Effects of Storage Conditions on Complete Blood Cell Count Parameters
|
|
|
- Joan King
- 7 years ago
- Views:
Transcription
1 Türk Biyokimya Dergisi [Turkish Journal of Biochemistry Turk J Biochem] 2011; 36 (2) ; Research Article [Araştırma Makalesi] Yayın tarihi 15 Haziran, 2011 TurkJBiochem.com [Published online 15 June, 2011] Effects of Storage Conditions on Complete Blood Cell Count Parameters [Saklama Şartlarının Tam Kan Sayım Parametrelerine Etkisi] Turan Turhan, Sevilay Sezer, Çiğdem Yücel, Yüksel Koca Ankara Numune Education and Research Hospital, Clinical Biochemistry Laboratory, Ankara, Turkey ABSTRACT Purpose: Delineating the changes occuring in various parameters of automated complete blood cell count of normal and abnormal specimens due to prolonged storage at different conditions. Methods: The study was conducted in Ankara Numune Education and Research Hospital Emergency Biochemistry Laboratory. 117 randomly selected K 2 EDTA anticoagulated blood specimens were processed through the Coulter Gen S hematology analyzer, before and after 24 and 48 h storage at room temperature and +4 o C. Among the 117 specimens; 48 were normal, 14 were leukopenic, 27 had leukocytosis, 19 were thrombocytopenic and 9 had thrombocytosis. Results: Room temperature storage caused MCV and MPV increase in all normal and pathalogical specimens. At 48 hours, Htc and RDW were increased in all specimens except the ones with leukocytosis. MCHC was decreased in all but leukopenic ones. Refrigerated storage caused a decline in WBC and platelet counts of normal specimens. MPV was increased in the specimens with leukocytosis whereas MCHC was decreased in leukopenic ones. Specimens with thrombocytosis and thrombocytopenia were stable up to 48 hours at + 4 o C. Conclusion: In CBC measurements, it is preferred to analyze the specimens in a short time after venipuncture. The only stable parameters seem to be RBCs and hemoglobin if the measurements are carried out following a delay. Key words: Hematology, blood cell count, refrigeration Yazışma Adresi [Correspondence Address] Sevilay Sezer Emergency Laboratory, Ankara Numune Education and Research Hospital, PO Box 06300, Samanpazarı, Ankara, Turkey Tel: Fax: adres: sevilaysezer@gmail.com Registered: 10 June 2010; Accepted: 2 May 2011 [Kayıt Tarihi : 10 Haziran 2010; Kabul Tarihi : 02 Mayıs 2011] ÖZET Amaç: Ankara Numune Eğitim ve Araştırma Hastanesi Acil Biyokimya Laboratuarı nda analiz edilen normal ve patolojik hemogram kanlarının, farklı saklama koşullarının tam kan sayım parametreleri üzerindeki değişimlerini saptamak. Gereç ve Yöntemler: Çalışma Ankara Numune Eğitim ve Araştırma Hastanesi Acil Biyokimya Laboratuarı nda yapılmıştır. Rastgele seçilmiş 117 K 2 EDTA lı kan örneği, oda sıcaklığında ve +4 o C de buzdolabında 2 gün saklanarak, gelir gelmez, 24 saat ve 48 saat sonra Coulter Gen S hemogram cihazında çalışılmıştır. 117 kan örneğinden 48 i normal, 14 ü lökopenik, 27 si lökositoz, 19 u trombositopenik, 9 u trombositoz lu idi. Bulgular: Oda sıcaklığında bekletilen normal ve patolojik tüm kan örneklerinde MCV ve MPV artmıştır. Ayrıca ikinci gün, lökositozlu örnekler haricinde tüm örneklerde Htc ve RDW artmış, lökopenik örnekler haricinde ise MCHC azalmıştır. Buzdolabında bekletme ile normal kan örneklerinde WBC ve trombosit sayısı azalmış olup patolojik kan örneklerinden lökositozlu olanlarda MPV artışı, lökopenik kan örneklerinde ise MCHC azalması görülmüştür. Trombositozlu ve trombositopenik kan örnekleri buzdolabında bekletildiğinde iki gün stabilitesini korumuştur. Sonuç: Tam kan sayım analizinde tercih edilen kanın alımından kısa bir süre sonra ölçülmesidir. Eğer ölçümlerde bir gecikme olursa sadece eritrosit ve hemoglobin stabil parametreler olarak görülmektedir. Anahtar kelimeler: Hematoloji, tam kan sayımı, soğutma ISSN X (electronic) (printed)
2 Introduction Laboratory practices carried out by centralized laboratories make it critical to know the specimen storage conditions well. Transport of collected blood specimens to centralized laboratories can lead to a delay in testing for several hours. Cellular elements are known to have a limited stability in blood containing ethylenediamine tetraacetate (EDTA) [1]. Excessive delays in processing, however, might compromise the reliability of results. Limited number of studies have been conducted with all the currently used hematology analyzers for the effects of storage on analysis [2-4]. Delays in analysis may have caused different results in both normal and pathological specimens [2]. Manufacturers of automated analyzers and published literature often state that blood specimens, kept at either room temperature or at +4 o C (refrigerated) for up to 24 hours, generally reveal reliable results for Complete Blood Cell Count (CBC) [2-4]. However, these studies may not be satisfactory as the high variety of analyzers used are considered. Besides, no consensus was reached about which paremeters can still be reliable in delays over 24 hours when the analyzers were out of order due to different reasons [2-4,6]. In order to decide whether to accept or reject the aged specimen, laboratory staff needs to be familiar with the changes known to occur in blood specimens during storage [3]. In the present study, Beckman Coulter Gen S (COUL- TER Corp, Miami, USA) hematology analyzer was used in order to compare the stability of normal and pathological blood specimens collected in K 2 EDTA tubes and stored at +4 o C and +25 o C up to 48 hours and evaluated if there is an advantage of storing at +4 o C. Materials and Methods A total of 117 randomly selected K 2 EDTA anticoagulated blood specimens were processed through the Coulter Gen S hematology analyzer at Ankara Numune Education and Research Hospital Emergency Biochemistry Laboratories. Two specimens were taken from each patient into two separate tubes. Among the 117 specimens; 48 of them were normal, 14 of them were leukopenic (white cell count, <4.0 x 10 3 /µl), 27 of them had leukocytosis (white cell count, >11.0 x 10 3 /µl), 19 of them were thrombocytopenic (platelet count, <140 x 10 3 /µl) and 9 of them had thrombocytosis (platelet count, >400 x 10 3 /µl). Blood specimens were kept at either room temperature or in refrigerator prior to analysis up to 48 hours. Each sample was drawn into 4 ml capacity K 2 EDTA BD Vacutainer tubes. Each sample was analyzed at time point of <20 min (baseline measurement). After this initial measurement, each sample was divided into two separate tubes; and stored at room temperature or at +4 o C throughout the study for stability evaluations. All specimens were re-analyzed at 24 and 48 hours. The specimens stored at +4 o C were allowed to equilibrate at room temperature for 30 minutes prior to analysis. Room temperatures were measured with a standard indoor thermometer and temperature followup charts were daily filled as a part of quality control standards. The range of temperature during the study was 21 ± 3 o C. According to the recommendations of the International Committee of Haematology Standardization; the maximized storage intervals for CBC and differential count were 6 hours at o C and 24 hours at 2-6 o C [5]. With the guidance of these information, it is known that no change occurs in the first 6 hours. Actually, we carried out the study up to 72 and 96 hours. But we neither splitted the specimens to aliquotes in different tubes nor made a specimen pool. So, the device could not make proper readings due to some inadequate specimen volumes. The number of specimens with no measured value at these hours were high enough to affect our statistical evaluations. For that reason we excluded these time intervals from the study. The Coulter Gen-S uses impedance technology so as to generate a 12-part CBC and volume (impedance), conductivity, and light scatter measurements, commonly referred to as VCS technology, to generate a 5-part differential. In addition; it gives the percent and absolute number of reticulocytes making a total of 24 parameters. However, we did not carry out reticulocyte count in this study. Because this device could not make a reticulocyte count. The Software version used in the study was 4A5. Calibration and quality control of the analyzer were performed using S-cal and 3 levels of 5C control (both from Beckman Coulter), respectively according to the manufacturer s instructions. External Quality Control was carried out with Bio-Rad Laboratories EQAS system. (External Quality Assurance Services). External quality control reports are among the goals of performance. Table 1 illustrates the reference values and within-run precision values for the CBC parameters used in this study. Within-subject and between-subject CV values of analytes and desirable analytical quality specifications for total error are shown (Table 2) [7]. Statistical analysis The groups were compared for each parameter and percent changes were calculated. The values of the initial measurements were taken into account for comparisons. The percent change value obtained for each parameter was compared with the pre-determined imprecision value calculated for individual biological variation. The allowable total error (TE a ) was accepted as the target value for imprecision. Between groups differences were evaluated with paired Student s T test. Values of p less than 0.05 were considered to be significant. 166
3 Table 1. Range of initial automated CBC and differential of specimens included in the study Parameter Range Mean %* WBC, x10 9 /L NE, % LY, % MO, % EO, % BA, % NE, x10 9 /L LY, x10 9 /L MO, x10 9 /L EO, x10 9 /L BA, x10 9 /L RBC, x10 12 /L Hemoglobin, g/dl Hematocrit, % MCV, fl MCH, pg MCHC, g/dl RDW-CV, % Platelet, x10 9 /L MPV, fl * within-run precision values from analyses of medium samples Results Among the normal specimens stored at room temperature; WBC, RBC, hemoglobin and platelets were relatively stable; the MCV, RDW and MPV each increased over time at day 1 and day 2; Htc increased at day 2 while MCHC descended over time; each change was statistically significant (p<0.05). When the specimens stored at room temperature were compared with regard to target value dependent on biological variation; obtained percent change values were higher than the target values determined for imprecision for only RDW in day 1, and Htc, MCV, RDW and MPV in day 2 (Table 3). Among the WBC differentials; the number of monocytes tended to decrease over time both statistically and with regard to biological variation. (especially at day 2). The 95% CIs for the monocyte number ranged between to throughout the 2 day period. The ranges for other WBC differential counts were from to for the number of neutrophils, from to for the number of lymphocytes, from to for the number of eosinophils, and from to for the number of basophils. The same specimens stored at +4 o C exhibited lower mean WBC and platelet count at day 1 and day 2. MCV was stable for the first day, but an increase was observed at day 2. MPV and MCH were increased (p<0.05). When compared with biological variation values, only the changes in WBC and MPV in day 2 were higher than Table 2. Within-subject and between-subject CV values of analytes and desirable analytical quality specifications for total error Parameter CV i CV g TE a WBC, x10 9 /L NE, x10 9 /L LY, x10 9 /L MO, x10 9 /L EO, x10 9 /L BA, x10 9 /L RBC, x10 12 /L Hemoglobin, g/dl Hematocrit, % MCV, fl MCH, pg MCHC, g/dl RDW-CV, % Platelet, x10 9 /L MPV, fl CV i : : within-subject biological variation CV g : between-subject biological variation TE a : allowable total error the predetermined imprecision values. From the results of the differential count; a downward trend was observed for lymphocytes and neutrophiles at day 2. (Table 4). The 95% CIs during the 2 day period ranged from to for the number of neutrophils. The ranges for other WBC differentials were from to for the number of monocytes, from to for the number of lymphocytes, from to for the number of eosinophils, from to for the number of basophils. Higher values for neutrophiles at day 1 and for basophiles at day 1 and day 2 were detected according to biological variation. Among the pathological specimens; MCV and MPV increased at day 1 and 2 when bloods having leukocytosis were kept at room temperature. MCHC was decreased. An increase was detected in Htc and MPV at day 1 and day 2; and in MCV, MCHC and RDW at day 2 in reference to biological variation values. Since the refrigerator stored counterpart of the same specimens, only MPV increased at day 1 and 2 (p<0.05). Among the samples with leukocytosis kept at refrigerator Htc, MCV, MCHC, RDW ve MPV were elevated when evaluated due to biological variation values (Table 5). In leukopenic specimens kept at room temperature, MCV, RDW, Htc and MPV increased at day 2, MPV at day 1 and RDW at day 1 and day 2 increased according to biological variation but in leukopenic specimes kept at +4 o C ; only MCHC was decreased at day 2 (p<0.05), RBC, HGB, Htc, MCHC and Plt were increased at day 2, MPV was increased at day 1 with regard to biological variation (Table 6). 167
4 Table 3. Mean percent changes induced by storage of normal specimens at room temperature and refrigerated Refrigerated Room Temperature Normal (n= 48) WBC ( x 10 9 /L) RBC (10 12 /L ) HGB HCT MCV ( fl) MCH (pg) MCHC RDW PLT (x 10 9 /L) MPV (fl) Fresh 24 h CI(95%) CI(95%) CI(95%) CI(95%) 48 h 24h 48h Lower Upper Lower Upper Lower Upper Lower Upper * * 25.97^ ^ * * * -4.54^ * * * * 3.89^ * -6.47^ * ^ * * * * -9.09^ * * ^ Table 4. Mean percent changes in WBC automated differential count with storage NORMAL (n= 48) Refrigerated Room Temperature Fresh 24 h CHANGE 48 h CHANGE 24 h CHANGE 48 h CHANGE NE ( x 10 9 /L) * ^ LY ( x 10 9 /L) * MO ( x 10 9 /L) * -30^ EO ( x 10 9 /L) BA ( x 10 9 /L) ^ ^ Counts are given as absolute count: the change is the percentage of change 168
5 Table 5. Mean percent changes induced by storage of specimens having leukocytosis at room temperature and +4 o C Refrigerated Room Temperature L.cytosis (n=27) Fresh 24 h CI(95%) 48 h CI(95%) 24h CI(95%) 48h CI(95%) Lower Upper Lower Upper Lower Upper Lower Upper WBC ( x 109/L) RBC (1012/L ) HGB HCT ^ ^ ^ * -7.21^ MCV * -5.54^ * -6.56^ * * -4.86^ ( fl) MCH (pg) MCHC ^ ^ * 4.38^ RDW ^ ^ ^ PLT ^ (x 109/L) MPV ^ ^ * -8.63^ * ^ (fl) 169
6 Table 6. Mean percent changes induced by storage of specimens having leukopenic at room temperature and +4 o C Refrigerated Room Temperature L.copenic (n=14) Fresh 24 h CI(95%) 48 h CI(95%) 24h CI(95%) 48h CI(95%) Lower Upper Lower Upper Lower Upper Lower Upper WBC ( x 10 9 /L) RBC (10 12 /L ) HGB HCT MCV ( fl) MCH (pg) MCHC RDW PLT (x 10 9 /L) MPV (fl) ^ ^ ^ * * * 10.50^ * -7.38^ ^ ^ ^ * ^
7 In specimens with thrombocytosis, storage at room temperature caused an increase in Htc, MCV, RDW, MPV and a decrease in MCHC (p<0.05). MCV, MCHC, RDW were increased at day 2 and MPV was increased at day 1 and day 2 according to biological variation. No significant changes were observed when same specimens were refrigerated. When the specimens were kept at refrigerator; WBC, RBC, HGB, Hct, MCH, MCHC, Plt and MPV were increased at day 2 (Table 7). In thrombocytopenic specimens room temperature storage caused an increase in Htc, MCV, RDW ve MPV at day 2 (p <0.05), MCHC was decreased. At days 1 and 2 in RDW, and at day 2 MCHC and MPV were increased due to biological variation, but refrigerated storage showed correlation with the initial measurements. In these specimens only MPV at day 1 was increased biologically (Table 8). Among the pathological specimens; results of the differential count were not significant changes. So their results were not shown in the tables. Discussion According to the recommendations of the Committee of International Haematology Standardization; Hematology analyzer manufacturers often quote that blood specimens kept at either at room temperature or at +4 o C up to 24 hours generally reveal reliable results for complete blood count and automated differential count [5]. The recent trend towards large centralized laboratories have brought some obligations about the transfer and storage times of the blood specimens properly. Laboratories now test specimens that have been dispatched over a long distance; and the testing is often delayed by several hours or in some cases for days [1,3]. Refrigerated storage of anticoagulated blood has been noted to improve the stability of CBC. However, the number of systematic reviews including all the commonly used hematology analyzers and patological specimens along with normal ones is limited [4,8]. The studies of Wood et al. with Cell-Dyne 3500, Hedberg et al. with Cell-Dyne Saphire and Warner et al. with Coulter STKS have all claimed that refrigerated storage improves stability with these analyzers [3,8,9]. However; the results of the study of Gulati et al. with Coulter Gen S analyzer have shown that stability of normal and pathological specimens at room temperature can be acceptable with some limitations, but the same can not be claimed for the differential count [2]. In this study, we also used Coulter Gen S analyzer and investigated if there were any advantages of refrigerated storage over room temperature storage. The evaluation of the study results was not only compared statistically but according to the biological variations of defined analytical goals as well. Each of the analytes within subject CV and between subject CV results and desirable analytical quality spesifications for total error values are given in Table 2. The parameters measured directly like WBC, RBC, hemoglobin and platelet remained stable for 2 days at room temperature as stated earlier in the study by Gulati et al. The degree of change observed in WBC count of both normal and pathological specimens throughout the study could be considered acceptable for clinical applications. However, a significant decrease in WBC and platelet count was observed when the same specimens were kept at + 4 o C. This downward trend was prevented by using impedance technology instead of optical measurement in the study of Wood et al [8]. We did not have the chance to compare optical and impedance measurement techniques as the Beckman Coulter Gen S analyzer uses only impedance technology. However; we used the impedance method of Coulter Gen S to analyze the specimens stored at +4 o C; and concluded that the decrease can not be prevented both for biological variation and statistically. Unexpectedly we obtained negative percentage change in HTC and positive percentage change in MCV in normal specimens. But the situation which was found at the first 24 hours period measurement was not significant for both biological variation and statistically (Table 3). This unsignificant change is considered as the calculations of Htc and MCV are in within run presicion values. The increase in MCV in the specimens stored at room temperature reflects the swelling of RBCs. As also indicated by Wood et al. this increase is largely prevented by refrigerated storage [8]. In our study; we also concluded that the increase in MCV at room temperature was significant both biologically and statistically; whereas in refrigerated storage the increase in MCV was significant statistically while the target imprecision values for biological variation could not be passed. An increase in MPV is also observed besides MCV. But the increase in MPV was over the biological variation limits in specimens kept at refrigerator. The decrease in MCHC is caused by hematocrit increase when the hemoglobin level is still stable. As it is commonly known, MCHC is a parameter calculated by the ratio of hemoglobin to hematocrit multiplied by 100. s in the components of this equation will also lead to a change in MCHC value. But the changes detected in MCHC were not higher than the biological variation target values. In our study, it is obviously seen that especially the storage of pathological specimens at +4 o C improves the stability when compared to normal specimens. However, we argue that this result is due to the higher number of studies with normal specimens than the pathological ones. As a result, we decided to increase the number of pathological specimens so as to make the changes more meaningful. In CBC measurements, it is preferred that the specimens are analyzed in a short time after venipuncture. The only stable parameters seem to be RBCs and hemoglobin if 171
8 Table 7. Mean percent changes induced by storage of specimens having thrombocytosis at room temperature and +4 o C Refrigerated Room Temperature T.cytosis (n=19) Fresh 24 h CI(95%) 48 h CI(95%) 24h CI(95%) 48h CI(95%) Lower Upper Lower Upper Lower Upper Lower Upper WBC ^ ( x 10 9 /L) RBC ^ (10 12 /L ) HGB ^ HCT ^ * MCV * -3.99^ ( fl) MCH ^ (pg) MCHC ^ ^ RDW * -7.74^ PLT ^ (x 10 9 /L) MPV ^ ^ ^ * ^ (fl) 172
9 Table 8. Mean percent changes induced by storage of specimens having thrombocytopenic at room temperature and +4 o C Refrigerated Room Temperature T.cytopeni (n=19) Fresh 24 h CI(95%) 48 h CI(95%) 24h CI(95%) 48h CI(95%) Lower Upper Lower Upper Lower Upper Lower Upper WBC ( x 109/L) RBC (1012/L ) HGB HCT * MCV * ( fl) MCH (pg) MCHC * 2.55^ RDW ^ * -9.96^ PLT (x 109/L) MPV ^ * ^ (fl) 173
10 the measurements are carried out following a delay especially for the evaluations we made with the normal specimens. For other parameters; delayed processing affects stability. The delays and the possible effect of these delays on the results should be indicated in the reports. However, in case of necessity; for Coulter Gen-S analyzer; it can be concluded that it is possible to maintain the stability of specimens by refrigerated storage up to two days with some limitations. Conflict of interest The authors declare that they have no conflict of interest. References [1] Lawrence AC, Bevington JM, Young M. (1975) Storage of blood and the mean corpuscular volume. J Clin Pathol. 28: [2] Gulati GL, Hyland LJ, Kocher W, Schwarting R. (2002) s in automated complete blood cell count and differential leukocyte count results induced by storage of blood at room temperature. Arch Pathol Lab Med. 126: [3] Hedberg P, Lehto T. (2009) Aging stability of complete blood count and white blood cell differential parameters analyzed by Abbott CELL-DYN Sapphire hematology analyzer. Int J Lab Hematol. 31: [4] Buttarello M. (2004) Quality specification in haematology: the automated blood cell count. Clin Chim Acta. 346: [5] International Council for Standardization of Haemotology (1993). Recommendations of the International Council for Standardization in Haemotology for ethylendiamine-tetraacetic acid anticoagulation of blood for blood cell counting and sizing. Am J Clin Pathol. 100: [6] Picard F, Gicquel C, Marnet L, Guesnu M, Levy JP. (1999) Preliminary evaluation of the new hematology analyzer COULTER GEN-S in a university hospital. Clin Chem Lab Med. 37(6): [7] Ricos C, Alvarez V, Cava F, Garcia-Lario JV, Hernandez A, Jimenez CV, Minchinela J, Perich C, Simon M. (1999) Current databases on biologic variation: pros, cons and progress. Scand J Clin Lab Invest. 59: [8] Wood BL, Andrews J, Miller S, Sabath DE. (1999) Refrigerated storage improves the stability of the complete blood cell count and automated differential. Am J Clin Pathol. 112(5): [9] Warner BA, Reardon DM. (1991) A field evaluation of the Coulter STKS. Am J Clin Pathol. 95(2):
Collect and label sample according to standard protocols. Gently invert tube 8-10 times immediately after draw. DO NOT SHAKE. Do not centrifuge.
 Complete Blood Count CPT Code: CBC with Differential: 85025 CBC without Differential: 85027 Order Code: CBC with Differential: C915 Includes: White blood cell, Red blood cell, Hematocrit, Hemoglobin, MCV,
Complete Blood Count CPT Code: CBC with Differential: 85025 CBC without Differential: 85027 Order Code: CBC with Differential: C915 Includes: White blood cell, Red blood cell, Hematocrit, Hemoglobin, MCV,
QUALITY CONTROL (QC) INFORMATION AND TROUBLESHOOTING GUIDE HEMATOLOGY
 QUALITY CONTROL (QC) INFORMATION AND TROUBLESHOOTING GUIDE HEMATOLOGY This document is not intended to replace the information in your Instructions for Use Manuals (IFU). Information in the Instructions
QUALITY CONTROL (QC) INFORMATION AND TROUBLESHOOTING GUIDE HEMATOLOGY This document is not intended to replace the information in your Instructions for Use Manuals (IFU). Information in the Instructions
BC-5800. Auto Hematology Analyzer
 BC-5800 Auto Hematology Analyzer BC-5800 Auto Hematology Analyzer 5-part differentiation, 29 parameters, 2 histograms + 2 scattergrams Up to 90 samples per hour Laser scatter + Chemical dye + Flow cytometry
BC-5800 Auto Hematology Analyzer BC-5800 Auto Hematology Analyzer 5-part differentiation, 29 parameters, 2 histograms + 2 scattergrams Up to 90 samples per hour Laser scatter + Chemical dye + Flow cytometry
Clinical Laboratory Parameters for Crl:CD(SD) Rats. March, 2006. Information Prepared by Mary L.A. Giknis, Ph.D. Charles B. Clifford, D.V.M., Ph.D.
 Clinical Laboratory Parameters for Crl:CD(SD) Rats March, 2006 Information Prepared by Mary L.A. Giknis, Ph.D. Charles B. Clifford, D.V.M., Ph.D. CHARLES RIVER LABORATORIES Clinical Laboratory Parameters
Clinical Laboratory Parameters for Crl:CD(SD) Rats March, 2006 Information Prepared by Mary L.A. Giknis, Ph.D. Charles B. Clifford, D.V.M., Ph.D. CHARLES RIVER LABORATORIES Clinical Laboratory Parameters
Department of Pathology and Laboratory Medicine Capital District Health Authority Nova Scotia
 Department of Pathology and Laboratory Medicine Capital District Health Authority Nova Scotia Doc #: 10902 Section: \\Management System\PLM\Point of Care Testing\CBC Diff\ Document Owner: ES Team Lead
Department of Pathology and Laboratory Medicine Capital District Health Authority Nova Scotia Doc #: 10902 Section: \\Management System\PLM\Point of Care Testing\CBC Diff\ Document Owner: ES Team Lead
XE-2100 Automated Hematology System. Fast, Accurate, Dependable
 XE-2100 Automated Hematology System Fast, Accurate, Dependable Advanced Technology Solutions to Meet your Lab s Needs Even with these challenges, the need for hematology testing has remained steady or
XE-2100 Automated Hematology System Fast, Accurate, Dependable Advanced Technology Solutions to Meet your Lab s Needs Even with these challenges, the need for hematology testing has remained steady or
Keywords: RBC histogram, Peripheral blood smear, red cell disorders.
 Research article Automated red blood cell analysis compared with routine red blood cell morphology by smear review Dr.Poonam Radadiya*, Dr.Nandita Mehta*, Dr.Hansa Goswami***, Dr.R.N.Gonsai**** *3 rd year
Research article Automated red blood cell analysis compared with routine red blood cell morphology by smear review Dr.Poonam Radadiya*, Dr.Nandita Mehta*, Dr.Hansa Goswami***, Dr.R.N.Gonsai**** *3 rd year
Hematology Morphology Critique
 Survey Slide: History: 60-year-old female presenting with pneumonia Further Laboratory Data: Hgb : 90 g/l RBC : 2.92 10 12 /L Hct : 0.25 L/L MCV : 87 fl MCH : 30.8 pg MCHC : 355 g/l RDW : 17.7 % WBC :
Survey Slide: History: 60-year-old female presenting with pneumonia Further Laboratory Data: Hgb : 90 g/l RBC : 2.92 10 12 /L Hct : 0.25 L/L MCV : 87 fl MCH : 30.8 pg MCHC : 355 g/l RDW : 17.7 % WBC :
Pentra DX Nexus. Process efficiency in Hematology
 Pentra DX Nexus Process efficiency in Hematology Pentra DX Nexus Be productive and flexible 120 samples per hour 28 parameters Automatic reflex testing Automatic validation of results Integrated cytology
Pentra DX Nexus Process efficiency in Hematology Pentra DX Nexus Be productive and flexible 120 samples per hour 28 parameters Automatic reflex testing Automatic validation of results Integrated cytology
Yvette Marie Miller, M.D. Executive Medical Officer American Red Cross October 20, 2012 45 th Annual Great Lakes Cancer Nursing Conference Troy, MI
 Yvette Marie Miller, M.D. Executive Medical Officer American Red Cross October 20, 2012 45 th Annual Great Lakes Cancer Nursing Conference Troy, MI Overview of Hematology, http://www.nu.edu.sa/userfiles/mhmorsy/h
Yvette Marie Miller, M.D. Executive Medical Officer American Red Cross October 20, 2012 45 th Annual Great Lakes Cancer Nursing Conference Troy, MI Overview of Hematology, http://www.nu.edu.sa/userfiles/mhmorsy/h
HEMATOCRIT/HCT AND CALCULATED HEMOGLOBIN/HB
 HEMATOCRIT/HCT AND CALCULATED HEMOGLOBIN/HB Hematocrit is determined conductometrically. The measured conductivity, after correction for electrolyte concentration, is inversely related to the hematocrit.
HEMATOCRIT/HCT AND CALCULATED HEMOGLOBIN/HB Hematocrit is determined conductometrically. The measured conductivity, after correction for electrolyte concentration, is inversely related to the hematocrit.
Preparation of Blood Films
 Preparation of Blood Films Principle: Blood film enables us to evaluate WBC, RBC, and PLT morphology, also, allows us to perform differential WBC count, furthermore estimation of WBC and platelets counts
Preparation of Blood Films Principle: Blood film enables us to evaluate WBC, RBC, and PLT morphology, also, allows us to perform differential WBC count, furthermore estimation of WBC and platelets counts
How To Collect A Leukapheresis Collection
 Leukapheresis Collection Insights in Unstimulated Patients with Synchronous Metastatic Renal Cell Carcinoma Receiving a Combination of Autologous Immunotherapy (AGS-003) and Sunitinib: From the Ongoing
Leukapheresis Collection Insights in Unstimulated Patients with Synchronous Metastatic Renal Cell Carcinoma Receiving a Combination of Autologous Immunotherapy (AGS-003) and Sunitinib: From the Ongoing
Reshaping compact automation. XN-Series Automated Hematology Systems
 Reshaping compact automation XN-Series Automated Hematology Systems Small, Smart, Compact Automation It s an approach to hematology testing that s never been done before. An approach that gives all laboratories
Reshaping compact automation XN-Series Automated Hematology Systems Small, Smart, Compact Automation It s an approach to hematology testing that s never been done before. An approach that gives all laboratories
HumaCount System Line A Better Way to Connect. A Better Way to Work.
 HumaCount System Line A Better Way to Connect. A Better Way to Work. > Integration into hospital management system > 2D barcode target transfer range > CoreLab DX Hematology Thousands of HumaCounts Installed
HumaCount System Line A Better Way to Connect. A Better Way to Work. > Integration into hospital management system > 2D barcode target transfer range > CoreLab DX Hematology Thousands of HumaCounts Installed
Young fetus: site of hematopoiesis together with the liver and bone marrow. Hgb WBC >----------< Plt Hct. Retic =
 Learning Objectives At the completion of this program, the participants will be able to: 1. Identify the components of the CBC and Differential and their clinical implications. 2. Identify normal pediatric
Learning Objectives At the completion of this program, the participants will be able to: 1. Identify the components of the CBC and Differential and their clinical implications. 2. Identify normal pediatric
ABSTRACT INTRODUCTION
 Performance of the PAXgene Blood DNA Tube for the Collection, Transport, and Storage of Whole Blood and the Purification of DNA Using the QIAsymphony Instrument L. Rainen 1, K. Guenther 2, E. Provencher
Performance of the PAXgene Blood DNA Tube for the Collection, Transport, and Storage of Whole Blood and the Purification of DNA Using the QIAsymphony Instrument L. Rainen 1, K. Guenther 2, E. Provencher
And Now, Presenting...
 Presentation and Handling of Clinical Laboratory Data From Test Tube to Table Randall K. Carlson, Wilmington, DE and Nate Freimark, Lakewood, NJ Omnicare Clinical Research. Inc. INTRODUCTION In human clinical
Presentation and Handling of Clinical Laboratory Data From Test Tube to Table Randall K. Carlson, Wilmington, DE and Nate Freimark, Lakewood, NJ Omnicare Clinical Research. Inc. INTRODUCTION In human clinical
The IFCC HbA 2 standardization
 The IFCC HbA 2 standardization program Andrea Mosca Centro Interdipartimentale per la Riferibilità Metrologica in Medicina di Laboratorio (CIRME) Dip. di Scienze e Tecnologie Biomediche Università degli
The IFCC HbA 2 standardization program Andrea Mosca Centro Interdipartimentale per la Riferibilità Metrologica in Medicina di Laboratorio (CIRME) Dip. di Scienze e Tecnologie Biomediche Università degli
Systems of Measurement
 Systems of Measurement Course Principles of Health Science Unit X Vital Signs Essential Question What s math got to do with it? TEKS 130.202 (c)1a, 1B, 1D Prior Student Learning Basic understanding of
Systems of Measurement Course Principles of Health Science Unit X Vital Signs Essential Question What s math got to do with it? TEKS 130.202 (c)1a, 1B, 1D Prior Student Learning Basic understanding of
Human Clinical Study for Free Testosterone & Muscle Mass Boosting
 Human Clinical Study for Free Testosterone & Muscle Mass Boosting GE Nutrients, Inc. 920 E. Orangethorpe Avenue, Suite B Anaheim, California 92801, USA Phone: +1-714-870-8723 Fax: +1-732-875-0306 Contact
Human Clinical Study for Free Testosterone & Muscle Mass Boosting GE Nutrients, Inc. 920 E. Orangethorpe Avenue, Suite B Anaheim, California 92801, USA Phone: +1-714-870-8723 Fax: +1-732-875-0306 Contact
RDW-- Interpreting the Full Blood Count
 RDW-- Interpreting the Full Blood Count The most important components of a Full Blood Count report are, of course, the Haemoglobin, the White Cell Count and Differential and the Platelet Count. However,
RDW-- Interpreting the Full Blood Count The most important components of a Full Blood Count report are, of course, the Haemoglobin, the White Cell Count and Differential and the Platelet Count. However,
Blood Testing Protocol
 Blood Testing Protocol IAAF Blood Testing Protocol Two main focus areas: Blood sampling for the detection of Prohibited Substances and Prohibited Methods, e.g. CERA or other ESAs, hgh, blood transfusion,
Blood Testing Protocol IAAF Blood Testing Protocol Two main focus areas: Blood sampling for the detection of Prohibited Substances and Prohibited Methods, e.g. CERA or other ESAs, hgh, blood transfusion,
Hematology Case Studies: Platelets
 California Association for Medical Laboratory Technology Distance Learning Program Hematology Case Studies: Platelets by Helen M. Sowers, MA, CLS Dept of Biological Science (ret.) California State University,
California Association for Medical Laboratory Technology Distance Learning Program Hematology Case Studies: Platelets by Helen M. Sowers, MA, CLS Dept of Biological Science (ret.) California State University,
MANUAL PLATELET COUNT
 MANUAL PLATELET COUNT LAB OBJECTIVE The student will be able to perform, within K20% accuracy as compared to the automated result, five manual platelet counts using the Unopette system. and the light microscope.
MANUAL PLATELET COUNT LAB OBJECTIVE The student will be able to perform, within K20% accuracy as compared to the automated result, five manual platelet counts using the Unopette system. and the light microscope.
Six Sigma Metric Analysis for Analytical Testing Processes
 Six Sigma Metric Analysis for Analytical Testing Processes Sten Westgard, MS, Westgard QC Introduction Laboratories seek objective assessment and comparison of analytical methods and instrumentation performance.
Six Sigma Metric Analysis for Analytical Testing Processes Sten Westgard, MS, Westgard QC Introduction Laboratories seek objective assessment and comparison of analytical methods and instrumentation performance.
MANUAL WBC LAB OBJECTIVE
 MANUAL WBC LAB OBJECTIVE The student will be able to perform, within K20% accuracy as compared to the automated result, five manual white blood cell counts using the Unopette system. PRINCIPLE Free-flowing
MANUAL WBC LAB OBJECTIVE The student will be able to perform, within K20% accuracy as compared to the automated result, five manual white blood cell counts using the Unopette system. PRINCIPLE Free-flowing
Clinical Laboratory Instrumentation
 Clinical Laboratory Instrumentation Responsible for analyzing patient specimens to provide information to aid in diagnosis of disease to evaluate effectiveness of therapy Major sections: Chemistry lab
Clinical Laboratory Instrumentation Responsible for analyzing patient specimens to provide information to aid in diagnosis of disease to evaluate effectiveness of therapy Major sections: Chemistry lab
R&D Systems Hematology Products
 R&D Systems Hematology Products Controls Calibrators Linearity Materials Quality Control Program TABLE OF CONTENTS R&D SYSTEMS HEMATOLOGY PRODUCTS R&D Systems, Inc., a specialty manufacturer of biological
R&D Systems Hematology Products Controls Calibrators Linearity Materials Quality Control Program TABLE OF CONTENTS R&D SYSTEMS HEMATOLOGY PRODUCTS R&D Systems, Inc., a specialty manufacturer of biological
BLOOD COUNT INTERPRETATION. J. Bačovský
 BLOOD COUNT INTERPRETATION J. Bačovský Sysmex XE 5000 + software + staining ) CellaVision DM 96 (automatic digitál morphology of peripheral blood smears Hematology Cell Counter Counts cells by electronic
BLOOD COUNT INTERPRETATION J. Bačovský Sysmex XE 5000 + software + staining ) CellaVision DM 96 (automatic digitál morphology of peripheral blood smears Hematology Cell Counter Counts cells by electronic
Content Sheet 7-1: Overview of Quality Control for Quantitative Tests
 Content Sheet 7-1: Overview of Quality Control for Quantitative Tests Role in quality management system Quality Control (QC) is a component of process control, and is a major element of the quality management
Content Sheet 7-1: Overview of Quality Control for Quantitative Tests Role in quality management system Quality Control (QC) is a component of process control, and is a major element of the quality management
Visual Acuity. Hearing. Height and Weight. Blood Pressure MEASURED VALUE
 TEST ITEM DESCRIPTION STANDARD LEVEL Standard level varies among different examination methods. Please check with your medical facility about normal level. MEASURED VALUE Visual Acuity You look at rings
TEST ITEM DESCRIPTION STANDARD LEVEL Standard level varies among different examination methods. Please check with your medical facility about normal level. MEASURED VALUE Visual Acuity You look at rings
PRINCIPLE. REF 442635 (200 tests/cartridge) REF 476836 (400 tests/cartridge) ANNUAL REVIEW Reviewed by: Date. Date INTENDED USE
 SYNCHRON System(s) Chemistry Information Sheet Copyright 2010 Beckman Coulter, Inc. Creatine Kinase REF 442635 (200 tests/cartridge) REF 476836 (400 tests/cartridge) For In Vitro Diagnostic Use ANNUAL
SYNCHRON System(s) Chemistry Information Sheet Copyright 2010 Beckman Coulter, Inc. Creatine Kinase REF 442635 (200 tests/cartridge) REF 476836 (400 tests/cartridge) For In Vitro Diagnostic Use ANNUAL
Georgia Northwestern Technical College Practical Nursing Program CLINICAL DAILY ASSESSMENT WORKSHEET FOR MODULES I-IV STUDENT: CLINICAL INSTRUCTOR:
 Georgia Northwestern Technical College Practical Nursing Program CLINICAL DAILY ASSESSMENT WORKSHEET FOR MODULES I-IV STUDENT: CLINICAL INSTRUCTOR: CLINICAL UNIT: ASSIGNMENT DATES: PATIENT (last initial):
Georgia Northwestern Technical College Practical Nursing Program CLINICAL DAILY ASSESSMENT WORKSHEET FOR MODULES I-IV STUDENT: CLINICAL INSTRUCTOR: CLINICAL UNIT: ASSIGNMENT DATES: PATIENT (last initial):
Interpretation of Laboratory Values
 Interpretation of Laboratory Values Konrad J. Dias PT, DPT, CCS Overview Electrolyte imbalances Renal Function Tests Complete Blood Count Coagulation Profile Fluid imbalance Sodium Electrolyte Imbalances
Interpretation of Laboratory Values Konrad J. Dias PT, DPT, CCS Overview Electrolyte imbalances Renal Function Tests Complete Blood Count Coagulation Profile Fluid imbalance Sodium Electrolyte Imbalances
Hepatitis and Retrovirus. LIAISON XL Accurate detection of HIV infection. HIV Ab/Ag FOR OUTSIDE THE US AND CANADA ONLY
 Hepatitis and Retrovirus LIAISON XL Accurate detection of HIV infection HIV Ab/Ag FOR OUTSIDE THE US AND CANADA ONLY LIAISON XL HIV Ab/Ag is Your solution LIAISON XL murex HIV Ab/Ag main features The LIAISON
Hepatitis and Retrovirus LIAISON XL Accurate detection of HIV infection HIV Ab/Ag FOR OUTSIDE THE US AND CANADA ONLY LIAISON XL HIV Ab/Ag is Your solution LIAISON XL murex HIV Ab/Ag main features The LIAISON
Abstract. NHL Journal of Medical Sciences/Jan 2014/Vol 3/Issue 1 32
 Abstract Research article Impedance platelet count in severe microcytosis-study of 161 patients N J Ninama*, Nirali K Shah** *Assistant Professor, Dept of Physiology; **Resident, Dept of Pathology Smt
Abstract Research article Impedance platelet count in severe microcytosis-study of 161 patients N J Ninama*, Nirali K Shah** *Assistant Professor, Dept of Physiology; **Resident, Dept of Pathology Smt
The comparison of blood parameters between morning and evening exercise
 Available online at www.pelagiaresearchlibrary.com European Journal of Experimental Biology, 2013, 3(1):559-563 ISSN: 2248 9215 CODEN (USA): EJEBAU The comparison of blood parameters between morning and
Available online at www.pelagiaresearchlibrary.com European Journal of Experimental Biology, 2013, 3(1):559-563 ISSN: 2248 9215 CODEN (USA): EJEBAU The comparison of blood parameters between morning and
For In Vitro Diagnostic Use
 SYNCHRON System(s) Chemistry Information Sheet Creatine Kinase REF (200 tests/cartridge) 442635 REF (400 tests/cartridge) 476836 For In Vitro Diagnostic Use ANNUAL REVIEW Reviewed by: Reviewed by: Date
SYNCHRON System(s) Chemistry Information Sheet Creatine Kinase REF (200 tests/cartridge) 442635 REF (400 tests/cartridge) 476836 For In Vitro Diagnostic Use ANNUAL REVIEW Reviewed by: Reviewed by: Date
BIOCHEMISTRY DEPARTMENT
 ALBUMIN 33 46 g/l ALK PHOSPHATASE Up to 11 months Male 133 426 U/L Female 196 553 U/L 11+ months 2 years Male 133 287 U/L Female 196 510 U/L 2+ 8 years Male 133 287 U/L Female 196 426 U/L 8+ 11 years Male
ALBUMIN 33 46 g/l ALK PHOSPHATASE Up to 11 months Male 133 426 U/L Female 196 553 U/L 11+ months 2 years Male 133 287 U/L Female 196 510 U/L 2+ 8 years Male 133 287 U/L Female 196 426 U/L 8+ 11 years Male
for Leucocyte Immunophenotyping Leukaemia Diagnosis Interpretation All Participants Date Issued: 08-September-2014 Closing Date: 26-September-2014
 for Leucocyte Immunophenotyping Leukaemia Interpretation All Participants Participant: 4xxxx Trial No: 141502 Date Issued: 08-September-2014 Closing Date: 26-September-2014 Trial Comments This was an electronic
for Leucocyte Immunophenotyping Leukaemia Interpretation All Participants Participant: 4xxxx Trial No: 141502 Date Issued: 08-September-2014 Closing Date: 26-September-2014 Trial Comments This was an electronic
Clinical Laboratory Parameters For Crl:WI(Han)Rats
 Clinical Laboratory Parameters For Crl:WI(Han)Rats INTRODUCTION... 2 PURPOSE... 2 COMMON STUDY PARAMETERS... 2 DATA PRESENTED... 3 NOTES AND ABBREVIATIONS... 3 ACKNOWLEDGMENTS... 4 REFERENCES... 4 REQUEST
Clinical Laboratory Parameters For Crl:WI(Han)Rats INTRODUCTION... 2 PURPOSE... 2 COMMON STUDY PARAMETERS... 2 DATA PRESENTED... 3 NOTES AND ABBREVIATIONS... 3 ACKNOWLEDGMENTS... 4 REFERENCES... 4 REQUEST
Pathology No: SHS-CASE No. Date of Procedure: Client Name Address
 TEL #: (650) 725-5604 FAX #: (650) 725-7409 Med. Rec. No.: Date of Procedure: Sex: A ge: Date Received: Date of Birth: Account No.: Physician(s): Client Name Address SPECIMEN SUBMITTED: LEFT PIC BONE MARROW,
TEL #: (650) 725-5604 FAX #: (650) 725-7409 Med. Rec. No.: Date of Procedure: Sex: A ge: Date Received: Date of Birth: Account No.: Physician(s): Client Name Address SPECIMEN SUBMITTED: LEFT PIC BONE MARROW,
 Blood Sticky, opaque fluid with a metallic taste (Fe 2+ ) Varies from scarlet (P O2 = 100) to dark red (P O2 = 40) ph is between 7.35 and 7.45 Average volume in an adult is 5 L (7% of body weight) 2 L
Blood Sticky, opaque fluid with a metallic taste (Fe 2+ ) Varies from scarlet (P O2 = 100) to dark red (P O2 = 40) ph is between 7.35 and 7.45 Average volume in an adult is 5 L (7% of body weight) 2 L
EXERCISE 5: ERYTHROCYTES SEDIMENTATION RATE - ESR, SED RATE
 EXERCISE 5: ERYTHROCYTES SEDIMENTATION RATE - ESR, SED RATE Textbook: Skills: None 10 points Objectives: 1. State the principle of the Erythrocytes Sedimentation Rate - ESR. 2. List two factors which may
EXERCISE 5: ERYTHROCYTES SEDIMENTATION RATE - ESR, SED RATE Textbook: Skills: None 10 points Objectives: 1. State the principle of the Erythrocytes Sedimentation Rate - ESR. 2. List two factors which may
Whole Blood Flow Cytometry
 Whole Blood Flow Cytometry y Nailin Li Department t of Medicine, i Clinical i l Pharmacology Unit Karolinska Institute/University Hospital, 171 76 Stockholm Department of Pathology & Pathophysiology Zhejiang
Whole Blood Flow Cytometry y Nailin Li Department t of Medicine, i Clinical i l Pharmacology Unit Karolinska Institute/University Hospital, 171 76 Stockholm Department of Pathology & Pathophysiology Zhejiang
Phlebotomy Handbook Blood Collection Essentials Seventh Edition
 Phlebotomy Handbook Blood Collection Essentials Seventh Edition Diana Garza Kathleen Becan-McBride Chapter Four The Cardiovascular System Introduction Circulatory system is a transport system. Contributes
Phlebotomy Handbook Blood Collection Essentials Seventh Edition Diana Garza Kathleen Becan-McBride Chapter Four The Cardiovascular System Introduction Circulatory system is a transport system. Contributes
James O. Westgard, PhD, 1,2 and Sten A. Westgard, MS 2. Abstract
 Clinical Chemistry / ANALYTIC QUALITY TODAY The Quality of Laboratory Testing Today An Assessment of σ Metrics for Analytic Quality Using Performance Data From Proficiency Testing Surveys and the CLIA
Clinical Chemistry / ANALYTIC QUALITY TODAY The Quality of Laboratory Testing Today An Assessment of σ Metrics for Analytic Quality Using Performance Data From Proficiency Testing Surveys and the CLIA
Laboratory Studies in the Diagnosis of Iron Deficiency, Latent Iron Deficiency and Iron Deficient Erythropoiesis
 Laboratory Studies in the Diagnosis of Iron Deficiency, Latent Iron Deficiency and Iron Deficient Erythropoiesis General Comments The laboratory studies listed below are helpful in the diagnosis and management
Laboratory Studies in the Diagnosis of Iron Deficiency, Latent Iron Deficiency and Iron Deficient Erythropoiesis General Comments The laboratory studies listed below are helpful in the diagnosis and management
COULTER A C T diff COULTER A C T diff 2
 COULTER A C T diff COULTER A C T diff 2 HEMATOLOGY ANALYZER ROUTINE OPERATION EASY REFERENCE GUIDE Beckman Coulter, Inc. INTRODUCTION READ ALL PRODUCT MANUALS BEFORE ATTEMPTING TO OPERATE INSTRUMENT This
COULTER A C T diff COULTER A C T diff 2 HEMATOLOGY ANALYZER ROUTINE OPERATION EASY REFERENCE GUIDE Beckman Coulter, Inc. INTRODUCTION READ ALL PRODUCT MANUALS BEFORE ATTEMPTING TO OPERATE INSTRUMENT This
Effect of Blood Collection Technique in Mice on Clinical Pathology Parameters
 HUMAN GENE THERAPY 13:155 162 (January 1, 2002) Mary Ann Liebert, Inc. Effect of Blood Collection Technique in Mice on Clinical Pathology Parameters MICHAEL A. SCHNELL, 1,2 CHRISTINE HARDY, 3 MELANIE HAWLEY,
HUMAN GENE THERAPY 13:155 162 (January 1, 2002) Mary Ann Liebert, Inc. Effect of Blood Collection Technique in Mice on Clinical Pathology Parameters MICHAEL A. SCHNELL, 1,2 CHRISTINE HARDY, 3 MELANIE HAWLEY,
USING CLSI GUIDELINES TO PERFORM METHOD EVALUATION STUDIES IN YOUR LABORATORY
 USING CLSI GUIDELINES TO PERFORM METHOD EVALUATION STUDIES IN YOUR LABORATORY Breakout Session 3B Tuesday, May 1 8:30 10 am James Blackwood, MS, CLSI David D. Koch, PhD, FACB, DABCC, Pathology & Laboratory
USING CLSI GUIDELINES TO PERFORM METHOD EVALUATION STUDIES IN YOUR LABORATORY Breakout Session 3B Tuesday, May 1 8:30 10 am James Blackwood, MS, CLSI David D. Koch, PhD, FACB, DABCC, Pathology & Laboratory
Laboratory 12 Blood Cells
 Laboratory 12 Blood Cells Objectives: Identify microscopically each of the following: erythrocytes (red blood cells or RBCs), the five types of leukocytes (white blood cells or WBCs), and thrombocytes
Laboratory 12 Blood Cells Objectives: Identify microscopically each of the following: erythrocytes (red blood cells or RBCs), the five types of leukocytes (white blood cells or WBCs), and thrombocytes
THE EFFECT OF α-thalassemia ON CORD BLOOD RED CELL INDICES AND INTERACTION WITH SICKLE CELL GENE
 THE EFFECT OF α-thalassemia ON CORD BLOOD RED CELL INDICES AND INTERACTION WITH SICKLE CELL GENE Mohammad I. Quadri, MD, MNAMS, PhD; Sherief I.A.M. Islam, FRCPath; Zaki Nasserullah, MD Background: α-thalassemia
THE EFFECT OF α-thalassemia ON CORD BLOOD RED CELL INDICES AND INTERACTION WITH SICKLE CELL GENE Mohammad I. Quadri, MD, MNAMS, PhD; Sherief I.A.M. Islam, FRCPath; Zaki Nasserullah, MD Background: α-thalassemia
NEW YORK STATE CYTOHEMATOLOGY PROFICIENCY TESTING PROGRAM Glass Slide Critique ~ November 2010
 NEW YORK STATE CYTOHEMATOLOGY PROFICIENCY TESTING PROGRAM Glass Slide Critique ~ November 2010 Slide 081 Available data: 72 year-old female Diagnosis: MDS to AML WBC 51.0 x 10 9 /L RBC 3.39 x 10 12 /L
NEW YORK STATE CYTOHEMATOLOGY PROFICIENCY TESTING PROGRAM Glass Slide Critique ~ November 2010 Slide 081 Available data: 72 year-old female Diagnosis: MDS to AML WBC 51.0 x 10 9 /L RBC 3.39 x 10 12 /L
Exercise 9: Blood. Readings: Silverthorn 5 th ed, 547 558, 804 805; 6 th ed, 545 557, 825 826.
 Exercise 9: Blood Readings: Silverthorn 5 th ed, 547 558, 804 805; 6 th ed, 545 557, 825 826. Blood Typing The membranes of human red blood cells (RBCs) contain a variety of cell surface proteins called
Exercise 9: Blood Readings: Silverthorn 5 th ed, 547 558, 804 805; 6 th ed, 545 557, 825 826. Blood Typing The membranes of human red blood cells (RBCs) contain a variety of cell surface proteins called
CHEMISTRY. Updated Monday June 25, 2007. CHEMISTRY - BASIC PANEL * may be asked to fast for this test. Range M 23-29 F 23-29
 The Center for Family Medicine - Minot laboratory provides many laboratory tests for our patients. A list of the laboratory tests performed, expected ranges and a brief explanation of the tests are listed
The Center for Family Medicine - Minot laboratory provides many laboratory tests for our patients. A list of the laboratory tests performed, expected ranges and a brief explanation of the tests are listed
Validation and Calibration. Definitions and Terminology
 Validation and Calibration Definitions and Terminology ACCEPTANCE CRITERIA: The specifications and acceptance/rejection criteria, such as acceptable quality level and unacceptable quality level, with an
Validation and Calibration Definitions and Terminology ACCEPTANCE CRITERIA: The specifications and acceptance/rejection criteria, such as acceptable quality level and unacceptable quality level, with an
Hepatitis C. Laboratory Tests and Hepatitis C
 Hepatitis C Laboratory Tests and Hepatitis C If you have hepatitis C, your doctor will use laboratory tests to check your health. This handout will help you understand what the major tests are and what
Hepatitis C Laboratory Tests and Hepatitis C If you have hepatitis C, your doctor will use laboratory tests to check your health. This handout will help you understand what the major tests are and what
510(k) SUBSTANTIAL EQUIVALENCE DETERMINATION DECISION SUMMARY
 510(k) SUBSTANTIAL EQUIVALENCE DETERMINATION DECISION SUMMARY A. 510(k) Number: k103555 B. Purpose for Submission: New submission C. Measurand: Platelet aggregation D. Type of Test: Platelet aggregometer
510(k) SUBSTANTIAL EQUIVALENCE DETERMINATION DECISION SUMMARY A. 510(k) Number: k103555 B. Purpose for Submission: New submission C. Measurand: Platelet aggregation D. Type of Test: Platelet aggregometer
Hematology Reagents and Controls Our products for your critical process
 Part of a pure process Hematology Reagents and Controls Our products for your critical process 2 Reagents you can count on Mallinckrodt Baker s chemical expertise and 30 years of experience are the foundation
Part of a pure process Hematology Reagents and Controls Our products for your critical process 2 Reagents you can count on Mallinckrodt Baker s chemical expertise and 30 years of experience are the foundation
Additional Blood Tests
 Additional Blood Tests CBC W/Auto Diff The CBC is used as a broad screening test to check for such disorders as anemia, infection, and many other diseases. It is actually panel of tests that examines different
Additional Blood Tests CBC W/Auto Diff The CBC is used as a broad screening test to check for such disorders as anemia, infection, and many other diseases. It is actually panel of tests that examines different
EDUCATIONAL COMMENTARY RED BLOOD CELLS AND WHITE BLOOD CELLS IN URINALYSIS
 URINALYSIS Educational commentary is provided through our affiliation with the American Society for Clinical Pathology (ASCP). To obtain FREE CME/CMLE credits click on Earn CE Credits under Continuing
URINALYSIS Educational commentary is provided through our affiliation with the American Society for Clinical Pathology (ASCP). To obtain FREE CME/CMLE credits click on Earn CE Credits under Continuing
Selected Topics in Electrical Engineering: Flow Cytometry Data Analysis
 Selected Topics in Electrical Engineering: Flow Cytometry Data Analysis Bilge Karaçalı, PhD Department of Electrical and Electronics Engineering Izmir Institute of Technology Outline Compensation and gating
Selected Topics in Electrical Engineering: Flow Cytometry Data Analysis Bilge Karaçalı, PhD Department of Electrical and Electronics Engineering Izmir Institute of Technology Outline Compensation and gating
Blood Plasma Sample Collection and Handling for Proteomics Analysis - A guide to obtain optimal plasma samples -
 Blood Plasma Sample Collection and Handling for Proteomics Analysis - A guide to obtain optimal plasma samples - 1 Introduction Blood can be regarded as a complex liquid tissue that comprises cells and
Blood Plasma Sample Collection and Handling for Proteomics Analysis - A guide to obtain optimal plasma samples - 1 Introduction Blood can be regarded as a complex liquid tissue that comprises cells and
LABORATORY 2 Staining and processing of blood parasites Differential counts of leukocytes (giemsa stains)
 LABORATORY 2 Staining and processing of blood parasites Differential counts of leukocytes (giemsa stains) SPECIMENS TO BE STAINED 1. Thin and thick blood smears from a patient returning from Africa - case
LABORATORY 2 Staining and processing of blood parasites Differential counts of leukocytes (giemsa stains) SPECIMENS TO BE STAINED 1. Thin and thick blood smears from a patient returning from Africa - case
NORIP - Haematology Preliminary results from the Nordic Reference Interval Project
 NORIP - Haematology Preliminary results from the Nordic Reference Interval Project 2003-06-26 Arne Mårtensson Clinical Biochemist, EQUALIS, Sweden Working group NORIP Haematology: Eeva-Riitta Savolainen,
NORIP - Haematology Preliminary results from the Nordic Reference Interval Project 2003-06-26 Arne Mårtensson Clinical Biochemist, EQUALIS, Sweden Working group NORIP Haematology: Eeva-Riitta Savolainen,
KEY CHAPTER 14: BLOOD OBJECTIVES. 1. Describe blood according to its tissue type and major functions.
 KEY CHAPTER 14: BLOOD OBJECTIVES 1. Describe blood according to its tissue type and major functions. TISSUE TYPE? MAJOR FUNCTIONS connective Transport Maintenance of body temperature 2. Define the term
KEY CHAPTER 14: BLOOD OBJECTIVES 1. Describe blood according to its tissue type and major functions. TISSUE TYPE? MAJOR FUNCTIONS connective Transport Maintenance of body temperature 2. Define the term
Reference Range: 0.5-1.6 mmol/l (arterial) 0.7-2.1 mmol/l (venous) CPT Code: 83605
 LACTIC ACID Fasting, arterial specimen preferred. Please note whether arterial or venous. 0.5 ml heparinized plasma. Green top or PST must be drawn, placed on ice, and spun within 15 minutes. Immediately
LACTIC ACID Fasting, arterial specimen preferred. Please note whether arterial or venous. 0.5 ml heparinized plasma. Green top or PST must be drawn, placed on ice, and spun within 15 minutes. Immediately
A COMPARATIVE STUDY OF SEVEN IN-HOUSE AND TWO LABORATORY HEMATOLOGY INSTRUMENTS
 A COMPARATIVE STUDY OF SEVEN IN-HOUSE AND TWO LABORATORY HEMATOLOGY INSTRUMENTS MARTINA BECKER INAUGURAL-DISSERTATION zur Erlangung des Grades eines Dr. med. vet. beim Fachbereich Veterinärmedizin der
A COMPARATIVE STUDY OF SEVEN IN-HOUSE AND TWO LABORATORY HEMATOLOGY INSTRUMENTS MARTINA BECKER INAUGURAL-DISSERTATION zur Erlangung des Grades eines Dr. med. vet. beim Fachbereich Veterinärmedizin der
UniCare Professional Reimbursement Policy
 UniCare Professional Reimbursement Policy Subject: Laboratory and Venipuncture Services Policy #: UniCare 0029 Adopted: 02/02/2010 Effective: 04/05/2016 Coverage is subject to the terms, conditions, and
UniCare Professional Reimbursement Policy Subject: Laboratory and Venipuncture Services Policy #: UniCare 0029 Adopted: 02/02/2010 Effective: 04/05/2016 Coverage is subject to the terms, conditions, and
Biochemistry Validation Form
 Biochemistry Validation Form Method: Piccolo Xpress Chemistry 13 panel Manufacturer: Abaxis Cat no: 400-0029 CE marked: Yes: x No: Location of bench book: X:\Bio\Patricia\Piccollo Xpress evaluation (August
Biochemistry Validation Form Method: Piccolo Xpress Chemistry 13 panel Manufacturer: Abaxis Cat no: 400-0029 CE marked: Yes: x No: Location of bench book: X:\Bio\Patricia\Piccollo Xpress evaluation (August
INMA LABORATORY MANUAL
 INMA LABORATORY MANUAL NUTRITION BIOMARKERS Cohorte-VALENCIA This manual describes the collection, separation, storage and transport of blood samples for the INMA study. Careful collection of samples is
INMA LABORATORY MANUAL NUTRITION BIOMARKERS Cohorte-VALENCIA This manual describes the collection, separation, storage and transport of blood samples for the INMA study. Careful collection of samples is
STAINING OF PBF AND INTERPRETATION OF NORMAL AND ABNORMAL RED CELL MORPHOLOGY
 9 STAINING OF PBF AND INTERPRETATION OF NORMAL AND ABNORMAL RED CELL MORPHOLOGY 9.1 INTRODUCTION A peripheral blood smear (peripheral blood film) is a glass microscope slide coated on one side with a thin
9 STAINING OF PBF AND INTERPRETATION OF NORMAL AND ABNORMAL RED CELL MORPHOLOGY 9.1 INTRODUCTION A peripheral blood smear (peripheral blood film) is a glass microscope slide coated on one side with a thin
PROCEDURE COVERSHEET. Document Section: CHEMISTRY Author: JOANNE WONG Date Authored: JAN 2011. In Use Date: 2/1/2011 Discontinued Date:
 PROCEDURE COVERSHEET Procedure Title: ph (Urine, Body Fluid, Platelet Samples) Document Section: CHEMISTRY Author: JOANNE WONG Date Authored: JAN 2011 Laboratory Director: TED KURTZ, MD (PARNASSUS & CHINA
PROCEDURE COVERSHEET Procedure Title: ph (Urine, Body Fluid, Platelet Samples) Document Section: CHEMISTRY Author: JOANNE WONG Date Authored: JAN 2011 Laboratory Director: TED KURTZ, MD (PARNASSUS & CHINA
Cell Counting Using the BD Accuri C6 Flow Cytometer
 Cell Counting Using the BD Accuri C6 Flow Cytometer Koen Verbrugghe, PhD Marketing Applications Specialist BD Biosciences 23-14763- The BD Accuri C6 Flow Cytometer System An affordable, full-featured,
Cell Counting Using the BD Accuri C6 Flow Cytometer Koen Verbrugghe, PhD Marketing Applications Specialist BD Biosciences 23-14763- The BD Accuri C6 Flow Cytometer System An affordable, full-featured,
Standardization of hemoglobin A 1c : myth or reality? Andrea Mosca
 Standardization of hemoglobin A 1c : myth or reality? Andrea Mosca Dept. Pathophysiology and Transplantation Centre for Traceability in Laboratory Medicine (CIRME) University of Milano, Milano (IT) contents
Standardization of hemoglobin A 1c : myth or reality? Andrea Mosca Dept. Pathophysiology and Transplantation Centre for Traceability in Laboratory Medicine (CIRME) University of Milano, Milano (IT) contents
IDEXX LaserCyte Hematology Analyzer Case Study Book and Technical Guide
 IDEXX LaserCyte Hematology Analyzer Case Study Book and Technical Guide Fold out for normal dot plot comparison Feline Canine Equine 496 496 496 12288 16384 496 12288 16384 Red Blood Cells White Blood
IDEXX LaserCyte Hematology Analyzer Case Study Book and Technical Guide Fold out for normal dot plot comparison Feline Canine Equine 496 496 496 12288 16384 496 12288 16384 Red Blood Cells White Blood
HAEMATOLOGY LABORATORY
 HAEMATOLOGY LABORATORY Head of Unit : Dr Raudhawati Osman Phone : +603-26155281 Email : raudhaosman@yahoo.com 1. INTRODUCTION The Haematology Unit, Department of Pathology, HKL provides tertiary diagnostic
HAEMATOLOGY LABORATORY Head of Unit : Dr Raudhawati Osman Phone : +603-26155281 Email : raudhaosman@yahoo.com 1. INTRODUCTION The Haematology Unit, Department of Pathology, HKL provides tertiary diagnostic
Laboratory Tests Interpretation
 Laboratory Tests Interpretation By NURSES REASEARCH PUBLICATION Continuing Education 2001 ALL RIGHTS RESERVED. ALL RIGHTS RESERVED. No part of this material may be reprinted, reproduced, transmitted, stored
Laboratory Tests Interpretation By NURSES REASEARCH PUBLICATION Continuing Education 2001 ALL RIGHTS RESERVED. ALL RIGHTS RESERVED. No part of this material may be reprinted, reproduced, transmitted, stored
Medical Necessity and Advanced Beneficiary Notice (ABN) Policy and Form
 Medical Necessity and Advanced Beneficiary Notice (ABN) Policy and Form Billings Clinic Laboratory believes all health-care providers should order only appropriate tests for the diagnosis and treatment
Medical Necessity and Advanced Beneficiary Notice (ABN) Policy and Form Billings Clinic Laboratory believes all health-care providers should order only appropriate tests for the diagnosis and treatment
Determination of Hematologic Reference Values of Neonates in Mashhad - Iran
 ULUSLARARASı HEMATOLOJI-ONKOLOJI DERGISI ARTICLE International Journal of Hematology and Oncology Determination of Hematologic Reference Values of Neonates in Mashhad - Iran Mohammad R. KERAMATI 1, Ashraf
ULUSLARARASı HEMATOLOJI-ONKOLOJI DERGISI ARTICLE International Journal of Hematology and Oncology Determination of Hematologic Reference Values of Neonates in Mashhad - Iran Mohammad R. KERAMATI 1, Ashraf
LABORATORY PROCEDURE MANUAL
 Exercise 2 Donor Physical Exam Textbook: Skills: 20 points Objectives 1. For each of the following state the acceptable values for blood donation using appropriate units: a. Pulse b. Temperature (Celsius
Exercise 2 Donor Physical Exam Textbook: Skills: 20 points Objectives 1. For each of the following state the acceptable values for blood donation using appropriate units: a. Pulse b. Temperature (Celsius
BioVisualization: Enhancing Clinical Data Mining
 BioVisualization: Enhancing Clinical Data Mining Even as many clinicians struggle to give up their pen and paper charts and spreadsheets, some innovators are already shifting health care information technology
BioVisualization: Enhancing Clinical Data Mining Even as many clinicians struggle to give up their pen and paper charts and spreadsheets, some innovators are already shifting health care information technology
Urinalysis and Body Fluids CRg
 Urinalysis and Body Fluids CRg Unit 2; Session 1 Urine Microscopic Examination The Complete Urinalysis Physical properties already covered Chemical analysis in the next unit Microscopic our current focus
Urinalysis and Body Fluids CRg Unit 2; Session 1 Urine Microscopic Examination The Complete Urinalysis Physical properties already covered Chemical analysis in the next unit Microscopic our current focus
SEED Haematology. Sysmex Educational Enhancement and Development February 2013
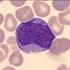 SEED Haematology Sysmex Educational Enhancement and Development February 2013 The role of the peripheral blood smear in the modern haematology laboratory Automated haematology cell counting The laboratory
SEED Haematology Sysmex Educational Enhancement and Development February 2013 The role of the peripheral blood smear in the modern haematology laboratory Automated haematology cell counting The laboratory
Statistical estimation using confidence intervals
 0894PP_ch06 15/3/02 11:02 am Page 135 6 Statistical estimation using confidence intervals In Chapter 2, the concept of the central nature and variability of data and the methods by which these two phenomena
0894PP_ch06 15/3/02 11:02 am Page 135 6 Statistical estimation using confidence intervals In Chapter 2, the concept of the central nature and variability of data and the methods by which these two phenomena
A&P 2 Blood Lab Guide In-Lab Exercises
 A&P 2 Blood Lab Guide In-Lab Exercises Have someone in your group read the following out loud, while the others read along: In this "Lab Guide", we will be looking at the blood and some common blood tests.
A&P 2 Blood Lab Guide In-Lab Exercises Have someone in your group read the following out loud, while the others read along: In this "Lab Guide", we will be looking at the blood and some common blood tests.
LIAISON XL HBsAg Quant
 Hepatitis and Retrovirus LIAISON XL HBsAg Quant Reliable detection of HBsAg mutants and genotypes for accurate differential diagnosis of the stage of infection FOR OUTSIDE THE US AND CANADA ONLY LIAISON
Hepatitis and Retrovirus LIAISON XL HBsAg Quant Reliable detection of HBsAg mutants and genotypes for accurate differential diagnosis of the stage of infection FOR OUTSIDE THE US AND CANADA ONLY LIAISON
Lab 02: Blood Cytology (20 points)
 Pierce College Putman/Biol 242 Name: Lab 02: Blood Cytology (20 points) Reference: Marieb & Mitchell 9 th Ed: 29A (Activities 1, 2, 3, 4, 7); 10 th Ed: Exercise 29 (Activities 1, 2, 3, 4, 7). Pierce College
Pierce College Putman/Biol 242 Name: Lab 02: Blood Cytology (20 points) Reference: Marieb & Mitchell 9 th Ed: 29A (Activities 1, 2, 3, 4, 7); 10 th Ed: Exercise 29 (Activities 1, 2, 3, 4, 7). Pierce College
Standardization, Calibration and Quality Control
 Standardization, Calibration and Quality Control Ian Storie Flow cytometry has become an essential tool in the research and clinical diagnostic laboratory. The range of available flow-based diagnostic
Standardization, Calibration and Quality Control Ian Storie Flow cytometry has become an essential tool in the research and clinical diagnostic laboratory. The range of available flow-based diagnostic
2015 RN.ORG, S.A., RN.ORG, LLC
 The Complete Blood Count and Associated Tests WWW.RN.ORG Reviewed December, 2015, Expires December, 2017 Provider Information and Specifics available on our Website Unauthorized Distribution Prohibited
The Complete Blood Count and Associated Tests WWW.RN.ORG Reviewed December, 2015, Expires December, 2017 Provider Information and Specifics available on our Website Unauthorized Distribution Prohibited
How to Verify Performance Specifications
 How to Verify Performance Specifications VERIFICATION OF PERFORMANCE SPECIFICATIONS In 2003, the Centers for Medicare and Medicaid Services (CMS) updated the CLIA 88 regulations. As a result of the updated
How to Verify Performance Specifications VERIFICATION OF PERFORMANCE SPECIFICATIONS In 2003, the Centers for Medicare and Medicaid Services (CMS) updated the CLIA 88 regulations. As a result of the updated
3. The Circulatory System
 3. The Circulatory System A. Introduction B. Blood 1. Circulatory system transports water, electrolytes, hormones, enzymes, antibodies, cell, gases and nutrients to all cells and carries away metabolic
3. The Circulatory System A. Introduction B. Blood 1. Circulatory system transports water, electrolytes, hormones, enzymes, antibodies, cell, gases and nutrients to all cells and carries away metabolic
Thrombin Formation for Children on Lovenox. Steven Ignell, BA
 Thrombin Formation for Children on Lovenox Steven Ignell, BA Definitions Anticoagulation Historically this refers to inhibiting thrombin formation Measured by PTT, INR, anti-xa Hypo and hypercoagulation
Thrombin Formation for Children on Lovenox Steven Ignell, BA Definitions Anticoagulation Historically this refers to inhibiting thrombin formation Measured by PTT, INR, anti-xa Hypo and hypercoagulation
laboratorymedicine> august 2001> number 8> volume 32
 interpretation [management/administration and training generalist] A Four-Part Approach to Competency Assessment Jennifer Schiffgens, MBA, MT(ASCP), 1 and Valerie Bush, PhD, MT(ASCP) 2 From 1 Clinical
interpretation [management/administration and training generalist] A Four-Part Approach to Competency Assessment Jennifer Schiffgens, MBA, MT(ASCP), 1 and Valerie Bush, PhD, MT(ASCP) 2 From 1 Clinical
LabNotes. The Hemolyzed Specimen: See Inside For Instructions On How To Obtain Your FREE Subscription. Causes, Effects, and Reduction
 LabNotes A Newsletter from BD Vacutainer Systems, Preanalytical Solutions Volume 13 No.1, Winter 2003 See Inside For Instructions On How To Obtain Your FREE Subscription The Hemolyzed Specimen: Causes,
LabNotes A Newsletter from BD Vacutainer Systems, Preanalytical Solutions Volume 13 No.1, Winter 2003 See Inside For Instructions On How To Obtain Your FREE Subscription The Hemolyzed Specimen: Causes,
USE OF REFERENCE MATERIALS IN THE LABORATORY
 USE OF REFERENCE MATERIALS IN THE LABORATORY What is a reference material? A general definition of a reference material is a material or substance one or more of whose property values are sufficiently
USE OF REFERENCE MATERIALS IN THE LABORATORY What is a reference material? A general definition of a reference material is a material or substance one or more of whose property values are sufficiently
WHOLE BLOOD LYSING SOLUTION FOR FLOW CYTOMETRIC APPLICATIONS
 FOR IN-VITRO DIAGNOSTIC USE INVITROGEN CAL-LYSE TM Lysing Solution WHOLE BLOOD LYSING SOLUTION FOR FLOW CYTOMETRIC APPLICATIONS CAL-LYSE TM CATALOG No. GAS-010 250 tests 25 ml CATALOG No. GAS-010S-100
FOR IN-VITRO DIAGNOSTIC USE INVITROGEN CAL-LYSE TM Lysing Solution WHOLE BLOOD LYSING SOLUTION FOR FLOW CYTOMETRIC APPLICATIONS CAL-LYSE TM CATALOG No. GAS-010 250 tests 25 ml CATALOG No. GAS-010S-100
Case 2:10-md-02179-CJB-SS Document 6427-14 Filed 05/03/12 Page 1 of 5 EXHIBIT 12
 Case 2:10-md-02179-CJB-SS Document 6427-14 Filed 05/03/12 Page 1 of 5 EXHIBIT 12 Case 2:10-md-02179-CJB-SS Document 6427-14 Filed 05/03/12 Page 2 of 5 Components of the PERIODIC MEDICAL CONSULTATION PROGRAM
Case 2:10-md-02179-CJB-SS Document 6427-14 Filed 05/03/12 Page 1 of 5 EXHIBIT 12 Case 2:10-md-02179-CJB-SS Document 6427-14 Filed 05/03/12 Page 2 of 5 Components of the PERIODIC MEDICAL CONSULTATION PROGRAM
