URSO 250 (ursodiol) tablets, for oral use URSO Forte (ursodiol) tablets, for oral use Initial U.S. Approval: 1997
|
|
|
- Gillian Kelley
- 7 years ago
- Views:
Transcription
1 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use URSO 250 or URSO FORTE safely and effectively. See full prescribing information for URSO 250 and URSO FORTE. URSO 250 (ursodiol) tablets, for oral use URSO Forte (ursodiol) tablets, for oral use Initial U.S. Approval: INDICATIONS AND USAGE URSO 250 and URSO Forte (ursodiol) tablets are bile acids indicated for the treatment of patients with primary biliary cirrhosis (1) DOSAGE AND ADMINISTRATION Recommended adult dosage: mg/kg/day administered in two to four divided doses with food (2.1) Scored URSO Forte tablet: scored tablet can be broken in halves to provide recommended dosage (2.2, 16.2) DOSAGE FORMS AND STRENGTHS URSO 250: 250 mg tablet (3) URSO Forte: 500 mg scored tablet (3) CONTRAINDICATIONS Patients with complete biliary obstruction and known hypersensitivity or intolerance to ursodiol or any of the components of the formulation (4) WARNINGS AND PRECAUTIONS Patients with variceal bleeding, hepatic encephalopathy, ascites or in need of an urgent liver transplant, should receive appropriate specific treatment (5). Liver function tests (γ-gt, alkaline phosphatase, AST, ALT ) and bilirubin level should be monitored. Treatment discontinuation should be considered if parameters increase to a level considered clinically significant in patients with stable historical liver function test levels (5). Caution should be exercised to maintain patients bile flow (5) ADVERSE REACTIONS Most common adverse reactions reported with the use of ursodiol during worldwide postmarketing and clinical experience ( 1%) are, in alphabetical order: abdominal discomfort, abdominal pain, alopecia, diarrhea, nausea, pruritus, and rash (6) To report SUSPECTED ADVERSE REACTIONS, contact Aptalis Pharma US, Inc. at or FDA at FDA-1088 or DRUG INTERACTIONS Bile Acid Sequestering Agents: May interfere with the action of URSO 250 and URSO Forte by reducing its absorption (7.1) Aluminum-based Antacids: May interfere with the action of URSO 250 and URSO Forte by reducing its absorption (7.2) Drugs that alter the metabolism of lipids or induce cholestasis may interfere with the action of URSO 250 and URSO Forte (7.3) See 17 for PATIENT COUNSELING INFORMATION. Revised: 6/2013 FULL PRESCRIBING INFORMATION: CONTENTS* 1 INDICATIONS AND USAGE 2 DOSAGE AND ADMINISTRATION 2.1 General Dosing Information 2.2 Liver Function Tests 2.3 Scoring the URSO Forte Tablet 3 DOSAGE FORMS AND STRENGTHS 4 CONTRAINDICATIONS 5 WARNINGS AND PRECAUTIONS 6 ADVERSE REACTIONS 6.1 Clinical Studies Experience 6.2 Postmarketing Experience 7 DRUG INTERACTIONS 7.1 Bile Acid Sequestering Agents 7.2 Aluminum-based Antacids 7.3 Drugs Affecting Lipid Metabolism 8 USE IN SPECIFIC POPULATIONS 8.1 Pregnancy 8.3 Nursing Mothers 8.4 Pediatric Use 10 OVERDOSAGE 11 DESCRIPTION 12 CLINICAL PHARMACOLOGY 12.1 Mechanism of Action 12.2 Pharmacodynamics 12.3 Pharmacokinetics 13 NONCLINICAL TOXICOLOGY 13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility 14 CLINICAL STUDIES 14.1 Efficacy of Ursodeoxycholic Acid Administered at 13 to 15 mg/kg/day in 3 or 4 Divided Doses to PBC Patients 14.2 Efficacy of Ursodiol Administered at 14 mg/kg/day as a Once Daily Dose to PBC Patients 14.3 Efficacy of URSO 250 Administered in Twice a Day Versus Four Times a Day Divided Dosing Schedules to PBC Patients 16 HOW SUPPLIED/STORAGE AND HANDLING 16.1 URSO URSO Forte 17 PATIENT COUNSELING INFORMATION 17.1 Appropriate Treatments 17.2 Drug Interactions *Sections or subsections omitted from the full prescribing information are not listed.
2 FULL PRESCRIBING INFORMATION 1 INDICATIONS AND USAGE URSO 250 and URSO Forte (ursodiol) tablets are indicated for the treatment of patients with primary biliary cirrhosis (PBC). 2 DOSAGE AND ADMINISTRATION 2.1 General Dosing Information The recommended adult dosage for URSO 250 and URSO Forte in the treatment of PBC is mg/kg/day administered in two to four divided doses with food. Dosing regimen should be adjusted according to each patient s need at the discretion of the physician. 2.2 Liver Function Tests Liver function tests (γ-gt, alkaline phosphatase, AST, ALT) and bilirubin levels should be monitored every month for three months after start of therapy, and every six months thereafter [see Warnings and Precautions (5.1)]. 2.3 Scoring the URSO Forte Tablet The URSO Forte scored tablet can be broken in halves to provide recommended dosage. To break URSO Forte scored tablet easily, place the tablet on a flat surface with the scored section on top. Hold the tablet with your thumbs placed close to the scored part of the tablet (groove). Then apply gentle pressure and snap the tablet segments apart (segments breaking incorrectly should not be used). The segments should be washed down unchewed, with water, keeping the segments in the mouth can reveal a bitter taste. Due to the bitter taste, segments should be stored separately from whole tablets. [see How Supplied/Storage and Handling (16.2)]. 3 DOSAGE FORMS AND STRENGTHS URSO 250: 250 mg tablet URSO Forte: 500 mg scored tablet 4 CONTRAINDICATIONS Patients with complete biliary obstruction and known hypersensitivity or intolerance to ursodiol or any of the components of the formulation. 5 WARNINGS AND PRECAUTIONS Patients with variceal bleeding, hepatic encephalopathy, ascites or in need of an urgent liver transplant, should receive appropriate specific treatment. 5.1 Abnormal Liver Function Tests Liver function tests (γ-gt, alkaline phosphatase, AST, ALT) and bilirubin levels should be monitored every month for three months after start of therapy, and every six months thereafter. This monitoring will allow the early detection of a possible deterioration of the hepatic function. Treatment discontinuation should be considered if the above parameters increase to a level considered clinically significant in patients with stable historical liver function test levels. Caution has to be exercised to maintain the bile flow of the patients taking ursodiol. 6 ADVERSE REACTIONS 6.1 Clinical Studies Experience Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice. The following table summarizes the adverse reactions observed in two placebo-controlled clinical trials. ADVERSE VISIT AT 12 VISIT AT 24 REACTIONS MONTHS MONTHS UDCA Placebo UDCA Diarrhea (1.32) --- Elevated creatinine (1.32) --- Elevated blood glucose 1 (1.18) (1.32) --- Leukopenia (2.63) --- Peptic ulcer (1.32) --- Skin rash (2.63) --- Thrombocytopenia (1.32) --- Placebo Note: Those adverse reactions occurring at the same or higher incidence in the placebo as in the UDCA group have been deleted from this table (this includes diarrhea and thrombocytopenia at 12 months, nausea/vomiting, fever and other toxicity). UDCA = Ursodeoxycholic acid = Ursodiol In a randomized, cross-over study in sixty PBC patients, seven patients (11.6%) reported nine adverse reactions: abdominal pain and asthenia (1 patient), nausea (3 patients), dyspepsia (2 patients) and anorexia and esophagitis (1 patient each). One patient on the twice a day regimen (total dose 1000 mg) withdrew due to nausea. All of these nine adverse reactions except esophagitis were observed with the twice a day regimen at a total daily dose of 1000 mg or greater. However, an adverse reaction may occur at any dose. 6.2 Postmarketing Experience The following adverse reactions, presented by system organ class in alphabetical order, have been identified during postapproval use of ursodiol. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. Gastrointestinal disorders: abdominal discomfort, abdominal pain, constipation, diarrhea, dyspepsia, nausea, vomiting. General disorders and administration site conditions: malaise, peripheral edema, pyrexia. Hepatobiliary disorders: jaundice (or aggravation of preexisting jaundice). Immune System Disorders: Drug hypersensitivity to include facial edema, urticaria, angioedema and laryngeal edema. Abnormal Laboratory Tests: ALT increased, AST increased, blood alkaline phosphatase increased, blood bilirubin increased, γ-gt increased, hepatic enzyme increased, liver function test abnormal, transaminases increased. Musculoskeletal and connective tissue disorders: myalgia Nervous system disorders: dizziness, headache.
3 Respiratory, thoracic and mediastinal disorders: cough. Skin and subcutaneous tissue disorder: alopecia, pruritus, rash. chemical name of ursodiol is 3α,7ß-dihydroxy-5ß-cholan-24-oic (C 24 H 40 O 4 ). Ursodiol has a molecular weight of Its structure is shown below. 7 DRUG INTERACTIONS 7.1 Bile Acid Sequestering Agents Bile acid sequestering agents such as cholestyramine and colestipol may interfere with the action of URSO 250 and URSO Forte by reducing its absorption. 7.2 Aluminum-based Antacids Aluminum-based antacids have been shown to adsorb bile acids in vitro and may be expected to interfere with URSO 250 and URSO Forte in the same manner as the bile acid sequestering agents. 7.3 Drugs Affecting Lipid Metabolism Estrogens, oral contraceptives, and clofibrate (and perhaps other lipid-lowering drugs) increase hepatic cholesterol secretion and encourage cholesterol gallstone formation and hence may counteract the effectiveness of URSO 250 and URSO Forte. 8 USE IN SPECIFIC POPULATIONS 8.1 Pregnancy Pregnancy Category B. Reproduction studies have been performed in pregnant rats at oral doses up to 22 times the recommended maximum human dose (based on body surface area) and in pregnant rabbits at oral doses up to 7 times the recommended maximum human dose (based on body surface area) and have revealed no evidence of impaired fertility or harm to the fetus due to ursodiol. There are no adequate or well-controlled studies in pregnant women. Because animal reproduction studies are not always predictive of human response, this drug should be used during pregnancy only if clearly needed. 8.3 Nursing Mothers It is not known whether ursodiol is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when URSO 250 and URSO Forte are administered to a nursing mother. 8.4 Pediatric Use The safety and effectiveness of URSO 250 and URSO Forte in pediatric patients have not been established. 10 OVERDOSAGE There have been no reports of accidental or intentional overdosage with ursodiol. Single oral doses of ursodiol at 10 g/kg in mice and dogs, and 5 g/kg in rats were not lethal. A single oral dose of ursodiol at 1.5 g/kg was lethal in hamsters. Symptoms of acute toxicity were salivation and vomiting in dogs, and ataxia, dyspnea, ptosis, agonal convulsions and coma in hamsters. 11 DESCRIPTION URSO 250 (ursodiol, 250 mg) is available as a film-coated tablet for oral administration. URSO Forte (ursodiol, 500 mg) is available as a scored film-coated tablet for oral administration. Ursodiol (ursodeoxycholic acid, UDCA) is a naturally occurring bile acid found in small quantities in normal human bile and in larger quantities in the biles of certain species of bears. It is a bitter-tasting white powder consisting of crystalline particles freely soluble in ethanol and glacial acetic acid, slightly soluble in chloroform, sparingly soluble in ether, and practically insoluble in water. The Inactive ingredients: microcrystalline cellulose, povidone, sodium starch glycolate, magnesium stearate, ethylcellulose, dibutyl sebacate, carnauba wax, hydroxypropyl methylcellulose, PEG 3350, PEG 8000, cetyl alcohol, sodium lauryl sulfate and hydrogen peroxide. 12 CLINICAL PHARMACOLOGY 12.1 Mechanism of Action Ursodiol, a naturally occurring hydrophilic bile acid, derived from cholesterol, is present as a minor fraction of the total human bile acid pool. Oral administration of ursodiol increases this fraction in a dose related manner, to become the major biliary acid, replacing/displacing toxic concentrations of endogenous hydrophobic bile acids that tend to accumulate in cholestatic liver disease. In addition to the replacement and displacement of toxic bile acids, other mechanisms of action include cytoprotection of the injured bile duct epithelial cells (cholangiocytes) against toxic effects of bile acids, inhibition of apotosis of hepatocytes, immunomodulatory effects, and stimulation of bile secretion by hepatocytes and cholangiocytes Pharmacodynamics Lithocholic acid, when administered chronically to animals, causes cholestatic liver injury that may lead to death from liver failure in certain species unable to form sulfate conjugates. Ursodiol is 7-dehydroxylated more slowly than chenodiol. For equimolar doses of ursodiol and chenodiol, steady state levels of lithocholic acid in biliary bile acids are lower during ursodiol administration than with chenodiol administration. Humans and chimpanzees can sulfate lithocholic acid. Although liver injury has not been associated with ursodiol therapy, a reduced capacity to sulfate may exist in some individuals Pharmacokinetics Ursodiol (UDCA) is normally present as a minor fraction of the total bile acids in humans (about 5%). Following oral administration, the majority of ursodiol is absorbed by passive diffusion and its absorption is incomplete. Once absorbed, ursodiol undergoes hepatic extraction to the extent of about 50% in the absence of liver disease. As the severity of liver disease increases, the extent of extraction decreases. In the liver, ursodiol is conjugated with glycine or taurine, then secreted into bile. These conjugates of ursodiol are absorbed in the small intestine by passive and active mechanisms. The conjugates can also be deconjugated in the ileum by intestinal enzymes, leading to the formation of free ursodiol that can be reabsorbed and reconjugated in the liver. Nonabsorbed ursodiol passes into the colon where it is mostly 7-dehydroxylated to lithocholic acid. Some ursodiol is epimerized to chenodiol (CDCA) via a 7-oxo intermediate. Chenodiol also undergoes 7- dehydroxylation to form lithocholic acid. These metabolites are poorly soluble and excreted in the feces. A small portion of lithocholic acid is reabsorbed, conjugated in the liver with glycine, or
4 taurine and sulfated at the 3 position. The resulting sulfated lithocholic acid conjugates are excreted in bile and then lost in feces. In healthy subjects, at least 70% of ursodiol (unconjugated) is bound to plasma protein. No information is available on the binding of conjugated ursodiol to plasma protein in healthy subjects or PBC patients. Its volume of distribution has not been determined, but is expected to be small since the drug is mostly distributed in the bile and small intestine. Ursodiol is excreted primarily in the feces. With treatment, urinary excretion increases, but remains less than 1% except in severe cholestatic liver disease. During chronic administration of ursodiol, it becomes a major biliary and plasma bile acid. At a chronic dose of 13 to 15 mg/kg/day, ursodiol constitutes 30-50% of biliary and plasma bile acids. 13 NONCLINICAL TOXICOLOGY 13.1 Carcinogenicity, Mutagenicity and Impairment of Fertility In two 24-month oral carcinogenicity studies in mice, ursodiol at doses up to 1,000 mg/kg/day (3,000 mg/m 2 /day) was not tumorigenic. Based on body surface area, for a 50 kg person of average height (1.46 m 2 body surface area), this dose represents 5.4 times the recommended maximum clinical dose of 15 mg/kg/day (555 mg/m 2 /day). In a two-year oral carcinogenicity study in Fischer 344 rats, ursodiol at doses up to 300 mg/kg/day (1,800 mg/m 2 /day, 3.2 times the recommended maximum human dose based on body surface area) was not tumorigenic. In a life-span ( weeks) oral carcinogenicity study, Sprague- Dawley rats were treated with doses of 33 to 300 mg/kg/day, 0.4 to 3.2 times the recommended maximum human dose based on body surface area. Ursodiol produced a significantly (p<0.5, Fisher's exact test) increased incidence of pheochromocytomas of the adrenal medulla in females of the highest dose group. In 103-week oral carcinogenicity studies of lithocholic acid, a metabolite of ursodiol, doses up to 250 mg/kg/day in mice and 500 mg/kg/day in rats did not produce any tumors. In a 78-week rat study, intrarectal instillation of lithocholic acid (1 mg/kg/day) for 13 months did not produce colorectal tumors. A tumor-promoting effect was observed when it was administered after a single intrarectal dose of a known carcinogen N-methyl-N'-nitro-N-nitrosoguanidine. On the other hand, in a 32-week rat study, ursodiol at a daily dose of 240 mg/kg (1,440 mg/m 2, 2.6 times the maximum recommended human dose based on body surface area) suppressed the colonic carcinogenic effect of another known carcinogen azoxymethane. Ursodiol was not genotoxic in the Ames test, the mouse lymphoma cell (L5178Y, TK+/-) forward mutation test, the human lymphocyte sister chromatid exchange test, the mouse spermatogonia chromosome aberration test, the Chinese hamster micronucleus test and the Chinese hamster bone marrow cell chromosome aberration test. Ursodiol at oral doses of up to 2,700 mg/kg/day (16,200 mg/m 2 /day, 29 times the recommended maximum human dose based on body surface area) was found to have no effect on fertility and reproductive performance of male and female rats. 14 CLINICAL STUDIES 14.1 Efficacy of Ursodeoxycholic Acid Administered at 13 to 15 mg/kg/day in 3 or 4 Divided Doses to PBC Patients A U.S., multicenter, randomized, double-blind, placebocontrolled study was conducted to evaluate the efficacy of ursodeoxycholic acid at a dose of 13 to 15 mg/kg/day, administered in 3 or 4 divided doses in 180 patients with PBC (78% received four times a day dosage). Upon completion of the double-blind portion, all patients entered an open-label active treatment extension phase. Treatment failure, the main efficacy end point measured during this study, was defined as death, need for liver transplantation, histologic progression by two stages or to cirrhosis, development of varices, ascites or encephalopathy, marked worsening of fatigue or pruritus, inability to tolerate the drug, doubling of serum bilirubin and voluntary withdrawal. After two years of double-blind treatment, the incidence of treatment failure was significantly (p 0.01) reduced in the URSO 250 mg group (20 of 86 (23%)) as compared to the placebo group (40 of 86 (47%)). Time to treatment failure, which excluded doubling of serum bilirubin and voluntary withdrawal, was also significantly (p 0.001) delayed in the URSO 250 treated group (n=86, 803.8±24.9 d vs ±24.4 d for the placebo group (n=86) on average) regardless of either histologic stage or baseline bilirubin levels (>1.8 or <1.8 mg/dl). Using a definition of treatment failure, which excluded doubling of serum bilirubin and voluntary withdrawal, time to treatment failure was significantly delayed in the URSO 250 group. In comparison with placebo, treatment with URSO 250 resulted in a significant improvement in the following serum hepatic biochemistries when compared to baseline: total bilirubin, SGOT, alkaline phosphatase and IgM Efficacy of Ursodiol Administered at 14 mg/kg/day as a Once Daily Dose to PBC Patients A second study conducted in Canada randomized 222 PBC patients to ursodiol, 14 mg/kg/day or placebo, administered as a once daily dose in a double-blind manner during a two-year period. At two years, a statistically significant (p 0.001) difference between the two treatments (n=106 for the URSO 250 group and n=106 for the placebo group), in favor of ursodiol, was demonstrated in the following: reduction in the proportion of patients exhibiting a more than 50% increase in serum bilirubin; median percent decrease in bilirubin (-17.12% for the URSO 250 group vs % for the placebo group), transaminases (-40.54% for the URSO 250 group vs % for the placebo group) and alkaline phosphatase (-47.61% for the URSO 250 group vs % for the placebo group); incidence of treatment failure; and time to treatment failure. The definition of treatment failure included: discontinuing the study for any reason; a total serum bilirubin level greater than or equal to 1.5 mg/dl or increasing to a level equal to or greater than two times the baseline level; and the development of ascites or encephalopathy. Evaluation of patients at 4 years or longer was inadequate due to the high drop out rate (n=10 withdrew from the URSO 250 group vs. n=15 from the placebo group) and small number of patients. Therefore, death, need for liver transplantation, histological progression by two stages or to cirrhosis, development of varices, ascites or encephalopathy, marked worsening of fatigue or pruritus, inability to tolerate the drug, doubling of serum bilirubin and voluntary withdrawal were not assessed Efficacy of URSO 250 Administered in Twice a Day Versus Four Times a Day Divided Dosing Schedules to PBC Patients A randomized, two-period crossover study in fifty PBC patients compared efficacy of URSO 250 (ursodiol) in twice a day versus four times a day divided dosing schedules in 50 patients for 6 months in each crossover period. Mean percent changes from baseline in liver test results and Mayo risk score (n=46) and serum enrichment with UDCA (n=34) were not statistically significant with any dosage at any time interval. This study demonstrated that UDCA (13 to 15 mg/kg/day) given twice a day is equally effective to UDCA given four times a day. In addition, URSO 250 was given as a single versus three times a day dosing schedules in 10 patients. Due to the small
5 number of patients in this arm of the study, it was not possible to conduct statistical comparisons between these regimens. 16 HOW SUPPLIED/STORAGE AND HANDLING 16.1 URSO 250 Each URSO 250 elliptical, biconvex, film-coated tablet, white, engraved with "URS785", contains 250 mg of ursodiol. Available in bottles of 100 tablets (NDC ) URSO Forte Each URSO Forte elliptical, biconvex, scored, film-coated tablet, white, engraved with "URS790", contains 500 mg of ursodiol. Available in bottles of 100 tablets (NDC ). Store at 20 C to 25 C (68 F to 77 F). Dispense in a tight container. Half-tablets (scored URSO Forte 500 mg tablets broken in half) maintain acceptable quality for up to 28 days when stored in the current packaging (bottles) at 20ºC to 25ºC (68ºF to 77ºF). Due to the bitter taste, the halved segments should be stored separately from the whole tablets [see Dosage and Administration (2.2)]. 17 PATIENT COUNSELING INFORMATION 17.1 Appropriate Treatments Patients with the following conditions should be instructed to receive appropriate management measures: variceal bleeding, hepatic encephalopathy, ascites, in need of an urgent liver transplant or hepatic function deterioration [see Warnings and Precautions (5)]. Caution has to be exercised to maintain the bile flow of the patients taking ursodiol Drug Interactions Patients should be informed that absorption of URSO 250 and URSO Forte may be reduced if they are taking bile acid sequestering agents, such as cholestyramine and colestipol, aluminum-based antacids, or drugs known to alter the metabolism of cholesterol [see Drug Interactions (7)]. Manufactured in Canada for: Aptalis Pharma US, Inc. 100 Somerset Corporate Boulevard Bridgewater, NJ 08807, USA URSO 250 and URSO Forte are registered trademarks of Aptalis Pharma US, Inc Aptalis Pharma US, Inc.
See 17 for PATIENT COUNSELING INFORMATION. Revised: 3/2016 FULL PRESCRIBING INFORMATION: CONTENTS* WARNING: ADRENAL CRISIS IN THE SETTING OF SHOCK OR
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use LYSODREN safely and effectively. See full prescribing information for LYSODREN. LYSODREN (mitotane)
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use LYSODREN safely and effectively. See full prescribing information for LYSODREN. LYSODREN (mitotane)
INITIATING ORAL AUBAGIO (teriflunomide) THERAPY
 FOR YOUR PATIENTS WITH RELAPSING FORMS OF MS INITIATING ORAL AUBAGIO (teriflunomide) THERAPY WARNING: HEPATOTOXICITY AND RISK OF TERATOGENICITY Severe liver injury including fatal liver failure has been
FOR YOUR PATIENTS WITH RELAPSING FORMS OF MS INITIATING ORAL AUBAGIO (teriflunomide) THERAPY WARNING: HEPATOTOXICITY AND RISK OF TERATOGENICITY Severe liver injury including fatal liver failure has been
Teriflunomide is the active metabolite of Leflunomide, a drug employed since 1994 for the treatment of rheumatoid arthritis (Baselt, 2011).
 Page 1 of 10 ANALYTE NAME AND STRUCTURE TERIFLUNOMIDE Teriflunomide TRADE NAME Aubagio CATEGORY Antimetabolite TEST CODE PURPOSE Therapeutic Drug Monitoring GENERAL RELEVANCY BACKGROUND sclerosis. The
Page 1 of 10 ANALYTE NAME AND STRUCTURE TERIFLUNOMIDE Teriflunomide TRADE NAME Aubagio CATEGORY Antimetabolite TEST CODE PURPOSE Therapeutic Drug Monitoring GENERAL RELEVANCY BACKGROUND sclerosis. The
2 DOSAGE AND ADMINISTRATION 2.1 Dosing and Dose Adjustment
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use Auryxia safely and effectively. See full prescribing information for Auryxia. Auryxia (ferric citrate)
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use Auryxia safely and effectively. See full prescribing information for Auryxia. Auryxia (ferric citrate)
Material Safety data sheet
 EMERGENCY OVERVIEW METFORMIN HYDROCHLORIDE TABLET contain Metformin and excipients generally considered to be non- toxic and non-hazardous in small quantities and under conditions of normal occupational
EMERGENCY OVERVIEW METFORMIN HYDROCHLORIDE TABLET contain Metformin and excipients generally considered to be non- toxic and non-hazardous in small quantities and under conditions of normal occupational
DO YOU HAVE ITCHY ALLERGY EYES? Find out about lasting relief
 DO YOU HAVE ITCHY ALLERGY EYES? Find out about lasting relief Common causes of itching due to eye allergies include: Pollen from trees, grasses, and ragweed Dust mites Cat dander Approximately 10 million
DO YOU HAVE ITCHY ALLERGY EYES? Find out about lasting relief Common causes of itching due to eye allergies include: Pollen from trees, grasses, and ragweed Dust mites Cat dander Approximately 10 million
VISTARIL (hydroxyzine pamoate) Capsules and Oral Suspension
 VISTARIL (hydroxyzine pamoate) Capsules and Oral Suspension DESCRIPTION Hydroxyzine pamoate is designated chemically as 1-(p-chlorobenzhydryl) 4- [2-(2-hydroxyethoxy) ethyl] diethylenediamine salt of 1,1
VISTARIL (hydroxyzine pamoate) Capsules and Oral Suspension DESCRIPTION Hydroxyzine pamoate is designated chemically as 1-(p-chlorobenzhydryl) 4- [2-(2-hydroxyethoxy) ethyl] diethylenediamine salt of 1,1
MATERIAL SAFETY DATA SHEET
 MATERIAL SAFETY DATA SHEET 1. IDENTIFICATION OF THE SUBSTANCE AND THE COMPANY Material Manufacturer Distributor Losartan Potassium and Hydrochlorothiazide Tablets 100 mg/12.5 mg Lupin Limited Goa 403 722
MATERIAL SAFETY DATA SHEET 1. IDENTIFICATION OF THE SUBSTANCE AND THE COMPANY Material Manufacturer Distributor Losartan Potassium and Hydrochlorothiazide Tablets 100 mg/12.5 mg Lupin Limited Goa 403 722
White, circular, biconvex, uncoated tablets with a score line on one side, plain on the other.
 Nausicalm Cyclizine hydrochloride Ph. Eur. 50 mg Presentation White, circular, biconvex, uncoated tablets with a score line on one side, plain on the other. Uses Actions The active ingredient-cyclizine
Nausicalm Cyclizine hydrochloride Ph. Eur. 50 mg Presentation White, circular, biconvex, uncoated tablets with a score line on one side, plain on the other. Uses Actions The active ingredient-cyclizine
EMEA PUBLIC STATEMENT ON LEFLUNOMIDE (ARAVA) - SEVERE AND SERIOUS HEPATIC REACTIONS -
 The European Agency for the Evaluation of Medicinal Products Post-authorisation evaluation of medicines for human use London, 12 March 2001 Doc. Ref: EMEA/H/5611/01/en EMEA PUBLIC STATEMENT ON LEFLUNOMIDE
The European Agency for the Evaluation of Medicinal Products Post-authorisation evaluation of medicines for human use London, 12 March 2001 Doc. Ref: EMEA/H/5611/01/en EMEA PUBLIC STATEMENT ON LEFLUNOMIDE
Nursing 113. Pharmacology Principles
 Nursing 113 Pharmacology Principles 1. The study of how drugs enter the body, reach the site of action, and are removed from the body is called a. pharmacotherapeutics b. pharmacology c. pharmacodynamics
Nursing 113 Pharmacology Principles 1. The study of how drugs enter the body, reach the site of action, and are removed from the body is called a. pharmacotherapeutics b. pharmacology c. pharmacodynamics
5.07.09. Aubagio. Aubagio (teriflunomide) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.07.09 Subject: Aubagio Page: 1 of 6 Last Review Date: December 5, 2014 Aubagio Description Aubagio (teriflunomide)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.07.09 Subject: Aubagio Page: 1 of 6 Last Review Date: December 5, 2014 Aubagio Description Aubagio (teriflunomide)
IMPORTANT DRUG WARNING Regarding Mycophenolate-Containing Products
 Dear Healthcare Provider: Mycophenolate REMS (Risk Evaluation and Mitigation Strategy) has been mandated by the FDA (Food and Drug Administration) due to postmarketing reports showing that exposure to
Dear Healthcare Provider: Mycophenolate REMS (Risk Evaluation and Mitigation Strategy) has been mandated by the FDA (Food and Drug Administration) due to postmarketing reports showing that exposure to
ZOVIRAX Cold Sore Cream
 Data Sheet ZOVIRAX Cold Sore Cream Aciclovir 5% w/w Presentation Topical cream Indications ZOVIRAX Cold Sore Cream is indicated for the treatment of Herpes simplex virus infections of the lips and face
Data Sheet ZOVIRAX Cold Sore Cream Aciclovir 5% w/w Presentation Topical cream Indications ZOVIRAX Cold Sore Cream is indicated for the treatment of Herpes simplex virus infections of the lips and face
Selected Requirements of Prescribing Information
 The Selected Requirements of Prescribing Information (SRPI) is a checklist of 42 important format prescribing information (PI) items based on labeling regulations [21 CFR 201.56(d) and 201.57] and guidances.
The Selected Requirements of Prescribing Information (SRPI) is a checklist of 42 important format prescribing information (PI) items based on labeling regulations [21 CFR 201.56(d) and 201.57] and guidances.
FOR ORAL OR RECTAL ADMINISTRATION FOR THE PREVENTION AND TREATMENT OF PORTAL-SYSTEMIC ENCEPHALOPATHY
 FORM NO. 1360 VC3074 Rev.2/08 LACTULOSE SOLUTION, USP 10 g/15 ml FOR ORAL OR RECTAL ADMINISTRATION FOR THE PREVENTION AND TREATMENT OF PORTAL-SYSTEMIC ENCEPHALOPATHY Rx Only DESCRIPTION Lactulose is a
FORM NO. 1360 VC3074 Rev.2/08 LACTULOSE SOLUTION, USP 10 g/15 ml FOR ORAL OR RECTAL ADMINISTRATION FOR THE PREVENTION AND TREATMENT OF PORTAL-SYSTEMIC ENCEPHALOPATHY Rx Only DESCRIPTION Lactulose is a
ENZAR FORTE TABLETS. (derived from Pancreatin USP) Sodium tauroglycocholate BPC 65mg (with sugar coating containing essential carminative oils)
 ENZAR FORTE TABLETS COMPOSITION Each enteric-coated tablet contains: Amylase activity -15,000 USP units Lipase activity -4,000 USP units Protease activity -15,000 USP units of (derived from Pancreatin
ENZAR FORTE TABLETS COMPOSITION Each enteric-coated tablet contains: Amylase activity -15,000 USP units Lipase activity -4,000 USP units Protease activity -15,000 USP units of (derived from Pancreatin
OCALIVA (obeticholic acid) tablets, for oral use Initial U.S. Approval: 2016
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use OCALIVA safely and effectively. See full prescribing information for OCALIVA. OCALIVA (obeticholic
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use OCALIVA safely and effectively. See full prescribing information for OCALIVA. OCALIVA (obeticholic
Reference ID: 3482803
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use EVZIO safely and effectively. See full prescribing information for EVZIO. EVZIO (naloxone hydrochloride
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use EVZIO safely and effectively. See full prescribing information for EVZIO. EVZIO (naloxone hydrochloride
1. NAME OF THE MEDICINAL PRODUCT. Impavido 10 mg capsules Impavido 50 mg capsules 2. QUALITATIVE AND QUANTITATIVE COMPOSITION. 1 capsule contains:
 1. NAME OF THE MEDICINAL PRODUCT Impavido 10 mg capsules Impavido 50 mg capsules 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 capsule contains: Impavido 10 mg capsules 10 mg Miltefosine. Impavido 50 mg
1. NAME OF THE MEDICINAL PRODUCT Impavido 10 mg capsules Impavido 50 mg capsules 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 capsule contains: Impavido 10 mg capsules 10 mg Miltefosine. Impavido 50 mg
ORAL MEDICATIONS FOR MS! Gilenya and Aubagio
 ORAL MEDICATIONS FOR MS! Gilenya and Aubagio Champions against MS 4/20/13 Alexandra Goodyear, MD Stanford University Oral Medications Since 2010, 3 new oral medications for MS: Gilenya 2010 Aubagio 2012
ORAL MEDICATIONS FOR MS! Gilenya and Aubagio Champions against MS 4/20/13 Alexandra Goodyear, MD Stanford University Oral Medications Since 2010, 3 new oral medications for MS: Gilenya 2010 Aubagio 2012
Learn More About Product Labeling
 Learn More About Product Labeling Product label The product label is developed during the formal process of review and approval by regulatory agencies of any medicine or medical product. There are specific
Learn More About Product Labeling Product label The product label is developed during the formal process of review and approval by regulatory agencies of any medicine or medical product. There are specific
PHOSPHATE-SANDOZ Tablets (High dose phosphate supplement)
 1 PHOSPHATE-SANDOZ Tablets (High dose phosphate supplement) PHOSPHATE-SANDOZ PHOSPHATE-SANDOZ Tablets are a high dose phosphate supplement containing sodium phosphate monobasic. The CAS registry number
1 PHOSPHATE-SANDOZ Tablets (High dose phosphate supplement) PHOSPHATE-SANDOZ PHOSPHATE-SANDOZ Tablets are a high dose phosphate supplement containing sodium phosphate monobasic. The CAS registry number
PRESCRIBING INFORMATION Refer to Summary of Product Characteristics (SmPC) before Prescribing
 EZETROL (ezetimibe) PRESCRIBING INFORMATION Refer to Summary of Product Characteristics (SmPC) before Prescribing Adverse events should be reported. Reporting forms and information can be found at www.mhra.gov.uk/yellowcard.
EZETROL (ezetimibe) PRESCRIBING INFORMATION Refer to Summary of Product Characteristics (SmPC) before Prescribing Adverse events should be reported. Reporting forms and information can be found at www.mhra.gov.uk/yellowcard.
Revised: 9/2014. LUMIGAN (bimatoprost ophthalmic solution) 0.01% for topical ophthalmic use. Initial U.S. Approval: 2001
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use LUMIGAN (bimatoprost ophthalmic solution) 0.01% safely and effectively. See full prescribing information
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use LUMIGAN (bimatoprost ophthalmic solution) 0.01% safely and effectively. See full prescribing information
ESBRIET (pirfenidone) capsules, for oral use Initial U.S. Approval: 2014
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use ESBRIET safely and effectively. See full prescribing information for ESBRIET. ESBRIET (pirfenidone)
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use ESBRIET safely and effectively. See full prescribing information for ESBRIET. ESBRIET (pirfenidone)
PARACETAMOL REXIDOL. 600 mg Tablet. Analgesic-Antipyretic. Paracetamol 600 mg
 (Insert Text) UL Consumer Health PARACETAMOL REXIDOL 600 mg Tablet Analgesic-Antipyretic FORMULATION Each tablet contains: Paracetamol 600 mg PRODUCT DESCRIPTION Rexidol is a round, yellow, flat, bevel-edged
(Insert Text) UL Consumer Health PARACETAMOL REXIDOL 600 mg Tablet Analgesic-Antipyretic FORMULATION Each tablet contains: Paracetamol 600 mg PRODUCT DESCRIPTION Rexidol is a round, yellow, flat, bevel-edged
Who are Levonorgestrel Tablets, 0.75mg for? Other things your patients should know: When are Levonorgestrel Tablets, 0.75mg effective?
 One-hour ACPE accredited continuing education program is available at no charge for pharmacists provided by the University of Illinois-Chicago College of Pharmacy at www.pharmacyce.uic.edu Accidents happen...
One-hour ACPE accredited continuing education program is available at no charge for pharmacists provided by the University of Illinois-Chicago College of Pharmacy at www.pharmacyce.uic.edu Accidents happen...
MATERIAL SAFETY DATA SHEET
 Page 1 of 7 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND THE COMPANY/UNDERTAKING Pfizer Inc Pfizer Pharmaceuticals Group 235 East 42nd Street New York, New York 10017 1-212-573-2222 Emergency telephone
Page 1 of 7 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND THE COMPANY/UNDERTAKING Pfizer Inc Pfizer Pharmaceuticals Group 235 East 42nd Street New York, New York 10017 1-212-573-2222 Emergency telephone
* Sections or subsections omitted from the full prescribing information are not listed
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use RECLAST safely and effectively. See full prescribing information for RECLAST. Reclast (zoledronic
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use RECLAST safely and effectively. See full prescribing information for RECLAST. Reclast (zoledronic
A guide to the accelerated elimination procedure
 A guide to the accelerated elimination procedure Prescribing information and information on adverse event reporting can be found on the last page. What is the accelerated elimination procedure? An accelerated
A guide to the accelerated elimination procedure Prescribing information and information on adverse event reporting can be found on the last page. What is the accelerated elimination procedure? An accelerated
Phase: IV. Study Period: 20 Jan. 2006-17 Sep. 2008
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
CLINICAL PHARMACOLOGY
 Emcyt estramustine phosphate sodium capsules DESCRIPTION Estramustine phosphate sodium, an antineoplastic agent, is an off-white powder readily soluble in water. EMCYT Capsules are white and opaque, each
Emcyt estramustine phosphate sodium capsules DESCRIPTION Estramustine phosphate sodium, an antineoplastic agent, is an off-white powder readily soluble in water. EMCYT Capsules are white and opaque, each
Absorption of Drugs. Transport of a drug from the GI tract
 Absorption of Drugs Absorption is the transfer of a drug from its site of administration to the bloodstream. The rate and efficiency of absorption depend on the route of administration. For IV delivery,
Absorption of Drugs Absorption is the transfer of a drug from its site of administration to the bloodstream. The rate and efficiency of absorption depend on the route of administration. For IV delivery,
LEFLUNOMIDE (Adults)
 Shared Care Guideline DRUG: Introduction: LEFLUNOMIDE (Adults) Indication: Disease modifying drug for rheumatoid arthritis and psoriatic arthritis Licensing Information: Disease modifying drug for active
Shared Care Guideline DRUG: Introduction: LEFLUNOMIDE (Adults) Indication: Disease modifying drug for rheumatoid arthritis and psoriatic arthritis Licensing Information: Disease modifying drug for active
Guidance for Industry Safety Testing of Drug Metabolites
 Guidance for Industry Safety Testing of Drug Metabolites U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research (CDER) February 2008 Pharmacology
Guidance for Industry Safety Testing of Drug Metabolites U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research (CDER) February 2008 Pharmacology
An Introduction to the Improved FDA Prescription Drug Labeling
 An Introduction to the Improved FDA Prescription Drug Labeling 1 Introduction Mary E. Kremzner, Pharm.D. CDR, U.S. Public Health Service Deputy Director, Division of Drug Information Center for Drug Evaluation
An Introduction to the Improved FDA Prescription Drug Labeling 1 Introduction Mary E. Kremzner, Pharm.D. CDR, U.S. Public Health Service Deputy Director, Division of Drug Information Center for Drug Evaluation
8.5 Geriatric Use * Sections or subsections omitted from the full prescribing information are not listed.
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use Injectafer safely and effectively. See full prescribing information for Injectafer. INJECTAFER (ferric
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use Injectafer safely and effectively. See full prescribing information for Injectafer. INJECTAFER (ferric
FRENCH AGENCY FOR VETERINARY MEDICINAL PRODUCTS DECENTRALISED PROCEDURE PUBLICLY AVAILABLE ASSESSMENT REPORT FOR A VETERINARY MEDICINAL PRODUCT
 FRENCH AGENCY FOR VETERINARY MEDICINAL PRODUCTS DECENTRALISED PROCEDURE FOR A VETERINARY MEDICINAL PRODUCT EMEDOG, 1 mg/ml, solution for injection for dogs Date: 20/08/2015 French agency for food, environnemental
FRENCH AGENCY FOR VETERINARY MEDICINAL PRODUCTS DECENTRALISED PROCEDURE FOR A VETERINARY MEDICINAL PRODUCT EMEDOG, 1 mg/ml, solution for injection for dogs Date: 20/08/2015 French agency for food, environnemental
Sponsor Novartis. Generic Drug Name Secukinumab. Therapeutic Area of Trial Psoriasis. Approved Indication investigational
 Clinical Trial Results Database Page 2 Sponsor Novartis Generic Drug Name Secukinumab Therapeutic Area of Trial Psoriasis Approved Indication investigational Clinical Trial Results Database Page 3 Study
Clinical Trial Results Database Page 2 Sponsor Novartis Generic Drug Name Secukinumab Therapeutic Area of Trial Psoriasis Approved Indication investigational Clinical Trial Results Database Page 3 Study
PLEGRIDY (peginterferon beta-1a) injection, for subcutaneous injection Initial U.S. Approval: 2014
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use PLEGRIDY safely and effectively. See full prescribing information for PLEGRIDY. PLEGRIDY (peginterferon
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use PLEGRIDY safely and effectively. See full prescribing information for PLEGRIDY. PLEGRIDY (peginterferon
MEDICATION GUIDE JUXTAPID (JUKS-tuh-pid) (lomitapide) capsules
 MEDICATION GUIDE JUXTAPID (JUKS-tuh-pid) (lomitapide) capsules Read this Medication Guide before your treatment. What is the most important information I should know about JUXTAPID? JUXTAPID is available
MEDICATION GUIDE JUXTAPID (JUKS-tuh-pid) (lomitapide) capsules Read this Medication Guide before your treatment. What is the most important information I should know about JUXTAPID? JUXTAPID is available
Medication for the Treatment of Alcohol Use Disorder. Pocket Guide
 Medication for the Treatment of Alcohol Use Disorder Pocket Guide Medications are underused in the treatment of alcohol use disorder. According to the National Survey on Drug Use and Health, of the estimated
Medication for the Treatment of Alcohol Use Disorder Pocket Guide Medications are underused in the treatment of alcohol use disorder. According to the National Survey on Drug Use and Health, of the estimated
Summary of the risk management plan (RMP) for Orkambi (lumacaftor and ivacaftor)
 EMA/662624/2015 Summary of the risk management plan (RMP) for Orkambi (lumacaftor and ivacaftor) This is a summary of the risk management plan (RMP) for Orkambi, which details the measures to be taken
EMA/662624/2015 Summary of the risk management plan (RMP) for Orkambi (lumacaftor and ivacaftor) This is a summary of the risk management plan (RMP) for Orkambi, which details the measures to be taken
Attached from the following page is the press release made by BMS for your information.
 July 22, 2015 CheckMate -025 (global clinical trial), a Pivotal Phase III Opdivo (nivolumab) Renal Cancer Trial Stopped Early (PRINCETON, NJ, July 20, 2015) Bristol-Myers Squibb Company (NYSE:BMY) announced
July 22, 2015 CheckMate -025 (global clinical trial), a Pivotal Phase III Opdivo (nivolumab) Renal Cancer Trial Stopped Early (PRINCETON, NJ, July 20, 2015) Bristol-Myers Squibb Company (NYSE:BMY) announced
EFFIMET 1000 XR Metformin Hydrochloride extended release tablet
 BRAND NAME: Effimet XR. THERAPEUTIC CATEGORY: Anti-Diabetic PHARMACOLOGIC CLASS: Biguanides EFFIMET 1000 XR Metformin Hydrochloride extended release tablet COMPOSITION AND PRESENTATION Composition Each
BRAND NAME: Effimet XR. THERAPEUTIC CATEGORY: Anti-Diabetic PHARMACOLOGIC CLASS: Biguanides EFFIMET 1000 XR Metformin Hydrochloride extended release tablet COMPOSITION AND PRESENTATION Composition Each
Billing and Coding Guide
 Billing and Coding Guide INDICATIONS Injectafer is an iron replacement product indicated for the treatment of iron deficiency anemia (IDA) in adult patients: who have intolerance to or have had unsatisfactory
Billing and Coding Guide INDICATIONS Injectafer is an iron replacement product indicated for the treatment of iron deficiency anemia (IDA) in adult patients: who have intolerance to or have had unsatisfactory
CheckMate -057, a Pivotal III Opdivo (nivolumab) Lung Cancer Trial, Stopped Early
 April 21, 2015 CheckMate -057, a Pivotal III Opdivo (nivolumab) Lung Cancer Trial, Stopped Early Opdivo Demonstrates Superior Overall Survival Compared to Docetaxel in Patients with Previously-Treated
April 21, 2015 CheckMate -057, a Pivotal III Opdivo (nivolumab) Lung Cancer Trial, Stopped Early Opdivo Demonstrates Superior Overall Survival Compared to Docetaxel in Patients with Previously-Treated
MATERIAL SAFETY DATA SHEET
 Page 1 of 7 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND THE COMPANY/UNDERTAKING Pfizer Inc Pfizer Pharmaceuticals Group 235 East 42nd Street New York, New York 10017 1-212-573-2222 Emergency telephone
Page 1 of 7 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND THE COMPANY/UNDERTAKING Pfizer Inc Pfizer Pharmaceuticals Group 235 East 42nd Street New York, New York 10017 1-212-573-2222 Emergency telephone
SR 5. W2364A_NT.XATRAL SR5 PAK/SA 5/12/05 9:47 Page 1
 W2364A_NT.XATRAL SR5 PAK/SA 5/12/05 9:47 Page 1 alfuzosin SR 5 mg (Alfuzosin HCI) COMPOSITION Each sustained-release, film-coated tablet contains: Alfuzosin hydrochloride......... 5 mg Excipients... q.s.
W2364A_NT.XATRAL SR5 PAK/SA 5/12/05 9:47 Page 1 alfuzosin SR 5 mg (Alfuzosin HCI) COMPOSITION Each sustained-release, film-coated tablet contains: Alfuzosin hydrochloride......... 5 mg Excipients... q.s.
NONCLINICAL EVALUATION FOR ANTICANCER PHARMACEUTICALS
 INTERNATIONAL CONFERENCE ON HARMONISATION OF TECHNICAL REQUIREMENTS FOR REGISTRATION OF PHARMACEUTICALS FOR HUMAN USE ICH HARMONISED TRIPARTITE GUIDELINE NONCLINICAL EVALUATION FOR ANTICANCER PHARMACEUTICALS
INTERNATIONAL CONFERENCE ON HARMONISATION OF TECHNICAL REQUIREMENTS FOR REGISTRATION OF PHARMACEUTICALS FOR HUMAN USE ICH HARMONISED TRIPARTITE GUIDELINE NONCLINICAL EVALUATION FOR ANTICANCER PHARMACEUTICALS
Special pediatric considerations are noted when applicable, otherwise adult provisions apply.
 DRUG NAME: Eribulin SYNONYM(S): Eribulin mesylate COMMON TRADE NAME(S): HALAVEN CLASSIFICATION: antimicrotubule agent Special pediatric considerations are noted when applicable, otherwise adult provisions
DRUG NAME: Eribulin SYNONYM(S): Eribulin mesylate COMMON TRADE NAME(S): HALAVEN CLASSIFICATION: antimicrotubule agent Special pediatric considerations are noted when applicable, otherwise adult provisions
National MS Society Information Sourcebook www.nationalmssociety.org/sourcebook
 National MS Society Information Sourcebook www.nationalmssociety.org/sourcebook Chemotherapy The literal meaning of the term chemotherapy is to treat with a chemical agent, but the term generally refers
National MS Society Information Sourcebook www.nationalmssociety.org/sourcebook Chemotherapy The literal meaning of the term chemotherapy is to treat with a chemical agent, but the term generally refers
LCD for Viral Hepatitis Serology Tests
 LCD for Viral Hepatitis Serology Tests Applicable CPT Code(s): 86692 Antibody; Hepatitis, Delta Agent 86704 Hepatitis B Core Antibody (HBcAb); Total 86705 Hepatitis B Core Antibody (HBcAb); IgM Antibody
LCD for Viral Hepatitis Serology Tests Applicable CPT Code(s): 86692 Antibody; Hepatitis, Delta Agent 86704 Hepatitis B Core Antibody (HBcAb); Total 86705 Hepatitis B Core Antibody (HBcAb); IgM Antibody
NIZORAL (KETOCONAZOLE) 2% SHAMPOO DESCRIPTION
 NIZORAL (KETOCONAZOLE) 2% SHAMPOO DESCRIPTION NIZORAL (ketoconazole) 2% Shampoo is a red-orange liquid for topical application, containing the broad spectrum synthetic antifungal agent ketoconazole in
NIZORAL (KETOCONAZOLE) 2% SHAMPOO DESCRIPTION NIZORAL (ketoconazole) 2% Shampoo is a red-orange liquid for topical application, containing the broad spectrum synthetic antifungal agent ketoconazole in
MANAGEMENT OF COMMON SIDE EFFECTS of INH (Isoniazid), RIF (Rifampin), PZA (Pyrazinamide), and EMB (Ethambutol)
 MANAGEMENT OF COMMON SIDE EFFECTS of INH (Isoniazid), RIF (Rifampin), PZA (Pyrazinamide), and EMB (Ethambutol) 1. Hepatotoxicity: In Active TB Disease a. Background: 1. Among the 4 standard anti-tb drugs,
MANAGEMENT OF COMMON SIDE EFFECTS of INH (Isoniazid), RIF (Rifampin), PZA (Pyrazinamide), and EMB (Ethambutol) 1. Hepatotoxicity: In Active TB Disease a. Background: 1. Among the 4 standard anti-tb drugs,
Adams Memorial Hospital Decatur, Indiana EXPLANATION OF LABORATORY TESTS
 Adams Memorial Hospital Decatur, Indiana EXPLANATION OF LABORATORY TESTS Your health is important to us! The test descriptions listed below are for educational purposes only. Laboratory test interpretation
Adams Memorial Hospital Decatur, Indiana EXPLANATION OF LABORATORY TESTS Your health is important to us! The test descriptions listed below are for educational purposes only. Laboratory test interpretation
DRUG NAME: Abiraterone
 DRUG NAME: Abiraterone SYNONYM(S): abiraterone acetate 1 COMMON TRADE NAME(S): ZYTIGA CLASSIFICATION: endocrine anti-hormone Special pediatric considerations are noted when applicable, otherwise adult
DRUG NAME: Abiraterone SYNONYM(S): abiraterone acetate 1 COMMON TRADE NAME(S): ZYTIGA CLASSIFICATION: endocrine anti-hormone Special pediatric considerations are noted when applicable, otherwise adult
160S01105, Page 1 of 7. Human Hepatitis B Immunoglobulin, solution for intramuscular injection.
 160S01105, Page 1 of 7 New Zealand Data Sheet Hepatitis B Immunoglobulin-VF NAME OF THE MEDICINE Human Hepatitis B Immunoglobulin, solution for intramuscular injection. DESCRIPTION Hepatitis B Immunoglobulin-VF
160S01105, Page 1 of 7 New Zealand Data Sheet Hepatitis B Immunoglobulin-VF NAME OF THE MEDICINE Human Hepatitis B Immunoglobulin, solution for intramuscular injection. DESCRIPTION Hepatitis B Immunoglobulin-VF
Adjunctive psychosocial intervention. Conditions requiring dose reduction. Immediate, peak plasma concentration is reached within 1 hour.
 Shared Care Guideline for Prescription and monitoring of Naltrexone Hydrochloride in alcohol dependence Author(s)/Originator(s): (please state author name and department) Dr Daly - Consultant Psychiatrist,
Shared Care Guideline for Prescription and monitoring of Naltrexone Hydrochloride in alcohol dependence Author(s)/Originator(s): (please state author name and department) Dr Daly - Consultant Psychiatrist,
Bile Duct Diseases and Problems
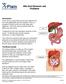 Bile Duct Diseases and Problems Introduction A bile duct is a tube that carries bile between the liver and gallbladder and the intestine. Bile is a substance made by the liver that helps with digestion.
Bile Duct Diseases and Problems Introduction A bile duct is a tube that carries bile between the liver and gallbladder and the intestine. Bile is a substance made by the liver that helps with digestion.
Acamprosate calcium APOLLO 91991 46 950 950
 Acamprosate calcium APOLLO 91991 46 950 950 Acamprosate calcium APOLLO 91991 46 950 950 Acamprosate calcium CAS Number : 77337-73-6 Molecular Weight : 400.48 g/mol Molecular Formula : 2C5H10NO4S.Ca Systematic
Acamprosate calcium APOLLO 91991 46 950 950 Acamprosate calcium APOLLO 91991 46 950 950 Acamprosate calcium CAS Number : 77337-73-6 Molecular Weight : 400.48 g/mol Molecular Formula : 2C5H10NO4S.Ca Systematic
Section 1: Identification Product information Product Name Active substance Intended Uses
 Section 1: Identification Product information Product Name Active substance Intended Uses Company Details Manufacturer SAFETY DATA SHEET Levetiracetam Tablets, 250 mg, 500 mg, 750 mg & 1000 mg Levetiracetam
Section 1: Identification Product information Product Name Active substance Intended Uses Company Details Manufacturer SAFETY DATA SHEET Levetiracetam Tablets, 250 mg, 500 mg, 750 mg & 1000 mg Levetiracetam
Wobenzym. Wobenzym Keeps you moving.
 Wobenzym PS Helps to maintain healthy joints, mobility and flexibility Relief from everyday aches, pains and muscle soreness Speeds recovery following exertion Supported by over 200 clinical trials Wobenzym
Wobenzym PS Helps to maintain healthy joints, mobility and flexibility Relief from everyday aches, pains and muscle soreness Speeds recovery following exertion Supported by over 200 clinical trials Wobenzym
(NON-PRESCRIPTION) LEAFLET: USER INFORMATION. CALTRATE VITAMIN D3 600mg/400 IU, film-coated tablet Calcium and Cholecalciferol
 (NON-PRESCRIPTION) LEAFLET: USER INFORMATION CALTRATE VITAMIN D3 600mg/400 IU, film-coated tablet Calcium and Cholecalciferol Read all of this leaflet carefully because it contains important information
(NON-PRESCRIPTION) LEAFLET: USER INFORMATION CALTRATE VITAMIN D3 600mg/400 IU, film-coated tablet Calcium and Cholecalciferol Read all of this leaflet carefully because it contains important information
VESIcare (solifenacin succinate) tablets Initial U.S. Approval: 2004
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use safely and effectively. See full prescribing information for. (solifenacin succinate) tablets Initial
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use safely and effectively. See full prescribing information for. (solifenacin succinate) tablets Initial
Prior Authorization Guideline
 Prior Authorization Guideline Guideline: CSD - Suboxone Therapeutic Class: Central Nervous System Agents Therapeutic Sub-Class: Analgesics and Antipyretics (Opiate Partial Agonists) Client: County of San
Prior Authorization Guideline Guideline: CSD - Suboxone Therapeutic Class: Central Nervous System Agents Therapeutic Sub-Class: Analgesics and Antipyretics (Opiate Partial Agonists) Client: County of San
Fexinidazole a new oral treatment for sleeping sickness update of development
 Fexinidazole a new oral treatment for sleeping sickness update of development SMe O 2 Me CH 2 O Antoine TARRAL Olaf Valverde Séverine Blesson Clélia Bardonneau Wilfried Mutumbo September 2011 Fexinidazole
Fexinidazole a new oral treatment for sleeping sickness update of development SMe O 2 Me CH 2 O Antoine TARRAL Olaf Valverde Séverine Blesson Clélia Bardonneau Wilfried Mutumbo September 2011 Fexinidazole
RISK EVALUATION AND MITIGATION STRATEGY (REMS)
 Initial REMS Approval: 9/2010 Most Recent Modification: 05/2015 0.5mg capsules Sphingosine 1-phosphate Receptor Modulator Novartis Pharmaceuticals Corporation One Health Plaza East Hanover, NJ 07936 RISK
Initial REMS Approval: 9/2010 Most Recent Modification: 05/2015 0.5mg capsules Sphingosine 1-phosphate Receptor Modulator Novartis Pharmaceuticals Corporation One Health Plaza East Hanover, NJ 07936 RISK
Clinical Trial Results Database Page 1
 Clinical Trial Results Database Page Sponsor Novartis Generic Drug Name BGT6 Therapeutic Area of Trial Advanced solid malignancies Approved Indication Investigational Study Number CBGT6A0 Title A phase
Clinical Trial Results Database Page Sponsor Novartis Generic Drug Name BGT6 Therapeutic Area of Trial Advanced solid malignancies Approved Indication Investigational Study Number CBGT6A0 Title A phase
1) Aug 2005 In Vitro Chromosomal Aberration Study Cytotoxicity (File: TS39) Conclusion No cytotoxicity can be detected at concentrations up to 100%.
 Endotoxicity and Cytotoxicity Studies 1) Aug 2005 In Vitro Chromosomal Aberration Study Cytotoxicity (File: TS39) Chinese Hamster Ovary (CHO) cells were grown in monolayer with and without metabolic activation.
Endotoxicity and Cytotoxicity Studies 1) Aug 2005 In Vitro Chromosomal Aberration Study Cytotoxicity (File: TS39) Chinese Hamster Ovary (CHO) cells were grown in monolayer with and without metabolic activation.
Oral Zinc Supplementation as an Adjunct Therapy in the Management of Hepatic Encephalopathy: A Randomized Controlled Trial
 Oral Zinc Supplementation as an Adjunct Therapy in the Management of Hepatic Encephalopathy: A Randomized Controlled Trial Marcus R. Pereira A. Study Purpose Hepatic encephalopathy is a common complication
Oral Zinc Supplementation as an Adjunct Therapy in the Management of Hepatic Encephalopathy: A Randomized Controlled Trial Marcus R. Pereira A. Study Purpose Hepatic encephalopathy is a common complication
Activity of pemetrexed in thoracic malignancies
 Activity of pemetrexed in thoracic malignancies Results of phase III clinical studies of pemetrexed in malignant pleural mesothelioma and non-small cell lung cancer show benefit P emetrexed (Alimta) is
Activity of pemetrexed in thoracic malignancies Results of phase III clinical studies of pemetrexed in malignant pleural mesothelioma and non-small cell lung cancer show benefit P emetrexed (Alimta) is
Lung Pathway Group Pemetrexed and Cisplatin in Non-Small Cell Lung Cancer (NSCLC)
 Indication: NICE TA181 First line treatment option in advanced or metastatic non-squamous NSCLC (histology confirmed as adenocarcinoma or large cell carcinoma) Performance status 0-1 Regimen details: Pemetrexed
Indication: NICE TA181 First line treatment option in advanced or metastatic non-squamous NSCLC (histology confirmed as adenocarcinoma or large cell carcinoma) Performance status 0-1 Regimen details: Pemetrexed
Teriflunomide (Aubagio) 14mg once daily tablet
 Teriflunomide (Aubagio) 14mg once daily tablet Exceptional healthcare, personally delivered Your Consultant Neurologist has suggested that you may benefit from treatment with Teriflunomide. The decision
Teriflunomide (Aubagio) 14mg once daily tablet Exceptional healthcare, personally delivered Your Consultant Neurologist has suggested that you may benefit from treatment with Teriflunomide. The decision
MATERIAL SAFETY DATA SHEET
 Page 1 of 6 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND THE COMPANY/UNDERTAKING Pfizer Inc Pfizer Pharmaceuticals Group 235 East 42nd Street New York, New York 10017 1-212-573-2222 Emergency telephone
Page 1 of 6 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND THE COMPANY/UNDERTAKING Pfizer Inc Pfizer Pharmaceuticals Group 235 East 42nd Street New York, New York 10017 1-212-573-2222 Emergency telephone
MEDICATION GUIDE ACTOPLUS MET (ak-tō-plus-met) (pioglitazone hydrochloride and metformin hydrochloride) tablets
 MEDICATION GUIDE (ak-tō-plus-met) (pioglitazone hydrochloride and metformin hydrochloride) tablets Read this Medication Guide carefully before you start taking and each time you get a refill. There may
MEDICATION GUIDE (ak-tō-plus-met) (pioglitazone hydrochloride and metformin hydrochloride) tablets Read this Medication Guide carefully before you start taking and each time you get a refill. There may
VITAMIN C AND INFECTIOUS DISEASE: A REVIEW OF THE LITERATURE AND THE RESULTS OF A RANDOMIZED, DOUBLE-BLIND, PROSPECTIVE STUDY OVER 8 YEARS
 39 Chapter 3 VITAMIN C AND INFECTIOUS DISEASE: A REVIEW OF THE LITERATURE AND THE RESULTS OF A RANDOMIZED, DOUBLE-BLIND, PROSPECTIVE STUDY OVER 8 YEARS Maxine Briggs TABLE OF CONTENTS I. Review of the
39 Chapter 3 VITAMIN C AND INFECTIOUS DISEASE: A REVIEW OF THE LITERATURE AND THE RESULTS OF A RANDOMIZED, DOUBLE-BLIND, PROSPECTIVE STUDY OVER 8 YEARS Maxine Briggs TABLE OF CONTENTS I. Review of the
Lung Pathway Group Nintedanib (Vargatef) in advanced Non-Small Cell Lung Cancer (NSCLC)
 Lung Pathway Group Nintedanib (Vargatef) in advanced Non-Small Cell Lung Cancer (NSCLC) Indication: In combination with docetaxel in locally advanced, metastatic or locally recurrent NSCLC of adenocarcinoma
Lung Pathway Group Nintedanib (Vargatef) in advanced Non-Small Cell Lung Cancer (NSCLC) Indication: In combination with docetaxel in locally advanced, metastatic or locally recurrent NSCLC of adenocarcinoma
嘉 義 長 庚 醫 院 藥 劑 科 Speaker : 翁 玟 雯
 The Clinical Efficacy and Safety of Sodium Glucose Cotransporter-2 (SGLT2) Inhibitors in Adults with Type 2 Diabetes Mellitus 嘉 義 長 庚 醫 院 藥 劑 科 Speaker : 翁 玟 雯 Diabetes Mellitus : A group of diseases characterized
The Clinical Efficacy and Safety of Sodium Glucose Cotransporter-2 (SGLT2) Inhibitors in Adults with Type 2 Diabetes Mellitus 嘉 義 長 庚 醫 院 藥 劑 科 Speaker : 翁 玟 雯 Diabetes Mellitus : A group of diseases characterized
LAMICTAL XR (lamotrigine) extended-release tablets, for oral use Initial U.S. Approval: 1994
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use LAMICTAL XR safely and effectively. See full prescribing information for LAMICTAL XR. LAMICTAL XR
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use LAMICTAL XR safely and effectively. See full prescribing information for LAMICTAL XR. LAMICTAL XR
PACKAGE LEAFLET: INFORMATION FOR THE USER Paracetamol 500 mg Effervescent Tablets Paracetamol
 PACKAGE LEAFLET: INFORMATION FOR THE USER Paracetamol 500 mg Effervescent Tablets Paracetamol Read all of this leaflet carefully because it contains important information for you. This medicine is available
PACKAGE LEAFLET: INFORMATION FOR THE USER Paracetamol 500 mg Effervescent Tablets Paracetamol Read all of this leaflet carefully because it contains important information for you. This medicine is available
See 17 for PATIENT COUNSELING INFORMATION and FDAapproved. Revised: 4/2015
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DUAC Gel safely and effectively. See full prescribing information for DUAC Gel. DUAC (clindamycin
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DUAC Gel safely and effectively. See full prescribing information for DUAC Gel. DUAC (clindamycin
Drug Coding and CPT (Current Procedural Terminology) Guidance
 Aridol (mannitol inhalation powder) Bronchial Challenge Test Kit NDC 44178-0552-1 Drug Coding and CPT (Current Procedural Terminology) Guidance Product Reimbursement Code: HCPCS Code 1 Description 1 J7665
Aridol (mannitol inhalation powder) Bronchial Challenge Test Kit NDC 44178-0552-1 Drug Coding and CPT (Current Procedural Terminology) Guidance Product Reimbursement Code: HCPCS Code 1 Description 1 J7665
See 17 for PATIENT COUNSELING INFORMATION (intraventricular or lumbar puncture) every 28 days for 4 doses
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DEPOCYT safely and effectively. See full prescribing information for DEPOCYT. DEPOCYT (cytarabine
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DEPOCYT safely and effectively. See full prescribing information for DEPOCYT. DEPOCYT (cytarabine
PRODUCT MONOGRAPH. Pr XENICAL. orlistat. Capsules 120 mg. Pharmaceutical Standard: Professed. Anti-Obesity Agent/Gastrointestinal Lipase Inhibitor
 PRODUCT MONOGRAPH Pr XENICAL orlistat Capsules 120 mg Pharmaceutical Standard: Professed Anti-Obesity Agent/Gastrointestinal Lipase Inhibitor Hoffmann-La Roche Limited Date of Revision: 7070 Mississauga
PRODUCT MONOGRAPH Pr XENICAL orlistat Capsules 120 mg Pharmaceutical Standard: Professed Anti-Obesity Agent/Gastrointestinal Lipase Inhibitor Hoffmann-La Roche Limited Date of Revision: 7070 Mississauga
Drug Excretion. Renal Drug Clearance. Drug Clearance and Half-Life. Glomerular Filtration II. Glomerular Filtration I. Drug Excretion and Clearance
 t/.drugexcretion AINTRAVENOUSDOSE 36848765430TIME(hours) t/ Drug Excretion Dr. Robert G. Lamb Professor Pharmacology & Toxicology Drug Excretion and Clearance Drug Excretion: is the movement of drug from
t/.drugexcretion AINTRAVENOUSDOSE 36848765430TIME(hours) t/ Drug Excretion Dr. Robert G. Lamb Professor Pharmacology & Toxicology Drug Excretion and Clearance Drug Excretion: is the movement of drug from
Nurse Aide Training Program Application Checklist
 Nurse Aide Training Program Application Checklist The following checklist must be completed before enrolling in the Nurse Aide Training course: Complete, sign, and date the Application Form Have the physical
Nurse Aide Training Program Application Checklist The following checklist must be completed before enrolling in the Nurse Aide Training course: Complete, sign, and date the Application Form Have the physical
MATERIAL SAFETY DATA SHEET
 Page 1 of 8 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND THE COMPANY/UNDERTAKING Pfizer Inc Pfizer Pharmaceuticals Group 235 East 42nd Street New York, New York 10017 1-212-573-2222 Emergency telephone
Page 1 of 8 1. IDENTIFICATION OF THE SUBSTANCE/PREPARATION AND THE COMPANY/UNDERTAKING Pfizer Inc Pfizer Pharmaceuticals Group 235 East 42nd Street New York, New York 10017 1-212-573-2222 Emergency telephone
FULL PRESCRIBING INFORMATION: CONTENTS*
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use PHOTREXA VISCOUS and PHOTREXA safely and effectively. See full prescribing information for PHOTREXA
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use PHOTREXA VISCOUS and PHOTREXA safely and effectively. See full prescribing information for PHOTREXA
SUMMARY OF PRODUCT CHARACTERISTICS. Paracetamol...10.00 mg for 1 ml of solution for infusion
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT PARACETAMOL MACOPHARMA 10 mg/ml, solution for infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Paracetamol...10.00 mg for 1 ml of
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT PARACETAMOL MACOPHARMA 10 mg/ml, solution for infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Paracetamol...10.00 mg for 1 ml of
Doxylamine succinate belongs to the ethanolamine class of antihistamines with sedative properties.
 Data Sheet MERSYNDOL Tablet Paracetamol 450mg per tablet Codeine Phosphate 9.75mg per tablet Doxylamine Succinate 5mg per tablet MERSYNDOL FORTE Tablet Paracetamol 450mg per tablet Codeine Phosphate 30mg
Data Sheet MERSYNDOL Tablet Paracetamol 450mg per tablet Codeine Phosphate 9.75mg per tablet Doxylamine Succinate 5mg per tablet MERSYNDOL FORTE Tablet Paracetamol 450mg per tablet Codeine Phosphate 30mg
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Progit 50 mg film-coated tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each film-coated tablet contains 50 mg of itopride hydrochloride.
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Progit 50 mg film-coated tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each film-coated tablet contains 50 mg of itopride hydrochloride.
Medication Guide Plavix (PLAV-iks) (clopidogrel bisulfate) tablets
 Medication Guide Plavix (PLAV-iks) (clopidogrel bisulfate) tablets Read this Medication Guide before you start taking Plavix and each time you get a refill. There may be new information. This Medication
Medication Guide Plavix (PLAV-iks) (clopidogrel bisulfate) tablets Read this Medication Guide before you start taking Plavix and each time you get a refill. There may be new information. This Medication
Guidance for Industry
 Guidance for Industry S9 Nonclinical Evaluation for Anticancer Pharmaceuticals U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research (CDER) Center
Guidance for Industry S9 Nonclinical Evaluation for Anticancer Pharmaceuticals U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research (CDER) Center
DIFICID (fidaxomicin) tablets, for oral use Initial U.S. Approval: 2011
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DIFICID safely and effectively. See full prescribing information for DIFICID. DIFICID (fidaxomicin)
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DIFICID safely and effectively. See full prescribing information for DIFICID. DIFICID (fidaxomicin)
Albumin. Prothrombin time. Total protein
 Hepatitis C Fact Sheet February 2016 www.hepatitis.va.gov Laboratory Tests and Hepatitis If you have hepatitis C, your doctor will use laboratory tests to about learn more about your individual hepatitis
Hepatitis C Fact Sheet February 2016 www.hepatitis.va.gov Laboratory Tests and Hepatitis If you have hepatitis C, your doctor will use laboratory tests to about learn more about your individual hepatitis
------------------------------ CONTRAINDICATIONS ------------------------------ Severe hepatic impairment (Child Pugh Class C).
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use ORENITRAM safely and effectively. See Full Prescribing Information for ORENITRAM. ORENITRAM (treprostinil)
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use ORENITRAM safely and effectively. See Full Prescribing Information for ORENITRAM. ORENITRAM (treprostinil)
DRUG NAME: Quinagolide
 DRUG NAME: Quinagolide SYNONYM(S): CV 205 502 1 COMMON TRADE NAME(S): NORPROLAC CLASSIFICATION: hormonal agent Special pediatric considerations are noted when applicable, otherwise adult provisions apply.
DRUG NAME: Quinagolide SYNONYM(S): CV 205 502 1 COMMON TRADE NAME(S): NORPROLAC CLASSIFICATION: hormonal agent Special pediatric considerations are noted when applicable, otherwise adult provisions apply.
Novartis Gilenya FDO Program Clinical Protocol and Highlights from Prescribing Information (PI)
 Novartis Gilenya FDO Program Clinical Protocol and Highlights from Prescribing Information (PI) Highlights from Prescribing Information - the link to the full text PI is as follows: http://www.pharma.us.novartis.com/product/pi/pdf/gilenya.pdf
Novartis Gilenya FDO Program Clinical Protocol and Highlights from Prescribing Information (PI) Highlights from Prescribing Information - the link to the full text PI is as follows: http://www.pharma.us.novartis.com/product/pi/pdf/gilenya.pdf
