Ecotoxicology. Biotransformation
|
|
|
- Charleen Gregory
- 9 years ago
- Views:
Transcription
1 Ecotoxicology Biotransformation
2 RÉSUMÉ UPTAKE I RGAISM DEPEDS : Lipophilicity (polarization, ionization) Route of uptake Concentration Molecular size UPTAKE I RGAS DEPEDS : Vascularization Binding mechanisms in blood Lipophilicity Binding sites in the cells of the organ
3 Target organ Absorption Bound Free Bound Bound Free Free Depot Adipose tissues Inert membranes Lipoprotein micells Lysosomes Skeleton (endo or exo) Excretion Urine Faeces (gall) Lungs or gills Secretion from surface
4 rganism s defence against xenobiotics Fast excretion Deposition in less susceptible organs (fat depots, skeleton) Deposition in intracellular organelles Formation of complexes (i.e. metallothionin and Se/g) Biotransformation
5 Uptake and excretion of hydrophilic and lipophilic compounds UPTAKE UPTAKE UPTAKE RGA RGA BITRAS- FRMATI EXCRETI EXCRETI EXCRETI Primarily biotransformation makes lipophilic compounds more hydrophilic
6 XEBITIC EXCRETI on-toxic metabolite BLD BITRAS- FRMATI Activation Detoxification Toxic metabolite Somatic effect DA damage Definition Biotransformation is the sum of all processes, whereby a compound is transformed chemically within a living organism
7 Phase I and phase II reactions PASE I PASE II Expose or add functional group XEBITIC xidation Reduction ydrolysis PRIMARY PRDUCT Conjugation SECUDARY PRDUCT EXCRETI LIPPILIC YDRPILIC
8 Phase I reactions
9 Mixed function oxidase enzymes (P450) are located in the endoplasmic reticulum (SER)
10 Important phase I enzymes Enzyme Co-factor Substrate Mixed-function oxidases ADP Most lipophilic substances (cytochrome P-450) (AD) with M.wt < 800 Carboxyl esterases Unknown Lipophilic carboxyl esters A esterases Ca ++ rganophosphate esteres Epoxide hydrolases Unknown rganic epoxids Reduktases AD rganic nitrous compounds ADP rganic halogens
11 P-450 system in the endoplasmic reticulum REDUCTASE P-450 P-450
12 Classification and evolution of the P-450 gene-family Millioner år før nutid 2,000 1,500 1,000 LI I II III IV XIX D A B C E XXI XVII CI XI A B I-IV involved in phase I reactions XI, XVII, XIX, XXI participate in the biosynthesis of steroid hormones
13 Cytochrome P-450 s catalytic cycle Xenobiotic Fe3+ Fe3+ CYT P- 450 ADP ADP + e - CYT P-450 reductase Fe3+ Fe Fe3+ eṉadp Fe2+ ADP ADP ADP+ Fp oxidized e - Fp reduced (R)-P450-(Fe2+) (R)-P450-(Fe3+) 2 e - R (R)-P450-(Fe2+) 2 P450 (Fe3+) R + 2
14 Examples of oxidations catalysed by P-450 Aliphatic hydroxylation Sulphur oxidation R - C 2 C 2 C 3 R C 2 C C 3 R -S-R R -S-R Aromatic hydroxylation R R Epoxidation R - C C - R R - C - C - R -, -, or S-dealkylation R - (,, S) - C 3 R ( 2,, S) + C 2 De-sulphurnation S R 1 R 2 P - X R 1 R 2 P - X + S xidative dehalogenation X X R - C - R - C - R - C - + X Deamination R C 2 2 R - C hydroxylation R - - C C 3 R - - C C 3
15 ther phase I enzymes C 2 C 2 S P C 2 5 C 2 5 M C 2 C 2 P C 2 5 C 2 5 C 3 Diazinone C 3 Diazoxon C 2 C 2 P C 2 5 C 2 5 A esterase C 2 C 2 P + C 2 5 C 2 5 C 3 C 3 Diazoxon
16 ther phase I enzymes Cl Cl CC 2 B esterase Cl Cl C 2 C Permethrine Epoxide hydrolase Benzo(a)pyrene 7,8 oxide 2 2 itroreductase itropyrene
17 Phase II reactions
18 PASE I PASE II Expose or add functional group XEBITIC xidation Reduction ydrolysis PRIMARY PRDUCT Conjugation SECUDARY PRDUCT EXCRETI LIPPILIC YDRPILIC
19 Two important co-factors in phase II conjugations UDP and PAPS C P P C 2 S P C 2 2 Uridine-5 -diphosphoα-d-glucuronic acid (UDP-GA) 3 -Phosphoadenosine- 5 -phosphosulfate (PAPS)
20 Glucuronyl transferase conjugations C C R + UDP Glucuronyl transferase R + UDP UDP (uridin diphosphate) delivers the energy to the conjugation process Important phase II reactions for both exo- and endogenous compounds Many forms with a wide range of substrates Localised in SER in close connection with the MF-system The resulting glucuronides are excreted in urine and faeces
21 Examples of Glucuronide conjugations -Glucuronid -C -G -C - -G - C = C - G -Glucuronide - C -G Alcohol Aliphatic Alicyclic Phenolic Carboxyl acid Aliphatic Aromatic α,β-unsaturated ketone Carbamate Trichloroethanol exobarbital Estrone α-ethylhexanoic acid o-aminobenzoic acid Progesterone Meprobamate R S 2 -G Sulfonamide Sulfadimethoxine S-Glucuronide Ar S - G Aryl thiol Thiophenol C-Glucuronide -C -G 1,3-Dicarbonyl system Phenylbutazone
22 Sulfotransferase conjugation R + PAPS Sulfotransferase R S + ADP PAPS (Phosphoadenine phosphosulphate) delivers the energy Localised in the cytosol Adds sulphate to -groups (phenols and aliphatic alcohols) Also important for the transformation of endogenous low-molecular compounds (catacholamins, hydroxy-steroids, bile salts) The conjugates are primarily excreted in the urine
23 Glutathione Glutamic acid Cysteine Glycine S + + S Glutathione
24 Glutathione S-transferase C - C C C - SG + GS Glutathion S-transferase 1,2-Epoxyetylbenzene GS = reduced glutathione (tripeptide) glutathione s S group attacks electrophilic (reactive) C-atoms predominantly localised in the cytosol several enzymatic cleavages of glutathione after conjugation ends with a derivate of mercapturic acid, which is excreted in the urine R SC 2 CC CC 3
25 Glutathione S-transferase reactions Glutathione S-alkyltransferase Glutathione S-alkenetransferase C 3 I + GS C 3 -SG + I Methyl iodide Glutathione S-aryltransferase CCC 2 5 CCC GS C 2 CC 2 5 GS-CCC 2 5 Diethyl maleate Cl Cl Glutathione S-aryl epoxidetransferase Cl SG + GS + Cl P-450 GS S 2 3,4-Dichloronitrobenzen 2 aphthalene aphthalene oxide Glutathione S-aralkyltransferase C 2 Cl C 2 SG + GS + Cl Benzyl chloride
26 Induction of biotransformation enzymes
27 Characteristics of the hepatic effects of Phenobarbital and Benzo[a]pyren (PA) CARACTERISTICS PEBARBITAL PA nset of effect 8-12 hours 3-6 hours Time of maximum effects 3-5 days hours Persistence of induction 5-7 days 5-12 days Liver enlargement marked slight Protein synthesis large increase small increase Phosphorlipid synthesis marked increase no effect Liver blood flow increased no effects Biliary flow increased no effect Enzyme components Cytochrome P-450 increased no effect Cytochrome P-448 no effect increased ADR-cytochrome reductase increased no effect Substrate specificity -Demethylation increased no effect Aliphatic hydroxylation increased no effect PA hydroxylation small increase increased Glucuronidation increased small increase Glutathione conjugation small increase small increase Epoxide hydrolase increased small increase
28 Examples of other inducers alogenated pesticides (DDT, aldrin, lindan, chlordan) PCB Steroids Chlorinated dioxins (TCDD) Alcohol and acetone
29 Induction of cytochrome P-450 Ah receptor-hsp90 Cell C (inducer) C C-AhR hsp90 C-AhR ucleus P450 gen XRE P450 protein P450 mra Bioactivation Detoxification Toxicity Elimination C: ydrocarbon (inducer) XRC: Regulator gene (stimulates transcription of P-450 gene)
30 Bioactivation
31 Bioactivation is define as: Enzymatically formed metabolites, which are more reactive than the mother substance and excreted metabolites The most significant toxicological effects of xenobiotics are reactive metabolites - can react with nucleophilic sites - S groups (glutathione, cystein) - 2 and C groups (DA, RA, proteins) Imbalance between formation and detoxification of reactive metabolites can arise from: - enzyme induction (increased biotransformation and formation of reactive metabolites) - high dose of xenobiotic depletion of cellular defence mechanisms saturation of non-toxic pathways
32 Examples of bioactivating compounds Reactive pathway Factors increasing Stof or intermediate product toxicity Acetaminophen -hydroxylation Sulphate and GS depletion Acetylhydrazine Aflatoxin B Benzen -hydroxylation Epoxidation Epoxidation Benzo[a]pyren Epoxidation Further metabolism PCB Epoxidation GS depletion Tetrachlorcarbon Free radicals Reductive metabolism alotane Free radicals Reductive metabolism Parathion xidation with sulphur formation
33 Activation of Paracetamol Acetaminophen (Paracetamol) ADP 2 CC 3 Activation of cyt. P-450 5% CC 3 Sulfotransferase 95% Glucuronosyltransferase CC 3 CJUGATE CC 3 Mercapturic acid CC 3 GS cellular macro molecule At overdose Glutathione (GS) is depleted CC 3 *+ -Acetyl-p-Benzoquinoneimin Cellular macro molecule Liver damage
BIOMARKERS AND TOXICITY MECHANISMS 06 Mechanisms Metabolism & Detoxification. Luděk Bláha, PřF MU, RECETOX www.recetox.cz
 BIOMARKERS AND TOXICITY MECHANISMS 06 Mechanisms Metabolism & Detoxification Luděk Bláha, PřF MU, RECETOX www.recetox.cz Metabolism and detoxification Chemicals enter body... mostly via food Pass directly
BIOMARKERS AND TOXICITY MECHANISMS 06 Mechanisms Metabolism & Detoxification Luděk Bláha, PřF MU, RECETOX www.recetox.cz Metabolism and detoxification Chemicals enter body... mostly via food Pass directly
Lecture 4 Sunday 30/9/2012
 Inactivation and Detoxification of Xenobiotics and Metabolites in the Liver The liver is one of the most important organs in the body when it comes to detoxifying or getting rid of foreign substances or
Inactivation and Detoxification of Xenobiotics and Metabolites in the Liver The liver is one of the most important organs in the body when it comes to detoxifying or getting rid of foreign substances or
Chapter 22 Detoxification and Biotransformational Imbalances
 Chapter 22 Detoxification and Biotransformational Imbalances DeAnn Liska, PhD, Michael Lyon, MD, David S. Jones, MD The Detoxification Systems Introduction The concept that toxins accumulate in the body
Chapter 22 Detoxification and Biotransformational Imbalances DeAnn Liska, PhD, Michael Lyon, MD, David S. Jones, MD The Detoxification Systems Introduction The concept that toxins accumulate in the body
UDP-Glucuronosyltransferases (UGTs): Overview
 UDP-Glucuronosyltransferases (UGTs): Overview Anna Radominska-Pandya Department of Biochemistry and Molecular Biology University of Arkansas for Medical Sciences Little Rock, Arkansas, US October 2010;
UDP-Glucuronosyltransferases (UGTs): Overview Anna Radominska-Pandya Department of Biochemistry and Molecular Biology University of Arkansas for Medical Sciences Little Rock, Arkansas, US October 2010;
Mitigation Strategies for Reactive Intermediates in
 Mitigation Strategies for Reactive Intermediates in Drug Discovery ew Perspectives in DMPK: Informing Drug Discovery Royal Society of Chemistry, London February 10-11, 2014 Thomas A. Baillie School of
Mitigation Strategies for Reactive Intermediates in Drug Discovery ew Perspectives in DMPK: Informing Drug Discovery Royal Society of Chemistry, London February 10-11, 2014 Thomas A. Baillie School of
QSAR. The following lecture has drawn many examples from the online lectures by H. Kubinyi
 QSAR The following lecture has drawn many examples from the online lectures by H. Kubinyi LMU Institut für Informatik, LFE Bioinformatik, Cheminformatics, Structure independent methods J. Apostolakis 1
QSAR The following lecture has drawn many examples from the online lectures by H. Kubinyi LMU Institut für Informatik, LFE Bioinformatik, Cheminformatics, Structure independent methods J. Apostolakis 1
Lecture 6: Cholesterol (Ch. 9.1e, 9.2b, 19.7b,c) & Lipoproteins (Ch. 10.3*, 19.1, 19.7b,c)
 Lecture 6: Cholesterol (Ch. 9.1e, 9.2b, 19.7b,c) & Lipoproteins (Ch. 10.3*, 19.1, 19.7b,c) Next lecture: Fatty Acid Oxidation (Ch. 19.2), Ketone Bodies (Ch. 19.3) and Fatty Acid Biosynthesis (Ch. 19.4)
Lecture 6: Cholesterol (Ch. 9.1e, 9.2b, 19.7b,c) & Lipoproteins (Ch. 10.3*, 19.1, 19.7b,c) Next lecture: Fatty Acid Oxidation (Ch. 19.2), Ketone Bodies (Ch. 19.3) and Fatty Acid Biosynthesis (Ch. 19.4)
1- Fatty acids are activated to acyl-coas and the acyl group is further transferred to carnitine because:
 Section 10 Multiple Choice 1- Fatty acids are activated to acyl-coas and the acyl group is further transferred to carnitine because: A) acyl-carnitines readily cross the mitochondrial inner membrane, but
Section 10 Multiple Choice 1- Fatty acids are activated to acyl-coas and the acyl group is further transferred to carnitine because: A) acyl-carnitines readily cross the mitochondrial inner membrane, but
The Detoxification System Part II: Hepatic Biotransformation
 The Detoxification System Part II: Hepatic Biotransformation by Mark J. Donohue Introduction By the early 21st century our society has increasingly been exposed to more and more toxic compounds in the
The Detoxification System Part II: Hepatic Biotransformation by Mark J. Donohue Introduction By the early 21st century our society has increasingly been exposed to more and more toxic compounds in the
Anatomy and Physiology Placement Exam 2 Practice with Answers at End!
 Anatomy and Physiology Placement Exam 2 Practice with Answers at End! General Chemical Principles 1. bonds are characterized by the sharing of electrons between the participating atoms. a. hydrogen b.
Anatomy and Physiology Placement Exam 2 Practice with Answers at End! General Chemical Principles 1. bonds are characterized by the sharing of electrons between the participating atoms. a. hydrogen b.
Detoxification Pathways. Theory and Application to Practice
 Detoxification Pathways Theory and Application to Practice 1 Detoxification Pathways Failure to detoxify Accumulation of Toxins Detoxification mechanism Importance to practitioner Phase 1 Biotransformation
Detoxification Pathways Theory and Application to Practice 1 Detoxification Pathways Failure to detoxify Accumulation of Toxins Detoxification mechanism Importance to practitioner Phase 1 Biotransformation
Ionization of amino acids
 Amino Acids 20 common amino acids there are others found naturally but much less frequently Common structure for amino acid COOH, -NH 2, H and R functional groups all attached to the a carbon Ionization
Amino Acids 20 common amino acids there are others found naturally but much less frequently Common structure for amino acid COOH, -NH 2, H and R functional groups all attached to the a carbon Ionization
The Urea Cycle. April 11, 2003 Bryant Miles
 The Urea ycle April 11, 2003 Bryant Miles I. Ammonia Toxicity Every amino acid contains at least one amino group. Therefore every amino acid degradation pathway has a key step where the amino group is
The Urea ycle April 11, 2003 Bryant Miles I. Ammonia Toxicity Every amino acid contains at least one amino group. Therefore every amino acid degradation pathway has a key step where the amino group is
General Principles of Pharmacology and Toxicology
 General Principles of Pharmacology and Toxicology Parisa Gazerani, Pharm D, PhD Assistant Professor Center for Sensory-Motor Interaction (SMI) Department of Health Science and Technology Aalborg University
General Principles of Pharmacology and Toxicology Parisa Gazerani, Pharm D, PhD Assistant Professor Center for Sensory-Motor Interaction (SMI) Department of Health Science and Technology Aalborg University
Overview of Lipid Metabolism
 Overview of Lipid Metabolism Learning Objectives By the end of this lecture the students should be able to understand: Classification of Lipids The digestion, absorption and utilization of dietary lipids
Overview of Lipid Metabolism Learning Objectives By the end of this lecture the students should be able to understand: Classification of Lipids The digestion, absorption and utilization of dietary lipids
Actions of Hormones on Target Cells Page 1. Actions of Hormones on Target Cells Page 2. Goals/ What You Need to Know Goals What You Need to Know
 Actions of Hormones on Target Cells Graphics are used with permission of: Pearson Education Inc., publishing as Benjamin Cummings (http://www.aw-bc.com) Page 1. Actions of Hormones on Target Cells Hormones
Actions of Hormones on Target Cells Graphics are used with permission of: Pearson Education Inc., publishing as Benjamin Cummings (http://www.aw-bc.com) Page 1. Actions of Hormones on Target Cells Hormones
Paracetamol (Acetaminophen) Poisoning Evidence Based Review
 Paracetamol (Acetaminophen) Poisoning Evidence Based Review Context Deliberate self harm is a common reason for presentation to the ED, with paracetamol poisoning being one of the most common methods used
Paracetamol (Acetaminophen) Poisoning Evidence Based Review Context Deliberate self harm is a common reason for presentation to the ED, with paracetamol poisoning being one of the most common methods used
Glutathione and Oxidative Stress - Part I
 Glutathione and Oxidative Stress - Part I By: James L. Holly, MD Oxidative Stress refers to effects from endogenous (produced in the body) toxins (free radicals) produced in the body by normal metabolism
Glutathione and Oxidative Stress - Part I By: James L. Holly, MD Oxidative Stress refers to effects from endogenous (produced in the body) toxins (free radicals) produced in the body by normal metabolism
DetoxiGenomic Profile Patient's Copy
 TM DetoxiGenomic Profile Patient: JANE DOE DOB: April 09, 1969 Sex: F MRN: Order Number: Completed: February 26, 2014 Received: February 19, 2014 Collected: February 18, 2014 Security Code: PHASE I Detoxification:
TM DetoxiGenomic Profile Patient: JANE DOE DOB: April 09, 1969 Sex: F MRN: Order Number: Completed: February 26, 2014 Received: February 19, 2014 Collected: February 18, 2014 Security Code: PHASE I Detoxification:
Unsaturated and Odd-Chain Fatty Acid Catabolism
 Unsaturated and dd-hain Fatty Acid atabolism March 24, 2003 Bryant Miles The complete oxidation of saturated fatty acids containing an even number of carbon atoms is accomplished by the β-oxidation pathway.
Unsaturated and dd-hain Fatty Acid atabolism March 24, 2003 Bryant Miles The complete oxidation of saturated fatty acids containing an even number of carbon atoms is accomplished by the β-oxidation pathway.
DRUG METABOLISM CHAPTER. Frank J. Gonzalez and Robert H. Tukey
 CHAPTER 3 DRUG METABOLISM Frank J. Gonzalez and Robert H. Tukey How Humans Cope with Exposure to Xenobiotics. The ability of humans to metabolize and clear drugs is a natural process that involves the
CHAPTER 3 DRUG METABOLISM Frank J. Gonzalez and Robert H. Tukey How Humans Cope with Exposure to Xenobiotics. The ability of humans to metabolize and clear drugs is a natural process that involves the
Exam 4 Outline CH 105 Spring 2012
 Exam 4 Outline CH 105 Spring 2012 You need to bring a pencil and your ACT card. Chapter 24: Lipids 1. Describe the properties and types of lipids a. All are hydrophobic b. Fatty acid-based typically contain
Exam 4 Outline CH 105 Spring 2012 You need to bring a pencil and your ACT card. Chapter 24: Lipids 1. Describe the properties and types of lipids a. All are hydrophobic b. Fatty acid-based typically contain
4/18/2011. 9.8 Substituent Effects in Electrophilic Substitutions. Substituent Effects in Electrophilic Substitutions
 9.8 Substituent effects in the electrophilic substitution of an aromatic ring Substituents affect the reactivity of the aromatic ring Some substituents activate the ring, making it more reactive than benzene
9.8 Substituent effects in the electrophilic substitution of an aromatic ring Substituents affect the reactivity of the aromatic ring Some substituents activate the ring, making it more reactive than benzene
Electrophilic Aromatic Substitution
 Electrophilic Aromatic Substitution Electrophilic substitution is the typical reaction type for aromatic rings. Generalized electrophilic aromatic substitution: E E Electrophile Lewis acid: may be or neutral.
Electrophilic Aromatic Substitution Electrophilic substitution is the typical reaction type for aromatic rings. Generalized electrophilic aromatic substitution: E E Electrophile Lewis acid: may be or neutral.
Carbohydrates, proteins and lipids
 Carbohydrates, proteins and lipids Chapter 3 MACROMOLECULES Macromolecules: polymers with molecular weights >1,000 Functional groups THE FOUR MACROMOLECULES IN LIFE Molecules in living organisms: proteins,
Carbohydrates, proteins and lipids Chapter 3 MACROMOLECULES Macromolecules: polymers with molecular weights >1,000 Functional groups THE FOUR MACROMOLECULES IN LIFE Molecules in living organisms: proteins,
Detoxification & Your Health
 Detoxification & Your Health Detoxification is the metabolic process your body uses to transform and eliminate toxins. The process can occur in two steps, called Phase I and Phase II. Phase I is our first
Detoxification & Your Health Detoxification is the metabolic process your body uses to transform and eliminate toxins. The process can occur in two steps, called Phase I and Phase II. Phase I is our first
COMMITTEE FOR VETERINARY MEDICINAL PRODUCTS
 The European Agency for the Evaluation of Medicinal Products EMEA/MRL/551/99-FINAL February 1999 COMMITTEE FOR VETERINARY MEDICINAL PRODUCTS PARACETAMOL SUMMARY REPORT 1. Paracetamol (synonym: acetaminophen)
The European Agency for the Evaluation of Medicinal Products EMEA/MRL/551/99-FINAL February 1999 COMMITTEE FOR VETERINARY MEDICINAL PRODUCTS PARACETAMOL SUMMARY REPORT 1. Paracetamol (synonym: acetaminophen)
Chapter 25: Metabolism and Nutrition
 Chapter 25: Metabolism and Nutrition Chapter Objectives INTRODUCTION 1. Generalize the way in which nutrients are processed through the three major metabolic fates in order to perform various energetic
Chapter 25: Metabolism and Nutrition Chapter Objectives INTRODUCTION 1. Generalize the way in which nutrients are processed through the three major metabolic fates in order to perform various energetic
H H N - C - C 2 R. Three possible forms (not counting R group) depending on ph
 Amino acids - 0 common amino acids there are others found naturally but much less frequently - Common structure for amino acid - C, -N, and functional groups all attached to the alpha carbon N - C - C
Amino acids - 0 common amino acids there are others found naturally but much less frequently - Common structure for amino acid - C, -N, and functional groups all attached to the alpha carbon N - C - C
Hormones & Chemical Signaling
 Hormones & Chemical Signaling Part 2 modulation of signal pathways and hormone classification & function How are these pathways controlled? Receptors are proteins! Subject to Specificity of binding Competition
Hormones & Chemical Signaling Part 2 modulation of signal pathways and hormone classification & function How are these pathways controlled? Receptors are proteins! Subject to Specificity of binding Competition
Absorption of Drugs. Transport of a drug from the GI tract
 Absorption of Drugs Absorption is the transfer of a drug from its site of administration to the bloodstream. The rate and efficiency of absorption depend on the route of administration. For IV delivery,
Absorption of Drugs Absorption is the transfer of a drug from its site of administration to the bloodstream. The rate and efficiency of absorption depend on the route of administration. For IV delivery,
Alcohols An alcohol contains a hydroxyl group ( OH) attached to a carbon chain. A phenol contains a hydroxyl group ( OH) attached to a benzene ring.
 Chapter : rganic Compounds with xygen Alcohols, Ethers Alcohols An alcohol contains a hydroxyl group ( H) attached to a carbon chain. A phenol contains a hydroxyl group ( H) attached to a benzene ring.
Chapter : rganic Compounds with xygen Alcohols, Ethers Alcohols An alcohol contains a hydroxyl group ( H) attached to a carbon chain. A phenol contains a hydroxyl group ( H) attached to a benzene ring.
(Woods) Chem-131 Lec-19 09-4 Lipids 1. Lipids:
 (Woods) Chem-131 Lec-19 09-4 Lipids 1 Lipids Classifying Lipids Triacylglycerols (triglycerides): a storage form of energy not required for immediate use. Phospholipids, p sphingolipids, p and cholesterol
(Woods) Chem-131 Lec-19 09-4 Lipids 1 Lipids Classifying Lipids Triacylglycerols (triglycerides): a storage form of energy not required for immediate use. Phospholipids, p sphingolipids, p and cholesterol
Integration of Metabolism
 I. Central Themes of Metabolism 1. ATP is the universal energy carrier. Integration of Metabolism Bryant Miles 2. ATP is generated by the oxidation of metabolic fuels Glucose Fatty Acids Amino Acids 3.
I. Central Themes of Metabolism 1. ATP is the universal energy carrier. Integration of Metabolism Bryant Miles 2. ATP is generated by the oxidation of metabolic fuels Glucose Fatty Acids Amino Acids 3.
The Citric Acid Cycle
 The itric Acid ycle February 14, 2003 Bryant Miles I. itrate Synthase + 3 SoA The first reaction of the citric acid cycle is the condensation of acetyloa and oxaloacetate to form citrate and oas. The enzyme
The itric Acid ycle February 14, 2003 Bryant Miles I. itrate Synthase + 3 SoA The first reaction of the citric acid cycle is the condensation of acetyloa and oxaloacetate to form citrate and oas. The enzyme
Regulation of enzyme activity
 1 Regulation of enzyme activity Regulation of enzyme activity is important to coordinate the different metabolic processes. It is also important for homeostasis i.e. to maintain the internal environment
1 Regulation of enzyme activity Regulation of enzyme activity is important to coordinate the different metabolic processes. It is also important for homeostasis i.e. to maintain the internal environment
Preliminary MFM Quiz
 Preliminary MFM Quiz 1. The major carrier of chemical energy in all cells is: A) adenosine monophosphate B) adenosine diphosphate C) adenosine trisphosphate D) guanosine trisphosphate E) carbamoyl phosphate
Preliminary MFM Quiz 1. The major carrier of chemical energy in all cells is: A) adenosine monophosphate B) adenosine diphosphate C) adenosine trisphosphate D) guanosine trisphosphate E) carbamoyl phosphate
Oxygen Metabolism and Oxygen Toxicity
 xygen Metabolism and xygen Toxicity February 26, 2003 Bryant Miles Chemiosmotic Theory Chemiosomitic Theory state that the free energy of electron transport is coupled to the pumping of protons from the
xygen Metabolism and xygen Toxicity February 26, 2003 Bryant Miles Chemiosmotic Theory Chemiosomitic Theory state that the free energy of electron transport is coupled to the pumping of protons from the
Drug Metabolism. Importance of Drug Metabolism. Importance of Drug Metabolism. Prof. Patrick Davis Basic Medicinal Chemical Principles
 Drug Metabolism Prof. Patrick Davis Basic Medicinal Chemical Principles PR 143M Fall-08 The basic premise: Lipophilic Drugs --> ydrophilic Metabolites (ot Excreted) (Excreted) Water soluble => increased
Drug Metabolism Prof. Patrick Davis Basic Medicinal Chemical Principles PR 143M Fall-08 The basic premise: Lipophilic Drugs --> ydrophilic Metabolites (ot Excreted) (Excreted) Water soluble => increased
Lipid Metabolism. Dr. Howaida Nounou Biochemistry department Sciences college
 Lipid Metabolism Dr. Howaida Nounou Biochemistry department Sciences college Lipids - Heterogenous group of biomolecules - Water insoluble (hydrophobic) - Soluble in organic and non-polar solvents acetone,
Lipid Metabolism Dr. Howaida Nounou Biochemistry department Sciences college Lipids - Heterogenous group of biomolecules - Water insoluble (hydrophobic) - Soluble in organic and non-polar solvents acetone,
Analytical Methods for Quantification of Drug Metabolites in Biological Samples
 Chapter 4 Analytical Methods for Quantification of Drug Metabolites in Biological Samples Robert Roškar and Tina Trdan Lušin Additional information is available at the end of the chapter http://dx.doi.org/10.5772/51676
Chapter 4 Analytical Methods for Quantification of Drug Metabolites in Biological Samples Robert Roškar and Tina Trdan Lušin Additional information is available at the end of the chapter http://dx.doi.org/10.5772/51676
THE LIVING CELL. Cells also have variety of shapes. Plant cells are often rectangular or polygonal, while egg cells are usually spherical.
 THE LIVING CELL A Tour of the cell The cell is the smallest and the basic unit of structure of all organisms. There are two main types or categories of cells: prokaryotic cells and eukaryotic cells. Prokaryotic
THE LIVING CELL A Tour of the cell The cell is the smallest and the basic unit of structure of all organisms. There are two main types or categories of cells: prokaryotic cells and eukaryotic cells. Prokaryotic
1. What has a higher stored energy potential per gram, glycogen or triglycerides? Explain.
 Lipid Metabolism 1. What has a higher stored energy potential per gram, glycogen or triglycerides? Explain. 2. How can excess acetyl CoA trapped in the mitochondria, be utilized as a substrate for fatty
Lipid Metabolism 1. What has a higher stored energy potential per gram, glycogen or triglycerides? Explain. 2. How can excess acetyl CoA trapped in the mitochondria, be utilized as a substrate for fatty
LIVER FUNCTION TESTS
 MODULE Liver Function Tests 17 LIVER FUNCTION TESTS 17.1 INTRODUCTION Liver function tests are a group of tests done to assess the functional capacity of the liver as well as any cellular damage to the
MODULE Liver Function Tests 17 LIVER FUNCTION TESTS 17.1 INTRODUCTION Liver function tests are a group of tests done to assess the functional capacity of the liver as well as any cellular damage to the
THE WHEREWITHAL TO DETOXIFY
 Nutrition Information Services Each Issue available at 2.50 from your distributor By Dr. Lawrence G. Plaskett B.A., Ph.D., C.Chem., F.R.I.C. THE WHEREWITHAL TO DETOXIFY Today those who grasp the principles
Nutrition Information Services Each Issue available at 2.50 from your distributor By Dr. Lawrence G. Plaskett B.A., Ph.D., C.Chem., F.R.I.C. THE WHEREWITHAL TO DETOXIFY Today those who grasp the principles
Lipids. Classifying Lipids
 (Woods) Chem-131 Lec-19 09-4 Lipids 1 Lipids Triacylglycerols (triglycerides): a storage form of energy not required for immediate use. Phospholipids, sphingolipids, and cholesterol (together with proteins)
(Woods) Chem-131 Lec-19 09-4 Lipids 1 Lipids Triacylglycerols (triglycerides): a storage form of energy not required for immediate use. Phospholipids, sphingolipids, and cholesterol (together with proteins)
Keystone Review Practice Test Module A Cells and Cell Processes. 1. Which characteristic is shared by all prokaryotes and eukaryotes?
 Keystone Review Practice Test Module A Cells and Cell Processes 1. Which characteristic is shared by all prokaryotes and eukaryotes? a. Ability to store hereditary information b. Use of organelles to control
Keystone Review Practice Test Module A Cells and Cell Processes 1. Which characteristic is shared by all prokaryotes and eukaryotes? a. Ability to store hereditary information b. Use of organelles to control
Reactions of Fats and Fatty Acids
 Reactions of Fats and Fatty Acids Outline Fats and Oils Fatty Acid Biosynthesis Biodiesel Homework We hear quite a lot about the place of fats and oils in human nutrition. Foods high in fat are at the
Reactions of Fats and Fatty Acids Outline Fats and Oils Fatty Acid Biosynthesis Biodiesel Homework We hear quite a lot about the place of fats and oils in human nutrition. Foods high in fat are at the
IDENTIFICATION OF ALCOHOLS
 IDENTIFICATION OF ALCOHOLS Alcohols are organic compounds that which considered as derivatives of water. One of the hydrogen atoms of water molecule (H-O-H) has been replaced by an alkyl or substituted
IDENTIFICATION OF ALCOHOLS Alcohols are organic compounds that which considered as derivatives of water. One of the hydrogen atoms of water molecule (H-O-H) has been replaced by an alkyl or substituted
Chem 306 Chapter 21 Bioenergetics Lecture Outline III
 Chem 306 Chapter 21 Bioenergetics Lecture Outline III I. HOW IS ATP GENERATED IN THE FINAL STAGE CATABOLISM? A. OVERVIEW 1. At the end of the citric acid cycle, all six carbons of glucose have been oxidized
Chem 306 Chapter 21 Bioenergetics Lecture Outline III I. HOW IS ATP GENERATED IN THE FINAL STAGE CATABOLISM? A. OVERVIEW 1. At the end of the citric acid cycle, all six carbons of glucose have been oxidized
for excitation to occur, there must be an exact match between the frequency of the applied radiation and the frequency of the vibration
 ! = 1 2"c k (m + M) m M wavenumbers! =!/c = 1/" wavelength frequency! units: cm 1 for excitation to occur, there must be an exact match between the frequency of the applied radiation and the frequency
! = 1 2"c k (m + M) m M wavenumbers! =!/c = 1/" wavelength frequency! units: cm 1 for excitation to occur, there must be an exact match between the frequency of the applied radiation and the frequency
AP BIOLOGY CHAPTER 7 Cellular Respiration Outline
 AP BIOLOGY CHAPTER 7 Cellular Respiration Outline I. How cells get energy. A. Cellular Respiration 1. Cellular respiration includes the various metabolic pathways that break down carbohydrates and other
AP BIOLOGY CHAPTER 7 Cellular Respiration Outline I. How cells get energy. A. Cellular Respiration 1. Cellular respiration includes the various metabolic pathways that break down carbohydrates and other
Waxes. From the head of sperm whales Structural material of beehives Coating on the leaves of Brazilian palm. Fats and Oils
 Lipids Lipids are organic compounds that contain hydrocarbons which are the foundation for the structure and function of living cells. Lipids are non polar so they are soluble in nonpolar environments
Lipids Lipids are organic compounds that contain hydrocarbons which are the foundation for the structure and function of living cells. Lipids are non polar so they are soluble in nonpolar environments
Drug Excretion. Renal Drug Clearance. Drug Clearance and Half-Life. Glomerular Filtration II. Glomerular Filtration I. Drug Excretion and Clearance
 t/.drugexcretion AINTRAVENOUSDOSE 36848765430TIME(hours) t/ Drug Excretion Dr. Robert G. Lamb Professor Pharmacology & Toxicology Drug Excretion and Clearance Drug Excretion: is the movement of drug from
t/.drugexcretion AINTRAVENOUSDOSE 36848765430TIME(hours) t/ Drug Excretion Dr. Robert G. Lamb Professor Pharmacology & Toxicology Drug Excretion and Clearance Drug Excretion: is the movement of drug from
Guidance for Industry Safety Testing of Drug Metabolites
 Guidance for Industry Safety Testing of Drug Metabolites U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research (CDER) February 2008 Pharmacology
Guidance for Industry Safety Testing of Drug Metabolites U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research (CDER) February 2008 Pharmacology
The Electron Transport Chain
 The Electron Transport hain February 19, 2003 Bryant Miles The citric acid cycle oxidizes acetate into two molecules of 2 while capturing the electrons in the form of 3 NAD molecules and one molecule of
The Electron Transport hain February 19, 2003 Bryant Miles The citric acid cycle oxidizes acetate into two molecules of 2 while capturing the electrons in the form of 3 NAD molecules and one molecule of
Fatty Acids carboxylic acids
 Triglycerides (TG) should actually be called triacylglycerols (TAG). TG or TAG are molecules with a glycerol (a carbohydrate) backbone to which are attached three acyl groups. They represent a concentrated
Triglycerides (TG) should actually be called triacylglycerols (TAG). TG or TAG are molecules with a glycerol (a carbohydrate) backbone to which are attached three acyl groups. They represent a concentrated
LAB 3: DIGESTION OF ORGANIC MACROMOLECULES
 LAB 3: DIGESTION OF ORGANIC MACROMOLECULES INTRODUCTION Enzymes are a special class of proteins that lower the activation energy of biological reactions. These biological catalysts change the rate of chemical
LAB 3: DIGESTION OF ORGANIC MACROMOLECULES INTRODUCTION Enzymes are a special class of proteins that lower the activation energy of biological reactions. These biological catalysts change the rate of chemical
Regulation of cholesterol synthesis pathway. Cholesterol Synthesis. Cholesterol biosynthesis. Regulation of HMG CoA reductase activity
 Cholesterol Synthesis Main function: ation of steroid nucleus Substrate: cytoplasmic acetyl CoA Endproduct: cholesterol Location: cytoplasm Regulation of cholesterol synthesis pathway HMG CoA reductase
Cholesterol Synthesis Main function: ation of steroid nucleus Substrate: cytoplasmic acetyl CoA Endproduct: cholesterol Location: cytoplasm Regulation of cholesterol synthesis pathway HMG CoA reductase
How To Understand The Human Body
 Introduction to Biology and Chemistry Outline I. Introduction to biology A. Definition of biology - Biology is the study of life. B. Characteristics of Life 1. Form and size are characteristic. e.g. A
Introduction to Biology and Chemistry Outline I. Introduction to biology A. Definition of biology - Biology is the study of life. B. Characteristics of Life 1. Form and size are characteristic. e.g. A
Chemical Bonds and Groups - Part 1
 hemical Bonds and Groups - Part 1 ARB SKELETS arbon has a unique role in the cell because of its ability to form strong covalent bonds with other carbon atoms. Thus carbon atoms can join to form chains.
hemical Bonds and Groups - Part 1 ARB SKELETS arbon has a unique role in the cell because of its ability to form strong covalent bonds with other carbon atoms. Thus carbon atoms can join to form chains.
Chapter 22 Carbonyl Alpha-Substitution Reactions
 John E. McMurry www.cengage.com/chemistry/mcmurry Chapter 22 Carbonyl Alpha-Substitution Reactions The α Position The carbon next to the carbonyl group is designated as being in the α position Electrophilic
John E. McMurry www.cengage.com/chemistry/mcmurry Chapter 22 Carbonyl Alpha-Substitution Reactions The α Position The carbon next to the carbonyl group is designated as being in the α position Electrophilic
Antioxidant Products. A comprehensive range of. Antioxidants
 Antioxidant Products A comprehensive range of ALB BIL FERR GR Ransel Ransod antioxidant TAS testing TIBC kits TF BIL FERR GR Ransel Ransod TAS TIBC TF UA A FERR GR Ransel Ransod TAS TIBC TF UA ALB GR Ransel
Antioxidant Products A comprehensive range of ALB BIL FERR GR Ransel Ransod antioxidant TAS testing TIBC kits TF BIL FERR GR Ransel Ransod TAS TIBC TF UA A FERR GR Ransel Ransod TAS TIBC TF UA ALB GR Ransel
INTRODUCTION TO HORMONES
 INTRODUCTION TO HORMONES UNIVERSITY OF PNG SCHOOL OF MEDICINE AND HEALTH SCIENCES DISCIPLINE OF BIOCHEMISTRY & MOLECULAR BIOLOGY PBL MBBS II SEMINAR VJ Temple What are hormones? Cells in multi-cellular
INTRODUCTION TO HORMONES UNIVERSITY OF PNG SCHOOL OF MEDICINE AND HEALTH SCIENCES DISCIPLINE OF BIOCHEMISTRY & MOLECULAR BIOLOGY PBL MBBS II SEMINAR VJ Temple What are hormones? Cells in multi-cellular
Biological molecules:
 Biological molecules: All are organic (based on carbon). Monomers vs. polymers: Monomers refer to the subunits that, when polymerized, make up a larger polymer. Monomers may function on their own in some
Biological molecules: All are organic (based on carbon). Monomers vs. polymers: Monomers refer to the subunits that, when polymerized, make up a larger polymer. Monomers may function on their own in some
Cell Membrane Structure (and How to Get Through One)
 Cell Membrane Structure (and How to Get Through One) A cell s membrane is a wall of sorts that defines the boundaries of a cell. The membrane provides protection and structure for the cell and acts as
Cell Membrane Structure (and How to Get Through One) A cell s membrane is a wall of sorts that defines the boundaries of a cell. The membrane provides protection and structure for the cell and acts as
ENZYME SCIENCE AND ENGINEERING PROF. SUBHASH CHAND DEPARTMENT OF BIOCHEMICAL ENGINEERING AND BIOTECHNOLOGY IIT DELHI LECTURE 4 ENZYMATIC CATALYSIS
 ENZYME SCIENCE AND ENGINEERING PROF. SUBHASH CHAND DEPARTMENT OF BIOCHEMICAL ENGINEERING AND BIOTECHNOLOGY IIT DELHI LECTURE 4 ENZYMATIC CATALYSIS We will continue today our discussion on enzymatic catalysis
ENZYME SCIENCE AND ENGINEERING PROF. SUBHASH CHAND DEPARTMENT OF BIOCHEMICAL ENGINEERING AND BIOTECHNOLOGY IIT DELHI LECTURE 4 ENZYMATIC CATALYSIS We will continue today our discussion on enzymatic catalysis
Energy Production In A Cell (Chapter 25 Metabolism)
 Energy Production In A Cell (Chapter 25 Metabolism) Large food molecules contain a lot of potential energy in the form of chemical bonds but it requires a lot of work to liberate the energy. Cells need
Energy Production In A Cell (Chapter 25 Metabolism) Large food molecules contain a lot of potential energy in the form of chemical bonds but it requires a lot of work to liberate the energy. Cells need
How to create and interpret the predictive analysis of a compound
 How to create and interpret the predictive analysis of a compound Platform with suite of tools Predict & understand biological effects of small molecules & compounds Predict targets and metabolites, potential
How to create and interpret the predictive analysis of a compound Platform with suite of tools Predict & understand biological effects of small molecules & compounds Predict targets and metabolites, potential
Chapter 8: Energy and Metabolism
 Chapter 8: Energy and Metabolism 1. Discuss energy conversions and the 1 st and 2 nd law of thermodynamics. Be sure to use the terms work, potential energy, kinetic energy, and entropy. 2. What are Joules
Chapter 8: Energy and Metabolism 1. Discuss energy conversions and the 1 st and 2 nd law of thermodynamics. Be sure to use the terms work, potential energy, kinetic energy, and entropy. 2. What are Joules
AMINO ACIDS & PEPTIDE BONDS STRUCTURE, CLASSIFICATION & METABOLISM
 AMINO ACIDS & PEPTIDE BONDS STRUCTURE, CLASSIFICATION & METABOLISM OBJECTIVES At the end of this session the student should be able to, recognize the structures of the protein amino acid and state their
AMINO ACIDS & PEPTIDE BONDS STRUCTURE, CLASSIFICATION & METABOLISM OBJECTIVES At the end of this session the student should be able to, recognize the structures of the protein amino acid and state their
What Do We Learn about Hepatotoxicity Using Coumarin-Treated Rat Model?
 What Do We Learn about Hepatotoxicity Using Coumarin-Treated Rat Model? authors M. David Ho 1, Bob Xiong 1, S. Stellar 2, J. Proctor 2, J. Silva 2, H.K. Lim 2, Patrick Bennett 1, and Lily Li 1 Tandem Labs,
What Do We Learn about Hepatotoxicity Using Coumarin-Treated Rat Model? authors M. David Ho 1, Bob Xiong 1, S. Stellar 2, J. Proctor 2, J. Silva 2, H.K. Lim 2, Patrick Bennett 1, and Lily Li 1 Tandem Labs,
The diagram below summarizes the effects of the compounds that cells use to regulate their own metabolism.
 Regulation of carbohydrate metabolism Intracellular metabolic regulators Each of the control point steps in the carbohydrate metabolic pathways in effect regulates itself by responding to molecules that
Regulation of carbohydrate metabolism Intracellular metabolic regulators Each of the control point steps in the carbohydrate metabolic pathways in effect regulates itself by responding to molecules that
Carl Rosow, M.D., Ph.D. 1 HST-151. Lecture 1 - Principles of Pharmacology: Introduction
 Harvard-MIT Division of Health Sciences and Technology HST.151: Principles of Pharmocology Instructor: Dr. Carl Rosow Carl Rosow, M.D., Ph.D. 1 HST-151 Lecture 1 - Principles of Pharmacology: Introduction
Harvard-MIT Division of Health Sciences and Technology HST.151: Principles of Pharmocology Instructor: Dr. Carl Rosow Carl Rosow, M.D., Ph.D. 1 HST-151 Lecture 1 - Principles of Pharmacology: Introduction
Structure of proteins
 Structure of proteins Primary structure: is amino acids sequence or the covalent structure (50-2500) amino acids M.Wt. of amino acid=110 Dalton (56 110=5610 Dalton). Single chain or more than one polypeptide
Structure of proteins Primary structure: is amino acids sequence or the covalent structure (50-2500) amino acids M.Wt. of amino acid=110 Dalton (56 110=5610 Dalton). Single chain or more than one polypeptide
Chemistry 20 Chapters 15 Enzymes
 Chemistry 20 Chapters 15 Enzymes Enzymes: as a catalyst, an enzyme increases the rate of a reaction by changing the way a reaction takes place, but is itself not changed at the end of the reaction. An
Chemistry 20 Chapters 15 Enzymes Enzymes: as a catalyst, an enzyme increases the rate of a reaction by changing the way a reaction takes place, but is itself not changed at the end of the reaction. An
Safe Nano Design Molecule Manufacturing Market
 August 14 16, 212 College of Nanoscale Science & Engineering (CNSE) of the University at Albany National Institute for Occupational Safety and Health (NIOSH) Prevention through Design Program Safe Nano
August 14 16, 212 College of Nanoscale Science & Engineering (CNSE) of the University at Albany National Institute for Occupational Safety and Health (NIOSH) Prevention through Design Program Safe Nano
William Shaw, Ph.D. The Great Plains Laboratory, Inc., Lenexa, Kansas, USA
 Inhibition of dopamine conversion to norepinephrine by Clostridia metabolites appears to be a (the) major cause of autism, schizophrenia, and other neuropsychiatric disorders. All these factors can now
Inhibition of dopamine conversion to norepinephrine by Clostridia metabolites appears to be a (the) major cause of autism, schizophrenia, and other neuropsychiatric disorders. All these factors can now
Amino Acid Metabolism (Chapter 20) Lecture 8:
 Amino Acid Metabolism (Chapter 20) Lecture 8: Nitrogen Fixation (20.7); Nitrite Assimilation (not in text?); Protein Digestion in the Gut (5.3b, 11.5, 20.2); Amino Acid Degradation in Cells (20.2); Next:
Amino Acid Metabolism (Chapter 20) Lecture 8: Nitrogen Fixation (20.7); Nitrite Assimilation (not in text?); Protein Digestion in the Gut (5.3b, 11.5, 20.2); Amino Acid Degradation in Cells (20.2); Next:
Pharmacokinetics CHAPTER 2 OVERVIEW DRUG ABSORPTION. Effect of ph on Absorption of Weak Acids and Bases. Processes of Absorption
 CHAPTE 2 Pharmacokinetics VEVIEW Pharmacokinetics is the study of drug disposition in the body and focuses on the changes in drug plasma concentration. For any given drug and dose, the plasma concentration
CHAPTE 2 Pharmacokinetics VEVIEW Pharmacokinetics is the study of drug disposition in the body and focuses on the changes in drug plasma concentration. For any given drug and dose, the plasma concentration
Glutathione The Miracle Molecule for Longevity By Dr. Sherrill Sellman 2009
 Glutathione The Miracle Molecule for Longevity By Dr. Sherrill Sellman 2009 Recent statistics have good news for us. Centenarians are the fastest growing age group in America. What s the secret to a long
Glutathione The Miracle Molecule for Longevity By Dr. Sherrill Sellman 2009 Recent statistics have good news for us. Centenarians are the fastest growing age group in America. What s the secret to a long
I N V E S T I C E D O R O Z V O J E V Z D Ě L Á V Á N Í ENZYMES
 = substances that... biological reactions 1. Provide an alternative reaction route which has a lower... energy 2. Reactions catalysed by enzymes occur under mild conditions + good yield + fast 3. Enzymes
= substances that... biological reactions 1. Provide an alternative reaction route which has a lower... energy 2. Reactions catalysed by enzymes occur under mild conditions + good yield + fast 3. Enzymes
MULTIPLE CHOICE QUESTIONS
 MULTIPLE CHOICE QUESTIONS 1. Most components of energy conversion systems evolved very early; thus, the most fundamental aspects of energy metabolism tend to be: A. quite different among a diverse group
MULTIPLE CHOICE QUESTIONS 1. Most components of energy conversion systems evolved very early; thus, the most fundamental aspects of energy metabolism tend to be: A. quite different among a diverse group
Enzymes. Enzyme Structure. Enzyme Classification. CHEM464/Medh, J.D. Reaction Rate and Enzyme Activity
 Enzymes Enzymes are biological catalysts They are not consumed or altered during the reaction They do not change the equilibrium, just reduce the time required to reach equilibrium. They increase the rate
Enzymes Enzymes are biological catalysts They are not consumed or altered during the reaction They do not change the equilibrium, just reduce the time required to reach equilibrium. They increase the rate
PHC 313 The 7 th. Lecture. Adrenergic Agents
 PHC 313 The 7 th. Lecture Adrenergic Agents Introduction Introduction Adrenergic agents are a broad class of agents employed in the treatment of many disorders. They are those chemical agents that exert
PHC 313 The 7 th. Lecture Adrenergic Agents Introduction Introduction Adrenergic agents are a broad class of agents employed in the treatment of many disorders. They are those chemical agents that exert
Detoxification & Your Health
 Detoxification & Your Health Detoxification is the metabolic process your body uses to transform and eliminate toxins. The process can occur in two steps, called Phase I and Phase II. Phase I is our first
Detoxification & Your Health Detoxification is the metabolic process your body uses to transform and eliminate toxins. The process can occur in two steps, called Phase I and Phase II. Phase I is our first
Cell Structure & Function!
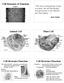 Cell Structure & Function! Chapter 3! The most exciting phrase to hear in science, the one that heralds new discoveries, is not 'Eureka!' but 'That's funny.! -- Isaac Asimov Animal Cell Plant Cell Cell
Cell Structure & Function! Chapter 3! The most exciting phrase to hear in science, the one that heralds new discoveries, is not 'Eureka!' but 'That's funny.! -- Isaac Asimov Animal Cell Plant Cell Cell
General Protein Metabolism
 General Protein Metabolism Protein Digestion Dietary proteins are very large complex molecules that cannot be absorbed from the intestine. To be absorbed, dietary proteins must be digested to small simple
General Protein Metabolism Protein Digestion Dietary proteins are very large complex molecules that cannot be absorbed from the intestine. To be absorbed, dietary proteins must be digested to small simple
7 Answers to end-of-chapter questions
 7 Answers to end-of-chapter questions Multiple choice questions 1 B 2 B 3 A 4 B 5 A 6 D 7 C 8 C 9 B 10 B Structured questions 11 a i Maintenance of a constant internal environment within set limits i Concentration
7 Answers to end-of-chapter questions Multiple choice questions 1 B 2 B 3 A 4 B 5 A 6 D 7 C 8 C 9 B 10 B Structured questions 11 a i Maintenance of a constant internal environment within set limits i Concentration
Chapter 5 Classification of Organic Compounds by Solubility
 Chapter 5 Classification of Organic Compounds by Solubility Deductions based upon interpretation of simple solubility tests can be extremely useful in organic structure determination. Both solubility and
Chapter 5 Classification of Organic Compounds by Solubility Deductions based upon interpretation of simple solubility tests can be extremely useful in organic structure determination. Both solubility and
Name Lab #3: Solubility of Organic Compounds Objectives: Introduction: soluble insoluble partially soluble miscible immiscible
 Lab #3: Solubility of rganic Compounds bjectives: - Understanding the relative solubility of organic compounds in various solvents. - Exploration of the effect of polar groups on a nonpolar hydrocarbon
Lab #3: Solubility of rganic Compounds bjectives: - Understanding the relative solubility of organic compounds in various solvents. - Exploration of the effect of polar groups on a nonpolar hydrocarbon
HOMEWORK PROBLEMS: IR SPECTROSCOPY AND 13C NMR. The peak at 1720 indicates a C=O bond (carbonyl). One possibility is acetone:
 HMEWRK PRBLEMS: IR SPECTRSCPY AND 13C NMR 1. You find a bottle on the shelf only labeled C 3 H 6. You take an IR spectrum of the compound and find major peaks at 2950, 1720, and 1400 cm -1. Draw a molecule
HMEWRK PRBLEMS: IR SPECTRSCPY AND 13C NMR 1. You find a bottle on the shelf only labeled C 3 H 6. You take an IR spectrum of the compound and find major peaks at 2950, 1720, and 1400 cm -1. Draw a molecule
Oxidative Phosphorylation
 Oxidative Phosphorylation NADH from Glycolysis must be transported into the mitochondrion to be oxidized by the respiratory electron transport chain. Only the electrons from NADH are transported, these
Oxidative Phosphorylation NADH from Glycolysis must be transported into the mitochondrion to be oxidized by the respiratory electron transport chain. Only the electrons from NADH are transported, these
Endocrine System: Practice Questions #1
 Endocrine System: Practice Questions #1 1. Removing part of gland D would most likely result in A. a decrease in the secretions of other glands B. a decrease in the blood calcium level C. an increase in
Endocrine System: Practice Questions #1 1. Removing part of gland D would most likely result in A. a decrease in the secretions of other glands B. a decrease in the blood calcium level C. an increase in
1. The diagram below represents a biological process
 1. The diagram below represents a biological process 5. The chart below indicates the elements contained in four different molecules and the number of atoms of each element in those molecules. Which set
1. The diagram below represents a biological process 5. The chart below indicates the elements contained in four different molecules and the number of atoms of each element in those molecules. Which set
Ch18_PT MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question.
 Ch18_PT MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) All of the following can be classified as biomolecules except A) lipids. B) proteins. C)
Ch18_PT MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) All of the following can be classified as biomolecules except A) lipids. B) proteins. C)
Anti-Atheroscrerotic Drugs
 Anti-Atheroscrerotic Drugs Masuko Ushio-Fukai, PhD, FAHA Dept. of Pharmacology University of Illinois at Chicago Anti-Atherogenic Drugs: Treatment of Hyperlipidemias Knowledge Objectives: 1) Know the mechanism
Anti-Atheroscrerotic Drugs Masuko Ushio-Fukai, PhD, FAHA Dept. of Pharmacology University of Illinois at Chicago Anti-Atherogenic Drugs: Treatment of Hyperlipidemias Knowledge Objectives: 1) Know the mechanism
Compartmentalization of the Cell. Objectives. Recommended Reading. Professor Alfred Cuschieri. Department of Anatomy University of Malta
 Compartmentalization of the Cell Professor Alfred Cuschieri Department of Anatomy University of Malta Objectives By the end of this session the student should be able to: 1. Identify the different organelles
Compartmentalization of the Cell Professor Alfred Cuschieri Department of Anatomy University of Malta Objectives By the end of this session the student should be able to: 1. Identify the different organelles
What affects an enzyme s activity? General environmental factors, such as temperature and ph. Chemicals that specifically influence the enzyme.
 CH s 8-9 Respiration & Metabolism Metabolism A catalyst is a chemical agent that speeds up a reaction without being consumed by the reaction. An enzyme is a catalytic protein. Hydrolysis of sucrose by
CH s 8-9 Respiration & Metabolism Metabolism A catalyst is a chemical agent that speeds up a reaction without being consumed by the reaction. An enzyme is a catalytic protein. Hydrolysis of sucrose by
