Contact Angle and Surface Energy Measurements on Steel
|
|
|
- Dortha Blair
- 7 years ago
- Views:
Transcription
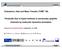
1 Contact Angle and Surface Energy Measurements on Steel July 19, 2003 Four precision steel coupons were tested on an FTA200 for contact angle and surface energy. The samples were ground smooth and appeared clean, which is what makes the following results interesting. The following image shows a typical sessile drop on the sample. The test liquid was water. Contact angle of water on steel sample The samples were tested with water, ethylene glycol and formamide. In all cases the test liquids spread after being deposited on the surface. This typically indicates one of three conditions: penetration into the surface adsorption of test liquid vapor onto surface, or the test liquid is dissolving the surface or reacting chemically with it The following graphs show the contact angles of the three test liquids on one of the samples.
2 2 Contact angle ( o symbols) and sessile drop base area ( x symbols) for water Contact angle and base area for ethylene glycol
3 3 Contact angle and base area for formamide All four samples behaved in a similar way. Notice that all three test liquids spread. And, finally, let us repeat the samples appeared clean. The samples were then cleaned in a 99% isopropyl alcohol (IPA) bath, dried, and then tested with water again. The following two graphs show the water contact angle data on sample #1, as received, and after the IPA wash. While the response is different, it still shows spreading over time. The other samples behaved similarly. From this we conclude the IPA wash affected the surface but did not clean it thoroughly. The samples were next cleaned with a methyl ethyl ketone (MEK) wash. This was more successful. The third graph in the series shows the water contact angle data after the MEK wash. The flatness of the graphs are remarkable: there is no spreading of the solvent on the sample after deposition. The contact angle is also lower, indicating a higher surface energy. We would normally expect organic contamination to have a lower surface energy and therefore a higher contact angle, but this is a rule-of-thumb and not as convincing as the change in the shape of the time curves. This set of data is an excellent example of contact angle data revealing contamination on precision parts that otherwise appear clean.
4 4 Water contact angle on sample as received, before any wash Water contact angle on sample after IPA wash
5 5 Water contact angle on sample after MEK wash The initial, before cleaning, and final, after cleaning, contact angles for the four samples are listed in the table. Sample number Initial CA ( ) Final CA ( ) The mean of the final contact angles is 75.7, the standard deviation 1.6, and the coefficient of variance is 2.1%. An attempt was made to characterize the initial (uncleaned) surfaces using Lewis acid/base analysis and Owens-Wendt analysis. We know that these analyses require thermal equilibrium and that we do not have equilibrium when the drops continue to spread. Nevertheless, may people try to apply these equations in circumstances where the underlying assumptions are not met. Sometimes workers feel that if they use either the initial contact angle (before the liquid spreads) or the final value after spreading, then the results will be better. The following table shows the acid/base and dispersive/polar components for each of the samples, using both the initial and final time values, all before any cleaning was performed. The energies computed using the final values for the contact angles (i.e., after spreading) are naturally different but not necessarily
6 6 more reliable. For Lewis acid/base analysis, negative values are obtained for the acid components when using the initial contact angles. These negative values are not seen when using the contact angles after spreading. Negative values would indicate repulsion and are considered suspect by many. A final point of contention is that the acid/base non-polar (i.e., dispersive) components should be similar to those for Owens-Wendt analysis. The Owens- Wendt analysis was performed using the water and ethylene glycol contact angle data. Sample Lewis A/B NonPolar (mn/m) Lewis A/B Acid (mn/m) Lewis A/B Base (mn/m) Owens-Wendt NonPolar (mn/m) Owens-Wendt Polar (mn/m) 1 initial final initial final initial final initial final E What should one make of this kind of situation? Understand that the surface energy equations are based on important thermal equilibrium assumptions and they should not be used when the contact angle time graphs are varying as they are with these (uncleaned) samples. However the contact angle data itself is valid and is very revealing. We learn from the spreading that there is some coating on the surface interacting with the test liquids. This is important information in itself. Secondly, after successfully cleaning the samples, we see that there is a correlation between contact angle and cleanliness. This is something that has been found to be true on many surfaces, although the exact details must be established by empirical means. File: ContactAnglesOnSteel.doc
Copyright 2010 Pearson Education, Inc. Chapter Fourteen 1
 An alcohol has an OH bonded to an alkyl group; a phenol has an OH bonded directly to an aromatic ring; and an ether has an O bonded to two organic groups. Chapter Fourteen 1 Ethyl alcohol, dimethyl ether,
An alcohol has an OH bonded to an alkyl group; a phenol has an OH bonded directly to an aromatic ring; and an ether has an O bonded to two organic groups. Chapter Fourteen 1 Ethyl alcohol, dimethyl ether,
Name Lab #3: Solubility of Organic Compounds Objectives: Introduction: soluble insoluble partially soluble miscible immiscible
 Lab #3: Solubility of rganic Compounds bjectives: - Understanding the relative solubility of organic compounds in various solvents. - Exploration of the effect of polar groups on a nonpolar hydrocarbon
Lab #3: Solubility of rganic Compounds bjectives: - Understanding the relative solubility of organic compounds in various solvents. - Exploration of the effect of polar groups on a nonpolar hydrocarbon
Experiment 446.1 SURFACE TENSION OF LIQUIDS. Experiment 1, page 1 Version of June 17, 2016
 Experiment 1, page 1 Version of June 17, 2016 Experiment 446.1 SURFACE TENSION OF LIQUIDS Theory To create a surface requires work that changes the Gibbs energy, G, of a thermodynamic system. dg = SdT
Experiment 1, page 1 Version of June 17, 2016 Experiment 446.1 SURFACE TENSION OF LIQUIDS Theory To create a surface requires work that changes the Gibbs energy, G, of a thermodynamic system. dg = SdT
Dispersing Powders in Liquid
 Dispersing Powders in Liquid Mark Bumiller mark.bumiller@horiba.com Definitions Typical powder: individual particles and clumps stuck together by weak or strong forces Agglomerates: assemblage of particles
Dispersing Powders in Liquid Mark Bumiller mark.bumiller@horiba.com Definitions Typical powder: individual particles and clumps stuck together by weak or strong forces Agglomerates: assemblage of particles
Experiment #8 properties of Alcohols and Phenols
 Introduction Experiment #8 properties of Alcohols and Phenols As has been mentioned before, over 20 million organic compounds have been identified. If each substance had to be studied as an entity completely
Introduction Experiment #8 properties of Alcohols and Phenols As has been mentioned before, over 20 million organic compounds have been identified. If each substance had to be studied as an entity completely
Chapter 5 Student Reading
 Chapter 5 Student Reading THE POLARITY OF THE WATER MOLECULE Wonderful water Water is an amazing substance. We drink it, cook and wash with it, swim and play in it, and use it for lots of other purposes.
Chapter 5 Student Reading THE POLARITY OF THE WATER MOLECULE Wonderful water Water is an amazing substance. We drink it, cook and wash with it, swim and play in it, and use it for lots of other purposes.
Chapter 13 Solution Dynamics. An Introduction to Chemistry by Mark Bishop
 Chapter 13 Solution Dynamics An Introduction to Chemistry by Mark Bishop Chapter Map Why Changes Happen Consider a system that can switch freely between two states, A and B. Probability helps us to predict
Chapter 13 Solution Dynamics An Introduction to Chemistry by Mark Bishop Chapter Map Why Changes Happen Consider a system that can switch freely between two states, A and B. Probability helps us to predict
ACRYLICS. The properties of the resulting acrylics vary depending on the nature of the alkyl groups both on the alcohol and the acrylic acid.
 AYLIS Acrylics are polyesters based on acrylic acid (propenoic acid - H 2 =H 2 H) formed from the polymerisation of an alkyl acrylate ester. They are widely used in the surface coatings industry (e.g.
AYLIS Acrylics are polyesters based on acrylic acid (propenoic acid - H 2 =H 2 H) formed from the polymerisation of an alkyl acrylate ester. They are widely used in the surface coatings industry (e.g.
Concepts in Investments Risks and Returns (Relevant to PBE Paper II Management Accounting and Finance)
 Concepts in Investments Risks and Returns (Relevant to PBE Paper II Management Accounting and Finance) Mr. Eric Y.W. Leung, CUHK Business School, The Chinese University of Hong Kong In PBE Paper II, students
Concepts in Investments Risks and Returns (Relevant to PBE Paper II Management Accounting and Finance) Mr. Eric Y.W. Leung, CUHK Business School, The Chinese University of Hong Kong In PBE Paper II, students
Scotch-Weld TM. Epoxy Adhesive 1838 B/A Green 1838 B/A Tan 1838-L B/A Translucent. Technical Data February, 2016. Product Description
 3 Scotch-Weld TM 1838 1838 1838-L Technical Data February, 2016 Description 3M TM Scotch-Weld TM s 1838 and are controlled flow products; Scotch-Weld 1838-L is flowable. These epoxy adhesives are two-part,
3 Scotch-Weld TM 1838 1838 1838-L Technical Data February, 2016 Description 3M TM Scotch-Weld TM s 1838 and are controlled flow products; Scotch-Weld 1838-L is flowable. These epoxy adhesives are two-part,
Separation by Solvent Extraction
 Experiment 3 Separation by Solvent Extraction Objectives To separate a mixture consisting of a carboxylic acid and a neutral compound by using solvent extraction techniques. Introduction Frequently, organic
Experiment 3 Separation by Solvent Extraction Objectives To separate a mixture consisting of a carboxylic acid and a neutral compound by using solvent extraction techniques. Introduction Frequently, organic
The chemical interactions of the template molecule are primarily dependent on the choice of polymer
 Study of the Surface Morphology of Methyl 4-nitrobenzoate Template Thin-film Molecularly Imprinted Polymers Gary Kaganas Dartmouth College and Center for Nanomaterials Research at Dartmouth, Hanover NH
Study of the Surface Morphology of Methyl 4-nitrobenzoate Template Thin-film Molecularly Imprinted Polymers Gary Kaganas Dartmouth College and Center for Nanomaterials Research at Dartmouth, Hanover NH
ACID-BASE TITRATIONS: DETERMINATION OF CARBONATE BY TITRATION WITH HYDROCHLORIC ACID BACKGROUND
 #3. Acid - Base Titrations 27 EXPERIMENT 3. ACID-BASE TITRATIONS: DETERMINATION OF CARBONATE BY TITRATION WITH HYDROCHLORIC ACID BACKGROUND Carbonate Equilibria In this experiment a solution of hydrochloric
#3. Acid - Base Titrations 27 EXPERIMENT 3. ACID-BASE TITRATIONS: DETERMINATION OF CARBONATE BY TITRATION WITH HYDROCHLORIC ACID BACKGROUND Carbonate Equilibria In this experiment a solution of hydrochloric
Phase Equilibrium: Fugacity and Equilibrium Calculations. Fugacity
 Phase Equilibrium: Fugacity and Equilibrium Calculations (FEC) Phase Equilibrium: Fugacity and Equilibrium Calculations Relate the fugacity and the chemical potential (or the partial molar Gibbs free energy)
Phase Equilibrium: Fugacity and Equilibrium Calculations (FEC) Phase Equilibrium: Fugacity and Equilibrium Calculations Relate the fugacity and the chemical potential (or the partial molar Gibbs free energy)
Physical Properties of a Pure Substance, Water
 Physical Properties of a Pure Substance, Water The chemical and physical properties of a substance characterize it as a unique substance, and the determination of these properties can often allow one to
Physical Properties of a Pure Substance, Water The chemical and physical properties of a substance characterize it as a unique substance, and the determination of these properties can often allow one to
Turbulence, Heat and Mass Transfer (THMT 09) Poiseuille flow of liquid methane in nanoscopic graphite channels by molecular dynamics simulation
 Turbulence, Heat and Mass Transfer (THMT 09) Poiseuille flow of liquid methane in nanoscopic graphite channels by molecular dynamics simulation Sapienza Università di Roma, September 14, 2009 M. T. HORSCH,
Turbulence, Heat and Mass Transfer (THMT 09) Poiseuille flow of liquid methane in nanoscopic graphite channels by molecular dynamics simulation Sapienza Università di Roma, September 14, 2009 M. T. HORSCH,
RTV162. Electronic Grade Silicone Adhesive Sealant
 RTV162 Product Description Electronic Grade Silicone Adhesive Sealant RTV162 adhesive sealant from GE Silicones is one-component, readyto-use electronic grade silicone sealant. It cures to a tough, resilient
RTV162 Product Description Electronic Grade Silicone Adhesive Sealant RTV162 adhesive sealant from GE Silicones is one-component, readyto-use electronic grade silicone sealant. It cures to a tough, resilient
CALCULATIONS & STATISTICS
 CALCULATIONS & STATISTICS CALCULATION OF SCORES Conversion of 1-5 scale to 0-100 scores When you look at your report, you will notice that the scores are reported on a 0-100 scale, even though respondents
CALCULATIONS & STATISTICS CALCULATION OF SCORES Conversion of 1-5 scale to 0-100 scores When you look at your report, you will notice that the scores are reported on a 0-100 scale, even though respondents
Peel Adhesion of Pressure Sensitive Tape
 Harmonized International Standard Call Letters PSTC 101 Date of Issuance 10/00 Revised 10/03 Revised 05/07 Peel Adhesion of Pressure Sensitive Tape 1. SCOPE 1.1 These methods cover the measurement of the
Harmonized International Standard Call Letters PSTC 101 Date of Issuance 10/00 Revised 10/03 Revised 05/07 Peel Adhesion of Pressure Sensitive Tape 1. SCOPE 1.1 These methods cover the measurement of the
SKEWNESS. Measure of Dispersion tells us about the variation of the data set. Skewness tells us about the direction of variation of the data set.
 SKEWNESS All about Skewness: Aim Definition Types of Skewness Measure of Skewness Example A fundamental task in many statistical analyses is to characterize the location and variability of a data set.
SKEWNESS All about Skewness: Aim Definition Types of Skewness Measure of Skewness Example A fundamental task in many statistical analyses is to characterize the location and variability of a data set.
The introduction of your report should be written on the on the topic of the role of indicators on acid base titrations.
 Experiment # 13A TITRATIONS INTRODUCTION: This experiment will be written as a formal report and has several parts: Experiment 13 A: Basic methods (accuracy and precision) (a) To standardize a base (~
Experiment # 13A TITRATIONS INTRODUCTION: This experiment will be written as a formal report and has several parts: Experiment 13 A: Basic methods (accuracy and precision) (a) To standardize a base (~
Solvents: Theory and Application
 Solvents: Theory and Application Revised: 20131107 Source: www.microchemicals.com/downloads/application_notes.html Dissolving While etching breaks the intramolecular bonds of a solid state, solving describes
Solvents: Theory and Application Revised: 20131107 Source: www.microchemicals.com/downloads/application_notes.html Dissolving While etching breaks the intramolecular bonds of a solid state, solving describes
Selection, Use, and Care of Brushes
 Wood Finishing & Refinishing Fact Sheet #2 Selection, Use, and Care of Brushes Dr. Leona K. Hawks Home Furnishings and Housing Specialist 1995 HI 22 Good results in wood refinishing depend to a large extent
Wood Finishing & Refinishing Fact Sheet #2 Selection, Use, and Care of Brushes Dr. Leona K. Hawks Home Furnishings and Housing Specialist 1995 HI 22 Good results in wood refinishing depend to a large extent
Thin Layer Chromatography.
 Thin Layer Chromatography. Thin layer chromatography, or TLC, is a method for analyzing mixtures by separating the compounds in the mixture. TLC can be used to help determine the number of components in
Thin Layer Chromatography. Thin layer chromatography, or TLC, is a method for analyzing mixtures by separating the compounds in the mixture. TLC can be used to help determine the number of components in
# 12 Condensation Polymerization: Preparation of Two Types of Polyesters
 # 12 Condensation Polymerization: Preparation of Two Types of Polyesters Submitted by: Arturo Contreras, Visiting Scholar, Center for Chemical Education, Miami University, Middletown, OH; 1996 1997. I.
# 12 Condensation Polymerization: Preparation of Two Types of Polyesters Submitted by: Arturo Contreras, Visiting Scholar, Center for Chemical Education, Miami University, Middletown, OH; 1996 1997. I.
experiment5 Understanding and applying the concept of limiting reagents. Learning how to perform a vacuum filtration.
 81 experiment5 LECTURE AND LAB SKILLS EMPHASIZED Synthesizing an organic substance. Understanding and applying the concept of limiting reagents. Determining percent yield. Learning how to perform a vacuum
81 experiment5 LECTURE AND LAB SKILLS EMPHASIZED Synthesizing an organic substance. Understanding and applying the concept of limiting reagents. Determining percent yield. Learning how to perform a vacuum
VC 104+ Rigid Grade / Rigid Grade Imagine VC 104 Rigid Grade Commercial Customised
 Page 1 of 6 instructions VC 104+ Rigid Grade / VC 104+ Rigid Grade Imagine / (for the rest of this document referred to as VC 104) can be applied to new and used trucks and trailers with painted rigid
Page 1 of 6 instructions VC 104+ Rigid Grade / VC 104+ Rigid Grade Imagine / (for the rest of this document referred to as VC 104) can be applied to new and used trucks and trailers with painted rigid
Experiment 1: Colligative Properties
 Experiment 1: Colligative Properties Determination of the Molar Mass of a Compound by Freezing Point Depression. Objective: The objective of this experiment is to determine the molar mass of an unknown
Experiment 1: Colligative Properties Determination of the Molar Mass of a Compound by Freezing Point Depression. Objective: The objective of this experiment is to determine the molar mass of an unknown
Chapter 4 Lecture Notes
 Chapter 4 Lecture Notes Chapter 4 Educational Goals 1. Given the formula of a molecule, the student will be able to draw the line-bond (Lewis) structure. 2. Understand and construct condensed structural
Chapter 4 Lecture Notes Chapter 4 Educational Goals 1. Given the formula of a molecule, the student will be able to draw the line-bond (Lewis) structure. 2. Understand and construct condensed structural
Acid Base Titrations
 Acid Base Titrations Introduction A common question chemists have to answer is how much of something is present in a sample or a product. If the product contains an acid or base, this question is usually
Acid Base Titrations Introduction A common question chemists have to answer is how much of something is present in a sample or a product. If the product contains an acid or base, this question is usually
Paper interaction with fountain solution during multi-color offset printing
 Paper interaction with fountain solution during multi-color offset printing F. Ciolacu, Elena Bobu PULP, PAPER AND PRINTING DEPARTMENT GH.ASACHI TECHNICAL UNIVERSITY OF IASI ROMANIA Offset printing Printing
Paper interaction with fountain solution during multi-color offset printing F. Ciolacu, Elena Bobu PULP, PAPER AND PRINTING DEPARTMENT GH.ASACHI TECHNICAL UNIVERSITY OF IASI ROMANIA Offset printing Printing
Neutralizing an Acid and a Base
 Balancing Act Teacher Information Objectives In this activity, students neutralize a base with an acid. Students determine the point of neutralization of an acid mixed with a base while they: Recognize
Balancing Act Teacher Information Objectives In this activity, students neutralize a base with an acid. Students determine the point of neutralization of an acid mixed with a base while they: Recognize
Acid Dissociation Constants and the Titration of a Weak Acid
 Acid Dissociation Constants and the Titration of a Weak Acid One of the most important applications of equilibria is the chemistry of acids and bases. The Brønsted-Lowry acid-base theory defines an acid
Acid Dissociation Constants and the Titration of a Weak Acid One of the most important applications of equilibria is the chemistry of acids and bases. The Brønsted-Lowry acid-base theory defines an acid
3 Destructible Vinyl Label Material 7613T / 7930T
 3 Destructible Vinyl Label Material Technical Data September, 2010 Product Description 3M Destructible Vinyl Label Materials 7613T and 7930T are consistent, smooth, topcoated films designed to exhibit
3 Destructible Vinyl Label Material Technical Data September, 2010 Product Description 3M Destructible Vinyl Label Materials 7613T and 7930T are consistent, smooth, topcoated films designed to exhibit
Activ ActivTM self-cleaning glass Activ
 Pilkington Activ TM Activ self-cleaning glass Pilkington Activ The world s first self-cleaning glass with advanced dual action for superior results A revolution in glass Self-cleaning glass has been described
Pilkington Activ TM Activ self-cleaning glass Pilkington Activ The world s first self-cleaning glass with advanced dual action for superior results A revolution in glass Self-cleaning glass has been described
TRIAL CHEMICAL CLEANING OF FOULED APH BASKETS
 TRIAL CHEMICAL CLEANING OF FOULED APH BASKETS Dr. Abhay Kumar Sahay, AGM(CC OS) Bijay Manjul, AGM( Operation) Kahalgaon Boiler has three inputs Steam generator 1. WATER 2. COAL 3. AIR Burner Air preheater
TRIAL CHEMICAL CLEANING OF FOULED APH BASKETS Dr. Abhay Kumar Sahay, AGM(CC OS) Bijay Manjul, AGM( Operation) Kahalgaon Boiler has three inputs Steam generator 1. WATER 2. COAL 3. AIR Burner Air preheater
PAPER CHROMATOGRAPHY
 PAPER CHROMATOGRAPHY INTRODUCTION Chromatography is a technique that is used to separate and to identify components of a mixture. This analytical technique has a wide range of applications in the real
PAPER CHROMATOGRAPHY INTRODUCTION Chromatography is a technique that is used to separate and to identify components of a mixture. This analytical technique has a wide range of applications in the real
The Theory of HPLC. Gradient HPLC
 The Theory of HPLC Gradient HPLC i Wherever you see this symbol, it is important to access the on-line course as there is interactive material that cannot be fully shown in this reference manual. Aims
The Theory of HPLC Gradient HPLC i Wherever you see this symbol, it is important to access the on-line course as there is interactive material that cannot be fully shown in this reference manual. Aims
Thermodynamics of Mixing
 Thermodynamics of Mixing Dependence of Gibbs energy on mixture composition is G = n A µ A + n B µ B and at constant T and p, systems tend towards a lower Gibbs energy The simplest example of mixing: What
Thermodynamics of Mixing Dependence of Gibbs energy on mixture composition is G = n A µ A + n B µ B and at constant T and p, systems tend towards a lower Gibbs energy The simplest example of mixing: What
RECITATION NOTES FOR EXPERIMENT # 5 A&B THIN LAYER CHROMATOGRAPHY
 RECITATION NOTES FOR EXPERIMENT # 5 A&B THIN LAYER CHROMATOGRAPHY Have your lab textbook available for quick reference to specific pages, indicated in red. BASIC PRINCIPLES OF CHROMATOGRAPHY Chromatography
RECITATION NOTES FOR EXPERIMENT # 5 A&B THIN LAYER CHROMATOGRAPHY Have your lab textbook available for quick reference to specific pages, indicated in red. BASIC PRINCIPLES OF CHROMATOGRAPHY Chromatography
To measure the solubility of a salt in water over a range of temperatures and to construct a graph representing the salt solubility.
 THE SOLUBILITY OF A SALT IN WATER AT VARIOUS TEMPERATURES 2007, 1995, 1991 by David A. Katz. All rights reserved. Permission for academic use provided the original copyright is included. OBJECTIVE To measure
THE SOLUBILITY OF A SALT IN WATER AT VARIOUS TEMPERATURES 2007, 1995, 1991 by David A. Katz. All rights reserved. Permission for academic use provided the original copyright is included. OBJECTIVE To measure
MEASURES OF VARIATION
 NORMAL DISTRIBTIONS MEASURES OF VARIATION In statistics, it is important to measure the spread of data. A simple way to measure spread is to find the range. But statisticians want to know if the data are
NORMAL DISTRIBTIONS MEASURES OF VARIATION In statistics, it is important to measure the spread of data. A simple way to measure spread is to find the range. But statisticians want to know if the data are
Discovering the Properties of Magic Sand
 NNIN Nanotechnology Education Teacher s Preparatory Guide Discovering the Properties of Magic Sand Purpose Explore the properties of molecular bonding Introduce students to the engineering of hydrophobic
NNIN Nanotechnology Education Teacher s Preparatory Guide Discovering the Properties of Magic Sand Purpose Explore the properties of molecular bonding Introduce students to the engineering of hydrophobic
Name Partners Date. Energy Diagrams I
 Name Partners Date Visual Quantum Mechanics The Next Generation Energy Diagrams I Goal Changes in energy are a good way to describe an object s motion. Here you will construct energy diagrams for a toy
Name Partners Date Visual Quantum Mechanics The Next Generation Energy Diagrams I Goal Changes in energy are a good way to describe an object s motion. Here you will construct energy diagrams for a toy
CHEMISTRY. Matter and Change. Section 13.1 Section 13.2 Section 13.3. The Gas Laws The Ideal Gas Law Gas Stoichiometry
 CHEMISTRY Matter and Change 13 Table Of Contents Chapter 13: Gases Section 13.1 Section 13.2 Section 13.3 The Gas Laws The Ideal Gas Law Gas Stoichiometry State the relationships among pressure, temperature,
CHEMISTRY Matter and Change 13 Table Of Contents Chapter 13: Gases Section 13.1 Section 13.2 Section 13.3 The Gas Laws The Ideal Gas Law Gas Stoichiometry State the relationships among pressure, temperature,
Chapter 14 Solutions
 Chapter 14 Solutions 1 14.1 General properties of solutions solution a system in which one or more substances are homogeneously mixed or dissolved in another substance two components in a solution: solute
Chapter 14 Solutions 1 14.1 General properties of solutions solution a system in which one or more substances are homogeneously mixed or dissolved in another substance two components in a solution: solute
Air Sampling for Gases and Vapors. Patrick N. Breysse, PhD, CIH Peter S.J. Lees, PhD, CIH Johns Hopkins University
 This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike License. Your use of this material constitutes acceptance of that license and the conditions of use of materials on this
This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike License. Your use of this material constitutes acceptance of that license and the conditions of use of materials on this
Exp 13 Volumetric Analysis: Acid-Base titration
 Exp 13 Volumetric Analysis: Acid-Base titration Exp. 13 video (time: 47:17 minutes) Titration - is the measurement of the volume of a standard solution required to completely react with a measured volume
Exp 13 Volumetric Analysis: Acid-Base titration Exp. 13 video (time: 47:17 minutes) Titration - is the measurement of the volume of a standard solution required to completely react with a measured volume
3M Thermally Conductive Adhesive Transfer Tapes
 Technical Data July 215 3M Thermally Conductive Adhesive Transfer Tapes 885 881 8815 882 Product Description 885, 881, 8815 and 882 are designed to provide a preferential heat-transfer path between heat-generating
Technical Data July 215 3M Thermally Conductive Adhesive Transfer Tapes 885 881 8815 882 Product Description 885, 881, 8815 and 882 are designed to provide a preferential heat-transfer path between heat-generating
Phase diagram of water. Note: for H 2 O melting point decreases with increasing pressure, for CO 2 melting point increases with increasing pressure.
 Phase diagram of water Note: for H 2 O melting point decreases with increasing pressure, for CO 2 melting point increases with increasing pressure. WATER Covers ~ 70% of the earth s surface Life on earth
Phase diagram of water Note: for H 2 O melting point decreases with increasing pressure, for CO 2 melting point increases with increasing pressure. WATER Covers ~ 70% of the earth s surface Life on earth
Molecular Geometry & Polarity
 Name AP Chemistry Molecular Geometry & Polarity Molecular Geometry A key to understanding the wide range of physical and chemical properties of substances is recognizing that atoms combine with other atoms
Name AP Chemistry Molecular Geometry & Polarity Molecular Geometry A key to understanding the wide range of physical and chemical properties of substances is recognizing that atoms combine with other atoms
9130 Kallo (Kieldrecht) Houston, TX 77015-6544 Phone: +32 3 570 28 78 Phone : 281-452-5951 Email : info@monumentchemical.com Fax : 281-457-1128
 Monument Chemical bvba Johann Haltermann Ltd. Haven 1972, Ketenislaan 3 16717 Jacintoport Blvd. 9130 Kallo (Kieldrecht) Houston, TX 77015-6544 Phone: +32 3 570 28 78 Phone : 281-452-5951 Email : info@monumentchemical.com
Monument Chemical bvba Johann Haltermann Ltd. Haven 1972, Ketenislaan 3 16717 Jacintoport Blvd. 9130 Kallo (Kieldrecht) Houston, TX 77015-6544 Phone: +32 3 570 28 78 Phone : 281-452-5951 Email : info@monumentchemical.com
Pre-Lab Notebook Content: Your notebook should include the title, date, purpose, procedure; data tables.
 Determination of Molar Mass by Freezing Point Depression M. Burkart & M. Kim Experimental Notes: Students work in pairs. Safety: Goggles and closed shoes must be worn. Dispose of all chemical in the plastic
Determination of Molar Mass by Freezing Point Depression M. Burkart & M. Kim Experimental Notes: Students work in pairs. Safety: Goggles and closed shoes must be worn. Dispose of all chemical in the plastic
Assessment and Solutions for Hole Wall Pull Away in High Tg and High Technology Laminate Materials
 Assessment and Solutions for Hole Wall Pull Away in High Tg and High Technology Laminate Materials Neil Patton Atotech Deutschland GmbH Erasmusstrasse 20 10553 Berlin Germany ABSTRACT Today the use of
Assessment and Solutions for Hole Wall Pull Away in High Tg and High Technology Laminate Materials Neil Patton Atotech Deutschland GmbH Erasmusstrasse 20 10553 Berlin Germany ABSTRACT Today the use of
Laboratory Exercise: Calibration of a Thermometer
 CHEM 109 Introduction themistry Revision 3.1 Laboratory Exercise: Calibration of a Thermometer In this exercise we will calibrate a stem-type thermometer and then use it to correctly measure the Air temperature
CHEM 109 Introduction themistry Revision 3.1 Laboratory Exercise: Calibration of a Thermometer In this exercise we will calibrate a stem-type thermometer and then use it to correctly measure the Air temperature
HEXANES. Insoluble in water, soluble in ether, alcohol, and acetone. Neutral to methyl orange (ph indicator) Not more than 0.
 HEXANES Prepared at the 51st JECFA (1998), published in FNP 52 Add 6 (1998) superseding specifications prepared at the 14th JECFA (1970), published in NMRS 48B (1971) and in FNP 52 (1992). ADI "limited
HEXANES Prepared at the 51st JECFA (1998), published in FNP 52 Add 6 (1998) superseding specifications prepared at the 14th JECFA (1970), published in NMRS 48B (1971) and in FNP 52 (1992). ADI "limited
Paper Chromatography: Separation and Identification of Five Metal Cations
 Paper Chromatography: Separation and Identification of Five Metal Cations Objectives Known and unknown solutions of the metal ions Ag +, Fe 3+, Co 2+, Cu 2+ and Hg 2+ will be analyzed using paper chromatography.
Paper Chromatography: Separation and Identification of Five Metal Cations Objectives Known and unknown solutions of the metal ions Ag +, Fe 3+, Co 2+, Cu 2+ and Hg 2+ will be analyzed using paper chromatography.
Chapter 10. Key Ideas Correlation, Correlation Coefficient (r),
 Chapter 0 Key Ideas Correlation, Correlation Coefficient (r), Section 0-: Overview We have already explored the basics of describing single variable data sets. However, when two quantitative variables
Chapter 0 Key Ideas Correlation, Correlation Coefficient (r), Section 0-: Overview We have already explored the basics of describing single variable data sets. However, when two quantitative variables
Selective Permeation of Solvents through Plastic Bottles
 PO. BOX 377, MS 4H-26 Seattle, WA 98124-227 Phone: (26) 544-1222 Fax: (26) 544-325 Introduction Loss of material, whether by spill, leakage, evaporation, etc. translates directly to loss on the bottom
PO. BOX 377, MS 4H-26 Seattle, WA 98124-227 Phone: (26) 544-1222 Fax: (26) 544-325 Introduction Loss of material, whether by spill, leakage, evaporation, etc. translates directly to loss on the bottom
Volumetric Analysis. Lecture 5 Experiment 9 in Beran page 109 Prelab = Page 115
 Volumetric Analysis Lecture 5 Experiment 9 in Beran page 109 Prelab = Page 115 Experimental Aims To prepare and standardize (determine concentration) a NaOH solution Using your standardized NaOH calculate
Volumetric Analysis Lecture 5 Experiment 9 in Beran page 109 Prelab = Page 115 Experimental Aims To prepare and standardize (determine concentration) a NaOH solution Using your standardized NaOH calculate
Photocatalyst Activity Indicator Ink for medium activity self-cleaning films such as glass. First Working Draft
 1 Photocatalyst Activity Indicator Ink for medium activity self-cleaning films such as glass. First Working Draft Contents 1. Scope 2. Normative references 3. Terms and Definitions 4. Symbols 5. Principle
1 Photocatalyst Activity Indicator Ink for medium activity self-cleaning films such as glass. First Working Draft Contents 1. Scope 2. Normative references 3. Terms and Definitions 4. Symbols 5. Principle
WORKSHEET: ph and Water 23 Lab Instructor: Lab Day & Time: Student name(s): drain cleaner flat soda pop window cleaner ammonia.
 WORKSHEET: ph and Water 23 Lab Instructor: Lab Day & Time: Student name(s): DATA AND ANALYSIS ph Substance ph Value Substance ph Value Substance ph Value pure water apple juice dish soap sugar water beer
WORKSHEET: ph and Water 23 Lab Instructor: Lab Day & Time: Student name(s): DATA AND ANALYSIS ph Substance ph Value Substance ph Value Substance ph Value pure water apple juice dish soap sugar water beer
Lab 25. Acid-Base Titration and Neutralization Reactions: What Is the Concentration of Acetic Acid in Each Sample of Vinegar?
 Lab 25. Acid-Base Titration and Neutralization Reactions: What Is the Concentration of Acetic Acid in Each Sample of Vinegar? Introduction Vinegar is basically a solution of acetic acid (CH3COOH). It is
Lab 25. Acid-Base Titration and Neutralization Reactions: What Is the Concentration of Acetic Acid in Each Sample of Vinegar? Introduction Vinegar is basically a solution of acetic acid (CH3COOH). It is
COMPOSITION / INFORMATION ON INGREDIENTS
 MATERIAL SAFETY DATA SHEET DAMTITE WATERPROOFER- WHITE 01071, 01211, 01451 DAMTITE POWDER FOUNDATION WATERPROOFER GRAY 02451 SECTION I. COMPANY & PRODUCT INFORMATION Product Identification Manufacturer
MATERIAL SAFETY DATA SHEET DAMTITE WATERPROOFER- WHITE 01071, 01211, 01451 DAMTITE POWDER FOUNDATION WATERPROOFER GRAY 02451 SECTION I. COMPANY & PRODUCT INFORMATION Product Identification Manufacturer
Chapter 5 Classification of Organic Compounds by Solubility
 Chapter 5 Classification of Organic Compounds by Solubility Deductions based upon interpretation of simple solubility tests can be extremely useful in organic structure determination. Both solubility and
Chapter 5 Classification of Organic Compounds by Solubility Deductions based upon interpretation of simple solubility tests can be extremely useful in organic structure determination. Both solubility and
Phenolphthalein-NaOH Kinetics
 Phenolphthalein-NaOH Kinetics Phenolphthalein is one of the most common acid-base indicators used to determine the end point in acid-base titrations. It is also the active ingredient in some laxatives.
Phenolphthalein-NaOH Kinetics Phenolphthalein is one of the most common acid-base indicators used to determine the end point in acid-base titrations. It is also the active ingredient in some laxatives.
Laboratory 22: Properties of Alcohols
 Introduction Alcohols represent and important class of organic molecules. In this experiment you will study the physical and chemical properties of alcohols. Solubility in water, and organic solvents,
Introduction Alcohols represent and important class of organic molecules. In this experiment you will study the physical and chemical properties of alcohols. Solubility in water, and organic solvents,
PREPARATION AND PROPERTIES OF A SOAP
 (adapted from Blackburn et al., Laboratory Manual to Accompany World of Chemistry, 2 nd ed., (1996) Saunders College Publishing: Fort Worth) Purpose: To prepare a sample of soap and to examine its properties.
(adapted from Blackburn et al., Laboratory Manual to Accompany World of Chemistry, 2 nd ed., (1996) Saunders College Publishing: Fort Worth) Purpose: To prepare a sample of soap and to examine its properties.
Chapter 2 Polar Covalent Bonds; Acids and Bases
 John E. McMurry http://www.cengage.com/chemistry/mcmurry Chapter 2 Polar Covalent Bonds; Acids and Bases Javier E. Horta, M.D., Ph.D. University of Massachusetts Lowell Polar Covalent Bonds: Electronegativity
John E. McMurry http://www.cengage.com/chemistry/mcmurry Chapter 2 Polar Covalent Bonds; Acids and Bases Javier E. Horta, M.D., Ph.D. University of Massachusetts Lowell Polar Covalent Bonds: Electronegativity
Objective: Preparation of Materials Lesson Instructions
 Smectite Clay Products Teaching Materials Audrey C. Rule Objective: Students will be able to match cards that tell smectite clay properties and uses with corresponding clay products to demonstrate their
Smectite Clay Products Teaching Materials Audrey C. Rule Objective: Students will be able to match cards that tell smectite clay properties and uses with corresponding clay products to demonstrate their
CHEMISTRY 101 EXAM 3 (FORM B) DR. SIMON NORTH
 1. Is H 3 O + polar or non-polar? (1 point) a) Polar b) Non-polar CHEMISTRY 101 EXAM 3 (FORM B) DR. SIMON NORTH 2. The bond strength is considerably greater in HF than in the other three hydrogen halides
1. Is H 3 O + polar or non-polar? (1 point) a) Polar b) Non-polar CHEMISTRY 101 EXAM 3 (FORM B) DR. SIMON NORTH 2. The bond strength is considerably greater in HF than in the other three hydrogen halides
Determination of Molar Mass by Freezing-Point Depression
 DETERMINATION OF MOLAR MASS BY FREEZING-POINT DEPRESSION 141 Determination of Molar Mass by Freezing-Point Depression OBJECTIVES: Gain familiarity with colligative properties of nonelectrolyte solutions
DETERMINATION OF MOLAR MASS BY FREEZING-POINT DEPRESSION 141 Determination of Molar Mass by Freezing-Point Depression OBJECTIVES: Gain familiarity with colligative properties of nonelectrolyte solutions
5. Which temperature is equal to +20 K? 1) 253ºC 2) 293ºC 3) 253 C 4) 293 C
 1. The average kinetic energy of water molecules increases when 1) H 2 O(s) changes to H 2 O( ) at 0ºC 3) H 2 O( ) at 10ºC changes to H 2 O( ) at 20ºC 2) H 2 O( ) changes to H 2 O(s) at 0ºC 4) H 2 O( )
1. The average kinetic energy of water molecules increases when 1) H 2 O(s) changes to H 2 O( ) at 0ºC 3) H 2 O( ) at 10ºC changes to H 2 O( ) at 20ºC 2) H 2 O( ) changes to H 2 O(s) at 0ºC 4) H 2 O( )
Simple Regression Theory II 2010 Samuel L. Baker
 SIMPLE REGRESSION THEORY II 1 Simple Regression Theory II 2010 Samuel L. Baker Assessing how good the regression equation is likely to be Assignment 1A gets into drawing inferences about how close the
SIMPLE REGRESSION THEORY II 1 Simple Regression Theory II 2010 Samuel L. Baker Assessing how good the regression equation is likely to be Assignment 1A gets into drawing inferences about how close the
Acids and Bases: A Brief Review
 Acids and : A Brief Review Acids: taste sour and cause dyes to change color. : taste bitter and feel soapy. Arrhenius: acids increase [H ] bases increase [OH ] in solution. Arrhenius: acid base salt water.
Acids and : A Brief Review Acids: taste sour and cause dyes to change color. : taste bitter and feel soapy. Arrhenius: acids increase [H ] bases increase [OH ] in solution. Arrhenius: acid base salt water.
Lecture 3: Models of Solutions
 Materials Science & Metallurgy Master of Philosophy, Materials Modelling, Course MP4, Thermodynamics and Phase Diagrams, H. K. D. H. Bhadeshia Lecture 3: Models of Solutions List of Symbols Symbol G M
Materials Science & Metallurgy Master of Philosophy, Materials Modelling, Course MP4, Thermodynamics and Phase Diagrams, H. K. D. H. Bhadeshia Lecture 3: Models of Solutions List of Symbols Symbol G M
Crystal Clear Sealant
 Crystal Clear Sealant Technical Data February, 2012 Product Description 3M Crystal Clear Sealant is a one component thermoplastic elastomer which forms permanent elastic bonds when used in surface sealing
Crystal Clear Sealant Technical Data February, 2012 Product Description 3M Crystal Clear Sealant is a one component thermoplastic elastomer which forms permanent elastic bonds when used in surface sealing
ISANE Isoparaffin Products
 ISANE Isoparaffin Products High Product Purity The Isane range is a unique range of products that is characterized by a high purity level, low odor, low surface tension, and relative chemical inertness.
ISANE Isoparaffin Products High Product Purity The Isane range is a unique range of products that is characterized by a high purity level, low odor, low surface tension, and relative chemical inertness.
PAINTING OVER HOT DIP GALVANIZED STEEL
 PAINTING OVER HOT DIP GALVANIZED STEEL Thomas J. Langill, Ph.D., Technical Director American Galvanizers Association 12200 E. Iliff Ave. Suite 204 Aurora, CO 80014 Abstract: Duplex systems, also known
PAINTING OVER HOT DIP GALVANIZED STEEL Thomas J. Langill, Ph.D., Technical Director American Galvanizers Association 12200 E. Iliff Ave. Suite 204 Aurora, CO 80014 Abstract: Duplex systems, also known
Waterproofing System for Wastewater Tanks in Petrochemical Industries and Refineries
 Waterproofing System for Wastewater Tanks in Petrochemical Industries and Refineries Introduction Wastewater of petrochemical industries and refineries contains high amounts of emulsified aliphatic or
Waterproofing System for Wastewater Tanks in Petrochemical Industries and Refineries Introduction Wastewater of petrochemical industries and refineries contains high amounts of emulsified aliphatic or
One-Way ANOVA using SPSS 11.0. SPSS ANOVA procedures found in the Compare Means analyses. Specifically, we demonstrate
 1 One-Way ANOVA using SPSS 11.0 This section covers steps for testing the difference between three or more group means using the SPSS ANOVA procedures found in the Compare Means analyses. Specifically,
1 One-Way ANOVA using SPSS 11.0 This section covers steps for testing the difference between three or more group means using the SPSS ANOVA procedures found in the Compare Means analyses. Specifically,
Lab #11: Determination of a Chemical Equilibrium Constant
 Lab #11: Determination of a Chemical Equilibrium Constant Objectives: 1. Determine the equilibrium constant of the formation of the thiocyanatoiron (III) ions. 2. Understand the application of using a
Lab #11: Determination of a Chemical Equilibrium Constant Objectives: 1. Determine the equilibrium constant of the formation of the thiocyanatoiron (III) ions. 2. Understand the application of using a
Introduction to Chemistry. Course Description
 CHM 1025 & CHM 1025L Introduction to Chemistry Course Description CHM 1025 Introduction to Chemistry (3) P CHM 1025L Introduction to Chemistry Laboratory (1) P This introductory course is intended to introduce
CHM 1025 & CHM 1025L Introduction to Chemistry Course Description CHM 1025 Introduction to Chemistry (3) P CHM 1025L Introduction to Chemistry Laboratory (1) P This introductory course is intended to introduce
99.37, 99.38, 99.38, 99.39, 99.39, 99.39, 99.39, 99.40, 99.41, 99.42 cm
 Error Analysis and the Gaussian Distribution In experimental science theory lives or dies based on the results of experimental evidence and thus the analysis of this evidence is a critical part of the
Error Analysis and the Gaussian Distribution In experimental science theory lives or dies based on the results of experimental evidence and thus the analysis of this evidence is a critical part of the
Determination of the Amount of Acid Neutralized by an Antacid Tablet Using Back Titration
 Determination of the Amount of Acid Neutralized by an Antacid Tablet Using Back Titration GOAL AND OVERVIEW Antacids are bases that react stoichiometrically with acid. The number of moles of acid that
Determination of the Amount of Acid Neutralized by an Antacid Tablet Using Back Titration GOAL AND OVERVIEW Antacids are bases that react stoichiometrically with acid. The number of moles of acid that
Acid-Base Extraction.
 Acid-Base Extraction. Extraction involves dissolving a compound or compounds either (1) from a solid into a solvent or (2) from a solution into another solvent. A familiar example of the first case is
Acid-Base Extraction. Extraction involves dissolving a compound or compounds either (1) from a solid into a solvent or (2) from a solution into another solvent. A familiar example of the first case is
CRYOCOOLER CONTAMINATION STUDY. S.W.K. Yuan, D.T.Kuo, and A.S. Loc. BEI Technologies, Cryocooler Group Sylmar, CA 91342 ABSTRACT
 CRYOCOOLER CONTAMINATION STUDY S.W.K. Yuan, D.T.Kuo, and A.S. Loc EI Technologies, Cryocooler Group Sylmar, CA 91342 ASTRACT Eliminating or reduction of contamination is essential for the success of cryocoolers
CRYOCOOLER CONTAMINATION STUDY S.W.K. Yuan, D.T.Kuo, and A.S. Loc EI Technologies, Cryocooler Group Sylmar, CA 91342 ASTRACT Eliminating or reduction of contamination is essential for the success of cryocoolers
The Properties of Water (Instruction Sheet)
 The Properties of Water (Instruction Sheet) Property : High Polarity Activity #1 Surface Tension: PILE IT ON. Materials: 1 DRY penny, 1 eye dropper, water. 1. Make sure the penny is dry. 2. Begin by estimating
The Properties of Water (Instruction Sheet) Property : High Polarity Activity #1 Surface Tension: PILE IT ON. Materials: 1 DRY penny, 1 eye dropper, water. 1. Make sure the penny is dry. 2. Begin by estimating
Prentice Hall. Chemistry (Wilbraham) 2008, National Student Edition - South Carolina Teacher s Edition. High School. High School
 Prentice Hall Chemistry (Wilbraham) 2008, National Student Edition - South Carolina Teacher s Edition High School C O R R E L A T E D T O High School C-1.1 Apply established rules for significant digits,
Prentice Hall Chemistry (Wilbraham) 2008, National Student Edition - South Carolina Teacher s Edition High School C O R R E L A T E D T O High School C-1.1 Apply established rules for significant digits,
KINETIC MOLECULAR THEORY OF MATTER
 KINETIC MOLECULAR THEORY OF MATTER The kinetic-molecular theory is based on the idea that particles of matter are always in motion. The theory can be used to explain the properties of solids, liquids,
KINETIC MOLECULAR THEORY OF MATTER The kinetic-molecular theory is based on the idea that particles of matter are always in motion. The theory can be used to explain the properties of solids, liquids,
An acid is a substance that produces H + (H 3 O + ) Ions in aqueous solution. A base is a substance that produces OH - ions in aqueous solution.
 Chapter 8 Acids and Bases Definitions Arrhenius definitions: An acid is a substance that produces H + (H 3 O + ) Ions in aqueous solution. A base is a substance that produces OH - ions in aqueous solution.
Chapter 8 Acids and Bases Definitions Arrhenius definitions: An acid is a substance that produces H + (H 3 O + ) Ions in aqueous solution. A base is a substance that produces OH - ions in aqueous solution.
Chemistry. The student will be able to identify and apply basic safety procedures and identify basic equipment.
 Chemistry UNIT I: Introduction to Chemistry The student will be able to describe what chemistry is and its scope. a. Define chemistry. b. Explain that chemistry overlaps many other areas of science. The
Chemistry UNIT I: Introduction to Chemistry The student will be able to describe what chemistry is and its scope. a. Define chemistry. b. Explain that chemistry overlaps many other areas of science. The
Chemical Basis of Life Module A Anchor 2
 Chemical Basis of Life Module A Anchor 2 Key Concepts: - Water is a polar molecule. Therefore, it is able to form multiple hydrogen bonds, which account for many of its special properties. - Water s polarity
Chemical Basis of Life Module A Anchor 2 Key Concepts: - Water is a polar molecule. Therefore, it is able to form multiple hydrogen bonds, which account for many of its special properties. - Water s polarity
SPE and HPLC. Dr Iva Chianella Lecturer in Analytical Chemistry Cranfield Health +44 (0) 1234 758322. i.chianella.1998@cranfield.ac.
 SPE and HPLC Dr Iva Chianella Lecturer in Analytical Chemistry Cranfield Health +44 (0) 1234 758322 i.chianella.1998@cranfield.ac.uk Solid-Phase Extraction- SPE Simple, fast and efficient sample preparation
SPE and HPLC Dr Iva Chianella Lecturer in Analytical Chemistry Cranfield Health +44 (0) 1234 758322 i.chianella.1998@cranfield.ac.uk Solid-Phase Extraction- SPE Simple, fast and efficient sample preparation
RTV159. RTV157 and RTV159 High Strength Silicone Adhesive Sealants
 RTV159 RTV157 and RTV159 High Strength Silicone Adhesive Sealants Product Description RTV157 and RTV159 are one-component, ready-to-use, high strength silicone rubber adhesive sealants. They cure to tough
RTV159 RTV157 and RTV159 High Strength Silicone Adhesive Sealants Product Description RTV157 and RTV159 are one-component, ready-to-use, high strength silicone rubber adhesive sealants. They cure to tough
Q.1 Classify the following according to Lewis theory and Brønsted-Lowry theory.
 Acid-base A4 1 Acid-base theories ACIDS & BASES - IONIC EQUILIBRIA 1. LEWIS acid electron pair acceptor H, AlCl 3 base electron pair donor NH 3, H 2 O, C 2 H 5 OH, OH e.g. H 3 N: -> BF 3 > H 3 N BF 3 see
Acid-base A4 1 Acid-base theories ACIDS & BASES - IONIC EQUILIBRIA 1. LEWIS acid electron pair acceptor H, AlCl 3 base electron pair donor NH 3, H 2 O, C 2 H 5 OH, OH e.g. H 3 N: -> BF 3 > H 3 N BF 3 see
CHEM 36 General Chemistry EXAM #1 February 13, 2002
 CHEM 36 General Chemistry EXAM #1 February 13, 2002 Name: Serkey, Anne INSTRUCTIONS: Read through the entire exam before you begin. Answer all of the questions. For questions involving calculations, show
CHEM 36 General Chemistry EXAM #1 February 13, 2002 Name: Serkey, Anne INSTRUCTIONS: Read through the entire exam before you begin. Answer all of the questions. For questions involving calculations, show
expectation as hot dip galvanizing?
 FREQUENTLY ASKED QUESTIONS ********************************* Q. 1.What exactly is Cold galvanizing? A. Cold galvanisation is an organic, single-component zinc coating of minimum 92% (weight) zinc in the
FREQUENTLY ASKED QUESTIONS ********************************* Q. 1.What exactly is Cold galvanizing? A. Cold galvanisation is an organic, single-component zinc coating of minimum 92% (weight) zinc in the
Quantifying Bacterial Concentration using a Calibrated Growth Curve
 BTEC 4200 Lab 2. Quantifying Bacterial Concentration using a Calibrated Growth Curve Background and References Bacterial concentration can be measured by several methods, all of which you have studied
BTEC 4200 Lab 2. Quantifying Bacterial Concentration using a Calibrated Growth Curve Background and References Bacterial concentration can be measured by several methods, all of which you have studied
CeramClean II Aluminum Safe
 1. Identification of substance: MATERIAL SAFETY DATA SHEET DIVISION Reviewed on 07/01/2004 Product details: Trade name: CeramClean II Ink Cleaner Product Code: 332506 Manufacturer/Supplier: Harper Corporation
1. Identification of substance: MATERIAL SAFETY DATA SHEET DIVISION Reviewed on 07/01/2004 Product details: Trade name: CeramClean II Ink Cleaner Product Code: 332506 Manufacturer/Supplier: Harper Corporation
