Biology Plant Physiology - Lab Exercise 2 Basic Lab Techniques
|
|
|
- Brice Powers
- 7 years ago
- Views:
Transcription
1 Biol3460 Lab 1 1 Biology Plant Physiology - Lab Exercise 2 Basic Lab Techniques Objectives: This lab is intended to: (1) provide an introduction to basic techniques used in biology labs (2) provide some experience with the equipment employed in future labs. Part A. Microscopy The compound light microscope is a very important tool in the study of biology. Typical compound light microscopes allow specimens to be viewed at 400 to 1000 times their actual size (total magnification is determined by multiplying the objective lens magnification by the ocular lens magnification). The light microscope is made up of a system of lenses aligned in such a way that allows magnification, as well as improved resolution. The resolution obtainable is also dependent on contrast in the specimen. Contrast can be adjusted with the iris diaphragm, located below the condenser lens, and can also be enhanced by the addition of stains to the specimen. Scientific drawings of specimens observed under the light microscope are often used to provide a record of what was viewed. Proper scientific drawings should be neatly drawn, with smooth lines, in pencil (they should never be in colour). More than one cell should be drawn to illustrate the cellular association to the observer (e.g. found singly or as part of a tissue). Labels should be added using straight horizontal lines to the right side of the drawing, and care should be taken with singular and plural forms of structural terminology. To demonstrate differences in density of specific structures, use stippling rather than shading. A figure caption that indicates the specimen viewed, how viewed, and the magnification of the drawing is always necessary. Practice your microscopy technique by viewing slides of Elodea, spinach leaf, and pea seeds. Work in pairs to set-up and view each slide preparation. 1. Place one leaflet of Elodea on a slide with a drop of water and look for the clearly visible chloroplasts. You should notice that the chloroplasts are circulating around in each cell. What is the purpose of this cytoplasmic streaming? 2. Prepare a slide of spinach leaf epidermis by peeling back a bit of the undersurface of the leaf tissue. You should get a thin section of only the clear epidermal tissue, leaving behind the parenchyma cells of the leaf itself. Find the guard cells and the stomata and make a labeled sketch of a section of epidermal tissue. Use an ocular micrometer to determine the actual size of the guard cells and then calculate the drawing magnification (drawing size actual size) for your figure and include it in your figure caption. 3. Slice a very thin section of imbibed pea seed tissue and place it on a slide with a drop of iodine stain. What do you think the bulk of the pea seed is composed of? What compound is commonly stored by plants as a food reserve?
2 Biol3460 Lab 1 2 Part B. Analytical Balance and Weighing Accuracy Careful use of all types of equipment is essential to obtaining accurate and repeatable results in a biology lab. In this exercise you will practice use of the balance, as well as perform a mini experiment that indicates the effect of imbibition on bean seeds. Care and use of the balance: Avoid shock and vibrations. Make sure the balance is level using the leveling bubble and the adjustable legs. Always use a weigh boat or weighing paper. Do not place any material directly on the balance pan. Use a damp paper towel to wipe up any spills and keep the pan and case clean at all times. Work in a group of four to complete this exercise. One pair of students should weigh the imbibed seeds then the other pair should weigh the dry seeds. 1. Place a weigh boat on the balance and press tare. 2. Gently blot each seed on a piece of paper towel. 3. Place the bean seed in the weigh boat and record its weight in Table Calculate the means and standard deviations for the weights of the dry and the imbibed seeds. 5. Perform a t-test on the data to determine if there is a significant effect of imbibition on bean seed weight. 6. What is the level of accuracy of the balance? Table 1. Weight of dry and imbibed bean seeds determined using analytical balance. Treatment Bean Seed Weights (g) Dry Imbibed Part C. A Bit of Chemistry and Solution Dilutions A basic understanding of general chemistry is very important to many topics in plant physiology. In the lab setting, in particular, it is necessary to be able to accurately prepare solutions of known concentrations and prepare dilutions from stock solutions. This exercise will give you practice with the preparation of solutions of various concentrations, as well as practice using pipettes to transport accurate volumes between solutions. Work in a group of four to complete the dilution series. 1. Obtain 7 test tubes from the side bench and label them with the appropriate protein standard concentrations (see Table 2). 2. Beginning with the 5mg/mL bovine serum albumin (BSA) stock solution, prepare the series of dilutions as outlined in Table 2. You will be responsible for calculating how much of each solution is required to prepare the next dilution in the series. 3. Prepare each solution by pipetting the appropriate amount of the previous concentration into the correctly labeled test tube and adding the appropriate volume of distilled water. 4. Mix each solution on the vortex for 15 seconds. 5. Use these solutions to continue with the procedure in Part D.
3 Biol3460 Lab 1 3 Table 2. Volume of protein solutions and distilled water to make a dilution series for protein analysis and standard curve construction. Protein concentration (mg/ml) Volume of protein solution (ml) Volume of water (ml) Total volume (ml) 1.0 ml of 5.0 mg/ml stock ml of 1.0 mg/ml solution ml of 0.75 mg/ml solution ml of 0.50 mg/ml solution ml of 0.25 mg/ml solution ml of 0.10 mg/ml solution Part D. Protein Assay and Spectrophotometer Use Many plant molecules such as the photosynthetic pigments, chlorophylls, anthocyanins, and carotenoids, naturally absorb light. Other colorless plant molecules can be converted to light-absorbing pigments by the addition of specific chemicals. A spectrophotometer is an instrument that can be used to objectively quantify the amounts and wavelengths of light that are absorbed by molecules in a solution. To practice using the spectrophotometer, you will conduct a Bradford protein assay using the solutions you prepared in Part C. Since protein is not highly coloured it can be reacted with another reagent to produce a coloured compound. Protein concentration is a useful measure to express various processes studied and allows comparison to other samples and to published values. Work as a group of four for this exercise. 1. Label 7 small tubes with the concentrations listed in Table 2, plus 2 test tubes with Unknown A and Unknown B. 2. Pipette 0.1mL of each protein solution into the appropriately labeled tube. 3. Add 5.0mL of the Bradford Reagent and vortex to mix. 4. Allow tubes to incubate for five minutes then use the spectrophotometer to read the absorbances at 595 nm. Spectrophotometer Procedure: 1. Set the wavelength to the appropriate value. 2. With no cuvette in the spectrophotometer set the spectrophotometer to zero on the percent transmittance scale, using the left-hand knob. 3. Always wipe the cuvette with a Kimwipe before inserting it into the spectrophotometer, to remove any dust, fingerprints, or water droplets that may affect how the sample absorbs light. 4. Place the reagent blank in the spectrophotometer and set to zero on the absorbance scale with the right-hand knob. The reagent blank is a solution that contains all the chemicals that are in the other samples except the molecule of interest. 5. Transfer your palest sample to the cuvette, insert it into the sample compartment, and read the absorbance value and record it in Table Pour the sample back into the test tube it came from and continue reading the absorbance of the other samples from palest to darkest (don t discard any samples until you are finished and are sure that your readings are accurate).
4 Biol3460 Lab 1 4 Table 3. Absorbance values for solutions of known BSA concentration, measured by spectrophotometer at 595 nm. BSA concentration (mg/ml) Absorbance (A 595 ) Part E. Standard Curves and Graphing Formats Proper graph construction is a must in nearly every course of biological study. Graphs provide a clear indication of general trends seen in a range of data and can be used to infer relationships between variables. A standard curve is a graph in which the absorbance of a sample is plotted as a function of various concentrations of the compound of interest. The equation of this curve can then be used to determine the concentration of unknown samples of the same compound, when you have measured their absorbance. Standard Curve Construction: 1. Plot the absorbance values obtained from Part D against the known concentrations of the seven protein solutions. 2. Be sure to properly label the axis and provide a descriptive figure caption for the graph. 3. Fit a straight line to the data and calculate the equation of the line. 4. Use the equation of the line to determine the concentration of the two unknown samples you ran with the protein assay in Part D. Graph Formatting Tips: Graphs should be constructed in Excel or a similar spreadsheet program, however you may use graph paper if you are very neat. The dependent variable is always plotted on the y-axis against the independent variable, which is plotted on the x-axis. The axes scales should be divided into equal intervals if the data is continuous. Choose intervals that are logically spaced and permit easy interpretation of the graph. If there are no data points at the low end of the scale it is not necessary to start the scale at zero. However, do not break the graph in the middle of the scale with data points on either side. Axes labels should be descriptive and should include the units for the measurement in parentheses. The graph should include a descriptive figure caption, placed below the graph. Label the graph as a figure and give it a number (e.g. Figure 1), based on when it is referred to in the text of your paper. The description should include all information necessary to understand the data presented in the graph (all variables, treatments, types of values reported, symbols, sample sizes, and organisms or cells studied). To illustrate trends in line graphs, use smooth curves or straight lines to fit the values for one data set. Do not force a straight line through a data set that does not appear to be linear and do not force a straight line through the origin (0,0) if it is not representative of the data series.
5 Biol3460 Lab 1 5 Conversion Factors Table 4. The calibrated values of an ocular unit for various light microscope objective lenses as determined by a stage micrometer. Objective Lens Single Ocular Unit Value (mm) 4X X X X Metric Conversion Values: Volume 1L = 1000 ml 1 ml = 1000 µl Length 1 m = 100 cm 1 cm = 10 mm 1 mm = 1000 µm 1 µm = 1000 nm Mass 1 kg = 1000 g 1 g = 1000 mg 1 mg = 1000 µg
Using the Spectrophotometer
 Using the Spectrophotometer Introduction In this exercise, you will learn the basic principals of spectrophotometry and and serial dilution and their practical application. You will need these skills to
Using the Spectrophotometer Introduction In this exercise, you will learn the basic principals of spectrophotometry and and serial dilution and their practical application. You will need these skills to
Laboratory 5: Properties of Enzymes
 Laboratory 5: Properties of Enzymes Technical Objectives 1. Accurately measure and transfer solutions with pipettes 2. Use a Spectrophotometer to study enzyme action. 3. Properly graph a set of data. Knowledge
Laboratory 5: Properties of Enzymes Technical Objectives 1. Accurately measure and transfer solutions with pipettes 2. Use a Spectrophotometer to study enzyme action. 3. Properly graph a set of data. Knowledge
Austin Peay State University Department of Chemistry Chem 1111. The Use of the Spectrophotometer and Beer's Law
 Purpose To become familiar with using a spectrophotometer and gain an understanding of Beer s law and it s relationship to solution concentration. Introduction Scientists use many methods to determine
Purpose To become familiar with using a spectrophotometer and gain an understanding of Beer s law and it s relationship to solution concentration. Introduction Scientists use many methods to determine
Measuring Protein Concentration through Absorption Spectrophotometry
 Measuring Protein Concentration through Absorption Spectrophotometry In this lab exercise you will learn how to homogenize a tissue to extract the protein, and then how to use a protein assay reagent to
Measuring Protein Concentration through Absorption Spectrophotometry In this lab exercise you will learn how to homogenize a tissue to extract the protein, and then how to use a protein assay reagent to
MICROSCOPY. To demonstrate skill in the proper utilization of a light microscope.
 MICROSCOPY I. OBJECTIVES To demonstrate skill in the proper utilization of a light microscope. To demonstrate skill in the use of ocular and stage micrometers for measurements of cell size. To recognize
MICROSCOPY I. OBJECTIVES To demonstrate skill in the proper utilization of a light microscope. To demonstrate skill in the use of ocular and stage micrometers for measurements of cell size. To recognize
EFFECT OF SALT ON CELL MEMBRANES
 EFFECT OF SALT ON CELL MEMBRANES LAB CELL 2 INTRODUCTION A eukaryotic cell, a cell with a nucleus, not only has a plasma membrane as its external boundary, but it also has a variety of membranes that divide
EFFECT OF SALT ON CELL MEMBRANES LAB CELL 2 INTRODUCTION A eukaryotic cell, a cell with a nucleus, not only has a plasma membrane as its external boundary, but it also has a variety of membranes that divide
Chem 131A: Absorbance of Riboflavin
 Chem 131A: Absorbance of Riboflavin Purpose: The purpose of this experiment is to: 1) Familiarize the student with the use of the HP 8452 diode array spectrophotometer, 2) examine the limitations of the
Chem 131A: Absorbance of Riboflavin Purpose: The purpose of this experiment is to: 1) Familiarize the student with the use of the HP 8452 diode array spectrophotometer, 2) examine the limitations of the
Chem 405 Biochemistry Lab I Experiment 2 Quantitation of an unknown protein solution.
 Chem 405 Biochemistry Lab I Experiment 2 Quantitation of an unknown protein solution. Introduction: The determination of protein concentration is frequently required in biochemical work. Several methods
Chem 405 Biochemistry Lab I Experiment 2 Quantitation of an unknown protein solution. Introduction: The determination of protein concentration is frequently required in biochemical work. Several methods
EFFECT OF ALCOHOL ON CELL MEMBRANES
 EFFECT OF ALCOHOL ON CELL MEMBRANES LAB CELL 1 INTRODUCTION A eukaryotic cell, a cell with a nucleus, not only has a plasma membrane as its external boundary, but it also has a variety of membranes that
EFFECT OF ALCOHOL ON CELL MEMBRANES LAB CELL 1 INTRODUCTION A eukaryotic cell, a cell with a nucleus, not only has a plasma membrane as its external boundary, but it also has a variety of membranes that
Name Class Date Laboratory Investigation 4B Chapter 4: Cell Structure
 Name Class Date Laboratory Investigation 4B Chapter 4: Cell Structure The Microscope: A Tool of the Scientist You may refer to pages 66-67, 72-73 in your textbook for a general discussion of microscopes.
Name Class Date Laboratory Investigation 4B Chapter 4: Cell Structure The Microscope: A Tool of the Scientist You may refer to pages 66-67, 72-73 in your textbook for a general discussion of microscopes.
Care and Use of the Compound Microscope
 Revised Fall 2011 Care and Use of the Compound Microscope Objectives After completing this lab students should be able to 1. properly clean and carry a compound and dissecting microscope. 2. focus a specimen
Revised Fall 2011 Care and Use of the Compound Microscope Objectives After completing this lab students should be able to 1. properly clean and carry a compound and dissecting microscope. 2. focus a specimen
Spectrophotometry and the Beer-Lambert Law: An Important Analytical Technique in Chemistry
 Spectrophotometry and the Beer-Lambert Law: An Important Analytical Technique in Chemistry Jon H. Hardesty, PhD and Bassam Attili, PhD Collin College Department of Chemistry Introduction: In the last lab
Spectrophotometry and the Beer-Lambert Law: An Important Analytical Technique in Chemistry Jon H. Hardesty, PhD and Bassam Attili, PhD Collin College Department of Chemistry Introduction: In the last lab
Factors Affecting Enzyme Activity
 INTRODUCTION Factors Affecting Enzyme Activity The chemical reactions occurring in living things are controlled by enzymes. An enzyme is a protein in the cell which lowers the activation energy of a catalyzed
INTRODUCTION Factors Affecting Enzyme Activity The chemical reactions occurring in living things are controlled by enzymes. An enzyme is a protein in the cell which lowers the activation energy of a catalyzed
A Beer s Law Experiment
 A Beer s Law Experiment Introduction There are many ways to determine concentrations of a substance in solution. So far, the only experiences you may have are acid-base titrations or possibly determining
A Beer s Law Experiment Introduction There are many ways to determine concentrations of a substance in solution. So far, the only experiences you may have are acid-base titrations or possibly determining
ENZYME KINETICS ENZYME-SUBSTRATE PRODUCTS
 ENZYME KINETICS INTRODUCTION The study of reaction rates catalyzed by enzymes and the factors affecting them is generally referred to as enzyme kinetics. The basic components of an enzyme catalyzed reaction
ENZYME KINETICS INTRODUCTION The study of reaction rates catalyzed by enzymes and the factors affecting them is generally referred to as enzyme kinetics. The basic components of an enzyme catalyzed reaction
Cell Biology Prokaryotic and eukaryotic cells
 Cell Biology Prokaryotic and eukaryotic cells Observation of cells and organelles In this lab you will be looking at an example of a Prokaryotic cell (Bacillus cereus) and a some examples of Eukaryotic
Cell Biology Prokaryotic and eukaryotic cells Observation of cells and organelles In this lab you will be looking at an example of a Prokaryotic cell (Bacillus cereus) and a some examples of Eukaryotic
Upon completion of this lab, the student will be able to:
 1 Learning Outcomes EXPERIMENT B4: CHEMICAL EQUILIBRIUM Upon completion of this lab, the student will be able to: 1) Analyze the absorbance spectrum of a sample. 2) Calculate the equilibrium constant for
1 Learning Outcomes EXPERIMENT B4: CHEMICAL EQUILIBRIUM Upon completion of this lab, the student will be able to: 1) Analyze the absorbance spectrum of a sample. 2) Calculate the equilibrium constant for
Colorimetry Extinction coefficient (ε) Lambda max (λ max ) Qualitative vs. quantitative analysis
 Lab Week 2 - Spectrophotometry Purpose: Introduce students to the use of spectrophotometry for qualitative (what is it) and quantitative (how much is there of it) analysis of biological samples and molecules.
Lab Week 2 - Spectrophotometry Purpose: Introduce students to the use of spectrophotometry for qualitative (what is it) and quantitative (how much is there of it) analysis of biological samples and molecules.
THE ACTIVITY OF LACTASE
 THE ACTIVITY OF LACTASE Lab VIS-8 From Juniata College Science in Motion Enzymes are protein molecules which act to catalyze the chemical reactions in living things. These chemical reactions make up the
THE ACTIVITY OF LACTASE Lab VIS-8 From Juniata College Science in Motion Enzymes are protein molecules which act to catalyze the chemical reactions in living things. These chemical reactions make up the
EXPERIMENT 11 UV/VIS Spectroscopy and Spectrophotometry: Spectrophotometric Analysis of Potassium Permanganate Solutions.
 EXPERIMENT 11 UV/VIS Spectroscopy and Spectrophotometry: Spectrophotometric Analysis of Potassium Permanganate Solutions. Outcomes After completing this experiment, the student should be able to: 1. Prepare
EXPERIMENT 11 UV/VIS Spectroscopy and Spectrophotometry: Spectrophotometric Analysis of Potassium Permanganate Solutions. Outcomes After completing this experiment, the student should be able to: 1. Prepare
EXPERIMENT #1: MICROSCOPY
 EXPERIMENT #1: MICROSCOPY Brightfield Compound Light Microscope The light microscope is an important tool in the study of microorganisms. The compound light microscope uses visible light to directly illuminate
EXPERIMENT #1: MICROSCOPY Brightfield Compound Light Microscope The light microscope is an important tool in the study of microorganisms. The compound light microscope uses visible light to directly illuminate
MITOSIS IN ONION ROOT TIP CELLS: AN INTRODUCTION TO LIGHT MICROSCOPY
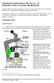 MITOSIS IN ONION ROOT TIP CELLS: AN INTRODUCTION TO LIGHT MICROSCOPY Adapted from Foundations of Biology I; Lab 6 Introduction to Microscopy Dr. John Robertson, Westminster College Biology Department,
MITOSIS IN ONION ROOT TIP CELLS: AN INTRODUCTION TO LIGHT MICROSCOPY Adapted from Foundations of Biology I; Lab 6 Introduction to Microscopy Dr. John Robertson, Westminster College Biology Department,
2 Spectrophotometry and the Analysis of Riboflavin
 2 Spectrophotometry and the Analysis of Riboflavin Objectives: A) To become familiar with operating the Platereader; B) to learn how to use the Platereader in determining the absorption spectrum of a compound
2 Spectrophotometry and the Analysis of Riboflavin Objectives: A) To become familiar with operating the Platereader; B) to learn how to use the Platereader in determining the absorption spectrum of a compound
OBJECTIVES PROCEDURE. Lab 2- Bio 160. Name:
 Lab 2- Bio 160 Name: Prokaryotic and Eukaryotic Cells OBJECTIVES To explore cell structure and morphology in prokaryotes and eukaryotes. To gain more experience using the microscope. To obtain a better
Lab 2- Bio 160 Name: Prokaryotic and Eukaryotic Cells OBJECTIVES To explore cell structure and morphology in prokaryotes and eukaryotes. To gain more experience using the microscope. To obtain a better
Experiment 13H THE REACTION OF RED FOOD COLOR WITH BLEACH 1
 Experiment 13H FV 1/25/2011(2-run) THE REACTION OF RED FOOD COLOR WITH BLEACH 1 PROBLEM: Determine the rate law for the chemical reaction between FD&C Red Dye #3 and sodium hypochlorite. LEARNING OBJECTIVES:
Experiment 13H FV 1/25/2011(2-run) THE REACTION OF RED FOOD COLOR WITH BLEACH 1 PROBLEM: Determine the rate law for the chemical reaction between FD&C Red Dye #3 and sodium hypochlorite. LEARNING OBJECTIVES:
Absorbance Spectrophotometry: Analysis of FD&C Red Food Dye #40
 Absorbance Spectrophotometry: Analysis of FD&C Red Food Dye #40 Note: there is a second document that goes with this one! 2046 - Absorbance Spectrophotometry - Calibration Curve Procedure. The second document
Absorbance Spectrophotometry: Analysis of FD&C Red Food Dye #40 Note: there is a second document that goes with this one! 2046 - Absorbance Spectrophotometry - Calibration Curve Procedure. The second document
Biology 3A Laboratory: Enzyme Function
 Biology 3A Laboratory: Enzyme Function Objectives To be able to list the general characteristics of enzymes. To study the effects of enzymes on the rate of chemical reactions. To demonstrate the effect
Biology 3A Laboratory: Enzyme Function Objectives To be able to list the general characteristics of enzymes. To study the effects of enzymes on the rate of chemical reactions. To demonstrate the effect
Biology 29 Cell Structure and Function Spring, 2009 Springer LABORATORY 2:CHLOROPLASTS AND PHOTOREDUCTION
 Biology 29 Cell Structure and Function Spring, 2009 Springer LABORATORY 2:CHLOROPLASTS AND PHOTOREDUCTION In this laboratory we will purify chloroplasts from spinach by differential centrifugation, then
Biology 29 Cell Structure and Function Spring, 2009 Springer LABORATORY 2:CHLOROPLASTS AND PHOTOREDUCTION In this laboratory we will purify chloroplasts from spinach by differential centrifugation, then
Prokaryotic and Eukaryotic Cells
 Lab 2- Bio 201 Prokaryotic and Eukaryotic Cells Name: OBJECTIVES To explore cell structure and morphology in prokaryotes and eukaryotes. To gain more experience using the microscope, and in particular,
Lab 2- Bio 201 Prokaryotic and Eukaryotic Cells Name: OBJECTIVES To explore cell structure and morphology in prokaryotes and eukaryotes. To gain more experience using the microscope, and in particular,
The Determination of an Equilibrium Constant
 The Determination of an Equilibrium Constant Computer 10 Chemical reactions occur to reach a state of equilibrium. The equilibrium state can be characterized by quantitatively defining its equilibrium
The Determination of an Equilibrium Constant Computer 10 Chemical reactions occur to reach a state of equilibrium. The equilibrium state can be characterized by quantitatively defining its equilibrium
Quantifying Bacterial Concentration using a Calibrated Growth Curve
 BTEC 4200 Lab 2. Quantifying Bacterial Concentration using a Calibrated Growth Curve Background and References Bacterial concentration can be measured by several methods, all of which you have studied
BTEC 4200 Lab 2. Quantifying Bacterial Concentration using a Calibrated Growth Curve Background and References Bacterial concentration can be measured by several methods, all of which you have studied
Solubility Product Constants
 Solubility Product Constants PURPOSE To measure the solubility product constant (K sp ) of copper (II) iodate, Cu(IO 3 ) 2. GOALS 1 To measure the molar solubility of a sparingly soluble salt in water.
Solubility Product Constants PURPOSE To measure the solubility product constant (K sp ) of copper (II) iodate, Cu(IO 3 ) 2. GOALS 1 To measure the molar solubility of a sparingly soluble salt in water.
Cellular Membranes I. BACKGROUND MATERIAL
 Cellular Membranes Objectives: 1. To explore the nature of cellular membranes by investigating environmental conditions which stress them. 2. To learn methods for measuring the extent of stress on the
Cellular Membranes Objectives: 1. To explore the nature of cellular membranes by investigating environmental conditions which stress them. 2. To learn methods for measuring the extent of stress on the
Determining the Quantity of Iron in a Vitamin Tablet. Evaluation copy
 Determining the Quantity of Iron in a Vitamin Tablet Computer 34 As biochemical research becomes more sophisticated, we are learning more about the role of metallic elements in the human body. For example,
Determining the Quantity of Iron in a Vitamin Tablet Computer 34 As biochemical research becomes more sophisticated, we are learning more about the role of metallic elements in the human body. For example,
Compound microscope (Hund)
 1 2 3 4 5 6 7 8 9 10 11 12 13 14 Compound microscope (Hund) 15 16 17 18 19 20 1) Eyepieces (magnifies 10x), one with diopter adjustment, 2) Interp[upillary adjustment, 3) Head, 4) Revolving nosepiece,
1 2 3 4 5 6 7 8 9 10 11 12 13 14 Compound microscope (Hund) 15 16 17 18 19 20 1) Eyepieces (magnifies 10x), one with diopter adjustment, 2) Interp[upillary adjustment, 3) Head, 4) Revolving nosepiece,
Lab 2. Spectrophotometric Measurement of Glucose
 Lab 2 Spectrophotometric Measurement of Glucose Objectives 1. Learn how to use a spectrophotometer. 2. Produce a glucose standard curve. 3. Perform a glucose assay. Safety Precautions Glucose Color Reagent
Lab 2 Spectrophotometric Measurement of Glucose Objectives 1. Learn how to use a spectrophotometer. 2. Produce a glucose standard curve. 3. Perform a glucose assay. Safety Precautions Glucose Color Reagent
Using Spectrophotometers to Examine Photosynthetic Rates Under Various Qualities of Light
 Purdue GK-12 Lesson Plan 2006-07 Using Spectrophotometers to Examine Photosynthetic Rates Under Various Qualities of Light Purdue University GK-12 2006-2007 Lead developer and contact: Amanda Deering Purdue
Purdue GK-12 Lesson Plan 2006-07 Using Spectrophotometers to Examine Photosynthetic Rates Under Various Qualities of Light Purdue University GK-12 2006-2007 Lead developer and contact: Amanda Deering Purdue
Determination of the Mass Percentage of Copper in a Penny. Introduction
 Determination of the Mass Percentage of Copper in a Penny Introduction This experiment will cost you one penny ($0.01). The penny must be minted after 1983. Any penny will do; for best results the penny
Determination of the Mass Percentage of Copper in a Penny Introduction This experiment will cost you one penny ($0.01). The penny must be minted after 1983. Any penny will do; for best results the penny
Students will identify these animal cell structures: Students should properly answer the pre-activity cell membrane, nucleus. questions.
 WHAT DO PLANT & ANIMAL CELLS LOOK LIKE? Grade Levels: 10-12 Time Frame: 2 periods Big Idea: Students will compare various plant epithelial cells (onion and elodea) with human epithelial cells (cheek lining
WHAT DO PLANT & ANIMAL CELLS LOOK LIKE? Grade Levels: 10-12 Time Frame: 2 periods Big Idea: Students will compare various plant epithelial cells (onion and elodea) with human epithelial cells (cheek lining
Determining the Free Chlorine Content of Swimming Pool Water. HOCl H + + OCl. Evaluation copy
 Determining the Free Chlorine Content of Swimming Pool Water Computer 33 Physicians in the nineteenth century used chlorine water as a disinfectant. Upon the discovery that certain diseases were transmitted
Determining the Free Chlorine Content of Swimming Pool Water Computer 33 Physicians in the nineteenth century used chlorine water as a disinfectant. Upon the discovery that certain diseases were transmitted
Osmosis Demonstration Lab
 Osmosis Demonstration Lab Objectives The student will: 1) Observe the effects of different concentrations of salt solutions on potato cores. 2) Infer the relationship between weight loss and rate of osmosis.
Osmosis Demonstration Lab Objectives The student will: 1) Observe the effects of different concentrations of salt solutions on potato cores. 2) Infer the relationship between weight loss and rate of osmosis.
A Fishy Tale. Observing the Circulatory System of a Goldfish with a Compound Light Microscope
 A Fishy Tale Observing the Circulatory System of a Goldfish with a Compound Light Microscope A Fishy Tale About this Lesson In this lesson, students will explore a computer animation of the human body
A Fishy Tale Observing the Circulatory System of a Goldfish with a Compound Light Microscope A Fishy Tale About this Lesson In this lesson, students will explore a computer animation of the human body
Comparing Plant and Animal Cells
 1.2 Comparing Plant and Animal Cells Here is a summary of what you will learn in this section: Plant and animal cell structures are called organelles. Plant and animal cells perform some similar functions,
1.2 Comparing Plant and Animal Cells Here is a summary of what you will learn in this section: Plant and animal cell structures are called organelles. Plant and animal cells perform some similar functions,
ANALYSIS OF PLANT PIGMENTS USING PAPER CHROMATOGRAPHY AND VISIBLE AND/OR UV SPECTROSCOPY (1-31-96)
 ANALYSIS OF PLANT PIGMENTS USING PAPER CHROMATOGRAPHY AND VISIBLE AND/OR UV SPECTROSCOPY (1-31-96) INTRODUCTION We have seen that all cells must constantly consume fuel molecules to maintain themselves,
ANALYSIS OF PLANT PIGMENTS USING PAPER CHROMATOGRAPHY AND VISIBLE AND/OR UV SPECTROSCOPY (1-31-96) INTRODUCTION We have seen that all cells must constantly consume fuel molecules to maintain themselves,
ATOMIC ABSORTION SPECTROSCOPY: rev. 4/2011 ANALYSIS OF COPPER IN FOOD AND VITAMINS
 1 ATOMIC ABSORTION SPECTROSCOPY: rev. 4/2011 ANALYSIS OF COPPER IN FOOD AND VITAMINS Buck Scientific Atomic Absorption Spectrophotometer, Model 200 Atomic absorption spectroscopy (AAS) has for many years
1 ATOMIC ABSORTION SPECTROSCOPY: rev. 4/2011 ANALYSIS OF COPPER IN FOOD AND VITAMINS Buck Scientific Atomic Absorption Spectrophotometer, Model 200 Atomic absorption spectroscopy (AAS) has for many years
Reaction Stoichiometry and the Formation of a Metal Ion Complex
 Reaction Stoichiometry and the Formation of a Metal Ion Complex Objectives The objectives of this laboratory are as follows: To use the method of continuous variation to determine the reaction stoichiometry
Reaction Stoichiometry and the Formation of a Metal Ion Complex Objectives The objectives of this laboratory are as follows: To use the method of continuous variation to determine the reaction stoichiometry
CHEM 161: Beer s Law and Analysis of a Sports Drink
 CHEM 161: Beer s Law and Analysis of a Sports Drink Introduction Although sunlight appears white, it contains a spectrum of colors. A rainbow actually shows this range of colors in visible light: violet,
CHEM 161: Beer s Law and Analysis of a Sports Drink Introduction Although sunlight appears white, it contains a spectrum of colors. A rainbow actually shows this range of colors in visible light: violet,
2C: One in a Million. Part 1: Making solutions. Name: Section: Date: Materials
 Name: Section: Date: 2C: One in a Million Drinking water can contain up to 1.3 parts per million (ppm) of copper and still be considered safe. What does parts per million mean? Both living things and the
Name: Section: Date: 2C: One in a Million Drinking water can contain up to 1.3 parts per million (ppm) of copper and still be considered safe. What does parts per million mean? Both living things and the
Photosynthesis and Respiration
 Photosynthesis and Respiration Experiment 31C Plants make sugar, storing the energy of the sun into chemical energy, by the process of photosynthesis. When they require energy, they can tap the stored
Photosynthesis and Respiration Experiment 31C Plants make sugar, storing the energy of the sun into chemical energy, by the process of photosynthesis. When they require energy, they can tap the stored
Spectrophotometry Practical Lesson on Medical Chemistry and Biochemistry
 Spectrophotometry Practical Lesson on Medical Chemistry and Biochemistry General Medicine Jiřina Crkovská (translated by Jan Pláteník) 2010/2011 1 Spectrophotometry is one of the most widely used instrumental
Spectrophotometry Practical Lesson on Medical Chemistry and Biochemistry General Medicine Jiřina Crkovská (translated by Jan Pláteník) 2010/2011 1 Spectrophotometry is one of the most widely used instrumental
Osmosis. Evaluation copy
 Osmosis Computer 5 In order to survive, all organisms need to move molecules in and out of their cells. Molecules such as gases (e.g., O 2, CO 2 ), water, food, and wastes pass across the cell membrane.
Osmosis Computer 5 In order to survive, all organisms need to move molecules in and out of their cells. Molecules such as gases (e.g., O 2, CO 2 ), water, food, and wastes pass across the cell membrane.
Phenolphthalein-NaOH Kinetics
 Phenolphthalein-NaOH Kinetics Phenolphthalein is one of the most common acid-base indicators used to determine the end point in acid-base titrations. It is also the active ingredient in some laxatives.
Phenolphthalein-NaOH Kinetics Phenolphthalein is one of the most common acid-base indicators used to determine the end point in acid-base titrations. It is also the active ingredient in some laxatives.
COMPARING PLANT AND ANIMAL CELLS
 COMPARING PLANT AND ANIMAL CELLS OBJECTIVES: Distinguish between plant and animals cells by their structures Demonstrate the benefit of stains Acquire ability to prepare wet mounts SAFETY: Methylene blue
COMPARING PLANT AND ANIMAL CELLS OBJECTIVES: Distinguish between plant and animals cells by their structures Demonstrate the benefit of stains Acquire ability to prepare wet mounts SAFETY: Methylene blue
Beer's Law: Colorimetry of Copper(II) Solutions
 Exercise 11 Page 1 Illinois Central College CHEMISTRY 130 Name: Beer's Law: Colorimetry of Copper(II) Solutions Objectives In this experiment, we will use Beer's Law to determine the unknown concentrations
Exercise 11 Page 1 Illinois Central College CHEMISTRY 130 Name: Beer's Law: Colorimetry of Copper(II) Solutions Objectives In this experiment, we will use Beer's Law to determine the unknown concentrations
Scientific Graphing in Excel 2010
 Scientific Graphing in Excel 2010 When you start Excel, you will see the screen below. Various parts of the display are labelled in red, with arrows, to define the terms used in the remainder of this overview.
Scientific Graphing in Excel 2010 When you start Excel, you will see the screen below. Various parts of the display are labelled in red, with arrows, to define the terms used in the remainder of this overview.
LAB 24 Transpiration
 Name: AP Biology Lab 24 LAB 24 Transpiration Objectives: To understand how water moves from roots to leaves in terms of the physical/chemical properties of water and the forces provided by differences
Name: AP Biology Lab 24 LAB 24 Transpiration Objectives: To understand how water moves from roots to leaves in terms of the physical/chemical properties of water and the forces provided by differences
Cells, Diffusion, Osmosis, and Biological Membranes
 Cells, Diffusion, Osmosis, and Biological Membranes A. Objectives Upon completion of this lab activity, you should be able to: 1. Define and correctly use the following terms: solute, solvent, selectively
Cells, Diffusion, Osmosis, and Biological Membranes A. Objectives Upon completion of this lab activity, you should be able to: 1. Define and correctly use the following terms: solute, solvent, selectively
EXPERIMENT 5. Molecular Absorption Spectroscopy: Determination of Iron With 1,10-Phenanthroline
 EXPERIMENT 5 Molecular Absorption Spectroscopy: Determination of Iron With 1,10-Phenanthroline UNKNOWN Submit a clean, labeled 100-mL volumetric flask to the instructor so that your unknown iron solution
EXPERIMENT 5 Molecular Absorption Spectroscopy: Determination of Iron With 1,10-Phenanthroline UNKNOWN Submit a clean, labeled 100-mL volumetric flask to the instructor so that your unknown iron solution
10/5/06 Lab 3. Protein Determination. Lab 3. PROTEIN DETERMINATION
 10/5/06 Lab 3. Protein Determination Lab 3. PROTEIN DETERMINATION I. INTRODUCTION Reading in Biology, 6 th ed. By Campbell: PROTEINS-MANY STRUCTURES, MANY FUNCTIONS p. 71-80 Blood is a connective tissue
10/5/06 Lab 3. Protein Determination Lab 3. PROTEIN DETERMINATION I. INTRODUCTION Reading in Biology, 6 th ed. By Campbell: PROTEINS-MANY STRUCTURES, MANY FUNCTIONS p. 71-80 Blood is a connective tissue
Procedure for Protein Analysis. Protein Extraction
 Name Section Lab 6 Protein, Fats, Sugars and Starch Plants are composed of a variety of compounds, many of which humans use for food, drugs oils and cosmetics. Besides water, certain plant organs have
Name Section Lab 6 Protein, Fats, Sugars and Starch Plants are composed of a variety of compounds, many of which humans use for food, drugs oils and cosmetics. Besides water, certain plant organs have
Animal & Plant Cell Slides
 Animal & Plant Cell Slides Category: Biology Type: Class Experiment, 60 min class Materials: 2 Glass Slides 2 Cover Slips 1 Bottle of methylene blue (optional) 1 Plastic tray 1 Bottle of iodine 1 Plastic
Animal & Plant Cell Slides Category: Biology Type: Class Experiment, 60 min class Materials: 2 Glass Slides 2 Cover Slips 1 Bottle of methylene blue (optional) 1 Plastic tray 1 Bottle of iodine 1 Plastic
Ultraviolet-Visible (UV-Vis) Spectroscopy Background Information
 1 Ultraviolet-Visible (UV-Vis) Spectroscopy Background Information Instructions for the Operation of the Cary 300 Bio UV-Visible Spectrophotometer See the Thermo OMNIC Help reference on page 49. Ultraviolet-Visible
1 Ultraviolet-Visible (UV-Vis) Spectroscopy Background Information Instructions for the Operation of the Cary 300 Bio UV-Visible Spectrophotometer See the Thermo OMNIC Help reference on page 49. Ultraviolet-Visible
Enzyme Kinetics: Properties of â-galactosidase
 Enzyme Kinetics: Properties of â-galactosidase Preparation for Laboratory: Read the introduction to this laboratory before doing the Web Tutorial - Beta Galactosidase. Additional background: Freeman, skim
Enzyme Kinetics: Properties of â-galactosidase Preparation for Laboratory: Read the introduction to this laboratory before doing the Web Tutorial - Beta Galactosidase. Additional background: Freeman, skim
Lab #11: Determination of a Chemical Equilibrium Constant
 Lab #11: Determination of a Chemical Equilibrium Constant Objectives: 1. Determine the equilibrium constant of the formation of the thiocyanatoiron (III) ions. 2. Understand the application of using a
Lab #11: Determination of a Chemical Equilibrium Constant Objectives: 1. Determine the equilibrium constant of the formation of the thiocyanatoiron (III) ions. 2. Understand the application of using a
Human Free Testosterone(F-TESTO) ELISA Kit
 Human Free Testosterone(F-TESTO) ELISA Kit Catalog Number. MBS700040 For the quantitative determination of human free testosterone(f-testo) concentrations in serum, plasma. This package insert must be
Human Free Testosterone(F-TESTO) ELISA Kit Catalog Number. MBS700040 For the quantitative determination of human free testosterone(f-testo) concentrations in serum, plasma. This package insert must be
THE LABORATORY NOTEBOOK
 THE LABORATORY NOTEBOOK In scientific work keeping a permanent record of all raw data, observations, calculations, et cetera obtained during an experiment is important. Therefore, a student must become
THE LABORATORY NOTEBOOK In scientific work keeping a permanent record of all raw data, observations, calculations, et cetera obtained during an experiment is important. Therefore, a student must become
What s in the Mix? Liquid Color Spectroscopy Lab (Randy Landsberg & Bill Fisher)
 What s in the Mix? Liquid Color Spectroscopy Lab (Randy Landsberg & Bill Fisher) Introduction: There is more to a color than a name. Color can tell us lots of information. In this lab you will use a spectrophotometer
What s in the Mix? Liquid Color Spectroscopy Lab (Randy Landsberg & Bill Fisher) Introduction: There is more to a color than a name. Color can tell us lots of information. In this lab you will use a spectrophotometer
Chemistry 111 Lab: Intro to Spectrophotometry Page E-1
 Chemistry 111 Lab: Intro to Spectrophotometry Page E-1 SPECTROPHOTOMETRY Absorption Measurements & their Application to Quantitative Analysis study of the interaction of light (or other electromagnetic
Chemistry 111 Lab: Intro to Spectrophotometry Page E-1 SPECTROPHOTOMETRY Absorption Measurements & their Application to Quantitative Analysis study of the interaction of light (or other electromagnetic
Evaluation copy. Case File 9. A Killer Cup of Coffee? GlobalTech manager dies
 Case File 9 Killer Cup of Coffee: Using colorimetry to determine concentration of a poison Determine the concentration of cyanide in the solution. A Killer Cup of Coffee? SOUTH PAINTER, Tuesday: It was
Case File 9 Killer Cup of Coffee: Using colorimetry to determine concentration of a poison Determine the concentration of cyanide in the solution. A Killer Cup of Coffee? SOUTH PAINTER, Tuesday: It was
Experiment 2 Kinetics II Concentration-Time Relationships and Activation Energy
 2-1 Experiment 2 Kinetics II Concentration-Time Relationships and Activation Energy Introduction: The kinetics of a decomposition reaction involving hydroxide ion and crystal violet, an organic dye used
2-1 Experiment 2 Kinetics II Concentration-Time Relationships and Activation Energy Introduction: The kinetics of a decomposition reaction involving hydroxide ion and crystal violet, an organic dye used
Exemplar for Internal Achievement Standard. Biology Level 2
 Exemplar for internal assessment resource Biology for Achievement Standard 91160 Exemplar for Internal Achievement Standard Biology Level 2 This exemplar supports assessment against: Achievement Standard
Exemplar for internal assessment resource Biology for Achievement Standard 91160 Exemplar for Internal Achievement Standard Biology Level 2 This exemplar supports assessment against: Achievement Standard
Impressions of a Stoma
 Huntington Library, Art Collections, and Botanical Gardens Impressions of a Stoma Overview Students use two different methods to view stomata on the underside of leaves. Introduction Plants exchange the
Huntington Library, Art Collections, and Botanical Gardens Impressions of a Stoma Overview Students use two different methods to view stomata on the underside of leaves. Introduction Plants exchange the
Rat creatine kinase MM isoenzyme (CK-MM) ELISA Kit
 Rat creatine kinase MM isoenzyme (CK-MM) ELISA Kit Catalog Number. CSB-E14405r For the quantitative determination of rat creatine kinase MM isoenzyme (CK-MM) concentrations in serum, plasma and tissue
Rat creatine kinase MM isoenzyme (CK-MM) ELISA Kit Catalog Number. CSB-E14405r For the quantitative determination of rat creatine kinase MM isoenzyme (CK-MM) concentrations in serum, plasma and tissue
QuickZyme Soluble Collagen Assay
 QuickZyme Soluble Collagen Assay Version June 2012 FOR RESEARCH USE ONLY NOT FOR USE IN DIAGNOSTIC PROCEDURES This package insert must be read in its entirety before using this product. Introduction Collagen
QuickZyme Soluble Collagen Assay Version June 2012 FOR RESEARCH USE ONLY NOT FOR USE IN DIAGNOSTIC PROCEDURES This package insert must be read in its entirety before using this product. Introduction Collagen
Enzyme Action: Testing Catalase Activity
 Enzyme Action: Testing Catalase Activity Experiment 6A Many organisms can decompose hydrogen peroxide (H 2 O 2 ) enzymatically. Enzymes are globular proteins, responsible for most of the chemical activities
Enzyme Action: Testing Catalase Activity Experiment 6A Many organisms can decompose hydrogen peroxide (H 2 O 2 ) enzymatically. Enzymes are globular proteins, responsible for most of the chemical activities
GlobalTech manager dies
 Case File 9 Killer Cup of Coffee: Using colorimetry to determine concentration of a poison Determine the concentration of cyanide in the solution. A Killer Cup of Coffee? GlobalTech manager dies SOUTH
Case File 9 Killer Cup of Coffee: Using colorimetry to determine concentration of a poison Determine the concentration of cyanide in the solution. A Killer Cup of Coffee? GlobalTech manager dies SOUTH
Lab 4: Osmosis and Diffusion
 Lab 4: Osmosis and Diffusion The plasma membrane enclosing every cell is the boundary that separates the cell from its external environment. It is not an impermeable barrier, but like all biological membranes,
Lab 4: Osmosis and Diffusion The plasma membrane enclosing every cell is the boundary that separates the cell from its external environment. It is not an impermeable barrier, but like all biological membranes,
Figure 1. Basic structure of the leaf, with a close up of the leaf surface showing Stomata and Guard cells.
 BIOL100 Laboratory Assignment 3: Analysis of Stomata Name: Stomata (singular=stoma) are the respiratory control structures in plants (see Figure 1 below). They are essentially small holes in the surface
BIOL100 Laboratory Assignment 3: Analysis of Stomata Name: Stomata (singular=stoma) are the respiratory control structures in plants (see Figure 1 below). They are essentially small holes in the surface
Microscope Lab Introduction to the Microscope Lab Activity
 Microscope Lab Introduction to the Microscope Lab Activity Wendy Kim 3B 24 Sep 2010 http://www.mainsgate.com/spacebio/modules/gs_resource/ CellDivisionMetaphase.jpeg 1 Introduction Microscope is a tool
Microscope Lab Introduction to the Microscope Lab Activity Wendy Kim 3B 24 Sep 2010 http://www.mainsgate.com/spacebio/modules/gs_resource/ CellDivisionMetaphase.jpeg 1 Introduction Microscope is a tool
Enzyme Activity Measuring the Effect of Enzyme Concentration
 6 Measuring the Effect of Enzyme Concentration Enzymes are proteins that serve as biological catalysts in a wide variety of life sustaining chemical reactions that take place in cells. As catalysts, enzymes
6 Measuring the Effect of Enzyme Concentration Enzymes are proteins that serve as biological catalysts in a wide variety of life sustaining chemical reactions that take place in cells. As catalysts, enzymes
Experiment #5: Qualitative Absorption Spectroscopy
 Experiment #5: Qualitative Absorption Spectroscopy One of the most important areas in the field of analytical chemistry is that of spectroscopy. In general terms, spectroscopy deals with the interactions
Experiment #5: Qualitative Absorption Spectroscopy One of the most important areas in the field of analytical chemistry is that of spectroscopy. In general terms, spectroscopy deals with the interactions
KINETIC DETERMINATION OF SELENIUM BY VISIBLE SPECTROSCOPY (VERSION 1.8)
 Selenium Determination, Page 1 KINETIC DETERMINATION OF SELENIUM BY VISIBLE SPECTROSCOPY I. BACKGROUND. (VERSION 1.8) The majority of reactions used in analytical chemistry possess the following characteristics:
Selenium Determination, Page 1 KINETIC DETERMINATION OF SELENIUM BY VISIBLE SPECTROSCOPY I. BACKGROUND. (VERSION 1.8) The majority of reactions used in analytical chemistry possess the following characteristics:
DNA Electrophoresis Lesson Plan
 DNA Electrophoresis Lesson Plan Primary Learning Outcomes: Students will learn how to properly load a well in an agarose gel. Students will learn how to analyze the results of DNA electrophoresis. Students
DNA Electrophoresis Lesson Plan Primary Learning Outcomes: Students will learn how to properly load a well in an agarose gel. Students will learn how to analyze the results of DNA electrophoresis. Students
To determine the mass of iron in one adult dose of either a ferrous sulfate or. ferrous gluconate iron supplement using a colorimetric technique.
 Lab: Colorimetric Analysis of Iron in Iron Supplements Purpose To determine the mass of iron in one adult dose of either a ferrous sulfate or ferrous gluconate iron supplement using a colorimetric technique.
Lab: Colorimetric Analysis of Iron in Iron Supplements Purpose To determine the mass of iron in one adult dose of either a ferrous sulfate or ferrous gluconate iron supplement using a colorimetric technique.
Table of Content. Enzymes and Their Functions Teacher Version 1
 Enzymes and Their Functions Jeisa Pelet, Cornell University Carolyn Wilczynski, Binghamton High School Cornell Learning Initiative in Medicine and Bioengineering (CLIMB) Table of Content Title Page Abstract..
Enzymes and Their Functions Jeisa Pelet, Cornell University Carolyn Wilczynski, Binghamton High School Cornell Learning Initiative in Medicine and Bioengineering (CLIMB) Table of Content Title Page Abstract..
Activity Sheets Enzymes and Their Functions
 Name: Date: Activity Sheets Enzymes and Their Functions amylase What are Enzymes? starch glucose Enzymes are compounds that assist chemical reactions by increasing the rate at which they occur. For example,
Name: Date: Activity Sheets Enzymes and Their Functions amylase What are Enzymes? starch glucose Enzymes are compounds that assist chemical reactions by increasing the rate at which they occur. For example,
Cells. Introduction WSBCTC 1
 Cells Cells are the fundamental unit of life. All living things are composed of cells. While there are several characteristics that are common to all cells, such as the presence of a cell membrane, cytoplasm,
Cells Cells are the fundamental unit of life. All living things are composed of cells. While there are several characteristics that are common to all cells, such as the presence of a cell membrane, cytoplasm,
Mouse glycated hemoglobin A1c(GHbA1c) ELISA Kit
 Mouse glycated hemoglobin A1c(GHbA1c) ELISA Kit Catalog Number. CSB-E08141m For the quantitative determination of mouse glycated hemoglobin A1c(GHbA1c) concentrations in lysate for RBC. This package insert
Mouse glycated hemoglobin A1c(GHbA1c) ELISA Kit Catalog Number. CSB-E08141m For the quantitative determination of mouse glycated hemoglobin A1c(GHbA1c) concentrations in lysate for RBC. This package insert
Chemistry 112 Laboratory Experiment 6: The Reaction of Aluminum and Zinc with Hydrochloric Acid
 Chemistry 112 Laboratory Experiment 6: The Reaction of Aluminum and Zinc with Hydrochloric Acid Introduction Many metals react with acids to form hydrogen gas. In this experiment, you will use the reactions
Chemistry 112 Laboratory Experiment 6: The Reaction of Aluminum and Zinc with Hydrochloric Acid Introduction Many metals react with acids to form hydrogen gas. In this experiment, you will use the reactions
Chemical reaction (slow): Enzyme-catalyzed reaction (much faster):
 1 Enzymes Introduction Enzymes are Biological Catalysts Recall that a catalyst is an agent which speeds up a chemical reaction without actually being consumed or changed by the reaction. Enzymes are proteins
1 Enzymes Introduction Enzymes are Biological Catalysts Recall that a catalyst is an agent which speeds up a chemical reaction without actually being consumed or changed by the reaction. Enzymes are proteins
Lab 5: Quantitative Analysis- Phosphates in Water By: A Generous Student. LBS 171L Section 9 TA: Dana October 27, 2005
 How uch Phosphate is the Body Being Exposed to During a Lifetime by Showering? Lab 5: Quantitative Analysis- Phosphates in Water By: A Generous Student LBS 171L Section 9 TA: Dana October 7, 005 [Note:
How uch Phosphate is the Body Being Exposed to During a Lifetime by Showering? Lab 5: Quantitative Analysis- Phosphates in Water By: A Generous Student LBS 171L Section 9 TA: Dana October 7, 005 [Note:
Enzymes: Amylase Activity in Starch-degrading Soil Isolates
 Enzymes: Amylase Activity in Starch-degrading Soil Isolates Introduction This week you will continue our theme of industrial microbiologist by characterizing the enzyme activity we selected for (starch
Enzymes: Amylase Activity in Starch-degrading Soil Isolates Introduction This week you will continue our theme of industrial microbiologist by characterizing the enzyme activity we selected for (starch
Bovine Vitamin B12 (VB12) ELISA Kit
 Bovine Vitamin B12 (VB12) ELISA Kit Catalog Number. CSB-E14089B For the quantitative determination of endogenic bovine vitamin B12 (VB12) concentrations in serum, plasma, tissue homogenates. This package
Bovine Vitamin B12 (VB12) ELISA Kit Catalog Number. CSB-E14089B For the quantitative determination of endogenic bovine vitamin B12 (VB12) concentrations in serum, plasma, tissue homogenates. This package
Rat creatine kinase MM isoenzyme (CK-MM) ELISA Kit
 Rat creatine kinase MM isoenzyme (CK-MM) ELISA Kit Catalog Number. CSB-E14405r For the quantitative determination of rat creatine kinase MM isoenzyme (CK-MM) concentrations in serum, plasma, tissue homogenates.
Rat creatine kinase MM isoenzyme (CK-MM) ELISA Kit Catalog Number. CSB-E14405r For the quantitative determination of rat creatine kinase MM isoenzyme (CK-MM) concentrations in serum, plasma, tissue homogenates.
HiPer Ion Exchange Chromatography Teaching Kit
 HiPer Ion Exchange Chromatography Teaching Kit Product Code: HTC001 Number of experiments that can be performed: 5 Duration of Experiment: Protocol: 5-6 hours Storage Instructions: The kit is stable for
HiPer Ion Exchange Chromatography Teaching Kit Product Code: HTC001 Number of experiments that can be performed: 5 Duration of Experiment: Protocol: 5-6 hours Storage Instructions: The kit is stable for
Use of Micropipettes
 Use of Micropipettes Prior to lab you should understand: The function of micropipettes in the laboratory Basic parts of micropipette What volumes are measured with P, P and P1 micopipettors How to read
Use of Micropipettes Prior to lab you should understand: The function of micropipettes in the laboratory Basic parts of micropipette What volumes are measured with P, P and P1 micopipettors How to read
Rat Creatine Kinase MB isoenzyme,ck-mb ELISA Kit
 Rat Creatine Kinase MB isoenzyme,ck-mb ELISA Kit Catalog No: E0479r 96 Tests Operating instructions www.eiaab.com FOR RESEARCH USE ONLY; NOT FOR THERAPEUTIC OR DIAGNOSTIC APPLICATIONS! PLEASE READ THROUGH
Rat Creatine Kinase MB isoenzyme,ck-mb ELISA Kit Catalog No: E0479r 96 Tests Operating instructions www.eiaab.com FOR RESEARCH USE ONLY; NOT FOR THERAPEUTIC OR DIAGNOSTIC APPLICATIONS! PLEASE READ THROUGH
Experiment 7 (Lab Period 8) Quantitative Determination of Phosphatase Activity
 Experiment 7 (Lab Period 8) Quantitative Determination of Phosphatase Activity Phosphatases are enzymes that catalyze the hydrolysis of organic-phosphate compounds, releasing inorganic phosphate from the
Experiment 7 (Lab Period 8) Quantitative Determination of Phosphatase Activity Phosphatases are enzymes that catalyze the hydrolysis of organic-phosphate compounds, releasing inorganic phosphate from the
NNIN Nanotechnology Education
 NNIN Nanotechnology Education Student Guide Part 1: Silver Nanoparticle Synthesis and Spectroscopy Introduction: In this lab you will synthesize silver nanoparticles one of the most commonly used nanoparticles
NNIN Nanotechnology Education Student Guide Part 1: Silver Nanoparticle Synthesis and Spectroscopy Introduction: In this lab you will synthesize silver nanoparticles one of the most commonly used nanoparticles
Photosynthesis and Respiration
 Photosynthesis and Respiration Experiment 31C Plants make sugar, storing the energy of the sun into chemical energy, by the process of photosynthesis. When they require energy, they can tap the stored
Photosynthesis and Respiration Experiment 31C Plants make sugar, storing the energy of the sun into chemical energy, by the process of photosynthesis. When they require energy, they can tap the stored
