APPLIED NUTRITIONAL INVESTIGATIONS INTRODUCTION
|
|
|
- Madeline Horn
- 10 years ago
- Views:
Transcription
1 APPLIED NUTRITIONAL INVESTIGATIONS Does Enteral Nutrition Compared to Parenteral Nutrition Result in Better Outcomes in Critically Ill Adult Patients? A Systematic Review of the Literature Leah Gramlich, MD, Krikor Kichian, MD, Jaime Pinilla, MD, Nadia J. Rodych, RD, Rupinder Dhaliwal, RD, and Daren K. Heyland, MD, MSc From the Department of Medicine, University of Alberta, Edmonton, Alberta, Canada; the Department of Surgery, Royal University Hospital, Saskatoon, Saskatchewan, Canada; and the Department of Medicine, Queens University, Kingston, Ontario, Canada OBJECTIVE: Nutritional support is part of the standard of care for the critically ill adult patient. In the average patient in the intensive care unit who has no contraindications to enteral nutrition (EN) or parenteral nutrition (PN), the choice of route for nutritional support may be influenced by several factors. Because EN and PN are associated with risks and benefits, we systematically reviewed and critically appraised the literature to compare EN with PN the critically ill patient. METHODS: We searched computerized bibliographic databases, personal files, and relevant reference lists to identify potentially eligible studies. Only randomized clinical trials that compared EN with PN in critically ill patients with respect to clinically important outcomes were included in this review. In an independent fashion, relevant data on the methodology and outcomes of primary studies were abstracted in duplicate. The studies were subsequently aggregated statistically. RESULTS: There were 13 studies that met the inclusion criteria and, hence, were included in our meta-analysis. The use of EN as opposed to PN was associated with a significant decrease in infectious complications (relative risk 0.64, 95% confidence interval 0.47 to 0.87, P 0.004) but not with any difference in mortality rate (relative risk 1.08, 95% confidence interval 0.70 to 1.65, P 0.7). There was no difference in the number of days on a ventilator or length of stay in the hospital between groups receiving EN or PN (Standardized Mean Difference [SMD] 0.07, 95% confidence interval 0.2 to 0.33, P 0.6). PN was associated with a higher incidence of hyperglycemia. Data that compared days on a ventilator and the development of diarrhea in patients who received EN versus PN were inconclusive. In the EN and PN groups, complications with enteral and parenteral access were seen. Four studies documented cost savings with EN as opposed to PN. CONCLUSION: The use of EN as opposed to PN results in an important decrease in the incidence of infectious complications in the critically ill and may be less costly. EN should be the first choice for nutritional support in the critically ill. Nutrition 2004;20: Elsevier Inc KEY WORDS: enteral nutrition, parenteral nutrition, critical illness, meta-analysis INTRODUCTION In the critically ill patient, malnutrition results in impaired immunologic function, impaired ventilatory drive, and weakened respiratory muscles leading to prolonged ventilator dependence and increased infectious morbidity and mortality rates. 1 Malnutrition is prevalent in patients in the intensive care unit (ICU), and its prevalence has been reported to be as high as 40% and is associated with poor outcome. 2 Recent reviews have documented evidence that nutritional support influences morbidity and mortality rates in critically ill patients 3. Parenteral nutrition (PN) is used in Dr. Heyland is a Career Scientist for the Ontario Ministry of Health in Ontario, Canada. Correspondence to: Daren K. Heyland, MD, MSc, Angada 3, Kingston General Hospital, 76 Stuart Street, Kingston ON K7L 2V7, Canada. [email protected] Nutrition 20: , 2004 Elsevier Inc., Printed in the United States. All rights reserved. 12% to 71% and enteral nutrition (EN) is used in 33% to 92% of critically ill patients who receive nutritional support. 4 9 The general benefits of nutritional support include improved wound healing, 3 a decreased catabolic response to injury, 10 improved gastrointestinal permeability, 11 decreased bacterial translocation, 12 and improved clinical outcomes, including a decrease in complication rates and length of stay with accompanying cost savings However, nutritional support is not without adverse effects and risks. Early EN may be associated with high gastric residuals, 17 bacterial colonization of the stomach, and increased risk of aspiration pneumonia. 18 PN has been associated with gut mucosal atrophy, overfeeding, hyperglycemia, an increased risk of infectious complications 16 and increased mortality rates 19 in critically ill patients. Both forms of nutritional support can affect cost and workload. Various factors influence the choice of EN or PN, one of which is the estimate of treatment benefit and risk of harm. Braunschweig et al. 20 conducted a meta-analysis to review prospective, randomized, controlled trials that randomly assigned patients to EN or PN and in which PN was provided at or above /04/$30.00 doi: /j.nut
2 844 Gramlich et al. Nutrition Volume 20, Number 10, 2004 estimated needs. Studies included patients identified as having compromised gastrointestinal function (pancreatitis, ulcerative colitis, or Crohn s disease), surgery, trauma, or multisystem organ failure. When the results of the trials were aggregated, tube feeding was associated with a lower risk of infection but a higher risk of complications associated with nutritional support. The strength of this association was questioned because of a significant test for heterogeneity (P 0.03). There was no treatment effect for EN on other complications or mortality rate. Koretz et al., in the AGA Technical Review on Parenteral Nutrition 21, performed a global meta-analysis on 82 randomized, controlled trials to assess the clinical efficacy of PN, including perioperative trials, oncologic therapy trials, alcoholic hepatitis trials, and trials in low-birthweight infants. They also tried to distinguish whether or not malnutrition was present in patient populations. They identified that PN did not influence mortality or overall complication rates but that it was associated with an increased risk for infection. One of the limitations of these previous reviews is the heterogeneity of the patient populations included in the meta-analyses. The treatment effect of nutritional support differs depending on the population studied. Heyland et al. 19 compared PN with standard care (oral diet plus intravenous dextrose) in surgical and critically ill patients and found that, overall, PN did not influence overall mortality rate, but that there was a trend toward decreased complication rates in malnourished patients. They identified that study results were influenced by patient populations. Patients undergoing major surgery were more likely to achieve a positive outcome with PN, whereas critically ill patients were more likely to have a complication and die. The differences in treatment effect across these groups were statistically significant. This suggests that the results of studies in patients who are not critically ill are not generalizable to those who are. Because there have been several small clinical studies that have compared EN with PN in specific populations of patients with critical illness, we systematically reviewed and statistically aggregated all studies that compared EN with PN in the critically ill to allow a more precise estimation of the treatment effect and to increase the power to identify a treatment effect that may not be apparent in smaller individual studies. MATERIALS AND METHODS Search Strategy We conducted a computerized bibliographic search of Medline, Embase, Cinahl, and Cochrane Library for studies from 1980 to August 2002 to locate all relevant articles. Search terms included nutritional support, dietary supplementation, enteral nutrition, parenteral nutrition, peripheral nutrition, total parenteral nutrition, nutritional support team, nutrition requirements, nutritional assessment, parenteral nutrition solutions, critical care, critical illness, and intensive care units. In addition, personal files and relevant review articles were searched for additional studies. Study Selection Criteria Studies were selected for inclusion in the review process it they met the following criteria: 1. Study design: randomized clinical trial and meta-analysis of randomized controlled trials (pseudo-randomized trials were excluded). 2. Population: critically ill, human adult patients (patients who underwent elective surgery were excluded). 3. Intervention: any form of EN or PN. 4. Outcomes: primary outcomes of interest were mortality rate (ICU, hospital, or long term) and infectious complications. Secondary endpoints included length of stay, quality of life, functional recovery, complications, and cost. We elected to include only randomized trials in this review. The trials evaluated the effect of PN administered at or above estimated energy needs compared with the effect of EN in the critically ill. We defined critically ill patients as those who would be routinely cared for in the critical care environment. We excluded studies of pediatric or neonatal patients. Studies were not limited to those that involved English-speaking adult patients. Studies that evaluated the effect of PN or EN on nutritional outcomes (i.e., nitrogen balance, amino acid profile) were not included in this review. Methodologic Quality of Primary Studies Each randomized trial was critically appraised according to an explicit procedure. The two appraisers (L.G. and J.P.) appraised the following descriptors: intervention, study population, nature of allocation, co-intervention, exclusion after randomization, double blinding, event rates, relative risk (RR), and other outcomes. Clinical trials were assigned level 1 if they reported information on concealed randomization, blinded outcome adjudication, and an intent-to-treat analysis. Trials were assigned level 2 if any one of those characteristics was unfulfilled. For the one meta-analysis included in the review process, the following descriptors were abstracted: intervention, number of trials, population selection criteria, search strategy, independent validity assessment, method of pooling results, assessment of homogeneity, pooled event rates, and other outcomes. Disagreement between appraisers was resolved by consensus. When data were missing, unclear, or not reported on a per-patient basis, we attempted to contact the primary investigator and request further information. One investigator 27 provided data on a subset of critically ill patients who were randomized to received EN or PN, and these data were included in the analysis. A priori, we considered that the harmful effect of PN might be associated with relative overfeeding and hyperglycemia. Accordingly, we conducted a subgroup analysis to determine the effect of excess calories (PN versus EN) and higher glucose levels (across groups). The primary outcomes were mortality rate and infectious complications. Data from all relevant studies were combined to estimate the common risk ratio and associated 95% confidence intervals (95% CIs). The common risk ratios and their confidence intervals were estimated by using the random effect model of DerSimonian and Laird 35 as implemented in RevMan We considered P 0.25 to be supportive of a trend and P 0.05 to be statistically significant. A test for heterogeneity was considered significant if P 0.05, indicating heterogeneity among studies, thereby weakening the estimate of overall treatment. RESULTS Study Identification and Selection Twenty-seven citations of randomized controlled trials were identified in the bibliographic search, our personal files, and review of references. Of these studies, there were 12 level 2 studies 5,11,16,22 26 and one level 1 study 27 that met the inclusion criteria and described a total of 856 critically ill patients. The 13 studies are presented in Table I, which lists study populations, designs, interventions, and outcomes (mortality rate, infections, length of stay in the ICU, days on a ventilator, and cost). Reasons for exclusions were studies 20,33,34,37 48 that examined a mix of patients who and were not critically ill or that examined patients who underwent elective surgery. The 13 studies included reflected a heterogeneous population of ICU patients who had head trauma and injuries, abdominal trauma, sepsis, cardiac bypass, or severe acute pancreatitis. In the study by Woodcock et al., 27 we abstracted data concerning only ICU patients, and 11 of 38 patients moved between groups after randomization. The data on mortality rate
3 Nutrition Volume 20, Number 10, 2004 Enteral Versus Parenteral Nutrition in Critically Ill Patients 845 TABLE I. RANDOMIZED CLINICAL TRIALS THAT COMPARED EN WITH PN IN CRITICALLY ILL PATIENTS Subjects Infection Mortality LOS VD References EN PN Population EN PN EN PN EN PN EN PN Trauma and laparotomy 15 (65) 17 (74) 1 (4) 3 (13) Closed-head injury N/A N/A 5 (18) 1 (5) N/A N/A Post sepsis N/A N/A 7 (22) 8 (23) N/A N/A N/A N/A Blunt trauma N/A N/A 1 (7) 1 (8) N/A N/A N/A N/A Cardiac bypass N/A N/A 2 (15) 6 (55) N/A N/A N/A N/A Head trauma 17 (80) 15 (63) 3 (14) 2 (8) N/A N/A N/A N/A Acute pancreatitis 5 (28) 10 (50) 1 (6) 2 (10) Abdominal trauma 9 (16) 18 (40) Abdominal trauma 5 (17) 11 (37) 0 0 N/A N/A N/A N/A High-risk surgical 19 (16) 39 (35) 8 (7) 11 (10) N/A N/A Head injury N/A N/A 9 (50) 3 (15) Malnutrition 6 (38) 11 (52) 9 (53) 5 (24) N/A N/A Brain injury 5 (18) 4 (17) 10 (36) 10 (43) N/A N/A N/A N/A EN, enteral nutrition; N/A, not available; PN, parenteral nutrition and infectious complications from the 1989 study by Moore et al. 26 were included in their 1992 meta-analysis, 25 whereas data on caloric intake, blood sugars, and non-septic complications were not and, hence, appeared in the tables of the 1989 study. 26 Effect of EN Versus PN on Clinical Outcomes Nine of the 13 studies reported data on infectious complications with EN versus PN. The nature of the infectious complications varied with the particular patient population and included pneumonia, aspiration pneumonia, urinary tract infections, bacteremia, wound infection, abdominal abscess, and line sepsis. When the data were aggregated from these studies (Figure 1), there was a significant decrease in the number of patients with infectious complications who had received EN rather than PN (RR 0.64, 95% CI 0.47 to 0.87). The test for heterogeneity of this aggregate was not statistically significant (P 0.22). All 13 studies reported mortality rate as an outcome. The result of this analysis (Figure 2) demonstrated no difference in mortality rate in critically ill patients on EN versus PN (RR 1.08, 95% CI 0.70 to 1.65), with a non-significant test for heterogeneity of 0.2. When a subgroup of studies in which the PN group was fed more calories than the EN group (non-isocaloric dosing across groups) were aggregated 16,26,27,30,31 (Figure 3), EN was associated with a trend toward an excessive mortality rate (RR 1.58, 95% CI 0.75 to 3.35, P 0.2) compared with PN. When the trials in which EN and PN were fed isocalorically were aggregated, there was no effect between EN and PN (RR 1.08, 95% CI 0.56 to 2.06, P 0.8). Mortality rate in the subgroup analysis that compared patients who received PN and had higher levels of blood sugar with those who received EN showed no effect (RR.093, 95% CI.021 to 4.15, P 0.90) when compared with studies in which patients levels of blood sugar were similar across groups. There was no difference in length of stay 16,22,25,27,29,30,32 or days on ventilation 16,29,30,32 between groups receiving EN or PN, but the information was not aggregated statistically due to insufficient data. Only three studies reported on baseline nutritional status, and data regarding the relation of nutritional status to outcome were not available. Of the studies that reported on nutritional intake, 5 of 11 associated PN with a larger caloric intake. 16,25,27,30,31 Three studies associated EN with an increase in diarrhea, 16,23,31 and one reported decreased diarrhea. 22 Four studies reported cost savings with the use of EN rather than PN. 22,23,29,32 FIG. 1. EN is associated with fewer infectious complications than is PN (RR 0.64, P 0.004). 95% CI, 95% confidence interval; EN, enteral nutrition; PN parenteral nutrition; RR, relative risk.
4 846 Gramlich et al. Nutrition Volume 20, Number 10, 2004 FIG. 2. EN does not differ from PN with respect to mortality rate (RR 1.08, P 0.7). 95% CI, 95% confidence interval; EN, enteral nutrition; PN parenteral nutrition; RR, relative risk. DISCUSSION The body of literature regarding nutritional support in critically ill patients continues to grow, but, because of methodic limitations and the small size of many of the studies, making inferences and generalizing results from individual trials are problematic. Because the treatment effects of EN and PN vary depending on the patient population, in contrast to previous reviews, we systematically examined all randomized trials that compared EN with PN specifically in critically ill patients. The data, when aggregated, demonstrated that patients on EN developed fewer infectious complications. Further, neither EN nor PN was associated with a survival advantage. Complications were seen with both forms of therapy. Although a meta-analysis does not replace a large, multicenter, randomized, controlled trial that compares EN with PN in the critically ill patient, it does provide useful information and can guide us in the development of such a trial to specifically assess treatment effects of EN versus PN. This would also require a change in how the nutrition community performs such studies, so that larger multicenter, randomized, controlled trials could be performed in this patient population. Among the limitations, we acknowledge the heterogeneity in the formulations and amount of energy provided by nutritional support in patients receiving EN and PN. We also recognize the difficulty of conducting studies in severely ill patients who often have an unpredictable course in the ICU and determining the effect of that course on outcome. Other important considerations for inclusion in future studies investigating nutritional support in critical illness would be a quantifiable assessment of disease severity (Injury Severity Score and Second Acute Physiology and Chronic Health Evaluation) and baseline nutritional status. The main clinical implication of our data concerns the use of nutritional support in critically ill patients who can tolerate some EN. Our findings suggest that EN is the preferred method to provide nutritional support to critically ill patients. Although we did not find any difference in mortality rate between patients administered EN and PN, the meta-analysis lacked power to detect a small but meaningful treatment effect. Moreover, a difference in infectious complications alone warrants a preferential recommendation of EN. Acquired infection, in particular ventilatorassociated pneumonia, is a major problem for critically ill patients, which results in increased morbidity and mortality rates and health care costs Perceived barriers to using EN for nutritional support include concerns over the risk of aspiration pneumonia, high gastric residuals and bowel irregularities, and an inability to reach targeted nutritional goal rates. In those patients on pressors, there is the added concern of the potential to increase the oxygen demand of the gastrointestinal tract in those who are fed with EN. 52 Recent guidelines that address decreasing the risks and maximizing the benefits of EN have been published. 53 These guidelines include an evidence-based evaluation of nutritional support in the FIG. 3. EN in non-isocaloric studies (in which the PN group received more calories than the EN group) is associated with a trend toward an excessive mortality rate. 95% CI, 95% confidence interval; EN, enteral nutrition; PN parenteral nutrition; RR, relative risk.
5 Nutrition Volume 20, Number 10, 2004 Enteral Versus Parenteral Nutrition in Critically Ill Patients 847 ventilated, critically ill patient and review EN versus PN compositions of nutritional support. Several measures have been shown to decrease the risk of aspiration pneumonia in critically ill patients on EN Gastrointestinal promorbidity agents in the ICU have been systematically reviewed recently; 58 although no study demonstrated a positive effect on clinical outcomes, promorbidity agents as a class appear to increase indexes of gastrointestinal transit and tolerance of feeding. Small bowel feeding, beyond the pylorus, may also be associated with a decrease in gastroesophageal regurgitation, an increase in nutrient delivery, and a lower rate of ventilator-associated pneumonia. 59 The use of an EN feeding protocol with a gastric residual threshold volume of 250 ml may also positively affect tolerance of tube feeds and achievement of goal rates. 60 Why is there an increased risk of infection associated with PN in the critically ill patient? Although perhaps controversial, 16,61 the adverse effects of PN have been attributed to hyperglycemia and subsequent increased infectious complications. 61 This attribution has been supported by Van den berghe et al. 62 who reported that intensive insulin therapy and tight control (glucose 4.4 to 6.1 mm/l) decrease morbidity and mortality rates in critically ill patients. All patients in this study received 200 to 300 g of glucose on day 1 and 60% went on to receive PN. An alternative explanation of this study s findings is that high glucose loading, as one would see with PN, with inadequate glycemic control is associated with increased morbidity and mortality rates. In our subgroup analysis, we found no difference in treatment effect between those studies in which the PN groups received more calories or had a higher incidence of hyperglycemia. McCowan et al. 61 compared hypocaloric PN (1000 kcal, 70 g of protein, and no lipid) with standard PN (25 kcal kg 1 d 1 with lipid, 1.5 g/kg) and found a trend toward fewer infections (P 0.2) in the hypocalorically fed group. Interestingly, the incidence of hyperglycemia in both groups was similar. Another hypothesis, not proved in human subjects, is that bacterial translocation in the setting of gut atrophy, secondary to its disuse with PN, is responsible for the increased risk of infection seen with PN. 63 There will be critically ill patients in whom EN is not possible, such as patients with bowel obstruction, short bowel syndrome, or abdominal compartment syndrome or those who could not tolerate EN over a prolonged period 64 and who may be at increased risk for mortality and morbidity. As such, it is imperative to consider strategies to optimize PN, 53 which would include optimization of glycemic control. 62 PN without lipid has been associated with fewer infections. 61,65 The addition of parenteral glutamine to PN may also be associated with decreased complication and mortality rates Although the combination of EN and PN does not confer a significant advantage over PN alone to satisfy patients needs, 53 it is reasonable to continue attempts at EN in patients who require PN. In conclusion, when EN and PN are compared in the critically ill patient, EN is associated with fewer infectious complications and, if possible, should be the chosen route for nutritional support. It is fundamental that, in the provision of EN and PN, strategies be adopted to optimize benefit and minimize potential harm. REFERENCES 1. Dark SK, Pingleton SK. Nutrition and nutritional support in critically ill patients. J Intensive Care Med 1993;8:16 2. Giner M, et al. In. a correlation between malnutrition and poor outcome in critically ill patients still exists. Nutrition 1996;1995:12:23 3. Shroeder D, et al. Effects of immediate postoperative enteral nutrition on body composition, muscle function, and wound healing. JPEN 1981;15: Payne JC, et al. Artificial nutrition support in hospitals inn the United Kingdom 1991: second national survey. Clin Nutr 1992;11: Hill SA, et al. Nutrition support in intensive care units in England and Wales: a survey. Eur J Clin Nutr 1995;49: De Jonghe B, et al. A prospective survey of nutritional support practices in intensive care units patients: what is prescribed? what is delivered? Crit Care Med 2001;29:8 7. Preiser JC, et al. Management of nutrition in European intensive care units: results of a questionnaire. Intensive Care Med 1999;25:95 8. Lipman TO. Grains or veins: is enteral nutrition really better then parenteral nutrition? A look at the evidence. JPEN 1998;22: Heyland D, Schroter-Noppe D, Drover JW, Jain M, Keefe L, Dhaliwal R, et al. Nutrition support in the critical care setting: current practice in Canadian ICUs opportunities for improvement? JPEN 2003;27: Mochizuki H, et al. Mechanism of prevention of postburn hypermetabolism and catabolism by early enteral feeding. Ann Surg 1984;200: Hadfield RJ, et al. Effects of enteral and parenteral nutrition on gut mucosal permeability in the critically ill. Am J Respir Crit Care Med 1995;152: Gianotti L, et al. Role of early enteral feeding and acute starvation on postburn bacterial translocation and host defence: prospective, randomized trials. Crit Care Med ;22: Carr C, et al. Randomized trial of safety and efficacy of immediate postoperative enteral feeding in patients undergoing gastrointestinal resection. BMJ 1996;312: Taylor SJ, et al. Prospective, randomized controlled trial to determine the effect of early enteral nutrition on clinical outcome in mechanically ventilated patients suffering head injury. Crit Care Med 1999;27: Delmi M, Rapin CH, Bengoa JM, et al. Dietary supplementation in elderly patients with fractured neck of the femur. Lancet 1990;335: Kudsk KA, et al. Enteral versus parenteral feeding: effects on septic morbidity after blunt and penetrating abdominal trauma. Ann Surg 1992;215: Heyland D, et al. How well do critically ill patients tolerate early intragastric enteral feeding? Results of a prospective, multicenter trial. NCP 1999;14: Rello J, et al. Incidence, etiology and outcome of nosocomial pneumonia in mechanically ventilated patients. Chest 1991;100: Heyland DK, Macdonald S, Keefe L, Drover JW. Total parenteral nutrition in the critically ill patient: a meta analysis. JAMA 1998;280: Braunschweig CL, et al. Enteral compared with parenteral nutrition: a meta analysis. Am J Clin Nutr 2001;74: Koretz RL, Lipman TO, Klein S. AGA technical review on parenteral nutrition. Gastroenterology 2001;121: Borzotta AP, Pennings J, Papasadero B, et al. Enteral versus parenteral nutrition after severe closed head injury. J Trauma 1994;37: Cerra FB, McPherson JP, Konstantinides FN, Konstantinides NN, Teasley KM. Enteral nutrition does not prevent multiple organ failure syndrome (MOFS) after sepsis. Surgery 1988;104: Hadley MN, Grahm TW, Harrington T. Nutritional support and neurotrauma: a critical review of early nutrition in forty-five acute head injury patients. Neurosurgery 1986;19: Moore FA, Feliciano DV, Andrassy RJ, et al. Early enteral feeding, compared with parenteral, reduces postoperative septic complications: the results of a meta-analysis. Ann Surg 1992;216: Moore FA, Moore EE, Jones TN, McCroskey BL, Peterson VM. TEN versus TPN following major abdominal trauma reduced septic morbidity. J Trauma 1989;29: Woodcock N, Ziegler D, Palmer M, et al. Enteral versus parenteral nutrition: a pragmatic study. Nutrition 2001;17:1 28. Dunham CM, Frankenfield D, Belzberg H, Wiles C, Cushing B, Grant Z. Gut failure predictor or contributor to mortality in mechanically ventilated blunt trauma patients? J Trauma 1994;37: Adams S, Dellinger EP, Wertz MJ. Enteral versus parenteral nutritional support following laparotomy for trauma: a randomized prospective trial. J Trauma 1986;26: Rapp RP, Young DB, Twyman D. The favorable effect of early parenteral feeding on survival in head-injured patients. J Neurosurg 1983;58: Young B, Ott L, Haack D. Effect of total parenteral nutrition upon intracranial pressure in severe head injury. J Neurol 1987;67: Kalfarentzos F, Kehiagias J, Mead N, Kokkinis K, Gogos CA. Enteral nutrition is superior to parenteral nutrition in severe acute pancreatitis: results of a randomized prospective trial. Br J Surg 1997;84: Suchner U, Senftleben U, Eckart T, et al. Enteral versus parenteral nutrition: effects on gastrointestinal function and metabolism. Nutrition 1996;12: Huang YC, Yen CE, Cheng CH, Jih KS, Kan MN. Nutritional status of mechanically ventilated critically ill patients: comparison of different types of nutritional support. Clin Nutr 2002;19: DerSimonian R, Laird N. Meta-analysis in clinical trials. Control Clin Trials 1986;7: RevMan 4.1 user s guide. The Cochrane collaboration; Seri S, Aquilio E. Effects of early nutritional support in patients with abdominal trauma. Ital J Surg Sci 1984;14:223
6 848 Gramlich et al. Nutrition Volume 20, Number 10, Hamaoui E, Lefkowitz R, Olender L, et al. Enteral nutrition in the early postoperative period: a new semi-elemental formula versus total parenteral nutrition. JPEN 1990;14: Bower RH, Talamini MA, Sax HC. Postoperative enteral vs parenteral nutrition: a randomized controlled trial. Arch Surg 1986;121: Pacelli F, Bossola M, Papa V, et al. Enteral vs parenteral nutrition after major abdominal surgery: an even match. Arch Surg 2001;136: Braga M, Gianotti L, Gentilini O, Parisi V, Salis C, Di CV. Early postoperative enteral nutrition improves gut oxygenation and reduces costs compared with total parenteral nutrition. Crit Care Med 2001;29: Bozzetti F, Braga M, Gianotti L, Gavazz C, Mariani L. Postoperative enteral versus parenteral nutrition in malnourished patients with gastrointestinal cancer: a randomized multicentre trial. Lancet 2001;358: Von Meyenfeldt MF, Meijerink WJHJ, Rouflart MMJ, Buil-Maassen MTHJ, Soeters PB. Perioperative nutritional support: a randomized clinical trial. Clin Nutr 1992;11: Fletcher JP, Little JM. A comparison of parenteral nutrition and early postoperative enteral feeding on the nitrogen balance after major surgery. Surgery 1986; 100: Wicks C, Somasundaram S, Bjarnason I, et al. Comparison of enteral feeding and total parenteral nutrition after liver transplantation. Lancet 2002;344: Windsor ACJ, Kanwar S, Li AGK, et al. Compared with parenteral nutrition, enteral feeding attenuates the acute phase response and improves disease severity in acute pancreatitis. Gut 1998;42: Braga M, Gianotti L, Vignali A, Cestari A, Bisagni P, Di CV. Artificial nutrition after major abdominal surgery: impact of route of administration and composition of the diet. Crit Care Med 1998;26: Hernandez-Aranda JC, Gallo-Chico B, Flores-Ramirez LA, Avalos-Huante R, Magos-Vazquez FJ. Treatment of enterocutaneous fistula with or without octreotide and parenteral nutrition. Nutr Hosp 1996;11: Girou E, Stephan F, Novara A, Safar M, Fagon JY. Risk factors and outcome of nosocomial infections: results of a matched case-control study of ICU patients. Am J Respir Crit Care 1998;157: Bueno-Cavanillas A, Delgado-Rodriguez M, Lopez-Luque A, Schaffino-Can S, Galvez-Vargas R. Influence of nosocomial infection on morality rate in an intensive care unit. Crit Care Med 1994;22: Cook DJ, Griffith L, Keenan S, Brun-Buisson C. The attributable morbidity and mortality of ventilator-associated pneumonia in the critically ill patient. Am J Respir Crit Care Med 1999;159: Kles KA, Wallig MA, Tappenden KA. Luminal nutrients exacerbate intestinal hypoxia in the hypoperfused jejunum. JPEN 2001;5: Heyland D, Dhaliwal R, Drover J, et al. Canadian clinical practice guidelines for nutrition support in mechanically ventilated critically ill adult patient. JPEN 2003;27: Drakulovic MB, Torres A, Bauer TT, Nicolas JM, Nogue S, Ferrer M. Supine body position as a risk factor for nosocomial pneumonia in mechanically ventilated patients: a randomised trial. Lancet 1999;354: Kortbeek JB, Haigh PI, Doig C. Duodenal versus gastric feeding in ventilated blunt trauma patients: a randomized controlled trial. J Trauma 1999;46: Montecalvo MA, Steger KA, Farber HW, et al. Nutritional outcome and pneumonia in critical care patients randomized to gastric versus jejunal tube feedings. The Critical Care Research Team. Crit Care Med 1992;20: Davies AR, Froomes PR, French CJ, Bellomo R, Gutteridge GA, Nyulasi L, et al. Randomized comparison of nasojejunal and nasogastric feeding in critically ill patients. Crit Care Med 2002;30: Booth CM, Heyland D, Paterson WG, et al. Gastrointestinal promotility drugs in the critical care setting: a systematic review of the evidence. Crit Care Med 2002;30: Heyland D, Drover JW, Dhaliwal R, Greenwood J, et al. Optimizing the benefits and minimizing the risks of enteral nutrition in the critically ill: role of small bowel feeding. JPEN 2002;26(suppl):S Pinilla JC, Samphire J, Arnold C, Liu L, Thiessen B, et al. Comparison of gastrointestinal tolerance of two enteral feeding protocols in critically ill patients: a prospective, randomized controlled trial. JPEN 2001;25: McCowen KC, Friel C, Sternberg J, et al. Hypocaloric total parenteral nutrition: effectiveness in prevention of hyperglycemia and infectious complications. A randomized clinical trial. Crit. Care Med 2000;28: Van den Berghe G, Wouters P, Weekers F, Verwaest C, Bruyninckx F, Schetz M, et al. Intensive insulin therapy in critically ill patients. N Engl J Med 2001;344: Miura S, et al. Changes in intestinal absorption of nutrients and brush border glycoproteins after total parenteral nutrition in rats. Gut 1992;33: Sandstrom R, Drott C, Hyltander A, et al. The effect of postoperative intravenous feeding (TPN) on outcome following major surgery; evaluated in a randomized study. Ann Surg 1993;217: Basttistella FD, Widergren JT, Anderson JT, et al. A prospective, randomized trial of intravenous fat emulsion administration in trauma victims requiring total parenteral nutrition. J Trauma 1997;43: Griffiths RD, Jones C, Palmer TE. Six-month outcome of critically ill patients given glutamine-supplemented parenteral nutrition. Nutrition;13: Dechelotte P, Bleichner G, Hasselmann M, et al. Improved clinical outcome in ICU patients receiving alanyl-glutamine (Dipeptiven) supplemented total parenteral nutrition (abstract). Clin Nutr 2002;21:S1 68. Wischmeyer P, Lynch J, Leidel J, et al. Glutamine administration reduces gram-negative bacteremia in severely burned patients: a prospective, randomized, double-blind trial versus isonitrogenous control. Crit Care Med 2001;29: Powell-Tuck J, Jamieson CP, Bettany GEA, et al. A double blind randomised controlled trial of glutamine supplementation in parenteral nutrition. Gut 1999; 45: Goeters C, Wenn A, Mertes N, et al. Parenteral L-alanyl-L-glutamine improves six month outcome in critically ill patients. Crit Care Med 2002;30: Novak F, Heyland D, Avenell A, Drover JW, Su X, et al. Glutamine supplementation in serious illness: a systematic review of the evidence. Crit Care Med 2002;30:2022
Omega-3 fatty acids improve the diagnosis-related clinical outcome. Critical Care Medicine April 2006;34(4):972-9
 Omega-3 fatty acids improve the diagnosis-related clinical outcome 1 Critical Care Medicine April 2006;34(4):972-9 Volume 34(4), April 2006, pp 972-979 Heller, Axel R. MD, PhD; Rössler, Susann; Litz, Rainer
Omega-3 fatty acids improve the diagnosis-related clinical outcome 1 Critical Care Medicine April 2006;34(4):972-9 Volume 34(4), April 2006, pp 972-979 Heller, Axel R. MD, PhD; Rössler, Susann; Litz, Rainer
Aims of Nutritional Support in Oncology (Parenteral) Part 2
 Aims of Nutritional Support in Oncology (Parenteral) Part 2 Rachel Barrett MSc, BSc (Hons) RD Principal Haematology-Oncology Dietitian Hong Kong Hospital Authority Commissioned Training November 20 th
Aims of Nutritional Support in Oncology (Parenteral) Part 2 Rachel Barrett MSc, BSc (Hons) RD Principal Haematology-Oncology Dietitian Hong Kong Hospital Authority Commissioned Training November 20 th
SINPE trial, Ann Surg 2009. Overall morbidity. Minor Major. Infectious Non infectious. Post-hoc analysis WL < %5 (n=379) WL between 5-10% (n=49)
 Chinese International Symposium on Nutritional Oncology Changchun, June 20-21 2014 IMMUNONUTRITION IN CANCER PATIENTS IMMUNONUTRITION IN SURGERY OBJECTIVE To get the patient undergoing major surgery for
Chinese International Symposium on Nutritional Oncology Changchun, June 20-21 2014 IMMUNONUTRITION IN CANCER PATIENTS IMMUNONUTRITION IN SURGERY OBJECTIVE To get the patient undergoing major surgery for
Applying the 2016 ASPEN/ SCCM Critical Care Guidelines to Your Practice. Susan Brantley, MS, RD, LDN
 Applying the 2016 ASPEN/ SCCM Critical Care Guidelines to Your Practice Susan Brantley, MS, RD, LDN Objectives: Upon completion of this presentation, participants should be able to: 1. Distinguish the
Applying the 2016 ASPEN/ SCCM Critical Care Guidelines to Your Practice Susan Brantley, MS, RD, LDN Objectives: Upon completion of this presentation, participants should be able to: 1. Distinguish the
TPN/ Enteral nutrition. Salsabil HADIRE Dietitian in Oncology Hematology Center of University Hospital Mohammed VI Marrakech-
 TPN/ Enteral nutrition Salsabil HADIRE Dietitian in Oncology Hematology Center of University Hospital Mohammed VI Marrakech- Work plan Part 1: Total Parenteral Nutrition (TPN) Part 2: Enteral Nutrition
TPN/ Enteral nutrition Salsabil HADIRE Dietitian in Oncology Hematology Center of University Hospital Mohammed VI Marrakech- Work plan Part 1: Total Parenteral Nutrition (TPN) Part 2: Enteral Nutrition
Nutritional support for cancer patients
 Nutritional support for cancer patients Patients who are receiving adequate nutrition have a better prognosis, respond better to chemotherapy and can tolerate higher doses of anticancer treatments. It
Nutritional support for cancer patients Patients who are receiving adequate nutrition have a better prognosis, respond better to chemotherapy and can tolerate higher doses of anticancer treatments. It
Dietary treatment of cachexia challenges of nutritional research in cancer patients
 Dietary treatment of cachexia challenges of nutritional research in cancer patients Trude R. Balstad 4th International Seminar of the PRC and EAPC RN, Amsterdam 2014 Outline Cancer cachexia Dietary treatment
Dietary treatment of cachexia challenges of nutritional research in cancer patients Trude R. Balstad 4th International Seminar of the PRC and EAPC RN, Amsterdam 2014 Outline Cancer cachexia Dietary treatment
Should Insulin be added to Parenteral Nutrition?
 ESPEN Congress Gothenburg 2011 Educational Session - Pharmaceutical session (in collaboration with ASPEN) Should Insulin be added to Parenteral Nutrition? Jay M Mirtallo Should Insulin be added to Parenteral
ESPEN Congress Gothenburg 2011 Educational Session - Pharmaceutical session (in collaboration with ASPEN) Should Insulin be added to Parenteral Nutrition? Jay M Mirtallo Should Insulin be added to Parenteral
John E. O Toole, Marjorie C. Wang, and Michael G. Kaiser
 Hypothermia and Human Spinal Cord Injury: Updated Position Statement and Evidence Based Recommendations from the AANS/CNS Joint Sections on Disorders of the Spine & Peripheral Nerves and Neurotrauma &
Hypothermia and Human Spinal Cord Injury: Updated Position Statement and Evidence Based Recommendations from the AANS/CNS Joint Sections on Disorders of the Spine & Peripheral Nerves and Neurotrauma &
ESCMID Online Lecture Library. by author
 Do statins improve outcomes of patients with sepsis and pneumonia? Jordi Carratalà Department of Infectious Diseases Statins for sepsis & community-acquired pneumonia Sepsis and CAP are major healthcare
Do statins improve outcomes of patients with sepsis and pneumonia? Jordi Carratalà Department of Infectious Diseases Statins for sepsis & community-acquired pneumonia Sepsis and CAP are major healthcare
- define enteral nutrition - to explore the different types of enteral formulas that are available - describe tube feeding administration guidelines
 Basics of Enteral Feeding 2004 Beth Mills, MS, RD, CNSD, LDN Objectives - define enteral nutrition - to explore the different types of enteral formulas that are available - describe tube feeding administration
Basics of Enteral Feeding 2004 Beth Mills, MS, RD, CNSD, LDN Objectives - define enteral nutrition - to explore the different types of enteral formulas that are available - describe tube feeding administration
Denominator Statement: Cardiac surgery patients with no evidence of prior infection.
 Last Updated: Version 4.3b NQF-ENDORSED VOLUNTARY CONSENSUS STANDARDS FOR HOSPITAL CARE Measure Information Form CMS/The Joint Commission: Suspended (Effective immediately beginning with July 1, 2014 discharges)
Last Updated: Version 4.3b NQF-ENDORSED VOLUNTARY CONSENSUS STANDARDS FOR HOSPITAL CARE Measure Information Form CMS/The Joint Commission: Suspended (Effective immediately beginning with July 1, 2014 discharges)
ESPEN Congress Geneva 2014 ESPEN GUIDELINES. ESPEN Guidelines: nutrition support in cancer J. Arends (DE)
 ESPEN Congress Geneva 2014 ESPEN GUIDELINES ESPEN Guidelines: nutrition support in cancer J. Arends (DE) espen and epaac guidelines nutrition in cancer Jann Arends Tumor Biology Center Freiburg Ethical
ESPEN Congress Geneva 2014 ESPEN GUIDELINES ESPEN Guidelines: nutrition support in cancer J. Arends (DE) espen and epaac guidelines nutrition in cancer Jann Arends Tumor Biology Center Freiburg Ethical
Oral Zinc Supplementation as an Adjunct Therapy in the Management of Hepatic Encephalopathy: A Randomized Controlled Trial
 Oral Zinc Supplementation as an Adjunct Therapy in the Management of Hepatic Encephalopathy: A Randomized Controlled Trial Marcus R. Pereira A. Study Purpose Hepatic encephalopathy is a common complication
Oral Zinc Supplementation as an Adjunct Therapy in the Management of Hepatic Encephalopathy: A Randomized Controlled Trial Marcus R. Pereira A. Study Purpose Hepatic encephalopathy is a common complication
Surgical Treatment of Obesity: A Surgeon s View
 Surgical Treatment of Obesity: A Surgeon s View Jenny J. Choi, MD Director of Bariatrics Associate Director of Clinical Affairs Assistant Professor of Surgery Albert Einstein School of Medicine Montefiore
Surgical Treatment of Obesity: A Surgeon s View Jenny J. Choi, MD Director of Bariatrics Associate Director of Clinical Affairs Assistant Professor of Surgery Albert Einstein School of Medicine Montefiore
TPN origin and calculations. Naureen Iqbal 01/09/13
 TPN origin and calculations Naureen Iqbal 01/09/13 TPN - History Glucose and electrolyte Protein hydrolysates in 30s Fat emulsion- Intralipid in 60s. Vitamins, minerals, trace elements Central venous catheter
TPN origin and calculations Naureen Iqbal 01/09/13 TPN - History Glucose and electrolyte Protein hydrolysates in 30s Fat emulsion- Intralipid in 60s. Vitamins, minerals, trace elements Central venous catheter
ENTERAL FORMULAE AND PARENTERAL NUTRITIONAL SOLUTIONS, DME
 ENTERAL FORMULAE AND PARENTERAL NUTRITIONAL SOLUTIONS, DME Policy NHP only reimburses participating DME vendors for the provision of medically necessary enteral and parenteral formulae and nutritional
ENTERAL FORMULAE AND PARENTERAL NUTRITIONAL SOLUTIONS, DME Policy NHP only reimburses participating DME vendors for the provision of medically necessary enteral and parenteral formulae and nutritional
ALESSANDRO PINTO. Dipartimento di Medicina Sperimentale. Sezione di Fisiopatologia Medica, Scienza dell Alimentazione ed Endocrinologia
 ALESSANDRO PINTO [email protected] Dipartimento di Medicina Sperimentale Sezione di Fisiopatologia Medica, Scienza dell Alimentazione ed Endocrinologia metabolic perturbations from inflammatory
ALESSANDRO PINTO [email protected] Dipartimento di Medicina Sperimentale Sezione di Fisiopatologia Medica, Scienza dell Alimentazione ed Endocrinologia metabolic perturbations from inflammatory
Feeding in Infants with Complex Congenital Heart Disease. Rachel Torok, MD Southeastern Pediatric Cardiology Society Conference September 6, 2014
 Feeding in Infants with Complex Congenital Heart Disease Rachel Torok, MD Southeastern Pediatric Cardiology Society Conference September 6, 2014 Objectives Discuss common feeding issues in patients with
Feeding in Infants with Complex Congenital Heart Disease Rachel Torok, MD Southeastern Pediatric Cardiology Society Conference September 6, 2014 Objectives Discuss common feeding issues in patients with
Inpatient Treatment of Diabetes
 Inpatient Treatment of Diabetes Alan J. Conrad, MD Medical Director Diabetes Services EVP, Physician Alignment Diabetes Symposium November 12, 2015 Objectives Explain Palomar Health goals for inpatient
Inpatient Treatment of Diabetes Alan J. Conrad, MD Medical Director Diabetes Services EVP, Physician Alignment Diabetes Symposium November 12, 2015 Objectives Explain Palomar Health goals for inpatient
Guidance for Industry Diabetes Mellitus Evaluating Cardiovascular Risk in New Antidiabetic Therapies to Treat Type 2 Diabetes
 Guidance for Industry Diabetes Mellitus Evaluating Cardiovascular Risk in New Antidiabetic Therapies to Treat Type 2 Diabetes U.S. Department of Health and Human Services Food and Drug Administration Center
Guidance for Industry Diabetes Mellitus Evaluating Cardiovascular Risk in New Antidiabetic Therapies to Treat Type 2 Diabetes U.S. Department of Health and Human Services Food and Drug Administration Center
Cancer Treatment Centers of America Treating the Whole Patient. Presented by: Jill Schuman, RD, CNSC, CSP, LCN Date: Spring 2012
 Cancer Treatment Centers of America Treating the Whole Patient Presented by: Jill Schuman, RD, CNSC, CSP, LCN Date: Spring 2012 What is Cancer Two types of tumors Liquid tumor Leukemia Lymphoma Aplastic
Cancer Treatment Centers of America Treating the Whole Patient Presented by: Jill Schuman, RD, CNSC, CSP, LCN Date: Spring 2012 What is Cancer Two types of tumors Liquid tumor Leukemia Lymphoma Aplastic
Health-Care Associated Infection Rates among Adult Patients in Bahrain Military Hospital: A Cross Sectional Survey
 Bahrain Medical Bulletin, Vol. 32, No. 1, March 2010 Health-Care Associated Infection Rates among Adult Patients in Bahrain Military Hospital: A Cross Sectional Survey Kelechi Austin Ofurum, M.Sc, B.Sc*,
Bahrain Medical Bulletin, Vol. 32, No. 1, March 2010 Health-Care Associated Infection Rates among Adult Patients in Bahrain Military Hospital: A Cross Sectional Survey Kelechi Austin Ofurum, M.Sc, B.Sc*,
What s new in cancer. NHMRC Evidence hierarchy. Evidence Based Guidelines 12/06/2013. Level I: systematic review of all RCTs
 What s new in cancer treatment? Dr Liz Isenring, AdvAPD Email: [email protected] Clinical Academic Fellow, Princess Alexandra Hospital Conjoint Senior Lecturer University of Queensland, Australia Evidence
What s new in cancer treatment? Dr Liz Isenring, AdvAPD Email: [email protected] Clinical Academic Fellow, Princess Alexandra Hospital Conjoint Senior Lecturer University of Queensland, Australia Evidence
Does Selenium protect against lung cancer? Do Selenium supplements reduce the incidence of lung cancer in healthy individuals?
 Does Selenium protect against lung cancer? Do Selenium supplements reduce the incidence of lung cancer in healthy individuals? Alexander Blakes and Eleanor Brunt October 2014 The question Mr Wilson s father
Does Selenium protect against lung cancer? Do Selenium supplements reduce the incidence of lung cancer in healthy individuals? Alexander Blakes and Eleanor Brunt October 2014 The question Mr Wilson s father
Components of CVC Care Bundle. selection
 Components of CVC Care Bundle Catheter site selection Site of insertion influences the subsequent risk for CR-BSI and phlebitis The influence of site is related in part to the risk for thrombophlebitis
Components of CVC Care Bundle Catheter site selection Site of insertion influences the subsequent risk for CR-BSI and phlebitis The influence of site is related in part to the risk for thrombophlebitis
Refeeding syndrome in anorexia nervosa
 ESPEN Congress Barcelona 2012 Is there a role for nutrition in psychiatric disorders? Refeeding syndrome in anorexia nervosa V. Haas (Germany) ESPEN - 2012 - Barcelona The refeeding syndrome in Anorexia
ESPEN Congress Barcelona 2012 Is there a role for nutrition in psychiatric disorders? Refeeding syndrome in anorexia nervosa V. Haas (Germany) ESPEN - 2012 - Barcelona The refeeding syndrome in Anorexia
Over 50% of hospitalized patients are malnourished. Coding for Malnutrition in the Adult Patient: What the Physician Needs to Know
 Carol Rees Parrish, M.S., R.D., Series Editor Coding for Malnutrition in the Adult Patient: What the Physician Needs to Know Wendy Phillips At least half of all hospitalized patients are malnourished,
Carol Rees Parrish, M.S., R.D., Series Editor Coding for Malnutrition in the Adult Patient: What the Physician Needs to Know Wendy Phillips At least half of all hospitalized patients are malnourished,
NUTRITION OF THE BODY
 5 Training Objectives:! Knowledge of the most important function of nutrients! Description of both, mechanism and function of gluconeogenesis! Knowledge of the difference between essential and conditionally
5 Training Objectives:! Knowledge of the most important function of nutrients! Description of both, mechanism and function of gluconeogenesis! Knowledge of the difference between essential and conditionally
The When, Where and Why of Permissive Underfeeding
 The When, Where and Why of Permissive Underfeeding Ainsley Malone, MS, RD, LD, CNSD Nutrition Support Team Mt. Carmel West Hospital Columbus, OH 43222 Introduction The importance of providing patients
The When, Where and Why of Permissive Underfeeding Ainsley Malone, MS, RD, LD, CNSD Nutrition Support Team Mt. Carmel West Hospital Columbus, OH 43222 Introduction The importance of providing patients
CME Test for AMDA Clinical Practice Guideline. Diabetes Mellitus
 CME Test for AMDA Clinical Practice Guideline Diabetes Mellitus Part I: 1. Which one of the following statements about type 2 diabetes is not accurate? a. Diabetics are at increased risk of experiencing
CME Test for AMDA Clinical Practice Guideline Diabetes Mellitus Part I: 1. Which one of the following statements about type 2 diabetes is not accurate? a. Diabetics are at increased risk of experiencing
Rehabilitation and Lung Cancer Resection. Roberto Benzo MD MS Mindful Breathing Laboratory Division of Pulmonary & CCM Mayo Clinic
 Rehabilitation and Lung Cancer Resection Roberto Benzo MD MS Mindful Breathing Laboratory Division of Pulmonary & CCM Mayo Clinic Disclosure Funded by the National Cancer Institute NIH for Preoperative
Rehabilitation and Lung Cancer Resection Roberto Benzo MD MS Mindful Breathing Laboratory Division of Pulmonary & CCM Mayo Clinic Disclosure Funded by the National Cancer Institute NIH for Preoperative
Nutritional Support of the Burn Patient
 Nutritional Support of the Burn Patient Objectives To understand the principles of normal nutrient utilization and the abnormalities caused by burn injury To be able to assess nutrient needs To be able
Nutritional Support of the Burn Patient Objectives To understand the principles of normal nutrient utilization and the abnormalities caused by burn injury To be able to assess nutrient needs To be able
Diabetes and Weight-Loss Surgery
 WHITE PAPER Diabetes and Weight-Loss Surgery Treat the cause. Cure the symptom. Center of Excellence BARIATRIC SURGERY Written July 2011 Bariatric Surgery: The Cure for Type II Diabetes? For most individuals
WHITE PAPER Diabetes and Weight-Loss Surgery Treat the cause. Cure the symptom. Center of Excellence BARIATRIC SURGERY Written July 2011 Bariatric Surgery: The Cure for Type II Diabetes? For most individuals
Nutrition Intervention for Spinal Cord Injury in Critical Care
 Nutrition Intervention for Spinal Cord Injury in Critical Care Dina Abdulrahman Muharib Critical Care Dietitian King Saud Medical City Riyadh, Saudi Arabia ICND 2015 : XIII International Conference on
Nutrition Intervention for Spinal Cord Injury in Critical Care Dina Abdulrahman Muharib Critical Care Dietitian King Saud Medical City Riyadh, Saudi Arabia ICND 2015 : XIII International Conference on
PATIENT CONSENT TO PROCEDURE - ROUX-EN-Y GASTRIC BYPASS
 As a patient you must be adequately informed about your condition and the recommended surgical procedure. Please read this document carefully and ask about anything you do not understand. Please initial
As a patient you must be adequately informed about your condition and the recommended surgical procedure. Please read this document carefully and ask about anything you do not understand. Please initial
The Link Between Obesity and Diabetes The Rapid Evolution and Positive Results of Bariatric Surgery
 The Link Between Obesity and Diabetes The Rapid Evolution and Positive Results of Bariatric Surgery Michael E. Farkouh, MD, MSc Peter Munk Chair in Multinational Clinical Trials Director, Heart and Stroke
The Link Between Obesity and Diabetes The Rapid Evolution and Positive Results of Bariatric Surgery Michael E. Farkouh, MD, MSc Peter Munk Chair in Multinational Clinical Trials Director, Heart and Stroke
Colocutaneous Fistula. Disclosures
 Colocutaneous Fistula Madhulika G. Varma MD Associate Professor Chief, Colorectal Surgery University of California, San Francisco Honoraria Applied Medical Covidien Disclosures 1 Colocutaneous Fistula
Colocutaneous Fistula Madhulika G. Varma MD Associate Professor Chief, Colorectal Surgery University of California, San Francisco Honoraria Applied Medical Covidien Disclosures 1 Colocutaneous Fistula
Nursing college, Second stage Microbiology Dr.Nada Khazal K. Hendi L14: Hospital acquired infection, nosocomial infection
 L14: Hospital acquired infection, nosocomial infection Definition A hospital acquired infection, also called a nosocomial infection, is an infection that first appears between 48 hours and four days after
L14: Hospital acquired infection, nosocomial infection Definition A hospital acquired infection, also called a nosocomial infection, is an infection that first appears between 48 hours and four days after
High Impact Intervention Care bundle to reduce ventilation-association pneumonia
 High Impact Intervention Care bundle to reduce ventilation-association pneumonia Aim To reduce the incidence of ventilation-associated pneumonia (VAP). Context The aim of the care bundle, as set out in
High Impact Intervention Care bundle to reduce ventilation-association pneumonia Aim To reduce the incidence of ventilation-associated pneumonia (VAP). Context The aim of the care bundle, as set out in
Clinical Study Synopsis
 Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
FOR THE PREVENTION OF ATRIAL FIBRILLATION RELATED STROKE
 www.bpac.org.nz keyword: warfarinaspirin FOR THE PREVENTION OF ATRIAL FIBRILLATION RELATED STROKE Key Concepts In atrial fibrillation (AF) warfarin is more effective than aspirin for stroke prevention.
www.bpac.org.nz keyword: warfarinaspirin FOR THE PREVENTION OF ATRIAL FIBRILLATION RELATED STROKE Key Concepts In atrial fibrillation (AF) warfarin is more effective than aspirin for stroke prevention.
ENTERAL. Sincerely, Professor Ferdinand Haschke, MD Chairman Nestlé Nutrition Institute. American Society for Parenteral and Enteral Nutrition
 ENTERAL It is with pleasure that I invite you to apply for the 2010 2011 Nestlé Institute Enteral. This competitive and prestigious award is designed to provide an unparalleled educational and clinical
ENTERAL It is with pleasure that I invite you to apply for the 2010 2011 Nestlé Institute Enteral. This competitive and prestigious award is designed to provide an unparalleled educational and clinical
INTERNAL MEDICINE RESIDENTS NOON CONFERENCE: INPATIENT GLYCEMIC CONTROL
 INTERNAL MEDICINE RESIDENTS NOON CONFERENCE: INPATIENT GLYCEMIC CONTROL Presented by: Leyda Callejas PGY5 Endocrinology, Diabetes and Metabolism Acknowledgements: Dr. P Orlander Dr. V Lavis Dr. N Shah
INTERNAL MEDICINE RESIDENTS NOON CONFERENCE: INPATIENT GLYCEMIC CONTROL Presented by: Leyda Callejas PGY5 Endocrinology, Diabetes and Metabolism Acknowledgements: Dr. P Orlander Dr. V Lavis Dr. N Shah
HOMEOPATHY AS AN ALTERNATIVE TO ANTIBIOTICS
 HOMEOPATHY AS AN ALTERNATIVE TO ANTIBIOTICS Professor Dr. Michael FRASS Medical University of Vienna, Department Medicine I Doctor s Asscociation for Classical Homeopathy www.aekh.at President, Umbrella
HOMEOPATHY AS AN ALTERNATIVE TO ANTIBIOTICS Professor Dr. Michael FRASS Medical University of Vienna, Department Medicine I Doctor s Asscociation for Classical Homeopathy www.aekh.at President, Umbrella
Critical appraisal of systematic reviews
 Critical appraisal of systematic reviews Abalos E, Carroli G, Mackey ME, Bergel E Centro Rosarino de Estudios Perinatales, Rosario, Argentina INTRODUCTION In spite of the increasingly efficient ways to
Critical appraisal of systematic reviews Abalos E, Carroli G, Mackey ME, Bergel E Centro Rosarino de Estudios Perinatales, Rosario, Argentina INTRODUCTION In spite of the increasingly efficient ways to
DIET AND EXERCISE STRATEGIES FOR WEIGHT LOSS AND WEIGHT MAINTENANCE
 DIET AND EXERCISE STRATEGIES FOR WEIGHT LOSS AND WEIGHT MAINTENANCE 40 yo woman, BMI 36. Motivated to begin diet therapy. Which of the following is contraindicated: Robert B. Baron MD MS Professor and
DIET AND EXERCISE STRATEGIES FOR WEIGHT LOSS AND WEIGHT MAINTENANCE 40 yo woman, BMI 36. Motivated to begin diet therapy. Which of the following is contraindicated: Robert B. Baron MD MS Professor and
Principles of Systematic Review: Focus on Alcoholism Treatment
 Principles of Systematic Review: Focus on Alcoholism Treatment Manit Srisurapanont, M.D. Professor of Psychiatry Department of Psychiatry, Faculty of Medicine, Chiang Mai University For Symposium 1A: Systematic
Principles of Systematic Review: Focus on Alcoholism Treatment Manit Srisurapanont, M.D. Professor of Psychiatry Department of Psychiatry, Faculty of Medicine, Chiang Mai University For Symposium 1A: Systematic
Liver, Gallbladder, Exocrine Pancreas KNH 406
 Liver, Gallbladder, Exocrine Pancreas KNH 406 2007 Thomson - Wadsworth LIVER Anatomy - functions With disease blood flow becomes obstructed Bile All bile drains into common hepatic duct Liver Bile complex
Liver, Gallbladder, Exocrine Pancreas KNH 406 2007 Thomson - Wadsworth LIVER Anatomy - functions With disease blood flow becomes obstructed Bile All bile drains into common hepatic duct Liver Bile complex
10. Treatment of peritoneal dialysis associated fungal peritonitis
 10. Treatment of peritoneal dialysis associated fungal peritonitis Date written: February 2003 Final submission: July 2004 Guidelines (Include recommendations based on level I or II evidence) The use of
10. Treatment of peritoneal dialysis associated fungal peritonitis Date written: February 2003 Final submission: July 2004 Guidelines (Include recommendations based on level I or II evidence) The use of
Clinical Policy Title: Air Ambulance Transport
 Clinical Policy Title: Air Ambulance Transport Clinical Policy Number: 18.02.02 Effective Date: Sept. 1, 2014 Initial Review Date: April 16, 2014 Most Recent Review Date: May 21, 2014 Next Review Date:
Clinical Policy Title: Air Ambulance Transport Clinical Policy Number: 18.02.02 Effective Date: Sept. 1, 2014 Initial Review Date: April 16, 2014 Most Recent Review Date: May 21, 2014 Next Review Date:
Medical Nutrition Therapy for Upper Gastrointestinal Tract Disorders. By: Jalal Hejazi PhD, MSc.
 Medical Nutrition Therapy for Upper Gastrointestinal Tract Disorders By: Jalal Hejazi PhD, MSc. Digestive Disorders Common problem; more than 50 million outpatient visits per year Dietary habits and nutrition
Medical Nutrition Therapy for Upper Gastrointestinal Tract Disorders By: Jalal Hejazi PhD, MSc. Digestive Disorders Common problem; more than 50 million outpatient visits per year Dietary habits and nutrition
CDEC FINAL RECOMMENDATION
 CDEC FINAL RECOMMENDATION RIVAROXABAN (Xarelto Bayer Inc.) New Indication: Pulmonary Embolism Note: The Canadian Drug Expert Committee (CDEC) previously reviewed rivaroxaban for the treatment of deep vein
CDEC FINAL RECOMMENDATION RIVAROXABAN (Xarelto Bayer Inc.) New Indication: Pulmonary Embolism Note: The Canadian Drug Expert Committee (CDEC) previously reviewed rivaroxaban for the treatment of deep vein
Dietary Composition for Weight Loss and Weight Loss Maintenance
 Dietary Composition for Weight Loss and Weight Loss Maintenance Bridget M. Hron, MD Instructor in Pediatrics, Harvard Medical School Staff Physician in Gastroenterology & Nutrition and New Balance Foundation
Dietary Composition for Weight Loss and Weight Loss Maintenance Bridget M. Hron, MD Instructor in Pediatrics, Harvard Medical School Staff Physician in Gastroenterology & Nutrition and New Balance Foundation
European Dietitians in Oncology: The Future
 European Dietitians in Oncology: The Future Carolina Bento Clare Shaw Julia Lobenwein 1 2 Oncology Workshop Introduction to the committee Clare Shaw (United Kingdom) Carolina Bento (Portugal) Julia Lobenwein
European Dietitians in Oncology: The Future Carolina Bento Clare Shaw Julia Lobenwein 1 2 Oncology Workshop Introduction to the committee Clare Shaw (United Kingdom) Carolina Bento (Portugal) Julia Lobenwein
TOTAL PARENTERAL NUTRITION (TPN) Revised January 2013
 TOTAL PARENTERAL NUTRITION (TPN) Revised January 2013 OBJECTIVES Definition Indications for TPN administration Composition of TPN solutions Access routes for TPN administration Monitoring TPN administration
TOTAL PARENTERAL NUTRITION (TPN) Revised January 2013 OBJECTIVES Definition Indications for TPN administration Composition of TPN solutions Access routes for TPN administration Monitoring TPN administration
CARDIAC SURGERY INTRAVENOUS INSULIN PROTOCOL PHYSICIAN ORDERS INDICATIONS EXCLUSIONS. Insulin allergy
 Page 1 of 5 INDICATIONS EXCLUSIONS 2 consecutive blood glucose measurements greater than 110 mg per dl AND NPO with a continuous caloric source AND Diagnosis of : Cardio-thoracic Surgery NOTE: This protocol
Page 1 of 5 INDICATIONS EXCLUSIONS 2 consecutive blood glucose measurements greater than 110 mg per dl AND NPO with a continuous caloric source AND Diagnosis of : Cardio-thoracic Surgery NOTE: This protocol
DCCT and EDIC: The Diabetes Control and Complications Trial and Follow-up Study
 DCCT and EDIC: The Diabetes Control and Complications Trial and Follow-up Study National Diabetes Information Clearinghouse U.S. Department of Health and Human Services NATIONAL INSTITUTES OF HEALTH What
DCCT and EDIC: The Diabetes Control and Complications Trial and Follow-up Study National Diabetes Information Clearinghouse U.S. Department of Health and Human Services NATIONAL INSTITUTES OF HEALTH What
Diabetes? Does Metabolic Surgery. Experts disagree about how surgery treats diabetes but agree more research needs to be done.
 Does Metabolic Surgery The combination of type 2 diabetes and being significantly overweight is a huge burden. Doctors tell you to lose weight, in essence, to save your life. Weight loss, in addition to
Does Metabolic Surgery The combination of type 2 diabetes and being significantly overweight is a huge burden. Doctors tell you to lose weight, in essence, to save your life. Weight loss, in addition to
Wound Healing. Outline. Normal Wound Healing. Wounds and nutrition refresher UPHS evidence-based guideline for. wounds
 Wound Healing Clinical Nutrition Support Service Hospital of the University of Pennsylvania Jung Kim, RD CNSD, LDN Tricia Stefankiewicz, MA, RD, CNSC, LDN Outline Wounds and nutrition refresher UPHS evidence-based
Wound Healing Clinical Nutrition Support Service Hospital of the University of Pennsylvania Jung Kim, RD CNSD, LDN Tricia Stefankiewicz, MA, RD, CNSC, LDN Outline Wounds and nutrition refresher UPHS evidence-based
Jill Malcolm, Karen Moir
 Evaluation of Fife- DICE: Type 2 diabetes insulin conversion Article points 1. Fife-DICE is an insulin conversion group education programme. 2. People with greater than 7.5% on maximum oral therapy are
Evaluation of Fife- DICE: Type 2 diabetes insulin conversion Article points 1. Fife-DICE is an insulin conversion group education programme. 2. People with greater than 7.5% on maximum oral therapy are
BIBLIOGRAPHICAL REVIEW ON COST OF PATIENT SAFETY FAILINGS IN ADMINISTRATION OF DRUGS. SUMMARY.
 BIBLIOGRAPHICAL REVIEW ON COST OF PATIENT SAFETY FAILINGS IN ADMINISTRATION OF DRUGS. SUMMARY. Bibliographical review on cost of Patient Safety Failings in administration of drugs. Summary This has been
BIBLIOGRAPHICAL REVIEW ON COST OF PATIENT SAFETY FAILINGS IN ADMINISTRATION OF DRUGS. SUMMARY. Bibliographical review on cost of Patient Safety Failings in administration of drugs. Summary This has been
Antibiotic Prophylaxis for Short-term Catheter Bladder Drainage in adults. A Systematic Review (Cochrane database August 2013)
 Antibiotic Prophylaxis for Short-term Catheter Bladder Drainage in adults A Systematic Review (Cochrane database August 2013) Gail Lusardi, Senior Lecturer Dr Allyson Lipp, Principal Lecturer, Dr Chris
Antibiotic Prophylaxis for Short-term Catheter Bladder Drainage in adults A Systematic Review (Cochrane database August 2013) Gail Lusardi, Senior Lecturer Dr Allyson Lipp, Principal Lecturer, Dr Chris
Learning Objectives. Introduction to Medical Careers. Vocabulary: Chapter 16 FACTS. Functions. Organs. Digestive System Chapter 16
 Learning Objectives Introduction to Medical Careers Digestive System Chapter 16 Define at least 10 terms relating to the digestive Describe the four functions of the digestive Identify different structures
Learning Objectives Introduction to Medical Careers Digestive System Chapter 16 Define at least 10 terms relating to the digestive Describe the four functions of the digestive Identify different structures
Pain and tissue-interface pressures during spineboard immobilization.
 Ann Emerg Med. 1995 Jul;26(1):31-6. Links Pain and tissue-interface pressures during spineboard immobilization. Cordell WH, Hollingsworth JC, Olinger ML, Stroman SJ, elson DR. Emergency Medicine and Trauma
Ann Emerg Med. 1995 Jul;26(1):31-6. Links Pain and tissue-interface pressures during spineboard immobilization. Cordell WH, Hollingsworth JC, Olinger ML, Stroman SJ, elson DR. Emergency Medicine and Trauma
Vitamin D. Sources of vitamin D
 1 has been in the news frequently this past year, including an article in The New York Times on November 16, 2009. So what is this vitamin? Why is it important? Most people have heard that vitamin D is
1 has been in the news frequently this past year, including an article in The New York Times on November 16, 2009. So what is this vitamin? Why is it important? Most people have heard that vitamin D is
Bariatric Surgery. OHTAC Recommendation. Bariatric Surgery
 OHTAC Recommendation Bariatric Surgery January 21, 2005 1 The Ontario Health Technology Advisory Committee (OHTAC) met on January 21, 2005 and reviewed bariatric surgery for morbid obesity. Obesity is
OHTAC Recommendation Bariatric Surgery January 21, 2005 1 The Ontario Health Technology Advisory Committee (OHTAC) met on January 21, 2005 and reviewed bariatric surgery for morbid obesity. Obesity is
How To Screen For Alcohol Dependence
 Commonwealth of Massachusetts Board of Registration in Medicine Quality and Patient Safety Division Advisory Alcohol Screening and Management Protocols July 2013 Background The Quality and Patient Safety
Commonwealth of Massachusetts Board of Registration in Medicine Quality and Patient Safety Division Advisory Alcohol Screening and Management Protocols July 2013 Background The Quality and Patient Safety
How To Decide If A Helicopter Is Right For A Patient
 Clinical Policy Title: Air Ambulance Transport Clinical Policy Number: 18.02.02 Effective Date: September 1, 2014 Initial Review Date: April 16, 2014 Most Recent Review Date: May 20, 2015 Next Review Date:
Clinical Policy Title: Air Ambulance Transport Clinical Policy Number: 18.02.02 Effective Date: September 1, 2014 Initial Review Date: April 16, 2014 Most Recent Review Date: May 20, 2015 Next Review Date:
Committee Approval Date: September 12, 2014 Next Review Date: September 2015
 Medication Policy Manual Policy No: dru361 Topic: Pradaxa, dabigatran Date of Origin: September 12, 2014 Committee Approval Date: September 12, 2014 Next Review Date: September 2015 Effective Date: November
Medication Policy Manual Policy No: dru361 Topic: Pradaxa, dabigatran Date of Origin: September 12, 2014 Committee Approval Date: September 12, 2014 Next Review Date: September 2015 Effective Date: November
Sepsis Awareness Month
 Aon Kenya Insurance Brokers Ltd Aon Hewitt Healthcare Division Sepsis Awareness Month Issue 11 September 2015 In this Issue 2 Getting to understand Sepsis 3 Stages in Sepsis Advancement 4 Diagnosis & Treatment
Aon Kenya Insurance Brokers Ltd Aon Hewitt Healthcare Division Sepsis Awareness Month Issue 11 September 2015 In this Issue 2 Getting to understand Sepsis 3 Stages in Sepsis Advancement 4 Diagnosis & Treatment
Presented by: Jean Yoo-Campbell, Matthew Konerman, Monica Konerman, Jean Yoo Campbell, Christian Gocke, Eunpi Cho Donald Lynch
 Bass N.M., et. al. N Engl J Med 2010; 362:1071-1081 Presented by: Jean Yoo-Campbell, Matthew Konerman, Monica Konerman, Jean Yoo Campbell, Christian Gocke, Eunpi Cho Donald Lynch Faculty Advisor: Dr. Fred
Bass N.M., et. al. N Engl J Med 2010; 362:1071-1081 Presented by: Jean Yoo-Campbell, Matthew Konerman, Monica Konerman, Jean Yoo Campbell, Christian Gocke, Eunpi Cho Donald Lynch Faculty Advisor: Dr. Fred
A: Nursing Knowledge. Alberta Licensed Practical Nurses Competency Profile 1
 A: Nursing Knowledge Alberta Licensed Practical Nurses Competency Profile 1 Competency: A-1 Anatomy and Physiology A-1-1 A-1-2 A-1-3 A-1-4 A-1-5 A-1-6 A-1-7 A-1-8 Identify the normal structures and functions
A: Nursing Knowledge Alberta Licensed Practical Nurses Competency Profile 1 Competency: A-1 Anatomy and Physiology A-1-1 A-1-2 A-1-3 A-1-4 A-1-5 A-1-6 A-1-7 A-1-8 Identify the normal structures and functions
FORMULA & SPECIALIZED FOOD
 FORMULA & SPECIALIZED FOOD ADMINISTRATIVE POLICY Policy Number: HOME 005.16 T2 Effective Date: December 1, 2014 Table of Contents CONDITIONS OF COVERAGE... COVERAGE RATIONALE BENEFIT CONSIDERATIONS...
FORMULA & SPECIALIZED FOOD ADMINISTRATIVE POLICY Policy Number: HOME 005.16 T2 Effective Date: December 1, 2014 Table of Contents CONDITIONS OF COVERAGE... COVERAGE RATIONALE BENEFIT CONSIDERATIONS...
ICD-9-CM/ICD-10-CM Codes for MNT
 / Codes for MNT ICD (International Classification of Diseases) codes are used by physicians and medical coders to assign medical diagnoses to individual patients. It is not within the scope of practice
/ Codes for MNT ICD (International Classification of Diseases) codes are used by physicians and medical coders to assign medical diagnoses to individual patients. It is not within the scope of practice
Measure #257 (NQF 1519): Statin Therapy at Discharge after Lower Extremity Bypass (LEB) National Quality Strategy Domain: Effective Clinical Care
 Measure #257 (NQF 1519): Statin Therapy at Discharge after Lower Extremity Bypass (LEB) National Quality Strategy Domain: Effective Clinical Care 2016 PQRS OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY
Measure #257 (NQF 1519): Statin Therapy at Discharge after Lower Extremity Bypass (LEB) National Quality Strategy Domain: Effective Clinical Care 2016 PQRS OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY
Treatment for Severely Obese Patients
 Treatment for Severely Obese Patients Associate Professor Jimmy So Senior Consultant Surgeon Director, Centre for Obesity Management and Surgery (COMS) National University Hospital Obesity Shortens Lives
Treatment for Severely Obese Patients Associate Professor Jimmy So Senior Consultant Surgeon Director, Centre for Obesity Management and Surgery (COMS) National University Hospital Obesity Shortens Lives
The first endoscopically-delivered device therapy for obese patients with type 2 diabetes
 DIABETES WEIGHT ENDOBARRIER THERAPY The first endoscopically-delivered device therapy for obese patients with type 2 diabetes Restore the metabolic health of your patients with EndoBarrier Therapy. Dual
DIABETES WEIGHT ENDOBARRIER THERAPY The first endoscopically-delivered device therapy for obese patients with type 2 diabetes Restore the metabolic health of your patients with EndoBarrier Therapy. Dual
Roux-en-Y Gastric Bypass
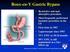 Roux-en-Y Gastric Bypass Restrictive and malabsorptive procedure Most frequently performed bariatric procedure in the US First done in 1967 Laparoscopic since 1993 75% EWL in 18-24 months 50% EWL is still
Roux-en-Y Gastric Bypass Restrictive and malabsorptive procedure Most frequently performed bariatric procedure in the US First done in 1967 Laparoscopic since 1993 75% EWL in 18-24 months 50% EWL is still
A study to Evaluate PPI s effect on vitamin D levels. Rani Hanna M.D., M.S. PGY-3 Joseph Grisanti, MD
 A study to Evaluate PPI s effect on vitamin D levels Rani Hanna M.D., M.S. PGY-3 Joseph Grisanti, MD The sunshine vitamin Existed over 500 million years. Prehormone, not only a vitamin. Two major sources:
A study to Evaluate PPI s effect on vitamin D levels Rani Hanna M.D., M.S. PGY-3 Joseph Grisanti, MD The sunshine vitamin Existed over 500 million years. Prehormone, not only a vitamin. Two major sources:
BOLETIN BIBLIOGRAFICO BIBLIOTECA MEDICA-HCG JUNIO 2014
 BOLETIN BIBLIOGRAFICO BIBLIOTECA MEDICA-HCG JUNIO 2014 Am J Clin Pathol 1. 2013 Statement on Human Papillomavirus DNA Test Utilization. AJCP 141 (4): 459-461, abr. 2014 2. Pathology Consultation on Electronic
BOLETIN BIBLIOGRAFICO BIBLIOTECA MEDICA-HCG JUNIO 2014 Am J Clin Pathol 1. 2013 Statement on Human Papillomavirus DNA Test Utilization. AJCP 141 (4): 459-461, abr. 2014 2. Pathology Consultation on Electronic
Types of Bariatric Procedures. Tejal Brahmbhatt, MD General Surgery Teaching Conference April 18, 2012
 Types of Bariatric Procedures Tejal Brahmbhatt, MD General Surgery Teaching Conference April 18, 2012 A Brief History of Bariatric Surgery First seen in pts with short bowel syndrome weight loss First
Types of Bariatric Procedures Tejal Brahmbhatt, MD General Surgery Teaching Conference April 18, 2012 A Brief History of Bariatric Surgery First seen in pts with short bowel syndrome weight loss First
FAILURE TO THRIVE What Is Failure to Thrive?
 FAILURE TO THRIVE The first few years of life are a time when most children gain weight and grow much more rapidly than they will later on. Sometimes, however, babies and children don't meet expected standards
FAILURE TO THRIVE The first few years of life are a time when most children gain weight and grow much more rapidly than they will later on. Sometimes, however, babies and children don't meet expected standards
Vasopressors. Judith Hellman, M.D. Associate Professor Anesthesia and Perioperative Care University of California, San Francisco
 Vasopressors Judith Hellman, M.D. Associate Professor Anesthesia and Perioperative Care University of California, San Francisco Overview Define shock states Review drugs commonly used to treat hypotension
Vasopressors Judith Hellman, M.D. Associate Professor Anesthesia and Perioperative Care University of California, San Francisco Overview Define shock states Review drugs commonly used to treat hypotension
Surgical Site Infection Prevention
 Surgical Site Infection Prevention 1 Objectives 1. Discuss risk factors for SSI 2. Describe evidence-based best practices for SSI prevention 3. State principles of antibiotic prophylaxis 4. Discuss novel
Surgical Site Infection Prevention 1 Objectives 1. Discuss risk factors for SSI 2. Describe evidence-based best practices for SSI prevention 3. State principles of antibiotic prophylaxis 4. Discuss novel
APPENDIX B SAMPLE PEDIATRIC CRITICAL CARE NURSE PRACTITIONER GOALS AND OBJECTIVES
 APPENDIX B SAMPLE PEDIATRIC CRITICAL CARE NURSE PRACTITIONER GOALS AND OBJECTIVES The critical care nurse practitioner orientation is an individualized process based on one s previous experiences and should
APPENDIX B SAMPLE PEDIATRIC CRITICAL CARE NURSE PRACTITIONER GOALS AND OBJECTIVES The critical care nurse practitioner orientation is an individualized process based on one s previous experiences and should
