FDA s Electronic Submissions Gateway (ESG)
|
|
|
- Johnathan Bridges
- 9 years ago
- Views:
Transcription
1 FDA s Electronic Submissions Gateway (ESG) April 19, 2016 Presenter: La Misha Fields, Program Manager FDA ESG Slide 1
2 Agenda What is ESG? ESG Process, Account Types Account Setup and Differences Submission Best Practices ESG Program Milestones and Growth Help Slide 2
3 ESG is: A conduit of receiving and delivering regulatory submissions to the appropriate center for further processing. The central transmission point for sending information electronically to the FDA and automatically routes them to the proper FDA Center or Office. Secure and used Agency-wide. ESG is NOT: A system for tracking the review and approval of a submission. It does not open or review the submissions. Slide 3
4 ESG Process and Account Types Web Interface (WebTrader) Web Interface sends submissions via Hyper Text Transfer Protocol Secure (HTTPS) through a web browser Uses Java applet Uses Tar functionality WebTrader Hosted solution: a portal to access WebTrader Applicability Statement 2 (AS2) Gateway to Gateway Secure HTTP Requires files to be tarred Attribute/Header and routing ID information will be used to route submission Requires an AS2 compliant gateway software All Submissions are signed using PKI certificates Slide 4
5 Account Setup ~1-2 Weeks 9. Prod Account Ready to Use! 1. Request an Account through ESG Help Desk ~1 Day 2. Obtain a Digital Certificate ~1-2 Weeks ~1 Day 8. Send Test Submission to Preprod Easy Steps for Setting up Your ESG Account with the FDA 3. Submit a Nonrepudiation Letter ~2-3 Days ~1 Day 7. Install JRE/JCE and Configure your Firewall (WT Only) ~1-2 Days 6. Register Account, Account Activation 5. Prepare Load Test Submissions Read User Guide and Tutorials ~1 Day 4. Prepare a Guidance Compliant Test Submission ~1 Day Slide 5
6 Account Setup Helpful Tips WebTrader accounts should not be shared. Certificates are associated with the accounts that determine the origin of the submission. However, the same account can be used to submit to different centers. Recommend having multiple WebTrader accounts if sending large number of submissions. Each account must send a guidance compliant test submission. Only one load test is required if the accounts requested are located in same physical location or on the same physical network as previous accounts established for the company. Third Party Submitters register for separate WT account for each of their clients. Letter of Authorization and Non-repudiation letter required. A Third Party Submitter can use one AS2 account to submit on behalf of multiple clients 2-4 weeks for Account Setup, so plan ahead! Slide 6
7 WebTrader Do not need to acquire any software Automatically creates tar/gzip file WebTrader vs. AS2 AS2 Need to acquire/buy AS2 compliant software Tar file is either created manually with an external tar utility or automatically within an application that has tar functionality. No special equipment is required, user can use their own desktop for WT Users will need to send one submission at a time. Submissions can t be automated Low Cost; Minimal technical expertise needed to use WT: Users need to acquire digital certificate and install JRE Suggested for low volume submitters Gzip is used after file tarring is complete. It is done either manually using an external utility or automatically within an application that has gzip functionality. Will need to acquire hardware/required for implementing AS2 system Submissions can be automated More technical expertise/help needed to setup and maintain AS2 system. Suggested for high volume submitters Slide 7
8 ESG Submission Best Practices 1. Do not use special characters in file names. a) Do not use /, \, &, #, etc. 2. File names should not contain more than one dot (.) in case of single files. Multi-files/folders should not contain any dots (.) other than in the extension. a) Example: testfile.txt or testfile.tar.gz 3. Encrypt (for AS2 only) files to enhance security. Slide 8
9 ESG Milestones: ~ 175K Submissions ~ 200 Users ESG Implemented: All Centers ~60K Submissions 2012 ~ 2 M Submissions ~ 15K Users WTHS (Citrix) 2nd Gen Planning ~ 7 M Subs (Proj) ~ 23K Users (Proj) ~ 2 nd Gen Execution 2016 ~ 1.4M Submissions ~ 3 M Submissions ~ 18K Users Collaboration with Industry Slide 9
10 ESG 2 nd Generation To ensure that the ESG is stable and can meet current demands and projected future increases in submission loads, FDA intends to address the following areas via the ESG 2 nd Generation Modernization project: Increase the capacity of the system to handle submission volume increases High availability; High Throughput for processing submissions and acknowledgements Estimated Increase in Submissions: 7 million submissions projected for million submissions per year by 2020 Production: Phase I - April 2016 Phase II Later 2016 Slide 10
11 ESG Potential Enhancements Functionality Timely and Informative Acknowledgements Process Account Set-up Process Communications Website User Guide and Other Documents 2-way Communications Slide 11
12 ESG Resources and Contact Information Website: Account Information System Requirements User Guide and Tutorials Digital Signatures and Certificates System Status, Planned Outages, Metrics and Help Desk Links to submission guidelines Account Setup: This is used for questions, system and submission related issues, and help in setting up test and production accounts FDA Program Manager: La Misha Fields Phone: Slide 12
13 Thank you! Slide 13
14 HL7 SPL SUBMISSION OPTION April 19, 2016 Varsha Thakar Data Steward Food And Drug Administration Slide 14
15 GUDID HL7 SPL Submission Option Submission of medical device information as HL7 SPL message o One device record data per xml file GUDID HL7 SPL submissions are submitted via the FDA ESG Testing required prior to submitting data to Production GUDID Ideal for labelers with large volume of submissions Slide 15
16 FDA Electronic Submissions Gateway Two Options for submission o WebTrader o Applicability Statement 2 (AS2) If you choose WebTrader o consider GUDID Web Interface instead of HL7 SPL submission option GUDID does NOT use the esubmitter tool Slide 16
17 Acknowledgements Center = CDRH Submission Type = GUDID Labelers Ack1 Ack2 ESG Ack3 CDRH Slide 17
18 Acknowledgement Types: Ack1 Ack1/Receipt/MDN Slide 18
19 Acknowledgement Types: Ack1, Ack2 Ack1/Receipt/MDN Ack2 Slide 19
20 Acknowledgement Types: Ack3 Slide 20
21 Acknowledgement Type: ACK3 Ack3 <status>failed</status> Slide 21
22 Acknowledgements (Ack) Ack1/2 Issues Ack1 Provide info Ack2 Provide messageid Ack3 Issues Provide coreid Contact appropriate help desk regarding issues prior to retransmitting Slide 22
23 Submission option: HL7 SPL Checklist for submitting in Production environment Obtain an ESG Test Account (contact FDA ESG Helpdesk) Obtain a GUDID Test Account (contact FDA UDI Helpdesk) Complete GUDID HL7 SPL testing Submit test scenarios to FDA UDI Helpdesk Slide 23
24 Request and Obtain an ESG Account Gather Data, Generate GUDID HL7 SPL XML Request and obtain a GUDID Test Account PROCESS Time Needed WebTrader 1-2 weeks AS2 2-4 weeks Complete ESG Testing FDA ESG Process Complete GUDID HL7 SPL Testing Submit Test Scenarios for FDA Review Request and Obtain a GUDID Production Account Time Needed 6-8 weeks GUDID Process Submit DI Records to Production GUDID Slide 24
25 Generate GUDID HL7 SPL XML Files Build and generate GUDID HL7 SPL XML files o One file for each DI record o Validate files against the GUDID schema Unparseable error o Most common error during GUDID testing from users o Validating files against the GUDID schema will save you time Slide 25
26 Request and obtain a GUDID Test Account Complete GUDID HL7 SPL Testing GUDID Testing Process 1. Request a GUDID test account 2. Submit XML files with test scenarios Submit Test Scenarios for FDA Review Request and Obtain a GUDID Production Account 3. Submit test results to UDI Helpdesk 4. FDA reviews and provides feedback 5. Submit to Production Submit DI Records to Production GUDID Slide 26
27 ESG and GUDID ESG serves the entire FDA ESG and GUDID have Test and Production areas Submissions sent via ESG Test Account Submissions sent via ESG Production Account Loads to GUDID Test System Loads to GUDID Production System Slide 27
28 Using Third-Party Submitters Provide Third-Party information during GUDID account request o If third-party is not associated to labeler s GUDID account, submission from third-party will be rejected Slide 28
29 Third-Parties may: Third-Parties Provide software solution/tool to labeler to generate HL7 SPL XML files; labeler sends submission via ESG o Labeler obtains ESG account Provide end-to-end solution: use labeler data to generate GUDID HL7 SPL XML files AND send submissions via ESG on behalf of labeler o Third-party has the ESG account o Labeler sends a letter of non-repudiation indicating third-party is authorized to submit on their behalf Slide 29
30 Labelers and Third-Party Submitters Labelers who intend to use the HL7 SPL submission option: o Must complete GUDID testing o Testing required even if using a third-party submitter Labelers are responsible for fulfilling GUDID submission requirements: o Ensure submissions are received and processed by FDA. o Login to GUDID and review your submissions o Report within the required timeframe o Maintain proper records Slide 30
31 Third-Party Solution Providers May test GUDID HL7 SPL submission solution independently of Labelers Request a GUDID test Account: indicate it is for HL7 SPL testing Dummy data for certain required attributes provided for testing purposes ONLY, upon request GUDID Web Interface and Production Accounts NOT provided Must complete GUDID HL7 SPL testing with each labeler Labelers must establish their own separate test GUDID account Slide 31
32 GUDID HL7 SPL Pointers Read FDA Guidance on GUDID nandguidance/guidancedocuments/ucm pdf Allow adequate time for testing ESG and GUDID GUDID testing completion criteria is the bare minimum Do thorough internal testing to ensure scenarios appropriate for your products are accounted for Slide 32
33 GUDID HL7 SPL Pointers Do not submit sample message in the HL7 SPL implementation package as a test submission it is not validated Specify Center = CDRH Submission Type = GUDID Slide 33
34 Key Points Submission folder structure must be followed o Top level folder must be uniquely named o Lower level folder must always be named spl ; only one spl folder o GUDID HL7 SPL xml submission file must be named submission.xml o Do not include any other files in the spl folder Only one submission (one DI record) in each folder structure Slide 34
35 Records can be submitted as DI Records o Unpublished = DI Record Publish Date is in the future o Published = DI Record Publish Date is today s date or earlier Review your submission via the Web Interface o Login into the GUDID Web Interface o Labeler DUNS number for that DI record should be assigned to you Slide 35
36 Editing HL7 SPL Submissions Submit the entire DI record, i.e., include changed and unchanged attributes o DI record will be over-written with the most recent file o document.id checks for the uniqueness of the GUDID submission o document.setid links all related submissions o document.versionnumber tracks versions; increment by 1 for each edit, even for failed submissions - First time submission, versionnumber = 1, Ack3 = Fail - Increment versionnumber = 2 o assign a new document.id, keep the same setid and increment the versionnumber before resubmitting Slide 36
37 Editing DI Records DI Records submitted using Web Interface o Draft, Unpublished, Published during Grace Period must edit via Web Interface o Published and past Grace Period may edit via Web Interface or HL7 SPL DI Records submitted using HL7 SPL o Any state, any time may edit via Web Interface or HL7 SPL Slide 37
38 Data Quality Before you move to production o Complete adequate internal testing o Verify test records are loaded correctly to GUDID by logging in After you move to production o Continue to monitor, review and correct records during grace period o Review information during Grace Period and make edits as needed o Use export feature to export all records in GUDID as XML files o Remember records go to AccessGUDID after Grace Period Slide 38
39 Submitting Batches in Production Start submissions in small batches and slowly ramp up Limit submissions to no more than 500 at one time If you do not receive Acknowledgements, do not automatically resend, contact us first Slide 39
40 Common questions: Please let me know the reason for the below error. Troubleshooting Errors STEP 2. Unable to parse XML STEP 2: UDI SPL submission already exists, submission is duplicate Slide 41
41 HelpDesk Do not contact multiple helpdesks or create multiple cases to report the same issue Please wait for 48 hours before reporting any ACK3 delays to the FDA UDI Helpdesk Do not send submissions prior to obtaining a GUDID account your submissions will be rejected with a failed Ack3 message Before submitting test scenarios review them and ensure that all the edits are accurately reflected in GUDID Slide 42
42 GUDID System Status Scheduled Downtimes o will be posted on o look for GUDID System Status Unscheduled Downtimes o visit for information o if no information, report issue via Help Desk Subscribe to GUDID Alerts: DeviceIdentification/GlobalUDIDatabaseGUDID/default.htm Slide 43
43 Resources - FDA Guidance on GUDID HL7 SPL Implementation Files GUDID Data Elements Reference Table CDRH Learn with GUDID Overview Slide 44
44 Questions Slide 45
FDA Electronic Submissions Gateway (ESG) Presenter: Michael B. Fauntleroy Program Manager FDA ESG
 FDA Electronic Submissions Gateway (ESG) Presenter: Michael B. Fauntleroy Program Manager FDA ESG What is the Gateway? FDA ESG - Submission Options WebTrader Gateway Cost ROI for your investment Registration
FDA Electronic Submissions Gateway (ESG) Presenter: Michael B. Fauntleroy Program Manager FDA ESG What is the Gateway? FDA ESG - Submission Options WebTrader Gateway Cost ROI for your investment Registration
GUDID Account Request: Preparation and Process
 GUDID Account Request: Preparation and Process April 19, 2016 Chris Diamant, UDI Program Analyst Food and Drug Administration Slide 1 The GUDID Data submission required by UDI System Final Rule Submission
GUDID Account Request: Preparation and Process April 19, 2016 Chris Diamant, UDI Program Analyst Food and Drug Administration Slide 1 The GUDID Data submission required by UDI System Final Rule Submission
Agenda The emdr Challenge
 The emdr Challenge Agenda The emdr Challenge Terminology High-Volume Basic Requirements Low volume Requirements Decision tree high volume vs. low volume Lessons Learned & Tips for Successful emdr Implementation
The emdr Challenge Agenda The emdr Challenge Terminology High-Volume Basic Requirements Low volume Requirements Decision tree high volume vs. low volume Lessons Learned & Tips for Successful emdr Implementation
Global Unique Device Identification Database (GUDID) Guidance for Industry and Food and Drug Administration Staff
 Global Unique Device Identification Database (GUDID) Guidance for Industry and Food and Drug Administration Staff Document issued on June 27, 2014. The draft of this document was issued on September 24,
Global Unique Device Identification Database (GUDID) Guidance for Industry and Food and Drug Administration Staff Document issued on June 27, 2014. The draft of this document was issued on September 24,
FDA Electronic Submissions Gateway (ESG) Presenter: Michael B. Fauntleroy Program Manager, FDA ESG June 13, 20013
 FDA Electronic Submissions Gateway (ESG) Presenter: Michael B. Fauntleroy Program Manager, FDA ESG June 13, 20013 Talking Points 1. What is the Gateway? 2. Receiving Units 3. Statistics 4. Submission Types
FDA Electronic Submissions Gateway (ESG) Presenter: Michael B. Fauntleroy Program Manager, FDA ESG June 13, 20013 Talking Points 1. What is the Gateway? 2. Receiving Units 3. Statistics 4. Submission Types
Food and Drug Administration
 Food and Drug Administration FDA Electronic Submissions Gateway Tutorial WebTrader Hosted Solution (WTHS): Making Electronic Submissions Document Version Date: 04/28/2014 Page 1 of 9 1. OVERVIEW Organizations
Food and Drug Administration FDA Electronic Submissions Gateway Tutorial WebTrader Hosted Solution (WTHS): Making Electronic Submissions Document Version Date: 04/28/2014 Page 1 of 9 1. OVERVIEW Organizations
FDA Electronic Submissions Gateway (ESG) Presenter: Michael B. Fauntleroy Program Manager FDA ESG June 17, 2014
 FDA Electronic Submissions Gateway (ESG) Presenter: Michael B. Fauntleroy Program Manager FDA ESG June 17, 2014 Talking Points 1. What is the Gateway? 2. Receiving Units 3. Submission Transaction Processing
FDA Electronic Submissions Gateway (ESG) Presenter: Michael B. Fauntleroy Program Manager FDA ESG June 17, 2014 Talking Points 1. What is the Gateway? 2. Receiving Units 3. Submission Transaction Processing
How to Create Your esubmitter Submissions for the Center for Tobacco Products
 How to Create Your esubmitter Submissions for the Center for Tobacco Products Last Updated December 22, 2009 Prepared by Office of Information Management (OIM) 1 Table of Contents 1. Center for Tobacco
How to Create Your esubmitter Submissions for the Center for Tobacco Products Last Updated December 22, 2009 Prepared by Office of Information Management (OIM) 1 Table of Contents 1. Center for Tobacco
Implementing a Complaint Management and Reporting System using TrackWise
 Implementing a Complaint Management and Reporting System using TrackWise Gambro Use Case Kai Kiefer Manager, IT Center of Excellence ECM & Quality Solutions Gambro Dialysatoren GmbH Yohay Yafe Director,
Implementing a Complaint Management and Reporting System using TrackWise Gambro Use Case Kai Kiefer Manager, IT Center of Excellence ECM & Quality Solutions Gambro Dialysatoren GmbH Yohay Yafe Director,
FDA Electronic Submissions Gateway (ESG) User Guide. July 1, 2009
 FDA Electronic Submissions Gateway (ESG) User Guide July 1, 2009 1. Introduction... 4 1.1 Objective... 5 2. Overview of the Registration Process... 5 2.1 Apply for a Test Account... 6 2.2 Test Account
FDA Electronic Submissions Gateway (ESG) User Guide July 1, 2009 1. Introduction... 4 1.1 Objective... 5 2. Overview of the Registration Process... 5 2.1 Apply for a Test Account... 6 2.2 Test Account
Guidance for Industry
 #108 Guidance for Industry How to Register with the CVM Electronic Submission System To Submit Information in Electronic Format Using the FDA Electronic Submissions Gateway This version of the guidance
#108 Guidance for Industry How to Register with the CVM Electronic Submission System To Submit Information in Electronic Format Using the FDA Electronic Submissions Gateway This version of the guidance
Stellar Phoenix Exchange Server Backup
 Stellar Phoenix Exchange Server Backup Version 1.0 Installation Guide Introduction This is the first release of Stellar Phoenix Exchange Server Backup tool documentation. The contents will be updated periodically
Stellar Phoenix Exchange Server Backup Version 1.0 Installation Guide Introduction This is the first release of Stellar Phoenix Exchange Server Backup tool documentation. The contents will be updated periodically
Deltek Touch Time & Expense for GovCon. User Guide for Triumph
 Deltek Touch Time & Expense for GovCon User Guide for Triumph November 25, 2014 While Deltek has attempted to verify that the information in this document is accurate and complete, some typographical or
Deltek Touch Time & Expense for GovCon User Guide for Triumph November 25, 2014 While Deltek has attempted to verify that the information in this document is accurate and complete, some typographical or
Implementation Guide Corporate egateway
 Implementation Guide Corporate egateway 2(16) Page Table of contents 1 Purpose of this guide... 3 1.1 Recommended information 4 1.2 How to get started? 4 2 Project preparation... 5 2.1 List of interested
Implementation Guide Corporate egateway 2(16) Page Table of contents 1 Purpose of this guide... 3 1.1 Recommended information 4 1.2 How to get started? 4 2 Project preparation... 5 2.1 List of interested
www.reedtech.com www.reedtech.com 1 800 772 8368 1
 www.reedtech.com www.reedtech.com 1 800 772 8368 1 / +1 215 441 6438 Agenda Reed Tech Company Profile FDA UDI / GUDID Refresher 12 Steps for Accurate GUDID Submissions Reed Tech GUDID Submission Solution
www.reedtech.com www.reedtech.com 1 800 772 8368 1 / +1 215 441 6438 Agenda Reed Tech Company Profile FDA UDI / GUDID Refresher 12 Steps for Accurate GUDID Submissions Reed Tech GUDID Submission Solution
VRIRSA Frequently Asked Questions (FAQs)
 VRIRSA Frequently Asked Questions (FAQs) Policy FAQs Q: What is VRIRSA? Q: Can a county choose to not participate in VRIRSA? Q: What years will be available in VRIRSA? Q: How soon will recent birth and
VRIRSA Frequently Asked Questions (FAQs) Policy FAQs Q: What is VRIRSA? Q: Can a county choose to not participate in VRIRSA? Q: What years will be available in VRIRSA? Q: How soon will recent birth and
Foreign Account Tax Compliance Act (FATCA) IDES Implementation Update. April 2015
 Foreign Account Tax Compliance Act (FATCA) IDES Implementation Update April 2015 IDES Implementation Developments 2 The IRS has made significant progress in developing and deploying technology capabilities
Foreign Account Tax Compliance Act (FATCA) IDES Implementation Update April 2015 IDES Implementation Developments 2 The IRS has made significant progress in developing and deploying technology capabilities
SFTP File Transmission for Section 111 Monday, October 5, 2015
 Slide 1 - of 23 Welcome to the Secure File Transfer Protocol (SFTP) File Transmission for Section 111 course. Note: This module only applies to Responsible Reporting Entities (RREs) that will be submitting
Slide 1 - of 23 Welcome to the Secure File Transfer Protocol (SFTP) File Transmission for Section 111 course. Note: This module only applies to Responsible Reporting Entities (RREs) that will be submitting
Taulia Supplier Portal User Guide
 Taulia Supplier Portal User Guide Version 2.1 Taulia Inc. Taulia GmbH 201 Mission Street, Ste. 900 Gladbacher Strasse 74 San Francisco, CA 94105 Phone +1 (415) 376 8280 40219 Düsseldorf Germany Fax +1
Taulia Supplier Portal User Guide Version 2.1 Taulia Inc. Taulia GmbH 201 Mission Street, Ste. 900 Gladbacher Strasse 74 San Francisco, CA 94105 Phone +1 (415) 376 8280 40219 Düsseldorf Germany Fax +1
Agenda. How to configure
 [email protected] Agenda Strongly Recommend: Knowledge of ArcGIS Server and Portal for ArcGIS Security in the context of ArcGIS Server/Portal for ArcGIS Access Authentication Authorization: securing web services
[email protected] Agenda Strongly Recommend: Knowledge of ArcGIS Server and Portal for ArcGIS Security in the context of ArcGIS Server/Portal for ArcGIS Access Authentication Authorization: securing web services
Child Care Provider Preparation Guide
 Child Care Provider Preparation Guide North Carolina Department of Health and Human Services Families Accessing Services through Technology (NC FAST) February 2016 Version 1.0 Page 1 of 17 TABLE OF CONTENTS
Child Care Provider Preparation Guide North Carolina Department of Health and Human Services Families Accessing Services through Technology (NC FAST) February 2016 Version 1.0 Page 1 of 17 TABLE OF CONTENTS
Preparing Electronic Drug Establishment Registration Submissions in SPL Format Medicated Animal Feed Mills. Version 1.0
 Preparing Electronic Drug Establishment Registration Submissions in SPL Format Medicated Animal Feed Mills Version 1.0 Preface This document provides basic instructions for using Structured Product Labeling
Preparing Electronic Drug Establishment Registration Submissions in SPL Format Medicated Animal Feed Mills Version 1.0 Preface This document provides basic instructions for using Structured Product Labeling
EMMA Application v. 4.9 User Manual
 EMMA Application v. 4.9 User Manual Prepared by: HP/DMDC 1600 N. Beauregard Street Alexandria, VA 22311 Abstract This guide describes how to use the EMMA system, which allows users to provision for required
EMMA Application v. 4.9 User Manual Prepared by: HP/DMDC 1600 N. Beauregard Street Alexandria, VA 22311 Abstract This guide describes how to use the EMMA system, which allows users to provision for required
Getting Started Guide
 Page 2 of 9 Introduction This guide is designed to provide you with the information you need to complete your Payment Gateway account set up and begin processing live payment transactions. As a quick overview,
Page 2 of 9 Introduction This guide is designed to provide you with the information you need to complete your Payment Gateway account set up and begin processing live payment transactions. As a quick overview,
Replacements TECHNICAL REFERENCE. DTCCSOLUTIONS Dec 2009. Copyright 2009 Depository Trust Clearing Corporation. All Rights Reserved.
 TECHNICAL REFERENCE Replacements Page 1 Table of Contents Table of Contents 1 Overview... 3 1.1 Replacements Features... 3 2 Roles and Responsibilities... 4 2.1 Sender (Receiving Carrier)... 4 2.2 Recipient
TECHNICAL REFERENCE Replacements Page 1 Table of Contents Table of Contents 1 Overview... 3 1.1 Replacements Features... 3 2 Roles and Responsibilities... 4 2.1 Sender (Receiving Carrier)... 4 2.2 Recipient
QUICK START FOR COURSES: USING BASIC COURSE SITE FEATURES
 collab.virginia.edu UVACOLLAB QUICK START FOR COURSES: USING BASIC COURSE SITE FEATURES UVaCollab Quick Start Series [email protected] Revised 5/20/2015 Quick Start for Courses Overview... 4
collab.virginia.edu UVACOLLAB QUICK START FOR COURSES: USING BASIC COURSE SITE FEATURES UVaCollab Quick Start Series [email protected] Revised 5/20/2015 Quick Start for Courses Overview... 4
HOW TO SUBMIT enbs IN THE KEY
 Page 1 of 12 HOW TO SUBMIT enbs IN THE KEY Introduction In order to submit an Electronic New Business Submission (enbs) through the Key, advisers need to first ensure their Sesame Web User ID is held in
Page 1 of 12 HOW TO SUBMIT enbs IN THE KEY Introduction In order to submit an Electronic New Business Submission (enbs) through the Key, advisers need to first ensure their Sesame Web User ID is held in
Salesforce Classic Guide for iphone
 Salesforce Classic Guide for iphone Version 37.0, Summer 16 @salesforcedocs Last updated: July 12, 2016 Copyright 2000 2016 salesforce.com, inc. All rights reserved. Salesforce is a registered trademark
Salesforce Classic Guide for iphone Version 37.0, Summer 16 @salesforcedocs Last updated: July 12, 2016 Copyright 2000 2016 salesforce.com, inc. All rights reserved. Salesforce is a registered trademark
UDI Regulatory 101. Erin (Fields) Quencer. UDI Conference October 28-29, 2014 Baltimore, MD. October 28, 2014. Regulatory Analyst, FDA
 UDI Regulatory 101 October 28, 2014 Erin (Fields) Quencer Regulatory Analyst, FDA Slide 1 Basic UDI Requirements Device label and device package must bear a UDI, 21 CFR 801.20 Devices intended to be used
UDI Regulatory 101 October 28, 2014 Erin (Fields) Quencer Regulatory Analyst, FDA Slide 1 Basic UDI Requirements Device label and device package must bear a UDI, 21 CFR 801.20 Devices intended to be used
Securing Web Services From Encryption to a Web Service Security Infrastructure
 Securing Web Services From Encryption to a Web Service Security Infrastructure Kerberos WS-Security X.509 TLS Gateway OWSM WS-Policy Peter Lorenzen WS-Addressing Agent SAML Policy Manager Technology Manager
Securing Web Services From Encryption to a Web Service Security Infrastructure Kerberos WS-Security X.509 TLS Gateway OWSM WS-Policy Peter Lorenzen WS-Addressing Agent SAML Policy Manager Technology Manager
LATITUDE Patient Management System
 LATITUDE PACEART INTEGRATION 1.01 GUIDE LATITUDE Patient Management System LATITUDE PACEART INTEGRATION SYSTEM DIAGRAM a. Patient environment b. LATITUDE environment c. Clinic environment d. Data retrieval
LATITUDE PACEART INTEGRATION 1.01 GUIDE LATITUDE Patient Management System LATITUDE PACEART INTEGRATION SYSTEM DIAGRAM a. Patient environment b. LATITUDE environment c. Clinic environment d. Data retrieval
Remote Online Support
 Remote Online Support STRONGVON Tournament Management System 1 Overview The Remote Online Support allow STRONGVON support personnel to log into your computer over the Internet to troubleshoot your system
Remote Online Support STRONGVON Tournament Management System 1 Overview The Remote Online Support allow STRONGVON support personnel to log into your computer over the Internet to troubleshoot your system
Citrix Access Gateway Enterprise Edition Citrix Access Gateway Plugin for Java User Guide. Citrix Access Gateway 8.1, Enterprise Edition
 Citrix Access Gateway Enterprise Edition Citrix Access Gateway Plugin for Java User Guide Citrix Access Gateway 8.1, Enterprise Edition Copyright and Trademark Notice Use of the product documented in this
Citrix Access Gateway Enterprise Edition Citrix Access Gateway Plugin for Java User Guide Citrix Access Gateway 8.1, Enterprise Edition Copyright and Trademark Notice Use of the product documented in this
ABB solar inverters. User s manual ABB Remote monitoring portal
 ABB solar inverters User s manual ABB Remote monitoring portal List of related manuals Title ABB Remote monitoring portal User s manual NETA-01 Ethernet adapter module User s manual Code (English) 3AUA0000098904
ABB solar inverters User s manual ABB Remote monitoring portal List of related manuals Title ABB Remote monitoring portal User s manual NETA-01 Ethernet adapter module User s manual Code (English) 3AUA0000098904
Secure Data Transfer
 Secure Data Transfer INSTRUCTIONS 3 Options to SECURELY TRANSMIT DATA 1. FTP 2. WinZip 3. Password Protection Version 2.0 Page 1 Table of Contents Acronyms & Abbreviations...1 Option 1: File Transfer Protocol
Secure Data Transfer INSTRUCTIONS 3 Options to SECURELY TRANSMIT DATA 1. FTP 2. WinZip 3. Password Protection Version 2.0 Page 1 Table of Contents Acronyms & Abbreviations...1 Option 1: File Transfer Protocol
Intelligent Process Management & Process Visualization. TAProViz 2014 workshop. Presenter: Dafna Levy
 Intelligent Process Management & Process Visualization TAProViz 2014 workshop Presenter: Dafna Levy The Topics Process Visualization in Priority ERP Planning Execution BI analysis (Built-in) Discovering
Intelligent Process Management & Process Visualization TAProViz 2014 workshop Presenter: Dafna Levy The Topics Process Visualization in Priority ERP Planning Execution BI analysis (Built-in) Discovering
VRIRSA Frequently Asked Questions (FAQs)
 Policy FAQs Q: What is VRIRSA? Q: Can a county choose to not participate in VRIRSA? Q: What initial years are available in VRIRSA? Q: How will I know which years are presently available in VRIRSA? Q: How
Policy FAQs Q: What is VRIRSA? Q: Can a county choose to not participate in VRIRSA? Q: What initial years are available in VRIRSA? Q: How will I know which years are presently available in VRIRSA? Q: How
Entrust Managed Services PKI Administrator s Quick Start Guide
 Entrust Managed Services PKI Administrator s Quick Start Guide Each Managed Services PKI organization requires an administrator also known as a local registration authority (LRA) whose duty it is to manage
Entrust Managed Services PKI Administrator s Quick Start Guide Each Managed Services PKI organization requires an administrator also known as a local registration authority (LRA) whose duty it is to manage
Configuring Infoblox DHCP
 Copyright 2008 Sophos Group. All rights reserved. No part of this publication may be reproduced, stored in retrieval system, or transmitted, in any form or by any means electronic, mechanical, photocopying,
Copyright 2008 Sophos Group. All rights reserved. No part of this publication may be reproduced, stored in retrieval system, or transmitted, in any form or by any means electronic, mechanical, photocopying,
INSTALLATION GUIDE 1.0.0 MC
 MC Extension Auto Review Reminder Emailer Irzoo Thank you for purchasing the Auto Review Reminder Emailer extension. Below are the installation instructions, please let us know if you have any questions
MC Extension Auto Review Reminder Emailer Irzoo Thank you for purchasing the Auto Review Reminder Emailer extension. Below are the installation instructions, please let us know if you have any questions
The Freelance Bookkeeper s Business Blueprint
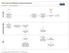 Transition to Transition BK101 Ongoing Set-Up Mgr Notify Ongoing new ready for transition. Set up time to meet Review client s folder and formulates questions prior to meeting Meet with Set-Up Mgr and
Transition to Transition BK101 Ongoing Set-Up Mgr Notify Ongoing new ready for transition. Set up time to meet Review client s folder and formulates questions prior to meeting Meet with Set-Up Mgr and
Establishment Registration SPL Technical Errors Training ebook
 Technical Errors Training ebook Interpretations & s for Technical Errors in Establishment Registration SPL Documents Submitted to FDA Version 1.0 Purpose: This SPL training ebook is to be utilized by SPL
Technical Errors Training ebook Interpretations & s for Technical Errors in Establishment Registration SPL Documents Submitted to FDA Version 1.0 Purpose: This SPL training ebook is to be utilized by SPL
Deposit Direct. Getting Started Guide
 Deposit Direct Getting Started Guide Table of Contents Before You Start... 3 Installing the Deposit Direct application for use with Microsoft Windows Vista... 4 Running Programs in Microsoft Windows Vista...
Deposit Direct Getting Started Guide Table of Contents Before You Start... 3 Installing the Deposit Direct application for use with Microsoft Windows Vista... 4 Running Programs in Microsoft Windows Vista...
Corporate Access File Transfer Service Description Version 1.0 01/05/2015
 Corporate Access File Transfer Service Description Version 1.0 01/05/2015 This document describes the characteristics and usage of the Corporate Access File Transfer service, which is for transferring
Corporate Access File Transfer Service Description Version 1.0 01/05/2015 This document describes the characteristics and usage of the Corporate Access File Transfer service, which is for transferring
Unique Device Identification Challenges for Manufacturers Focus UDID
 Unique Device Identification Challenges for Manufacturers Focus UDID GS1 Healthcare Conference Copenhagen, Oct 22, 2014 Volker Zeinar Global UDI Project Leader FDA s UDI Requirements (Medical Devices)
Unique Device Identification Challenges for Manufacturers Focus UDID GS1 Healthcare Conference Copenhagen, Oct 22, 2014 Volker Zeinar Global UDI Project Leader FDA s UDI Requirements (Medical Devices)
Before submitting claims online you must complete the following form(s): Online Provider Services Account Request Form (www.valueoptions.
 EDI RESOURCE DOCUMENT/ E-SUPPORT SERVICES PROVIDERCONNECT AND ELECTRONIC CLAIMS ValueOptions is committed to helping our providers manage administrative functions more efficiently and conveniently, and
EDI RESOURCE DOCUMENT/ E-SUPPORT SERVICES PROVIDERCONNECT AND ELECTRONIC CLAIMS ValueOptions is committed to helping our providers manage administrative functions more efficiently and conveniently, and
MUTUAL OF OMAHA SECURE EMAIL SYSTEM CLIENT/PARTNER USER GUIDE
 MUTUAL OF OMAHA SECURE EMAIL SYSTEM CLIENT/PARTNER USER GUIDE Mutual of Omaha Secure Email Client/Partner User Guide April 2015 TABLE OF CONTENTS INTRODUCTION 3 About this Guide 3 CREATING A MUTUAL OF
MUTUAL OF OMAHA SECURE EMAIL SYSTEM CLIENT/PARTNER USER GUIDE Mutual of Omaha Secure Email Client/Partner User Guide April 2015 TABLE OF CONTENTS INTRODUCTION 3 About this Guide 3 CREATING A MUTUAL OF
SCOPE OF SERVICE Hosted Cloud Storage Service: Scope of Service
 Hosted Cloud Storage Service: Scope of Service 1. Definitions 1.1 For the purposes of this Schedule: Access Account is an End User account with Data Storage requiring authentication via a username and
Hosted Cloud Storage Service: Scope of Service 1. Definitions 1.1 For the purposes of this Schedule: Access Account is an End User account with Data Storage requiring authentication via a username and
Online Payroll System Bureau Manual
 Table of Contents Introduction... 2 Setting Up A New Company... 3 View Company Status... 5 Reset A Company... 8 Bureau Maintenance... 9 Select A Company... 10 Bureau Contacts... 12 General Messages...
Table of Contents Introduction... 2 Setting Up A New Company... 3 View Company Status... 5 Reset A Company... 8 Bureau Maintenance... 9 Select A Company... 10 Bureau Contacts... 12 General Messages...
GoToAssist Corporate Integration White Paper
 Corporate Integration White Paper Corporate Integration Services offer a variety of methods to integrate Citrix Corporate, the industry-leading remote-support solution, into an existing infrastructure
Corporate Integration White Paper Corporate Integration Services offer a variety of methods to integrate Citrix Corporate, the industry-leading remote-support solution, into an existing infrastructure
GoToAssist Integration White Paper
 White paper Integration White Paper Integration Services offer a variety of methods to integrate Citrix, the industry-leading remote-support solution, into an existing infrastructure quickly and easily.
White paper Integration White Paper Integration Services offer a variety of methods to integrate Citrix, the industry-leading remote-support solution, into an existing infrastructure quickly and easily.
SECTION 3: TMHP ELECTRONIC DATA INTERCHANGE (EDI) TEXAS MEDICAID PROVIDER PROCEDURES MANUAL: VOL. 1
 SECTION 3: TMHP ELECTRONIC DATA INTERCHANGE (EDI) TEXAS MEDICAID PROVIDER PROCEDURES MANUAL: VOL. 1 AUGUST 2015 TEXAS MEDICAID PROVIDER PROCEDURES MANUAL: VOL. 1 AUGUST 2015 SECTION 3: TMHP ELECTRONIC
SECTION 3: TMHP ELECTRONIC DATA INTERCHANGE (EDI) TEXAS MEDICAID PROVIDER PROCEDURES MANUAL: VOL. 1 AUGUST 2015 TEXAS MEDICAID PROVIDER PROCEDURES MANUAL: VOL. 1 AUGUST 2015 SECTION 3: TMHP ELECTRONIC
ARGUS SUPPORT: INSTALLATION AND CONFIGURATION GUIDE FOR BEST PRACTICE
 ARGUS SUPPORT: (03) 5335 2221 or [email protected] INSTALLATION AND CONFIGURATION GUIDE FOR BEST PRACTICE VERSION 1.6.1.x ArgusConnect Pty Ltd: Phone: (03) 5335 2220 Support: (03) 5335 2221 Email:
ARGUS SUPPORT: (03) 5335 2221 or [email protected] INSTALLATION AND CONFIGURATION GUIDE FOR BEST PRACTICE VERSION 1.6.1.x ArgusConnect Pty Ltd: Phone: (03) 5335 2220 Support: (03) 5335 2221 Email:
Enrollment Guide for Electronic Services
 Enrollment Guide for Electronic Services 2014 Kareo, Inc. Rev. 3/11 1 Table of Contents 1. Introduction...1 1.1 An Overview of the Kareo Enrollment Process... 1 2. Services Offered... 2 2.1 Electronic
Enrollment Guide for Electronic Services 2014 Kareo, Inc. Rev. 3/11 1 Table of Contents 1. Introduction...1 1.1 An Overview of the Kareo Enrollment Process... 1 2. Services Offered... 2 2.1 Electronic
HP Service Manager. Software Version: 9.40 For the supported Windows and Linux operating systems. Request Management help topics for printing
 HP Service Manager Software Version: 9.40 For the supported Windows and Linux operating systems Request Management help topics for printing Document Release Date: December 2014 Software Release Date: December
HP Service Manager Software Version: 9.40 For the supported Windows and Linux operating systems Request Management help topics for printing Document Release Date: December 2014 Software Release Date: December
My HealtheVet: Secure Messaging
 My HealtheVet: Secure Messaging User Manual Version 5.0 January 2011 Department of Veterans Affairs Veterans and Consumer Health Informatics Office 1 Revision History The most recent entries in this list
My HealtheVet: Secure Messaging User Manual Version 5.0 January 2011 Department of Veterans Affairs Veterans and Consumer Health Informatics Office 1 Revision History The most recent entries in this list
How to use SURA in three simple steps:
 How to use SURA in three simple steps: Most of SURA s functionality can be accessed through these three steps. 1) Download SURA to your computer Go to the SU Downloads page to obtain the SURA utility.
How to use SURA in three simple steps: Most of SURA s functionality can be accessed through these three steps. 1) Download SURA to your computer Go to the SU Downloads page to obtain the SURA utility.
CRAY GOLD SUPPORT SUPPORT OPERATIONS HANDBOOK. GOLD-SOHB 2.2-90213 Page 1 of 22 www.cray.com
 CRAY GOLD SUPPORT SUPPORT OPERATIONS HANDBOOK GOLD-SOHB 2.2-90213 Page 1 of 22 www.cray.com Table of Contents Document Version History... 4 About Cray Customer Service... 5 About this document... 6 1.
CRAY GOLD SUPPORT SUPPORT OPERATIONS HANDBOOK GOLD-SOHB 2.2-90213 Page 1 of 22 www.cray.com Table of Contents Document Version History... 4 About Cray Customer Service... 5 About this document... 6 1.
Entrust Managed Services PKI Administrator Guide
 Entrust Managed Services PKI Entrust Managed Services PKI Administrator Guide Document issue: 3.0 Date of issue: May 2009 Copyright 2009 Entrust. All rights reserved. Entrust is a trademark or a registered
Entrust Managed Services PKI Entrust Managed Services PKI Administrator Guide Document issue: 3.0 Date of issue: May 2009 Copyright 2009 Entrust. All rights reserved. Entrust is a trademark or a registered
Automated Vendor Form for Disbursement Voucher Vendors Instruction Manual
 Automated Vendor Form for Disbursement Voucher Vendors Instruction Manual Table of Contents Eligible Vendor Types for Use with the Automated Vendor Form... 2 Search KFS for Vendors Before Using the Automated
Automated Vendor Form for Disbursement Voucher Vendors Instruction Manual Table of Contents Eligible Vendor Types for Use with the Automated Vendor Form... 2 Search KFS for Vendors Before Using the Automated
Government buyer user manual System Requirements and Administration Version 2.0
 Queensland Government etendering website Government buyer user manual System Requirements and Administration Version 2.0 July 2011 etender Help Desk phone 07 3836 0141 Table of contents 1 Introduction...
Queensland Government etendering website Government buyer user manual System Requirements and Administration Version 2.0 July 2011 etender Help Desk phone 07 3836 0141 Table of contents 1 Introduction...
TMS Phone Books Troubleshoot Guide
 TMS Phone Books Troubleshoot Guide Document ID: 118705 Contributed by Adam Wamsley and Magnus Ohm, Cisco TAC Engineers. Jan 05, 2015 Contents Introduction Prerequisites Requirements Components Used Related
TMS Phone Books Troubleshoot Guide Document ID: 118705 Contributed by Adam Wamsley and Magnus Ohm, Cisco TAC Engineers. Jan 05, 2015 Contents Introduction Prerequisites Requirements Components Used Related
Technology @ QMU. QuickStartIT: QMU OWA (webmail) service
 Technology @ QMU QuickStartIT: QMU OWA (webmail) service v1.1, September 2013 What s in this guide? Page Introduction to the QMU OWA (webmail) service 2 Logging in to the QMU OWA (webmail) service 2 OWA
Technology @ QMU QuickStartIT: QMU OWA (webmail) service v1.1, September 2013 What s in this guide? Page Introduction to the QMU OWA (webmail) service 2 Logging in to the QMU OWA (webmail) service 2 OWA
Basic ACD Planning. Agent Skill Group Planner ACD. Overflow Time
 Basic ACD Planning Agent Skill Group Planner The Agent Skill Group Planner assists in planning the distribution of workload between agent skill groups. Using the information collected from customer requirements
Basic ACD Planning Agent Skill Group Planner The Agent Skill Group Planner assists in planning the distribution of workload between agent skill groups. Using the information collected from customer requirements
JPMorgan Chase Treasury Workstation. Certification Setup Guide Version 2.0
 EMENTS JPMorgan Chase Treasury Workstation Certification Setup Guide Version 2.0 December 2010 TABLE OF CONTENTS Introduction... 1 About this Guide... 1 When to Create the Certificates... 2 Getting Help...
EMENTS JPMorgan Chase Treasury Workstation Certification Setup Guide Version 2.0 December 2010 TABLE OF CONTENTS Introduction... 1 About this Guide... 1 When to Create the Certificates... 2 Getting Help...
Re-associating.ica file extension on Vista/Windows 7 machines
 Troubleshooting This page provides answers to common problems you may have encountered when using TENGRAPH Online. Re-associating.ica file extension on Vista/Windows 7 machines There is a known issue that
Troubleshooting This page provides answers to common problems you may have encountered when using TENGRAPH Online. Re-associating.ica file extension on Vista/Windows 7 machines There is a known issue that
Cloud Voice Quick Reference Guide
 Cloud Voice Quick Reference Guide CLtel 107 N. 4th Street Clear Lake, Iowa 50428 Welcome! Welcome to Cloud Voice from CLtel! This guide is intended to get you up-and-running for the basic features associated
Cloud Voice Quick Reference Guide CLtel 107 N. 4th Street Clear Lake, Iowa 50428 Welcome! Welcome to Cloud Voice from CLtel! This guide is intended to get you up-and-running for the basic features associated
CRM Global Search: Installation & Configuration
 Installation ***Important: It is highly recommended that you first take a backup of your current CRM Application Ribbons prior to importing this solution. Please do so by navigating to Settings > Solutions
Installation ***Important: It is highly recommended that you first take a backup of your current CRM Application Ribbons prior to importing this solution. Please do so by navigating to Settings > Solutions
POINT OF SALES SYSTEM (POSS) USER MANUAL
 Page 1 of 24 POINT OF SALES SYSTEM (POSS) USER MANUAL System Name : POSI-RAD System Release Version No. : V4.0 Total pages including this covering : 23 Page 2 of 24 Table of Contents 1 INTRODUCTION...
Page 1 of 24 POINT OF SALES SYSTEM (POSS) USER MANUAL System Name : POSI-RAD System Release Version No. : V4.0 Total pages including this covering : 23 Page 2 of 24 Table of Contents 1 INTRODUCTION...
Claims Training Guide
 Claims Training Guide For exclusive use by Last Revised on 6-13-2007 10:50:00 AM Welcome... 3 Rejected Claims Dashboard... 6 Claims... 8 Editing Claims... 13 Working Claim Rejections... 16 Batches... 20
Claims Training Guide For exclusive use by Last Revised on 6-13-2007 10:50:00 AM Welcome... 3 Rejected Claims Dashboard... 6 Claims... 8 Editing Claims... 13 Working Claim Rejections... 16 Batches... 20
DRAFT US-EDI Early Development Instrument. Teacher s User Manual
 DRAFT US-EDI Early Development Instrument A Population Based Measure for Communities Teacher s User Manual EDI Implementation 2009-2010 The UCLA Center for Healthier Children, Families and Communities,
DRAFT US-EDI Early Development Instrument A Population Based Measure for Communities Teacher s User Manual EDI Implementation 2009-2010 The UCLA Center for Healthier Children, Families and Communities,
JMC Next Generation Web-based Server Install and Setup
 JMC Next Generation Web-based Server Install and Setup This document will discuss the process to install and setup a JMC Next Generation Web-based Windows Server 2008 R2. These instructions also work for
JMC Next Generation Web-based Server Install and Setup This document will discuss the process to install and setup a JMC Next Generation Web-based Windows Server 2008 R2. These instructions also work for
Application notes for SIPERA UC-Sec 4.0 Remote User Enablement Solution with Avaya Multimedia Communication System 5100 release 4.0 Issue 1.
 Avaya Solution & Interoperability Test Lab Application notes for SIPERA UC-Sec 4.0 Remote User Enablement Solution with Avaya Multimedia Communication System 5100 release 4.0 Issue 1.0 Abstract These Application
Avaya Solution & Interoperability Test Lab Application notes for SIPERA UC-Sec 4.0 Remote User Enablement Solution with Avaya Multimedia Communication System 5100 release 4.0 Issue 1.0 Abstract These Application
Emdeon Office General User Guide Table of Contents. Emdeon Office. General User Guide. February 22, 2011. Page 1
 Table of Contents Emdeon Office General User Guide February 22, 2011 Page 1 Table of Contents Table of Contents About Office... 4 System Requirements... 5 Minimum Requirements... 5 Browser Requirements...
Table of Contents Emdeon Office General User Guide February 22, 2011 Page 1 Table of Contents Table of Contents About Office... 4 System Requirements... 5 Minimum Requirements... 5 Browser Requirements...
Network/Floating License Installation Instructions
 Network/Floating License Installation Instructions Installation steps: On the Windows PC that will act as License Manager (SERVER): 1. Install HASP Run-time environment, SERVER 2. Plug in the red USB hardware
Network/Floating License Installation Instructions Installation steps: On the Windows PC that will act as License Manager (SERVER): 1. Install HASP Run-time environment, SERVER 2. Plug in the red USB hardware
Frequently Asked Questions about EDI and Item Setup
 Purchase Order Transmissions: Frequently Asked Questions about EDI and Item Setup Q. When do we transmit Purchase Orders to our Vendors? A. After the nightly batch process completes. This is typically
Purchase Order Transmissions: Frequently Asked Questions about EDI and Item Setup Q. When do we transmit Purchase Orders to our Vendors? A. After the nightly batch process completes. This is typically
SysPatrol - Server Security Monitor
 SysPatrol Server Security Monitor User Manual Version 2.2 Sep 2013 www.flexense.com www.syspatrol.com 1 Product Overview SysPatrol is a server security monitoring solution allowing one to monitor one or
SysPatrol Server Security Monitor User Manual Version 2.2 Sep 2013 www.flexense.com www.syspatrol.com 1 Product Overview SysPatrol is a server security monitoring solution allowing one to monitor one or
CT Provider Electronic Solutions. Presented by The Department of Social Services & EDS for Billing Providers
 CT Provider Electronic Solutions Presented by The Department of Social Services & EDS for Billing Providers 1 Provider Electronic Solutions New User Agenda Provider Electronic Solutions Software System
CT Provider Electronic Solutions Presented by The Department of Social Services & EDS for Billing Providers 1 Provider Electronic Solutions New User Agenda Provider Electronic Solutions Software System
EMC Documentum Quality and Manufacturing
 EMC Documentum Quality and Manufacturing Version 3.1 User Guide EMC Corporation Corporate Headquarters Hopkinton, MA 01748-9103 1-508-435-1000 www.emc.com Legal Notice Copyright 2012-2016 EMC Corporation.
EMC Documentum Quality and Manufacturing Version 3.1 User Guide EMC Corporation Corporate Headquarters Hopkinton, MA 01748-9103 1-508-435-1000 www.emc.com Legal Notice Copyright 2012-2016 EMC Corporation.
State of Michigan Data Exchange Gateway. Web-Interface Users Guide 12-07-2009
 State of Michigan Data Exchange Gateway Web-Interface Users Guide 12-07-2009 Page 1 of 21 Revision History: Revision # Date Author Change: 1 8-14-2009 Mattingly Original Release 1.1 8-31-2009 MM Pgs 4,
State of Michigan Data Exchange Gateway Web-Interface Users Guide 12-07-2009 Page 1 of 21 Revision History: Revision # Date Author Change: 1 8-14-2009 Mattingly Original Release 1.1 8-31-2009 MM Pgs 4,
Customer Portal User Guide: Transition to Delegation
 NEW GTLD PROGRAM Customer Portal User Guide: Transition to Delegation Version 0.8 Table of Contents About this User Guide... 2 Introduction to the Customer Portal... 3 Logging in with your User Name and
NEW GTLD PROGRAM Customer Portal User Guide: Transition to Delegation Version 0.8 Table of Contents About this User Guide... 2 Introduction to the Customer Portal... 3 Logging in with your User Name and
Help. F-Secure Online Backup
 Help F-Secure Online Backup F-Secure Online Backup Help... 3 Introduction... 3 What is F-Secure Online Backup?... 3 How does the program work?... 3 Using the service for the first time... 3 Activating
Help F-Secure Online Backup F-Secure Online Backup Help... 3 Introduction... 3 What is F-Secure Online Backup?... 3 How does the program work?... 3 Using the service for the first time... 3 Activating
EDI Testing System (ETS) User Guide
 EDI Testing System (ETS) Page 1 of 26 Table of Contents 1Introduction...3 2Accessing BCF EDI Testing Suite... 4 3Create User Account... 5 4Login... 6 5Change Password... 7 6Forgot Password...8 7Forgot
EDI Testing System (ETS) Page 1 of 26 Table of Contents 1Introduction...3 2Accessing BCF EDI Testing Suite... 4 3Create User Account... 5 4Login... 6 5Change Password... 7 6Forgot Password...8 7Forgot
webforms Quick Reference
 Page 1 of 9 webforms Quick Reference Version 6.0 Background & Overview webforms is the latest addition to the Progressive B2B product range. Ideal for s who are yet to become EDI (Electronic Data Interchange)
Page 1 of 9 webforms Quick Reference Version 6.0 Background & Overview webforms is the latest addition to the Progressive B2B product range. Ideal for s who are yet to become EDI (Electronic Data Interchange)
QUICK START FOR COURSES: CREATE A COURSE SITE
 UVACOLLAB QUICK START FOR COURSES: CREATE A COURSE SITE UVaCollab Quick Start Series [email protected] Revised 5/20/2015 Quick Start for Courses Overview... 3 Objectives... 3 The UVaCollab Gateway...
UVACOLLAB QUICK START FOR COURSES: CREATE A COURSE SITE UVaCollab Quick Start Series [email protected] Revised 5/20/2015 Quick Start for Courses Overview... 3 Objectives... 3 The UVaCollab Gateway...
Fannie Mae launched the Inquiry Response Tool (IRT) vendor portal in 2015. The IRT portal enhances the way the Fannie Mae Expense Reimbursement team
 Fannie Mae launched the Inquiry Response Tool (IRT) vendor portal in 2015. The IRT portal enhances the way the Fannie Mae Expense Reimbursement team manages customer service inquiries related to claims
Fannie Mae launched the Inquiry Response Tool (IRT) vendor portal in 2015. The IRT portal enhances the way the Fannie Mae Expense Reimbursement team manages customer service inquiries related to claims
Cayman Islands Automatic Exchange of Information (AEOI) Portal. User Guide v2.0
 Department for International Tax Cooperation CAYMAN ISLANDS Cayman Islands Automatic Exchange of Information (AEOI) Portal User Guide v2.0 (7 July 2016) Table of Contents Glossary of terms... 4 Introduction...
Department for International Tax Cooperation CAYMAN ISLANDS Cayman Islands Automatic Exchange of Information (AEOI) Portal User Guide v2.0 (7 July 2016) Table of Contents Glossary of terms... 4 Introduction...
CA Clarity Integration
 CA Clarity Integration Delivering Complete Cost, Resource and Change Visibility for IT Management CA has led the industry in providing integrated solutions. Enterprise IT Management (EITM) is CA s vision
CA Clarity Integration Delivering Complete Cost, Resource and Change Visibility for IT Management CA has led the industry in providing integrated solutions. Enterprise IT Management (EITM) is CA s vision
technical brief browsing to an installation of HP Web Jetadmin. Internal Access HTTP Port Access List User Profiles HTTP Port
 technical brief in HP Overview HP is a powerful webbased software utility for installing, configuring, and managing networkconnected devices. Since it can install and configure devices, it must be able
technical brief in HP Overview HP is a powerful webbased software utility for installing, configuring, and managing networkconnected devices. Since it can install and configure devices, it must be able
Vodafone PC SMS 2010. (Software version 4.7.1) User Manual
 Vodafone PC SMS 2010 (Software version 4.7.1) User Manual July 19, 2010 Table of contents 1. Introduction...4 1.1 System Requirements... 4 1.2 Reply-to-Inbox... 4 1.3 What s new?... 4 2. Installation...6
Vodafone PC SMS 2010 (Software version 4.7.1) User Manual July 19, 2010 Table of contents 1. Introduction...4 1.1 System Requirements... 4 1.2 Reply-to-Inbox... 4 1.3 What s new?... 4 2. Installation...6
Digital Voice Services User Guide
 Digital Voice Services User Guide * Feature Access Codes *72 Call Forwarding Always Activation *73 Call Forwarding Always Deactivation *90 Call Forwarding Busy Activation *91 Call Forwarding Busy Deactivation
Digital Voice Services User Guide * Feature Access Codes *72 Call Forwarding Always Activation *73 Call Forwarding Always Deactivation *90 Call Forwarding Busy Activation *91 Call Forwarding Busy Deactivation
PaperCut Payment Gateway Module - Heartland Quick Start Guide
 PaperCut Payment Gateway Module - Heartland Quick Start Guide This guide is designed to supplement the Payment Gateway Module documentation and provides a guide to installing, setting up and testing the
PaperCut Payment Gateway Module - Heartland Quick Start Guide This guide is designed to supplement the Payment Gateway Module documentation and provides a guide to installing, setting up and testing the
Volume AGKSOFT. Gilbarco Passport Back Office Software. Gilbarco Passport Guide
 Volume P AGKSOFT Gilbarco Passport Back Office Software Gilbarco Passport Guide Introduction T he Gilbarco Passport can be connected to your Windows PC using a straight CAT5/RJ45 cable connected to the
Volume P AGKSOFT Gilbarco Passport Back Office Software Gilbarco Passport Guide Introduction T he Gilbarco Passport can be connected to your Windows PC using a straight CAT5/RJ45 cable connected to the
http://www.nwea.org/support/course/enrolling-test-term
 Script: Enrolling for a Test Term: Creating Your Class Roster File and Special Programs File. This document is the script containing the narration text for the online training Enrolling for a Test Term:
Script: Enrolling for a Test Term: Creating Your Class Roster File and Special Programs File. This document is the script containing the narration text for the online training Enrolling for a Test Term:
EXPERT STRATEGIES FOR LOG COLLECTION, ROOT CAUSE ANALYSIS, AND COMPLIANCE
 EXPERT STRATEGIES FOR LOG COLLECTION, ROOT CAUSE ANALYSIS, AND COMPLIANCE A reliable, high-performance network is critical to your IT infrastructure and organization. Equally important to network performance
EXPERT STRATEGIES FOR LOG COLLECTION, ROOT CAUSE ANALYSIS, AND COMPLIANCE A reliable, high-performance network is critical to your IT infrastructure and organization. Equally important to network performance
