NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE
|
|
|
- August Parsons
- 7 years ago
- Views:
Transcription
1 NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE INTERVENTIONAL PROCEDURES PROGRAMME Interventional procedure overview of endovenous mechanochemical ablation for varicose veins Treating varicose veins using a rotating wire and an injected chemical Varicose veins are veins that have become wider than normal and are unable to transport blood properly so that blood collects in them. This can cause heaviness, aching, throbbing, itching, cramps and fatigue in the legs. In severe cases, patients may develop skin discoloration or inflammation, and skin ulcers. In endovenous mechanochemical ablation, a tube with a rotating hollow wire at its tip is inserted into the affected vein in the leg. As the tube is pulled back out of the vein, the wire is rotated, damaging the lining of the vein. At the same time, a chemical is injected through the hollow wire into the vein. This causes the vein to become inflamed, then shrivel and close. Introduction The National Institute for Health and Clinical Excellence (NICE) has prepared this overview to help members of the Interventional Procedures Advisory Committee (IPAC) make recommendations about the safety and efficacy of an interventional procedure. It is based on a rapid review of the medical literature and specialist opinion. It should not be regarded as a definitive assessment of the procedure. Date prepared This overview was prepared in May 2012 and updated in November Procedure name Endovenous mechanochemical ablation for varicose veins Specialist societies British Society of Interventional Radiology The Vascular Society of Great Britain and Ireland Page 1 of 16
2 Description Indications and current treatment Varicose veins are a sign of underlying venous insufficiency and affect 20 30% of adults. Most people with varicose veins have no symptoms, but venous insufficiency may cause fatigue, heaviness, aching, throbbing, itching and cramps in the legs. Chronic venous insufficiency can lead to skin discoloration, inflammatory dermatitis and ulceration. Long (or great) saphenous vein insufficiency is the most common form of venous insufficiency in people presenting with symptoms. Because most varicose veins do not cause serious health problems, treatment is not usually needed on medical grounds. Conservative methods such as compression hosiery (support stockings or tights) may help people with symptomatic varicose veins. If symptoms are severe the main treatment options include sclerotherapy, endovenous laser treatment, radiofrequency ablation and surgery (usually stripping and ligation of the long and short saphenous veins, and phlebectomies). What the procedure involves Endovenous mechanochemical ablation for varicose veins combines mechanical and chemical ablation to close veins without the need for tumescent anaesthesia (infusion of a large volume of dilute local anaesthetic around and along the entire length of vein to be treated). The procedure is carried out using local anaesthesia at the catheter insertion site. Ultrasound imaging is used to identify the target vein (usually the long saphenous vein), its diameter and the treatment length, which depends on perforators and tributaries. An infusion catheter with a motor drive is introduced percutaneously into the distal end of the target vein and, in the case of the long saphenous vein, the catheter tip is advanced to the saphenofemoral junction. A dispersion wire that extends through the catheter lumen is rotated to damage the epithelium and a sclerosant is infused simultaneously as the catheter is slowly pulled back through the vein. After the procedure, ultrasound may be used to check for target vein occlusion and patency of the common femoral vein. Patients are advised to wear compression stockings for approximately 2 weeks after the procedure. Clinical assessment The CEAP (clinical, etiological, anatomic and pathophysiologic) classification from the American Venous Forum is often used to classify venous disease of the lower limb. Clinical signs are classified as: C0 no signs of venous disease; C1 telangiectases or reticular veins; C2 varicose veins; C3 oedema; C4a pigmentation or eczema; C4b lipodermatosclerosis or atrophie blanche; C5 healed venous ulcer; C6 active venous ulcer. Page 2 of 16
3 Literature review Rapid review of literature The medical literature was searched to identify studies and reviews relevant to endovenous mechanochemical ablation for varicose veins. Searches were conducted of the following databases, covering the period from their commencement to 25 September 2012: MEDLINE, PREMEDLINE, EMBASE, Cochrane Library and other databases. Trial registries and the Internet were also searched. No language restriction was applied to the searches (see appendix C for details of search strategy). Relevant published studies identified during consultation or resolution that are published after this date may also be considered for inclusion. The following selection criteria (table 1) were applied to the abstracts identified by the literature search. Where selection criteria could not be determined from the abstracts the full paper was retrieved. Table 1 Inclusion criteria for identification of relevant studies Characteristic Publication type Patient Intervention/test Outcome Language Criteria Clinical studies were included. Emphasis was placed on identifying good-quality studies. Abstracts were excluded where no clinical outcomes were reported, or where the paper was a review, an editorial, or a laboratory or animal study. Conference abstracts were also excluded because of the difficulty of appraising study methodology, unless they reported specific adverse events that were not available in the published literature. Patients with varicose veins. Endovenous mechanochemical ablation. Articles were retrieved if the abstract contained information relevant to the safety and/or efficacy. Non-English-language articles were excluded unless they were thought to add substantively to the English-language evidence base. List of studies included in the overview This overview is based on approximately 60 patients from 2 case series and 6 case reports 1 3. Other studies that were considered to be relevant to the procedure but were not included in the main extraction table (table 2) have been listed in appendix A. Page 3 of 16
4 Table 2 Summary of key efficacy and safety findings on endovenous mechanochemical ablation for varicose veins Abbreviations used: CEAP, clinical, etiological, anatomical and pathophysiologic elements classification; DVT, deep vein thrombosis; GSV, great saphenous vein; IQR, interquartile range Study details Key efficacy findings Key safety findings Comments Elias S (2012) 1 Case series USA Recruitment period: 2009 Study population: patients with significant GSV reflux n=29 patients (30 veins) Mean age: 54 years (range 31 90) Sex: 60% female Mean length of treated vein: 37.5 cm (range 24 47) Patient selection criteria: Reflux as documented by ultrasound extending for 0.5 seconds or longer. CEAP Class 1 patients were excluded. Other exclusion criteria were acute DVT, immobility, anticoagulation and GSV diameters >12 mm. Technique: Clarivein system (Vascular Insights LLC, Madison, USA) was used with 1.5% liquid sodium tetradecyl sulphate. Procedures were limited to the GSV. No secondary treatments used. Mean follow-up: 9 months (range days) Conflict of interest/source of funding: The research was sponsored in part by Vascular Insights LLC. Number of patients analysed: 29 (30 veins) Average treatment time=14 minutes Primary closure rate at mean follow-up of 260 days (based on ultrasound)=96.7% (29/30) One vein recanalised between the 1-week and 1-month visits (recanalisation was seen on ultrasound but the treated vein was not refluxing). Patients did not complain of pain during the procedure. Three minor thigh ecchymoses were observed at levels where the rotating wire may have caught on a valve cusp or vein wall. No DVT, nerve or skin injury occurred. No patient complained of paraesthesia, hypoaesthesia or motor dysfunction. Follow-up issues: There were no losses to followup. Study design issues: The primary objectives of the study were to determine the overall safety of the procedure and to define recanalisation of treated veins at 6 months. Study population issues: 77% of patients were in Class 2 according to the CEAP classification, 7% were in Class 3 and 16% were in Class 4a. Page 4 of 16
5 Abbreviations used: CEAP, clinical, etiological, anatomical and pathophysiologic elements classification; DVT, deep vein thrombosis; GSV, great saphenous vein; IQR, interquartile range Study details Key efficacy findings Key safety findings Comments Van Eekeren R (2011) 2 Case series The Netherlands Recruitment period: not stated Study population: patients with varicose veins and primary GSV incompetence n=25 patients (30 veins) Mean age: 52 years Sex: 72% (18/25) female Mean length of treated vein: 40 cm Patient selection criteria: age over 18 years; C2 C6 varicose veins; primary GSV incompetence (reflux was defined as retrograde flow of >0.5 seconds after calf compression measured in an upright position). Exclusion criteria included pregnancy and lactation, allergy or contraindication to the sclerosant, previous surgical treatment of varicose veins, history of DVT, anticoagulation, severe tortuosity of the GSV, and GSV diameters less than 4 mm or greater than 12 mm measured in the supine position. Technique: Clarivein system (Vascular Insights LLC, Madison, USA) was used with 1.5% polidocanol. Procedures were limited to the GSV. Mean follow-up: 6 weeks Conflict of interest/source of funding: 1 of the authors was Principal Investigator for the US clinical study that was sponsored in part by Vascular Insights LLC. Number of patients analysed: 25 (30 veins) Average treatment time=20 minutes Technical success Initial technical success (occlusion identified by ultrasound)=100% Recanalisation Complete occlusion at 6 weeks=87% (26/30) of veins (assessed by duplex ultrasound). Two patients had partial recanalisation of the proximal GSV 15 and 18 cm, respectively, from the saphenofemoral junction; 1 patient had partial recanalisation of the distal GSV. One patient had total recanalisation of the GSV, which was successfully treated by a repeat procedure at 7 weeks after initial treatment. Median venous clinical severity score (scale 0 30, lower scores indicating less severe disease) Baseline=3.0 (IQR ) 6 weeks after the procedure=1.0 (IQR ), p<0.001 In 1 patient with superficial phlebitis, the venous clinical severity score worsened from 5 to 11. Patient satisfaction After 6 weeks, median patient satisfaction=8.5 (IQR 8 9, on a scale of 0 10) No major adverse events were observed. There was no deep vein thrombosis, nerve injury, skin necrosis, infection, or hyperpigmentation. Minor complications : localised ecchymosis at the puncture site=30% (9/30) transient superficial phlebitis of distal tributaries (resolved within a week)=13% (4/30) During the procedure, the median maximal pain score was 4 (on a scale of 0 10). On day 1 after the procedure, the mean maximal pain was 9 mm (on a scale of mm), which decreased to 2 mm at 7 days after the procedure. All 4 patients with superficial phlebitis had prolonged pain for >1 week. At 6 weeks, no additional complications were observed clinically or with duplex ultrasonography. Follow-up issues: There were no losses to followup. Study design issues: The primary outcome measures were immediate occlusion and postprocedural complications. Pain was measured during the procedure using a scale of 0 10 (where 0 indicated no pain). After the procedure, a visual analogue scale of mm was used. Patient satisfaction was determined on a scale of 0 10, with higher scores indicating greater satisfaction. The venous clinical severity score includes 9 clinical characteristics of chronic venous disease, measured on a scale of Study population issues: 60% (18/30) of veins were in Class 2 according to the CEAP classification, 30% (9/30) were in Class 3 and 10% (3/30) were in Class 4. Five patients had bilateral GSV insufficiency. Other issues The authors note that they have changed several aspects of the procedure following the results of this initial series of patients (including an increase in the concentration of sclerosant). Page 5 of 16
6 Abbreviations used: CEAP, clinical, etiological, anatomical and pathophysiologic elements classification; DVT, deep vein thrombosis; GSV, great saphenous vein; IQR, interquartile range Study details Key efficacy findings Key safety findings Comments FDA (MAUDE database) 3 Case reports Report dates: adverse events reported in relation to the use of Clarivein Infusion catheter (Vascular Insights LLC) 4 of the 6 events occurred after treatment of the short saphenous vein (events 1,2,3 and 5). 1. At the first post-procedure followup, venous duplex ultrasound showed a clot in the right popliteal vein. The patient was asymptomatic and was treated conservatively. 2. Four days after the procedure, venous duplex ultrasound showed a left popliteal DVT. The patient was asymptomatic and was treated conservatively. 3. Four days after the procedure, venous duplex ultrasound showed a right popliteal DVT with clot adherent to the vein wall. The patient was treated conservatively. Follow-up ultrasound showed no further propagation of DVT and the patient remains asymptomatic. 4. Three days after the procedure, the patient developed shortness of breath, left-sided pain on inspiration, and marked fatigue. She was treated for CT-confirmed pulmonary embolism and subsequently discharged from hospital with a good prognosis. 5. At the first postprocedure follow-up, venous duplex ultrasound showed subocclusive acute thrombus. The patient was asymptomatic and was treated conservatively. At a second follow-up, the patient was asymptomatic. The clot was still present but had retracted. 6. At the first postprocedure follow-up, venous duplex ultrasound showed DVT. The patient was asymptomatic and was treated conservatively. Page 6 of 16
7 Efficacy Occlusion rates A case series of 29 patients reported a primary closure rate of 97% (29/30 veins) at a mean follow-up of 260 days (assessed by ultrasound) 1. A case series of 25 patients reported complete occlusion in 87% (26/30) of veins at 6-week follow-up (assessed by duplex ultrasound) 2. In this case series, 3 patients had a partial recanalisation and 1 patient had a total recanalisation (which was successfully treated by a repeat procedure 7 weeks after the initial procedure). It should be noted that the 2 case series used different sclerosants. Venous clinical severity score The case series of 25 patients reported a statistically significant reduction in median venous clinical severity score (scale 0 30, higher scores indicating more severe disease) from 3 (interquartile range 2 5) at baseline to 1 (interquartile range 0.3 3) at 6-week follow-up (p<0.001) 2. Patient satisfaction The case series of 25 patients reported a median patient satisfaction score of 8.5 (scale 0 10, higher scores indicating increased patient satisfaction) 2. Safety Deep vein thrombosis Deep vein thrombosis after the procedure was reported in 4 patients (reports submitted to the Food and Drug Administration [FDA] manufacturer and user facility device experience [MAUDE] database); 3 of these thromboses occurred after treatment of the short saphenous vein 3. All 4 patients were asymptomatic and were treated conservatively. Pulmonary embolism Pulmonary embolism after the procedure was reported in 1 patient (report submitted to the FDA MAUDE database) 3. After treatment, the patient was discharged from hospital with a good prognosis. Ecchymosis Minor thigh ecchymosis was reported in 10% (3/30) of limbs in the case series of 29 patients 1. Localised ecchymosis at the puncture site was reported in 30% (9/30) of limbs in the case series of 25 patients 2. Page 7 of 16
8 Phlebitis Transient superficial phlebitis of distal tributaries was reported in 13% (4/30) of limbs in the case series of 25 patients 2. Validity and generalisability of the studies The evidence consists of 2 small case series with short-term follow-up. The 2 studies used different sclerosants; the type and concentration of sclerosant is likely to have an effect on the efficacy of the procedure. Both the studies report results from the initial series of patients treated by the procedure at the study centres. Existing assessments of this procedure There were no published assessments from other organisations identified at the time of the literature search. Related NICE guidance Below is a list of NICE guidance related to this procedure. Appendix B gives details of the recommendations made in each piece of guidance listed. Interventional procedures Ultrasound-guided foam sclerotherapy for varicose veins. NICE interventional procedure guidance 314 (2009). Available from Endovenous laser treatment of the long saphenous vein. NICE interventional procedure guidance 52 (2004). Available from Transilluminated powered phlebectomy for varicose veins. NICE interventional procedure guidance 37 (2004). Available from Radiofrequency ablation of varicose veins. NICE interventional procedure guidance 8 (2003). Available from Specialist Advisers opinions Specialist advice was sought from consultants who have been nominated or ratified by their Specialist Society or Royal College. The advice received is their individual opinion and does not represent the view of the society. Mr M Gaunt (British Society of Interventional Radiology); Mr E Chaloner, Mr I Franklin, Mr T Lees, Mr I Nyamekye, Professor R Sayers, Professor M Thompson (The Vascular Society) Page 8 of 16
9 Three Specialist Advisers perform the procedure regularly; 4 have never performed the procedure. Three Specialist Advisers described the procedure as the first in a new class of procedure, 2 described it as a minor variation of an existing procedure, 1 described it as established practice and 1 described it as novel and of uncertain safety and efficacy. The comparators would be conventional open surgery, endovenous thermal ablation (radiofrequency or laser), and liquid or foam sclerotherapy. One Specialist Adviser reported that he had seen 2 cases of deep vein thrombosis, both in patients having short saphenous veins treated, and has since stopped treating short saphenous veins. Adverse events reported in the literature include ecchymosis and phlebitis. Additional theoretical adverse events include visual and neurological disturbance, allergy to sclerosant, haemorrhage, infection, embolisation, vein wall perforation and skin pigmentation. Key efficacy outcomes include vein occlusion (up to 5 years after treatment), absence of full or partial recanalisations after the treatment (seen on duplex scan but not necessarily clinically apparent), length of treated truncal vein occluded by initial treatment, reduction of clinical symptoms, improved periand post-procedural pain scores and improved quality of life. There are no data on long-term vein occlusion rates. Two Specialist Advisers noted that a UK trial is due to start shortly (at Charing Cross Hospital). One Specialist Adviser noted that there are uncertainties with regard to the incidence and extent of recanalisation of treated truncal veins at 1 and 2 years and the clinical significance of this. Three Specialist Advisers considered the procedure to have a potentially moderate impact on the NHS, in terms of patients and use of resources; 2 Specialist Advisers thought the impact would be minor. Patient Commentators opinions NICE s Patient and Public Involvement Programme was unable to gather patient commentary for this procedure. Issues for consideration by IPAC None other than those described above. Page 9 of 16
10 References 1. Elias S, Raines JK (2012) Mechanochemical tumescentless endovenous ablation: final results of the initial clinical trial. Phlebology 27: van Eekeren RRJP, Boersma D, Elias S et al. (2011) Endovenous mechanochemical ablation of great saphenous vein incompetence using the ClariVein device: a safety study. Journal of Endovascular Therapy 18: Food and Drug Administration (FDA). Manufacturer and user facility device experience (MAUDE) database. Available from: sttimeout=500 [accessed 13 June 2012] Page 10 of 16
11 Appendix A: Additional papers on endovenous mechanochemical ablation for varicose veins There were no additional papers identified. Page 11 of 16
12 Appendix B: Related NICE guidance for endovenous mechanochemical ablation for varicose veins Guidance Interventional procedures Recommendations Ultrasound-guided foam sclerotherapy for varicose veins. NICE interventional procedure guidance 314 (2009). 1.1 Current evidence on ultrasound-guided foam sclerotherapy for varicose veins shows that it is efficacious in the short term. The evidence on safety includes systemic side effects in some patients. Therefore, this procedure should only be used with special arrangements for clinical governance, consent and audit. 1.2 Clinicians wishing to undertake ultrasound-guided foam sclerotherapy for varicose veins should take the following actions. Inform the clinical governance leads in their Trusts. Ensure that patients understand the potential side effects of the procedure, including those thought to be associated with foam embolisation, and provide them with clear written information, which should include other treatment options. In addition, the use of NICE s information for patients ( Understanding NICE guidance ) is recommended (available from Audit and review clinical outcomes of all patients having ultrasoundguided foam sclerotherapy for varicose veins (see section 3.1). 1.3 The procedure should only be undertaken using sclerosants licensed for varicose veins. The practice of mixing liquid sclerosant with air or other gas constitutes off-label use. 1.4 Current evidence of long-term efficacy is limited and clinicians are encouraged to collect longer-term follow-up data. Endovenous laser treatment of the long saphenous vein. NICE interventional procedure guidance 52 (2004). 1.1 Current evidence on the safety and efficacy of endovenous laser treatment of the long saphenous vein appears adequate to support the use of this procedure provided that the normal arrangements are in place for consent, audit and clinical governance. Current evidence on the efficacy of this procedure is limited to case series with up to 3 years follow-up. Clinicians are encouraged to collect longer-term follow up data. Transilluminated powered phlebectomy for varicose veins. NICE interventional procedure guidance 37 (2004). 1.1 Current evidence on the safety and efficacy of transilluminated powered phlebectomy for varicose veins includes small numbers of patients and is of limited quality. It does not appear adequate to support the use of this procedure without special arrangements for consent and for audit or research. Clinicians wishing to undertake transilluminated powered phlebectomy for varicose veins should inform the clinical governance leads in their Trusts. They should ensure that patients offered it understand the uncertainty about the Page 12 of 16
13 procedure s safety and efficacy and should provide them with clear written information. Use of the Institute s Information for the Public is recommended. Clinicians should ensure that appropriate arrangements are in place for audit or research. Publication of safety and efficacy outcomes will be useful in reducing the current uncertainty. NICE is not undertaking further investigation at present. Radiofrequency ablation of varicose veins. NICE interventional procedure guidance 8 (2003). 1.1 Current evidence on the safety and efficacy of radiofrequency ablation of varicose veins appears adequate to support the use of this procedure as an alternative to saphenofemoral ligation and stripping, provided that the normal arrangements are in place for consent, audit and clinical governance. Page 13 of 16
14 Appendix C: Literature search for endovenous mechanochemical ablation for varicose veins Databases Cochrane Database of Systematic Reviews CDSR (Cochrane Library) Date searched Version/files 25/09/12 Issue 9 of 12, Sept 2012 Database of Abstracts of Reviews of 25/09/12 - Effects DARE (CRD website) HTA database (CRD website) 25/09/12 - Cochrane Central Database of Controlled Trials CENTRAL (Cochrane Library) 25/09/12 Issue 9 of 12, Sept 2012 MEDLINE (Ovid) 25/09/ to September Week MEDLINE In-Process (Ovid) 25/09/12 September 24, 2012 EMBASE (Ovid) 25/09/ to 2012 Week 38 CINAHL (NLH Search 2.0 or EBSCOhost) 25/09/12 - Trial sources searched: Current Controlled Trials metaregister of Controlled Trials mrct Clinicaltrials.gov National Institute for Health Research Clinical Research Network Coordinating Centre (NIHR CRN CC) Portfolio Database Websites searched: National Institute for Health and Clinical Excellence (NICE) Food and Drug Administration (FDA) MAUDE database French Health Authority (FHA) Australian Safety and Efficacy Register of New Interventional Procedures Surgical (ASERNIP S) Australia and New Zealand Horizon Scanning Network (ANZHSN) Conference search General internet search The following search strategy was used to identify papers in MEDLINE. A similar strategy was used to identify papers in other databases. 1 exp Venous Insufficiency/ 2 ((venous or vein*) adj3 (incomp* or insuffic*)).tw. Page 14 of 16
15 3 ((venous or vein*) adj3 ulcer*).tw. 4 telangiectasis/ 5 telangiect*.tw. 6 ((reticular or thread or spider) adj3 (vein* or venous)).tw. 7 or/1-6 8 exp lower extremity/ 9 (lower limb* or lower extremit* or leg* or calf or valves or thigh* or membrum inferius).tw. 10 or/ and exp varicose veins/ 13 (varicos* adj3 vein*).tw. 14 (varix or varices or microvaricosity or phlebarteriectasia or phlebectas* or prevaricos* or vein ectasia or venectasia).tw. 15 Saphenous Vein/ 16 ((saphenous or perforator) adj3 (vein* or vena or incomp* or insuffic*)).tw. 17 GSV.tw. 18 or/ clarivein.tw. 20 MOCA.tw. 21 ((mechanochemical or mechano-chemical or mechanical) adj3 ablat*).tw. 22 ((non-thermal or nonthermal or "non thermal") adj3 ablat*).tw. 23 (infus* adj3 catheter*).tw. 24 ((damag* or disrupt* or disturb* or destroy* or break* or destruct*) adj3 (endothelium or endothelial or lining)).tw. 25 (rotat* adj3 (wire* or tip*)).tw. 26 tumescentless.tw. 27 ((spasm* adj2 vein*) or venospasm).tw. Page 15 of 16
16 28 or/ and animals/ not humans/ not 30 Page 16 of 16
Surgical Options for Venous Disease. Sandra C Carr MD Vascular Surgery Meriter Wisconsin Heart
 Surgical Options for Venous Disease Sandra C Carr MD Vascular Surgery Meriter Wisconsin Heart Chronic Venous Disease Approximately 23% of adults in the US have varicose veins Estimated 22 million women
Surgical Options for Venous Disease Sandra C Carr MD Vascular Surgery Meriter Wisconsin Heart Chronic Venous Disease Approximately 23% of adults in the US have varicose veins Estimated 22 million women
TREATMENT OF VARICOSE VEINS: CAN IT BE IMPROVED BY MECHANOCHEMICAL ABLATION USING THE CLARIVEIN DEVICE?
 TREATMENT OF VARICOSE VEINS: CAN IT BE IMPROVED BY MECHANOCHEMICAL ABLATION USING THE CLARIVEIN DEVICE? Michel MJP Reijnen Rijnstate Hospital, Arnhem The Netherlands mmpj.reijnen@gmail.com Disclosures
TREATMENT OF VARICOSE VEINS: CAN IT BE IMPROVED BY MECHANOCHEMICAL ABLATION USING THE CLARIVEIN DEVICE? Michel MJP Reijnen Rijnstate Hospital, Arnhem The Netherlands mmpj.reijnen@gmail.com Disclosures
Medicare C/D Medical Coverage Policy
 Varicose Vein Treatment Origination Date: June 1, 1993 Review Date: July 20, 2016 Next Review: July, 2018 Medicare C/D Medical Coverage Policy DESCRIPTION OF PROCEDURE OR SERVICE Varicose veins of the
Varicose Vein Treatment Origination Date: June 1, 1993 Review Date: July 20, 2016 Next Review: July, 2018 Medicare C/D Medical Coverage Policy DESCRIPTION OF PROCEDURE OR SERVICE Varicose veins of the
Medicare C/D Medical Coverage Policy
 Varicose Vein Treatment Medicare C/D Medical Coverage Policy Origination Date: June 1, 1993 Review Date: September 16, 2015 Next Review: September, 2017 DESCRIPTION OF PROCEDURE OR SERVICE Varicose veins
Varicose Vein Treatment Medicare C/D Medical Coverage Policy Origination Date: June 1, 1993 Review Date: September 16, 2015 Next Review: September, 2017 DESCRIPTION OF PROCEDURE OR SERVICE Varicose veins
Effective Date: March 2, 2016
 Medical Review Criteria Varicose Vein Procedures Effective Date: March 2, 2016 Treatmetn Subject: Varicose Vein Procedures VeinTreatment of Varicose Policy: Veins HPHC covers specific non-experimental
Medical Review Criteria Varicose Vein Procedures Effective Date: March 2, 2016 Treatmetn Subject: Varicose Vein Procedures VeinTreatment of Varicose Policy: Veins HPHC covers specific non-experimental
Yes when meets criteria below
 Vein Disease Treatment MP9241 Covered Service: Prior Authorization Required: Additional Information: Medicare Policy: BadgerCare Plus Policy: Yes when meets criteria below Yes None Dean Health Plan covers
Vein Disease Treatment MP9241 Covered Service: Prior Authorization Required: Additional Information: Medicare Policy: BadgerCare Plus Policy: Yes when meets criteria below Yes None Dean Health Plan covers
PROVIDER POLICIES & PROCEDURES
 PROVIDER POLICIES & PROCEDURES SCLEROTHERAPY TREATMENT OF SUPERFICIAL VARICOSE VEINS OF THE LEGS The purpose of this document is to provide guidance to providers enrolled in the Connecticut Medical Assistance
PROVIDER POLICIES & PROCEDURES SCLEROTHERAPY TREATMENT OF SUPERFICIAL VARICOSE VEINS OF THE LEGS The purpose of this document is to provide guidance to providers enrolled in the Connecticut Medical Assistance
Clinical Medical Policy Varicose Vein Treatment
 Benefit Coverage Covered Benefit for lines of business including: Health Benefits Exchange (HBE), Rite Care (MED), Children with Special Needs (CSN), Substitute Care (SUB), Rhody Health Partners (RHP),
Benefit Coverage Covered Benefit for lines of business including: Health Benefits Exchange (HBE), Rite Care (MED), Children with Special Needs (CSN), Substitute Care (SUB), Rhody Health Partners (RHP),
Spotlight Series: Interventional Radiology. Varicose Veins and Venous Insufficiency
 Spotlight Series: Interventional Radiology Varicose Veins and Venous Insufficiency What is venous insufficiency? Spectrum of Disease Spider veins and telangiectasias Small reddish and purple veins near
Spotlight Series: Interventional Radiology Varicose Veins and Venous Insufficiency What is venous insufficiency? Spectrum of Disease Spider veins and telangiectasias Small reddish and purple veins near
Non-surgical treatment of severe varicose veins
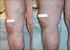 Non-surgical treatment of severe varicose veins Yasu Harasaki UCHSC Department of Surgery General Surgery Grand Rounds March 19, 2007 Definition Dilated, palpable, subcutaneous veins generally >3mm in
Non-surgical treatment of severe varicose veins Yasu Harasaki UCHSC Department of Surgery General Surgery Grand Rounds March 19, 2007 Definition Dilated, palpable, subcutaneous veins generally >3mm in
Clinical Review Criteria
 Clinical Review Criteria Treatment of Varicose Veins Radiofrequency Catheter Closure Sclerotherapy Surgical Stripping Trivex System for Outpatient Varicose Vein Surgery VenaSeal Closure System VNUS Closure
Clinical Review Criteria Treatment of Varicose Veins Radiofrequency Catheter Closure Sclerotherapy Surgical Stripping Trivex System for Outpatient Varicose Vein Surgery VenaSeal Closure System VNUS Closure
MEDICAL COVERAGE POLICY. SERVICE: Varicose Veins of the Lower Extremities. PRIOR AUTHORIZATION: Required.
 Page 1 of 5 MEDICAL COVERAGE POLICY Important note Even though this policy may indicate that a particular service or supply may be considered covered, this conclusion is not based upon the terms of your
Page 1 of 5 MEDICAL COVERAGE POLICY Important note Even though this policy may indicate that a particular service or supply may be considered covered, this conclusion is not based upon the terms of your
Peninsula Commissioning Priorities Group. Commissioning Policy Varicose Vein Referral
 NHS Devon NHS Plymouth Torbay Care Trust Peninsula Commissioning Priorities Group Commissioning Policy Varicose Vein Referral Varicose Vein Referral Guidelines 1. Description of service/treatment Most
NHS Devon NHS Plymouth Torbay Care Trust Peninsula Commissioning Priorities Group Commissioning Policy Varicose Vein Referral Varicose Vein Referral Guidelines 1. Description of service/treatment Most
Modern Management of Varicose Veins
 Modern Management of Varicose Veins GPCME 2008 David Ferrar Vascular and Endovascular Surgeon Vascular Ultrasound Specialist Multimodality treatment Compression stockings Sclerotherapy Ultrasound guided
Modern Management of Varicose Veins GPCME 2008 David Ferrar Vascular and Endovascular Surgeon Vascular Ultrasound Specialist Multimodality treatment Compression stockings Sclerotherapy Ultrasound guided
AMERICAN VENOUS FORUM
 Revised Venous Clinical Severity Score AMERICAN VENOUS FORUM Pain : 0 Mild: 1 or other discomfort (ie, aching, heaviness, fatigue, soreness, burning) origin Occasional pain or other discomfort (ie, not
Revised Venous Clinical Severity Score AMERICAN VENOUS FORUM Pain : 0 Mild: 1 or other discomfort (ie, aching, heaviness, fatigue, soreness, burning) origin Occasional pain or other discomfort (ie, not
treatment of varicose and spider veins patient information SAMPLE a publication by advancing vein care
 treatment of varicose and spider veins patient information a publication by advancing vein care Since most veins lie deep to the skin s surface, vein disorders are not always visible to the naked eye.
treatment of varicose and spider veins patient information a publication by advancing vein care Since most veins lie deep to the skin s surface, vein disorders are not always visible to the naked eye.
Treating varicose veins with foam injections using ultrasound guidance
 Issue date February 2013 Information for the public NICE interventional procedures guidance advises the NHS on when and how new procedures can be used in clinical practice. Treating varicose veins with
Issue date February 2013 Information for the public NICE interventional procedures guidance advises the NHS on when and how new procedures can be used in clinical practice. Treating varicose veins with
Health Technology Assessment of Scheduled Surgical Procedures
 HTA of Scheduled Surgical Procedures April 2013 Health Technology Assessment of Scheduled Surgical Procedures Varicose Vein Surgery April 2013 Safer Better Care About the The (HIQA) is the independent
HTA of Scheduled Surgical Procedures April 2013 Health Technology Assessment of Scheduled Surgical Procedures Varicose Vein Surgery April 2013 Safer Better Care About the The (HIQA) is the independent
TREATMENT OF VARICOSE AND SPIDER VEINS Patient Info
 TREATMENT OF VARICOSE AND SPIDER VEINS Patient Info www.heartofthevillages.com TIRED & ACHING LEGS? If you suffer from varicose and spider veins, you are not alone. It is estimated that there are more
TREATMENT OF VARICOSE AND SPIDER VEINS Patient Info www.heartofthevillages.com TIRED & ACHING LEGS? If you suffer from varicose and spider veins, you are not alone. It is estimated that there are more
Varicose veins - 1 -
 Varicose veins - 1 - Varicose Veins About 3 in 10 adults develop varicose veins at some time in their life. Most people with varicose veins do not have an underlying disease and they usually occur for
Varicose veins - 1 - Varicose Veins About 3 in 10 adults develop varicose veins at some time in their life. Most people with varicose veins do not have an underlying disease and they usually occur for
CHAPTER 15 SCLEROTHERAPY FOR VENOUS DISEASE
 Introduction CHAPTER 15 SCLEROTHERAPY FOR VENOUS DISEASE Original authors: Niren Angle, John J. Bergan, Joshua I. Greenberg, and J. Leonel Villavicencio Abstracted by Teresa L. Carman New technology has
Introduction CHAPTER 15 SCLEROTHERAPY FOR VENOUS DISEASE Original authors: Niren Angle, John J. Bergan, Joshua I. Greenberg, and J. Leonel Villavicencio Abstracted by Teresa L. Carman New technology has
Microfoam Ultrasound-Guided Sclerotherapy of Varicose Veins in 100 Legs
 Microfoam Ultrasound-Guided Sclerotherapy of Varicose Veins in 1 Legs JOHN M. BARRETT, FRNZCGP, n BRUCE ALLEN, FRACR, n ANNE OCKELFORD, FRNZCGP, n AND MITCHEL P. GOLDMAN, MD w n Palm Clinic, Auckland,
Microfoam Ultrasound-Guided Sclerotherapy of Varicose Veins in 1 Legs JOHN M. BARRETT, FRNZCGP, n BRUCE ALLEN, FRACR, n ANNE OCKELFORD, FRNZCGP, n AND MITCHEL P. GOLDMAN, MD w n Palm Clinic, Auckland,
Medical Affairs Policy
 Service: Varicose Vein Treatments PUM 250-0032 Medical Affairs Policy Implemented 04/04/14, 04/01/15, 4/1/16 Reviewed 12/12/14 Revised 12/12/14, 12/11/15 Developed Arise/WPS Policy 12/12/14, 12/11/15 Committee
Service: Varicose Vein Treatments PUM 250-0032 Medical Affairs Policy Implemented 04/04/14, 04/01/15, 4/1/16 Reviewed 12/12/14 Revised 12/12/14, 12/11/15 Developed Arise/WPS Policy 12/12/14, 12/11/15 Committee
Corporate Medical Policy
 Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: varicose_veins_treatment_for 12/2000 11/2014 11/2015 11/2014 Description of Procedure or Service A variety
Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: varicose_veins_treatment_for 12/2000 11/2014 11/2015 11/2014 Description of Procedure or Service A variety
MEDICAL COVERAGE POLICY SERVICE: Varicose Veins of the Lower Extremities. SERVICE: Treatment of Varicose Veins of the Lower Extremities
 Important note Even though this policy may indicate that a particular service or supply may be considered covered, this conclusion is not based upon the terms of your particular benefit plan. Each benefit
Important note Even though this policy may indicate that a particular service or supply may be considered covered, this conclusion is not based upon the terms of your particular benefit plan. Each benefit
Information for patients who require Foam Sclerotherapy for Varicose Veins.
 Information for patients who require Foam Sclerotherapy for Varicose Veins. Why do I need this procedure? Everybody has two sets of veins in the legs. These include the superficial and deep veins. Their
Information for patients who require Foam Sclerotherapy for Varicose Veins. Why do I need this procedure? Everybody has two sets of veins in the legs. These include the superficial and deep veins. Their
Recurrent Varicose Veins. Vineet Mishra, MD Director of Mohs Surgery and Procedural Dermatology University of Texas Health Science Center San Antonio
 Recurrent Varicose Veins Vineet Mishra, MD Director of Mohs Surgery and Procedural Dermatology University of Texas Health Science Center San Antonio Disclosures None Possible Causes of Recurrence DNA:
Recurrent Varicose Veins Vineet Mishra, MD Director of Mohs Surgery and Procedural Dermatology University of Texas Health Science Center San Antonio Disclosures None Possible Causes of Recurrence DNA:
Patient Information Understanding Varicose Veins
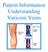 Patient Information Understanding Varicose Veins The Circulatory System Arteries carry oxygenated blood to your legs and the veins carry de-oxygenated blood away from your legs. The blood returns to the
Patient Information Understanding Varicose Veins The Circulatory System Arteries carry oxygenated blood to your legs and the veins carry de-oxygenated blood away from your legs. The blood returns to the
Medical Coverage Policy Treatment for Varicose Veins-PREAUTH
 Medical Coverage Policy Treatment for Varicose Veins-PREAUTH Device/Equipment Drug Medical Surgery Test Other Effective Date: 9/1/2001 Policy Last Updated: 12/20/2011 Prospective review is recommended/required.
Medical Coverage Policy Treatment for Varicose Veins-PREAUTH Device/Equipment Drug Medical Surgery Test Other Effective Date: 9/1/2001 Policy Last Updated: 12/20/2011 Prospective review is recommended/required.
Medical Coverage Policy Varicose Vein Treatment sad
 Medical Coverage Policy Varicose Vein Treatment sad EFFECTIVE DATE: 10 28 2001 POLICY LAST UPDATED: 05 07 2013 OVERVIEW Varicose veins are large superficial veins that have become swollen. These veins
Medical Coverage Policy Varicose Vein Treatment sad EFFECTIVE DATE: 10 28 2001 POLICY LAST UPDATED: 05 07 2013 OVERVIEW Varicose veins are large superficial veins that have become swollen. These veins
Varicose Vein Therapy: An Introduction to Surgical and Endovascular Treatment
 Varicose Vein Therapy: An Introduction to Surgical and Endovascular Treatment Lower Extremity Venous Anatomy Deep, Superficial, Perforating, Reticular veins Located in two separate compartments (deep and
Varicose Vein Therapy: An Introduction to Surgical and Endovascular Treatment Lower Extremity Venous Anatomy Deep, Superficial, Perforating, Reticular veins Located in two separate compartments (deep and
Australian Safety and Efficacy Register of New Interventional Procedures Surgical. Rapid review ASERNIP-S REPORT NO. 66
 ASERNIP S Australian Safety and Efficacy Register of New Interventional Procedures Surgical Rapid review Treatments for varicose veins ASERNIP-S REPORT NO. 66 Australian Safety & Efficacy Register of New
ASERNIP S Australian Safety and Efficacy Register of New Interventional Procedures Surgical Rapid review Treatments for varicose veins ASERNIP-S REPORT NO. 66 Australian Safety & Efficacy Register of New
SEDICO Newsletter Issue 8. Varicose veins
 SEDICO Newsletter Issue 8 Definition: Varicose veins Varicose veins are veins that have become enlarged and twisted. The term commonly refers to the veins on the leg, although varicose veins occur elsewhere.
SEDICO Newsletter Issue 8 Definition: Varicose veins Varicose veins are veins that have become enlarged and twisted. The term commonly refers to the veins on the leg, although varicose veins occur elsewhere.
VARICOSE VEINS. Information Leaflet. Your Health. Our Priority. VTE Ambulatory Clinic Stepping Hill Hospital
 VARICOSE VEINS Information Leaflet Your Health. Our Priority. Page 2 of 7 Varicose Veins There are no accurate figures for the number of people with varicose veins. Some studies suggest that 3 in 100 people
VARICOSE VEINS Information Leaflet Your Health. Our Priority. Page 2 of 7 Varicose Veins There are no accurate figures for the number of people with varicose veins. Some studies suggest that 3 in 100 people
Venous Reflux Disease and Current Treatments VN20-87-A 01/06
 Venous Reflux Disease and Current Treatments Leg Vein Anatomy Your legs are made up of a network of veins and vessels that carry blood back to the heart The venous system is comprised of: Deep veins Veins
Venous Reflux Disease and Current Treatments Leg Vein Anatomy Your legs are made up of a network of veins and vessels that carry blood back to the heart The venous system is comprised of: Deep veins Veins
Varicose Veins: Causes, Symptoms and Management. Andrew C. Stanley MD Section of Vascular Surgery
 Varicose Veins: Causes, Symptoms and Management Andrew C. Stanley MD Section of Vascular Surgery Circulation Heart (Pump)-MI, Sudden Death Arteries (Stroke, Aneurysms, Walking dysfunction, Limb loss) Veins
Varicose Veins: Causes, Symptoms and Management Andrew C. Stanley MD Section of Vascular Surgery Circulation Heart (Pump)-MI, Sudden Death Arteries (Stroke, Aneurysms, Walking dysfunction, Limb loss) Veins
Endovenous Laser Therapy
 Information for patients Endovenous Laser Therapy Northern General Hospital You have been given this leaflet because you will be having Endovenous Laser Therapy (EVLT). This leaflet explains more about
Information for patients Endovenous Laser Therapy Northern General Hospital You have been given this leaflet because you will be having Endovenous Laser Therapy (EVLT). This leaflet explains more about
The Treatment of Varicose Veins and Spider Veins
 The Treatment of Varicose Veins and Spider Veins St. Joseph Vein Center Surgical Specialists of St. Joseph, P.C. Glen H. Hastings, M.D., F.A.C.S. 820 Lester Ave., Suite 105 St. Joseph, MI 49085 (269) 983-3368
The Treatment of Varicose Veins and Spider Veins St. Joseph Vein Center Surgical Specialists of St. Joseph, P.C. Glen H. Hastings, M.D., F.A.C.S. 820 Lester Ave., Suite 105 St. Joseph, MI 49085 (269) 983-3368
Treatment by LASER. Understanding Varicose veins and. Dr. Rajesh Mundhada (MD. DNB) Dr. Atul Rewatkar (MD)
 Understanding Varicose veins and Treatment by LASER Dr. Rajesh Mundhada (MD. DNB) Dr. Atul Rewatkar (MD) Orange city Hospital & Research Institute 19, Pandey Lay out, Veer Sawarkar Square, Nagpur 440015
Understanding Varicose veins and Treatment by LASER Dr. Rajesh Mundhada (MD. DNB) Dr. Atul Rewatkar (MD) Orange city Hospital & Research Institute 19, Pandey Lay out, Veer Sawarkar Square, Nagpur 440015
LOWER EXTREMITY VENOUS DUPLEX ULTRASOUND:
 LOWER EXTREMITY VENOUS DUPLEX ULTRASOUND: Chronic Venous Insufficiency Phillip J Bendick, PhD William Beaumont Hospital Royal Oak, Michigan Prevalence: Carotid ASO ~ 3M Peripheral Arterial Dz ~ 5M + CAD
LOWER EXTREMITY VENOUS DUPLEX ULTRASOUND: Chronic Venous Insufficiency Phillip J Bendick, PhD William Beaumont Hospital Royal Oak, Michigan Prevalence: Carotid ASO ~ 3M Peripheral Arterial Dz ~ 5M + CAD
Modern Varicose Vein Treatments: What Every Patient Should Know
 The Skin and Vein Center Oneonta Laser Derm & Day Spa Natural Good Looks and Leg Veins Our Specialty Dr Eric Dohner, MD 41-45 Dietz St Oneonta, NY 13820 607/431-2525 www.oneontalaserderm.com Modern Varicose
The Skin and Vein Center Oneonta Laser Derm & Day Spa Natural Good Looks and Leg Veins Our Specialty Dr Eric Dohner, MD 41-45 Dietz St Oneonta, NY 13820 607/431-2525 www.oneontalaserderm.com Modern Varicose
Medical Policy Manual. Date of Origin: October 11, 1999. Topic: Varicose Vein Treatment. Last Reviewed Date: May 2015.
 Medical Policy Manual Topic: Varicose Vein Treatment Section: Surgery Policy No: 104 Date of Origin: October 11, 1999 Last Reviewed Date: May 2015 Effective Date: September 1, 2015 IMPORTANT REMINDER Medical
Medical Policy Manual Topic: Varicose Vein Treatment Section: Surgery Policy No: 104 Date of Origin: October 11, 1999 Last Reviewed Date: May 2015 Effective Date: September 1, 2015 IMPORTANT REMINDER Medical
X-Plain Varicose Veins Reference Summary
 X-Plain Varicose Veins Reference Summary Introduction Varicose veins are very common, in both women and men. Varicose veins can be painful and unattractive. Vein doctors use non-invasive ultrasound imaging
X-Plain Varicose Veins Reference Summary Introduction Varicose veins are very common, in both women and men. Varicose veins can be painful and unattractive. Vein doctors use non-invasive ultrasound imaging
Varicose veins and spider veins
 Varicose veins and spider veins Summary Varicose veins are knobbly, twisted and darkish-blue in appearance, and are most commonly found on people s legs. Varicose veins are caused by faulty valves within
Varicose veins and spider veins Summary Varicose veins are knobbly, twisted and darkish-blue in appearance, and are most commonly found on people s legs. Varicose veins are caused by faulty valves within
V03 Varicose Veins Surgery
 V03 Varicose Veins Surgery What are varicose veins? Varicose veins are enlarged and twisted veins in the leg. They are common and affect up to 3 in 10 people. More women than men ask for treatment, with
V03 Varicose Veins Surgery What are varicose veins? Varicose veins are enlarged and twisted veins in the leg. They are common and affect up to 3 in 10 people. More women than men ask for treatment, with
Australian Safety and Efficacy Register of New Interventional Procedures Surgical. Systematic review ASERNIP-S REPORT NO. 69
 ASERNIP S Australian Safety and Efficacy Register of New Interventional Procedures Surgical Systematic review Treatments for varicose veins ASERNIP-S REPORT NO. 69 Australian Safety & Efficacy Register
ASERNIP S Australian Safety and Efficacy Register of New Interventional Procedures Surgical Systematic review Treatments for varicose veins ASERNIP-S REPORT NO. 69 Australian Safety & Efficacy Register
Beaumont Hospital. Varicose Veins. and their TREATMENT. Professor Austin Leahy, MCh, FRCS, FRCSI WWW.VEINCLINICSOFIRELAND.COM
 Beaumont Hospital Varicose Veins and their TREATMENT Professor Austin Leahy, MCh, FRCS, FRCSI WWW.VEINCLINICSOFIRELAND.COM Department of Surgery Beaumont Hospital and Royal College of Surgeons in Ireland
Beaumont Hospital Varicose Veins and their TREATMENT Professor Austin Leahy, MCh, FRCS, FRCSI WWW.VEINCLINICSOFIRELAND.COM Department of Surgery Beaumont Hospital and Royal College of Surgeons in Ireland
Understanding Varicose Veins
 Understanding Varicose Veins Professor Bruce Campbell Published by Family Doctor Publications Limited in association with the British Medical Association IMPORTANT NOTICE This book is intended not as a
Understanding Varicose Veins Professor Bruce Campbell Published by Family Doctor Publications Limited in association with the British Medical Association IMPORTANT NOTICE This book is intended not as a
LASER TREATMENT FOR VARICOSE VEINS
 LASER TREATMENT FOR VARICOSE VEINS How can varicose veins be treated by laser? Laser treatment of varicose veins, Endovascular Laser Ablation (known as EVLA), is a minimally invasive procedure for treating
LASER TREATMENT FOR VARICOSE VEINS How can varicose veins be treated by laser? Laser treatment of varicose veins, Endovascular Laser Ablation (known as EVLA), is a minimally invasive procedure for treating
CHINOOK VASCULAR - ENDOVENOUS LASER ABLATION PROCEDURE
 CHINOOK VASCULAR - ENDOVENOUS LASER ABLATION PROCEDURE Endovenous Laser Ablation is a minimally invasive option for treating great saphenous vein incompetence (leaky valves). The first stage of your surgery
CHINOOK VASCULAR - ENDOVENOUS LASER ABLATION PROCEDURE Endovenous Laser Ablation is a minimally invasive option for treating great saphenous vein incompetence (leaky valves). The first stage of your surgery
Institute of Health Economics ENDOVENOUS ABLATION INTERVENTIONS FOR SYMPTOMATIC VARICOSE VEINS OF THE LEGS
 Institute of Health Economics ENDOVENOUS ABLATION INTERVENTIONS FOR SYMPTOMATIC VARICOSE VEINS OF THE LEGS September 2014 INSTITUTE OF HEALTH ECONOMICS The Institute of Health Economics (IHE) is an independent,
Institute of Health Economics ENDOVENOUS ABLATION INTERVENTIONS FOR SYMPTOMATIC VARICOSE VEINS OF THE LEGS September 2014 INSTITUTE OF HEALTH ECONOMICS The Institute of Health Economics (IHE) is an independent,
Greater Manchester EUR Policy Statement. Title/Topic: Varicose Veins Reference: GM003 Date: January 2015
 Greater Manchester EUR Policy Statement Title/Topic: Varicose Veins Reference: GM003 Date: January 2015 VERSION CONTROL Version Date Details Page number 0.1 23/10/2013 Initial Draft for consideration by
Greater Manchester EUR Policy Statement Title/Topic: Varicose Veins Reference: GM003 Date: January 2015 VERSION CONTROL Version Date Details Page number 0.1 23/10/2013 Initial Draft for consideration by
Varicose Vein Treatment (Endovenous Ablation of Varicose Veins)
 Scan for mobile link. Varicose Vein Treatment (Endovenous Ablation of Varicose Veins) Varicose vein treatment, also known as endovenous ablation, uses radiofrequency or laser energy to cauterize and close
Scan for mobile link. Varicose Vein Treatment (Endovenous Ablation of Varicose Veins) Varicose vein treatment, also known as endovenous ablation, uses radiofrequency or laser energy to cauterize and close
Local Coverage Determination (LCD): Varicose Veins of the Lower Extremities (L31796)
 Local Coverage Determination (LCD): Varicose Veins of the Lower Extremities (L31796) Contractor Information Contractor Name Palmetto GBA LCD Information Document Information LCD ID L31796 LCD Title Varicose
Local Coverage Determination (LCD): Varicose Veins of the Lower Extremities (L31796) Contractor Information Contractor Name Palmetto GBA LCD Information Document Information LCD ID L31796 LCD Title Varicose
Varicose veins and venous thrombosis: The latest treatment options
 Varicose veins and venous thrombosis: The latest treatment options DR ROBERT McBANE: I'm Rob McBane from the Vascular Center at Mayo Clinic in Rochester. Today we have the pleasure of talking with my esteemed
Varicose veins and venous thrombosis: The latest treatment options DR ROBERT McBANE: I'm Rob McBane from the Vascular Center at Mayo Clinic in Rochester. Today we have the pleasure of talking with my esteemed
Confirmed Deep Vein Thrombosis (DVT)
 Confirmed Deep Vein Thrombosis (DVT) Information for patients What is deep vein thrombosis? Blood clotting provides us with essential protection against severe loss of blood from an injury to a vein or
Confirmed Deep Vein Thrombosis (DVT) Information for patients What is deep vein thrombosis? Blood clotting provides us with essential protection against severe loss of blood from an injury to a vein or
Suffering from varicose veins? Patient Information. ELVeS Radial Minimally invasive laser therapy of venous insufficiency
 Suffering from varicose veins? Patient Information ELVeS Radial Minimally invasive laser therapy of venous insufficiency Do you suffer from heavy legs or visible veins? This makes diseases of the veins
Suffering from varicose veins? Patient Information ELVeS Radial Minimally invasive laser therapy of venous insufficiency Do you suffer from heavy legs or visible veins? This makes diseases of the veins
MEDICAL POLICY SUBJECT: VARICOSITIES, TREATMENT ALTERNATIVES TO VEIN STRIPPING AND LIGATION
 MEDICAL POLICY SUBJECT: VARICOSITIES, TREATMENT PAGE: 1 OF: 10 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including
MEDICAL POLICY SUBJECT: VARICOSITIES, TREATMENT PAGE: 1 OF: 10 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including
Treatment of Varicose Veins/Venous Insufficiency
 MEDICAL POLICY POLICY RELATED POLICIES POLICY GUIDELINES DESCRIPTION SCOPE BENEFIT APPLICATION RATIONALE REFERENCES CODING APPENDIX HISTORY Treatment of Varicose Veins/Venous Insufficiency Number 7.01.519
MEDICAL POLICY POLICY RELATED POLICIES POLICY GUIDELINES DESCRIPTION SCOPE BENEFIT APPLICATION RATIONALE REFERENCES CODING APPENDIX HISTORY Treatment of Varicose Veins/Venous Insufficiency Number 7.01.519
POLICY PRODUCT VARIATIONS DESCRIPTION/BACKGROUND RATIONALE DEFINITIONS BENEFIT VARIATIONS DISCLAIMER CODING INFORMATION REFERENCES POLICY HISTORY
 Original Issue Date (Created): 7/12/2003 Most Recent Review Date (Revised): 3/29/2016 Effective Date: 8/1/2016 I. POLICY Great or Small Saphenous Veins Treatment of the greater small saphenous veins by
Original Issue Date (Created): 7/12/2003 Most Recent Review Date (Revised): 3/29/2016 Effective Date: 8/1/2016 I. POLICY Great or Small Saphenous Veins Treatment of the greater small saphenous veins by
Venous leg ulcers. Venous Ulcers. Treat the source, not just the symptom
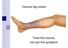 Venous leg ulcers Venous Ulcers Treat the source, not just the symptom Topics What causes venous ulcers (VU) Treating the source, not just the symptom Diagnosis with venous duplex ultrasound scan Treatment
Venous leg ulcers Venous Ulcers Treat the source, not just the symptom Topics What causes venous ulcers (VU) Treating the source, not just the symptom Diagnosis with venous duplex ultrasound scan Treatment
The anthropometric characteristics in the two groups were not significantly different.
 Studio prospettico, randomizzato monocentrico per il trattamento sintomatico dell insufficienza venosa cronica degli arti inferiori in pazienti trattati con calze preventive con filato SpikEnergy rispetto
Studio prospettico, randomizzato monocentrico per il trattamento sintomatico dell insufficienza venosa cronica degli arti inferiori in pazienti trattati con calze preventive con filato SpikEnergy rispetto
DEBATE ON Management of primary Varicose Veins SURGERY. Adel Husseiny, Kamhawy Vascular Surgery Unit Tanta University
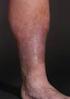 DEBATE ON Management of primary Varicose Veins SURGERY Adel Husseiny, Kamhawy Vascular Surgery Unit Tanta University Efficacy of treatment alternatives Durability of results Cosmetically acceptable results
DEBATE ON Management of primary Varicose Veins SURGERY Adel Husseiny, Kamhawy Vascular Surgery Unit Tanta University Efficacy of treatment alternatives Durability of results Cosmetically acceptable results
Recurrent Varicose Veins
 Information for patients Recurrent Varicose Veins Sheffield Vascular Institute Northern General Hospital You have been diagnosed as having Varicose Veins that have recurred (come back). This leaflet explains
Information for patients Recurrent Varicose Veins Sheffield Vascular Institute Northern General Hospital You have been diagnosed as having Varicose Veins that have recurred (come back). This leaflet explains
SURGICAL AND ABLATIVE PROCEDURES FOR VENOUS INSUFFICIENCY AND VARICOSE VEINS
 SURGICAL AND ABLATIVE PROCEDURES FOR VENOUS INSUFFICIENCY AND VARICOSE VEINS Protocol: SUR037 Effective Date: 5/1/2015 Table of Contents COMMERCIAL INDICATIONS FOR COVERAGE DEFINITIONS MEDICARE AND MEDICAID
SURGICAL AND ABLATIVE PROCEDURES FOR VENOUS INSUFFICIENCY AND VARICOSE VEINS Protocol: SUR037 Effective Date: 5/1/2015 Table of Contents COMMERCIAL INDICATIONS FOR COVERAGE DEFINITIONS MEDICARE AND MEDICAID
RANDOMIZED CONTROL TRIALS (RCT s) on VARICOSE VEIN ENDOVENOUS TREATMENT
 RANDOMIZED CONTROL TRIALS (RCT s) on VARICOSE VEIN ENDOVENOUS TREATMENT M. PERRIN Lyon - France CONTROVERSIES AND UPDATES IN VASCULAR SURGERY JANUARY 19-21, 2012 Paris B. Eklof Helsingborg, Sweden Thirty
RANDOMIZED CONTROL TRIALS (RCT s) on VARICOSE VEIN ENDOVENOUS TREATMENT M. PERRIN Lyon - France CONTROVERSIES AND UPDATES IN VASCULAR SURGERY JANUARY 19-21, 2012 Paris B. Eklof Helsingborg, Sweden Thirty
THE RELATIONSHIP BETWEEN PATIENT STATUS AND RECURRENCY RATE FOLLOWING LASER SURGERY OF VARICOSE VEINS
 THE RELATIONSHIP BETWEEN PATIENT STATUS AND RECURRENCY RATE FOLLOWING LASER SURGERY OF VARICOSE VEINS Dr Imre Bihari PhD Budapest, Hungary What can influence laser surgery results? Instrument and method
THE RELATIONSHIP BETWEEN PATIENT STATUS AND RECURRENCY RATE FOLLOWING LASER SURGERY OF VARICOSE VEINS Dr Imre Bihari PhD Budapest, Hungary What can influence laser surgery results? Instrument and method
Jose Almeida MD, FACS, RVT Jeff Raines PhD, RVT. Oliver Göckeritz MD Christian Wentzel MD. Tim Richards MD Daniel F. Hoheim, MD.
 Final Results 1 Jose Almeida MD, FACS, RVT Jeff Raines PhD, RVT Miami Vein Center Miami, Florida Oliver Göckeritz MD Christian Wentzel MD Venenzentrum Elserpark Leipzig, Germany John Kaufman MD Dotter
Final Results 1 Jose Almeida MD, FACS, RVT Jeff Raines PhD, RVT Miami Vein Center Miami, Florida Oliver Göckeritz MD Christian Wentzel MD Venenzentrum Elserpark Leipzig, Germany John Kaufman MD Dotter
Varicose Veins Operation. Patient information Leaflet
 Varicose Veins Operation Patient information Leaflet 22 nd August 2014 WHAT IS VARICOSE VEIN SURGERY (HIGH LIGATION AND MULTIPLE AVULSIONS) The operation varies from case to case, depending on where the
Varicose Veins Operation Patient information Leaflet 22 nd August 2014 WHAT IS VARICOSE VEIN SURGERY (HIGH LIGATION AND MULTIPLE AVULSIONS) The operation varies from case to case, depending on where the
Corporate Medical Policy Treatment of Varicose Veins/Venous Insufficiency. Medical Policy
 Corporate Medical Policy Treatment of Varicose Veins/Venous Insufficiency File name: Treatment of Varicose Veins/ Venous Insufficiency File code: UM.SURG.03 Origination: 9/01/2010 Last Review: 07/15/2010,
Corporate Medical Policy Treatment of Varicose Veins/Venous Insufficiency File name: Treatment of Varicose Veins/ Venous Insufficiency File code: UM.SURG.03 Origination: 9/01/2010 Last Review: 07/15/2010,
Varicose Veins & Pregnancy
 A Guide to Varicose Veins & Pregnancy What are varicose veins? Varicose veins are bulging and unsightly blue veins that occur on the legs. They can occur at any age in both men and women, but are very
A Guide to Varicose Veins & Pregnancy What are varicose veins? Varicose veins are bulging and unsightly blue veins that occur on the legs. They can occur at any age in both men and women, but are very
THE VEIN CENTER. State-of-the-Art Treatment for Varicose Veins and Spider Veins
 THE VEIN CENTER State-of-the-Art Treatment for Varicose Veins and Spider Veins Vein Disorders Nearly 50 percent of the adult population suffers from undesirable, sometimes painful vein disease. The most
THE VEIN CENTER State-of-the-Art Treatment for Varicose Veins and Spider Veins Vein Disorders Nearly 50 percent of the adult population suffers from undesirable, sometimes painful vein disease. The most
Evidence review. Endovascular treatment of varicose veins CEP09017
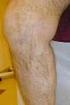 Evidence review Endovascular treatment of varicose veins CEP09017 August 2009 Contents 2 Summary... 3 Introduction... 4 Methods... 11 Evidence review... 19 Conclusions... 27 Acknowledgements... 28 Glossary...
Evidence review Endovascular treatment of varicose veins CEP09017 August 2009 Contents 2 Summary... 3 Introduction... 4 Methods... 11 Evidence review... 19 Conclusions... 27 Acknowledgements... 28 Glossary...
National Medical Policy
 National Medical Policy Subject: Policy Number: Varicose Veins Surgical Interventions NMP148 Effective Date*: September 2003 Updated: July 2016 Please contact the Medical Policy Department for an archive
National Medical Policy Subject: Policy Number: Varicose Veins Surgical Interventions NMP148 Effective Date*: September 2003 Updated: July 2016 Please contact the Medical Policy Department for an archive
Dr Marc Vuylsteke Sint-Andriesziekenhuis Tielt Belgium
 Endovenous Laser Treatment of non-gsv truncs Dr Marc Vuylsteke Sint-Andriesziekenhuis Tielt Belgium Superficial venous insufficiency Very common Evolution to chronic venous insufficiency Varicose Disease
Endovenous Laser Treatment of non-gsv truncs Dr Marc Vuylsteke Sint-Andriesziekenhuis Tielt Belgium Superficial venous insufficiency Very common Evolution to chronic venous insufficiency Varicose Disease
Venous and Lymphatic Disorders
 Venous and Lymphatic Disorders. ก. Venous and Lymphatic Disorders Varicose Veins Deep Vein Thrombosis (DVT) Lymphedema What Is Varicose Veins? Latin: Varicose = Varix = twisted Abnormal venous dilatation
Venous and Lymphatic Disorders. ก. Venous and Lymphatic Disorders Varicose Veins Deep Vein Thrombosis (DVT) Lymphedema What Is Varicose Veins? Latin: Varicose = Varix = twisted Abnormal venous dilatation
LONG TERM FOLLOW-UP AFTER ENDOVENOUS LASER ABLATION
 LONG TERM FOLLOW-UP AFTER ENDOVENOUS LASER ABLATION Giorgio Spreafico Centro Multidisciplinare Day Surgery Azienda Ospedaliera Università Padova - Italy Faculty Disclosure I have no financial relationships
LONG TERM FOLLOW-UP AFTER ENDOVENOUS LASER ABLATION Giorgio Spreafico Centro Multidisciplinare Day Surgery Azienda Ospedaliera Università Padova - Italy Faculty Disclosure I have no financial relationships
Veins. Zapping Varicose. Venous diseases affect many people and represent a major cost to the health-care system.
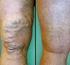 Zapping Varicose Veins Improving the Quality of Life for Sufferers By Jeannette Soriano, MD Venous diseases affect many people and represent a major cost to the health-care system. Hippocrates used compression
Zapping Varicose Veins Improving the Quality of Life for Sufferers By Jeannette Soriano, MD Venous diseases affect many people and represent a major cost to the health-care system. Hippocrates used compression
Provided by the American Venous Forum: veinforum.org
 CHAPTER 17 SURGICAL THERAPY FOR DEEP VALVE INCOMPETENCE Original author: Seshadri Raju Abstracted by Gary W. Lemmon Introduction Deep vein valvular incompetence happens when the valves in the veins (tubes
CHAPTER 17 SURGICAL THERAPY FOR DEEP VALVE INCOMPETENCE Original author: Seshadri Raju Abstracted by Gary W. Lemmon Introduction Deep vein valvular incompetence happens when the valves in the veins (tubes
CMS Limitations Guide - Radiology Services
 CMS Limitations Guide - Radiology Services Starting October 1, 2015, CMS will update their existing medical necessity limitations on tests and procedures to correspond to ICD-10 codes. This limitations
CMS Limitations Guide - Radiology Services Starting October 1, 2015, CMS will update their existing medical necessity limitations on tests and procedures to correspond to ICD-10 codes. This limitations
SURGICAL AND ABLATIVE PROCEDURES FOR VENOUS INSUFFICIENCY AND VARICOSE VEINS
 CLINICAL POLICY SURGICAL AND ABLATIVE PROCEDURES FOR VENOUS INSUFFICIENCY AND VARICOSE VEINS Policy Number: OUTPATIENT 013.24 T2 Effective Date: July 1, 2014 Table of Contents CONDITIONS OF COVERAGE...
CLINICAL POLICY SURGICAL AND ABLATIVE PROCEDURES FOR VENOUS INSUFFICIENCY AND VARICOSE VEINS Policy Number: OUTPATIENT 013.24 T2 Effective Date: July 1, 2014 Table of Contents CONDITIONS OF COVERAGE...
Diagnosis and Treatment of Venous Insufficiency and Varicose Veins
 Diagnosis and Treatment of Venous Insufficiency and Varicose Veins Joseph Conley, MD Scott and White Memorial Hospital Temple, TX September 22, 2012 Objectives Anatomy and Pathophysiology of venous insufficiency
Diagnosis and Treatment of Venous Insufficiency and Varicose Veins Joseph Conley, MD Scott and White Memorial Hospital Temple, TX September 22, 2012 Objectives Anatomy and Pathophysiology of venous insufficiency
EndoVenous Laser Therapy (EVLT) Information Booklet. Dr. Dueck. Varicose Veins
 EndoVenous Laser Therapy (EVLT) Information Booklet Dr. Dueck Varicose Veins Varicose veins are abnormally large veins that protrude from under the skin in the legs. They can be associated with a brown
EndoVenous Laser Therapy (EVLT) Information Booklet Dr. Dueck Varicose Veins Varicose veins are abnormally large veins that protrude from under the skin in the legs. They can be associated with a brown
INFORMATION DOCUMENT. Minimal Invasive Therapy of Leg Veins. Types of Veins. What is the mechanism by which veins develop?
 INFORMATION DOCUMENT Minimal Invasive Therapy of Leg Veins (Injection Microsclerotherapy / Ultrasound Guided Sclerotherapy / Endovenous Laser Therapy-EVLA and Modified Ambulatory Phlebectomy). This document
INFORMATION DOCUMENT Minimal Invasive Therapy of Leg Veins (Injection Microsclerotherapy / Ultrasound Guided Sclerotherapy / Endovenous Laser Therapy-EVLA and Modified Ambulatory Phlebectomy). This document
The veins of the lower limbs can be subdivided into three subtypes: deep veins, superficial veins, and connecting veins (figure 1).
 VEIN DISEASE Vein disease may seem insignificant and harmless at first, but if left untreated, it can develop into a major problem. It occurs when veins have trouble transporting blood back to the heart
VEIN DISEASE Vein disease may seem insignificant and harmless at first, but if left untreated, it can develop into a major problem. It occurs when veins have trouble transporting blood back to the heart
SURGICAL AND ABLATIVE PROCEDURES FOR VENOUS INSUFFICIENCY AND VARICOSE VEINS
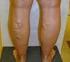 COVERAGE POLICY SURGICAL AND ABLATIVE PROCEDURES FOR VENOUS INSUFFICIENCY AND VARICOSE VEINS Policy Number: 237.E Effective Date: March 1, 2015 Table of Contents BENEFIT CONSIDERATIONS. COVERAGE RATIONALE...
COVERAGE POLICY SURGICAL AND ABLATIVE PROCEDURES FOR VENOUS INSUFFICIENCY AND VARICOSE VEINS Policy Number: 237.E Effective Date: March 1, 2015 Table of Contents BENEFIT CONSIDERATIONS. COVERAGE RATIONALE...
Varicose Veins and Spider Veins
 page 1 Varicose Veins and Spider Veins Q: What are varicose veins and spider A: Varicose (VAR-i-kos) veins are enlarged veins that can be blue, red, or fleshcolored. They often look like cords and appear
page 1 Varicose Veins and Spider Veins Q: What are varicose veins and spider A: Varicose (VAR-i-kos) veins are enlarged veins that can be blue, red, or fleshcolored. They often look like cords and appear
Varicose veins in the legs
 National Clinical Guideline Centre. Varicose veins in the legs The diagnosis and management of varicose veins Clinical guideline Methods, evidence and recommendations July 203 Final Version Commissioned
National Clinical Guideline Centre. Varicose veins in the legs The diagnosis and management of varicose veins Clinical guideline Methods, evidence and recommendations July 203 Final Version Commissioned
Successful Endovenous Ablation
 Successful Endovenous Ablation Is it the technology or the technique? PANEL Moderator: Steve Elias, MD, is Director, Center for Vein Disease, Englewood Hospital and Medical Center, New Jersey; and Assistant
Successful Endovenous Ablation Is it the technology or the technique? PANEL Moderator: Steve Elias, MD, is Director, Center for Vein Disease, Englewood Hospital and Medical Center, New Jersey; and Assistant
Varicose Vein Information Pack
 Varicose Vein Information Pack What are Varicose Veins? Varicose veins are veins under the skin of the legs which have become widened, bulging and tortuous. They are very common and do not cause medical
Varicose Vein Information Pack What are Varicose Veins? Varicose veins are veins under the skin of the legs which have become widened, bulging and tortuous. They are very common and do not cause medical
Varicose Vein Surgery Criteria Access Based Protocol Supporting people in Dorset to lead healthier lives
 NHS Dorset Clinical Commissioning Group Varicose Vein Surgery Criteria Access Based Protocol Supporting people in Dorset to lead healthier lives NHS DORSET CLINICAL COMMISSIONING GROUP VARICOSE VEIN SUREGRY
NHS Dorset Clinical Commissioning Group Varicose Vein Surgery Criteria Access Based Protocol Supporting people in Dorset to lead healthier lives NHS DORSET CLINICAL COMMISSIONING GROUP VARICOSE VEIN SUREGRY
Col league. SMMC Vascular Center Opens A PUBLICATION FOR SOUTHERN MAINE PHYSICIANS
 A PUBLICATION FOR SOUTHERN MAINE PHYSICIANS Col league 8 2012 SMMC Vascular Center Opens By Frank Lavoie, MD, Executive Vice President and Chief Operating Officer During the last year, Southern Maine Medical
A PUBLICATION FOR SOUTHERN MAINE PHYSICIANS Col league 8 2012 SMMC Vascular Center Opens By Frank Lavoie, MD, Executive Vice President and Chief Operating Officer During the last year, Southern Maine Medical
Tired, Aching Legs? Swollen Ankles? Varicose Veins? An informative guide for patients
 Tired, Aching Legs? Swollen Ankles? Varicose Veins? An informative guide for patients Are You at Risk? Leg problems are widespread throughout the world, but what most people don t know is that approximately
Tired, Aching Legs? Swollen Ankles? Varicose Veins? An informative guide for patients Are You at Risk? Leg problems are widespread throughout the world, but what most people don t know is that approximately
Prevalence and surgical outcomes of varicose veins at Regional Institute of Medical Sciences, Imphal
 ORIGINAL ARTICLE JIACM 2013; 14(3-4): 209-13 Prevalence and surgical outcomes of varicose veins at Regional Institute of Medical Sciences, Imphal Ksh Kala Singh*, A Surjyalal Sharma**, L Sunil Singh***,
ORIGINAL ARTICLE JIACM 2013; 14(3-4): 209-13 Prevalence and surgical outcomes of varicose veins at Regional Institute of Medical Sciences, Imphal Ksh Kala Singh*, A Surjyalal Sharma**, L Sunil Singh***,
SURGICAL AND ABLATIVE PROCEDURES FOR VENOUS INSUFFICIENCY AND VARICOSE VEINS
 MEDICAL POLICY SURGICAL AND ABLATIVE PROCEDURES FOR VENOUS INSUFFICIENCY AND VARICOSE VEINS Policy Number: 2015T0447O Effective Date: March 1, 2015 Table of Contents BENEFIT CONSIDERATIONS COVERAGE RATIONALE
MEDICAL POLICY SURGICAL AND ABLATIVE PROCEDURES FOR VENOUS INSUFFICIENCY AND VARICOSE VEINS Policy Number: 2015T0447O Effective Date: March 1, 2015 Table of Contents BENEFIT CONSIDERATIONS COVERAGE RATIONALE
VARICOSE VEINS AND VENOUS INSUFFICIENCY
 VARICOSE VEINS AND VENOUS INSUFFICIENCY SURGICAL AND ABLATION TREATMENTS Policy Number: 2014M0064A Effective Date: September 1, 2014 Table of Contents: Page: Cross Reference Policy: POLICY DESCRIPTION
VARICOSE VEINS AND VENOUS INSUFFICIENCY SURGICAL AND ABLATION TREATMENTS Policy Number: 2014M0064A Effective Date: September 1, 2014 Table of Contents: Page: Cross Reference Policy: POLICY DESCRIPTION
The Treatment of Saphenous Vein Occlusion by Endovenous Laser Ablation (EVLA) with a 1064 nm VSP Nd:YAG Laser
 The Treatment of Saphenous Vein Occlusion by Endovenous Laser Ablation (EVLA) with a 1064 nm VSP Nd:YAG Laser Andrej Šikovec, MD, MSc Avelana Vein Clinic, Otočec, Slovenia Abstract This paper presents
The Treatment of Saphenous Vein Occlusion by Endovenous Laser Ablation (EVLA) with a 1064 nm VSP Nd:YAG Laser Andrej Šikovec, MD, MSc Avelana Vein Clinic, Otočec, Slovenia Abstract This paper presents
Treatment of Varicose Veins
 Varicose Vein Treatment of Varicose Veins JMAJ 47(3): 146 151, 2004 Osamu SATO Associate Professor, Department of Surgery, Saitama Medical Center Abstract: Varicose vein is a condition that was first described
Varicose Vein Treatment of Varicose Veins JMAJ 47(3): 146 151, 2004 Osamu SATO Associate Professor, Department of Surgery, Saitama Medical Center Abstract: Varicose vein is a condition that was first described
SPECIALISTS IN NON-SURGICAL TREATMENT OF VARICOSE VEINS. Clinics throughout the UK
 SPECIALISTS IN NON-SURGICAL TREATMENT OF VARICOSE VEINS Clinics throughout the UK 2 BE HAPPY WITH YOUR LEGS The way our legs look and feel profoundly affects our enjoyment of life. Up to a third of the
SPECIALISTS IN NON-SURGICAL TREATMENT OF VARICOSE VEINS Clinics throughout the UK 2 BE HAPPY WITH YOUR LEGS The way our legs look and feel profoundly affects our enjoyment of life. Up to a third of the
Verification of Participation & Certificate Request 28 th Annual Congress Phoenix, Arizona November 6 9, 2014
 Complete this form in its entirety to claim the credits commensurate with your participation at the ACP s 28 th Annual Congress. You can only claim credit for one session if it runs concurrently with others.
Complete this form in its entirety to claim the credits commensurate with your participation at the ACP s 28 th Annual Congress. You can only claim credit for one session if it runs concurrently with others.
