Metabolism: is the total chemical changes which takes place in the living cell
|
|
|
- Miles Wade
- 7 years ago
- Views:
Transcription
1
2 Metabolism: is the total chemical changes which takes place in the living cell Anabolic and catabolic reaction are collectively called metabolism metabolism Anabolic reaction catabolic reaction (formation complex end (breakdown complex molecule Product from simple compound) such as polysaccharide, protein lipid to a few simple molecules like CO2, H2O,NH3)
3
4 Digestion of carbohydrate Location of digestion enzyme substrate product Mouth ptyalin starch maltose+ dextrine isomaltose Stomach no enzyme Small intestine amylase starch+dextrin maltose Intestinal mucosal maltase maltose glucose +glucose lactase lactose glucose + galactose invertase sucrose glucose +fructose dextrinase isomaltose glucose +glucose
5 Digestion Pre-stomach Salivary amylase : a 1-4 endoglycosidase a 1-6 link amylase Limit dextrins a 1-4 link maltose
6 Stomach Not much carbohydrate digestion
7 Small Intestine Pancreatic enzymes a-amylase maltose amylose a amylase amylopectin
8 a Limit dextrins sucrase maltase lucoamylase (maltase) or a-dextrinase a-dextrinase
9 A. More than 60% of our foods are carbohydrates. Starch, glycogen, sucrose, lactose and cellulose are the chief carbohydrates in our food. Before intestinal absorption, they are hydrolysed to hexose sugars (glucose, galactose and fructose). B. A family of a glycosidases that degrade carbohydrate into their monohexose components catalyzes hydrolysis of glycocidic bonds. These enzymes are usually specific to the type of bond to be broken.
10 Digestion of carbohydrate by salivary α -amylase (ptylin) in the mouth: A. This enzyme is produced by salivary glands. Its optimum ph is 6.7. B. It is activated by chloride ions (cl-). C. It acts on cooked starch and glycogen breaking α 1-4 bonds, converting them into maltose [a disaccharide containing two glucose molecules attached by α 1-4 linkage]. This bond is not attacked by - amylase. Because both starch and glycogen also contain 1-6 bonds, the resulting digest contains isomaltose [a disaccharide in which two glucose molecules are attached by 1-6 linkage]. E. Because food remains for a short time in the mouth, digestion of starch and glycogen may be incomplete and gives a partial digestion products called: starch dextrins (amylodextrin, erythrodextrin and achrodextrin). F. Therefore, digestion of starch and glycogen in the mouth gives maltose, isomaltose and starch dextrins.
11 III. ln the stomach: carbohydrate digestion stops temporarily due to the high acidity which inactivates the salivary - amylase. IV. Digestion of carbohydrate by the pancreatic - amylase small intestine in the small intestine. A. α-amylase enzyme is produced by pancreas and acts in small intestine. Its optimum ph is 7.1. B. It is also activated by chloride ions. C. It acts on cooked and uncooked starch, hydrolysing them into maltose and isomaltose. Final carbohydrate digestion by intestinal enzymes: A. The final digestive processes occur at the small intestine and include the action of several disaccharidases. These enzymes are secreted through and remain associated with the brush border of the intestinal mucosal cells.
12 B. The disaccharidases include: 1. Lactase (β-galactosidase) which hydrolyses lactose into two molecules of glucose and galactose: Lactose Lactase lucose + alactose 2. Maltase ( α-glucosidase), which hydrolyses maltose into two molecules of glucose: Maltose Maltase lucose + lucose 3. Sucrose (α-fructofuranosidase), which hydrolyses sucrose into two molecules of glucose and fructose: Sucrose Sucrose lucose + Fructose 4. α - dextrinase (oligo-1,6 glucosidase) which hydrolyze (1,6) linkage of isomaltose. Isomaltose Dextrinase lucose + lucose
13 VI. Digestion of cellulose: A. Cellulose contains β(1-4) bonds between glucose molecules. B. In humans, there is no β (1-4) glucosidase that can digest such bonds. So cellulose passes as such in stool. C. Cellulose helps water retention during the passage of food along the intestine producing larger and softer feces preventing constipation.
14 Absorption Portal for transport of virtually all nutrients
15 Absorption of carbohydrate A.The end products of carbohydrate digestion are monosaccharides: glucose, galactose and fructose. They are absorbed from the jejunum to portal veins to the liver, where fructose and galactose are transformed into glucose. B.Two mechanisms are responsible for absorption of monosaccharides: active transport (against concentration gradient i.e. from low to high concentration) and passive transport (by facilitated diffusion). C. For active transport to take place, the structure of sugar should have: 1. Hexose ring. 2. OH group at position 2 at the right side. Both of which are present in glucose and galactose. Fructose, which does not contain -OH group to the right at position 2 is absorbed more slowly than glucose and galactose by passive diffusion (slow process).
16 II. Mechanisms of absorption: A. Active transport: 1. Mechanism of active transport: a) In the cell membrane of the intestinal cells, there is a mobile carrier protein called sodium dependant glucose transporter (SL T-1) It transports glucose to inside the cell using energy. The energy is derived from sodium-potassium pump. The transporter has 2 separate sites, one for sodium and the other for glucose. It transports them from the intestinal lumen across cell membrane to the cytoplasm. Then both glucose and sodium are released into the cytoplasm allowing the carrier to return for more transport of glucose and sodium.
17 b) The sodium is transported from high to low concentration (with concentration gradient) and at the same time causes the carrier to transport glucose against its concentration gradient. The Na+ is expelled outside the cell by sodium pump. Which needs ATP as a source of energy. The reaction is catalyzed by an enzyme called "Adenosine triphosphatase (ATPase)". Active transport is much more faster than passive transport. c) Insulin increases the number of glucose transporters in tissues containing insulin receptors e.g. muscles and adipose tissue.
18 2. Inhibitors of active transport: a) Ouabin (cardiac glycoside): Inhibits adenosine triphosphatase (ATPase) necessary for hydrolysis of ATP that produces energy of sodium pump. b) Phlorhizin; Inhibits the binding of sodium in the carrier protein. B. Passive transport (facilitated diffusion): Sugars pass with concentration gradient i.e. from high to low concentration. It needs no energy. It occurs by means of a sodium independent facilitative transporter (LUT -5). Fructose and pentoses are absorbed by this mechanism. lucose and galactose can also use the same transporter if the concentration gradient is favorable.
19 Carbohydrates Serve as primary source of energy in the cell Central to all metabolic processes Cytosol - anaerobic lucose Hexokinase Pentose Phosphate Shunt lucose-6-p lc-1- phosphate glycolysis glycogen Pyruvate
20 cytosol mitochondria (aerobic) FATTY ACIDS Pyruvate Aceytl CoA AMINO ACIDS Krebs cycle Reducing equivalents Oxidative Phosphorylation (ATP)
21 IV. Fate of absorbed sugars: Monosaccharides (glucose, galactose and fructose) resulting from carbohydrate digestion are absorbed and undergo the following: A. Uptake by tissues (liver): After absorption the liver takes up sugars, where galactose and fructose are converted into glucose. B. lucose utilization by tissues: lucose may undergo one of the following fate: 1. Oxidation: through a) Major pathways (glycolysis and Krebs' cycle) for production of energy. b) Hexose monophosphate pathway: for production of ribose, deoxyribose and NADPH + H+ c) Uronic acid pathway, for production of glucuronic acid, which is used in detoxication and enters in the formation of mucopolysaccharide.
22 2. Storage: in the form of: a) lycogen: glycogenesis. b) Fat: lipogenesis. 3. Conversion: to substances of biological importance: a) Ribose, deoxyribose RNA and DNA. b) Lactose milk. c) lucosamine, galactosamine mucopolysaccharides. d) lucoronic acid mucopolysaccharides. e) Fructose in semen.
23 lucose Oxidation major Pathway
24 oxidation of glucose anaerobic oxidation (glycolysis) 2- aerobic oxidation (Krebs cycle) I. lycolysis (Embden Meyerhof Pathway): A. Definition: 1. lycolysis means oxidation of glucose to give pyruvate (in the presence of oxygen) or lactate (in the absence of oxygen).
25 B. Steps: Stages of glycolysis 1. Stage one (the energy requiring stage): a) One molecule of glucose is converted into two molecules of glycerosldhyde-3-phosphate. b) These steps requires 2 molecules of ATP (energy loss) 2. Stage two (the energy producing stage(: a) The 2 molecules of glyceroaldehyde-3-phosphate are converted into pyruvate (aerobic glycolysis) or lactate (anaerobic glycolysis(. b) These steps produce ATP molecules (energy production). C). Energy (ATP) production of glycolysis: ATP production = ATP produced - ATP utilized
26 Energy Investment Phase (steps 1-5)
27 Energy-Payoff Phase (Steps 6-10)
28 Energy production of glycolysis: ATP produced ATP utilized Net energy In absence of oxygen (anaerobic glycolysis) 4 ATP (Substrate level phosphorylation) 2ATP from 1,3 DP. 2ATP from phosphoenol pyruvate 2ATP From glucose to glucose -6-p. From fructose -6- p to fructose 1,6 p. 2 ATP In presence of oxygen (aerobic glycolysis) 4 ATP (substrate level phosphorylation) 2ATP from 1,3 BP. 2ATP from phosphoenol pyruvate. 2ATP -From glucose to glucose -6-p. From fructose -6- p to fructose 1,6 p. 6 ATP Or 8 ATP + 4ATP or 6ATP (from oxidation of 2 NADH + H in mitochondria).
29 D. oxidation of extramitochondrial NADH+H+: 1. cytoplasmic NADH+H+ cannot penetrate mitochondrial membrane, however, it can be used to produce energy (4 or 6 ATP) by respiratory chain phosphorylation in the mitochondria. 2. This can be done by using special carriers for hydrogen of NADH+H+ These carriers are either dihydroxyacetone phosphate (lycerophosphate shuttle) or oxaloacetate (aspartate malate shuttle). a) lycerophosphate shuttle: 1) It is important in certain muscle and nerve cells. 2) The final energy produced is 4 ATP. 3) Mechanism: - The coenzyme of cytoplasmic glycerol-3- phosphate dehydrogenase is NAD+. - The coenzyme of mitochodrial glycerol-3-phosphate dehydogenase is FAD. - Oxidation of FADH, in respiratory chain gives 2 ATP. As glycolysis gives 2 cytoplasmic NADH + H+ 2 mitochondrial FADH, 2 x 2 ATP = 4 ATP. b) Malate aspartate shuttle: 1) It is important in other tissues patriculary liver and heart. 2) The final energy produced is 6 ATP.
30 Pyruvate is converted to acetyl CoA under aerobic conditions and to lactate under anaerobic conditions.
31 Aerobic oxidation
32 lycogenesis & glycogenolysis In the liver and muscles, most of the glucose is changed into glycogen by the process of glycogenesis (anabolism). lycogen is stored in the liver and muscles until needed at some later time when glucose levels are low. If blood glucose levels are low, then eqinephrine and glucogon hormones are secreted to stimulate the conversion of glycogen to glucose. This process is called glycogenolysis (catabolism).
33
34 Blood glucose Sources of blood glucose diet (carbohydrate) 2- breakdown of glycogen (glycogenolysis ) 3- non carbohydrate sources (gluconeogenesis ) A- amino acid B- glycerol
35 Q- what are the benefits of blood glucose?
36 Abnormalities in blood glucose level hyperglycemia 2- diabetes mellitus 3- hypoglycemia Q-what are the causes of hypoglycemia, hyperglycemia?
37 Determination of blood glucose level fasting blood sugar (F.B.S ) 2- random blood sugar (R.B.S) 3- post prandial test (p.p.t) 4- oral glucose tolerance test (O..T.T)
38 Thank you
MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question.
 Ch23_PT MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) All of the following statements concerning digestion are correct except A) The major physical
Ch23_PT MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) All of the following statements concerning digestion are correct except A) The major physical
Copyright 2010 Pearson Education, Inc. Chapter Twenty Three 1
 23.2 Glucose Metabolism: An Overview When glucose enters a cell from the bloodstream, it is immediately converted to glucose 6- phosphate. Once this phosphate is formed, glucose is trapped within the cell
23.2 Glucose Metabolism: An Overview When glucose enters a cell from the bloodstream, it is immediately converted to glucose 6- phosphate. Once this phosphate is formed, glucose is trapped within the cell
AP BIOLOGY CHAPTER 7 Cellular Respiration Outline
 AP BIOLOGY CHAPTER 7 Cellular Respiration Outline I. How cells get energy. A. Cellular Respiration 1. Cellular respiration includes the various metabolic pathways that break down carbohydrates and other
AP BIOLOGY CHAPTER 7 Cellular Respiration Outline I. How cells get energy. A. Cellular Respiration 1. Cellular respiration includes the various metabolic pathways that break down carbohydrates and other
Energy Production In A Cell (Chapter 25 Metabolism)
 Energy Production In A Cell (Chapter 25 Metabolism) Large food molecules contain a lot of potential energy in the form of chemical bonds but it requires a lot of work to liberate the energy. Cells need
Energy Production In A Cell (Chapter 25 Metabolism) Large food molecules contain a lot of potential energy in the form of chemical bonds but it requires a lot of work to liberate the energy. Cells need
Chapter 15 Lecture Notes: Metabolism
 Chapter 15 Lecture Notes: Metabolism Educational Goals 1. Define the terms metabolism, metabolic pathway, catabolism, and anabolism. 2. Understand how ATP is formed from ADP and inorganic phosphate (P
Chapter 15 Lecture Notes: Metabolism Educational Goals 1. Define the terms metabolism, metabolic pathway, catabolism, and anabolism. 2. Understand how ATP is formed from ADP and inorganic phosphate (P
Cellular Respiration and Fermentation
 LECTURE PRESENTATIONS For CAMPBELL BIOLOGY, NINTH EDITION Jane B. Reece, Lisa A. Urry, Michael L. Cain, Steven A. Wasserman, Peter V. Minorsky, Robert B. Jackson Chapter 9 Cellular Respiration and Fermentation
LECTURE PRESENTATIONS For CAMPBELL BIOLOGY, NINTH EDITION Jane B. Reece, Lisa A. Urry, Michael L. Cain, Steven A. Wasserman, Peter V. Minorsky, Robert B. Jackson Chapter 9 Cellular Respiration and Fermentation
The correct answer is d C. Answer c is incorrect. Reliance on the energy produced by others is a characteristic of heterotrophs.
 1. An autotroph is an organism that a. extracts energy from organic sources b. converts energy from sunlight into chemical energy c. relies on the energy produced by other organisms as an energy source
1. An autotroph is an organism that a. extracts energy from organic sources b. converts energy from sunlight into chemical energy c. relies on the energy produced by other organisms as an energy source
The diagram below summarizes the effects of the compounds that cells use to regulate their own metabolism.
 Regulation of carbohydrate metabolism Intracellular metabolic regulators Each of the control point steps in the carbohydrate metabolic pathways in effect regulates itself by responding to molecules that
Regulation of carbohydrate metabolism Intracellular metabolic regulators Each of the control point steps in the carbohydrate metabolic pathways in effect regulates itself by responding to molecules that
1. The diagram below represents a biological process
 1. The diagram below represents a biological process 5. The chart below indicates the elements contained in four different molecules and the number of atoms of each element in those molecules. Which set
1. The diagram below represents a biological process 5. The chart below indicates the elements contained in four different molecules and the number of atoms of each element in those molecules. Which set
Special organ structures and functions conduct these tasks through the successive parts of the overall system.
 Chapter 5 Digestion, Absorption, and Metabolism Chapter 5 Lesson 5.1 Key Concepts Through a balanced system of mechanical and chemical digestion, food is broken down into smaller substances and the nutrients
Chapter 5 Digestion, Absorption, and Metabolism Chapter 5 Lesson 5.1 Key Concepts Through a balanced system of mechanical and chemical digestion, food is broken down into smaller substances and the nutrients
Chapter 25: Metabolism and Nutrition
 Chapter 25: Metabolism and Nutrition Chapter Objectives INTRODUCTION 1. Generalize the way in which nutrients are processed through the three major metabolic fates in order to perform various energetic
Chapter 25: Metabolism and Nutrition Chapter Objectives INTRODUCTION 1. Generalize the way in which nutrients are processed through the three major metabolic fates in order to perform various energetic
Todays Outline. Metabolism. Why do cells need energy? How do cells acquire energy? Metabolism. Concepts & Processes. The cells capacity to:
 and Work Metabolic Pathways Enzymes Features Factors Affecting Enzyme Activity Membrane Transport Diffusion Osmosis Passive Transport Active Transport Bulk Transport Todays Outline -Releasing Pathways
and Work Metabolic Pathways Enzymes Features Factors Affecting Enzyme Activity Membrane Transport Diffusion Osmosis Passive Transport Active Transport Bulk Transport Todays Outline -Releasing Pathways
What affects an enzyme s activity? General environmental factors, such as temperature and ph. Chemicals that specifically influence the enzyme.
 CH s 8-9 Respiration & Metabolism Metabolism A catalyst is a chemical agent that speeds up a reaction without being consumed by the reaction. An enzyme is a catalytic protein. Hydrolysis of sucrose by
CH s 8-9 Respiration & Metabolism Metabolism A catalyst is a chemical agent that speeds up a reaction without being consumed by the reaction. An enzyme is a catalytic protein. Hydrolysis of sucrose by
Digestive System Module 7: Chemical Digestion and Absorption: A Closer Look
 OpenStax-CNX module: m49457 1 Digestive System Module 7: Chemical Digestion and Absorption: A Closer Look Donna Browne Based on Chemical Digestion and Absorption: A Closer Look by OpenStax This work is
OpenStax-CNX module: m49457 1 Digestive System Module 7: Chemical Digestion and Absorption: A Closer Look Donna Browne Based on Chemical Digestion and Absorption: A Closer Look by OpenStax This work is
10.1 The function of Digestion pg. 402
 10.1 The function of Digestion pg. 402 Macromolecules and Living Systems The body is made up of more than 60 % water. The water is found in the cells cytoplasm, the interstitial fluid and the blood (5
10.1 The function of Digestion pg. 402 Macromolecules and Living Systems The body is made up of more than 60 % water. The water is found in the cells cytoplasm, the interstitial fluid and the blood (5
Harvesting Energy: Glycolysis and Cellular Respiration. Chapter 8
 Harvesting Energy: Glycolysis and Cellular Respiration Chapter 8 Overview of Glucose Breakdown The overall equation for the complete breakdown of glucose is: C 6 H 12 O 6 + 6O 2 6CO 2 + 6H 2 O + ATP The
Harvesting Energy: Glycolysis and Cellular Respiration Chapter 8 Overview of Glucose Breakdown The overall equation for the complete breakdown of glucose is: C 6 H 12 O 6 + 6O 2 6CO 2 + 6H 2 O + ATP The
CHAPTER 15: ANSWERS TO SELECTED PROBLEMS
 CHAPTER 15: ANSWERS T SELECTED PRBLEMS SAMPLE PRBLEMS ( Try it yourself ) 15.1 ur bodies can carry out the second reaction, because it requires less energy than we get from breaking down a molecule of
CHAPTER 15: ANSWERS T SELECTED PRBLEMS SAMPLE PRBLEMS ( Try it yourself ) 15.1 ur bodies can carry out the second reaction, because it requires less energy than we get from breaking down a molecule of
Chapter 7 Active Reading Guide Cellular Respiration and Fermentation
 Name: AP Biology Mr. Croft Chapter 7 Active Reading Guide Cellular Respiration and Fermentation Overview: Before getting involved with the details of cellular respiration and photosynthesis, take a second
Name: AP Biology Mr. Croft Chapter 7 Active Reading Guide Cellular Respiration and Fermentation Overview: Before getting involved with the details of cellular respiration and photosynthesis, take a second
008 Chapter 8. Student:
 008 Chapter 8 Student: 1. Some bacteria are strict aerobes and others are strict anaerobes. Some bacteria, however, are facultative anaerobes and can live with or without oxygen. If given the choice of
008 Chapter 8 Student: 1. Some bacteria are strict aerobes and others are strict anaerobes. Some bacteria, however, are facultative anaerobes and can live with or without oxygen. If given the choice of
Summary of Metabolism. Mechanism of Enzyme Action
 Summary of Metabolism Mechanism of Enzyme Action 1. The substrate contacts the active site 2. The enzyme-substrate complex is formed. 3. The substrate molecule is altered (atoms are rearranged, or the
Summary of Metabolism Mechanism of Enzyme Action 1. The substrate contacts the active site 2. The enzyme-substrate complex is formed. 3. The substrate molecule is altered (atoms are rearranged, or the
Anabolic and Catabolic Reactions are Linked by ATP in Living Organisms
 Chapter 5: Microbial Metabolism Microbial Metabolism Metabolism refers to all chemical reactions that occur within a living a living organism. These chemical reactions are generally of two types: Catabolic:
Chapter 5: Microbial Metabolism Microbial Metabolism Metabolism refers to all chemical reactions that occur within a living a living organism. These chemical reactions are generally of two types: Catabolic:
1. Enzymes. Biochemical Reactions. Chapter 5: Microbial Metabolism. 1. Enzymes. 2. ATP Production. 3. Autotrophic Processes
 Chapter 5: Microbial Metabolism 1. Enzymes 2. ATP Production 3. Autotrophic Processes 1. Enzymes Biochemical Reactions All living cells depend on biochemical reactions to maintain homeostasis. All of the
Chapter 5: Microbial Metabolism 1. Enzymes 2. ATP Production 3. Autotrophic Processes 1. Enzymes Biochemical Reactions All living cells depend on biochemical reactions to maintain homeostasis. All of the
Biochemistry of Cells
 Biochemistry of Cells 1 Carbon-based Molecules Although a cell is mostly water, the rest of the cell consists mostly of carbon-based molecules Organic chemistry is the study of carbon compounds Carbon
Biochemistry of Cells 1 Carbon-based Molecules Although a cell is mostly water, the rest of the cell consists mostly of carbon-based molecules Organic chemistry is the study of carbon compounds Carbon
1. Essay: The Digestive and Absorption Processes of Macronutrients
 Jenny Kim Professor Rosario Nutrition: Macronutrients Project June 26, 2014 1. Essay: The Digestive and Absorption Processes of Macronutrients Whenever we eat, the foods we ingest in our bodies undergo
Jenny Kim Professor Rosario Nutrition: Macronutrients Project June 26, 2014 1. Essay: The Digestive and Absorption Processes of Macronutrients Whenever we eat, the foods we ingest in our bodies undergo
LAB 3: DIGESTION OF ORGANIC MACROMOLECULES
 LAB 3: DIGESTION OF ORGANIC MACROMOLECULES INTRODUCTION Enzymes are a special class of proteins that lower the activation energy of biological reactions. These biological catalysts change the rate of chemical
LAB 3: DIGESTION OF ORGANIC MACROMOLECULES INTRODUCTION Enzymes are a special class of proteins that lower the activation energy of biological reactions. These biological catalysts change the rate of chemical
Digestive System Functions
 Digestive System Functions A. Gastrointestinal Processes 1. Ingestion: placing food in mouth (voluntary) 2. Propulsion: moving food through GI tract a. Peristalsis: alternating waves of contraction and
Digestive System Functions A. Gastrointestinal Processes 1. Ingestion: placing food in mouth (voluntary) 2. Propulsion: moving food through GI tract a. Peristalsis: alternating waves of contraction and
1. Explain the difference between fermentation and cellular respiration.
 : Harvesting Chemical Energy Name Period Overview: Before getting involved with the details of cellular respiration and photosynthesis, take a second to look at the big picture. Photosynthesis and cellular
: Harvesting Chemical Energy Name Period Overview: Before getting involved with the details of cellular respiration and photosynthesis, take a second to look at the big picture. Photosynthesis and cellular
CHEMISTRY AND BIOLOGICAL ROLE OF CARBOHYDRATES IN THE BODY-1
 CHEMISTRY AND BIOLOGICAL ROLE OF CARBOHYDRATES IN THE BODY-1 Chiral centers: Asymmetric carbons, i.e carbon atom with four different substituents Enantiomers : Mirror images Stereoisomers MONOSACCHARIDE
CHEMISTRY AND BIOLOGICAL ROLE OF CARBOHYDRATES IN THE BODY-1 Chiral centers: Asymmetric carbons, i.e carbon atom with four different substituents Enantiomers : Mirror images Stereoisomers MONOSACCHARIDE
Lactic Acid Dehydrogenase
 Lactic Acid Dehydrogenase Pyruvic Acid Dehydrogenase Complex Pyruvate to ACETYL coa CC CoA + CO 2 Mitochondria 3 carbon Pyruvate to 2 carbon ACETYL Coenzyme A Pyruvate Acetyl CoA + CO 2 + NADH + H + CO2
Lactic Acid Dehydrogenase Pyruvic Acid Dehydrogenase Complex Pyruvate to ACETYL coa CC CoA + CO 2 Mitochondria 3 carbon Pyruvate to 2 carbon ACETYL Coenzyme A Pyruvate Acetyl CoA + CO 2 + NADH + H + CO2
How Cells Release Chemical Energy Cellular Respiration
 How Cells Release Chemical Energy Cellular Respiration Overview of Carbohydrate Breakdown Pathways Photoautotrophs make ATP during photosynthesis and use it to synthesize glucose and other carbohydrates
How Cells Release Chemical Energy Cellular Respiration Overview of Carbohydrate Breakdown Pathways Photoautotrophs make ATP during photosynthesis and use it to synthesize glucose and other carbohydrates
Chapter 14- RESPIRATION IN PLANTS
 Chapter 14- RESPIRATION IN PLANTS Living cells require a continuous supply of energy for maintaining various life activities. This energy is obtained by oxidizing the organic compounds (carbohydrates,
Chapter 14- RESPIRATION IN PLANTS Living cells require a continuous supply of energy for maintaining various life activities. This energy is obtained by oxidizing the organic compounds (carbohydrates,
Worksheet 13.1. Chapter 13: Human biochemistry glossary
 Worksheet 13.1 Chapter 13: Human biochemistry glossary α-helix Refers to a secondary structure of a protein where the chain is twisted to form a regular helix, held by hydrogen bonds between peptide bonds
Worksheet 13.1 Chapter 13: Human biochemistry glossary α-helix Refers to a secondary structure of a protein where the chain is twisted to form a regular helix, held by hydrogen bonds between peptide bonds
Chapter 9 Review Worksheet Cellular Respiration
 1 of 5 11/9/2011 8:11 PM Name: Hour: Chapter 9 Review Worksheet Cellular Respiration Energy in General 1. Differentiate an autotroph from a hetertroph as it relates to obtaining energy and the processes
1 of 5 11/9/2011 8:11 PM Name: Hour: Chapter 9 Review Worksheet Cellular Respiration Energy in General 1. Differentiate an autotroph from a hetertroph as it relates to obtaining energy and the processes
Chapter 7 Cellular Respiration
 Phases of aerobic cellular respiration 1. Glycolysis 2. Transition or Acetyl-CoA reaction 3. Krebs cycle 4. Electron transport system Chapter 7 Cellular Respiration These phases are nothing more than metabolic
Phases of aerobic cellular respiration 1. Glycolysis 2. Transition or Acetyl-CoA reaction 3. Krebs cycle 4. Electron transport system Chapter 7 Cellular Respiration These phases are nothing more than metabolic
Photosynthesis (CO 2 + H 2 O C 6 H 12 O 6 + O 2 )
 The vital role of A This is the energy-rich compound that is the source of energy for all living things. It is a nucleotide, comprising a 5C sugar (ribose); an organic base (adenosine); and 3 phosphate
The vital role of A This is the energy-rich compound that is the source of energy for all living things. It is a nucleotide, comprising a 5C sugar (ribose); an organic base (adenosine); and 3 phosphate
Chapter 14 Glycolysis. Glucose. 2 Pyruvate 2 Lactate (sent to liver to be converted back to glucose) TCA Cycle
 Chapter 14 Glycolysis Requires mitochondria and O 2 Glucose glycolysis anaerobic respiration 2 Pyruvate 2 Lactate (sent to liver to be converted back to glucose) pyruvate dehydrogenase acetyl-coa TCA Cycle
Chapter 14 Glycolysis Requires mitochondria and O 2 Glucose glycolysis anaerobic respiration 2 Pyruvate 2 Lactate (sent to liver to be converted back to glucose) pyruvate dehydrogenase acetyl-coa TCA Cycle
Photosynthesis takes place in three stages:
 Photosynthesis takes place in three stages: Light-dependent reactions Light-independent reactions The Calvin cycle 1. Capturing energy from sunlight 2. Using energy to make ATP and NADPH 3. Using ATP and
Photosynthesis takes place in three stages: Light-dependent reactions Light-independent reactions The Calvin cycle 1. Capturing energy from sunlight 2. Using energy to make ATP and NADPH 3. Using ATP and
The Digestive System. You are what you eat!
 The Digestive System You are what you eat! Try to label the diagram (PENCIL!!) What is Digestion? Digestion: the breakdown of large macromolecules (proteins, fats, carbohydrates) into smaller molecules
The Digestive System You are what you eat! Try to label the diagram (PENCIL!!) What is Digestion? Digestion: the breakdown of large macromolecules (proteins, fats, carbohydrates) into smaller molecules
Chapter 49 - Nutrients and the Digestive System I. Nutrients (chemical substances necessary for organisms to grow and function properly)
 Chapter 49 - Nutrients and the Digestive System I. Nutrients (chemical substances necessary for organisms to grow and function properly) 6 basic nutrients - 4 food groups (milk, meat, fruit and vegetable,
Chapter 49 - Nutrients and the Digestive System I. Nutrients (chemical substances necessary for organisms to grow and function properly) 6 basic nutrients - 4 food groups (milk, meat, fruit and vegetable,
Chapter 8: Energy and Metabolism
 Chapter 8: Energy and Metabolism 1. Discuss energy conversions and the 1 st and 2 nd law of thermodynamics. Be sure to use the terms work, potential energy, kinetic energy, and entropy. 2. What are Joules
Chapter 8: Energy and Metabolism 1. Discuss energy conversions and the 1 st and 2 nd law of thermodynamics. Be sure to use the terms work, potential energy, kinetic energy, and entropy. 2. What are Joules
AP Bio Photosynthesis & Respiration
 AP Bio Photosynthesis & Respiration Multiple Choice Identify the letter of the choice that best completes the statement or answers the question. 1. What is the term used for the metabolic pathway in which
AP Bio Photosynthesis & Respiration Multiple Choice Identify the letter of the choice that best completes the statement or answers the question. 1. What is the term used for the metabolic pathway in which
SOME Important Points About Cellular Energetics by Dr. Ty C.M. Hoffman
 SOME Important Points About Cellular Energetics by Dr. Ty C.M. Hoffman An Introduction to Metabolism Most biochemical processes occur as biochemical pathways, each individual reaction of which is catalyzed
SOME Important Points About Cellular Energetics by Dr. Ty C.M. Hoffman An Introduction to Metabolism Most biochemical processes occur as biochemical pathways, each individual reaction of which is catalyzed
Integration of Metabolism
 I. Central Themes of Metabolism 1. ATP is the universal energy carrier. Integration of Metabolism Bryant Miles 2. ATP is generated by the oxidation of metabolic fuels Glucose Fatty Acids Amino Acids 3.
I. Central Themes of Metabolism 1. ATP is the universal energy carrier. Integration of Metabolism Bryant Miles 2. ATP is generated by the oxidation of metabolic fuels Glucose Fatty Acids Amino Acids 3.
Cellular Respiration An Overview
 Why? Cellular Respiration An Overview What are the phases of cellular respiration? All cells need energy all the time, and their primary source of energy is ATP. The methods cells use to make ATP vary
Why? Cellular Respiration An Overview What are the phases of cellular respiration? All cells need energy all the time, and their primary source of energy is ATP. The methods cells use to make ATP vary
PHOTOSYNTHESIS AND CELLULAR RESPIRATION
 reflect Wind turbines shown in the photo on the right are large structures with blades that move in response to air movement. When the wind blows, the blades rotate. This motion generates energy that is
reflect Wind turbines shown in the photo on the right are large structures with blades that move in response to air movement. When the wind blows, the blades rotate. This motion generates energy that is
Biological molecules:
 Biological molecules: All are organic (based on carbon). Monomers vs. polymers: Monomers refer to the subunits that, when polymerized, make up a larger polymer. Monomers may function on their own in some
Biological molecules: All are organic (based on carbon). Monomers vs. polymers: Monomers refer to the subunits that, when polymerized, make up a larger polymer. Monomers may function on their own in some
Chapter 16 The Citric Acid Cycle
 Chapter 16 The Citric Acid Cycle Multiple Choice Questions 1. Which of the following is not true of the reaction catalyzed by the pyruvate dehydrogenase complex? A) Biotin participates in the decarboxylation.
Chapter 16 The Citric Acid Cycle Multiple Choice Questions 1. Which of the following is not true of the reaction catalyzed by the pyruvate dehydrogenase complex? A) Biotin participates in the decarboxylation.
BCOR 011 Exam 2, 2004
 BCOR 011 Exam 2, 2004 Name: Section: MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1. According to the first law of thermodynamics, A. the universe
BCOR 011 Exam 2, 2004 Name: Section: MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1. According to the first law of thermodynamics, A. the universe
4. Power: Pathways that make ATP
 Page 1 of 40 4. Power: Pathways that make ATP 4.1 The human body has a duel power system In hybrid cars, such as a Prius TM, power is supplied by two systems. For long-term travel, gasoline is used to
Page 1 of 40 4. Power: Pathways that make ATP 4.1 The human body has a duel power system In hybrid cars, such as a Prius TM, power is supplied by two systems. For long-term travel, gasoline is used to
1. What has a higher stored energy potential per gram, glycogen or triglycerides? Explain.
 Lipid Metabolism 1. What has a higher stored energy potential per gram, glycogen or triglycerides? Explain. 2. How can excess acetyl CoA trapped in the mitochondria, be utilized as a substrate for fatty
Lipid Metabolism 1. What has a higher stored energy potential per gram, glycogen or triglycerides? Explain. 2. How can excess acetyl CoA trapped in the mitochondria, be utilized as a substrate for fatty
How To Understand The Chemistry Of Organic Molecules
 CHAPTER 3 THE CHEMISTRY OF ORGANIC MOLECULES 3.1 Organic Molecules The chemistry of carbon accounts for the diversity of organic molecules found in living things. Carbon has six electrons, four of which
CHAPTER 3 THE CHEMISTRY OF ORGANIC MOLECULES 3.1 Organic Molecules The chemistry of carbon accounts for the diversity of organic molecules found in living things. Carbon has six electrons, four of which
10.2 The Human Digestive System pg. 411
 10.2 The Human Digestive System pg. 411 The human digestive system is made up of a group of organs working together. The digestive tract is made up of the mouth, esophagus, stomach, small intestine, and
10.2 The Human Digestive System pg. 411 The human digestive system is made up of a group of organs working together. The digestive tract is made up of the mouth, esophagus, stomach, small intestine, and
Chapter 16 The Citric Acid Cycle
 Chapter 16 The Citric Acid Cycle Multiple Choice Questions 1. Production of acetyl-coa (activated acetate) Page: 603 Difficulty: 2 Ans: A Which of the following is not true of the reaction catalyzed by
Chapter 16 The Citric Acid Cycle Multiple Choice Questions 1. Production of acetyl-coa (activated acetate) Page: 603 Difficulty: 2 Ans: A Which of the following is not true of the reaction catalyzed by
Chapter 3 Molecules of Cells
 Bio 100 Molecules of cells 1 Chapter 3 Molecules of Cells Compounds containing carbon are called organic compounds Molecules such as methane that are only composed of carbon and hydrogen are called hydrocarbons
Bio 100 Molecules of cells 1 Chapter 3 Molecules of Cells Compounds containing carbon are called organic compounds Molecules such as methane that are only composed of carbon and hydrogen are called hydrocarbons
Advanced Subsidiary GCE Biology
 Advanced Subsidiary GCE Biology Unit F214 - Communication, Homeostasis and Energy - High banded OCR has produced these candidate style answers to support teachers in interpreting the assessment criteria
Advanced Subsidiary GCE Biology Unit F214 - Communication, Homeostasis and Energy - High banded OCR has produced these candidate style answers to support teachers in interpreting the assessment criteria
7 Answers to end-of-chapter questions
 7 Answers to end-of-chapter questions Multiple choice questions 1 B 2 B 3 A 4 B 5 A 6 D 7 C 8 C 9 B 10 B Structured questions 11 a i Maintenance of a constant internal environment within set limits i Concentration
7 Answers to end-of-chapter questions Multiple choice questions 1 B 2 B 3 A 4 B 5 A 6 D 7 C 8 C 9 B 10 B Structured questions 11 a i Maintenance of a constant internal environment within set limits i Concentration
Work and Energy in Muscles
 Work and Energy in Muscles Why can't I sprint forever? I'll start this section with that silly question. What lies behind the undisputable observation that we must reduce speed if we want to run longer
Work and Energy in Muscles Why can't I sprint forever? I'll start this section with that silly question. What lies behind the undisputable observation that we must reduce speed if we want to run longer
Essentials of Anatomy and Physiology, 5e (Martini/Nath) Chapter 17 Nutrition and Metabolism. Multiple-Choice Questions
 Essentials of Anatomy and Physiology, 5e (Martini/Nath) Chapter 17 Nutrition and Metabolism Multiple-Choice Questions 1) The sum of all of the biochemical processes going on within the human body at any
Essentials of Anatomy and Physiology, 5e (Martini/Nath) Chapter 17 Nutrition and Metabolism Multiple-Choice Questions 1) The sum of all of the biochemical processes going on within the human body at any
Chemistry 20 Chapters 15 Enzymes
 Chemistry 20 Chapters 15 Enzymes Enzymes: as a catalyst, an enzyme increases the rate of a reaction by changing the way a reaction takes place, but is itself not changed at the end of the reaction. An
Chemistry 20 Chapters 15 Enzymes Enzymes: as a catalyst, an enzyme increases the rate of a reaction by changing the way a reaction takes place, but is itself not changed at the end of the reaction. An
-Loss of energy -Loss of hydrogen from carbons. -Gain of energy -Gain of hydrogen to carbons
 Cellular Respiration- Equation C6H12O6 + 6O2 6CO2 +6H20 and energy -The energy is released from the chemical bonds in the complex organic molecules -The catabolic process of releasing energy from food
Cellular Respiration- Equation C6H12O6 + 6O2 6CO2 +6H20 and energy -The energy is released from the chemical bonds in the complex organic molecules -The catabolic process of releasing energy from food
Name: Hour: Elements & Macromolecules in Organisms
 Name: Hour: Elements & Macromolecules in Organisms Most common elements in living things are carbon, hydrogen, nitrogen, and oxygen. These four elements constitute about 95% of your body weight. All compounds
Name: Hour: Elements & Macromolecules in Organisms Most common elements in living things are carbon, hydrogen, nitrogen, and oxygen. These four elements constitute about 95% of your body weight. All compounds
Macromolecules 1 Carbohydrates, Lipids & Nucleic Acids
 VEA Bringing Learning to Life Program Support Notes Macromolecules 1 Carbohydrates, Lipids & Nucleic Acids Grades 10 - College 25mins Teacher Notes by Sue Wright, B. Sc., Dip. Ed. Produced by VEA Pty Ltd
VEA Bringing Learning to Life Program Support Notes Macromolecules 1 Carbohydrates, Lipids & Nucleic Acids Grades 10 - College 25mins Teacher Notes by Sue Wright, B. Sc., Dip. Ed. Produced by VEA Pty Ltd
Methods of Grading S/N Style of grading Percentage Score 1 Attendance, class work and assignment 10 2 Test 20 3 Examination 70 Total 100
 COURSE: MIB 303 Microbial Physiology and Metabolism (3 Units- Compulsory) Course Duration: Three hours per week for 15 weeks (45 hours). Lecturer: Jimoh, S.O. B.Sc., M.Sc, Ph.D Microbiology (ABU, Zaria)
COURSE: MIB 303 Microbial Physiology and Metabolism (3 Units- Compulsory) Course Duration: Three hours per week for 15 weeks (45 hours). Lecturer: Jimoh, S.O. B.Sc., M.Sc, Ph.D Microbiology (ABU, Zaria)
Cellular Respiration & Metabolism. Metabolism. Coupled Reactions: Bioenergetics. Cellular Respiration: ATP is the cell s rechargable battery
 Cellular Respiration & Metabolism Metabolic Pathways: a summary Metabolism Bioenergetics Flow of energy in living systems obeys: 1 st law of thermodynamics: Energy can be transformed, but it cannot be
Cellular Respiration & Metabolism Metabolic Pathways: a summary Metabolism Bioenergetics Flow of energy in living systems obeys: 1 st law of thermodynamics: Energy can be transformed, but it cannot be
BIOLOGICAL MOLECULES OF LIFE
 BIOLOGICAL MOLECULES OF LIFE C A R B O H Y D R A T E S, L I P I D S, P R O T E I N S, A N D N U C L E I C A C I D S The Academic Support Center @ Daytona State College (Science 115, Page 1 of 29) Carbon
BIOLOGICAL MOLECULES OF LIFE C A R B O H Y D R A T E S, L I P I D S, P R O T E I N S, A N D N U C L E I C A C I D S The Academic Support Center @ Daytona State College (Science 115, Page 1 of 29) Carbon
Absorption and Transport of Nutrients
 Page1 Digestion Food travels from mouth esophagus stomach small intestine colon rectum anus. Food mixes with digestive juices, moving it through the digestive tract Large molecules of food are broken into
Page1 Digestion Food travels from mouth esophagus stomach small intestine colon rectum anus. Food mixes with digestive juices, moving it through the digestive tract Large molecules of food are broken into
Chapter 5: The Structure and Function of Large Biological Molecules
 Name Period Concept 5.1 Macromolecules are polymers, built from monomers 1. The large molecules of all living things fall into just four main classes. Name them. 2. Circle the three classes that are called
Name Period Concept 5.1 Macromolecules are polymers, built from monomers 1. The large molecules of all living things fall into just four main classes. Name them. 2. Circle the three classes that are called
The Human Digestive System
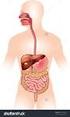 The Human Digestive System Name: Section: Date: Page 1 of 10 Page 2 of 10 Page 3 of 10 Page 4 of 10 Page 5 of 10 Page 6 of 10 Putting it All Together Digestive Enzymes Page 7 of 10 Page 8 of 10 Page 9
The Human Digestive System Name: Section: Date: Page 1 of 10 Page 2 of 10 Page 3 of 10 Page 4 of 10 Page 5 of 10 Page 6 of 10 Putting it All Together Digestive Enzymes Page 7 of 10 Page 8 of 10 Page 9
Organic Compounds. Essential Questions: What is Organic? What are the 4 major Organic Compounds? How are they made? What are they used for?
 Organic Compounds Essential Questions: What is Organic? What are the 4 major Organic Compounds? How are they made? What are they used for? Aristotle: Francesco Redi: What do we already know? Spontaneous
Organic Compounds Essential Questions: What is Organic? What are the 4 major Organic Compounds? How are they made? What are they used for? Aristotle: Francesco Redi: What do we already know? Spontaneous
Microbial Metabolism. Chapter 5. Enzymes. Enzyme Components. Mechanism of Enzymatic Action
 Chapter 5 Microbial Metabolism Metabolism is the sum of all chemical reactions within a living organism, including anabolic (biosynthetic) reactions and catabolic (degradative) reactions. Anabolism is
Chapter 5 Microbial Metabolism Metabolism is the sum of all chemical reactions within a living organism, including anabolic (biosynthetic) reactions and catabolic (degradative) reactions. Anabolism is
Biology 20 Cellular Respiration Review NG Know the process of Cellular Respiration (use this picture if it helps):
 Biology 20 Cellular Respiration Review NG Know the process of Cellular Respiration (use this picture if it helps): 1) How many ATP molecules are produced for each glucose molecule used in fermentation?
Biology 20 Cellular Respiration Review NG Know the process of Cellular Respiration (use this picture if it helps): 1) How many ATP molecules are produced for each glucose molecule used in fermentation?
Anatomy and Physiology Placement Exam 2 Practice with Answers at End!
 Anatomy and Physiology Placement Exam 2 Practice with Answers at End! General Chemical Principles 1. bonds are characterized by the sharing of electrons between the participating atoms. a. hydrogen b.
Anatomy and Physiology Placement Exam 2 Practice with Answers at End! General Chemical Principles 1. bonds are characterized by the sharing of electrons between the participating atoms. a. hydrogen b.
Chapter 9 Mitochondrial Structure and Function
 Chapter 9 Mitochondrial Structure and Function 1 2 3 Structure and function Oxidative phosphorylation and ATP Synthesis Peroxisome Overview 2 Mitochondria have characteristic morphologies despite variable
Chapter 9 Mitochondrial Structure and Function 1 2 3 Structure and function Oxidative phosphorylation and ATP Synthesis Peroxisome Overview 2 Mitochondria have characteristic morphologies despite variable
Digestive System Lecture 5 Winter 2014
 Digestive System Lecture 5 Winter 2014 This lecture tells the story of the Flow of Matter from Food to Cells. The pictures are only there to help you visualize structures don t worry about names of structures
Digestive System Lecture 5 Winter 2014 This lecture tells the story of the Flow of Matter from Food to Cells. The pictures are only there to help you visualize structures don t worry about names of structures
Electron transport chain, oxidative phosphorylation & mitochondrial transport systems. Joško Ivica
 Electron transport chain, oxidative phosphorylation & mitochondrial transport systems Joško Ivica Electron transport chain & oxidative phosphorylation collects e - & -H Oxidation of foodstuffs oxidizes
Electron transport chain, oxidative phosphorylation & mitochondrial transport systems Joško Ivica Electron transport chain & oxidative phosphorylation collects e - & -H Oxidation of foodstuffs oxidizes
Multiple Choice Identify the choice that best completes the statement or answers the question.
 AP bio fall 2014 final exam prep Multiple Choice Identify the choice that best completes the statement or answers the question. 1. According to the first law of thermodynamics, a. the energy of a system
AP bio fall 2014 final exam prep Multiple Choice Identify the choice that best completes the statement or answers the question. 1. According to the first law of thermodynamics, a. the energy of a system
Chapter 9 Cellular Respiration
 Chapter 9 Cellular Respiration Electrons carried in NADH Mitochondrion Glucose Glycolysis Pyruvic acid Krebs Cycle Electrons carried in NADH and FADH 2 Electron Transport Chain Cytoplasm Mitochondrion
Chapter 9 Cellular Respiration Electrons carried in NADH Mitochondrion Glucose Glycolysis Pyruvic acid Krebs Cycle Electrons carried in NADH and FADH 2 Electron Transport Chain Cytoplasm Mitochondrion
Copyright 2000-2003 Mark Brandt, Ph.D. 54
 Pyruvate Oxidation Overview of pyruvate metabolism Pyruvate can be produced in a variety of ways. It is an end product of glycolysis, and can be derived from lactate taken up from the environment (or,
Pyruvate Oxidation Overview of pyruvate metabolism Pyruvate can be produced in a variety of ways. It is an end product of glycolysis, and can be derived from lactate taken up from the environment (or,
Cellular Respiration Worksheet 1. 1. What are the 3 phases of the cellular respiration process? Glycolysis, Krebs Cycle, Electron Transport Chain.
 Cellular Respiration Worksheet 1 1. What are the 3 phases of the cellular respiration process? Glycolysis, Krebs Cycle, Electron Transport Chain. 2. Where in the cell does the glycolysis part of cellular
Cellular Respiration Worksheet 1 1. What are the 3 phases of the cellular respiration process? Glycolysis, Krebs Cycle, Electron Transport Chain. 2. Where in the cell does the glycolysis part of cellular
- Oxygen is needed for cellular respiration [OVERHEAD, fig. 6.2, p. 90 / 4th: 6.1] - lungs provide oxygen to blood, blood brings oxygen to the cells.
![- Oxygen is needed for cellular respiration [OVERHEAD, fig. 6.2, p. 90 / 4th: 6.1] - lungs provide oxygen to blood, blood brings oxygen to the cells. - Oxygen is needed for cellular respiration [OVERHEAD, fig. 6.2, p. 90 / 4th: 6.1] - lungs provide oxygen to blood, blood brings oxygen to the cells.](/thumbs/40/21014327.jpg) Cellular respiration - how cells make energy - Oxygen is needed for cellular respiration [OVERHEAD, fig. 6.2, p. 90 / 4th: 6.1] - ATP - this is provided by the lungs - lungs provide oxygen to blood, blood
Cellular respiration - how cells make energy - Oxygen is needed for cellular respiration [OVERHEAD, fig. 6.2, p. 90 / 4th: 6.1] - ATP - this is provided by the lungs - lungs provide oxygen to blood, blood
WATER CHAPTER 3 - BIOCHEMISTRY "THE CHEMISTRY OF LIFE" POLARITY HYDROGEN BONDING
 CHAPTER 3 - BIOCHEMISTRY "THE CHEMISTRY OF LIFE" WATER Compare the body of the jellyfish with our own bodies. The jellyfish will die if it is removed from its water environment, yet we can live in the
CHAPTER 3 - BIOCHEMISTRY "THE CHEMISTRY OF LIFE" WATER Compare the body of the jellyfish with our own bodies. The jellyfish will die if it is removed from its water environment, yet we can live in the
Regulation of the Citric Acid Cycle
 Regulation of the itric Acid ycle I. hanges in Free Energy February 17, 2003 Bryant Miles kj/mol 40 20 0 20 40 60 80 Reaction DGo' DG TA Free Energy hanges 1 2 3 4 5 6 7 8 9 1.) itrate Synthase 2.) Aconitase
Regulation of the itric Acid ycle I. hanges in Free Energy February 17, 2003 Bryant Miles kj/mol 40 20 0 20 40 60 80 Reaction DGo' DG TA Free Energy hanges 1 2 3 4 5 6 7 8 9 1.) itrate Synthase 2.) Aconitase
RESPIRATION AND FERMENTATION: AEROBIC AND ANAEROBIC OXIDATION OF ORGANIC MOLECULES. Bio 171 Week 6
 RESPIRATION AND FERMENTATION: AEROBIC AND ANAEROBIC OXIDATION OF ORGANIC MOLECULES Bio 171 Week 6 Procedure Label test tubes well, including group name 1) Add solutions listed to small test tubes 2) For
RESPIRATION AND FERMENTATION: AEROBIC AND ANAEROBIC OXIDATION OF ORGANIC MOLECULES Bio 171 Week 6 Procedure Label test tubes well, including group name 1) Add solutions listed to small test tubes 2) For
Figure 5. Energy of activation with and without an enzyme.
 Biology 20 Laboratory ENZYMES & CELLULAR RESPIRATION OBJECTIVE To be able to list the general characteristics of enzymes. To study the effects of enzymes on the rate of chemical reactions. To demonstrate
Biology 20 Laboratory ENZYMES & CELLULAR RESPIRATION OBJECTIVE To be able to list the general characteristics of enzymes. To study the effects of enzymes on the rate of chemical reactions. To demonstrate
Regulation of Metabolism. By Dr. Carmen Rexach Physiology Mt San Antonio College
 Regulation of Metabolism By Dr. Carmen Rexach Physiology Mt San Antonio College Energy Constant need in living cells Measured in kcal carbohydrates and proteins = 4kcal/g Fats = 9kcal/g Most diets are
Regulation of Metabolism By Dr. Carmen Rexach Physiology Mt San Antonio College Energy Constant need in living cells Measured in kcal carbohydrates and proteins = 4kcal/g Fats = 9kcal/g Most diets are
Carbon-organic Compounds
 Elements in Cells The living substance of cells is made up of cytoplasm and the structures within it. About 96% of cytoplasm and its included structures are composed of the elements carbon, hydrogen, oxygen,
Elements in Cells The living substance of cells is made up of cytoplasm and the structures within it. About 96% of cytoplasm and its included structures are composed of the elements carbon, hydrogen, oxygen,
Chapter 5. The Structure and Function of Macromolecule s
 Chapter 5 The Structure and Function of Macromolecule s Most Macromolecules are polymers: Polymer: (poly: many; mer: part) Large molecules consisting of many identical or similar subunits connected together.
Chapter 5 The Structure and Function of Macromolecule s Most Macromolecules are polymers: Polymer: (poly: many; mer: part) Large molecules consisting of many identical or similar subunits connected together.
Cellular Energy: ATP & Enzymes. What is it? Where do organism s get it? How do they use it?
 Cellular Energy: ATP & Enzymes What is it? Where do organism s get it? How do they use it? Where does Energy come from? Ultimately, from the sun. It is transferred between organisms in the earth s lithosphere,
Cellular Energy: ATP & Enzymes What is it? Where do organism s get it? How do they use it? Where does Energy come from? Ultimately, from the sun. It is transferred between organisms in the earth s lithosphere,
CHAPTER 6 AN INTRODUCTION TO METABOLISM. Section B: Enzymes
 CHAPTER 6 AN INTRODUCTION TO METABOLISM Section B: Enzymes 1. Enzymes speed up metabolic reactions by lowering energy barriers 2. Enzymes are substrate specific 3. The active site in an enzyme s catalytic
CHAPTER 6 AN INTRODUCTION TO METABOLISM Section B: Enzymes 1. Enzymes speed up metabolic reactions by lowering energy barriers 2. Enzymes are substrate specific 3. The active site in an enzyme s catalytic
CITRIC ACID (KREB S, TCA) CYCLE
 ITRI AID (KREB S, TA) YLE Date: September 2, 2005 * Time: 10:40 am 11:30 am * Room: G202 Biomolecular Building Lecturer: Steve haney 515A Mary Ellen Jones Building stephen_chaney@med.unc.edu 9663286 *Please
ITRI AID (KREB S, TA) YLE Date: September 2, 2005 * Time: 10:40 am 11:30 am * Room: G202 Biomolecular Building Lecturer: Steve haney 515A Mary Ellen Jones Building stephen_chaney@med.unc.edu 9663286 *Please
ATP accounting so far ELECTRON TRANSPORT CHAIN & CHEMIOSMOSIS. The Essence of ETC: The Electron Transport Chain O 2
 accounting so far The final stage of cellular respiration: ELECTRON TRANSPORT CHAIN & CHEMIOSMOSIS Glycolysis 2 Kreb s cycle 2 Life takes a lot of energy to run, need to extract more energy than 4! There
accounting so far The final stage of cellular respiration: ELECTRON TRANSPORT CHAIN & CHEMIOSMOSIS Glycolysis 2 Kreb s cycle 2 Life takes a lot of energy to run, need to extract more energy than 4! There
Get It Right. Answers. Chapter 1: The Science of Life. A biologist studies all living things.
 Discover Biology 'N' Level Science Chapter 1 Chapter 1: The Science of Life A biologist studies all living things. In order to carry out the scientific method, we need to ask questions. Discover Biology
Discover Biology 'N' Level Science Chapter 1 Chapter 1: The Science of Life A biologist studies all living things. In order to carry out the scientific method, we need to ask questions. Discover Biology
Metabolism Poster Questions
 Metabolism Poster Questions Answer the following questions concerning respiration. 1. Consider the mitochondrial electron transport chain. a. How many hydrogen ions can be pumped for every NADH? b. How
Metabolism Poster Questions Answer the following questions concerning respiration. 1. Consider the mitochondrial electron transport chain. a. How many hydrogen ions can be pumped for every NADH? b. How
Topic 4: Digestion and Nutrition
 Topic 4: Digestion and Nutrition THE CONTENTS OF FOOD Food contains nutrients: Nutrients include: 1. 2. 3. 4. 5. Nutrients must be small enough to enter our cells. If they are too large they must be digested
Topic 4: Digestion and Nutrition THE CONTENTS OF FOOD Food contains nutrients: Nutrients include: 1. 2. 3. 4. 5. Nutrients must be small enough to enter our cells. If they are too large they must be digested
Microbial Metabolism. Biochemical diversity
 Microbial Metabolism Biochemical diversity Metabolism Define Requirements Energy Enzymes Rate Limiting step Reaction time Types Anabolic Endergonic Dehydration Catabolic Exergonic Hydrolytic Metabolism
Microbial Metabolism Biochemical diversity Metabolism Define Requirements Energy Enzymes Rate Limiting step Reaction time Types Anabolic Endergonic Dehydration Catabolic Exergonic Hydrolytic Metabolism
Medical Physiology Z.H.Al-Zubaydi
 Lec.13 Medical Physiology Z.H.Al-Zubaydi Functions of the Digestive System The major functions of the digestive tract include the following six processes, summarized in Figure 1: 1. Ingestion Food must
Lec.13 Medical Physiology Z.H.Al-Zubaydi Functions of the Digestive System The major functions of the digestive tract include the following six processes, summarized in Figure 1: 1. Ingestion Food must
I. Chapter 5 Summary. II. Nucleotides & Nucleic Acids. III. Lipids
 I. Chapter 5 Summary A. Simple Sugars (CH 2 O) n : 1. One C contains a carbonyl (C=O) rest contain - 2. Classification by functional group: aldoses & ketoses 3. Classification by number of C's: trioses,
I. Chapter 5 Summary A. Simple Sugars (CH 2 O) n : 1. One C contains a carbonyl (C=O) rest contain - 2. Classification by functional group: aldoses & ketoses 3. Classification by number of C's: trioses,
Carbon Hydrogen Oxygen Nitrogen
 Concept 1 - Thinking Practice 1. If the following molecules were to undergo a dehydration synthesis reaction, what molecules would result? Circle the parts of each amino acid that will interact and draw
Concept 1 - Thinking Practice 1. If the following molecules were to undergo a dehydration synthesis reaction, what molecules would result? Circle the parts of each amino acid that will interact and draw
AP BIOLOGY 2015 SCORING GUIDELINES
 AP BIOLOGY 2015 SCORING GUIDELINES Question 2 Figure 1. Glycolysis and pyruvate oxidation Figure 2. Krebs cycle Figure 3. Electron transport chain Cellular respiration includes the metabolic pathways of
AP BIOLOGY 2015 SCORING GUIDELINES Question 2 Figure 1. Glycolysis and pyruvate oxidation Figure 2. Krebs cycle Figure 3. Electron transport chain Cellular respiration includes the metabolic pathways of
PRESTWICK ACADEMY NATIONAL 5 BIOLOGY CELL BIOLOGY SUMMARY
 Name PRESTWICK ACADEMY NATIONAL 5 BIOLOGY CELL BIOLOGY SUMMARY Cell Structure Identify animal, plant, fungal and bacterial cell ultrastructure and know the structures functions. Plant cell Animal cell
Name PRESTWICK ACADEMY NATIONAL 5 BIOLOGY CELL BIOLOGY SUMMARY Cell Structure Identify animal, plant, fungal and bacterial cell ultrastructure and know the structures functions. Plant cell Animal cell
