Oral riboflavin versus oral propranolol in migraine prophylaxis: An open label randomized controlled trial
|
|
|
- Winfred Copeland
- 7 years ago
- Views:
Transcription
1 Neurology Asia 2011; 16(3) : Oral riboflavin versus oral propranolol in migraine prophylaxis: An open label randomized controlled trial 1 *Natasha J Nambiar MBBS MD, 1 *Cuckoo Aiyappa PhD, 2 R Srinivasa MD DM 1 Department of pharmacology and 2 Department of Neurology, MS Ramaiah Medical College Bangalore, India *These authors contributed equally to this work Abstract Background: Migraine is a chronic, often debilitating disease. The treatment of migraine with propranolol ( mg/day) is limited by side effects and lack of tolerability. Riboflavin (vitamin B2) is the precursor of flavin mononucleotide and flavin adenine dinucleotide which are involved in mitochondrial transport chain. The use of riboflavin in migraine prophylaxis is based on the hypothesis of a deficient mitochondrial energy reserve as a causal factor in migraine pathogenesis, and on the findings of its safety and effectiveness at high doses (400 mg/day) in the treatment of migraine like headaches in classic mitochondriopathies. Objectives: To compare the efficacy of lower dose oral riboflavin at 100 mg/day with oral propranolol 80 mg/day in the reduction of migraine frequency and severity. Methods: One hundred patients diagnosed with migraine were randomized to receive either oral riboflavin 100 mg/day or propranolol 80 mg/day for a period of 3 months. Patients were issued a migraine diary and explained how to record the frequency of migraine attacks, headache intensity, duration and to report any side effects. Follow-up was at the end of 3 and 6 months. Results: Both study groups showed a reduction of migraine frequency, duration and severity of headache by visual analogue scale (VAS), and disability by migraine disability questionnaire (MIDAS) score at 3 months. No significant difference was seen between the two study groups in most of the measures. Side effects were significantly less in the riboflavin group (P=0.035). Conclusion: Oral riboflavin 100mg/day is a safe, equally effective and well tolerated alternative in migraine prophylaxis. INTRODUCTION Migraine is a chronic, often debilitating disease that affects 12% of the general population 1, 18% of women and 6% of men. 2 WHO cited migraine as the 19 th leading cause of years lived with disability among both males and females of all ages combined. 3 The high prevalence, the marked impairment of the sufferer s quality of life culminate in a major societal burden with high medical bills and indirect costs such as unemployment and work productivity loss. 4 The goal of preventive treatment should be to prevent or reduce the frequency of migraine attacks, to improve response to acute medications, to improve patient function and to reduce disability. 5 To maximize compliance, prophylaxis treatment should be safe, well tolerated, and inexpensive. The theoretical basis for using riboflavin is its ameliorating effect on the mitochondrial dysfunction that might be involved in the pathophysiology of migraine. A previous study of 49 patients treated with 400 mg daily of riboflavin showed a mean global improvement of 68.2%. 6 The dose of 400 mg was chosen in the previous trials because comparable doses were previously used to treat mitochondriopathies, and the lack of toxicity. Because of its high efficacy, excellent tolerability, and low cost, riboflavin is an interesting option for migraine prophylaxis, and a candidate for a comparative trial with an established prophylactic drug. Since the use of riboflavin as a migraine preventive medication is supported by a few clinical trials which have shown its safety and efficacy, the present study intends to explore the use of the drug in comparison with propanolol, a gold standard prophylactic drug. There is no evidence that riboflavin interacts with abortive medication, as do many other prophylactic agents. 7 It may also be worthwhile for cost reasons to explore the antimigraine effect of lower doses of riboflavin. Furthermore, no such studies have been done Address correspondence to: Dr.Natasha JB Nambiar, Department of pharmacology, M.S. Ramaiah Medical College, Bangalore , India. dr.natasha7@gmail.com 223
2 Neurology Asia September 2011 in the Indian population. Therefore, this study is undertaken to investigate the effectiveness of riboflavin in a lower dose of 100mg, and to compare its role as prophylactic agent with propranolol in migraine patients. METHODS The study was conducted for a period of one year starting from November 2008 to October 2009 on patients diagnosed with migraine attending Neurology outpatient department at M.S. Ramaiah Medical Teaching Hospital and in M.S. Ramaiah Memorial Hospital, Bangalore. Consent was taken from the subjects before their inclusion in the study. The ethical clearance was obtained from the Institutional Ethical Review Board of the Institute. The inclusion criteria were: (1). History of migraine of more than one year diagnosed according to IHS criteria; (2). Had 2-8 headache attacks per month; (3). Age between 18 to 65 years. The exclusion criteria were: (1). Use of prophylactic medication 3 months prior to the study; (2). Tension type headache more than five days per month; (3). Pregnancy or lactation; (4). Anemia; (5). Use of vitamin supplements; (6). Alcohol abuse; (7). Abnormal liver or renal function tests; (8). Patients diagnosed to have transformed migraine, chronic daily headache, cluster headache or cluster migraine; (9). Any significant cardiovascular, central nervous system, renal or respiratory dysfunction; (10). Patient who refused consent. Prior to randomization patients were counseled regarding migraine preventive therapy and the need for taking the treatment for at least 3 months. Patients were randomly assigned using computerized randomization table to either of the two groups: Group 1: Tablet propranolol 80mg once daily; Group 2: Tablet riboflavin 100mg once daily. All patients were issued a migraine diary and explained how to record the number and duration of attacks, severity of migraine headaches according to the visual analogue scale (VAS) and the disability using the migraine disability questionnaire (MIDAS). The patients were asked to come for follow-up at the end of first, third and sixth month. All patients were reminded of the follow-up through telephone. During the followup migraine diaries were cross-checked with compliance to the drug. Patients were allowed to use concomitant rescue medication to abort migraine attacks. Statistical methods Descriptive statistical analysis was carried out in the present study. Results on continuous measurements are presented as Mean ± SD (Min- Max) and results on categorical measurements are presented in Number (%). Significance is assessed at 5% level of significance. 2x3 Repeated measures Analysis of variance (RANOVA) has been used to find the significance of study parameters between three or more groups of patients, Student t test (two tailed, independent) has been used to find the significance of study parameters on continuous scale between two groups (Inter group analysis) Chi-square/ Fisher Exact test has been used to find the significance of study parameters on categorical scale between two or more groups. The Statistical software SAS 9.0, SPSS 15.0, Stata 8.0, MedCalc and Systat 11.0 were used for the analysis of the data. RESULTS Patients in both the study arms are age and sex matched. The mean age was 31 years with higher preponderance of females. Type of migraine was similar between the two groups with majority having migraine without aura. A positive family history was slightly higher in the riboflavin group. Baseline frequency and duration of headache, VAS and MIDAS scores were also similar in both study groups (Table 1). Both study groups showed reducetion of frequency of headache at the end of 3 months, and at sixth month follow up. There was a significant reduction in the frequency of migraine headaches in the propanolol relative to the riboflavin group at the end of the first month, but there was no difference in the two groups at the end of the third month and sixth month follow-up (Table 2). As for the duration of migraine headache, both groups showed reduction in the duration of headache at the third month. There was no significant difference between the two study groups at the end of three months of treatment, and follow-up at sixth month (Table 3). As for severity of the headache as measured by the VAS score, both groups showed reduction in the VAS scores at 3 months and at the sixth month follow-up. There was a also significantly lower VAS score in the riboflavin as compared to the propranolol group in the third month of treatment. (Table 4). As for disability as measured by the MIDAS score, both groups showed reduction at 3 months, but there was no significant difference between the two groups (Table 5). As for side effects, overall 224
3 Table 1: Baseline demographic and clinical characteristics of study patients Characteristic Propranolol 80mg/day Riboflavin100mg/day P value (n =50) (n =50) Age 31.5± ± Female gender n (%) 27(54%) 28(56%) Type of migraine n (%) With aura 16(32%) 15(30%) Without aura 34(68%) 35(70%) Duration of migraine in years 6.7± ± Positive family history 11(22%) 15(30%) Frequency of headaches/month 4.0± ± Duration of attack in hours 2.9± ± VAS at baseline (0 month) 5.3± ± MIDAS at baseline (0 month) 4.7± ± VAS: visual analogue score MIDAS: migraine disability questionnaire Table 2: The frequency of headache per month in the study period Frequency of headache Propranolol Riboflavin P value Baseline 4.0± ±1.0 NS 1 st month 3.3± ±1.0 < nd month 2.8± ±0.7 NS 3 rd month 2.3± ±0.7 NS 6 th month follow up 2.8± ±0.7 NS % change at 3 rd month 42.1% 43.2% - % change at 6 th month follow up 30.2% 28.1% - Repeated measures ANOVA Model up to 3 rd month evaluation Group* F=0.862; P=0.355 F=258.54; P<0.001 F=11.385; P<
4 Neurology Asia September 2011 Table 3: The duration of headache in hour in the study period Duration of headache Propranolol Riboflavin P value Baseline 2.9± ±1.0 NS 1 st month 2.9± ±1.0 NS 2 nd month 2.4± ±0.6 NS 3 rd month 2.0± ±0.7 NS 6 th month follow up 2.9± ±0.8 NS % change at 3 rd month 32.2% 26.2% - % change at 6 th month follow up 2.05% 9.2% - Repeated measures ANOVA Model up to 3 rd month evaluation Group F=0.009; P=0.925 F= ; P<0.001 F=1.630; P=0.182 Table 4: The severity of headache by visual analogue score (VAS) in the study period VAS score Propranolol Riboflavin P value Baseline 5.3± ±1.1 NS 1 st month 5.3± ±1.1 NS 2 nd month 4.4± ±0.9 NS 3 rd month 4.0± ± th month follow up 4.4± ±0.9 NS % change at 3 rd month 25.8% 27.1% - % change at 6 th month follow up 17.2% 13.1% - Repeated measures ANOVA Model up to 3 rd month evaluation Group F=3.278; P=0.073 F= ; P<0.001 F=1.428; P=
5 Table 5: The disability of headache by migraine disability questionnaire (MIDAS) score in the study period MIDAS score Propranolol Riboflavin P value Baseline 4.7± ±1.5 NS 3 rd month 3.8± ±1.4 NS % change at 3 rd month 18.6% 21.3% - Repeated measures ANOVA Model up to 3 rd month evaluation Group F=0.001; P=0.972 F= ; P<0.001 F=1.136; P=0.289 side effects were significantly less in the riboflavin as compared to the propranolol group. Patients on riboflavin treatment complained of orange discoloration of urine, nausea and diarrhea while in the propranolol group the common side effects reported were fatigue, dizziness and nausea (Table 6). DISCUSSION Riboflavin (vitamin B2) is the precursor of flavin mononucleotide and flavin adenine dinucleotide. These are coenzymes required for the activity of flavoenzymes involved in the transfer of electrons in oxidation-reduction reactions and Table 6: Side effects of the study patients Side effects Propranolol 80mg/day Riboflavin100mg/day (n=50) (n=50) No % No % Absent Present Fatigue Dizziness Orange discoloration of urine Vomiting Nausea Diarrhoea Side effects are significantly less in Group 2 with P=
6 Neurology Asia September 2011 in the production of energy in the mitochondria. A beneficial clinical response to riboflavin has been observed in patients with syndrome of mitochondrial encephalomyopathy, lactic acidosis and stroke-like episodes (MELAS), who experience headaches similar to headaches in migraine. 8,9 The present study suggests that oral riboflavin in the dose 100 mg/day is comparable with oral propranolol 80 mg/day in reducing the frequency of migraine headaches at the end of 3 months, and at sixth month follow up.,this study is in accordance with a non randomized trial by Sandor et al in which 26 patients with migraine received 4 months migraine prophylaxis with either riboflavin or beta blockers. Clinical improvement was seen with both treatments with patients in the riboflavin group demonstrating a reduction from 3.5 to 1.7 attacks/month. Based on auditory evoked cortical potential responses before and after treatment, the authors concluded that β-blockers and riboflavin act through different mechanisms. 10 Though both study drugs reduced the migraine headache frequency, our study showed that the propranolol group had a faster effect, with significantly greater reduction in the headache frequency by the end of the first month. In an open pilot study conducted by Schoenen et al, riboflavin at 400mg/day was superior to placebo in reducing headache attack frequency. The effect of riboflavin was found to set in after 1 month and was maximal after 3 months of treatment. 11,12 In another open label study by Boehnke et al, riboflavin 400mg/day significantly reduced the headache frequency from 4 to 2 days/month after 3 and 6 months. 13 The lag before reduction in headache frequency in the riboflavin group may be because impaired mitochondrial energy metabolism is likely to be only one functional abnormality that predisposes to migraine, and may not be crucial in all patients. It is also conceivable that a clinical effect due to pharmacologic intervention on mitochondrial metabolism builds up more slowly than one brought about by receptor blockade. The efficacy of riboflavin beyond 3 months of treatment is yet to be determined. Both of our study group patients showed reduction of duration of headache at 3 months, although there was no no significant difference in the reduction between the two groups at the end of 3 months and the sixth month follow-up. In the randomized double blind placebo controlled trial conducted by Schoenen et al, there was significant reduction of the migraine headache in the 28 patients on riboflavin 400 mg/day. 11 Both of our study group patients also showed reduction in severity of headache as measured by the VAS scores at 3 months and at the sixth month follow-up. There was a also significantly lower VAS score in the riboflavin as compared to the propranolol group in the third month of treatment. The percentage reduction of VAS scores at the end of three months and at sixth month follow-up was however similar in both study groups. Schoenen et al also found a significant reduction in migraine severity in the riboflavin arm as assessed by a four point scale in their placebo control study. 11 Boenhke et al on the other hand, using riboflavin 400 mg/day showed no effect on the migraine severity. 13 Although there are no human studies, animal studies conducted by Franca et al demonstrated a dose dependent analgesic effect of riboflavin against formaldehyde induced nociception. A similar animal study conducted by Granados-Soto et al also demonstrated the ability of riboflavin to produce antinociception and anti-inflammatory effect. This was thought due to the activation of K + channels or nitric oxide release, but not activation of opioid mechanisms. 14,15 The above observations may explain the effect of riboflavin on migraine severity. We have shown that both study groups showed reduction at 3 months of disability as measured by the MIDAS score, but there was no significant difference between the two groups So far no studies using riboflavin have assessed this parameter. The side effects in the present study were significantly less in the riboflavin group with orange discoloration of urine, vomiting and diarrhoea being the most reported. Only 24 % complained of adverse events in the riboflavin group as compared to 44% in the propranolol group. The present study is in accordance with the trial by Schoenen et al 11 and Boehnke et al, the later using higher dose of riboflavin 400mg/ day, with mild adverse effects such as diarrhoea, upper abdominal pain and facial erythema. 13 This was a prospective study with clear inclusion criteria, statistical analysis plan, closely matched study groups, and the patient follow-up was high. Among the limitations of the study is the open label study design and the relatively small sample size. We believe that the small sample size does not affect the internal validity of this study. Though noninferior, the results are unbiased. 228
7 In conclusion, we have demonstrated that oral riboflavin 100mg/day is comparable with oral propranolol 80 mg/day in reducing the frequency, duration and severity of migraine headaches and may be preferred over the latter due to its safety and tolerability. DISCLOSURE Disclaimer: None Source of support: Nil ACKNOWLEDGEMENT I would like to thank Dr. K. P. Suresh, Scientist (Biostatistics), National Institute of Animal Nutrition & Physiology, Bangalore for providing assistance with the statistical analysis for the study. I would also like to thank Dr. S. Kumar, Principal and Dean of the M.S. Ramaiah Medical College for providing the facilities to undertake research activities in the Institute. REFERENCES 1. Salomone S, Caraci F, Capasso A. Migraine: An overview. The Open Neurology Journal 2009; 3: Silberstein SD, Saper JR, Freitag FG. Migraine: diagnosis and treatment. In: Silberstein SD, Lipton RB, Dalessio DJ, eds. Wolff s headache and other head pain. 7th ed. New York: Oxford University Press, 2001: World Health Organization. The World Health Report 2001: Mental Health: New Understanding, New Hope. index.html. Accessed January 12, Berg J, Ramadan NM. Societal burden of the headache. In: Olesen J, Goadsby PJ, Ramadan NM, Tfelt-Hansen P, Welch KMA, eds: The Headaches. 3rd ed. Philadelphia: Lippincott Williams and Wilkin 2006; Silberstein SD. Practice parameter: evidence-based guidelines for migraine headache (an evidence based review): report of the Quality Standards Subcommittee of the American Academy of Neurology. Neurology 2000; 55: Schoenen J, Lenaerts M, Bastings E. High-dose riboflavin as a prophylactic treat ment of migraine: results of an open pilot study. Cephalalgia 1994; 14(5): Ferrari A, Sternieri E, Ferraris E, Bertolini A. Emerging problems in the pharmacotherapy of migraine: interactions between triptans and drugs for prophylaxis. Pharmacol Res 2003; 48: Montagna P, Gallassi R, Medori R, et al. MELAS syndrome: characteristic migrainous and epileptic features and maternal transmission. Neurology 1998; 38: Goto Y, Horai S, Matsuoka T, et al. Mitochondrial myopathy, encephalopathy, lactic acidosis, and strokelike episodes (MELAS): a correlative study of the clinical features and mitochondrial DNA mutation. Neurology 1992; 42: Sandor PS, Afra J, Ambrosini A, Schoenen J. Prophylactic treatment of migraine with beta-blockers and riboflavin: differential effects on the intensity dependence of auditory evoked cortical potentials. Headache 2000; 40(1): Schoenen J, Leneaerts M, Bastings E. High dose riboflavin as a prophylactic treatment of migraine: results of an open pilot study. Cephalalgia 1994; 14: Schoenen J, Jacquy J, Lenaerts M. Effectiveness of highdose riboflavin in migraine prophylaxis. A randomized controlled trial. Neurology 1998; 50: Boehnke C, Reuter U, Flach U, et al. High-dose riboflavin treatment is efficacious in migraine prophylaxis:an open study in a tertiary care centre. Eur J Neurol 2004; 11: Franca DS, Souza ALS, Almeida KR, Dolabella SS, Martinelli C, Coelho MM. B vitamins induce an antinociceptive effect in the acetic acid and formaldehyde models of nociception in mice. Eur J Pharmacol 2001; 421: Granados-Soto V, Terán-Rosales F, Rocha-González HI, et al. Riboflavin reduces hyperalgesia and inflammation but not tactile allodynia in the rat. Eur J Pharmacol 2004; 492:
New appendix criteria open for a broader concept of chronic migraine
 Blackwell Publishing LtdOxford, UKCHACephalalgia0333-1024Blackwell Science, 20062006266742746Original ArticleA broader concept of chronic migrainej Olesen et al. BRIEF REPORT New appendix criteria open
Blackwell Publishing LtdOxford, UKCHACephalalgia0333-1024Blackwell Science, 20062006266742746Original ArticleA broader concept of chronic migrainej Olesen et al. BRIEF REPORT New appendix criteria open
2.0 Synopsis. Vicodin CR (ABT-712) M05-765 Clinical Study Report R&D/07/095. (For National Authority Use Only) to Part of Dossier: Volume:
 2.0 Synopsis Abbott Laboratories Name of Study Drug: Vicodin CR Name of Active Ingredient: Hydrocodone/Acetaminophen Extended Release (ABT-712) Individual Study Table Referring to Part of Dossier: Volume:
2.0 Synopsis Abbott Laboratories Name of Study Drug: Vicodin CR Name of Active Ingredient: Hydrocodone/Acetaminophen Extended Release (ABT-712) Individual Study Table Referring to Part of Dossier: Volume:
Frequent headache is defined as headaches 15 days/month and daily. Course of Frequent/Daily Headache in the General Population and in Medical Practice
 DISEASE STATE REVIEW Course of Frequent/Daily Headache in the General Population and in Medical Practice Egilius L.H. Spierings, MD, PhD, Willem K.P. Mutsaerts, MSc Department of Neurology, Brigham and
DISEASE STATE REVIEW Course of Frequent/Daily Headache in the General Population and in Medical Practice Egilius L.H. Spierings, MD, PhD, Willem K.P. Mutsaerts, MSc Department of Neurology, Brigham and
BOTOX Injection (Onabotulinumtoxin A) for Migraine Headaches [Preauthorization Required]
![BOTOX Injection (Onabotulinumtoxin A) for Migraine Headaches [Preauthorization Required] BOTOX Injection (Onabotulinumtoxin A) for Migraine Headaches [Preauthorization Required]](/thumbs/29/13417512.jpg) BOTOX Injection (Onabotulinumtoxin A) for Migraine Headaches [Preauthorization Required] Medical Policy: MP-RX-01-11 Original Effective Date: March 24, 2011 Reviewed: Revised: This policy applies to products
BOTOX Injection (Onabotulinumtoxin A) for Migraine Headaches [Preauthorization Required] Medical Policy: MP-RX-01-11 Original Effective Date: March 24, 2011 Reviewed: Revised: This policy applies to products
Tension Type Headaches
 Tension Type Headaches Research Review by : Dr. Ian MacIntyre Physiotherapy for tension-type Headache: A Controlled Study P. Torelli, R. Jenson, J. Olsen: Cephalalgia, 2004, 24, 29-36 Tension-type headache
Tension Type Headaches Research Review by : Dr. Ian MacIntyre Physiotherapy for tension-type Headache: A Controlled Study P. Torelli, R. Jenson, J. Olsen: Cephalalgia, 2004, 24, 29-36 Tension-type headache
Appendix 1 (as supplied by the authors): Framework and content of the intervention of the LIMIT study
 Appendix 1 (as supplied by the authors): Framework and content of the intervention of the LIMIT study Background The training was based on the learning cycle of Kolb 1. According to this cycle effective
Appendix 1 (as supplied by the authors): Framework and content of the intervention of the LIMIT study Background The training was based on the learning cycle of Kolb 1. According to this cycle effective
PROTOCOL SYNOPSIS Evaluation of long-term opioid efficacy for chronic pain
 P a g e 1 PROTOCOL SYNOPSIS Evaluation of long-term opioid efficacy for chronic pain Clinical Phase 4 Study Centers Study Period 25 U.S. sites identified and reviewed by the Steering Committee and Contract
P a g e 1 PROTOCOL SYNOPSIS Evaluation of long-term opioid efficacy for chronic pain Clinical Phase 4 Study Centers Study Period 25 U.S. sites identified and reviewed by the Steering Committee and Contract
VITAMIN C AND INFECTIOUS DISEASE: A REVIEW OF THE LITERATURE AND THE RESULTS OF A RANDOMIZED, DOUBLE-BLIND, PROSPECTIVE STUDY OVER 8 YEARS
 39 Chapter 3 VITAMIN C AND INFECTIOUS DISEASE: A REVIEW OF THE LITERATURE AND THE RESULTS OF A RANDOMIZED, DOUBLE-BLIND, PROSPECTIVE STUDY OVER 8 YEARS Maxine Briggs TABLE OF CONTENTS I. Review of the
39 Chapter 3 VITAMIN C AND INFECTIOUS DISEASE: A REVIEW OF THE LITERATURE AND THE RESULTS OF A RANDOMIZED, DOUBLE-BLIND, PROSPECTIVE STUDY OVER 8 YEARS Maxine Briggs TABLE OF CONTENTS I. Review of the
INITIATING ORAL AUBAGIO (teriflunomide) THERAPY
 FOR YOUR PATIENTS WITH RELAPSING FORMS OF MS INITIATING ORAL AUBAGIO (teriflunomide) THERAPY WARNING: HEPATOTOXICITY AND RISK OF TERATOGENICITY Severe liver injury including fatal liver failure has been
FOR YOUR PATIENTS WITH RELAPSING FORMS OF MS INITIATING ORAL AUBAGIO (teriflunomide) THERAPY WARNING: HEPATOTOXICITY AND RISK OF TERATOGENICITY Severe liver injury including fatal liver failure has been
Handicap after acute whiplash injury A 1-year prospective study of risk factors
 1 Handicap after acute whiplash injury A 1-year prospective study of risk factors Neurology 2001;56:1637-1643 (June 26, 2001) Helge Kasch, MD, PhD; Flemming W Bach, MD, PhD; Troels S Jensen, MD, PhD From
1 Handicap after acute whiplash injury A 1-year prospective study of risk factors Neurology 2001;56:1637-1643 (June 26, 2001) Helge Kasch, MD, PhD; Flemming W Bach, MD, PhD; Troels S Jensen, MD, PhD From
Natural Modality in the Treatment of Primary Headaches. William S. Mihin, D.C. Catharine Helms, M.S. Michelle M. Anderson, M.S.N., F.N.P.
 Natural Modality in the Treatment of Primary Headaches William S. Mihin, D.C. Catharine Helms, M.S. Michelle M. Anderson, M.S.N., F.N.P. Abstract Headaches are both a prevalent and disabling condition.
Natural Modality in the Treatment of Primary Headaches William S. Mihin, D.C. Catharine Helms, M.S. Michelle M. Anderson, M.S.N., F.N.P. Abstract Headaches are both a prevalent and disabling condition.
The Prevalence of Neck Pain in Migrainehead_1608
 1..5 Headache 2010 the Authors Journal compilation 2010 American Headache Society ISSN 0017-8748 doi: 10.1111/j.1526-4610.2009.01608.x Published by Wiley Periodicals, Inc. Research Submission The Prevalence
1..5 Headache 2010 the Authors Journal compilation 2010 American Headache Society ISSN 0017-8748 doi: 10.1111/j.1526-4610.2009.01608.x Published by Wiley Periodicals, Inc. Research Submission The Prevalence
Update and Review of Medication Assisted Treatments
 Update and Review of Medication Assisted Treatments for Opiate and Alcohol Use Disorders Richard N. Whitney, MD Medical Director Addiction Services Shepherd Hill Newark, Ohio Medication Assisted Treatment
Update and Review of Medication Assisted Treatments for Opiate and Alcohol Use Disorders Richard N. Whitney, MD Medical Director Addiction Services Shepherd Hill Newark, Ohio Medication Assisted Treatment
There are two different types of migraines: migraines without aura and migraines with aura.
 What is migraine? A migraine is a relatively common medical condition that can severely affect the quality of life of the sufferer and his or her family and friends. 1 Almost 8% of Canadians over the age
What is migraine? A migraine is a relatively common medical condition that can severely affect the quality of life of the sufferer and his or her family and friends. 1 Almost 8% of Canadians over the age
Double-Blind Trial of Fluoxetine: Chronic Daily Headache and Migraine
 Double-Blind Trial of Fluoxetine: Chronic Daily Headache and Migraine Joel R. Saper, M.D., 1 Stephen D. Silberstein, M.D., 2 Alvin E. Lake III, Ph.D., 1 Marjorie E. Winters, R.N., B.S.N. 1 1 Michigan Head-Pain
Double-Blind Trial of Fluoxetine: Chronic Daily Headache and Migraine Joel R. Saper, M.D., 1 Stephen D. Silberstein, M.D., 2 Alvin E. Lake III, Ph.D., 1 Marjorie E. Winters, R.N., B.S.N. 1 1 Michigan Head-Pain
Oral Zinc Supplementation as an Adjunct Therapy in the Management of Hepatic Encephalopathy: A Randomized Controlled Trial
 Oral Zinc Supplementation as an Adjunct Therapy in the Management of Hepatic Encephalopathy: A Randomized Controlled Trial Marcus R. Pereira A. Study Purpose Hepatic encephalopathy is a common complication
Oral Zinc Supplementation as an Adjunct Therapy in the Management of Hepatic Encephalopathy: A Randomized Controlled Trial Marcus R. Pereira A. Study Purpose Hepatic encephalopathy is a common complication
Prednisone for the treatment of withdrawal headache in patients with medication overuse headache: A randomized, double-blind, placebo-controlled study
 Brief Report Prednisone for the treatment of withdrawal headache in patients with medication overuse headache: A randomized, double-blind, placebo-controlled study Cephalalgia 33(3) 202 207! International
Brief Report Prednisone for the treatment of withdrawal headache in patients with medication overuse headache: A randomized, double-blind, placebo-controlled study Cephalalgia 33(3) 202 207! International
Guidance for Industry Migraine: Developing Drugs for Acute Treatment
 Guidance for Industry Migraine: Developing Drugs for Acute Treatment DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments and suggestions regarding this draft
Guidance for Industry Migraine: Developing Drugs for Acute Treatment DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments and suggestions regarding this draft
Outcome of Drug Counseling of Outpatients in Chronic Obstructive Pulmonary Disease Clinic at Thawangpha Hospital
 Mahidol University Journal of Pharmaceutical Sciences 008; 35(14): 81. Original Article Outcome of Drug Counseling of Outpatients in Chronic Obstructive Pulmonary Disease Clinic at Thawangpha Hospital
Mahidol University Journal of Pharmaceutical Sciences 008; 35(14): 81. Original Article Outcome of Drug Counseling of Outpatients in Chronic Obstructive Pulmonary Disease Clinic at Thawangpha Hospital
Adjunctive psychosocial intervention. Conditions requiring dose reduction. Immediate, peak plasma concentration is reached within 1 hour.
 Shared Care Guideline for Prescription and monitoring of Naltrexone Hydrochloride in alcohol dependence Author(s)/Originator(s): (please state author name and department) Dr Daly - Consultant Psychiatrist,
Shared Care Guideline for Prescription and monitoring of Naltrexone Hydrochloride in alcohol dependence Author(s)/Originator(s): (please state author name and department) Dr Daly - Consultant Psychiatrist,
Behavioral and Physical Treatments for Tension-type and Cervicogenic Headache
 Evidence Report: Behavioral and Physical Treatments for Tension-type and Cervicogenic Headache Douglas C. McCrory, MD, MHSc Donald B. Penzien, PhD Vic Hasselblad, PhD Rebecca N. Gray, DPhil Duke University
Evidence Report: Behavioral and Physical Treatments for Tension-type and Cervicogenic Headache Douglas C. McCrory, MD, MHSc Donald B. Penzien, PhD Vic Hasselblad, PhD Rebecca N. Gray, DPhil Duke University
Headache - What is Your Migraine Size?
 Headache The Pharmacist s Role in Assessment & Peter Loewen, B.Sc.(Pharm), Pharm.D. Vancouver Hospital & Health Sciences Centre University of British Columbia ETC, Headache. Nan Quintin www.vhpharmsci.com
Headache The Pharmacist s Role in Assessment & Peter Loewen, B.Sc.(Pharm), Pharm.D. Vancouver Hospital & Health Sciences Centre University of British Columbia ETC, Headache. Nan Quintin www.vhpharmsci.com
NIMULID MD. 1. Introduction. 2. Nimulid MD Drug delivery system
 NIMULID MD 1. Introduction Nimulid MD is a flavoured dispersible Nimesulide tablet with fast mouth dissolving characteristics thereby providing immediate relief. Nimesulide is a non-steroidal antiinflammatory
NIMULID MD 1. Introduction Nimulid MD is a flavoured dispersible Nimesulide tablet with fast mouth dissolving characteristics thereby providing immediate relief. Nimesulide is a non-steroidal antiinflammatory
How To Treat The Sando Syndrome
 Consultation on Drugs for Rare Diseases Rare Disease Day Symposium Hannover Medical School (MHH), Solidarity Rare but Strong Together Roland Seifert, MHH @ Dear Sir, I am writing hoping that there might
Consultation on Drugs for Rare Diseases Rare Disease Day Symposium Hannover Medical School (MHH), Solidarity Rare but Strong Together Roland Seifert, MHH @ Dear Sir, I am writing hoping that there might
This clinical study synopsis is provided in line with Boehringer Ingelheim s Policy on Transparency and Publication of Clinical Study Data.
 abcd Clinical Study for Public Disclosure This clinical study synopsis is provided in line with s Policy on Transparency and Publication of Clinical Study Data. The synopsis which is part of the clinical
abcd Clinical Study for Public Disclosure This clinical study synopsis is provided in line with s Policy on Transparency and Publication of Clinical Study Data. The synopsis which is part of the clinical
Guidelines for controlled trials of prophylactic treatment of chronic migraine in adults
 doi:10.1111/j.1468-2982.2008.01555.x REVIEW Guidelines for controlled trials of prophylactic treatment of chronic migraine in adults S Silberstein 1 (Chairman, USA), P Tfelt-Hansen 2 (Co-Chairman, Denmark),
doi:10.1111/j.1468-2982.2008.01555.x REVIEW Guidelines for controlled trials of prophylactic treatment of chronic migraine in adults S Silberstein 1 (Chairman, USA), P Tfelt-Hansen 2 (Co-Chairman, Denmark),
Test Content Outline Effective Date: June 9, 2014. Pain Management Nursing Board Certification Examination
 Pain Management Nursing Board Certification Examination There are 175 questions on this examination. Of these, 150 are scored questions and 25 are pretest questions that are not scored. Pretest questions
Pain Management Nursing Board Certification Examination There are 175 questions on this examination. Of these, 150 are scored questions and 25 are pretest questions that are not scored. Pretest questions
Substance Abuse Treatment. Naltrexone for Extended-Release Injectable Suspension for Treatment of Alcohol Dependence
 Spring 2007 Volume 6 Issue 1 ADVISORY News for the Treatment Field Naltrexone for Extended-Release Injectable Suspension for Treatment of Alcohol Dependence What is naltrexone for extendedrelease injectable
Spring 2007 Volume 6 Issue 1 ADVISORY News for the Treatment Field Naltrexone for Extended-Release Injectable Suspension for Treatment of Alcohol Dependence What is naltrexone for extendedrelease injectable
Migraine The Problem: Common Symptoms:
 Migraine The Problem: A combination of genetic and environmental factors alter pain mechanisms in your brain Transient changes in brain chemicals such as serotonin and neuropeptides affect the membranes
Migraine The Problem: A combination of genetic and environmental factors alter pain mechanisms in your brain Transient changes in brain chemicals such as serotonin and neuropeptides affect the membranes
Post Traumatic and other Headache Syndromes. Danielle L. Erb, MD Brain Rehabilitation Medicine, LLC Brain Injury Rehab Center, PRA
 Post Traumatic and other Headache Syndromes Danielle L. Erb, MD Brain Rehabilitation Medicine, LLC Brain Injury Rehab Center, PRA Over 45 million Americans have chronic, recurring headaches 62% of these
Post Traumatic and other Headache Syndromes Danielle L. Erb, MD Brain Rehabilitation Medicine, LLC Brain Injury Rehab Center, PRA Over 45 million Americans have chronic, recurring headaches 62% of these
Maintenance of abstinence in alcohol dependence
 Shared Care Guideline for Prescription and monitoring of Acamprosate Calcium Author(s)/Originator(s): (please state author name and department) Dr Daly - Consultant Psychiatrist, Alcohol Services Dr Donnelly
Shared Care Guideline for Prescription and monitoring of Acamprosate Calcium Author(s)/Originator(s): (please state author name and department) Dr Daly - Consultant Psychiatrist, Alcohol Services Dr Donnelly
Medication Policy Manual. Topic: Aubagio, teriflunomide Date of Origin: November 9, 2012
 Medication Policy Manual Policy No: dru283 Topic: Aubagio, teriflunomide Date of Origin: November 9, 2012 Committee Approval Date: December 12, 2014 Next Review Date: December 2015 Effective Date: January
Medication Policy Manual Policy No: dru283 Topic: Aubagio, teriflunomide Date of Origin: November 9, 2012 Committee Approval Date: December 12, 2014 Next Review Date: December 2015 Effective Date: January
HEADACHES IN CHILDREN AND ADOLESCENTS. Brian D. Ryals, M.D.
 HEADACHES IN CHILDREN AND ADOLESCENTS Brian D. Ryals, M.D. Frequency and Type of Headaches in Schoolchildren 8993 children age 7-15 in Sweden Migraine in 4% Frequent Nonmigrainous in 7% Infrequent Nonmigrainous
HEADACHES IN CHILDREN AND ADOLESCENTS Brian D. Ryals, M.D. Frequency and Type of Headaches in Schoolchildren 8993 children age 7-15 in Sweden Migraine in 4% Frequent Nonmigrainous in 7% Infrequent Nonmigrainous
CLINICAL STUDY REPORT SYNOPSIS
 CLINICAL STUDY REPORT SYNOPSIS Document No.: EDMS-PSDB-6511351:2.0 Name of Sponsor/Company Name of Finished Product Name of Active Ingredient(s) Protocol No.: CR002353 Johnson & Johnson Pharmaceutical
CLINICAL STUDY REPORT SYNOPSIS Document No.: EDMS-PSDB-6511351:2.0 Name of Sponsor/Company Name of Finished Product Name of Active Ingredient(s) Protocol No.: CR002353 Johnson & Johnson Pharmaceutical
Progress in MS: Current and Emerging Therapies
 Progress in MS: Current and Emerging Therapies Presented by: Dr. Kathryn Giles, MD MSc FRCPC The MS Society gratefully acknowledges the grant received from Biogen Idec Canada, which makes possible the
Progress in MS: Current and Emerging Therapies Presented by: Dr. Kathryn Giles, MD MSc FRCPC The MS Society gratefully acknowledges the grant received from Biogen Idec Canada, which makes possible the
Classification of Chronic Headache
 Chronic Headache Classification of Chronic Headache JMAJ 47(3): 112 117, 2004 Mitsunori MORIMATSU Professor, Department of Neurology and Clinical Neuroscience, Yamaguchi University School of Medicine Abstract:
Chronic Headache Classification of Chronic Headache JMAJ 47(3): 112 117, 2004 Mitsunori MORIMATSU Professor, Department of Neurology and Clinical Neuroscience, Yamaguchi University School of Medicine Abstract:
Presented by: Jean Yoo-Campbell, Matthew Konerman, Monica Konerman, Jean Yoo Campbell, Christian Gocke, Eunpi Cho Donald Lynch
 Bass N.M., et. al. N Engl J Med 2010; 362:1071-1081 Presented by: Jean Yoo-Campbell, Matthew Konerman, Monica Konerman, Jean Yoo Campbell, Christian Gocke, Eunpi Cho Donald Lynch Faculty Advisor: Dr. Fred
Bass N.M., et. al. N Engl J Med 2010; 362:1071-1081 Presented by: Jean Yoo-Campbell, Matthew Konerman, Monica Konerman, Jean Yoo Campbell, Christian Gocke, Eunpi Cho Donald Lynch Faculty Advisor: Dr. Fred
SYNOPSIS. Risperidone: Clinical Study Report CR003274
 SYNOPSIS Protocol No: CR003274 Title of Study: An Open-Label, Long-Term Trial of Risperidone Long-Acting Microspheres in the Treatment of Subjects Diagnosed with Schizophrenia Coordinating Investigator:
SYNOPSIS Protocol No: CR003274 Title of Study: An Open-Label, Long-Term Trial of Risperidone Long-Acting Microspheres in the Treatment of Subjects Diagnosed with Schizophrenia Coordinating Investigator:
Naltrexone Shared Care Guideline for the treatment of alcohol dependence and opioid dependance
 Naltrexone Shared Care Guideline for the treatment of alcohol dependence and opioid dependance Introduction Indication/Licensing information: Naltrexone is licensed for use as an additional therapy, within
Naltrexone Shared Care Guideline for the treatment of alcohol dependence and opioid dependance Introduction Indication/Licensing information: Naltrexone is licensed for use as an additional therapy, within
ROUNDTABLE DISCUSSION
 THE ROLE OF OPIOID THERAPY IN MANAGING CHRONIC DAILY HEADACHE * The following includes a summary of a roundtable discussion with panel members Joel R. Saper, MD ; John F. Rothrock, MD ; and Jeffrey D.
THE ROLE OF OPIOID THERAPY IN MANAGING CHRONIC DAILY HEADACHE * The following includes a summary of a roundtable discussion with panel members Joel R. Saper, MD ; John F. Rothrock, MD ; and Jeffrey D.
Views and Perspectives
 Headache 2010 American Headache Society ISSN 0017-8748 doi: 10.1111/j.1526-4610.2010.01764.x Published by Wiley Periodicals, Inc. Views and Perspectives Defining the Pharmacologically Intractable Headache
Headache 2010 American Headache Society ISSN 0017-8748 doi: 10.1111/j.1526-4610.2010.01764.x Published by Wiley Periodicals, Inc. Views and Perspectives Defining the Pharmacologically Intractable Headache
A Phase 2 Study of HTX-011 in the Management of Post-Operative Pain Positive Top-Line Results
 A Phase 2 Study of HTX-011 in the Management of Post-Operative Pain Positive Top-Line Results September 22, 2015 Forward-Looking Statements This presentation contains "forward-looking statements" as defined
A Phase 2 Study of HTX-011 in the Management of Post-Operative Pain Positive Top-Line Results September 22, 2015 Forward-Looking Statements This presentation contains "forward-looking statements" as defined
Simplifying the measurement of co-morbidities and their influence on chemotherapy toxicity
 Simplifying the measurement of co-morbidities and their influence on chemotherapy toxicity Dr Rajesh Sinha BSc MBBS MRCP, Clinical Research Fellow in Medical Oncology Brighton and Sussex University Hospitals
Simplifying the measurement of co-morbidities and their influence on chemotherapy toxicity Dr Rajesh Sinha BSc MBBS MRCP, Clinical Research Fellow in Medical Oncology Brighton and Sussex University Hospitals
Headaches in Children How to Manage Difficult Headaches
 Headaches in Children How to Manage Difficult Headaches Peter Procopis Childhood headaches Differential diagnosis Migraine Psychological Raised Pressure Childhood headaches Other causes: Constitutional
Headaches in Children How to Manage Difficult Headaches Peter Procopis Childhood headaches Differential diagnosis Migraine Psychological Raised Pressure Childhood headaches Other causes: Constitutional
Chronic daily headache with analgesic overuse Epidemiology and impact on quality of life. NEUROLOGY April, 2004;62:1338 1342
 Chronic daily headache with analgesic overuse Epidemiology and impact on quality of life 1 NEUROLOGY April, 2004;62:1338 1342 R. Colás, MD; P. Muñoz, MD; R. Temprano, MD; C. Gómez, SW; and J. Pascual,
Chronic daily headache with analgesic overuse Epidemiology and impact on quality of life 1 NEUROLOGY April, 2004;62:1338 1342 R. Colás, MD; P. Muñoz, MD; R. Temprano, MD; C. Gómez, SW; and J. Pascual,
Not All Clinical Trials Are Created Equal Understanding the Different Phases
 Not All Clinical Trials Are Created Equal Understanding the Different Phases This chapter will help you understand the differences between the various clinical trial phases and how these differences impact
Not All Clinical Trials Are Created Equal Understanding the Different Phases This chapter will help you understand the differences between the various clinical trial phases and how these differences impact
EFFECT OF DAILY VERSUS WEEKLY IRON FOLIC ACID SUPPLEMENTATION ON THE HAEMOGLOBIN LEVELS OF CHILDREN 6 TO 36 MONTHS OF URBAN SLUMS OF VADODARA
 ORIGINAL ARTICLE. EFFECT OF DAILY VERSUS WEEKLY IRON FOLIC ACID SUPPLEMENTATION ON THE HAEMOGLOBIN LEVELS OF CHILDREN 6 TO 36 MONTHS OF URBAN SLUMS OF VADODARA K Sharma 1, P Parikh 2, F Desai 3 1 PhD Advisor,
ORIGINAL ARTICLE. EFFECT OF DAILY VERSUS WEEKLY IRON FOLIC ACID SUPPLEMENTATION ON THE HAEMOGLOBIN LEVELS OF CHILDREN 6 TO 36 MONTHS OF URBAN SLUMS OF VADODARA K Sharma 1, P Parikh 2, F Desai 3 1 PhD Advisor,
Sponsor. Novartis Generic Drug Name. Vildagliptin. Therapeutic Area of Trial. Type 2 diabetes. Approved Indication. Investigational.
 Clinical Trial Results Database Page 1 Sponsor Novartis Generic Drug Name Vildagliptin Therapeutic Area of Trial Type 2 diabetes Approved Indication Investigational Study Number CLAF237A2386 Title A single-center,
Clinical Trial Results Database Page 1 Sponsor Novartis Generic Drug Name Vildagliptin Therapeutic Area of Trial Type 2 diabetes Approved Indication Investigational Study Number CLAF237A2386 Title A single-center,
Spine Vol. 30 No. 16; August 15, 2005, pp 1799-1807
 A Randomized Controlled Trial of an Educational Intervention to Prevent the Chronic Pain of Whiplash Associated Disorders Following Rear-End Motor Vehicle Collisions 1 Spine Vol. 30 No. 16; August 15,
A Randomized Controlled Trial of an Educational Intervention to Prevent the Chronic Pain of Whiplash Associated Disorders Following Rear-End Motor Vehicle Collisions 1 Spine Vol. 30 No. 16; August 15,
Committee Approval Date: December 12, 2014 Next Review Date: December 2015
 Medication Policy Manual Policy No: dru299 Topic: Tecfidera, dimethyl fumarate Date of Origin: May 16, 2013 Committee Approval Date: December 12, 2014 Next Review Date: December 2015 Effective Date: January
Medication Policy Manual Policy No: dru299 Topic: Tecfidera, dimethyl fumarate Date of Origin: May 16, 2013 Committee Approval Date: December 12, 2014 Next Review Date: December 2015 Effective Date: January
The Application of Migraine Disability Assessment Questionnaire (MIDAS)
 Continuing Medical Education 43 The Application of Migraine Disability Assessment Questionnaire () Pei-Hua Hung, Jong-Ling Fuh, and Shuu-Jiun Wang Abstract- Migraine is a recurring and disabling pain disorder
Continuing Medical Education 43 The Application of Migraine Disability Assessment Questionnaire () Pei-Hua Hung, Jong-Ling Fuh, and Shuu-Jiun Wang Abstract- Migraine is a recurring and disabling pain disorder
How Is OnabotulinumtoxinA Reimbursed For Chronic Migraine? Impact Of FDA Approval And The New CPT Code
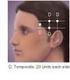 How Is OnabotulinumtoxinA Reimbursed For Chronic Migraine? Impact Of FDA Approval And The New CPT Code Effective January 1, 2013, physicians will be able to report the new CPT code 64615 when performing
How Is OnabotulinumtoxinA Reimbursed For Chronic Migraine? Impact Of FDA Approval And The New CPT Code Effective January 1, 2013, physicians will be able to report the new CPT code 64615 when performing
Novartis Gilenya FDO Program Clinical Protocol and Highlights from Prescribing Information (PI)
 Novartis Gilenya FDO Program Clinical Protocol and Highlights from Prescribing Information (PI) Highlights from Prescribing Information - the link to the full text PI is as follows: http://www.pharma.us.novartis.com/product/pi/pdf/gilenya.pdf
Novartis Gilenya FDO Program Clinical Protocol and Highlights from Prescribing Information (PI) Highlights from Prescribing Information - the link to the full text PI is as follows: http://www.pharma.us.novartis.com/product/pi/pdf/gilenya.pdf
Drugs for MS.Drug fact box cannabis extract (Sativex) Version 1.0 Author
 Version History Policy Title Drugs for MS.Drug fact box cannabis extract (Sativex) Version 1.0 Author West Midlands Commissioning Support Unit Publication Date Jan 2013 Review Date Supersedes/New (Further
Version History Policy Title Drugs for MS.Drug fact box cannabis extract (Sativex) Version 1.0 Author West Midlands Commissioning Support Unit Publication Date Jan 2013 Review Date Supersedes/New (Further
When the Pain Won t Stop: Managing Chronic Daily Headache
 When the Pain Won t Stop: Managing Chronic Daily Headache Arnolda Eloff, MB, ChB, Mmed Presented at the University of Calgary s Wednesday Evening Course Program, Calgary, Alberta. Copyright Chronic daily
When the Pain Won t Stop: Managing Chronic Daily Headache Arnolda Eloff, MB, ChB, Mmed Presented at the University of Calgary s Wednesday Evening Course Program, Calgary, Alberta. Copyright Chronic daily
Evaluation of a Morphine Weaning Protocol in Pediatric Intensive Care Patients
 Evaluation of a Morphine Weaning Protocol in Pediatric Intensive Care Patients Jennifer Kuhns, Pharm.D. Pharmacy Practice Resident Children s Hospital of Michigan **The speaker has no actual or potential
Evaluation of a Morphine Weaning Protocol in Pediatric Intensive Care Patients Jennifer Kuhns, Pharm.D. Pharmacy Practice Resident Children s Hospital of Michigan **The speaker has no actual or potential
Version History. Previous Versions. Policy Title. Drugs for MS.Drug facts box Glatiramer Acetate Version 1.0 Author
 Version History Policy Title Drugs for MS.Drug facts box Glatiramer Acetate Version 1.0 Author West Midlands Commissioning Support Unit Publication Date Jan 2013 Review Date Supersedes/New (Further fields
Version History Policy Title Drugs for MS.Drug facts box Glatiramer Acetate Version 1.0 Author West Midlands Commissioning Support Unit Publication Date Jan 2013 Review Date Supersedes/New (Further fields
Results of all scientific investigations with the A.Vogel Sore Throat Spray. Issue 2 - June 2008
 Results of all scientific investigations with the A.Vogel Sore Issue 2 - June 2008 Antiviral Antibacterial Anti-inflammatory Analgetic Immunemodulatory Authors: Andy Suter, Medical Department Roland Schoop,
Results of all scientific investigations with the A.Vogel Sore Issue 2 - June 2008 Antiviral Antibacterial Anti-inflammatory Analgetic Immunemodulatory Authors: Andy Suter, Medical Department Roland Schoop,
WAT ALS HOOFDPIJN CHRONISCH WORDT? Prof Jan Versijpt, MD PhD Kliniek voor Hoofd- en Aangezichtspijn Neurologie, UZ Brussel
 WAT ALS HOOFDPIJN CHRONISCH WORDT? Prof Jan Versijpt, MD PhD Kliniek voor Hoofd- en Aangezichtspijn Neurologie, UZ Brussel Stovner, IHC 2009 New daily persistent headache Primair = dagelijkse and continue
WAT ALS HOOFDPIJN CHRONISCH WORDT? Prof Jan Versijpt, MD PhD Kliniek voor Hoofd- en Aangezichtspijn Neurologie, UZ Brussel Stovner, IHC 2009 New daily persistent headache Primair = dagelijkse and continue
Tara Stevermuer (MAppStat), Centre for Health Service Development, University of Wollongong.
 ENAR - PAIN RELIEF THAT S FAST AND LASTS Tara Stevermuer (MAppStat), Centre for Health Service Development, University of Wollongong. Introducing ENAR The ENAR (Electro-Neuro-Adaptive-Regulator) is a Russian
ENAR - PAIN RELIEF THAT S FAST AND LASTS Tara Stevermuer (MAppStat), Centre for Health Service Development, University of Wollongong. Introducing ENAR The ENAR (Electro-Neuro-Adaptive-Regulator) is a Russian
Medications for Alcohol and Opioid Use Disorders
 Medications for Alcohol and Opioid Use Disorders Andrew J. Saxon, M.D. Center of Excellence in Substance Abuse Treatment and Education (CESATE) VA Puget Sound Health Care System Alcohol Pharmacotherapy
Medications for Alcohol and Opioid Use Disorders Andrew J. Saxon, M.D. Center of Excellence in Substance Abuse Treatment and Education (CESATE) VA Puget Sound Health Care System Alcohol Pharmacotherapy
HEADACHE. as. MUDr. Rudolf Černý, CSc. doc. MUDr. Petr Marusič, Ph.D.
 HEADACHE as. MUDr. Rudolf Černý, CSc. doc. MUDr. Petr Marusič, Ph.D. Dpt. of Neurology Charles University in Prague, 2nd Faculty of Medicine Motol University Hospital History of headache 1200 years B.C.
HEADACHE as. MUDr. Rudolf Černý, CSc. doc. MUDr. Petr Marusič, Ph.D. Dpt. of Neurology Charles University in Prague, 2nd Faculty of Medicine Motol University Hospital History of headache 1200 years B.C.
Prior Authorization Guideline
 Prior Authorization Guideline Guideline: PDP IBT Inj - Vivitrol Therapeutic Class: Central Nervous System Agents Therapeutic Sub-Class: Opiate Antagonist Client: 2007 PDP IBT Inj Approval Date: 2/20/2007
Prior Authorization Guideline Guideline: PDP IBT Inj - Vivitrol Therapeutic Class: Central Nervous System Agents Therapeutic Sub-Class: Opiate Antagonist Client: 2007 PDP IBT Inj Approval Date: 2/20/2007
Childhood Migraine A Complementary and Alternative Approach
 Childhood Migraine A Complementary and Alternative Approach Thomas K. Koch, MD Faculty Disclosure Information Childhood Migraine: A Complementary and Alternative Approach Presented by Thomas K. Koch, MD
Childhood Migraine A Complementary and Alternative Approach Thomas K. Koch, MD Faculty Disclosure Information Childhood Migraine: A Complementary and Alternative Approach Presented by Thomas K. Koch, MD
Introduction. Jean-Christophe Cuvellier 1 Frédéric Couttenier 2 Stéphane Auvin 1 Louis Vallée 1 ORIGINAL RESEARCH
 ORIGINAL RESEARCH The classifi cation of chronic daily headache in French children and adolescents: A comparison between the second edition of the International Classifi cation of Headache Disorders and
ORIGINAL RESEARCH The classifi cation of chronic daily headache in French children and adolescents: A comparison between the second edition of the International Classifi cation of Headache Disorders and
Expectancy Effects of Performance Enhancing Supplements on Motivation to Exercise. Chris Dawson and Alfonso Ribero.
 Running Head: EXPECTANCY EFFECTS ON MOTIVATION Expectancy Effects of Performance Enhancing Supplements on Motivation to Exercise Chris Dawson and Alfonso Ribero Hanover College Expectancy Effects 2 Abstract
Running Head: EXPECTANCY EFFECTS ON MOTIVATION Expectancy Effects of Performance Enhancing Supplements on Motivation to Exercise Chris Dawson and Alfonso Ribero Hanover College Expectancy Effects 2 Abstract
Updated guidelines on headache management for use by the pharmacist
 M I P C A MIGRAINE IN PRIMARY CARE ADVISORS Updated guidelines on headache management for use by the pharmacist Introduction N U M B E R 2 1, O C T O B E R 2 0 1 2 Headache is a major public health problem,
M I P C A MIGRAINE IN PRIMARY CARE ADVISORS Updated guidelines on headache management for use by the pharmacist Introduction N U M B E R 2 1, O C T O B E R 2 0 1 2 Headache is a major public health problem,
Headaches and Kids. Jennifer Bickel, MD Assistant Professor of Neurology Co-Director of Headache Clinic Children s Mercy Hospital
 Headaches and Kids Jennifer Bickel, MD Assistant Professor of Neurology Co-Director of Headache Clinic Children s Mercy Hospital Overview Headache classifications and diagnosis Address common headache
Headaches and Kids Jennifer Bickel, MD Assistant Professor of Neurology Co-Director of Headache Clinic Children s Mercy Hospital Overview Headache classifications and diagnosis Address common headache
Sponsor Novartis Pharmaceuticals
 Clinical Trial Results Database Page 1 Sponsor Novartis Pharmaceuticals Generic Drug Name Indacaterol Therapeutic Area of Trial Chronic Obstructive Pulmonary Disease (COPD) Indication studied: COPD Study
Clinical Trial Results Database Page 1 Sponsor Novartis Pharmaceuticals Generic Drug Name Indacaterol Therapeutic Area of Trial Chronic Obstructive Pulmonary Disease (COPD) Indication studied: COPD Study
Clinical Study Synopsis
 Clinical Study Synopsis This file is posted on the Bayer HealthCare Clinical Trials Registry and Results website. It is provided for patients and healthcare professionals to increase the transparency of
Clinical Study Synopsis This file is posted on the Bayer HealthCare Clinical Trials Registry and Results website. It is provided for patients and healthcare professionals to increase the transparency of
Alcohol Withdrawal. Introduction. Blood Alcohol Concentration. DSM-IV Criteria/Alcohol Abuse. Pharmacologic Effects of Alcohol
 Pharmacologic Effects of Alcohol Alcohol Withdrawal Kristi Theobald, Pharm.D., BCPS Therapeutics III Fall 2003 Inhibits glutamate receptor function (NMDA receptor) Inhibits excitatory neurotransmission
Pharmacologic Effects of Alcohol Alcohol Withdrawal Kristi Theobald, Pharm.D., BCPS Therapeutics III Fall 2003 Inhibits glutamate receptor function (NMDA receptor) Inhibits excitatory neurotransmission
Guidelines for trials of drug treatments in tension-type headache
 Guidelines for trials of drug treatments in tension-type headache First Edition: International Headache Society Committee on Clinical Trials in Tension-type Headache COMMITTEE MEMBERS J SCHOENEN, Belgium
Guidelines for trials of drug treatments in tension-type headache First Edition: International Headache Society Committee on Clinical Trials in Tension-type Headache COMMITTEE MEMBERS J SCHOENEN, Belgium
EFFIMET 1000 XR Metformin Hydrochloride extended release tablet
 BRAND NAME: Effimet XR. THERAPEUTIC CATEGORY: Anti-Diabetic PHARMACOLOGIC CLASS: Biguanides EFFIMET 1000 XR Metformin Hydrochloride extended release tablet COMPOSITION AND PRESENTATION Composition Each
BRAND NAME: Effimet XR. THERAPEUTIC CATEGORY: Anti-Diabetic PHARMACOLOGIC CLASS: Biguanides EFFIMET 1000 XR Metformin Hydrochloride extended release tablet COMPOSITION AND PRESENTATION Composition Each
Emergency and inpatient treatment of migraine: An American Headache Society
 Emergency and inpatient treatment of migraine: An American Headache Society survey. The objective of this study was to determine the practice preferences of AHS members for acute migraine treatment in
Emergency and inpatient treatment of migraine: An American Headache Society survey. The objective of this study was to determine the practice preferences of AHS members for acute migraine treatment in
Teriflunomide is the active metabolite of Leflunomide, a drug employed since 1994 for the treatment of rheumatoid arthritis (Baselt, 2011).
 Page 1 of 10 ANALYTE NAME AND STRUCTURE TERIFLUNOMIDE Teriflunomide TRADE NAME Aubagio CATEGORY Antimetabolite TEST CODE PURPOSE Therapeutic Drug Monitoring GENERAL RELEVANCY BACKGROUND sclerosis. The
Page 1 of 10 ANALYTE NAME AND STRUCTURE TERIFLUNOMIDE Teriflunomide TRADE NAME Aubagio CATEGORY Antimetabolite TEST CODE PURPOSE Therapeutic Drug Monitoring GENERAL RELEVANCY BACKGROUND sclerosis. The
Metabolic Syndrome with Prediabetic Factors Clinical Study Summary Concerning the Efficacy of the GC Control Natural Blood Sugar Support Supplement
 CLINICALLY T E S T E D Natural Blood Sugar Metabolic Syndrome with Prediabetic Factors Clinical Study Summary Concerning the Efficacy of the GC Control Natural Blood Sugar Metabolic Syndrome with Prediabetic
CLINICALLY T E S T E D Natural Blood Sugar Metabolic Syndrome with Prediabetic Factors Clinical Study Summary Concerning the Efficacy of the GC Control Natural Blood Sugar Metabolic Syndrome with Prediabetic
Omega-3 fatty acids improve the diagnosis-related clinical outcome. Critical Care Medicine April 2006;34(4):972-9
 Omega-3 fatty acids improve the diagnosis-related clinical outcome 1 Critical Care Medicine April 2006;34(4):972-9 Volume 34(4), April 2006, pp 972-979 Heller, Axel R. MD, PhD; Rössler, Susann; Litz, Rainer
Omega-3 fatty acids improve the diagnosis-related clinical outcome 1 Critical Care Medicine April 2006;34(4):972-9 Volume 34(4), April 2006, pp 972-979 Heller, Axel R. MD, PhD; Rössler, Susann; Litz, Rainer
Antipsychotic drugs are the cornerstone of treatment
 Article Effectiveness of Olanzapine, Quetiapine, Risperidone, and Ziprasidone in Patients With Chronic Schizophrenia Following Discontinuation of a Previous Atypical Antipsychotic T. Scott Stroup, M.D.,
Article Effectiveness of Olanzapine, Quetiapine, Risperidone, and Ziprasidone in Patients With Chronic Schizophrenia Following Discontinuation of a Previous Atypical Antipsychotic T. Scott Stroup, M.D.,
Ultram (tramadol), Ultram ER (tramadol extended-release tablets); Conzip (tramadol extended-release capsules), Ultracet (tramadol / acetaminophen)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.02.35 Subject: Tramadol Acetaminophen Page: 1 of 8 Last Review Date: September 18, 2015 Tramadol Acetaminophen
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.02.35 Subject: Tramadol Acetaminophen Page: 1 of 8 Last Review Date: September 18, 2015 Tramadol Acetaminophen
Examination Content Blueprint
 Examination Content Blueprint Overview The material on NCCPA s certification and recertification exams can be organized in two dimensions: (1) organ systems and the diseases, disorders and medical assessments
Examination Content Blueprint Overview The material on NCCPA s certification and recertification exams can be organized in two dimensions: (1) organ systems and the diseases, disorders and medical assessments
Preetha selva et al. / International Journal of Phytopharmacology. 6(1), 2015, 42-46. International Journal of Phytopharmacology
 International Journal of Phytopharmacology Journal homepage: www.onlineijp.com 42 e- ISSN 0975 9328 Print ISSN 2229 7472 IJP A CLINICAL STUDY TO EVALUATE THE EFFECT OF TOPICAL TAZAROTENE IN THE TREATMENT
International Journal of Phytopharmacology Journal homepage: www.onlineijp.com 42 e- ISSN 0975 9328 Print ISSN 2229 7472 IJP A CLINICAL STUDY TO EVALUATE THE EFFECT OF TOPICAL TAZAROTENE IN THE TREATMENT
Tension-type headache Non-pharmacological and pharmacological treatment
 Danish Headache Center Tension-type headache Non-pharmacological and pharmacological treatment Lars Bendtsen Associate professor, MD, PhD, Dr Med Sci Danish Headache Center, Department of Neurology Glostrup
Danish Headache Center Tension-type headache Non-pharmacological and pharmacological treatment Lars Bendtsen Associate professor, MD, PhD, Dr Med Sci Danish Headache Center, Department of Neurology Glostrup
Clinical Trial Results Database Page 1
 Clinical Trial Results Database Page Sponsor Novartis Generic Drug Name BGT6 Therapeutic Area of Trial Advanced solid malignancies Approved Indication Investigational Study Number CBGT6A0 Title A phase
Clinical Trial Results Database Page Sponsor Novartis Generic Drug Name BGT6 Therapeutic Area of Trial Advanced solid malignancies Approved Indication Investigational Study Number CBGT6A0 Title A phase
Medical management of CHF: A New Class of Medication. Al Timothy, M.D. Cardiovascular Institute of the South
 Medical management of CHF: A New Class of Medication Al Timothy, M.D. Cardiovascular Institute of the South Disclosures Speakers Bureau for Amgen Background Chronic systolic congestive heart failure remains
Medical management of CHF: A New Class of Medication Al Timothy, M.D. Cardiovascular Institute of the South Disclosures Speakers Bureau for Amgen Background Chronic systolic congestive heart failure remains
The effects of caffeine on alertness: a randomized trial Morrocona MM, Smith A, Jones FH
 RESEARCH The effects of caffeine on alertness: a randomized trial Morrocona MM, Smith A, Jones FH TRIAL DESIGN A randomized controlled trial was employed in which participants were randomly allocated to
RESEARCH The effects of caffeine on alertness: a randomized trial Morrocona MM, Smith A, Jones FH TRIAL DESIGN A randomized controlled trial was employed in which participants were randomly allocated to
New York State Office of Alcoholism & Substance Abuse Services Addiction Services for Prevention, Treatment, Recovery
 New York State Office of Alcoholism & Substance Abuse Services Addiction Services for Prevention, Treatment, Recovery USING THE 48 HOUR OBSERVATION BED USING THE 48 HOUR OBSERVATION BED Detoxification
New York State Office of Alcoholism & Substance Abuse Services Addiction Services for Prevention, Treatment, Recovery USING THE 48 HOUR OBSERVATION BED USING THE 48 HOUR OBSERVATION BED Detoxification
Naltrexone and Alcoholism Treatment Test
 Naltrexone and Alcoholism Treatment Test Following your reading of the course material found in TIP No. 28. Please read the following statements and indicate the correct answer on the answer sheet. A score
Naltrexone and Alcoholism Treatment Test Following your reading of the course material found in TIP No. 28. Please read the following statements and indicate the correct answer on the answer sheet. A score
The submission positioned dimethyl fumarate as a first-line treatment option.
 Product: Dimethyl Fumarate, capsules, 120 mg and 240 mg, Tecfidera Sponsor: Biogen Idec Australia Pty Ltd Date of PBAC Consideration: July 2013 1. Purpose of Application The major submission sought an
Product: Dimethyl Fumarate, capsules, 120 mg and 240 mg, Tecfidera Sponsor: Biogen Idec Australia Pty Ltd Date of PBAC Consideration: July 2013 1. Purpose of Application The major submission sought an
Issues Regarding Use of Placebo in MS Drug Trials. Peter Scott Chin, MD Novartis Pharmaceuticals Corporation
 Issues Regarding Use of Placebo in MS Drug Trials Peter Scott Chin, MD Novartis Pharmaceuticals Corporation Context of the Guidance The draft EMA Guidance mentions placebo as a comparator for superiority
Issues Regarding Use of Placebo in MS Drug Trials Peter Scott Chin, MD Novartis Pharmaceuticals Corporation Context of the Guidance The draft EMA Guidance mentions placebo as a comparator for superiority
Phase: IV. Study Period: 20 Jan. 2006-17 Sep. 2008
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP)
 The European Agency for the Evaluation of Medicinal Products Evaluation of Medicines for Human Use London, 19 March 2003 CPMP/EWP/785/97 COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP) POINTS TO CONSIDER
The European Agency for the Evaluation of Medicinal Products Evaluation of Medicines for Human Use London, 19 March 2003 CPMP/EWP/785/97 COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP) POINTS TO CONSIDER
Clinical Algorithm & Preferred Medications to Treat Pain in Dialysis Patients
 Clinical Algorithm & Preferred Medications to Treat Pain in Dialysis Patients Developed by the Mid Atlantic Renal Coalition and the Kidney End of Life Coalition September 2009 This project was supported,
Clinical Algorithm & Preferred Medications to Treat Pain in Dialysis Patients Developed by the Mid Atlantic Renal Coalition and the Kidney End of Life Coalition September 2009 This project was supported,
GUIDELINES FOR TUBERCULOSIS PREVENTIVE THERAPY AMONG HIV INFECTED INDIVIDUALS IN SOUTH AFRICA
 GUIDELINES FOR TUBERCULOSIS PREVENTIVE THERAPY AMONG HIV INFECTED INDIVIDUALS IN SOUTH AFRICA 2010 1 TB prophylaxis GUIDELINES FOR TUBERCULOSIS PREVENTIVE THERAPY AMONG HIV INFECTED INDIVIDUALS Background
GUIDELINES FOR TUBERCULOSIS PREVENTIVE THERAPY AMONG HIV INFECTED INDIVIDUALS IN SOUTH AFRICA 2010 1 TB prophylaxis GUIDELINES FOR TUBERCULOSIS PREVENTIVE THERAPY AMONG HIV INFECTED INDIVIDUALS Background
Human Clinical Study for Free Testosterone & Muscle Mass Boosting
 Human Clinical Study for Free Testosterone & Muscle Mass Boosting GE Nutrients, Inc. 920 E. Orangethorpe Avenue, Suite B Anaheim, California 92801, USA Phone: +1-714-870-8723 Fax: +1-732-875-0306 Contact
Human Clinical Study for Free Testosterone & Muscle Mass Boosting GE Nutrients, Inc. 920 E. Orangethorpe Avenue, Suite B Anaheim, California 92801, USA Phone: +1-714-870-8723 Fax: +1-732-875-0306 Contact
Abdominal Migraine: An Under-Diagnosed Cause of Recurrent Abdominal Pain in Childrenhead_1855 707..712
 Headache 2011 American Headache Society ISSN 0017-8748 doi: 10.1111/j.1526-4610.2011.01855.x Published by Wiley Periodicals, Inc. Research Submission Abdominal Migraine: An Under-Diagnosed Cause of Recurrent
Headache 2011 American Headache Society ISSN 0017-8748 doi: 10.1111/j.1526-4610.2011.01855.x Published by Wiley Periodicals, Inc. Research Submission Abdominal Migraine: An Under-Diagnosed Cause of Recurrent
Frequently Asked Questions (FAQs)
 Frequently Asked Questions (FAQs) Research Rationale 1. What does PrEP stand for? There is scientific evidence that antiretroviral (anti-hiv) medications may be able to play an important role in reducing
Frequently Asked Questions (FAQs) Research Rationale 1. What does PrEP stand for? There is scientific evidence that antiretroviral (anti-hiv) medications may be able to play an important role in reducing
Proposal for deletion Codeine phosphate tablets for pain in children
 Introduction Proposal for deletion Codeine phosphate tablets for pain in children Codeine is a phenanthrene opioid derivative. It is listed in the 2010 WHO Model List of Essential Medicines for Children
Introduction Proposal for deletion Codeine phosphate tablets for pain in children Codeine is a phenanthrene opioid derivative. It is listed in the 2010 WHO Model List of Essential Medicines for Children
Journal Club: Niacin in Patients with Low HDL Cholesterol Levels Receiving Intensive Statin Therapy by the AIM-HIGH Investigators
 Journal Club: Niacin in Patients with Low HDL Cholesterol Levels Receiving Intensive Statin Therapy by the AIM-HIGH Investigators Shaikha Al Naimi Doctor of Pharmacy Student College of Pharmacy Qatar University
Journal Club: Niacin in Patients with Low HDL Cholesterol Levels Receiving Intensive Statin Therapy by the AIM-HIGH Investigators Shaikha Al Naimi Doctor of Pharmacy Student College of Pharmacy Qatar University
5.07.09. Aubagio. Aubagio (teriflunomide) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.07.09 Subject: Aubagio Page: 1 of 6 Last Review Date: December 5, 2014 Aubagio Description Aubagio (teriflunomide)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.07.09 Subject: Aubagio Page: 1 of 6 Last Review Date: December 5, 2014 Aubagio Description Aubagio (teriflunomide)
33 % of whiplash patients develop. headaches originating from the upper. cervical spine
 33 % of whiplash patients develop headaches originating from the upper cervical spine - Dr Nikolai Bogduk Spine, 1995 1 Physical Treatments for Headache: A Structured Review Headache: The Journal of Head
33 % of whiplash patients develop headaches originating from the upper cervical spine - Dr Nikolai Bogduk Spine, 1995 1 Physical Treatments for Headache: A Structured Review Headache: The Journal of Head
