendations & Electrical Safety...
01.05 Cleaning, Sterilization, High-Level Disinfection and Storage of Patient Care Devices and Other Items Page 1 of 36 INDEX Page Purpose, Audience, Policy, General Recommendations & Electrical Safety...
KGI MEDICAL WASTE MANAGEMENT PLAN
 1revised 2012 I. Facility Information KGI MEDICAL WASTE MANAGEMENT PLAN a. Contact Person: Barbara Erwin Director Research Operations/Chemical and Biological Laboratory Safety/Chair. 909-607-0160 i. Facility:
1revised 2012 I. Facility Information KGI MEDICAL WASTE MANAGEMENT PLAN a. Contact Person: Barbara Erwin Director Research Operations/Chemical and Biological Laboratory Safety/Chair. 909-607-0160 i. Facility:
Top Ten Sterilization Issues in an Ambulatory Surgery Center
 10 Top Ten Sterilization Issues in an Ambulatory Surgery Center by Peggy Prinz Luebbert, MS, MT(ASCP), CIC, CHSP Objectives After completion of this self-study activity, the learner will be able to: 1.
10 Top Ten Sterilization Issues in an Ambulatory Surgery Center by Peggy Prinz Luebbert, MS, MT(ASCP), CIC, CHSP Objectives After completion of this self-study activity, the learner will be able to: 1.
TECHNIQUES FOR STEAM STERILIZING LABORATORY WASTE
 TECHNIQUES FOR STEAM STERILIZING LABORATORY WASTE by Timothy E. Cooney Research Scientist AMSCO Research and Development SUMMARY The traditional method for steam sterilizing petri dishes and other laboratory
TECHNIQUES FOR STEAM STERILIZING LABORATORY WASTE by Timothy E. Cooney Research Scientist AMSCO Research and Development SUMMARY The traditional method for steam sterilizing petri dishes and other laboratory
WHITE PAPER April 18, 2016
 WHITE PAPER April 18, 2016 Nitrogen Dioxide Biodecontamination: A New, Effective and Cost-saving Option for Biodecontaminating Syringe Tubs Prior to the Filling Line Prepared by: David Opie, PhD Maura
WHITE PAPER April 18, 2016 Nitrogen Dioxide Biodecontamination: A New, Effective and Cost-saving Option for Biodecontaminating Syringe Tubs Prior to the Filling Line Prepared by: David Opie, PhD Maura
EH&S. Sheet. Fact. Safe and Effective Use of Autoclaves. What are autoclaves? Factors for effective sterilization. Dry heat cycle - when to use
 Please post or circulate Fact heet nvironment, ealth and afety Information for the Berkeley Campus No. 33 Revised 04/04/11 afe and ffective Use of Autoclaves Autoclaves are easy to use but can pose a safety
Please post or circulate Fact heet nvironment, ealth and afety Information for the Berkeley Campus No. 33 Revised 04/04/11 afe and ffective Use of Autoclaves Autoclaves are easy to use but can pose a safety
Laboratory Autoclave SN210/310/510,SQ510/810
 1/5 Laboratory Autoclave SN210/310/510,SQ510/810 SN210/310/510,SQ510/810 The highest temperature of 135deg.C Upwards opening lid SN210 SN310 SN510 Effective Capacity : 20LEffective Capacity : 32L Effective
1/5 Laboratory Autoclave SN210/310/510,SQ510/810 SN210/310/510,SQ510/810 The highest temperature of 135deg.C Upwards opening lid SN210 SN310 SN510 Effective Capacity : 20LEffective Capacity : 32L Effective
Steam Sterilization Principles
 Reprinted from PHARMACEUTICAL ENGINEERING THE OFFICIAL TECHNICAL MAGAZINE OF ISPE NOVEMBER/DECEMBER 2013, VOL 33, NO 6 Copyright ISPE 2013 www.pharmaceuticalengineering.org facilities and equipment by
Reprinted from PHARMACEUTICAL ENGINEERING THE OFFICIAL TECHNICAL MAGAZINE OF ISPE NOVEMBER/DECEMBER 2013, VOL 33, NO 6 Copyright ISPE 2013 www.pharmaceuticalengineering.org facilities and equipment by
Sterilization of narrow channels: how to ensure effectiveness in daily routine? Dr.ir. Josephus Paulus Clemens Maria Van Doornmalen Gomez Hoyos
 April 2015 Sterilization of narrow channels: how to ensure effectiveness in daily routine? Dr.ir. Josephus Paulus Clemens Maria Van Doornmalen Gomez Hoyos 3M Deutschand GmbH, IPD, Decontamination University
April 2015 Sterilization of narrow channels: how to ensure effectiveness in daily routine? Dr.ir. Josephus Paulus Clemens Maria Van Doornmalen Gomez Hoyos 3M Deutschand GmbH, IPD, Decontamination University
Lab: Observing Osmosis in Gummi Bears
 Name Period Date Points Lab: Observing Osmosis in Gummi Bears Haribo macht Kinder froh und Erwachsene ebenso! 1 Laboratory: Observing Osmosis in Gummy Bears (28 points) Purpose: To investigate the movement
Name Period Date Points Lab: Observing Osmosis in Gummi Bears Haribo macht Kinder froh und Erwachsene ebenso! 1 Laboratory: Observing Osmosis in Gummy Bears (28 points) Purpose: To investigate the movement
SENTRY AIR SYSTEMS, INC. PHARMACEUTICAL COMPOUNDING CONTAINMENT SYSTEMS SIMPLE SOLUTIONS FOR CLEANER AIR. www.sentryair.com 1.800.799.
 SENTRY AIR SYSTEMS, INC. PHARMACEUTICAL COMPOUNDING CONTAINMENT SYSTEMS SIMPLE SOLUTIONS FOR CLEANER AIR 1.800.799.4609 www.sentryair.com Pharmaceutical Compounding Safety Solutions Sentry Air Systems
SENTRY AIR SYSTEMS, INC. PHARMACEUTICAL COMPOUNDING CONTAINMENT SYSTEMS SIMPLE SOLUTIONS FOR CLEANER AIR 1.800.799.4609 www.sentryair.com Pharmaceutical Compounding Safety Solutions Sentry Air Systems
The Real Costs of Surgical Instrument Training in Sterile Processing Revisited NANCY CHOBIN, RN, AAS, ACSP, CSPDM
 The Real Costs of Surgical Instrument Training in Sterile Processing Revisited NANCY CHOBIN, RN, AAS, ACSP, CSPDM ABSTRACT The need for properly trained sterile processing staff members has grown with
The Real Costs of Surgical Instrument Training in Sterile Processing Revisited NANCY CHOBIN, RN, AAS, ACSP, CSPDM ABSTRACT The need for properly trained sterile processing staff members has grown with
LOW-TEMPERATURE STERILIZATION IS AN EFFECTIVE MEANS TO
 CRCST Self-Study Lesson Plan Lesson No. CRCST 138 (Technical Continuing Education - TCE) Sponsored by: by Susan Klacik, ACE, CHL, CRCST, CIS, FCS, CSS Manager, St. Elizabeth Health Center, Youngstown,
CRCST Self-Study Lesson Plan Lesson No. CRCST 138 (Technical Continuing Education - TCE) Sponsored by: by Susan Klacik, ACE, CHL, CRCST, CIS, FCS, CSS Manager, St. Elizabeth Health Center, Youngstown,
Because You Can t See Sterile.SM
 Because You Can t See Sterile.SM Sterilization EVERY LOAD. Sure-Check Sterilization Pouches 3 / 29 PMS 201C EVERY DAY. 50% BLACK STEAMPlus Class 5 Sterilization Integrators EVERY WEEK. ConFirm 10 In-Office
Because You Can t See Sterile.SM Sterilization EVERY LOAD. Sure-Check Sterilization Pouches 3 / 29 PMS 201C EVERY DAY. 50% BLACK STEAMPlus Class 5 Sterilization Integrators EVERY WEEK. ConFirm 10 In-Office
Medical Market Laboratory EMEA
 Medical Market Laboratory EMEA Space for 3M Montage Penetration tests according to the ISO 11140 Josephus P.C.M van Doornmalen Gomez Hoyos Croatia, March 27, 2009 Outline Purpose penetration tests Original
Medical Market Laboratory EMEA Space for 3M Montage Penetration tests according to the ISO 11140 Josephus P.C.M van Doornmalen Gomez Hoyos Croatia, March 27, 2009 Outline Purpose penetration tests Original
Laboratory Line. Vertical and Bench-Top Life Science Autoclaves
 Laboratory Line Vertical and Bench-Top Life Science Autoclaves The Advanced Laboratory Line The Full Spectrum of Laboratory Sterilizers Sterilization in a laboratory environment has its unique requirements.
Laboratory Line Vertical and Bench-Top Life Science Autoclaves The Advanced Laboratory Line The Full Spectrum of Laboratory Sterilizers Sterilization in a laboratory environment has its unique requirements.
Body Art Facility Infection Prevention And Control Plan Guideline
 Body Art Facility Infection Prevention And Control Plan Guideline In accordance with the California Health and Safety Code, Section 119313, a body art facility shall maintain and follow a written Infection
Body Art Facility Infection Prevention And Control Plan Guideline In accordance with the California Health and Safety Code, Section 119313, a body art facility shall maintain and follow a written Infection
Getting a Handle on Loaner Instrumentation
 Getting a Handle on Loaner Instrumentation Essentials for a successful loaner program by Rose Seavey, RN, MBA, CNOR, CRCST, CSPDT Objectives After completion of this self-study activity, the learner will
Getting a Handle on Loaner Instrumentation Essentials for a successful loaner program by Rose Seavey, RN, MBA, CNOR, CRCST, CSPDT Objectives After completion of this self-study activity, the learner will
Using Observation and Audit Tools to Improve Quality Outcomes
 Using Observation and Audit Tools to Improve Quality Outcomes by Cynthia Hubbard, RN, BS Objectives After completion of this self-study activity the learner will be able to: 1. Identify the infection risk
Using Observation and Audit Tools to Improve Quality Outcomes by Cynthia Hubbard, RN, BS Objectives After completion of this self-study activity the learner will be able to: 1. Identify the infection risk
STATIM G4 Cassette Autoclaves From start to dry in the blink of an eye.
 STATIM G4 Cassette Autoclaves From start to dry in the blink of an eye. STATIM, the world s fastest autoclave from start to sterile Drawing on over 20 years of innovation, Healthcare practitioners in over
STATIM G4 Cassette Autoclaves From start to dry in the blink of an eye. STATIM, the world s fastest autoclave from start to sterile Drawing on over 20 years of innovation, Healthcare practitioners in over
Guidance on safe use of Autoclaves
 Safety Office Reviewed: Dec 2013 Reviewed: DD Month Year Guidance on safe use of Autoclaves 1. Hazards & Uses There are several different types of hazard associated with the use of autoclaves. The main
Safety Office Reviewed: Dec 2013 Reviewed: DD Month Year Guidance on safe use of Autoclaves 1. Hazards & Uses There are several different types of hazard associated with the use of autoclaves. The main
Microbiological Evaluation of the STI Series 2000 Medical Waste Treatment Process
 WNWN International,Inc. WNWN International Phone: 860-675-1217 Fax 860-675-1311 PO Box 1164 Burlington, CT. 06013 USA Microbiological Evaluation of the STI Series 2000 Medical Waste Treatment Process January
WNWN International,Inc. WNWN International Phone: 860-675-1217 Fax 860-675-1311 PO Box 1164 Burlington, CT. 06013 USA Microbiological Evaluation of the STI Series 2000 Medical Waste Treatment Process January
Copyright 2011 Casa Software Ltd. www.casaxps.com. Centre of Mass
 Centre of Mass A central theme in mathematical modelling is that of reducing complex problems to simpler, and hopefully, equivalent problems for which mathematical analysis is possible. The concept of
Centre of Mass A central theme in mathematical modelling is that of reducing complex problems to simpler, and hopefully, equivalent problems for which mathematical analysis is possible. The concept of
Revised 4-3-2012 EHS Biosafety. 1 Select appropriate containers/bags for autoclaving.
 Requirements for Decontamination by Autoclaving Summary: Steam sterilization (autoclaving) is one of the most effective methods for decontaminating biohazardous material. Autoclaves use saturated steam
Requirements for Decontamination by Autoclaving Summary: Steam sterilization (autoclaving) is one of the most effective methods for decontaminating biohazardous material. Autoclaves use saturated steam
Open-cell spray foam & what every contractor
 Open-cell spray foam & what every contractor should know Overview Open-cell or Closed-cell? The age old question that many stumped - from industry bloggers to homeowners - is whether to choose opencell
Open-cell spray foam & what every contractor should know Overview Open-cell or Closed-cell? The age old question that many stumped - from industry bloggers to homeowners - is whether to choose opencell
Sterilization Packaging Systems, Preparation and Loading for Steam Sterilization
 Sterilization Packaging Systems, Preparation and Loading for Steam Sterilization by Rose Seavey, RN, MBA, CNOR, ACSP Many thanks to the team at 3M Health Care for working with Managing Infection Contro
Sterilization Packaging Systems, Preparation and Loading for Steam Sterilization by Rose Seavey, RN, MBA, CNOR, ACSP Many thanks to the team at 3M Health Care for working with Managing Infection Contro
Effective Heat Sterilization in CO 2 Incubators
 Effective Heat Sterilization in CO 2 Incubators Volume 4, Number 3 Key Words Key Words Class 100 air Contamination control Contamination elimination HEPA filtration Incubator Heat Sterilization White Paper
Effective Heat Sterilization in CO 2 Incubators Volume 4, Number 3 Key Words Key Words Class 100 air Contamination control Contamination elimination HEPA filtration Incubator Heat Sterilization White Paper
Reprocessing. Critical & Semi-Critical Equipment
 Reprocessing (Cleaning, Disinfection & Sterilization) Critical & Semi-Critical Equipment - A Physician Toolkit - Reprocessing Critical & Semi-Critical Equipment - A Physician Tool Kit Table of Contents
Reprocessing (Cleaning, Disinfection & Sterilization) Critical & Semi-Critical Equipment - A Physician Toolkit - Reprocessing Critical & Semi-Critical Equipment - A Physician Tool Kit Table of Contents
Improving the Quality of Surgical Trays. By: Stephen M. Kovach
 Improving the Quality of Surgical Trays By: Stephen M. Kovach Performance measurement in healthcare represents what is done and how well it is done. The goal is to accurately understand the basis for current
Improving the Quality of Surgical Trays By: Stephen M. Kovach Performance measurement in healthcare represents what is done and how well it is done. The goal is to accurately understand the basis for current
Medical Market Laboratory EMEA
 Medical Market Laboratory EMEA Space for 3M Montage Penetration tests in according to the ISO 11140 Josephus P.C.M van Doornmalen Gomez Hoyos Croatia, March 27, 2009 Outline Purpose penetration tests Original
Medical Market Laboratory EMEA Space for 3M Montage Penetration tests in according to the ISO 11140 Josephus P.C.M van Doornmalen Gomez Hoyos Croatia, March 27, 2009 Outline Purpose penetration tests Original
STANDARD OPERATING PROCEDURES Safe Autoclave Operations
 Department of Environmental Health & Safety Biosafety & Biosecurity STANDARD OPERATING PROCEDURES Safe Autoclave Operations The purpose of this document is to provide standard operating procedures for
Department of Environmental Health & Safety Biosafety & Biosecurity STANDARD OPERATING PROCEDURES Safe Autoclave Operations The purpose of this document is to provide standard operating procedures for
LABOKLAV. Autoclaves Made in Germany. www.shp-steriltechnik.de
 LABOKLAV Autoclaves Made in Germany www.shp-steriltechnik.de 2 LABOKLAV STEAM STERILIZERS Steam sterilization is the sterilization method with the widest range of applications efficient, safe, reproducible
LABOKLAV Autoclaves Made in Germany www.shp-steriltechnik.de 2 LABOKLAV STEAM STERILIZERS Steam sterilization is the sterilization method with the widest range of applications efficient, safe, reproducible
Heating oven with mechanical control
 ED series 53 Drying ovens / Heating ovens Heating oven with mechanical control The strength of the BINDER ED series heating oven: routine drying and sterilization applications up to 300 C (572 F). Thanks
ED series 53 Drying ovens / Heating ovens Heating oven with mechanical control The strength of the BINDER ED series heating oven: routine drying and sterilization applications up to 300 C (572 F). Thanks
STERILIZATION WRAP. STERISHEET user guide for the validation of the packaging THE BACTERIAL BARRIER FOR HUMAN PROTECTION
 STERILIZATION WRAP STERISHEET user guide for the validation of the packaging ARJOWIGGINS in few words A large range of sterilization wrap from standard creped paper to non-woven A large range of standard
STERILIZATION WRAP STERISHEET user guide for the validation of the packaging ARJOWIGGINS in few words A large range of sterilization wrap from standard creped paper to non-woven A large range of standard
The Supplier Perspective: Sterilization. Presented by: Thad Wroblewski STERIS Isomedix Services
 The Supplier Perspective: Sterilization Presented by: Thad Wroblewski STERIS Isomedix Services Agenda Introduction Contractor selection criteria Pre-planning with sterilization in mind Key elements of
The Supplier Perspective: Sterilization Presented by: Thad Wroblewski STERIS Isomedix Services Agenda Introduction Contractor selection criteria Pre-planning with sterilization in mind Key elements of
FDA and the Compounding Pharmacy
 FDA and the Compounding Pharmacy Scott Sutton, Ph.D. scott.sutton@microbiol.org 41 Overview of Presentation The Recent Events GCP and GMP Basics the 483 Review H.R. 3204 Outsourcing Facility Preparation
FDA and the Compounding Pharmacy Scott Sutton, Ph.D. scott.sutton@microbiol.org 41 Overview of Presentation The Recent Events GCP and GMP Basics the 483 Review H.R. 3204 Outsourcing Facility Preparation
Accurate Convenient Affordable Results in 24 Hours. Mail-to-Lab Spore Testing. Mail-to-Lab Spore Testing
 How Mail-To-Lab Spore Testing Works: Sign up for a mail-in test kit by contacting ATS. A custom kit will be shipped to your facility. Each spore is accompanied with a test form, Class 4 chemical indicator,
How Mail-To-Lab Spore Testing Works: Sign up for a mail-in test kit by contacting ATS. A custom kit will be shipped to your facility. Each spore is accompanied with a test form, Class 4 chemical indicator,
2. Laboratory equipment
 2. Laboratory equipment The choice of laboratory equipment must take into account national regulations and the technical premises that determine the requirements for its appropriate use. The more com plex
2. Laboratory equipment The choice of laboratory equipment must take into account national regulations and the technical premises that determine the requirements for its appropriate use. The more com plex
Federal Wage System Job Grading Standard For Laboratory Working, 3511. Table of Contents
 Federal Wage System Job Grading Standard For Laboratory Working, 3511 Table of Contents WORK COVERED... 2 WORK NOT COVERED...2 TITLES... 2 GRADE LEVELS... 2 NOTE TO USERS... 3 LABORATORY WORKER, GRADE
Federal Wage System Job Grading Standard For Laboratory Working, 3511 Table of Contents WORK COVERED... 2 WORK NOT COVERED...2 TITLES... 2 GRADE LEVELS... 2 NOTE TO USERS... 3 LABORATORY WORKER, GRADE
EUROTUBO DELTALAB 4. PETRI DISHES AND LOOPS
 77 78 90 x 14 mm Petri Dish Made in polystyrene. Vented. Supplied in groups of 20 units, packaged in heat sealed bags. Code 200200 is ASEPTIC. Code 200209 is sterile by gamma radiation. Suitable for automatic
77 78 90 x 14 mm Petri Dish Made in polystyrene. Vented. Supplied in groups of 20 units, packaged in heat sealed bags. Code 200200 is ASEPTIC. Code 200209 is sterile by gamma radiation. Suitable for automatic
TECHNIQUES FOR NATURAL GAS SAMPLING A DISCUSSION OF FIELD METHODS FOR OBTAINING SPOT SAMPLES. Randall Messman SW Regional Sales Manager
 TECHNIQUES FOR NATURAL GAS SAMPLING A DISCUSSION OF FIELD METHODS FOR OBTAINING SPOT SAMPLES Randall Messman SW Regional Sales Manager PGI International 16101 Vallen Drive Houston, TX 77041 Purpose Natural
TECHNIQUES FOR NATURAL GAS SAMPLING A DISCUSSION OF FIELD METHODS FOR OBTAINING SPOT SAMPLES Randall Messman SW Regional Sales Manager PGI International 16101 Vallen Drive Houston, TX 77041 Purpose Natural
Micron-Master EL Series Fogger
 Micron-Master EL Series Fogger Instruction Manual Please read and keep these instructions For Models: EL2 Wall Mount EL3 Wall Mount EL4 Wall Mount EL2 Cart Mount EL3 Cart Mount EL4 Cart Mount EL3 Generator
Micron-Master EL Series Fogger Instruction Manual Please read and keep these instructions For Models: EL2 Wall Mount EL3 Wall Mount EL4 Wall Mount EL2 Cart Mount EL3 Cart Mount EL4 Cart Mount EL3 Generator
A GUIDELINE FOR THE SAFE USE OF AUTOCLAVES UNIVERSITY OF OTTAWA. Environmental Health and Safety Service
 A GUIDELINE FOR THE SAFE USE OF AUTOCLAVES UNIVERSITY OF OTTAWA Environmental Health and Safety Service Last date of revision July 9, 2003 TABLE OF CONTENT 1.0 PURPOSE...1 2.0 INTRODUCTION...1 3.0 PERSONAL
A GUIDELINE FOR THE SAFE USE OF AUTOCLAVES UNIVERSITY OF OTTAWA Environmental Health and Safety Service Last date of revision July 9, 2003 TABLE OF CONTENT 1.0 PURPOSE...1 2.0 INTRODUCTION...1 3.0 PERSONAL
Fact Sheet on Steam Sterilizers at Stanford University
 Fact Sheet on Steam Sterilizers at Stanford University By: Environmental Quality and Water Efficiency Group, Stanford University; December 2013 Jennifer Fitch, Marty Laporte, Julia Nussbaum, Adam Kern,
Fact Sheet on Steam Sterilizers at Stanford University By: Environmental Quality and Water Efficiency Group, Stanford University; December 2013 Jennifer Fitch, Marty Laporte, Julia Nussbaum, Adam Kern,
ReadyKart Mobile Processing Station Instructions for Use
 GE Healthcare Instructions 28-9606-47 AB ReadyKart Mobile Processing Station Instructions for Use Contents ReadyToProcess 1 Introduction... 1 2 General Description... 2 3 Description of Components... 4
GE Healthcare Instructions 28-9606-47 AB ReadyKart Mobile Processing Station Instructions for Use Contents ReadyToProcess 1 Introduction... 1 2 General Description... 2 3 Description of Components... 4
Packaging Validation according to ISO 11607
 Synergy makes sence Packaging Validation according to ISO 11607 MG-FSI72-104 Last revision: March 2011 5, Chemin du Catupolan - 69120 Vaulx en Velin - France - Tel. 33 (0)4 72 81 22 62 - Fax : 33 (0)4
Synergy makes sence Packaging Validation according to ISO 11607 MG-FSI72-104 Last revision: March 2011 5, Chemin du Catupolan - 69120 Vaulx en Velin - France - Tel. 33 (0)4 72 81 22 62 - Fax : 33 (0)4
Case Study in Test Method Validation - ASTM F1886 (2004), Visual Examination
 Case Study in Test Method Validation - ASTM F1886 (2004), Visual Examination Todd Dehmer CPP Senior Packaging Engineer St. Jude Medical, AFD Division March 3-5, 2009 www.healthpack.net 1 Presentation Overview
Case Study in Test Method Validation - ASTM F1886 (2004), Visual Examination Todd Dehmer CPP Senior Packaging Engineer St. Jude Medical, AFD Division March 3-5, 2009 www.healthpack.net 1 Presentation Overview
Vertical and Benchtop Autoclaves for Life Sciences. Laboratory Line
 Vertical and Benchtop Autoclaves for Life Sciences Laboratory Line Designed for Your Laboratory Applications Advanced Laboratory Autoclaves Tuttnauer laboratory autoclaves have been designed to provide
Vertical and Benchtop Autoclaves for Life Sciences Laboratory Line Designed for Your Laboratory Applications Advanced Laboratory Autoclaves Tuttnauer laboratory autoclaves have been designed to provide
Receiving and Storage
 Receiving and Storage Lesson 10 Overview Receiving and Storage Time 30 minutes Purpose To explain the relationship between receiving, food storage, and food safety Objectives At the conclusion of this
Receiving and Storage Lesson 10 Overview Receiving and Storage Time 30 minutes Purpose To explain the relationship between receiving, food storage, and food safety Objectives At the conclusion of this
Electric Kettle. Model: SK-1717 INSTRUCTIONS MANUAL
 Electric Kettle Model: SK-1717 INSTRUCTIONS MANUAL Thank you for your purchase. Please read this Instruction Manual thoroughly before use and keep in a safe place for future reference. READ ALL INSTRUCTIONS
Electric Kettle Model: SK-1717 INSTRUCTIONS MANUAL Thank you for your purchase. Please read this Instruction Manual thoroughly before use and keep in a safe place for future reference. READ ALL INSTRUCTIONS
Sterilization A REVIEW OF THE BASICS. By Gayla Tilton and Myrna Kauffman 66 MANAGING INFECTION CONTROL
 Sterilization A REVIEW OF THE BASICS By Gayla Tilton and Myrna Kauffman 66 MANAGING INFECTION CONTROL Although sterilization has been performed in healthcare facilities for many decades, the process is
Sterilization A REVIEW OF THE BASICS By Gayla Tilton and Myrna Kauffman 66 MANAGING INFECTION CONTROL Although sterilization has been performed in healthcare facilities for many decades, the process is
Sample Pharmacy CLEANING AND SANITIZING PROCEDURE P-304.1
 P-304.1 1.0 Definition and Purpose: To standardize and define the process by which the controlled Cleanroom environments (ISO Class 5, 7, and 8) and the general pharmacy prep area is cleaned, disinfected,
P-304.1 1.0 Definition and Purpose: To standardize and define the process by which the controlled Cleanroom environments (ISO Class 5, 7, and 8) and the general pharmacy prep area is cleaned, disinfected,
Bench Autoclave. Standard Operating Procedure. For Installation, Use and Maintenance
 Bench Autoclave Standard Operating Procedure For Installation, Use and Maintenance 1. Introduction This SOP is intended for use with the following model, in a laboratory context: Type: Nuve Bench Top Steam
Bench Autoclave Standard Operating Procedure For Installation, Use and Maintenance 1. Introduction This SOP is intended for use with the following model, in a laboratory context: Type: Nuve Bench Top Steam
EPSON Perfection 2450 PHOTO. Scanner Parts. Scanner Specifications. Basic Specifications. US letter or A4 size (8.5 11.7 inches [216 297 mm])
![EPSON Perfection 2450 PHOTO. Scanner Parts. Scanner Specifications. Basic Specifications. US letter or A4 size (8.5 11.7 inches [216 297 mm]) EPSON Perfection 2450 PHOTO. Scanner Parts. Scanner Specifications. Basic Specifications. US letter or A4 size (8.5 11.7 inches [216 297 mm])](/thumbs/40/21209055.jpg) Scanner Parts Start button power button (TPU) under lid IEEE 1394 port Scanner Specifications Basic Specifications Scanner type Photoelectric device Effective pixels USB port indicator light document cover
Scanner Parts Start button power button (TPU) under lid IEEE 1394 port Scanner Specifications Basic Specifications Scanner type Photoelectric device Effective pixels USB port indicator light document cover
BESTPRACTICE. HTM01-05 Q&A: You can now follow us on Twitter! We have launched our HTM01-05 questions and answers service on Twitter.
 ISSUE 4 BESTPRACTICE For Essential Requirements, hand washing is still a viable method of cleaning your instruments, but how do you validate for compliance with HTM01-05? Ensuring your instruments are
ISSUE 4 BESTPRACTICE For Essential Requirements, hand washing is still a viable method of cleaning your instruments, but how do you validate for compliance with HTM01-05? Ensuring your instruments are
MELA quick 12+ Compact. Fast. Strong.
 MELA quick 12+ Compact. Fast. Strong. The requirement. Requirements for the preparation of dental instruments are defined by official hygiene experts. Instruments for semicritical use (restorative treatment)
MELA quick 12+ Compact. Fast. Strong. The requirement. Requirements for the preparation of dental instruments are defined by official hygiene experts. Instruments for semicritical use (restorative treatment)
Understanding High Perm vs. Low Perm
 Understanding High Perm vs. Low Perm Building Science Bulletin Figure A High perm(eability) vs. low perm(eability) within the housewrap category what does it mean and how does it affect your building practices?
Understanding High Perm vs. Low Perm Building Science Bulletin Figure A High perm(eability) vs. low perm(eability) within the housewrap category what does it mean and how does it affect your building practices?
Sterile Processing University, LLC MODULE 25: INVENTORY CONTROL & MANAGEMENT [Ambulatory Surgery]
![Sterile Processing University, LLC MODULE 25: INVENTORY CONTROL & MANAGEMENT [Ambulatory Surgery] Sterile Processing University, LLC MODULE 25: INVENTORY CONTROL & MANAGEMENT [Ambulatory Surgery]](/thumbs/34/13094255.jpg) Sterile Processing University, LLC MODULE 25: INVENTORY CONTROL & MANAGEMENT [Ambulatory Surgery] Copyright Sterile Processing University, LLC 2012 All Rights Reserved. This material may not be copied
Sterile Processing University, LLC MODULE 25: INVENTORY CONTROL & MANAGEMENT [Ambulatory Surgery] Copyright Sterile Processing University, LLC 2012 All Rights Reserved. This material may not be copied
ISO Training Program
 ISO Training Program A Quality Control Program for Central Service Supported by New Jersey Department of Health and Senior Services and New Jersey Health Care Central Service Association and Mid-Atlantic
ISO Training Program A Quality Control Program for Central Service Supported by New Jersey Department of Health and Senior Services and New Jersey Health Care Central Service Association and Mid-Atlantic
STEAM STERILIZERS. DLOV & DLOM series. more than fifty years of experience in sterilization. DE LAMA s Quality Management System is certified:
 STEAM STERILIZERS DLOV & DLOM series DE LAMA s Quality Management System is certified: more than fifty years of experience in sterilization UNI EN ISO 9001:2000 - Cert. n 0351 UNI EN ISO 13485 - Cert.
STEAM STERILIZERS DLOV & DLOM series DE LAMA s Quality Management System is certified: more than fifty years of experience in sterilization UNI EN ISO 9001:2000 - Cert. n 0351 UNI EN ISO 13485 - Cert.
Sterile E Online NetworkTutorials
 3M Sterilization Assurance Products EO Blue bar must enter area Sterile E Online NetworkTutorials EO Blue bar must enter area Understanding the Differences Between Class 5 s and Class 6 Emulating Indicators
3M Sterilization Assurance Products EO Blue bar must enter area Sterile E Online NetworkTutorials EO Blue bar must enter area Understanding the Differences Between Class 5 s and Class 6 Emulating Indicators
CENTRAL SERVICE REGULATIONS FOR AMBULATORY SURGERY CENTERS
 CENTRAL SERVICE REGULATIONS FOR AMBULATORY SURGERY CENTERS Last updated July 2014 Legal disclaimer: The International Association of Healthcare Central Service Materiel Management (IAHCSMM) provides the
CENTRAL SERVICE REGULATIONS FOR AMBULATORY SURGERY CENTERS Last updated July 2014 Legal disclaimer: The International Association of Healthcare Central Service Materiel Management (IAHCSMM) provides the
Oxivir Tb and Accelerated Hydrogen Peroxide (AHP) Frequently Asked Questions
 Oxivir Tb and Accelerated Hydrogen Peroxide (AHP) Frequently Asked Questions What is Accelerated Hydrogen Peroxide (AHP)? AHP is a patented synergistic blend of commonly used, safe ingredients that when
Oxivir Tb and Accelerated Hydrogen Peroxide (AHP) Frequently Asked Questions What is Accelerated Hydrogen Peroxide (AHP)? AHP is a patented synergistic blend of commonly used, safe ingredients that when
Xerox Supplies Recycling Program Instructions
 Xerox Supplies Recycling Program Instructions This document outlines the instructions on how to create a return shipment on Xerox.ca Step 1 - Login to www.xerox.ca/about-xerox/recycling/enca.html Go to
Xerox Supplies Recycling Program Instructions This document outlines the instructions on how to create a return shipment on Xerox.ca Step 1 - Login to www.xerox.ca/about-xerox/recycling/enca.html Go to
SUPPLEMENTARY MATERIAL
 SUPPLEMENTARY MATERIAL (Student Instructions) Determination of the Formula of a Hydrate A Greener Approach Objectives To experimentally determine the formula of a hydrate salt. To learn to think in terms
SUPPLEMENTARY MATERIAL (Student Instructions) Determination of the Formula of a Hydrate A Greener Approach Objectives To experimentally determine the formula of a hydrate salt. To learn to think in terms
E X P E R I M E N T 8
 E X P E R I M E N T 8 Torque, Equilibrium & Center of Gravity Produced by the Physics Staff at Collin College Copyright Collin College Physics Department. All Rights Reserved. University Physics, Exp 8:
E X P E R I M E N T 8 Torque, Equilibrium & Center of Gravity Produced by the Physics Staff at Collin College Copyright Collin College Physics Department. All Rights Reserved. University Physics, Exp 8:
Be on the Look-Out for These Top-10 risks in Sterile Processing
 Be on the Look-Out for These Top-10 risks in Sterile Processing Martha Young, BS, MS, CSPDT Martha L. Young, LLC, Woodbury, MN Providing Savvy Sterilization Solutions for Healthcare marthalyoung1@aol.com
Be on the Look-Out for These Top-10 risks in Sterile Processing Martha Young, BS, MS, CSPDT Martha L. Young, LLC, Woodbury, MN Providing Savvy Sterilization Solutions for Healthcare marthalyoung1@aol.com
How to Prepare Powdered Infant Formula in Care Settings
 How to Prepare Powdered Infant Formula in Care Settings This booklet contains new information to help you prepare powdered infant formula for bottle-feeding and cup-feeding as safely as possible. Powdered
How to Prepare Powdered Infant Formula in Care Settings This booklet contains new information to help you prepare powdered infant formula for bottle-feeding and cup-feeding as safely as possible. Powdered
ARCO Electric Products Installation and Maintenance Manual Low Voltage Automatic Power Factor Correction Capacitor Systems 2013
 ARCO Electric Products Installation and Maintenance Manual Low Voltage Automatic Power Factor Correction Capacitor Systems 2013 READ CAREFULLY These instructions are intended to cover good practices in
ARCO Electric Products Installation and Maintenance Manual Low Voltage Automatic Power Factor Correction Capacitor Systems 2013 READ CAREFULLY These instructions are intended to cover good practices in
OCCUPATIONAL SAFETY AND ENVIRONMENTAL HEALTH GUIDELINE
 OSEH Occupational Safety & Environmental Health OCCUPATIONAL SAFETY AND ENVIRONMENTAL HEALTH GUIDELINE Subject: Biohazardous (Medical) Waste Disposal Date: 08/19/09 Revision: 03 Page: 1 of 7 TABLE OF Section
OSEH Occupational Safety & Environmental Health OCCUPATIONAL SAFETY AND ENVIRONMENTAL HEALTH GUIDELINE Subject: Biohazardous (Medical) Waste Disposal Date: 08/19/09 Revision: 03 Page: 1 of 7 TABLE OF Section
AUTOCLAVE STEAM STERILIZERS
 AUTOCLAVE STEAM STERILIZERS (BENCH TOP) Image: HS-1606VD Hanshin Bench Top Steam Sterilizer Small and easy to operate Three preset sterilization cycles Adjustable cycle presets Rapid heat up times Option
AUTOCLAVE STEAM STERILIZERS (BENCH TOP) Image: HS-1606VD Hanshin Bench Top Steam Sterilizer Small and easy to operate Three preset sterilization cycles Adjustable cycle presets Rapid heat up times Option
DIESEL FUEL CONDITIONING
 COD FILTRATION DIESEL FUEL CONDITIONING REMOVE WATER & PARTICLES TO EXTEND FUEL INJECTOR LIFE & INCREASE COMBUSTION ENGINE FUEL EFFICIENCY Remove free water to saturation point with high single pass efficiency
COD FILTRATION DIESEL FUEL CONDITIONING REMOVE WATER & PARTICLES TO EXTEND FUEL INJECTOR LIFE & INCREASE COMBUSTION ENGINE FUEL EFFICIENCY Remove free water to saturation point with high single pass efficiency
What is C-TPAT? Customs Trade Partnership Against Terrorism
 Learning Objectives What is C-TPAT? Terrorist Awareness Supply Chain Security Container Security Program 7 Point Inspection Seal Program Locks, Keys & Access Control / Visitor Controls Pre-Employment Verifications
Learning Objectives What is C-TPAT? Terrorist Awareness Supply Chain Security Container Security Program 7 Point Inspection Seal Program Locks, Keys & Access Control / Visitor Controls Pre-Employment Verifications
Biological Sciences Initiative
 Biological Sciences Initiative HHMI Student Activities Measuring Antibiotic Resistance Introduction: You might be aware that antibiotics were once thought of as a magic bullet; a nearly perfect drug for
Biological Sciences Initiative HHMI Student Activities Measuring Antibiotic Resistance Introduction: You might be aware that antibiotics were once thought of as a magic bullet; a nearly perfect drug for
Medical Waste Autoclaves. Medical Waste Autoclaves
 Medical Waste Autoclaves Medical Waste Autoclaves Safe, Cost-Effective, Medical Waste Sterilization Treat Medical Waste with high performance Tuttnauer Medical Waste autoclave systems designed for small,
Medical Waste Autoclaves Medical Waste Autoclaves Safe, Cost-Effective, Medical Waste Sterilization Treat Medical Waste with high performance Tuttnauer Medical Waste autoclave systems designed for small,
www.gbo.com/bioscience Tissue Culture 1 Cell/ Microplates 2 HTS- 3 Immunology/ HLA 4 Microbiology/ Bacteriology Purpose Beakers 5 Tubes/Multi-
 www.gbo.com/bioscience 11 Cryotechnics Technical Information 11 I 2 Cryo.s TM 11 I 3 Cryo.s TM 1 ml 11 I 3 Cryo.s TM 2 ml 11 I 3 Cryo.s TM 4 ml 11 I 4 Cryo.s TM 5 ml 11 I 4 Support Rack 11 I 5 Cryo Storage
www.gbo.com/bioscience 11 Cryotechnics Technical Information 11 I 2 Cryo.s TM 11 I 3 Cryo.s TM 1 ml 11 I 3 Cryo.s TM 2 ml 11 I 3 Cryo.s TM 4 ml 11 I 4 Cryo.s TM 5 ml 11 I 4 Support Rack 11 I 5 Cryo Storage
