2) Remove lemma using fingers (the palea adheres too tightly to be removed without damage).
|
|
|
- Nickolas Morrison
- 7 years ago
- Views:
Transcription
1 Brachypodium transformation Brachypodium transformation protocol Page 1 John Vogel, modified 2/13/2008 Reference for this method: Vogel, J. and Hill, T. (2008) High-efficiency Agrobacterium-mediated transformation of Brachypodium distachyon inbred line Bd21-3, Plant Cell Reports, in press. DOI /s y Note: Modifications to the published method are briefly listed below. Addition of CuSO 4 to the CIM Eliminate the addition of water to the filter paper in petri dishes used for the 3 day cocultivation/dessication step Transfer callus pieces to selective media after the 3 day cocultivation/dessication step. Begin moving callus to regeneration media at after 3 weeks selection rather than after 4. Callus initiation from excised embryos Materials needed: Bleach (5.25% NaOCl) 10% Triton X-100 stock solution 22.5 mm (5 mg/ml) 2,4-D stock solution (2000 X stock) dissolve 50 mg 2,4-D in 10 ml 95% ethanol 0.6 mg/ml CuSO 4 (1000X stock) Callus initiation media (CIM) per L: 4.43 g LS salts 30 g sucrose 1 ml 0.6 mg/ml CuSO 4 ph 5.8 with 0.1 N KOH 2g phytagel for plates (be sure bottles are dry before adding phytagel or it will clump. Also, phytagel cannot be re-melted like agar) 0.5ml of 5 mg/ml 2,4-D stock solution (add after autoclaving) This method produces callus from a much higher percentage of starting seeds faster than the whole seed method. We use this method to initiate callus from Bd ) Select immature seeds when most of the seeds are just starting to fill out. Harvest seed heads into 15ml falcon tubes and move to lab. Cap the tubes to keep seeds from drying out. 2) Remove lemma using fingers (the palea adheres too tightly to be removed without damage). 3) Surface sterilize by soaking in a solution of 10% bleach plus 0.1% triton X-100 for 4 minutes. Gently mix tubes while soaking. 4) Rinse 3 times in sterile water in the sterile hood.
2 Page 2 5) Dissect embryos out of immature seeds using fine forceps and a dissecting microscope in a laminar flow hood. Smaller embryos work best and large embryos (>0.7 mm) work very poorly. You can expect >90% of small (<0.3 mm) embryos to produce embryogenic callus and >50% of medium sized ( mm) embryos to form callus. A little practice is required to find the small embryos as they are clear. Older embryos are large and white. See figure 1A. 6) Place embryos on CIM scutellar side down and incubate at 28 o C in the dark. Seal plates with parafilm. 7) Callus produced from excised Bd21-3 embryos displays a number of distinct morphologies. First, the embryos start to from an amorphous whitish soft callus. After 1-2 weeks on CIM yellowish callus with organized structures sometimes interspersed with amorphous white callus begins to form. After 3-4 weeks the yellowish callus makes up greater than half of the callus volume (Fig 1 b). The yellowish organized callus and the callus with yellowish organized structures interspersed with amorphous callus both regenerate and are suitable for transformation. At the first subculture it is important to select only yellowish callus to transfer. At the second subculture yellowish callus is again selectively transferred. Probably the best way to get a feel for the system is to put some callus on regeneration media to test it. You can also look at some of the callus under a microscope to verify what you see with the naked eye. 8) Increase callus by subculturing at least every 2 weeks. Don t subculture more than a few times because regeneration becomes a problem with extended time in culture. The regenerability of Bd21-3 seems to decline over time so we try to transform it as soon after callus initiation as we can. Our typical subculturing routine is: dissect embryos subculture subculture transform 3-4 wks 2 wks 1 wk Figure 1. Embryogenic callus initiation. (A) Immature embryos on CI medium. The four embryos on the left are small enough to produce good embryogenic callus and the two large embryos on the right are too mature. Scale 1 mm. (B) Embryo after 3 weeks on CI medium. The yellowish organized callus (arrow) is picked for subculture. Unorganized white, watery callus (asterisk) is discarded. Scale 5 mm. (D) Closeup of embryogenic callus. (E) Closeup of non-embryogenic callus. Scale in D and E is 2 mm.
3 Transformation starting with embryogenic callus Brachypodium transformation protocol Page 3 Materials needed: antibiotic stock solutions 150 mg/ml timentin solution in water (filter sterilize) 200 mm acetosyringone stock solution prepare by dissolving g in 10 ml DMSO and filter sterilizing (use polypropylene filters). Aliquot and freeze to avoid repeated freeze-thaw cycles mm (5 mg/ml) 2,4-D stock solution (2000 X) dissolve 50 mg 2,4-D in 10 ml 95% ethanol 0.6 mg/ml CuSO 4 (1000 X stock) 9.3 mm (0.2 mg/ml) kinetin stock solution (1000 X) (dilute from 20 mg/ml stock in DMSO and filter sterilize with nylon filter) 10% Synperonic PE/F68 (Sigma, old name Pluronic F68) filter sterilize MG/L media per L: 5 g tryptone 2.5g yeast extract 5g NaCl 5 g mannitol 0.1 g MgSO g K 2 HPO g glutamic acid ph to 7.2 with 1N NaOH for plates add 15 g agar Callus initiation media (CIM) per L: 4.43 g LS salts 30 g sucrose 1 ml 0.6 mg/ml CuSO 4 ph 5.8 with 0.1 N KOH 2g phytagel for plates (be sure bottles are dry before adding phytagel or it will clump. Also, phytagel cannot be re-melted like agar) 0.5ml of 5 mg/ml 2,4-D stock solution (add after autoclaving) Regeneration media per L: 4.43 g LS salts 30 g maltose ph 5.8 with 0.1 N KOH 2 g Phytagel 1 ml of 0.2 mg/ml Kinetin stock solution (added after autoclaving) MS media per L: 4.42 g MS salts plus vitamins 30 g sucrose ph 5.7 with 0.1 N KOH 2 g Phytagel
4 Page 4 1) One week prior to co-cultivation subculture small pieces (2 mm) callus to fresh CIM and incubate at 28 o C. 2) Two days prior to co-cultivation streak out Agrobacterium strain from frozen perm on solid MG/L with appropriate antibiotics and incubate at o. You do not want single colonies and you do not need to add acetosyringone. 3) Prepare the Agro suspension by scraping Agro off the plates and resuspending in liquid CIM to OD 600 = 0.6. An easy way to do this is to add an excess of Agro, check the OD and dilute as necessary. Add acetosyringone stock solution so the Agro suspension has a final concentration of 200 μm. Add 10uL 10% Synperonic PE/F68 (Sigma, old name Pluronic F68) per 1 ml inoculation media. One plate of Agro should make at least 20 ml of suspension. 4) Immediately transfer the callus pieces from the plates to tubes and add an excess of Agrobacterium suspension. Six plates of callus fit easily in a 15 ml Falcon tube and 10 ml of suspension is sufficient. Incubate the callus in the Agro suspension for 5 minutes. I don t think this time is at all critical. 5) Place 1 sterile Whatman filter paper 7cm in a 100 X 15 mm petri plate to the center of the filter paper. 6) Pour off the inoculation media and dump calluses into large petri dish. Carefully suck off excess liquid with 1 ml pipette. Remove as much liquid as possible. 7) Place a reasonable size clump of calluses into each petri dish with filter paper. We typically place all the callus from 2 plates (50 callus pieces) of starting callus on one filter paper (Fig 2A). Spread the calluses around to lightly cover the filter paper. 8) Incubate in dark at 22 o C for 3 days. 9) Transfer callus pieces to CIM containing 150 mg/l Timentin (to kill Agro) and appropriate selective agent to kill untransformed plant tissue.** Incubate in dark at 28 o for 1 week. The calli recover quickly and grow rapidly. 10) Transfer callus pieces to fresh CIM containing 150 mg/l Timentin (to kill Agro) and appropriate selective agent to kill untransformed plant tissue.** Incubate in dark at 28 o for 2 weeks. 11) Continue to subculture and bulk up healthy callus every 2 weeks until you have a few healthy pieces for regeneration. You want to get them onto regeneration media as soon as possible. We typically start to move callus onto regeneration media after 3 weeks on selection and finish moving callus onto regeneration after 5 weeks. Keep callus lines separate so that you can ensure transformants are independent. 12) Transfer healthy callus to regeneration media containing selective agent and incubate in the light (we use 16 hr light 8 hr dark cycle) at 28 o. Shoots should appear in 2-4 weeks. 10) When plantlets are large enough to handle safely, transfer and separate plantlets to sundae cups containing MS sucrose for continued growth. (Note, the sundae cups from Solo are inexpensive cups are used for food and although not guaranteed sterile, we have not observed any contamination. If you are planning to do a lot of transformations you might want to try something like these.)
5 Page 5 11) When plantlets are large enough, carefully transplant to soil and slowly acclimate to outside conditions. We cover the plants with a plastic dome in a growth chamber (20hr light 4 hr dark). Wait until new growth is obvious, usually about a week, before removing the dome moving to normal growth conditions. Even rootless plantlets have a high survival rate. *Agro is typically grown with two antibiotics, one for the helper plasmid in the Agro strain and one for the binary plasmid you put in. For derivatives of: strain AGL1 use 100 mg/l carbinicillin plus for the following vectors: pwbvec8 use 25 mg/l spectinomycin liquid and 50 mg/ L for plates pct914-1a and pjj86 use 10 mg/l kanamycin for liquid and 50 mg/ L for plates ** For pwbvec8 and pjj86 derivatives use 40 mg/l hygromycin (note hygromycin is unstable in water so don t use old stock solutions.) General notes: Seal all plates with parafilm. Figure 2. Stages of transformation. (A) Co-cultivation on filter paper. Approximately 50 callus pieces are placed on each filter. The filter paper is 7 cm in diameter. (B) Selection of hygromycin resistant callus. Scale 5 mm. (C) After 2-4 weeks on regeneration media some of the callus produces shoots. Scale 10 mm. (D) GUS stained leaves of seven independent transgenic lines. The non-transgenic control is on the left.
6 Page 6 Summary of Brachypodium growth conditions Callus subculturing and selection Growth chamber at 28 o C, dark T 0 plant regeneration and rooting Growth chamber (28 o C, hours light / 8-4 hours dark) (Note: for rooting we use inexpensive sundae cups (Solo Cup Company) instead of tissue culture boxes.) T 0 plant growth Growth chamber 20 hr light : 4 hr dark photoperiod, 24 o C during the day and 18 o C at night Cool-white fluorescent lighting at a level of 150 mem-2s-1 Cover plants with clear plastic dome for the first 3 days No vernalization required for flowering Greenhouse no shading 24 o C in the day and 18 o C at night; supplemental lighting to extend day length to 16 hours requires 2 weeks vernalization at 4 o C to induce flowering T 1 plant growth Growth chamber 22 o C, cycle 20 hours light/4 hours dark 1 week stratification at 4 o C Greenhouse: requires 3 weeks vernalization at 4 o C for flowering
Agrobacterium tumefaciens-mediated transformation of Colletotrichum graminicola and Colletotrichum sublineolum
 Agrobacterium tumefaciens-mediated transformation of Colletotrichum graminicola and Colletotrichum sublineolum Flowers and Vaillancourt, 2005. Current Genetics 48: 380-388 NOTE added by L. Vaillancourt:
Agrobacterium tumefaciens-mediated transformation of Colletotrichum graminicola and Colletotrichum sublineolum Flowers and Vaillancourt, 2005. Current Genetics 48: 380-388 NOTE added by L. Vaillancourt:
PLANT TISSUE CULTURE
 PLANT TISSUE CULTURE Background Plant research often involves growing new plants in a controlled environment. These may be plants that we have genetically altered in some way or may be plants of which
PLANT TISSUE CULTURE Background Plant research often involves growing new plants in a controlled environment. These may be plants that we have genetically altered in some way or may be plants of which
Transformation Protocol
 To make Glycerol Stocks of Plasmids ** To be done in the hood and use RNase/DNase free tips** 1. In a 10 ml sterile tube add 3 ml autoclaved LB broth and 1.5 ul antibiotic (@ 100 ug/ul) or 3 ul antibiotic
To make Glycerol Stocks of Plasmids ** To be done in the hood and use RNase/DNase free tips** 1. In a 10 ml sterile tube add 3 ml autoclaved LB broth and 1.5 ul antibiotic (@ 100 ug/ul) or 3 ul antibiotic
Transformation of the bacterium E. coli. using a gene for Green Fluorescent Protein
 Transformation of the bacterium E. coli using a gene for Green Fluorescent Protein Background In molecular biology, transformation refers to a form of genetic exchange in which the genetic material carried
Transformation of the bacterium E. coli using a gene for Green Fluorescent Protein Background In molecular biology, transformation refers to a form of genetic exchange in which the genetic material carried
TransformAid Bacterial Transformation Kit
 Home Contacts Order Catalog Support Search Alphabetical Index Numerical Index Restriction Endonucleases Modifying Enzymes PCR Kits Markers Nucleic Acids Nucleotides & Oligonucleotides Media Transfection
Home Contacts Order Catalog Support Search Alphabetical Index Numerical Index Restriction Endonucleases Modifying Enzymes PCR Kits Markers Nucleic Acids Nucleotides & Oligonucleotides Media Transfection
HARVESTING AND CRYOPRESERVATION OF HUMAN EMBRYONIC STEM CELLS (hescs)
 HARVESTING AND CRYOPRESERVATION OF HUMAN EMBRYONIC STEM CELLS (hescs) OBJECTIVE: can be cryopreserved in a liquid nitrogen (LN 2 ) freezer for long-term storage. This Standard Operating Procedure (SOP)
HARVESTING AND CRYOPRESERVATION OF HUMAN EMBRYONIC STEM CELLS (hescs) OBJECTIVE: can be cryopreserved in a liquid nitrogen (LN 2 ) freezer for long-term storage. This Standard Operating Procedure (SOP)
Transformation Kit BACTERIAL TRANSFORMATION: GREEN FLUORESCENT PROTEIN. Partnership for Biotechnology and Genomics Education
 Transformation Kit BACTERIAL TRANSFORMATION: GREEN FLUORESCENT PROTEIN Partnership for Biotechnology and Genomics Education Barbara Soots Linda Curro Education Coordinator University of California Davis
Transformation Kit BACTERIAL TRANSFORMATION: GREEN FLUORESCENT PROTEIN Partnership for Biotechnology and Genomics Education Barbara Soots Linda Curro Education Coordinator University of California Davis
Day -1 RETRIEVAL OF OVARIES AND OOCYTE COLLECTION AND MATURATION
 Day -1 RETRIEVAL OF OVARIES AND OOCYTE COLLECTION AND MATURATION OVARY COLLECTION Materials and Equipment Needed Thermos with 2 containers with 0.5 L of transport saline in each. Appropriate attire as
Day -1 RETRIEVAL OF OVARIES AND OOCYTE COLLECTION AND MATURATION OVARY COLLECTION Materials and Equipment Needed Thermos with 2 containers with 0.5 L of transport saline in each. Appropriate attire as
Plant Responses to Different Growth Regulators (Hormones) in Tissue Culture
 Plant Responses to Different Growth Regulators (Hormones) in Tissue Culture (A Classroom Exercise) Plant growth regulators (sometimes referred to as plant hormones) induce various responses in plant pieces
Plant Responses to Different Growth Regulators (Hormones) in Tissue Culture (A Classroom Exercise) Plant growth regulators (sometimes referred to as plant hormones) induce various responses in plant pieces
Related topics: Application Note 27 Data Analysis of Tube Formation Assays.
 Tube Formation Assays in µ-slide Angiogenesis Related topics: Application Note 27 Data Analysis of Tube Formation Assays. Contents 1. General Information... 1 2. Material... 2 3. Work Flow Overview...
Tube Formation Assays in µ-slide Angiogenesis Related topics: Application Note 27 Data Analysis of Tube Formation Assays. Contents 1. General Information... 1 2. Material... 2 3. Work Flow Overview...
UTILIZATION of PLASMA ACTIVATED WATER in Biotechnology, Pharmacology and Medicine. JSC TECHNOSYSTEM-ECO Moscow, Russia April, 2009
 UTILIZATION of PLASMA ACTIVATED WATER in Biotechnology, Pharmacology and Medicine JSC TECHNOSYSTEM-ECO Moscow, Russia April, 2009 METHOD of WATER ACTIVATION with PLASMA of GAS DISCHARGE ANODE VACUUM WATER
UTILIZATION of PLASMA ACTIVATED WATER in Biotechnology, Pharmacology and Medicine JSC TECHNOSYSTEM-ECO Moscow, Russia April, 2009 METHOD of WATER ACTIVATION with PLASMA of GAS DISCHARGE ANODE VACUUM WATER
Bacterial Transformation with Green Fluorescent Protein. Table of Contents Fall 2012
 Bacterial Transformation with Green Fluorescent Protein pglo Version Table of Contents Bacterial Transformation Introduction..1 Laboratory Exercise...3 Important Laboratory Practices 3 Protocol...... 4
Bacterial Transformation with Green Fluorescent Protein pglo Version Table of Contents Bacterial Transformation Introduction..1 Laboratory Exercise...3 Important Laboratory Practices 3 Protocol...... 4
Microbiological Testing of the Sawyer Mini Filter. 16 December 2013. Summary
 Microbiological Testing of the Sawyer Mini Filter 16 December 2013 Summary The Sawyer Mini Filter was tested for its ability to remove three microorganisms Raoultella terrigena, Bacillus subtilis, and
Microbiological Testing of the Sawyer Mini Filter 16 December 2013 Summary The Sawyer Mini Filter was tested for its ability to remove three microorganisms Raoultella terrigena, Bacillus subtilis, and
Cellartis Protocol Culturing of hes Cells
 Cellartis Protocol Culturing of hes Cells This material was cultured and frozen using Cellartis protocols. WiCell recommends that stem cells should be thawed and established in the conditions in which
Cellartis Protocol Culturing of hes Cells This material was cultured and frozen using Cellartis protocols. WiCell recommends that stem cells should be thawed and established in the conditions in which
PROTOCOL. Immunocytochemistry (ICC) MATERIALS AND EQUIPMENT REQUIRED
 PROTOCOL Immunocytochemistry (ICC) 1850 Millrace Drive, Suite 3A Eugene, Oregon 97403 11-07 MATERIALS AND EQUIPMENT REQUIRED Materials: MitoSciences primary monoclonal antibody/antibodies Fluorophore-conjugated
PROTOCOL Immunocytochemistry (ICC) 1850 Millrace Drive, Suite 3A Eugene, Oregon 97403 11-07 MATERIALS AND EQUIPMENT REQUIRED Materials: MitoSciences primary monoclonal antibody/antibodies Fluorophore-conjugated
Lab Exercise 3: Media, incubation, and aseptic technique
 Lab Exercise 3: Media, incubation, and aseptic technique Objectives 1. Compare the different types of media. 2. Describe the different formats of media, plate, tube etc. 3. Explain how to sterilize it,
Lab Exercise 3: Media, incubation, and aseptic technique Objectives 1. Compare the different types of media. 2. Describe the different formats of media, plate, tube etc. 3. Explain how to sterilize it,
Agrobacterium-mediated transformation of soybean and recovery of transgenic soybean plants
 Agrobacterium-mediated transformation of soybean and recovery of transgenic soybean plants Paz, M, Martinez, J C, Kalvig, A, Fonger, T, Wang, K Improved cotyledonary node method using an alternative explant
Agrobacterium-mediated transformation of soybean and recovery of transgenic soybean plants Paz, M, Martinez, J C, Kalvig, A, Fonger, T, Wang, K Improved cotyledonary node method using an alternative explant
Running protein gels and detection of proteins
 Running protein gels and detection of proteins 1. Protein concentration determination using the BIO RAD reagent This assay uses a colour change reaction to give a direct measurement of protein concentration.
Running protein gels and detection of proteins 1. Protein concentration determination using the BIO RAD reagent This assay uses a colour change reaction to give a direct measurement of protein concentration.
Instructions. Torpedo sirna. Material. Important Guidelines. Specifications. Quality Control
 is a is a state of the art transfection reagent, specifically designed for the transfer of sirna and mirna into a variety of eukaryotic cell types. is a state of the art transfection reagent, specifically
is a is a state of the art transfection reagent, specifically designed for the transfer of sirna and mirna into a variety of eukaryotic cell types. is a state of the art transfection reagent, specifically
Green Fluorescent Protein (GFP): Genetic Transformation, Synthesis and Purification of the Recombinant Protein
 Green Fluorescent Protein (GFP): Genetic Transformation, Synthesis and Purification of the Recombinant Protein INTRODUCTION Green Fluorescent Protein (GFP) is a novel protein produced by the bioluminescent
Green Fluorescent Protein (GFP): Genetic Transformation, Synthesis and Purification of the Recombinant Protein INTRODUCTION Green Fluorescent Protein (GFP) is a novel protein produced by the bioluminescent
MGC premier Expression-Ready cdna clones TCH1103, TCM1104, TCR1105, TCB1106, TCH1203, TCM1204, TCR1205, TCB1206, TCH1303, TCM1304, TCR1305
 MGC premier Expression-Ready cdna clones TCH1103, TCM1104, TCR1105, TCB1106, TCH1203, TCM1204, TCR1205, TCB1206, TCH1303, TCM1304, TCR1305 The MGC premier Expression-Ready collection has the high quality,
MGC premier Expression-Ready cdna clones TCH1103, TCM1104, TCR1105, TCB1106, TCH1203, TCM1204, TCR1205, TCB1206, TCH1303, TCM1304, TCR1305 The MGC premier Expression-Ready collection has the high quality,
Lab 10: Bacterial Transformation, part 2, DNA plasmid preps, Determining DNA Concentration and Purity
 Lab 10: Bacterial Transformation, part 2, DNA plasmid preps, Determining DNA Concentration and Purity Today you analyze the results of your bacterial transformation from last week and determine the efficiency
Lab 10: Bacterial Transformation, part 2, DNA plasmid preps, Determining DNA Concentration and Purity Today you analyze the results of your bacterial transformation from last week and determine the efficiency
National Food Safety Standard Food microbiological examination: Aerobic plate count
 National Food Safety Standard of the People s Republic of China GB4789.2-2010 National Food Safety Standard Food microbiological examination: Aerobic plate count Issued by 2010-03-26 Implemented by 2010-06-01
National Food Safety Standard of the People s Republic of China GB4789.2-2010 National Food Safety Standard Food microbiological examination: Aerobic plate count Issued by 2010-03-26 Implemented by 2010-06-01
The Influence of Carbon Dioxide on Algae Growth
 The Influence of Carbon Dioxide on Algae Growth The first objective of this experiment is to show that increased atmospheric concentrations of carbon dioxide, CO 2, can stimulate algae growth. The second
The Influence of Carbon Dioxide on Algae Growth The first objective of this experiment is to show that increased atmospheric concentrations of carbon dioxide, CO 2, can stimulate algae growth. The second
Chromatin Immunoprecipitation (ChIP)
 Chromatin Immunoprecipitation (ChIP) Day 1 A) DNA shearing 1. Samples Dissect tissue (One Mouse OBs) of interest and transfer to an eppendorf containing 0.5 ml of dissecting media (on ice) or PBS but without
Chromatin Immunoprecipitation (ChIP) Day 1 A) DNA shearing 1. Samples Dissect tissue (One Mouse OBs) of interest and transfer to an eppendorf containing 0.5 ml of dissecting media (on ice) or PBS but without
Amaxa Mouse T Cell Nucleofector Kit
 Amaxa Mouse T Cell Nucleofector Kit For T cells isolated from C57BL/6 & BALB/c mice Evaluated for murine T cells isolated from C57BL/6 & BALB/c mice This protocol is designed for murine lymphocytes or
Amaxa Mouse T Cell Nucleofector Kit For T cells isolated from C57BL/6 & BALB/c mice Evaluated for murine T cells isolated from C57BL/6 & BALB/c mice This protocol is designed for murine lymphocytes or
Dose/Response Experiments on Lettuce Seeds
 Dose/Response Experiments on Lettuce Seeds Name: Date: Lab# Bioassays: Background Information on Dose/Response Experiments A bioassay involves use of a biological organism to test for chemical toxicity.
Dose/Response Experiments on Lettuce Seeds Name: Date: Lab# Bioassays: Background Information on Dose/Response Experiments A bioassay involves use of a biological organism to test for chemical toxicity.
Transferring a Broth Culture to Fresh Broth
 Sterile Technique It is very important in microbiology to work with pure cultures. Unfortunately this is difficult. The world around us is covered with microorganisms. Microorganisms are even carried on
Sterile Technique It is very important in microbiology to work with pure cultures. Unfortunately this is difficult. The world around us is covered with microorganisms. Microorganisms are even carried on
Fluoride Strengthens Teeth
 Fluoride Strengthens Teeth Two hard-boiled eggs Fluoride gel or solution, 4 to 6 oz. (from dental office) Three clean plastic containers Several cans of dark soda Water 1. Place a hard-boiled egg in one
Fluoride Strengthens Teeth Two hard-boiled eggs Fluoride gel or solution, 4 to 6 oz. (from dental office) Three clean plastic containers Several cans of dark soda Water 1. Place a hard-boiled egg in one
BUFFERS and MEDIAS Coomassie Blue Staining Solution Coomassie blue Destaining Solution DMEM Normal Cell Culture Media
 BUFFERS and MEDIAS Coomassie Blue Staining Solution 2 g Coomassie Blue 2 L Methanol or Ethanol * 1.6 L 400 ml Glacial acetic acid *If you will be microwaving the gel in staining solution for rapid staining
BUFFERS and MEDIAS Coomassie Blue Staining Solution 2 g Coomassie Blue 2 L Methanol or Ethanol * 1.6 L 400 ml Glacial acetic acid *If you will be microwaving the gel in staining solution for rapid staining
Procedure for RNA isolation from human muscle or fat
 Procedure for RNA isolation from human muscle or fat Reagents, all Rnase free: 20% SDS DEPC-H2O Rnase ZAP 75% EtOH Trizol Chloroform Isopropanol 0.8M NaCitrate/1.2M NaCl TE buffer, ph 7.0 1. Homogenizer-probe
Procedure for RNA isolation from human muscle or fat Reagents, all Rnase free: 20% SDS DEPC-H2O Rnase ZAP 75% EtOH Trizol Chloroform Isopropanol 0.8M NaCitrate/1.2M NaCl TE buffer, ph 7.0 1. Homogenizer-probe
Environmental Water Testing: Surface Water, Groundwater, Hard Water, Wastewater, & Seawater
 Document: AND Sol Env 08 2013 Environmental Water Testing: Surface Water, Groundwater, Hard Water, Wastewater, & Seawater Matrix specific sample preparation and testing methods for environmental waters
Document: AND Sol Env 08 2013 Environmental Water Testing: Surface Water, Groundwater, Hard Water, Wastewater, & Seawater Matrix specific sample preparation and testing methods for environmental waters
BD PuraMatrix Peptide Hydrogel
 BD PuraMatrix Peptide Hydrogel Catalog No. 354250 Guidelines for Use FOR RESEARCH USE ONLY NOT FOR CLINICAL, DIAGNOSTIC OR THERAPEUTIC USE SPC-354250-G rev 2.0 1 TABLE OF CONTENTS Intended Use... 2 Materials
BD PuraMatrix Peptide Hydrogel Catalog No. 354250 Guidelines for Use FOR RESEARCH USE ONLY NOT FOR CLINICAL, DIAGNOSTIC OR THERAPEUTIC USE SPC-354250-G rev 2.0 1 TABLE OF CONTENTS Intended Use... 2 Materials
Test Method for the Continuous Reduction of Bacterial Contamination on Copper Alloy Surfaces
 Test Method for the Continuous Reduction of Bacterial Contamination on Copper Alloy Surfaces Test Organisms: Staphylococcus aureus (ATCC 6538) Enterobacter aerogenes (ATCC 13048) Pseudomonas aeruginosa
Test Method for the Continuous Reduction of Bacterial Contamination on Copper Alloy Surfaces Test Organisms: Staphylococcus aureus (ATCC 6538) Enterobacter aerogenes (ATCC 13048) Pseudomonas aeruginosa
MTT Cell Proliferation Assay
 ATCC 30-1010K Store at 4 C This product is intended for laboratory research purposes only. It is not intended for use in humans, animals or for diagnostics. Introduction Measurement of cell viability and
ATCC 30-1010K Store at 4 C This product is intended for laboratory research purposes only. It is not intended for use in humans, animals or for diagnostics. Introduction Measurement of cell viability and
LAB 4. Cultivation of Bacteria INTRODUCTION
 LAB 4. Cultivation of Bacteria Protocols for use of cultivation of bacteria, use of general growth, enriched, selective and differential media, plate pouring, determination of temperature range for growth
LAB 4. Cultivation of Bacteria Protocols for use of cultivation of bacteria, use of general growth, enriched, selective and differential media, plate pouring, determination of temperature range for growth
EXPERIMENT 9 - IDENTIFYING FEATURES OF MUTANT EMBRYO USING NOMARSKI MICROSCOPY (GENE TWO)
 EXPERIMENT 9 - IDENTIFYING FEATURES OF MUTANT EMBRYO USING NOMARSKI MICROSCOPY (GENE TWO) Purpose: To introduce Differential Interference Contrast (DIC) or Nomarski Interference Contrast (NIC) microscopy
EXPERIMENT 9 - IDENTIFYING FEATURES OF MUTANT EMBRYO USING NOMARSKI MICROSCOPY (GENE TWO) Purpose: To introduce Differential Interference Contrast (DIC) or Nomarski Interference Contrast (NIC) microscopy
Concert Plant RNA Reagent
 General Information Concert Plant RNA Reagent Catalog no. 12322-012 Description Invitrogen s Concert Plant RNA Reagent is a proprietary RNA isolation reagent that allows isolation of high quality total
General Information Concert Plant RNA Reagent Catalog no. 12322-012 Description Invitrogen s Concert Plant RNA Reagent is a proprietary RNA isolation reagent that allows isolation of high quality total
BioResearch. RAFT 3D Cell Culture Kit Protocol
 BioResearch RAFT 3D Cell Culture Kit Protocol RAFT 3D Cell Culture Kit This protocol describes in detail how to produce RAFT 3D Cell Cultures in 96-well plates, 24-well plates and 24-well inserts. It is
BioResearch RAFT 3D Cell Culture Kit Protocol RAFT 3D Cell Culture Kit This protocol describes in detail how to produce RAFT 3D Cell Cultures in 96-well plates, 24-well plates and 24-well inserts. It is
Southern Blot Analysis (from Baker lab, university of Florida)
 Southern Blot Analysis (from Baker lab, university of Florida) DNA Prep Prepare DNA via your favorite method. You may find a protocol under Mini Yeast Genomic Prep. Restriction Digest 1.Digest DNA with
Southern Blot Analysis (from Baker lab, university of Florida) DNA Prep Prepare DNA via your favorite method. You may find a protocol under Mini Yeast Genomic Prep. Restriction Digest 1.Digest DNA with
Chromatin Immunoprecipitation
 Chromatin Immunoprecipitation A) Prepare a yeast culture (see the Galactose Induction Protocol for details). 1) Start a small culture (e.g. 2 ml) in YEPD or selective media from a single colony. 2) Spin
Chromatin Immunoprecipitation A) Prepare a yeast culture (see the Galactose Induction Protocol for details). 1) Start a small culture (e.g. 2 ml) in YEPD or selective media from a single colony. 2) Spin
Disc Diffusion Susceptibility Methods
 Disc Diffusion Susceptibility Methods Introduction When a filter paper disc impregnated with a chemical is placed on agar the chemical will diffuse from the disc into the agar. This diffusion will place
Disc Diffusion Susceptibility Methods Introduction When a filter paper disc impregnated with a chemical is placed on agar the chemical will diffuse from the disc into the agar. This diffusion will place
Determine Osmolarity Activity Using RFP under PompC
 Determine Osmolarity Activity Using RFP under PompC Materials and Equipment: E. coli : o containing RFP under Plac o (blank) Isogenic strains containing mrfp under PompC: o 21153 o 3367-3 ΔenvZ Isogenic
Determine Osmolarity Activity Using RFP under PompC Materials and Equipment: E. coli : o containing RFP under Plac o (blank) Isogenic strains containing mrfp under PompC: o 21153 o 3367-3 ΔenvZ Isogenic
TERRESTRIAL ORCHID SEED SOWING KIT
 TERRESTRIAL ORCHID SEED SOWING KIT Product No. O788 PhytoTechnology Laboratories P.O. Box 12205 Shawnee Mission, KS 66282-2205 Phone: 888-749-8682 or 1-913-341-5343 Fax: 888-449-8682 or 1-913-341-5442
TERRESTRIAL ORCHID SEED SOWING KIT Product No. O788 PhytoTechnology Laboratories P.O. Box 12205 Shawnee Mission, KS 66282-2205 Phone: 888-749-8682 or 1-913-341-5343 Fax: 888-449-8682 or 1-913-341-5442
RiboZol RNA Extraction Reagents
 RiboZol RNA Extraction Reagents Code Description Size N580-30ML-SAMPLE Ribozol TM RNA Extraction Reagent 30 ml N580-30ML Ribozol TM RNA Extraction Reagent 30 ml N580-100ML Ribozol TM RNA Extraction Reagent
RiboZol RNA Extraction Reagents Code Description Size N580-30ML-SAMPLE Ribozol TM RNA Extraction Reagent 30 ml N580-30ML Ribozol TM RNA Extraction Reagent 30 ml N580-100ML Ribozol TM RNA Extraction Reagent
MGC premier full length cdna and ORF clones
 MGC premier full length cdna and ORF clones TCH1003, TCM1004, TCR1005, TCB1006, TCL1007, TCT1008, TCZ1009, TOH6003, TOM6004, TOZ6009, TCHS1003, TCMS1004, TCRS1005, TCBS1006, TCLS1007, TCTS1008 MGC premier
MGC premier full length cdna and ORF clones TCH1003, TCM1004, TCR1005, TCB1006, TCL1007, TCT1008, TCZ1009, TOH6003, TOM6004, TOZ6009, TCHS1003, TCMS1004, TCRS1005, TCBS1006, TCLS1007, TCTS1008 MGC premier
EFFECT OF SOME TREATMENTS ON SEEDS GERMINATION, SHOOTS MULTIPLICATION AND ROOTING OF DAHLIA PLANTS VIA IN VITRO CULTURE
 EFFECT OF SOME TREATMENTS ON SEEDS GERMINATION, SHOOTS MULTIPLICATION AND ROOTING OF DAHLIA PLANTS VIA IN VITRO CULTURE Majid A. Ibrahim* and Israa A. Daraj Department of Horticulture and Landscape Design,
EFFECT OF SOME TREATMENTS ON SEEDS GERMINATION, SHOOTS MULTIPLICATION AND ROOTING OF DAHLIA PLANTS VIA IN VITRO CULTURE Majid A. Ibrahim* and Israa A. Daraj Department of Horticulture and Landscape Design,
ENUMERATION OF MICROORGANISMS. To learn the different techniques used to count the number of microorganisms in a sample.
 ENUMERATION OF MICROORGANISMS I. OBJECTIVES To learn the different techniques used to count the number of microorganisms in a sample. To be able to differentiate between different enumeration techniques
ENUMERATION OF MICROORGANISMS I. OBJECTIVES To learn the different techniques used to count the number of microorganisms in a sample. To be able to differentiate between different enumeration techniques
UltraClean Soil DNA Isolation Kit
 PAGE 1 UltraClean Soil DNA Isolation Kit Catalog # 12800-50 50 preps New improved PCR inhibitor removal solution (IRS) included Instruction Manual (New Alternative Protocol maximizes yields) Introduction
PAGE 1 UltraClean Soil DNA Isolation Kit Catalog # 12800-50 50 preps New improved PCR inhibitor removal solution (IRS) included Instruction Manual (New Alternative Protocol maximizes yields) Introduction
2D gel Protocol. 2. Determining Protein Concentration of cell lysates
 2D gel Protocol 1. Lysis and Protein Extraction from cells Prepare cell lysates with Trizol extraction by following Kathleen Lyons s protocol: AfCS Procedure Protocol PP00000155, Version 1, 05/12/03 (Ref.1).
2D gel Protocol 1. Lysis and Protein Extraction from cells Prepare cell lysates with Trizol extraction by following Kathleen Lyons s protocol: AfCS Procedure Protocol PP00000155, Version 1, 05/12/03 (Ref.1).
GRS Plasmid Purification Kit Transfection Grade GK73.0002 (2 MaxiPreps)
 1 GRS Plasmid Purification Kit Transfection Grade GK73.0002 (2 MaxiPreps) (FOR RESEARCH ONLY) Sample : Expected Yield : Endotoxin: Format : Operation Time : Elution Volume : 50-400 ml of cultured bacterial
1 GRS Plasmid Purification Kit Transfection Grade GK73.0002 (2 MaxiPreps) (FOR RESEARCH ONLY) Sample : Expected Yield : Endotoxin: Format : Operation Time : Elution Volume : 50-400 ml of cultured bacterial
Paper Chromatography: Separation and Identification of Five Metal Cations
 Paper Chromatography: Separation and Identification of Five Metal Cations Objectives Known and unknown solutions of the metal ions Ag +, Fe 3+, Co 2+, Cu 2+ and Hg 2+ will be analyzed using paper chromatography.
Paper Chromatography: Separation and Identification of Five Metal Cations Objectives Known and unknown solutions of the metal ions Ag +, Fe 3+, Co 2+, Cu 2+ and Hg 2+ will be analyzed using paper chromatography.
Enzyme Pre-Lab. Using the Enzyme worksheet and Enzyme lab handout answer the Pre-Lab questions the pre-lab must be complete before beginning the lab.
 Enzyme Pre-Lab Using the Enzyme worksheet and Enzyme lab handout answer the Pre-Lab questions the pre-lab must be complete before beginning the lab. Background: In this investigation, you will study several
Enzyme Pre-Lab Using the Enzyme worksheet and Enzyme lab handout answer the Pre-Lab questions the pre-lab must be complete before beginning the lab. Background: In this investigation, you will study several
Cell Culture Experiments
 Cell Culture Experiments Cell cultures are another way to observe and analyze biological systems. These in vitro (in glass; outside the living body and in an artificial environment) models allow experimental
Cell Culture Experiments Cell cultures are another way to observe and analyze biological systems. These in vitro (in glass; outside the living body and in an artificial environment) models allow experimental
Radius 24-Well Cell Migration Assay (Laminin Coated)
 Product Manual Radius 24-Well Cell Migration Assay (Laminin Coated) Catalog Number CBA-125-LN 24 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Cell migration is a highly
Product Manual Radius 24-Well Cell Migration Assay (Laminin Coated) Catalog Number CBA-125-LN 24 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Cell migration is a highly
Northern blot analysis for microrna. (Narry Kim s lab)
 Northern blot analysis for microrna (Narry Kim s lab) Materials 1. 10~50 μg of total RNA extracted from HeLa cells treated with sirna 2. RNA loading buffer 3. Probe: DNA oligonucleotide complementary to
Northern blot analysis for microrna (Narry Kim s lab) Materials 1. 10~50 μg of total RNA extracted from HeLa cells treated with sirna 2. RNA loading buffer 3. Probe: DNA oligonucleotide complementary to
Amaxa 4D-Nucleofector Protocol for Mouse Embryonic Stem [ES] Cells For 4D-Nucleofector X Unit Transfection in suspension
![Amaxa 4D-Nucleofector Protocol for Mouse Embryonic Stem [ES] Cells For 4D-Nucleofector X Unit Transfection in suspension Amaxa 4D-Nucleofector Protocol for Mouse Embryonic Stem [ES] Cells For 4D-Nucleofector X Unit Transfection in suspension](/thumbs/30/14702458.jpg) For 4D-Nucleofector X Unit Transfection in suspension Cells derived from mouse blastocysts; round cells, growing in clumps s 1. This protocol is meant to provide an outline for the handling and the Nucleofection
For 4D-Nucleofector X Unit Transfection in suspension Cells derived from mouse blastocysts; round cells, growing in clumps s 1. This protocol is meant to provide an outline for the handling and the Nucleofection
Classic Immunoprecipitation
 292PR 01 G-Biosciences 1-800-628-7730 1-314-991-6034 technical@gbiosciences.com A Geno Technology, Inc. (USA) brand name Classic Immunoprecipitation Utilizes Protein A/G Agarose for Antibody Binding (Cat.
292PR 01 G-Biosciences 1-800-628-7730 1-314-991-6034 technical@gbiosciences.com A Geno Technology, Inc. (USA) brand name Classic Immunoprecipitation Utilizes Protein A/G Agarose for Antibody Binding (Cat.
Mesenchymal Stem Cells
 Mesenchymal Stem Cells Instruction Manual Product Size Catalog Number from Bone Marrow (hmsc-bm) from Umbilical Cord Matrix (hmsc-uc) from Adipose Tissue (hmsc-at) 500,000 cryopreserved cells 500,000 proliferating
Mesenchymal Stem Cells Instruction Manual Product Size Catalog Number from Bone Marrow (hmsc-bm) from Umbilical Cord Matrix (hmsc-uc) from Adipose Tissue (hmsc-at) 500,000 cryopreserved cells 500,000 proliferating
Screening lambda cdna or genomic libraries
 Screening lambda cdna or genomic libraries by Michael Koelle 5/13/97 Media Need LB plates (9 cm or 14 cm) to plate the library and do secondary screens. I typically screen about 500,000-1 million plaques
Screening lambda cdna or genomic libraries by Michael Koelle 5/13/97 Media Need LB plates (9 cm or 14 cm) to plate the library and do secondary screens. I typically screen about 500,000-1 million plaques
Microbiology BIOL 275 DILUTIONS
 DILUTIONS Occasionally a solution is too concentrated to be used as is. For example, when one is performing manual blood counts, the blood contains too many cells to be counted as such. Or when performing
DILUTIONS Occasionally a solution is too concentrated to be used as is. For example, when one is performing manual blood counts, the blood contains too many cells to be counted as such. Or when performing
Name Section Lab 5 Photosynthesis, Respiration and Fermentation
 Name Section Lab 5 Photosynthesis, Respiration and Fermentation Plants are photosynthetic, which means that they produce their own food from atmospheric CO 2 using light energy from the sun. This process
Name Section Lab 5 Photosynthesis, Respiration and Fermentation Plants are photosynthetic, which means that they produce their own food from atmospheric CO 2 using light energy from the sun. This process
Quantifying Bacterial Concentration using a Calibrated Growth Curve
 BTEC 4200 Lab 2. Quantifying Bacterial Concentration using a Calibrated Growth Curve Background and References Bacterial concentration can be measured by several methods, all of which you have studied
BTEC 4200 Lab 2. Quantifying Bacterial Concentration using a Calibrated Growth Curve Background and References Bacterial concentration can be measured by several methods, all of which you have studied
TransIT -2020 Transfection Reagent
 Quick Reference Protocol, MSDS and Certificate of Analysis available at mirusbio.com/5400 INTRODUCTION TransIT -2020 Transfection Reagent is a high-performance, animal-origin free, broad spectrum reagent
Quick Reference Protocol, MSDS and Certificate of Analysis available at mirusbio.com/5400 INTRODUCTION TransIT -2020 Transfection Reagent is a high-performance, animal-origin free, broad spectrum reagent
Lab Exercise 2 Media and Culture
 Lab Exercise 2 Media and Culture Lab Exercise #2 Bacterial Media & Culture I. OBJECTIVES: Practice microbial collection techniques Describe colony morphology and the relationship to microbial identification.
Lab Exercise 2 Media and Culture Lab Exercise #2 Bacterial Media & Culture I. OBJECTIVES: Practice microbial collection techniques Describe colony morphology and the relationship to microbial identification.
Follicle Dermal Papilla Cell
 Follicle Dermal Papilla Cell Instruction Manual Product Size Catalog Number Human Follicle Dermal Papilla Cells (HFDPC) 500,000 cryopreserved cells 500,000 proliferating cells C-12071 C-12072 Product Description
Follicle Dermal Papilla Cell Instruction Manual Product Size Catalog Number Human Follicle Dermal Papilla Cells (HFDPC) 500,000 cryopreserved cells 500,000 proliferating cells C-12071 C-12072 Product Description
Blood Collection and Processing SOP
 Brisbane Breast Bank Blood Collection and Processing SOP Breast Pathology Laboratory University of Queensland Centre for Clinical Research Blood Collection We collect 30ml of blood from patients who have
Brisbane Breast Bank Blood Collection and Processing SOP Breast Pathology Laboratory University of Queensland Centre for Clinical Research Blood Collection We collect 30ml of blood from patients who have
Islet Viability Assessment by Single Cell Flow Cytometry
 Islet Viability Assessment by Single Cell Flow Cytometry Page 1 of 8 Purpose: To comprehensively assess the viability of the islet cell preparation prior to transplantation. Tissue Samples: A sample containing
Islet Viability Assessment by Single Cell Flow Cytometry Page 1 of 8 Purpose: To comprehensively assess the viability of the islet cell preparation prior to transplantation. Tissue Samples: A sample containing
RPCI 004 v.002 Staining Procedure For all Directly Conjugated Reagents (Whole Blood Method)
 Immune Tolerance Network RPCI 004 v.002 Staining Procedure For all Directly Conjugated Reagents (Whole Blood Method) Author: Paul Wallace, Director, RPCI Laboratory of Flow Cytometry Approved by: Paul
Immune Tolerance Network RPCI 004 v.002 Staining Procedure For all Directly Conjugated Reagents (Whole Blood Method) Author: Paul Wallace, Director, RPCI Laboratory of Flow Cytometry Approved by: Paul
RAGE. Plugs for RAGE/PFGE
 1 RAGE Rotating Field Gel Electrophoresis (RAGE) is a variation on Pulsed Field Gel Electrophoresis (PFGE) and gives similar results. We use equipment that was only briefly marketed by Stratagene, at far
1 RAGE Rotating Field Gel Electrophoresis (RAGE) is a variation on Pulsed Field Gel Electrophoresis (PFGE) and gives similar results. We use equipment that was only briefly marketed by Stratagene, at far
LABORATORY ORGANIZATION
 LABORATORY ORGANIZATION A laboratory designed for invitro cultures with plant tissues should have sufficient space for performing several functions maintaining sterile conditions. A plant tissue culture
LABORATORY ORGANIZATION A laboratory designed for invitro cultures with plant tissues should have sufficient space for performing several functions maintaining sterile conditions. A plant tissue culture
EPIPHYTIC ORCHID SEED SOWING KIT
 EPIPHYTIC ORCHID SEED SOWING KIT Product No. O799 PhytoTechnology Laboratories P.O. Box 12205 Shawnee Mission, KS 66282-2205 Phone: 888-749-8682 or 1-913-341-5343 Fax: 888-449-8682 or 1-913-341-5442 Web
EPIPHYTIC ORCHID SEED SOWING KIT Product No. O799 PhytoTechnology Laboratories P.O. Box 12205 Shawnee Mission, KS 66282-2205 Phone: 888-749-8682 or 1-913-341-5343 Fax: 888-449-8682 or 1-913-341-5442 Web
DETECTION OF BACTERIAL MOTILITY. To demonstrate bacterial motility by microscopic and macroscopic techniques.
 DETECTION OF BACTERIAL MOTILITY I. OBJECTIVES To demonstrate bacterial motility by microscopic and macroscopic techniques. To observe flagella in prepared slides stained by specific flagellar stains. II.
DETECTION OF BACTERIAL MOTILITY I. OBJECTIVES To demonstrate bacterial motility by microscopic and macroscopic techniques. To observe flagella in prepared slides stained by specific flagellar stains. II.
Agencourt RNAdvance Blood Kit for Free Circulating DNA and mirna/rna Isolation from 200-300μL of Plasma and Serum
 SUPPLEMENTAL PROTOCOL WHITE PAPER Agencourt RNAdvance Blood Kit for Free Circulating DNA and mirna/rna Isolation from 200-300μL of Plasma and Serum Bee Na Lee, Ph.D., Beckman Coulter Life Sciences Process
SUPPLEMENTAL PROTOCOL WHITE PAPER Agencourt RNAdvance Blood Kit for Free Circulating DNA and mirna/rna Isolation from 200-300μL of Plasma and Serum Bee Na Lee, Ph.D., Beckman Coulter Life Sciences Process
TEACHER ACTIVITY GUIDE
 Page 1/5 EXPECTED OUTCOMES TEACHER ACTIVITY GUIDE ROOT BEER PRODUCTION Taken from IFT Experiments in Food Science Series This activity will allow student an opportunity to explore yeast fermentation by
Page 1/5 EXPECTED OUTCOMES TEACHER ACTIVITY GUIDE ROOT BEER PRODUCTION Taken from IFT Experiments in Food Science Series This activity will allow student an opportunity to explore yeast fermentation by
CHEF Genomic DNA Plug Kits Instruction Manual
 CHEF Genomic DNA Plug Kits Instruction Manual Catalog Numbers 170-3591 170-3592 170-3593 For Technical Service Call Your Local Bio-Rad Office or in the U.S. Call 1-800-4BIORAD (1-800-424-6723) Table of
CHEF Genomic DNA Plug Kits Instruction Manual Catalog Numbers 170-3591 170-3592 170-3593 For Technical Service Call Your Local Bio-Rad Office or in the U.S. Call 1-800-4BIORAD (1-800-424-6723) Table of
LAB 11 PLASMID DNA MINIPREP
 LAB 11 PLASMID DNA MINIPREP STUDENT GUIDE GOAL The objective of this lab is to perform extraction of plasmid DNA and analyze the results. OBJECTIVES After completion, the student should be able to: 1.
LAB 11 PLASMID DNA MINIPREP STUDENT GUIDE GOAL The objective of this lab is to perform extraction of plasmid DNA and analyze the results. OBJECTIVES After completion, the student should be able to: 1.
Poietics human mesenchymal stem cells Instructions for use
 Poietics human mesenchymal stem cells Instructions for use www.lonza.com U.S. Scientific Support: 800-521-0390 scientific.support@lonza.com EU/ROW Scientific Support: +49-221-99199-400 scientific.support.eu@lonza.com
Poietics human mesenchymal stem cells Instructions for use www.lonza.com U.S. Scientific Support: 800-521-0390 scientific.support@lonza.com EU/ROW Scientific Support: +49-221-99199-400 scientific.support.eu@lonza.com
Potato Microbiology. Sarah Follenweider, The English High School 2009 Summer Research Internship Program
 Potato Microbiology Sarah Follenweider, The English High School 2009 Summer Research Internship Program Introduction: A number of microorganisms thrive on the nutrients that can be found in a potato. My
Potato Microbiology Sarah Follenweider, The English High School 2009 Summer Research Internship Program Introduction: A number of microorganisms thrive on the nutrients that can be found in a potato. My
DNA SPOOLING 1 ISOLATION OF DNA FROM ONION
 DNA SPOOLING 1 ISOLATION OF DNA FROM ONION INTRODUCTION This laboratory protocol will demonstrate several basic steps required for isolation of chromosomal DNA from cells. To extract the chromosomal DNA,
DNA SPOOLING 1 ISOLATION OF DNA FROM ONION INTRODUCTION This laboratory protocol will demonstrate several basic steps required for isolation of chromosomal DNA from cells. To extract the chromosomal DNA,
Amaxa Mouse/Rat Hepatocyte Nucleofector Kit
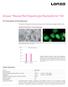 Amaxa Mouse/Rat Hepatocyte Nucleofector Kit For Primary Mouse and Rat Hepatocytes This protocol is designed for freshly isolated mouse and rat hepatocytes; polygonal, adherent cells Example for Nucleofection
Amaxa Mouse/Rat Hepatocyte Nucleofector Kit For Primary Mouse and Rat Hepatocytes This protocol is designed for freshly isolated mouse and rat hepatocytes; polygonal, adherent cells Example for Nucleofection
Standard Operating Procedure (SOP)
 Standard Operating Procedure (SOP) Title: Direct Fluorescent Antibody Test (DFAT) for the detection of Renibacterium salmoninarum in tissues Number: BACT-3 Version: 02 Created December 14, 2011 Approval:
Standard Operating Procedure (SOP) Title: Direct Fluorescent Antibody Test (DFAT) for the detection of Renibacterium salmoninarum in tissues Number: BACT-3 Version: 02 Created December 14, 2011 Approval:
GASTRIC ORGANOID CULTURE PROTOCOL
 GASTRIC ORGANOID CULTURE PROTOCOL THIS PROTOCOL PROVIDES THE PROCEDURE FOR SUBCULTURING NORMAL HUMAN GASTRIC ORGANOIDS WHICH WAS DERIVED FROM THE SUBMERGED METHOD AS DESCRIBED IN BARKER N, ET AL. LGR5+VE
GASTRIC ORGANOID CULTURE PROTOCOL THIS PROTOCOL PROVIDES THE PROCEDURE FOR SUBCULTURING NORMAL HUMAN GASTRIC ORGANOIDS WHICH WAS DERIVED FROM THE SUBMERGED METHOD AS DESCRIBED IN BARKER N, ET AL. LGR5+VE
Measuring Cell Viability/Cytotoxicity: Cell Counting Kit-F
 Introduction The Cell Counting Kit-F is a fluorometic assay for the determination of viable cell numbers. Calcein-AM in this kit passes through the cell membrane and is hydrolized by the esterase in the
Introduction The Cell Counting Kit-F is a fluorometic assay for the determination of viable cell numbers. Calcein-AM in this kit passes through the cell membrane and is hydrolized by the esterase in the
Frozen-EZ Yeast Transformation II Catalog No. T2001
 INSTRUCTIONS Frozen-EZ Yeast Transformation II Catalog No. T2001 Highlights High transformation efficiency that yields approximately 10 5-10 6 transformants per μg plasmid DNA (circular). Frozen storage
INSTRUCTIONS Frozen-EZ Yeast Transformation II Catalog No. T2001 Highlights High transformation efficiency that yields approximately 10 5-10 6 transformants per μg plasmid DNA (circular). Frozen storage
Enzyme Action: Testing Catalase Activity
 Enzyme Action: Testing Catalase Activity Experiment 6A Many organisms can decompose hydrogen peroxide (H 2 O 2 ) enzymatically. Enzymes are globular proteins, responsible for most of the chemical activities
Enzyme Action: Testing Catalase Activity Experiment 6A Many organisms can decompose hydrogen peroxide (H 2 O 2 ) enzymatically. Enzymes are globular proteins, responsible for most of the chemical activities
pmod2-puro A plasmid containing a synthetic Puromycin resistance gene Catalog # pmod2-puro For research use only Version # 11H29-MM
 pmod2-puro A plasmid containing a synthetic Puromycin resistance gene Catalog # pmod2-puro For research use only Version # 11H29-MM PrOduct information content: - 20 mg of lyophilized pmod2-puro plasmid
pmod2-puro A plasmid containing a synthetic Puromycin resistance gene Catalog # pmod2-puro For research use only Version # 11H29-MM PrOduct information content: - 20 mg of lyophilized pmod2-puro plasmid
An In-Gel Digestion Protocol
 An In-Gel Digestion Protocol This protocol describes the digestion of a protein present in an SDS-PAGE gel band with trypsin. The band can be taken from either a 1D or 2D electrophoresis gel. Reagents
An In-Gel Digestion Protocol This protocol describes the digestion of a protein present in an SDS-PAGE gel band with trypsin. The band can be taken from either a 1D or 2D electrophoresis gel. Reagents
STANDARD OPERATING PROCEDURE
 STANDARD OPERATING PROCEDURE Title: Antibody Production at Strategic Diagnostics Inc. SOP#: M-119 Version #: 1 Date Approved: August 6, 2009 Author: Strategic Diagnostic Inc. Date Modified: 1. PURPOSE
STANDARD OPERATING PROCEDURE Title: Antibody Production at Strategic Diagnostics Inc. SOP#: M-119 Version #: 1 Date Approved: August 6, 2009 Author: Strategic Diagnostic Inc. Date Modified: 1. PURPOSE
Enzyme Action: Testing Catalase Activity
 Enzyme Action: Testing Catalase Activity Experiment 6A Many organisms can decompose hydrogen peroxide (H 2 O 2 ) enzymatically. Enzymes are globular proteins, responsible for most of the chemical activities
Enzyme Action: Testing Catalase Activity Experiment 6A Many organisms can decompose hydrogen peroxide (H 2 O 2 ) enzymatically. Enzymes are globular proteins, responsible for most of the chemical activities
Aseptic Technique. A GMP/GTP Training Module
 Aseptic Technique A GMP/GTP Training Module Aseptic Technique The GMP Facility manufactures products for clinical use These products must meet a number of requirements, one of which is that they are sterile
Aseptic Technique A GMP/GTP Training Module Aseptic Technique The GMP Facility manufactures products for clinical use These products must meet a number of requirements, one of which is that they are sterile
The fastest spin-column based procedure for purifying up to 10 mg of ultra-pure endotoxin-free transfection-grade plasmid DNA.
 INSTRUCTION MANUAL ZymoPURE Plasmid Gigaprep Kit Catalog Nos. D4204 (Patent Pending) Highlights The fastest spin-column based procedure for purifying up to 10 mg of ultra-pure endotoxin-free transfection-grade
INSTRUCTION MANUAL ZymoPURE Plasmid Gigaprep Kit Catalog Nos. D4204 (Patent Pending) Highlights The fastest spin-column based procedure for purifying up to 10 mg of ultra-pure endotoxin-free transfection-grade
BACTERIAL ENUMERATION
 BACTERIAL ENUMERATION In the study of microbiology, there are numerous occasions when it is necessary to either estimate or determine the number of bacterial cells in a broth culture or liquid medium.
BACTERIAL ENUMERATION In the study of microbiology, there are numerous occasions when it is necessary to either estimate or determine the number of bacterial cells in a broth culture or liquid medium.
GROWING BACTERIA INTRODUCTION
 GROWING BACTERIA INTRODUCTION E. coli is a normal part of the bacterial flora of the human gut. It is not generally considered pathogenic, although some strains are highly toxic (recent food poisonings
GROWING BACTERIA INTRODUCTION E. coli is a normal part of the bacterial flora of the human gut. It is not generally considered pathogenic, although some strains are highly toxic (recent food poisonings
Enrichment for Phototrophic "Non-sulfur" and "Sulfur" Bacteria
 Enrichment for Phototrophic "Non-sulfur" and "Sulfur" Bacteria Content 1. Media Preparation 2. Stock Solutions 3. Procedures 4. Enrichment Conditions 1. Media Preparation 1a. Medium for Phototrophic "non-sulfur"
Enrichment for Phototrophic "Non-sulfur" and "Sulfur" Bacteria Content 1. Media Preparation 2. Stock Solutions 3. Procedures 4. Enrichment Conditions 1. Media Preparation 1a. Medium for Phototrophic "non-sulfur"
Plant Genomic DNA Extraction using CTAB
 Plant Genomic DNA Extraction using CTAB Introduction The search for a more efficient means of extracting DNA of both higher quality and yield has lead to the development of a variety of protocols, however
Plant Genomic DNA Extraction using CTAB Introduction The search for a more efficient means of extracting DNA of both higher quality and yield has lead to the development of a variety of protocols, however
CHITOSAN FILM PREPARATION Instructions for laboratory experiments. Tuija Annala 29.11.2007 Rev 0
 CHITOSAN FILM PREPARATION Instructions for laboratory experiments Tuija Annala 29.11.2007 Rev 0 Table of Contents 1 Introduction...3 2 Equipment and materials...3 3 Recipe...4 2 (6) 1 INTRODUCTION Chitosan
CHITOSAN FILM PREPARATION Instructions for laboratory experiments Tuija Annala 29.11.2007 Rev 0 Table of Contents 1 Introduction...3 2 Equipment and materials...3 3 Recipe...4 2 (6) 1 INTRODUCTION Chitosan
Transfection reagent for visualizing lipofection with DNA. For ordering information, MSDS, publications and application notes see www.biontex.
 METAFECTENE FluoR Transfection reagent for visualizing lipofection with DNA For ordering information, MSDS, publications and application notes see www.biontex.com Description Cat. No. Size METAFECTENE
METAFECTENE FluoR Transfection reagent for visualizing lipofection with DNA For ordering information, MSDS, publications and application notes see www.biontex.com Description Cat. No. Size METAFECTENE
One Shot TOP10 Competent Cells
 USER GUIDE One Shot TOP10 Competent Cells Catalog Numbers C4040-10, C4040-03, C4040-06, C4040-50, and C4040-52 Document Part Number 280126 Publication Number MAN0000633 Revision A.0 For Research Use Only.
USER GUIDE One Shot TOP10 Competent Cells Catalog Numbers C4040-10, C4040-03, C4040-06, C4040-50, and C4040-52 Document Part Number 280126 Publication Number MAN0000633 Revision A.0 For Research Use Only.
PureZOL RNA Isolation Reagent Instruction Manual Catalog #732-6890
 PureZOL RNA Isolation Reagent Instruction Manual Catalog #732-6890 For technical support, call your local Bio-Rad office, or in the US, call 1-800-4BIORAD (1-800-424-6723) Table of Contents Section 1 Introduction...1
PureZOL RNA Isolation Reagent Instruction Manual Catalog #732-6890 For technical support, call your local Bio-Rad office, or in the US, call 1-800-4BIORAD (1-800-424-6723) Table of Contents Section 1 Introduction...1
