Add-Aspirin Clinical Trial We are inviting you to take part in a research study called the Add-Aspirin trial
|
|
|
- Amberlynn Bryan
- 7 years ago
- Views:
Transcription
1 (To be presented on local headed paper) Version 4.0, 11-Nov-2014 Add-Aspirin Clinical Trial We are inviting you to take part in a research study called the Add-Aspirin trial Before you decide whether to take part, it is important for you to understand why the research is being done and what it will involve. Please take time to read the following information carefully. Discuss it with friends and relatives if you wish. Take time to decide if you wish to take part. You are free to decide if you want to take part in this research study. If you choose not to take part, this will not affect the care you get from your own doctors in any way. You can decide to stop taking part in the study at any time without giving a reason. Ask your study doctor if anything is not clear or if you would like more information. Thank you for reading this information. If you decide to take part, we will ask you to sign a form Part to ONE give your consent for each part of the study. Important things that you need to know We are testing whether taking aspirin regularly after treatment for early stage cancer stops or delays the cancer coming back. We are testing different doses of aspirin. Some people will receive a dummy drug (placebo). Like all medicines, aspirin can have side-effects. The most common side-effects of aspirin are: - irritation of the stomach - indigestion - bleeding - this a serious side-effect of aspirin but it is uncommon. We expect that most people who take part in this study will need to visit the hospital approximately 12 times over 5 years depending on your cancer type. Whenever possible, this will be at the same time as your regular check-up visit. We will also ask if you would like to donate a small amount of blood, urine and tissue from your cancer for future research. Contents 1 Why are we doing this study? 2 Why am I being asked to take part? 3 What do I need to know about the medicines used in this study? 4 What will I need to do if I take part? 5 What are the possible sideeffects? 6 Possible advantages and disadvantages of taking part 7 Samples and genetic tests 8 More information about taking part 9 Contacts for further information How to contact us If you have any questions about this study, please talk to your doctor at Hospital Department Hospital Address Address Tel: XXX XXX Page 1 of 8
2 1Why are we doing this study? 2 Why am I being asked to take part? This study is for people who have had or have started treatment for cancer of the breast, stomach, oesophagus (food pipe), prostate or bowel. We are aiming to find out whether taking aspirin regularly after treatment for cancers that have not spread widely (early stage cancer), stops or delays the cancer coming back. This study will compare groups of people who take aspirin and those who take placebo tablets. What is a placebo? A placebo is a dummy tablet. It looks like an aspirin tablet but it doesn t contain any medicine. We are using a placebo in this study so that we can make as clear an assessment as possible about the effects of aspirin. Keeping everything exactly the same apart from what is contained in the tablet allows us to study just the effects of aspirin. The placebo used in Add-Aspirin contains lactose. People who are lactose intolerant will be excluded from taking part in the study. What is the usual treatment? Usually, people like you, who have had or have started treatment for early stage cancer, will come into hospital for regular check-ups once the treatment has finished. People who have had prostate cancer sometimes also take long-term hormone treatment. People who have had breast cancer sometimes also take long-term hormone treatment or Herceptin. This will continue if you take part in the study. People who have had stomach, oesophagus or bowel cancer don t usually take any other long-term treatments. You are being invited to take part in the Add- Aspirin study because you have had or are having treatment for cancer of the breast, stomach, oesophagus, prostate or bowel. Although your doctors believe that they are able to successfully treat your cancer, some cancers unfortunately may come back. This study will find out whether or not taking aspirin regularly after treatment can stop or delay this happening. 3 What do I need to know about the medicines used in this study? Aspirin is a common drug that is used as a painkiller and to prevent further heart attacks and strokes in some people. Some studies have suggested that people who regularly take aspirin may be less likely to be diagnosed with cancer than those who don t take aspirin. Also, in studies testing the beneficial effect of aspirin on heart disease, aspirin appeared to reduce the number of people who developed cancer, and if people did develop cancer, it appeared to be less likely to spread. So researchers believe that aspirin may stop cancer coming back in people who have had treatment for early stage cancer. But, importantly, there is not any reliable evidence yet since previous studies were not specifically designed to answer this question. A clinical trial or study is the best way to test this. It will look at both the benefits and the side-effects of taking aspirin in a large group of people who have had cancer. The study will look at two different doses of aspirin. This is because little is known about how much aspirin may be needed to have an effect, if any, against cancer. Page 2 of 8
3 4 What will I need to do if I take part? If you agree to take part in this study, you will have a blood test and be asked questions about your medical history to check your suitability to take part. In the study, you will be asked to take the study medicines for at least 5 years. We know that it can be difficult to take medicines every day over a long period so everyone will be asked to take 100mg of aspirin for about 8 weeks at the beginning of the study. This is called the runin period and will help us to identify people who may have problems taking aspirin. If you find it difficult to take aspirin regularly, it may not be recommended that you continue. After 8 weeks, if taking aspirin every day hasn t caused you any problems, you will then be randomly allocated by computer to one of the three treatments: mg of aspirin every day (one tablet per day) or mg of aspirin every day (one tablet per day) or 3. A placebo tablet every day (one tablet per day). If you are 75 years old or over, you will only be allocated to 100mg of aspirin or placebo. This is because you may be more likely to have side-effects from aspirin. To make sure the results of this study are as reliable as possible, neither you, nor your doctor, will know which treatment you get. If your doctor needs to find out which treatment you are taking at some point during the study, they will be able to do so. Whichever treatment you get, we will ask you to take one tablet a day for at least 5 years. It is very important that you take your tablet daily. It is usually easier to remember to do this if you take the tablet at the same time each day. If you forget to take a tablet or number of tablets do not take additional tablets to catch up, you should only take one tablet a day. Please let your study doctor or nurse know if you have missed any tablets next time you see them. Who decides which treatment I will get? A computer will choose which treatment you get by a process called randomisation. This means you will have an equal chance of getting any one of the 3 treatments and ensures that the groups of people being compared in the study are as similar as possible to start with except for the treatment they take. This in turn ensures that any differences between these groups are only due to the treatments in the study. Everyone joining this study will have a two out of three chance of getting aspirin. Other medicines It is extremely important that you do not take any other medication that contains aspirin regularly and you should not buy aspirin from a chemist or shop while taking part in the study. You should also not take drugs such as ibuprofen or Nurofen (non-steroidal antiinflammatory drugs) on a regular basis but they can be used occasionally where Page 3 of 8
4 absolutely necessary for no more than two weeks at a time. If you need to use a painkiller for more than two weeks, please discuss an alternative with your doctor e.g. paracetamol. They will also give you more information about this if you decide to take part in this study. Checks and tests Before you start taking your study treatment, you will have a blood test. After the run-in period you will have a check-up and blood test to see if you have had any problems taking aspirin. You will then have a check-up, blood tests and other checks depending on your cancer type every three months until one year and then every six months after that. If you were diagnosed with bowel cancer, you will have some CT scans. These would all be part of your normal care whether or not you participate in the study. The total dose of ionising radiation from these scans is approximately the same as 30 years of natural background radiation in the UK to which we are all exposed. The risk of a secondary cancer occurring from these scans is approximately 1 in 300, which is considerably less than our lifetime risk for developing cancer which is 1 in 3. If you were diagnosed with breast cancer, you will have some mammograms. These would all be part of your normal care whether or not you participate in the study. The total dose of ionising radiation from these mammograms is approximately the same as three years of natural background radiation in the UK to which we are all exposed. The risk of a secondary cancer occurring from these mammograms is approximately 1 in Some studies have suggested that aspirin may help protect against age-related changes in memory and thought processes (cognitive status). In order to find out more about this, we will ask you to complete a questionnaire at the beginning of the study, after one year and after five years. You will have your blood pressure checked at each trial visit. Your study nurse or doctor will be able to give you more information about these checks and tests. 5 What are the possible sideeffects? Aspirin is a common, frequently used drug and most people take it without experiencing any side effects. We will ask you some questions about your medical history to check that aspirin is a suitable treatment for you but some people might experience some side effects. Most side effects are mild but, for a very small number of people, they can be serious. Common side-effects The most common side-effects of aspirin include indigestion and irritation of the stomach. Between 1 in 10 and 1 in 100 people will experience these side-effects. Aspirin can also make you more prone to bruising. Un-common side-effects Aspirin can cause minor bleeding from the stomach or bowel. It is very important to tell your study doctor or nurse or GP if you have any dark or black stools or any vomiting of blood. This is uncommon (between 1 in 100 and 1 in 1000 people will experience it). Aspirin can also cause severe bleeding, but this is rare and will affect between 1 in 1000 and 1 in 10,000 people. Rare side-effects Irritation of the stomach or bowel can cause a small break in the lining called an ulcer. It is very important to tell your study doctor or nurse or GP if you have any dark or black Page 4 of 8
5 stools or any vomiting of blood. Rarely, bleeding in the brain occurs. It is important to have your blood pressure checked regularly while you are taking part in the study and this will be done at each visit. Aspirin can rarely cause other areas of bleeding for example, nose bleeds or bleeding gums. Hypersensitivity reaction (allergic reaction) is also a rare side-effect. Rare side-effects will affect between 1 in 1000 and 1 in 10,000 people. Very rare side-effects Kidney and liver impairment are very rare side-effects of aspirin. Very rare side-effects affect less than 1 in 10,000 people. Other side-effects Some people have experienced tinnitus (ringing sound in ears) when taking aspirin. Some recent evidence has suggested that a condition called macular degeneration (age related loss of sight) may be more common in people taking aspirin. Please tell your GP or study doctor as soon as possible if you become concerned about any potential side-effects at any stage, or if you are concerned about any medical problems you are experiencing. For a more detailed list of possible side-effects, please see the patient information leaflet provided with your medication. If you become pregnant or decide you want to during the study, please inform your study doctor or nurse as there are risks associated with taking aspirin during pregnancy. 6 What are the possible benefits and disadvantages of taking part? What are the possible benefits of taking part in this study? We hope that you will be helped by taking part in this study, but we can t guarantee this. However, the information we get from this study will help us to improve future treatments for people like you who have had treatment for cancer and help us find out more about the overall healthcare benefits that aspirin might provide such as preventing heart disease. What are the possible disadvantages and risks of taking part in this study? You might experience side-effects from the treatment that you take in this study. Sideeffects of aspirin are listed in section 5. You will be asked to have a blood test before joining the study and then asked for your permission to have additional blood tests during the study. You may also be asked to have some extra appointments at the hospital if you take part in the study. During the study, you will not be able to take drugs such as ibuprofen or Nurofen (nonsteroidal anti-inflammatory drugs) on a regular basis. If you have private medical insurance or require travel insurance, your policy may be affected. You should check this with your insurance provider. Page 5 of 8
6 7Samples and genetic tests Everybody that participates in this study will be asked to donate samples which may include small samples of blood, urine and tissue from their cancer. We would like to collect samples from everybody participating in the study, even if their study medication is stopped because it allows us to study the side-effects of aspirin. If you choose not to give permission for this you can still take part in the study. What samples will I be asked to donate? Cancer samples: samples of your cancer may have been stored in the hospital pathology laboratory. If you decide to take part in this study, we will ask your permission to retrieve your samples. You will not be asked to have any additional samples taken for this to happen. Blood samples: we will ask your permission to take a small amount of blood when you register for the study. This part of the study is optional and if you do not give your permission for this, you can still take part in the main study. What will happen to samples I give? Your donated samples will be stored in a laboratory for future research on the anticancer effects of aspirin and other projects aimed at improving outcomes for cancer patients. Any research using your donated samples would only be carried out after an independent research ethics committee has approved it. Will any genetic tests be done? Depending on your cancer type and your hospital, you may have routine genetic tests done. You will receive personal results from these tests. Your study doctor can tell you more about these tests. In the future, cancer researchers would like to use the blood and cancer samples that you donate (including your DNA) for separately approved research to help find out how genetics influence the risk of cancer and responses to treatments (including aspirin). You will not receive any personal results from these non-routine genetic tests unless we discover genetic information which has significant implications for your ongoing care, your future health or for that of your family. If this happens, we will contact you or the doctors looking after you with this information. 8 More information about taking part Do I have to take part in the Add- Aspirin study? No, it is up to you to decide whether or not to take part. If you decide to take part, we will ask you to sign a form to give your consent before the 8 week run-in period and again if you decide to continue in the study after the run-in period and to be randomised to one of the three treatments. You can stop taking part in the study at any time and without giving a reason. You can also decide not to take part. This will not affect the standard of care you receive. If you do decide to stop taking part, please discuss this with your doctor. If you decide to stop taking your study tablets, we would like to continue to collect some information about you. This will help us to test whether aspirin does stop cancer coming back. This is important, as it helps us to Page 6 of 8
7 ensure that the results of the study are reliable. If you decide not to take part in this study, you will continue to have your regular checkups and any other medication that you are receiving (for example, hormone treatment if you have had breast or prostate cancer), without taking the study medications. Who is organising and funding the study? University College London (UCL) has overall responsibility for this study. It is organised by the Medical Research Council Clinical Trials Unit (MRC CTU at UCL), which has run studies of this kind for many years. MRC CTU at UCL will manage the study and will collect and analyse the information. You can find out more about the MRC CTU at UCL here: This study is being paid for by the charity Cancer Research UK, the National Institute for Health Research s Health Technology Assessment Programme (part of the UK Department of Health) and the MRC CTU at UCL. Bayer Pharmaceuticals have supplied the aspirin tablets and placebo tablets for this study. You will not be paid for taking part in this study. Your doctor is not receiving any money or other payment for asking you to take part in the study. Who has reviewed the Add-Aspirin study? The study has been reviewed by international scientists on behalf of the funders. It has been authorised by the regulatory body in the UK, Medicines and Healthcare products Regulatory Agency (MHRA), as well as by South Central - Oxford C Research Ethics Committee and your hospital s Research and Development office. What will happen to information about me collected during the study? If you agree to take part in the study, your doctor will send information about you, your cancer and your progress to the MRC CTU. This information will be put into a computer and analysed by Add-Aspirin researchers. With your permission, we will also link to your details at the NHS Central Register (NHSCR), so we can check your health status if you lose touch with your study doctor or stop visiting the hospital. To do this, we will need to keep your name and NHS number on file, but these will be kept separately from other information about you. Some participants may be asked to send a health update directly to the MRC CTU. You do not have to take part in this aspect of the study if you do not wish. Also, if you agree to take part in the study, your medical records may be looked at by selected hospital staff or the lead doctor for the study (or his/her nominee). Results from the study and any future research will be published but will be anonymous. We will follow all legal requirements to make sure that all information about you is treated in confidence. If you take part in this study, we will also tell your GP that you are taking part in this study so that s/he is aware that you might be taking aspirin. We would also like to keep you updated with information about the study. If you would like us to send you information, we will take your contact details and keep them separately from other information about you. You do not have to give us your contact details if you do not want us to send you updates. Page 7 of 8
8 What will happen to the results of the Add-Aspirin study? If you give us permission, we will contact you to tell you the results of this study when it is completed. We will also publish a summary of the results on the study website: and in a medical journal, so that other doctors can see them. You can ask your doctor for a copy of any publication. Your identity and any personal details will be kept confidential. No named information about you will be published in any report of this study. What if new information becomes available during the course of the study? Sometimes during a study, new information becomes available about the medicines that are being studied. An independent committee will look at any new information and will also look at the data collected so far in the study and decide if any changes are needed. If this happens, your doctor will tell you about it and discuss whether you want to continue in the study. If you decide to stop taking part in the study, your doctor will arrange for your care to continue. Also, on receiving new information or because of a change in your health status, your doctor might consider that it is in your best interests to stop taking part in the study. Your doctor will explain the reasons and arrange for your care to continue. What if something goes wrong? If you have any concerns about the way you have been approached or treated during the study, or wish to complain, please use the normal NHS complaints process. If you are harmed by taking part, or if you are harmed due to someone s negligence, then you may be able to take legal action. 9 Contacts for further information If you want further information about the study, contact your study doctor or nurse (see below). <<Insert address and telephone number of study doctor and/or nurse>> You can also find information about the study on the following websites: Thank you for taking the time to consider taking part in this study. Page 8 of 8
The RADICALS trial Radiotherapy Timing Randomisation (RADICALS-RT) Clinical trial of treatment after surgery for prostate cancer
 Stoke Mandeville Hospital Mandeville Road Aylesbury Buckinghamshire HP21 8AL Tel: 01296 315 908 www.buckshealthcare.nhs.uk The RADICALS trial Radiotherapy Timing Randomisation (RADICALS-RT) Clinical trial
Stoke Mandeville Hospital Mandeville Road Aylesbury Buckinghamshire HP21 8AL Tel: 01296 315 908 www.buckshealthcare.nhs.uk The RADICALS trial Radiotherapy Timing Randomisation (RADICALS-RT) Clinical trial
U.K. Familial Ovarian Cancer Screening Study (UK FOCSS) Phase 2 Patient Information Sheet
 U.K. Familial Ovarian Cancer Screening Study (UK FOCSS) Phase 2 Patient Information Sheet 1. Invitation You are being invited to take part in a research study. Before you decide it is important for you
U.K. Familial Ovarian Cancer Screening Study (UK FOCSS) Phase 2 Patient Information Sheet 1. Invitation You are being invited to take part in a research study. Before you decide it is important for you
Patient Information Leaflet: Part 1 select-d
 Patient Information Leaflet: Part 1 select-d Anticoagulation Therapy in SELECTeD Cancer Patients at Risk of Recurrence of Venous Thromboembolism Introduction This
Patient Information Leaflet: Part 1 select-d Anticoagulation Therapy in SELECTeD Cancer Patients at Risk of Recurrence of Venous Thromboembolism Introduction This
Taking Part in Research at University Hospitals Birmingham
 University Hospitals Birmingham NHS Foundation Trust The Trust provides free monthly health talks on a variety of medical conditions and treatments. For more information visit www.uhb.nhs.uk or call 0121
University Hospitals Birmingham NHS Foundation Trust The Trust provides free monthly health talks on a variety of medical conditions and treatments. For more information visit www.uhb.nhs.uk or call 0121
Understanding Clinical Trials
 Understanding Clinical Trials The UK Clinical Research Collaboration (UKCRC) is a partnership of organisations working to establish the UK as a world leader in clinical research, by harnessing the power
Understanding Clinical Trials The UK Clinical Research Collaboration (UKCRC) is a partnership of organisations working to establish the UK as a world leader in clinical research, by harnessing the power
1. Study title Is the title self explanatory to a layperson? If not, a simplified title should be included.
 These guidelines apply to all research projects where human subjects are involved in the study GUIDELINES FOR RESEARCHERS PATIENT INFORMATION SHEET & CONSENT FORM The guidance, which follows, applies primarily
These guidelines apply to all research projects where human subjects are involved in the study GUIDELINES FOR RESEARCHERS PATIENT INFORMATION SHEET & CONSENT FORM The guidance, which follows, applies primarily
swine flu vaccination:
 swine flu vaccination: what you need to know Flu. Protect yourself and others. Contents What is swine flu?............... 3 About the swine flu vaccine....... 4 What else do I need to know?...... 8 What
swine flu vaccination: what you need to know Flu. Protect yourself and others. Contents What is swine flu?............... 3 About the swine flu vaccine....... 4 What else do I need to know?...... 8 What
PATIENT INFORMATION SHEET KEY FACTS
 PATIENT INFORMATION SHEET KEY FACTS Please read this carefully and refer to the full information sheet You are invited to take part in a research study, comparing subcutaneously (injection under skin)
PATIENT INFORMATION SHEET KEY FACTS Please read this carefully and refer to the full information sheet You are invited to take part in a research study, comparing subcutaneously (injection under skin)
MEDICATION GUIDE COUMADIN (COU-ma-din) (warfarin sodium)
 MEDICATION GUIDE COUMADIN (COU-ma-din) (warfarin sodium) Read this Medication Guide before you start taking COUMADIN (warfarin sodium) and each time you get a refill. There may be new information. This
MEDICATION GUIDE COUMADIN (COU-ma-din) (warfarin sodium) Read this Medication Guide before you start taking COUMADIN (warfarin sodium) and each time you get a refill. There may be new information. This
PATIENT INFORMATION SHEET
 PATIENT INFORMATION SHEET Surgical and large bore pleural procedures in Malignant pleural Mesothelioma And Radiotherapy Trial (SMART trial) Stoke Mandeville Hospital Mandeville Road Aylesbury Buckinghamshire
PATIENT INFORMATION SHEET Surgical and large bore pleural procedures in Malignant pleural Mesothelioma And Radiotherapy Trial (SMART trial) Stoke Mandeville Hospital Mandeville Road Aylesbury Buckinghamshire
What is Helicobacter pylori? hat problems can H. pylori cause? Does H. pylori cause cancer? ight H. pylori even be good for us?
 In association with: Primary Care Society for Gastroenterology INFORMATION ABOUT Helicobacter pylori? What is Helicobacter pylori? hat problems can H. pylori cause? Does H. pylori cause cancer? ight H.
In association with: Primary Care Society for Gastroenterology INFORMATION ABOUT Helicobacter pylori? What is Helicobacter pylori? hat problems can H. pylori cause? Does H. pylori cause cancer? ight H.
Nonsteroidal. Drugs (NSAIDs) Anti-Inflammatory. North American Spine Society Public Education Series
 Nonsteroidal Anti-Inflammatory Drugs (NSAIDs) North American Spine Society Public Education Series Nonsteroidal Anti- Inflammatory Drugs Your healthcare provider has recommended that you take a nonsteroidal
Nonsteroidal Anti-Inflammatory Drugs (NSAIDs) North American Spine Society Public Education Series Nonsteroidal Anti- Inflammatory Drugs Your healthcare provider has recommended that you take a nonsteroidal
ORAL ANTICOAGULANTS - RIVAROXABAN (XARELTO) FOR ATRIAL FIBRILLATION
 ORAL ANTICOAGULANTS - RIVAROXABAN (XARELTO) FOR ATRIAL FIBRILLATION Information Leaflet Your Health. Our Priority. Page 2 of 6 What Are Anticoagulants And What Do They Do? This information leaflet has
ORAL ANTICOAGULANTS - RIVAROXABAN (XARELTO) FOR ATRIAL FIBRILLATION Information Leaflet Your Health. Our Priority. Page 2 of 6 What Are Anticoagulants And What Do They Do? This information leaflet has
Rivaroxaban for the treatment of deep vein thrombosis (DVT) and pulmonary embolism (PE)
 Rivaroxaban for the treatment of deep vein thrombosis (DVT) and pulmonary embolism (PE) This leaflet aims to answer your questions about rivaroxaban that may be prescribed for you when you are diagnosed
Rivaroxaban for the treatment of deep vein thrombosis (DVT) and pulmonary embolism (PE) This leaflet aims to answer your questions about rivaroxaban that may be prescribed for you when you are diagnosed
Aspirin Dosing: A Patient Centric Trial Assessing Benefits and Long term Effectiveness (ADAPTABLE)
 Aspirin Dosing: A Patient Centric Trial Assessing Benefits and Long term Effectiveness (ADAPTABLE) We are asking you to join a research study called ADAPTABLE. The information below explains the study
Aspirin Dosing: A Patient Centric Trial Assessing Benefits and Long term Effectiveness (ADAPTABLE) We are asking you to join a research study called ADAPTABLE. The information below explains the study
A guide to prostate cancer clinical trials
 1 A guide to prostate cancer clinical trials In this fact sheet: What is a clinical trial? Why are trials done? What are trials looking into at the moment? How are clinical trials done? Should I take part
1 A guide to prostate cancer clinical trials In this fact sheet: What is a clinical trial? Why are trials done? What are trials looking into at the moment? How are clinical trials done? Should I take part
Medication Guide Plavix (PLAV-iks) (clopidogrel bisulfate) tablets
 Medication Guide Plavix (PLAV-iks) (clopidogrel bisulfate) tablets Read this Medication Guide before you start taking Plavix and each time you get a refill. There may be new information. This Medication
Medication Guide Plavix (PLAV-iks) (clopidogrel bisulfate) tablets Read this Medication Guide before you start taking Plavix and each time you get a refill. There may be new information. This Medication
PACKAGE LEAFLET: INFORMATION FOR THE USER. Dalacin C 150 mg Capsules. clindamycin hydrochloride. Dalacin C 150mg Capsules clindamycin hydrochloride
 PACKAGE LEAFLET: INFORMATION FOR THE USER PFIZER Dalacin C 150 mg Capsules clindamycin hydrochloride Dalacin C 150mg Capsules clindamycin hydrochloride PFIZER Read all of this leaflet carefully before
PACKAGE LEAFLET: INFORMATION FOR THE USER PFIZER Dalacin C 150 mg Capsules clindamycin hydrochloride Dalacin C 150mg Capsules clindamycin hydrochloride PFIZER Read all of this leaflet carefully before
Phasel clinical trials:
 Phasel clinical trials: what are they all about? Information for people wanting to know more about early clinical trials in cancer, Belfast City Hospital NORTHERN IRELAND CANCER TRIALS CENTRE Introduction
Phasel clinical trials: what are they all about? Information for people wanting to know more about early clinical trials in cancer, Belfast City Hospital NORTHERN IRELAND CANCER TRIALS CENTRE Introduction
Getting the most from blood pressure medicines
 P R E S S U R E P O I N T S S E R I E S : NO. 4 Getting the most from blood pressure medicines B L O O D P R E S S U R E A S S O C I AT I O N Pressure Points series Pressure Points is a series of booklets
P R E S S U R E P O I N T S S E R I E S : NO. 4 Getting the most from blood pressure medicines B L O O D P R E S S U R E A S S O C I AT I O N Pressure Points series Pressure Points is a series of booklets
ORAL ANTICOAGULANTS - RIVAROXABAN (XARELTO) FOR DEEP VEIN THROMBOSIS (DVT)
 ORAL ANTICOAGULANTS - RIVAROXABAN (XARELTO) FOR DEEP VEIN THROMBOSIS (DVT) Information Leaflet Your Health. Our Priority. Page 2 of 6 What Are Anticoagulants And What Do They Do? This information leaflet
ORAL ANTICOAGULANTS - RIVAROXABAN (XARELTO) FOR DEEP VEIN THROMBOSIS (DVT) Information Leaflet Your Health. Our Priority. Page 2 of 6 What Are Anticoagulants And What Do They Do? This information leaflet
There is a risk of renal impairment in dehydrated children and adolescents.
 PACKAGE LEAFLET: INFORMATION FOR THE USER MELFEN 200mg FILM-COATED TABLETS MELFEN 400mg FILM-COATED TABLETS Ibuprofen Read all of this leaflet carefully before you start taking this medicine because it
PACKAGE LEAFLET: INFORMATION FOR THE USER MELFEN 200mg FILM-COATED TABLETS MELFEN 400mg FILM-COATED TABLETS Ibuprofen Read all of this leaflet carefully before you start taking this medicine because it
Information for Egg Donors for Research
 Newcastle Fertility Centre at BioScience Centre, International Centre for Life Times Square, Newcastle upon Tyne, NE1 4EP Telephone: 0191 2138213 Fax: 0191 2138124 www.nfc-life.org.uk Information for Egg
Newcastle Fertility Centre at BioScience Centre, International Centre for Life Times Square, Newcastle upon Tyne, NE1 4EP Telephone: 0191 2138213 Fax: 0191 2138124 www.nfc-life.org.uk Information for Egg
Temozolomide (oral) with concurrent radiotherapy to the brain
 Temozolomide (oral) with concurrent radiotherapy to the brain Temozolomide (oral) with concurrent radiotherapy to the brain This leaflet is offered as a guide to you and your family. You will find it useful
Temozolomide (oral) with concurrent radiotherapy to the brain Temozolomide (oral) with concurrent radiotherapy to the brain This leaflet is offered as a guide to you and your family. You will find it useful
Warfarin therapy for stroke patients with atrial fibrillation
 Warfarin therapy for stroke patients with atrial fibrillation Delivering the best in care UHB is a no smoking Trust To see all of our current patient information leaflets please visit www.uhb.nhs.uk/patient-information-leaflets.htm
Warfarin therapy for stroke patients with atrial fibrillation Delivering the best in care UHB is a no smoking Trust To see all of our current patient information leaflets please visit www.uhb.nhs.uk/patient-information-leaflets.htm
A PATIENT S GUIDE TO DEEP VEIN THROMBOSIS TREATMENT
 A PATIENT S GUIDE TO DEEP VEIN THROMBOSIS TREATMENT This medicine is subject to additional monitoring. This will allow quick identification of new safety information. If you get any side effects, talk
A PATIENT S GUIDE TO DEEP VEIN THROMBOSIS TREATMENT This medicine is subject to additional monitoring. This will allow quick identification of new safety information. If you get any side effects, talk
Script/Notes for PowerPoint Presentation. Medication Use Safety Training for Seniors (MUST for Seniors)
 Script/Notes for PowerPoint Presentation Medication Use Safety Training for Seniors (MUST for Seniors) Instructions: You can use the following script to help you prepare your remarks to your organization
Script/Notes for PowerPoint Presentation Medication Use Safety Training for Seniors (MUST for Seniors) Instructions: You can use the following script to help you prepare your remarks to your organization
Alcohol and older people. What you need to know
 Alcohol and older people What you need to know Alcohol and older people 1 Contents As we age, we become more sensitive to alcohol s effects...3 Alcohol can cause problems if you re taking certain medicines...4
Alcohol and older people What you need to know Alcohol and older people 1 Contents As we age, we become more sensitive to alcohol s effects...3 Alcohol can cause problems if you re taking certain medicines...4
Identifying and treating long-term kidney problems (chronic kidney disease)
 Understanding NICE guidance Information for people who use NHS services Identifying and treating long-term kidney problems (chronic kidney disease) NICE clinical guidelines advise the NHS on caring for
Understanding NICE guidance Information for people who use NHS services Identifying and treating long-term kidney problems (chronic kidney disease) NICE clinical guidelines advise the NHS on caring for
Anticoagulation in Atrial Fibrillation Patient information
 Anticoagulation in Atrial Fibrillation Patient information RSBAY013 Anticoag A5 V2.indd 1 21/02/2014 17:13 Introduction This leaflet will help you understand your new medicine. Your new medicine is commonly
Anticoagulation in Atrial Fibrillation Patient information RSBAY013 Anticoag A5 V2.indd 1 21/02/2014 17:13 Introduction This leaflet will help you understand your new medicine. Your new medicine is commonly
Immune Cell Studies in Type 1 Diabetes (ISTID) INFORMATION SHEET FOR ADULTS WITH NEWLY- DIAGNOSED DIABETES. Sponsored by Cardiff University
 Immune Cell Studies in Type 1 Diabetes (ISTID) INFORMATION SHEET FOR ADULTS WITH NEWLY- DIAGNOSED DIABETES Sponsored by Cardiff University Chief Investigator Wales: Professor Colin Dayan Address: Centre
Immune Cell Studies in Type 1 Diabetes (ISTID) INFORMATION SHEET FOR ADULTS WITH NEWLY- DIAGNOSED DIABETES Sponsored by Cardiff University Chief Investigator Wales: Professor Colin Dayan Address: Centre
ORAL ANTICOAGULANTS RIVAROXABAN (XARELTO) FOR PULMONARY EMBOLISM (PE)
 ORAL ANTICOAGULANTS RIVAROXABAN (XARELTO) FOR PULMONARY EMBOLISM (PE) Information Leaflet Your Health. Our Priority. Page 2 of 6 What Are Anticoagulants And What Do They Do? This information leaflet has
ORAL ANTICOAGULANTS RIVAROXABAN (XARELTO) FOR PULMONARY EMBOLISM (PE) Information Leaflet Your Health. Our Priority. Page 2 of 6 What Are Anticoagulants And What Do They Do? This information leaflet has
Thinking of getting pregnant?
 Thinking of getting pregnant? Make sure you re protected against German measles the safest way to protect the health of you and your baby Catching German measles (rubella) can be very serious for your
Thinking of getting pregnant? Make sure you re protected against German measles the safest way to protect the health of you and your baby Catching German measles (rubella) can be very serious for your
PATIENT INFORMATION SHEET Study name: CT morphology of lung parenchyma pre and post bariatric surgery: correlation with pulmonary function.
 Version 2. 8.09.2011 Hammersmith Hospital Du Cane Rd London W12 0HS Tel: 020 8383 1000 www.imperial.nhs.uk PATIENT INFORMATION SHEET Study name: CT morphology of lung parenchyma pre and post bariatric
Version 2. 8.09.2011 Hammersmith Hospital Du Cane Rd London W12 0HS Tel: 020 8383 1000 www.imperial.nhs.uk PATIENT INFORMATION SHEET Study name: CT morphology of lung parenchyma pre and post bariatric
University College Hospital. Contrast agent for radiotherapy CT (computed tomography) scans. Radiotherapy Department Patient information series
 University College Hospital Contrast agent for radiotherapy CT (computed tomography) scans Radiotherapy Department Patient information series 11 2 If you need a large print, audio or translated copy of
University College Hospital Contrast agent for radiotherapy CT (computed tomography) scans Radiotherapy Department Patient information series 11 2 If you need a large print, audio or translated copy of
Secondary liver cancer Patient Information Booklet
 Secondary liver cancer Patient Information Booklet Delivering the best in care UHB is a no smoking Trust To see all of our current patient information leaflets please visit www.uhb.nhs.uk/patient-information-leaflets.htm
Secondary liver cancer Patient Information Booklet Delivering the best in care UHB is a no smoking Trust To see all of our current patient information leaflets please visit www.uhb.nhs.uk/patient-information-leaflets.htm
Summary of the role and operation of NHS Research Management Offices in England
 Summary of the role and operation of NHS Research Management Offices in England The purpose of this document is to clearly explain, at the operational level, the activities undertaken by NHS R&D Offices
Summary of the role and operation of NHS Research Management Offices in England The purpose of this document is to clearly explain, at the operational level, the activities undertaken by NHS R&D Offices
Peptic Ulcer. Anatomy The stomach is a hollow organ. It is located in the upper abdomen, under the ribs.
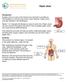 Peptic Ulcer Introduction A peptic ulcer is a sore in the lining of your stomach or duodenum. The duodenum is the first part of your small intestine. Peptic ulcers may also develop in the esophagus. Nearly
Peptic Ulcer Introduction A peptic ulcer is a sore in the lining of your stomach or duodenum. The duodenum is the first part of your small intestine. Peptic ulcers may also develop in the esophagus. Nearly
INFORMATION LEAFLET. If anything is not clear, or if you would like more information, please
 BIO INFOBK 1492 0410:Layout 1 21/04/2010 15:24 Page 1 INFORMATION LEAFLET You are being invited to take part in a major medical research project called UK Biobank. The purpose of UK Biobank is to set up
BIO INFOBK 1492 0410:Layout 1 21/04/2010 15:24 Page 1 INFORMATION LEAFLET You are being invited to take part in a major medical research project called UK Biobank. The purpose of UK Biobank is to set up
A Guide to Breast Screening
 A Guide to Breast Screening Contents Information for Carers, Family, Friends and Medical Guardians of Care Introduction Information on BreastCheck Information on Attending Screening The Screening Story
A Guide to Breast Screening Contents Information for Carers, Family, Friends and Medical Guardians of Care Introduction Information on BreastCheck Information on Attending Screening The Screening Story
A Guide to Clinical Trials
 A Guide to Clinical Trials For young people with cancer and their parents Children s Cancer and Leukaemia Group www.cclg.org.uk Original booklet produced in conjunction with the CCLG Patient Advocacy Committee.
A Guide to Clinical Trials For young people with cancer and their parents Children s Cancer and Leukaemia Group www.cclg.org.uk Original booklet produced in conjunction with the CCLG Patient Advocacy Committee.
Participant Invitation and Information Sheet MRI Test Run
 ON QE TRUST HEADED PAPER A randomised controlled trial of bariatric surgery versus a community weight loss programme for the sustained treatment of Idiopathic Intracranial Hypertension: the IIH:WT Trial
ON QE TRUST HEADED PAPER A randomised controlled trial of bariatric surgery versus a community weight loss programme for the sustained treatment of Idiopathic Intracranial Hypertension: the IIH:WT Trial
MEDICATION GUIDE XARELTO (zah-rel-toe) (rivaroxaban) Tablets
 MEDICATION GUIDE XARELTO (zah-rel-toe) (rivaroxaban) Tablets Read this Medication Guide before you start taking XARELTO and each time you get a refill. There may be new information. This Medication Guide
MEDICATION GUIDE XARELTO (zah-rel-toe) (rivaroxaban) Tablets Read this Medication Guide before you start taking XARELTO and each time you get a refill. There may be new information. This Medication Guide
Medicals c i e n t i f i c study
 Medicals c i e n t i f i c study design risks medical-ethics review board 2 Table of contents M e d i c a l - s c i e n t i f i c study Preface 2 Introduction 4 Medical-scientific study 5 Why participate?
Medicals c i e n t i f i c study design risks medical-ethics review board 2 Table of contents M e d i c a l - s c i e n t i f i c study Preface 2 Introduction 4 Medical-scientific study 5 Why participate?
Patient Guide. Important information for patients starting therapy with LEMTRADA (alemtuzumab)
 Patient Guide Important information for patients starting therapy with LEMTRADA (alemtuzumab) This medicinal product is subject to additional monitoring. This will allow quick identification of new safety
Patient Guide Important information for patients starting therapy with LEMTRADA (alemtuzumab) This medicinal product is subject to additional monitoring. This will allow quick identification of new safety
The Care Record Guarantee Our Guarantee for NHS Care Records in England
 The Care Record Guarantee Our Guarantee for NHS Care Records in England Introduction In the National Health Service in England, we aim to provide you with the highest quality of healthcare. To do this,
The Care Record Guarantee Our Guarantee for NHS Care Records in England Introduction In the National Health Service in England, we aim to provide you with the highest quality of healthcare. To do this,
Allopurinol Allopurinol
 Drug information Allopurinol Allopurinol This leaflet provides information on allopurinol and will answer any questions you have about the treatment. Arthritis Research UK produce and print our booklets
Drug information Allopurinol Allopurinol This leaflet provides information on allopurinol and will answer any questions you have about the treatment. Arthritis Research UK produce and print our booklets
PATIENT INFORMATION SHEET. Version 5, March 2015
 King s College Hospital NHS Foundation Trust King s College Hospital Denmark Hill London SE5 9RS Tel: 020 3299 9000 Fax: 020 3299 3445 www.kch.nhs.uk PATIENT INFORMATION SHEET Version 5, March 2015 Study
King s College Hospital NHS Foundation Trust King s College Hospital Denmark Hill London SE5 9RS Tel: 020 3299 9000 Fax: 020 3299 3445 www.kch.nhs.uk PATIENT INFORMATION SHEET Version 5, March 2015 Study
Breast Screening Explained. We can supply this information in other languages, in large print, on audio or in Braille.
 Breast Screening Explained We can supply this information in other languages, in large print, on audio or in Braille. Breast Screening Explained This leaflet tells you about free breast screening. Breast
Breast Screening Explained We can supply this information in other languages, in large print, on audio or in Braille. Breast Screening Explained This leaflet tells you about free breast screening. Breast
PARTICIPANT INFORMATION
 PARTICIPANT INFORMATION IMPERIAL POPULATION BIORESOURCE TISSUE BANK (PART OF NIHR BIORESOURCE) In the UK around 20 million people are said to be living with a long-term condition such as diabetes or heart
PARTICIPANT INFORMATION IMPERIAL POPULATION BIORESOURCE TISSUE BANK (PART OF NIHR BIORESOURCE) In the UK around 20 million people are said to be living with a long-term condition such as diabetes or heart
Leflunomide Leflunomide
 Drug information Leflunomide Leflunomide This leaflet provides information on leflunomide and will answer any questions you have about the treatment. Arthritis Research UK produce and print our booklets
Drug information Leflunomide Leflunomide This leaflet provides information on leflunomide and will answer any questions you have about the treatment. Arthritis Research UK produce and print our booklets
Screening for ovarian cancer Page 1 of 5 Ovacome
 Fact sheet 9 Screening for ovarian cancer Ovacome is a national charity providing advice and support to women with ovarian cancer. We give information about symptoms, diagnosis, treatment, research and
Fact sheet 9 Screening for ovarian cancer Ovacome is a national charity providing advice and support to women with ovarian cancer. We give information about symptoms, diagnosis, treatment, research and
UW MEDICINE PATIENT EDUCATION. Xofigo Therapy. For metastatic prostate cancer. What is Xofigo? How does it work?
 UW MEDICINE PATIENT EDUCATION Xofigo Therapy For metastatic prostate cancer This handout explains how the drug Xofigo is used to treat metastatic prostate cancer. What is Xofigo? Xofigo is a radioactive
UW MEDICINE PATIENT EDUCATION Xofigo Therapy For metastatic prostate cancer This handout explains how the drug Xofigo is used to treat metastatic prostate cancer. What is Xofigo? Xofigo is a radioactive
Fortrose Medical Practice
 Fortrose Medical Practice GP Partners: Dr Sandy MacGregor, Dr Will Fraser, Dr Iain Forth & Dr Jude Watmough Associate GP: Dr Shona Forth Station Road Fortrose Ross-shire IV10 8SY Phone: Fax: Email: Website:
Fortrose Medical Practice GP Partners: Dr Sandy MacGregor, Dr Will Fraser, Dr Iain Forth & Dr Jude Watmough Associate GP: Dr Shona Forth Station Road Fortrose Ross-shire IV10 8SY Phone: Fax: Email: Website:
(Form to be on hospital/institution headed paper) INCA TRIAL PATIENT INFORMATION SHEET
 (Form to be on hospital/institution headed paper) INCA TRIAL PATIENT INFORMATION SHEET A multicentre randomised phase II clinical trial of Inotuzumab Ozogamicin plus Rituximab and CVP (IO-R-CVP) versus
(Form to be on hospital/institution headed paper) INCA TRIAL PATIENT INFORMATION SHEET A multicentre randomised phase II clinical trial of Inotuzumab Ozogamicin plus Rituximab and CVP (IO-R-CVP) versus
Alzheimer s and. memory loss
 Alzheimer s and memory loss This leaflet aims to help you understand about memory loss, Alzheimer s disease and other forms of dementia. You can also find out how to get more information. It s for anyone
Alzheimer s and memory loss This leaflet aims to help you understand about memory loss, Alzheimer s disease and other forms of dementia. You can also find out how to get more information. It s for anyone
UNDERGOING TERMINATION OF PREGNANCY AFTER 12 WEEKS
 UNDERGOING TERMINATION OF PREGNANCY AFTER 12 WEEKS Information Leaflet Your Health. Our Priority. Page 2 of 5 The staff at Stepping Hill Hospital will support you as much as we can during this sad time.
UNDERGOING TERMINATION OF PREGNANCY AFTER 12 WEEKS Information Leaflet Your Health. Our Priority. Page 2 of 5 The staff at Stepping Hill Hospital will support you as much as we can during this sad time.
A PATIENT S GUIDE TO PULMONARY EMBOLISM TREATMENT
 A PATIENT S GUIDE TO PULMONARY EMBOLISM TREATMENT This medicine is subject to additional monitoring. This will allow quick identification of new safety information. If you get any side effects, talk to
A PATIENT S GUIDE TO PULMONARY EMBOLISM TREATMENT This medicine is subject to additional monitoring. This will allow quick identification of new safety information. If you get any side effects, talk to
Obstetric Cholestasis (itching liver disorder) Information for parents-to-be
 Oxford University Hospitals NHS Trust Obstetric Cholestasis (itching liver disorder) Information for parents-to-be page 2 You have been given this leaflet because you have been diagnosed with (or are suspected
Oxford University Hospitals NHS Trust Obstetric Cholestasis (itching liver disorder) Information for parents-to-be page 2 You have been given this leaflet because you have been diagnosed with (or are suspected
OGD (Gastroscopy) Information for patients. Liver, Renal & Surgery. Confirming your identity
 Liver, Renal & Surgery OGD (Gastroscopy) Information for patients This leaflet answers some of the questions you may have about having an OGD. It explains the risks and the benefits of the test and what
Liver, Renal & Surgery OGD (Gastroscopy) Information for patients This leaflet answers some of the questions you may have about having an OGD. It explains the risks and the benefits of the test and what
Always take this medicine exactly as described in this leaflet or as your doctor, pharmacist or nurse have told you.
 leaflet: Information for the user Macrogol 4000 10 g powder for oral solution in sachet Macrogol 4000
leaflet: Information for the user Macrogol 4000 10 g powder for oral solution in sachet Macrogol 4000
Safety Information Card for Xarelto Patients
 Safety Information Card for Xarelto Patients 15mg Simply Protecting More Patients 20mg Simply Protecting More Patients Keep this card with you at all times Present this card to every physician or dentist
Safety Information Card for Xarelto Patients 15mg Simply Protecting More Patients 20mg Simply Protecting More Patients Keep this card with you at all times Present this card to every physician or dentist
NHS Cervical Screening Having a colposcopy
 NHS Cervical Screening Having a colposcopy What is a colposcopy? 1 Why do I need a colposcopy? 1 What does a colposcopy involve? 2 Colposcopy results 4 Treatment to remove abnormal cells in the cervix
NHS Cervical Screening Having a colposcopy What is a colposcopy? 1 Why do I need a colposcopy? 1 What does a colposcopy involve? 2 Colposcopy results 4 Treatment to remove abnormal cells in the cervix
FAQs about Warfarin (brand name Coumadin )
 FAQs about Warfarin (brand name Coumadin ) What is warfarin? Warfarin is the most commonly used anticoagulant in the US. An anticoagulant is a drug used to prevent unwanted and harmful blood clots. Although
FAQs about Warfarin (brand name Coumadin ) What is warfarin? Warfarin is the most commonly used anticoagulant in the US. An anticoagulant is a drug used to prevent unwanted and harmful blood clots. Although
Teriflunomide (Aubagio) 14mg once daily tablet
 Teriflunomide (Aubagio) 14mg once daily tablet Exceptional healthcare, personally delivered Your Consultant Neurologist has suggested that you may benefit from treatment with Teriflunomide. The decision
Teriflunomide (Aubagio) 14mg once daily tablet Exceptional healthcare, personally delivered Your Consultant Neurologist has suggested that you may benefit from treatment with Teriflunomide. The decision
Imaging Markers of Brain Network Dysfunction in Multiple Sclerosis
 Faculty of Medicine & Health Sciences School of Medicine Radiological Sciences Research Group The University of Nottinham University Park Nottingham NG7 2RD t: +44 (0)115 823 0018 f: +44 (0)115 823 0004
Faculty of Medicine & Health Sciences School of Medicine Radiological Sciences Research Group The University of Nottinham University Park Nottingham NG7 2RD t: +44 (0)115 823 0018 f: +44 (0)115 823 0004
A guide for the patient
 Understanding series LUNG CANCER CLINICAL TRIALS 1-800-298-2436 LungCancerAlliance.org A guide for the patient TABLE OF CONTENTS The Basics What is a Clinical Trial?...3 Types of Clinical Trials... 3 Phases
Understanding series LUNG CANCER CLINICAL TRIALS 1-800-298-2436 LungCancerAlliance.org A guide for the patient TABLE OF CONTENTS The Basics What is a Clinical Trial?...3 Types of Clinical Trials... 3 Phases
Pain relief at home. Information for adult patients
 Pain relief at home Information for adult patients 3 Contents Page 3 Page 3 Page 5 Page 7 Page 8 Introduction to pain relief Common pain relief medicines and their side effects More information about pain
Pain relief at home Information for adult patients 3 Contents Page 3 Page 3 Page 5 Page 7 Page 8 Introduction to pain relief Common pain relief medicines and their side effects More information about pain
CMF: Cyclophosphamide, Methotrexate and Fluorouracil
 PATIENT EDUCATION patienteducation.osumc.edu CMF: Cyclophosphamide, Methotrexate and Fluorouracil What is CMF? This is the short name for the drugs used for this chemotherapy treatment. The three drugs
PATIENT EDUCATION patienteducation.osumc.edu CMF: Cyclophosphamide, Methotrexate and Fluorouracil What is CMF? This is the short name for the drugs used for this chemotherapy treatment. The three drugs
Tuberculosis and You A Guide to Tuberculosis Treatment and Services
 Tuberculosis and You A Guide to Tuberculosis Treatment and Services Tuberculosis (TB) is a serious disease that can damage the lungs or other parts of the body like the brain, kidneys or spine. There are
Tuberculosis and You A Guide to Tuberculosis Treatment and Services Tuberculosis (TB) is a serious disease that can damage the lungs or other parts of the body like the brain, kidneys or spine. There are
TC: Docetaxel and Cyclophosphamide
 PATIENT EDUCATION patienteducation.osumc.edu What is TC? It is the short name for the drugs used for this chemotherapy treatment. The two drugs you will receive during this treatment are Docetaxel (Taxotere
PATIENT EDUCATION patienteducation.osumc.edu What is TC? It is the short name for the drugs used for this chemotherapy treatment. The two drugs you will receive during this treatment are Docetaxel (Taxotere
Having denervation of the renal arteries for treatment of high blood pressure
 Having denervation of the renal arteries for treatment of high blood pressure The aim of this information sheet is to help answer some of the questions you may have about having denervation of the renal
Having denervation of the renal arteries for treatment of high blood pressure The aim of this information sheet is to help answer some of the questions you may have about having denervation of the renal
Excision or Open Biopsy of a Breast Lump Your Operation Explained
 Excision or Open Biopsy of a Breast Lump Your Operation Explained Patient Information Introduction This leaflet tells you about the procedure known as excision or open biopsy of a breast lump. It explains
Excision or Open Biopsy of a Breast Lump Your Operation Explained Patient Information Introduction This leaflet tells you about the procedure known as excision or open biopsy of a breast lump. It explains
PACKAGE LEAFLET: INFORMATION FOR THE USER. PARACETAMOL MACOPHARMA 10 mg/ml, solution for infusion. Paracetamol
 PACKAGE LEAFLET: INFORMATION FOR THE USER PARACETAMOL MACOPHARMA 10 mg/ml, solution for infusion Paracetamol Read all of this leaflet carefully before you start using this medicine. Keep this leaflet.
PACKAGE LEAFLET: INFORMATION FOR THE USER PARACETAMOL MACOPHARMA 10 mg/ml, solution for infusion Paracetamol Read all of this leaflet carefully before you start using this medicine. Keep this leaflet.
NHS cervical screening Helping you decide
 NHS cervical screening Helping you decide What is cervical cancer? 2 What causes cervical cancer? 2 What is cervical screening? 3 Cervical screening results 6 What is a colposcopy? 8 What are the benefits
NHS cervical screening Helping you decide What is cervical cancer? 2 What causes cervical cancer? 2 What is cervical screening? 3 Cervical screening results 6 What is a colposcopy? 8 What are the benefits
Mifamurtide (Mepact ) for Osteosarcoma
 Mifamurtide (Mepact ) for Osteosarcoma Mifamurtide (Mepact ) for Osteosarcoma This leaflet is offered as a guide to you and your family. Your treatment will be fully explained to you by your doctor or
Mifamurtide (Mepact ) for Osteosarcoma Mifamurtide (Mepact ) for Osteosarcoma This leaflet is offered as a guide to you and your family. Your treatment will be fully explained to you by your doctor or
Information for you Abortion care
 Information for you Abortion care Published in February 2012 This information is for you if you are considering having an abortion. It tells you: how you can access abortion services the care you can expect
Information for you Abortion care Published in February 2012 This information is for you if you are considering having an abortion. It tells you: how you can access abortion services the care you can expect
Breast Cancer Questions to Ask
 Ask about your medicines for primary (early) breast cancer Questions to help you get the best from your drug treatments Ask about your medicines for primary (early) breast cancer This guide is to help
Ask about your medicines for primary (early) breast cancer Questions to help you get the best from your drug treatments Ask about your medicines for primary (early) breast cancer This guide is to help
PATIENT INFORMATION LEAFLET. Calcium Sandoz Syrup calcium glubionate and calcium lactobionate
 PATIENT INFORMATION LEAFLET Calcium Sandoz Syrup calcium glubionate and calcium lactobionate Read all of this leaflet carefully before you start taking this medicine Keep this leaflet. You may need to
PATIENT INFORMATION LEAFLET Calcium Sandoz Syrup calcium glubionate and calcium lactobionate Read all of this leaflet carefully before you start taking this medicine Keep this leaflet. You may need to
Suspected pulmonary embolism (PE) in pregnant women
 Suspected pulmonary embolism (PE) in pregnant women What is a pulmonary embolus? A deep vein thrombosis (DVT) is a blood clot that forms in one of the deep veins of the leg. If the clot moves to the lung,
Suspected pulmonary embolism (PE) in pregnant women What is a pulmonary embolus? A deep vein thrombosis (DVT) is a blood clot that forms in one of the deep veins of the leg. If the clot moves to the lung,
Fact sheet 9. Screening for ovarian cancer
 Fact sheet 9 Screening for ovarian cancer Ovacome is a national charity providing advice and support to women with ovarian cancer. We give information about symptoms, diagnosis, treatment, research and
Fact sheet 9 Screening for ovarian cancer Ovacome is a national charity providing advice and support to women with ovarian cancer. We give information about symptoms, diagnosis, treatment, research and
Paclitaxel and Carboplatin
 PATIENT EDUCATION patienteducation.osumc.edu What is Paclitaxel (pak-li-tax-el) and how does it work? Paclitaxel is a chemotherapy drug known as an anti-microtubule inhibitor. Another name for this drug
PATIENT EDUCATION patienteducation.osumc.edu What is Paclitaxel (pak-li-tax-el) and how does it work? Paclitaxel is a chemotherapy drug known as an anti-microtubule inhibitor. Another name for this drug
PROCEDURE- SPECIFIC INFORMATION FOR PATIENTS
 The British Association of Urological Surgeons 35-43 Lincoln s Inn Fields London WC2A 3PE Phone: Fax: Website: E- mail: +44 (0)20 7869 6950 +44 (0)20 7404 5048 www.baus.org.uk admin@baus.org.uk PROCEDURE-
The British Association of Urological Surgeons 35-43 Lincoln s Inn Fields London WC2A 3PE Phone: Fax: Website: E- mail: +44 (0)20 7869 6950 +44 (0)20 7404 5048 www.baus.org.uk admin@baus.org.uk PROCEDURE-
Package leaflet: Information for the patient. Laxido Orange, powder for oral solution
 Package leaflet: Information for the patient Laxido Orange, powder for oral solution Read all of this leaflet carefully before you start taking this medicine because it contains important information for
Package leaflet: Information for the patient Laxido Orange, powder for oral solution Read all of this leaflet carefully before you start taking this medicine because it contains important information for
TCH: Docetaxel, Carboplatin and Trastuzumab
 PATIENT EDUCATION patienteducation.osumc.edu TCH: Docetaxel, Carboplatin and Trastuzumab What is TCH? It is the short name for the drugs used for this chemotherapy treatment. The three drugs you will receive
PATIENT EDUCATION patienteducation.osumc.edu TCH: Docetaxel, Carboplatin and Trastuzumab What is TCH? It is the short name for the drugs used for this chemotherapy treatment. The three drugs you will receive
Gemcitabine and Cisplatin
 PATIENT EDUCATION patienteducation.osumc.edu What is Gemcitabine (jem-site-a been)? Gemcitabine is a chemotherapy medicine known as an anti-metabolite. Another name for this drug is Gemzar. This drug is
PATIENT EDUCATION patienteducation.osumc.edu What is Gemcitabine (jem-site-a been)? Gemcitabine is a chemotherapy medicine known as an anti-metabolite. Another name for this drug is Gemzar. This drug is
Paracetamol apollo +9191 46 950 950. Paracetamol apollo +9191 46 950 950. Paracetamol
 Paracetamol apollo +9191 46 950 950 Paracetamol apollo +9191 46 950 950 Paracetamol CAS Number : 103-90-2 Molecular Weight : 151.17 g/mol Molecular Formula : C8H9NO2 Systematic (IUPAC) : N-(4- hydroxyphenyl)ethanamide
Paracetamol apollo +9191 46 950 950 Paracetamol apollo +9191 46 950 950 Paracetamol CAS Number : 103-90-2 Molecular Weight : 151.17 g/mol Molecular Formula : C8H9NO2 Systematic (IUPAC) : N-(4- hydroxyphenyl)ethanamide
Consent Form: Example 2 (DNA Sequencing)
 Consent Form: Example 2 (DNA Sequencing) Important note: This model language was developed for the NHGRI Medical Sequencing Project (MSP). It is included here only as an example of how to describe a sequencing
Consent Form: Example 2 (DNA Sequencing) Important note: This model language was developed for the NHGRI Medical Sequencing Project (MSP). It is included here only as an example of how to describe a sequencing
Gynaecology Service. Saint Mary s Hospital. The Whitworth Clinic. Information for patients
 Gynaecology Service Saint Mary s Hospital The Whitworth Clinic Information for patients Welcome to the Whitworth Clinic at Saint Mary s Hospital This leaflet will give you information about the service,
Gynaecology Service Saint Mary s Hospital The Whitworth Clinic Information for patients Welcome to the Whitworth Clinic at Saint Mary s Hospital This leaflet will give you information about the service,
Rubella. Questions and answers
 Rubella Questions and answers What is rubella? Rubella, or German measles, is a mild disease caused by a virus. Before the MMR (measles, mumps and rubella) vaccine was used, rubella used to be common in
Rubella Questions and answers What is rubella? Rubella, or German measles, is a mild disease caused by a virus. Before the MMR (measles, mumps and rubella) vaccine was used, rubella used to be common in
medicineupdate Tramadol for pain Asking the right questions about new medicines Page Section 1: What tramadol is 1 Section 2: What tramadol is for 1
 medicineupdate Asking the right questions about new medicines Tramadol for pain Page Section 1: What tramadol is 1 Section 2: What tramadol is for 1 Section 3: Who can take tramadol 2 Section 4: What does
medicineupdate Asking the right questions about new medicines Tramadol for pain Page Section 1: What tramadol is 1 Section 2: What tramadol is for 1 Section 3: Who can take tramadol 2 Section 4: What does
Atrial fibrillation (non-valvular) and reducing the risk of a stroke Management Options Brief Decision Aid
 Atrial fibrillation (non-valvular) and reducing the risk of a stroke Management Options Brief Decision Aid Atrial fibrillation (AF) increases your risk of having a stroke (a blood clot in the brain). From
Atrial fibrillation (non-valvular) and reducing the risk of a stroke Management Options Brief Decision Aid Atrial fibrillation (AF) increases your risk of having a stroke (a blood clot in the brain). From
Warfarin. (Coumadin, Jantoven ) Taking your medication safely
 Warfarin (Coumadin, Jantoven ) Taking your medication safely Welcome This booklet is designed to provide you with important information about warfarin to help you take this medication safely and effectively.
Warfarin (Coumadin, Jantoven ) Taking your medication safely Welcome This booklet is designed to provide you with important information about warfarin to help you take this medication safely and effectively.
Brand Name Marevan Pradaxa Xarelto Eliquis
 Medicines for stroke prevention in atrial fibrillation Choosing the right one for you Atrial fibrillation (AF) is a condition that affects the heart, causing it to beat irregularly and too fast. When this
Medicines for stroke prevention in atrial fibrillation Choosing the right one for you Atrial fibrillation (AF) is a condition that affects the heart, causing it to beat irregularly and too fast. When this
pregnant pregnant pregnant pregnant pregnan pregnant ant pregnant pregnant pregnant egnant pregnant pregnant pregnant pregnant pregna
 Pregnant and don t know what to do? A guide to your options pregna regnant ant pregnan egnant 2 3 Are you but not sure you want to have the baby? Do you need more information about the options available
Pregnant and don t know what to do? A guide to your options pregna regnant ant pregnan egnant 2 3 Are you but not sure you want to have the baby? Do you need more information about the options available
Rivaroxaban (Xarelto) for preventing blood clots after hip or knee replacement surgery
 medicineupdate Asking the right questions about new medicines Rivaroxaban (Xarelto) for preventing blood clots after hip or knee replacement surgery This Medicine Update is for people who have been prescribed
medicineupdate Asking the right questions about new medicines Rivaroxaban (Xarelto) for preventing blood clots after hip or knee replacement surgery This Medicine Update is for people who have been prescribed
Treatment of colon cancer
 Treatment of colon cancer This information is an extract from the booklet, Understanding colon cancer. You may find the full booklet helpful. We can send you a copy free see page 5. Contents How treatment
Treatment of colon cancer This information is an extract from the booklet, Understanding colon cancer. You may find the full booklet helpful. We can send you a copy free see page 5. Contents How treatment
It can be devastating to be diagnosed with a cancer like multiple myeloma. But there are treatments that can help you live longer and feel better.
 Patient information from the BMJ Group It can be devastating to be diagnosed with a cancer like multiple myeloma. But there are treatments that can help you live longer and feel better. What is multiple
Patient information from the BMJ Group It can be devastating to be diagnosed with a cancer like multiple myeloma. But there are treatments that can help you live longer and feel better. What is multiple
