Neoadjuvant (Primary) Systemic Therapy
|
|
|
- Vernon Martin
- 7 years ago
- Views:
Transcription
1 Diagnosis and Treatment of Patients with Primary and Metastatic Breast Cancer Version Neoadjuvant (Primary) Systemic Therapy
2 Neoadjuvant Systemic Therapy Version Version 2002: Costa Versions : Bauerfeind / Blohmer / Dall / Fersis / Göhring / Harbeck / Heinrich / Huober / Jackisch / Kaufmann / Loibl / Lux / von Minckwitz / Müller / Nitz / Schneeweiss / Schütz / Solomayer / Untch Version 2015: Friedrich / Schneeweiss
3 Neoadjuvant Systemic Chemotherapy Indications Version Oxford / AGO LoE / GR Inflammatory breast cancer 2b B ++ Inoperable breast cancer 1c A ++ Large operable breast cancer primarily requiring mastectomy and adjuvant chemotherapy with the goal of breast conservation 1b B + If similar postoperative adjuvant chemotherapy is indicated 1b A +
4 Subtype-specific General Systemic Strategies Version In case of indication for chemotherapy, AGO consider neoadjuvant approach ++ HR+/HER2- and low risk : Endocrine therapy without chemotherapy ++ HR+/HER2- and high risk Conventionally dosed AT-based chemotherapy ++ Dose dense & escalated in case of high tumor burden + Followed by endocrine therapy ++ HER2+ Trastuzumab plus ++ Sequential A/T-based regimen with concurrent T + H ++ Anthracycline-free, carboplatin-cont. regimen + Dose dense & escalated in case of high tumor burden + TNBC Conventionally dosed AT-based chemotherapy ++ Dose dense & escalated + Plus Carboplatin in case of family history for BC/OC or gbrca alteration +
5 Anteil Patienten mit pcr (%) ypt0/is ypn0 ypt0/is ypn0 ypt0/is ypn0 ypt0/is ypn0 1. Cortazar P, et al. Lancet 2014; 2. Ando M, et al. BCRT 2014; 3. Sikov WM, et al. J Clin Oncol 2015; 4. von Minckwitz G, et al. Lancet Oncol 2014; 5. Petrelli F, et al. BCRT 2014 ypt0/is ypn0 ypt0 ypn0 ypt0 ypn0 ypt0 ypn0 ypt0 ypn0 Verbesserung der pcr-raten durch Platin beim TNBC 70 p=0, p=0, p=0, P<0, TNBC HR (N=992)
6 pcr als Surrogat für Überleben? Überträgt sich die höhere pcr-rate in ein besseres Überleben? Wenn ja, welches Δ pcr brauchen wir für eine signifikante Verbesserung des Überlebens? Gibt es Subgruppen mit einem größeren Δ pcr?
7 GeparSixto (Phase II): pcr abhängig von genetischer Belastung nach PMB + Carbo vs. PMB beim TNBC (N=294) Von Minckwitz G, et al. ASCO 2014 (abs 1005), oral abstract session
8 Neoadjuvant Systemic Chemotherapy Recommended Regimens and Schedules Version Oxford / AGO LoE / GR Standard regimens used in the adjuvant setting with a duration of at least 18 weeks 1a A ++ AC or EC D q3w or P q1w 2b A ++ DAC 2b B ++ AP CMF 1b A + Taxane followed by anthracycline sequence 1a A + Dose-dense regimen (e.g. E -P-CMF, E-P-C) 1b B +* Platinum in TNBC 1a A +/- in case of family history of BC/OC or BRCA alteration 2b B + *Study participation recommended
9 Superior Carboplatin Containing Regimens in the Neoadjuvant Setting Version Author Study Regimen pcr rate Sikov WM, et al. (JCO 2015) von Minckwitz G, et al. (Lancet Oncol 2014) Ando M, et al. (BCRT 2014) CALGB Phase II Gepar Sixto Phase II Paclitaxel 80mg/m² qw x12 + Carboplatin AUC 6 q3w x4 dd AC q2w x4 NPLD 20mg/m² qw x18 + Paclitaxel 80mg/m² qw x18 + Carboplatin AUC 1.5 qw x18 + Bev 15mg/kg q3w x6 Phase II Paclitaxel 80mg/m² qw x12 + Carboplatin AUC 5 q3w x4 FEC q3w x4 TNBC ± Cb: 54% vs 41% (ypt0/is ypn0) TNBC ± Cb: 53% vs. 37% (ypt0 ypn0) TNBC ± Cb: 61% vs. 26%
10 Neoadjuvant Systemic Chemotherapy Recommended Regimens and Schedules Version Oxford / AGO LoE / GR Standard regimens used in the adjuvant setting with a duration of at least 18 weeks 1a A ++ AC or EC D q3w or P q1w 2b A ++ DAC 2b B ++ AP CMF 1b A + Taxane followed by anthracycline sequence 1a A + Dose-dense regimen (e.g. E -P-CMF, E-P-C) 1b B +* Platinum in TNBC 1a A +/- in case of family history of BC/OC or BRCA alteration 2b B + *Study participation recommended
11 Neo-tAnGo (Phase III): Neoadj. Pac ± Gem EC vs. EC Pac ± Gem beim HER2- EBC > 2cm (N=831) pcr, Überleben pcr (ypt0/is ypn0): 20% vs. 15%, p=0.03 Disease-free Survival Overall Survival Earl HM, et al. Lancet Oncol 15: , 2014
12 Review: (Neo)adj. Taxan Anthrazyklin vs. Anthrazyklin Taxan beim EBC Seven studies in the adjuvant setting and eight in the neoadjuvant setting (10 randomized trials) Nearly 5000 patients None of the clinical trials has shown disadvantages in terms of efficacy or toxicity for taxane first. Similar or increased pathological complete response rates for sequences in which the taxane first. Conclusion: There seems to be sufficient evidence to suggest that a taxane followed by an anthracycline is a sequence option that can be incorporated into daily clinical practice. Bines J, et al. Ann Oncol 25: , 2014
13 Neoadjuvant Systemic Therapy Response Prediction II Version Factor LoE 2009 CTS GR AGO Multigene signature III C B +/- Ki-67 I B A + Tumour infiltrating lymphocytes I B B +/- PIK3CA mutation II B B +/-
14 Standards für die Evaluation von Tumor-infiltrierenden Lymphozyten (TILs) Nur stromale TILs Als % des Tumorstroma-Areals Innerhalb der Tumorgrenzen Semiquantitativ Bei heterogener Verteilung gilt der Durchschnitt Salgado R, Denkert C, Demaria S, et al. Ann Oncol 2014 [Epub ahead of print]
15 Stromale TILs scheinen prädiktiv für das Erreichen einer pcr nach neoadjuvanter CT + Trastuzumab Retrospektive Analyse der GeparQuattro-Studie (N=156) Loi S, et al. SABCS 2013 (S1-05), oral presentation
16 Stromale TILs scheinen prädiktiv für eine Sensitivität gegenüber der adjuvanten CT aber nicht gegenüber Trastuzumab Retrospektive Analyse der N9831-Studie (N=945; 9,9% 60% stils = LPBC): TILs scheinen nicht prädiktiv für eine adjuvante Trastuzumabwirkung Perez EA, et al. SABCS 2014 (S1-06), oral pesentation
17 Neoadjuvant Systemic Therapy Response Prediction II Version Factor LoE 2009 CTS GR AGO Multigene signature III C B +/- Ki-67 I B A + Tumour infiltrating lymphocytes I B B +/- PIK3CA mutation II B B +/-
18 PIK3CA-Mutation und PTEN-Verlust scheinen prädiktiv für eine Resistenz auf CT + Anti-HER2-Therapie neoadjuvant Loibl S, et al. J Clin Oncol 2014; Baselga J, et al. ECC 2013; Guarneri V, et al. ESMO 2014
19 PIK3CA-Mutation scheinen nicht prädiktiv für eine Resistenz auf Trastuzumab adjuvant (retrospektive Analyse der FinHER-Studie, N=157) Loi S, et al. J Natl Cancer Inst 105: , 2013
20 Neoadjuvant Targeted Therapy in HER2 Positive Tumors Version Oxford / AGO LoE / GR Trastuzumab in combination with chemotherapy 1b A ++ Lapatinib in combination with chemotherapy 1a B - Lapatinib + Trastuzumab in combination with chemotherapy 1a B +/- Pertuzumab + Trastuzumab in combination with chemotherapy 1a B +* Two anti-her2 agents without chemotherapy 2b B +/- Anti-HER2 agent in combination with endocrine treatment 2b C +/- * Study participation recommended
21 Anteil Patienten mit pcr (%) Verbesserung der pcr-raten (ypt0/is ypn0) durch duale Anti-HER2 Therapie beim HER2+ EBC pcr pcr HR+ pcr HR Cortazar. Lancet 2014; 2 Carey. ASCO 2013; 3 Baselga. Lancet 2012; 4 Robidoux. Lancet Oncol 2013; 5 Von Minckwitz. Lancet Oncol 2014; 6 Gianni. Lancet Oncol 2012; 7 Schneeweiss. Ann Oncol 2013
22 Anteil Patienten mit pcr (%) pcr-raten (ypt0/is ypn0) beim HER2+ EBC Getrennt nach HR-Status % pcr pcr HR+ pcr HR HER2+ HR- HR Cortazar. Lancet 2014; 2 Carey. ASCO 2013; 3 Baselga. Lancet 2012; 4 Robidoux. Lancet Oncol 2013; 5 Von Minckwitz. Lancet Oncol 2014; 6 Gianni. Lancet Oncol 2012; 7 Schneeweiss. Ann Oncol 2013
23 Neoadjuvant Targeted Therapy in HER2 Positive Tumors Version Oxford / AGO LoE / GR Trastuzumab in combination with chemotherapy 1b A ++ Lapatinib in combination with chemotherapy 1a B - Lapatinib + Trastuzumab in combination with chemotherapy 1a B +/- Pertuzumab + Trastuzumab in combination with chemotherapy 1a B +* Two anti-her2 agents without chemotherapy 2b B +/- Anti-HER2 agent in combination with endocrine treatment 2b C +/- * Study participation recommended
24 Neoadjuvant Targeted Therapy in HER2 Negative Tumors Version Bevacizumab in combination with chemotherapy Oxford / AGO LoE / GR In hormone receptor positive BC 1b B - In TNBC 1b B +/-
25 GeparQuinto (Phase III): Neoadj. EC Doc vs. ECB DocBev beim HER2- EBC 2cm (N=1.925) Überleben Disease-free Survival Overall Survival Gerber B et al. SABCS 2014 (P ), poster discussion
26 Keine Verbesserung des invasiven DFS durch Bevacizumab adjuvant beim TNBC Studie N (TNBC) Therapie Primärer Endpunkt HR p BEATRICE x CT± B B (1y) E AC-P ± B B (1y) IDFS 0,87 0,18 IDFS 0,77 0,08 AC, Doxorubicin/Cyclophosphamid; B, Bevacizumab; CT, Chemotherapie; HR, hazard ratio; IDFS, invasive disease-free survival; P, Paclitaxel 38% TNBC Cameron D, et al. Lancet Oncol 2013; Miller K, et al. ASCO 2014 (abs 500), oral abstract session
27 Neoadjuvant Systemic Chemotherapy Clinical Benefit Version Oxford / AGO LoE / GR Survival is similar after neoadjuvant (preoperative, primary) and adjuvant systemic therapy 1a A Pathological complete response is associated with improved survival in particular subgroups 1b A Can achieve operability in primary inoperable tumors 1b A ++ Improved options for breast conserving surgery 1b A ++ Allows individualization of therapy according to mid-course treatment effect 1b B +* Allows individualization of post-neoadjuvant management according to refined risk assessment after neoadjuvant treatment and surgery 2b B +/-* * Study participation recommended
28 Adjuvant Systemic Therapy after Neoadjuvant Systemic Treatment Version Endocrine treatment in endocrine responsive disease 1a A ++ Complete trastuzumab treatment for 1 year in HER2-positive disease 2b B ++ In case of insufficient response Oxford / AGO LoE / GR Further chemotherapy 3 C - Experimental therapies in clinical trials 5 D +
29 KATHERINE (Phase III): Studiendesign und Endpunkte Operation ct1-4, cn0-3, M0 HER2+ Neoadjuvante Therapie: Anti-HER2/ Taxan ± Anthrazyklin Residualer invasiver Tumor 14 x Trastuzumab 6mg/kg i.v. q3w 14 x T-DM1 3,6mg/kg i.v. q3w Bestrahlung gemäß Leitlinien; endokrine Therapie falls ER/PgR pos Primärer Endpunkt: Invasives Disease-free Survival (IDFS); 3y-IDFS 70% 76,5% (HR 0,75) Sekundäre Endpunkte: IDFS, inklusive Zweitkarzinome; DFS; OS; distantes Rezidiv-freies Interval (DRFI) PI: G. von Minckwitz
30 PENELOPE B (Phase III): Studiendesign und primärer Endpunkt Operation ct1-4, cn0-3, M0 HR+ HER2- Neoadjuvante Chemotherapie inklusive Taxan Residualer invasiver Tumor mit CPS-EG Score 3 13 x Placebo p.o. d1-21 q4w 13 x Palbociclib 125mg p.o. d1-21 q4w Bestrahlung und endokrine Therapie gemäß Leitlinien Primärer Endpunkt: Invasives Disease-free Survival (IDFS) PI: G. von Minckwitz
31 Neoadjuvant Systemic Therapy Version Vielen Dank für Ihre Aufmerksamkeit
32 Neoadjuvant Systemic Chemotherapy Response Prediction I Version Factor CTS LoE Ox2001 GR AGO Young age B 1a A + ct1 / ct2 tumors o. N0 o. G3 B 1a A ++ Negative ER and PgR status B 1a A ++ Triple negative breast cancer (TNBC) B 1a A ++ Positive HER2 status B 1a A ++ Non-lobular tumor type B 1a A + Early clinical response B 1b A +
33 Neoadjuvant Systemic Chemotherapy Recommended Methods of Monitoring of Response Version Oxford / AGO LoE / GR Breast ultrasound 2b B ++ Palpation 2b B ++ Mammography 2b B ++ MRI 2b B + PET(-CT) 2b B +/- Clip tumour region 5 D ++
34 Neoadjuvant Systemic Therapy Procedures in Case of Early Response Version In case of early response following 6 to 12 weeks of neoadjuvant chemotherapy: Oxford / AGO LoE / GR Complete all chemotherapy before surgery i.e. 18 weeks of treatment 1b A ++ In case of response after 2 cycles of DAC in HR positive breast cancer consider 8 instead of 6 cycles of DAC 2b C +
35 Neoadjuvant Systemic Therapy Procedures in Case of No Early Response Version In case of no change: Completion of NST, followed by surgery 2b C ++ Continuation of NST with non cross-resistant regimen 2b B + AC or EC x 4 D x 4 or Pw x 12 2b B + DAC x 2 NX x 4 1b B + In case of progressive disease: Oxford / AGO LoE / GR Stop of NST and immediate surgery or radiotherapy 4 D ++* Additional adjuvant chemotherapy with non cross-resistant regimen 4 D +/-* * Study participation recommended
36 Local/Regional Procedure after Neoadjuvant Therapy Version Oxford / AGO LoE / GR Mark previous tumor region 5 D ++ Surgery 2b C ++ Microscopically clear margins 5 D ++ Tumor resection in the new margins 3b C + Sentinel node biopsy (see chapter Surgery )
37 Surgical Procedure of the Axilla Before or After NACT Version SLNB before or after NACT in cn0 SLNB before NACT SLNB after NACT Oxford / AGO LoE / GR 2b 2a B B + +/- Further surgical procedures depending on SLNB cn-status (before NST) pn-status (before NST) cn-status (after NST) Surgical procedure cn0 pn0(sn) - nihil 1a A + cn0 cn0 pn+(sn) analogue ACOZOG pn+(sn) not analogue ACOZOG cn+ cn+ (CNB/FNA) ycn0 ycn0 ALND 3 B +/- ycn0 ALND 2b B + SNB ALND 2a 2b B B +/- + ycn+ (CNB/FNA) ALND 2b B ++
38 Neoadjuvant Systemic Therapy Indications for Mastectomy Version Oxford / AGO LoE / GR Positive margins after repeated excisions 3b C ++ Radiotherapy not feasible 5 D ++ In case of clinical complete response Inflammatory breast cancer 2b C + In case of pcr +/- Multicentric lesions 2b C +/- ct4a-c breast cancer 2b B +/-
39 Neoadjuvant Systemic Therapy Timing of Surgery and Radiotherapy Version Oxford / AGO LoE / GR Surgery 4 C ++ After the nadir of the leucocyte count (2 to 4 weeks after last course of chemotherapy) Radiotherapy after surgery 2b B weeks after surgery BCS
40 Neoadjuvant Endocrine Therapy in patients with endocrine-responsive breast cancer Version Postmenopausal patients: Oxford / AGO LoE / GR who are inoperable and can / will not receive chemotherapy 2a B + Optimizes the option for breast conserving therapy 1b A + Aromatase inhibitors (for > 3 months) 1a a B + Aromatase inhibitor + lapatinib (HER2+ BC) 2b B +/- Premenopausal patients who are inoperable and can / will not receive chemotherapy 5 C + Tamoxifen 2b C + Aromatase inhibitors + LHRH 1b C +/- Concurrent chemo-endocrine therapy 1b A - Prognostic factors during/after NST: quantitative ERexpression, level of Ki-67, N status, T status 1b B + Optimal duration of neoadjuvant endocrine therapy is unknown No long term results for neoadjuvant endocrine therapy (vs. adjuvant endocrine therapy)
Neoadjuvant (Primary) Systemic Therapy
 Diagnosis and Treatment of Patients with Primary and Metastatic Breast Cancer AGO e. V. in der DGGG e.v. sowie in der DKG e.v. Guidelines Breast Version 2015.1 Neoadjuvant (Primary) Systemic Therapy Neoadjuvant
Diagnosis and Treatment of Patients with Primary and Metastatic Breast Cancer AGO e. V. in der DGGG e.v. sowie in der DKG e.v. Guidelines Breast Version 2015.1 Neoadjuvant (Primary) Systemic Therapy Neoadjuvant
Neoadjuvant (Primary) Systemic Therapy
 Diagnosis and Treatment of Patients with Primary and Metastatic Breast Cancer AGO e. V. in der DGGG e.v. sowie in der DKG e.v. Guidelines Breast Version 2016.1 Neoadjuvant (Primary) Systemic Therapy Neoadjuvant
Diagnosis and Treatment of Patients with Primary and Metastatic Breast Cancer AGO e. V. in der DGGG e.v. sowie in der DKG e.v. Guidelines Breast Version 2016.1 Neoadjuvant (Primary) Systemic Therapy Neoadjuvant
The current treatment landscape for early breast cancer: Advances in cytotoxic and endocrine treatment
 The current treatment landscape for early breast cancer: Advances in cytotoxic and endocrine treatment Ahmad Awada, MD, PhD Head of Medical Oncology Clinic Institut Jules Bordet Université Libre de Bruxelles
The current treatment landscape for early breast cancer: Advances in cytotoxic and endocrine treatment Ahmad Awada, MD, PhD Head of Medical Oncology Clinic Institut Jules Bordet Université Libre de Bruxelles
Breast Cancer Update 2014 Prevention, Risk, and Treatment of Early Stage Breast Cancer. Kevin R. Fox, MD University of Pennsylvania
 Breast Cancer Update 2014 Prevention, Risk, and Treatment of Early Stage Breast Cancer Kevin R. Fox, MD University of Pennsylvania Prevention of Breast Cancer Accepted treatments Tamoxifen (premenopausal
Breast Cancer Update 2014 Prevention, Risk, and Treatment of Early Stage Breast Cancer Kevin R. Fox, MD University of Pennsylvania Prevention of Breast Cancer Accepted treatments Tamoxifen (premenopausal
Cytotoxic Therapy in Metastatic Breast Cancer
 Diagnosis and Treatment of Patients with Primary and Metastatic Breast Cancer Cytotoxic Therapy in Metastatic Breast Cancer Cytotoxic Therapy in Metastatic Breast Cancer Version 2002: von Minckwitz Versions
Diagnosis and Treatment of Patients with Primary and Metastatic Breast Cancer Cytotoxic Therapy in Metastatic Breast Cancer Cytotoxic Therapy in Metastatic Breast Cancer Version 2002: von Minckwitz Versions
Chemotherapy or Not? Anthracycline or Not? Taxane or Not? Does Density Matter? Chemotherapy in Luminal Breast Cancer: Choice of Regimen.
 Chemotherapy in Luminal Breast Cancer: Choice of Regimen Andrew D. Seidman, MD Attending Physician Breast Cancer Medicine Service Memorial Sloan Kettering Cancer Center Professor of Medicine Weill Cornell
Chemotherapy in Luminal Breast Cancer: Choice of Regimen Andrew D. Seidman, MD Attending Physician Breast Cancer Medicine Service Memorial Sloan Kettering Cancer Center Professor of Medicine Weill Cornell
OI PARP ΑΝΑΣΤΟΛΕΙΣ ΣΤΟΝ ΚΑΡΚΙΝΟ ΤΟΥ ΜΑΣΤΟΥ ΝΙΚΟΛΑΙΔΗ ΑΔΑΜΑΝΤΙΑ ΠΑΘΟΛΟΓΟΣ-ΟΓΚΟΛΟΓΟΣ Β ΟΓΚΟΛΟΓΙΚΗ ΚΛΙΝΙΚΗ ΝΟΣ. ΜΗΤΕΡΑ
 OI PARP ΑΝΑΣΤΟΛΕΙΣ ΣΤΟΝ ΚΑΡΚΙΝΟ ΤΟΥ ΜΑΣΤΟΥ ΝΙΚΟΛΑΙΔΗ ΑΔΑΜΑΝΤΙΑ ΠΑΘΟΛΟΓΟΣ-ΟΓΚΟΛΟΓΟΣ Β ΟΓΚΟΛΟΓΙΚΗ ΚΛΙΝΙΚΗ ΝΟΣ. ΜΗΤΕΡΑ Study Overview Inhibition of poly(adenosine diphosphate [ADP]-ribose) polymerase
OI PARP ΑΝΑΣΤΟΛΕΙΣ ΣΤΟΝ ΚΑΡΚΙΝΟ ΤΟΥ ΜΑΣΤΟΥ ΝΙΚΟΛΑΙΔΗ ΑΔΑΜΑΝΤΙΑ ΠΑΘΟΛΟΓΟΣ-ΟΓΚΟΛΟΓΟΣ Β ΟΓΚΟΛΟΓΙΚΗ ΚΛΙΝΙΚΗ ΝΟΣ. ΜΗΤΕΡΑ Study Overview Inhibition of poly(adenosine diphosphate [ADP]-ribose) polymerase
Update on neoadjuvant treatment of breast cancer
 Update on neoadjuvant treatment of breast cancer «IS PATHOLOGIC COMPLETE RESPONSE STILL A GOOD SURROGATE OF SURVIVAL?» Complete histological response varies according to tumoral type pcr (%) 40 35 30 25
Update on neoadjuvant treatment of breast cancer «IS PATHOLOGIC COMPLETE RESPONSE STILL A GOOD SURROGATE OF SURVIVAL?» Complete histological response varies according to tumoral type pcr (%) 40 35 30 25
Loco-regional Recurrence
 Diagnosis and Treatment of Patients with Primary and Metastatic Breast Cancer AGO AGO e. e. V. V. Loco-regional Recurrence Loco-regional Recurrence Version 2002: Brunnert / Simon Versions 2003 2012: Audretsch
Diagnosis and Treatment of Patients with Primary and Metastatic Breast Cancer AGO AGO e. e. V. V. Loco-regional Recurrence Loco-regional Recurrence Version 2002: Brunnert / Simon Versions 2003 2012: Audretsch
ESMO 2014 Summary Breast Cancer
 ESMO 2014 Summary Breast Cancer 1 7. 1 0. 2 0 1 4 A N NA D U R I G OVA M E D I C A L O N CO LO GY U N I V E R S I T Y H O S P I TA L S O F G E N E VA Outline 1. Early Breast Cancer Her2+ Neoadjuvant: Lapatax
ESMO 2014 Summary Breast Cancer 1 7. 1 0. 2 0 1 4 A N NA D U R I G OVA M E D I C A L O N CO LO GY U N I V E R S I T Y H O S P I TA L S O F G E N E VA Outline 1. Early Breast Cancer Her2+ Neoadjuvant: Lapatax
Breast Cancer Educational Program. June 5-6, 2015
 Breast Cancer Educational Program June 5-6, 2015 Adjuvant Systemic Therapy For Early Breast Cancer: Who, What and for How Long? Debjani Grenier MD, FRCPC Medical Oncologist Disclosures Advisory Board Member:
Breast Cancer Educational Program June 5-6, 2015 Adjuvant Systemic Therapy For Early Breast Cancer: Who, What and for How Long? Debjani Grenier MD, FRCPC Medical Oncologist Disclosures Advisory Board Member:
Treatment of Metastatic Breast Cancer: Endocrine Therapies. Robert W. Carlson, M.D. Professor of Medicine Stanford University
 Treatment of Metastatic Breast Cancer: Endocrine Therapies Robert W. Carlson, M.D. Professor of Medicine Stanford University MDACC Experience with FAC in Chemotherapy-Naive MBC Greenberg et al, J Clin
Treatment of Metastatic Breast Cancer: Endocrine Therapies Robert W. Carlson, M.D. Professor of Medicine Stanford University MDACC Experience with FAC in Chemotherapy-Naive MBC Greenberg et al, J Clin
SAMO FoROMe Post-ESMO 2013 Breast Cancer
 SAMO FoROMe Post-ESMO 2013 Breast Cancer Dr. med. Manuela Rabaglio Klinik und Poliklinik für Medizinische Onkologie Breast Cancer Track 300 Abstracts 142 Poster 11 Proffered paper 4 late breaking news
SAMO FoROMe Post-ESMO 2013 Breast Cancer Dr. med. Manuela Rabaglio Klinik und Poliklinik für Medizinische Onkologie Breast Cancer Track 300 Abstracts 142 Poster 11 Proffered paper 4 late breaking news
Advances in Neoadjuvant and Adjuvant Therapy
 Advances in Neoadjuvant and Adjuvant Therapy Kathy S. Albain, MD, FACP Director, Breast Clinical Research Program Co-Director, Breast Oncology Center Director of the Thoracic Oncology Program Professor
Advances in Neoadjuvant and Adjuvant Therapy Kathy S. Albain, MD, FACP Director, Breast Clinical Research Program Co-Director, Breast Oncology Center Director of the Thoracic Oncology Program Professor
New Approval Mechanism for Breast Cancer using pathologic Complete Response
 New Approval Mechanism for Breast Cancer using pathologic Complete Response Sandra M. Swain, MD, FACP Medical Director, Washington Cancer Institute MedStar Washington Hospital Center Professor of Medicine
New Approval Mechanism for Breast Cancer using pathologic Complete Response Sandra M. Swain, MD, FACP Medical Director, Washington Cancer Institute MedStar Washington Hospital Center Professor of Medicine
Basics and limitations of adjuvant online an internet based decision tool
 Basics and limitations of adjuvant online an internet based decision tool J. Huober SAKK, Bern 31.10.2013 Univ.-Frauenklinik Ulm Integratives Tumorzentrum des Universitätsklinikums und der Medizinischen
Basics and limitations of adjuvant online an internet based decision tool J. Huober SAKK, Bern 31.10.2013 Univ.-Frauenklinik Ulm Integratives Tumorzentrum des Universitätsklinikums und der Medizinischen
Clinical Management Protocol Chemotherapy Breast Cancer. Protocol for Planning and Treatment
 Protocol for Planning and Treatment The process to be followed when a course of chemotherapy is required to treat: BREAST CANCER Patient information given at each stage following agreed information pathway
Protocol for Planning and Treatment The process to be followed when a course of chemotherapy is required to treat: BREAST CANCER Patient information given at each stage following agreed information pathway
Metastatic Breast Cancer 201. Carolyn B. Hendricks, MD October 29, 2011
 Metastatic Breast Cancer 201 Carolyn B. Hendricks, MD October 29, 2011 Overview Is rebiopsy necessary at the time of recurrence or progression of disease? How dose a very aggressive treatment upfront compare
Metastatic Breast Cancer 201 Carolyn B. Hendricks, MD October 29, 2011 Overview Is rebiopsy necessary at the time of recurrence or progression of disease? How dose a very aggressive treatment upfront compare
Breast Cancer Treatment Guidelines
 Breast Cancer Treatment Guidelines DCIS Stage 0 TisN0M0 Tamoxifen for 5 years for patients with ER positive tumors treated with: -Breast conservative therapy (lumpectomy) and radiation therapy -Excision
Breast Cancer Treatment Guidelines DCIS Stage 0 TisN0M0 Tamoxifen for 5 years for patients with ER positive tumors treated with: -Breast conservative therapy (lumpectomy) and radiation therapy -Excision
New Treatment Options for Breast Cancer
 New Treatment Options for Breast Cancer Brandon Vakiner, PharmD., BCOP Clinical Pharmacy Specialist - Oncology The University of Iowa Hospitals and Clinics Assistant Professor (Clinical) University of
New Treatment Options for Breast Cancer Brandon Vakiner, PharmD., BCOP Clinical Pharmacy Specialist - Oncology The University of Iowa Hospitals and Clinics Assistant Professor (Clinical) University of
Adjuvant Therapy with Trastuzumab
 Adjuvant Therapy with Trastuzumab Hiroji Iwata, M.D. Department of Breast Oncology, Aichi Cancer Center Hospital Although this presentation includes information regarding pharmaceuticals (including products
Adjuvant Therapy with Trastuzumab Hiroji Iwata, M.D. Department of Breast Oncology, Aichi Cancer Center Hospital Although this presentation includes information regarding pharmaceuticals (including products
Neoadjuvant Breast Cancer Therapy and Drug Development
 Neoadjuvant Breast Cancer Therapy and Drug Development Patricia Cortazar, MD, and Paul G. Kluetz, MD The authors are affiliated with the Office of Hematology and Oncology Products at the Center for Drug
Neoadjuvant Breast Cancer Therapy and Drug Development Patricia Cortazar, MD, and Paul G. Kluetz, MD The authors are affiliated with the Office of Hematology and Oncology Products at the Center for Drug
La Terapia Personalizzata in Oncologia
 AZIENDA OSPEDALIERO-UNIVERSITARIA DI MODENA La Terapia Personalizzata in Oncologia Roma, 25-26 Ottobre 2011 Stato dell arte e prospettive della Target Therapy nei tumori mammari PierFranco Conte Department
AZIENDA OSPEDALIERO-UNIVERSITARIA DI MODENA La Terapia Personalizzata in Oncologia Roma, 25-26 Ottobre 2011 Stato dell arte e prospettive della Target Therapy nei tumori mammari PierFranco Conte Department
trastuzumab, 600mg/5mL solution for injection (Herceptin ) SMC No. (928/13) Roche Products Ltd
 trastuzumab, 600mg/5mL solution for injection (Herceptin ) SMC No. (928/13) Roche Products Ltd 06 December 2013 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product
trastuzumab, 600mg/5mL solution for injection (Herceptin ) SMC No. (928/13) Roche Products Ltd 06 December 2013 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product
Jennifer Diamond M.D. Assistant Professor Developmental Therapeutics and Breast Oncology University of Colorado Anschutz Medical Campus SUMO Fall
 Jennifer Diamond M.D. Assistant Professor Developmental Therapeutics and Breast Oncology University of Colorado Anschutz Medical Campus SUMO Fall Meeting September 26, 2015 To understand the biology and
Jennifer Diamond M.D. Assistant Professor Developmental Therapeutics and Breast Oncology University of Colorado Anschutz Medical Campus SUMO Fall Meeting September 26, 2015 To understand the biology and
What is the optimal sequence of anti-her2 therapy in metastatic breast cancer?
 What is the optimal sequence of anti-her2 therapy in metastatic breast cancer? David Miles Mount Vernon Cancer Centre Northwood Middlesex UKBCM mee)ng: London 2013 Herceptin plus a taxoid extends survival
What is the optimal sequence of anti-her2 therapy in metastatic breast cancer? David Miles Mount Vernon Cancer Centre Northwood Middlesex UKBCM mee)ng: London 2013 Herceptin plus a taxoid extends survival
La Chemioterapia Adiuvante Dose-Dense. Lo studio GIM 2. Alessandra Fabi
 La Chemioterapia Adiuvante Dose-Dense Lo studio GIM 2 Alessandra Fabi San Antonio Breast Cancer Symposium -December 10-14, 2013 GIM 2 study Epirubicin and Cyclophosphamide (EC) followed by Paclitaxel (T)
La Chemioterapia Adiuvante Dose-Dense Lo studio GIM 2 Alessandra Fabi San Antonio Breast Cancer Symposium -December 10-14, 2013 GIM 2 study Epirubicin and Cyclophosphamide (EC) followed by Paclitaxel (T)
Avastin in breast cancer: Summary of clinical data
 Avastin in breast cancer: Summary of clinical data Worldwide, over one million people are diagnosed with breast cancer every year 1. It is the most frequently diagnosed cancer in women 1,2, and the leading
Avastin in breast cancer: Summary of clinical data Worldwide, over one million people are diagnosed with breast cancer every year 1. It is the most frequently diagnosed cancer in women 1,2, and the leading
Pathologic Assessment Of The Breast And Axilla After Preoperative Therapy
 Pathologic Assessment Of The Breast And Axilla After Preoperative Therapy W. Fraser Symmans, M.D. Associate Professor of Pathology UT M.D. Anderson Cancer Center Pathologic Complete Response (pcr) Proof
Pathologic Assessment Of The Breast And Axilla After Preoperative Therapy W. Fraser Symmans, M.D. Associate Professor of Pathology UT M.D. Anderson Cancer Center Pathologic Complete Response (pcr) Proof
Gynäkologische Onkologie-Klinische Studien
 Gynäkologische Onkologie-Klinische Studien Breast cancer A randomized, phase 2 trial of AEZS-108 in chemotherapy refractory triple negative (ER/PR/HER2-negative) LHRH-R positive metastatic breast cancer
Gynäkologische Onkologie-Klinische Studien Breast cancer A randomized, phase 2 trial of AEZS-108 in chemotherapy refractory triple negative (ER/PR/HER2-negative) LHRH-R positive metastatic breast cancer
Integrating Chemotherapy and Liver Surgery for the Management of Colorectal Metastases
 I Congresso de Oncologia D Or July 5-6, 2013 Integrating Chemotherapy and Liver Surgery for the Management of Colorectal Metastases Michael A. Choti, MD, MBA, FACS Department of Surgery Johns Hopkins University
I Congresso de Oncologia D Or July 5-6, 2013 Integrating Chemotherapy and Liver Surgery for the Management of Colorectal Metastases Michael A. Choti, MD, MBA, FACS Department of Surgery Johns Hopkins University
Miquel Àngel Seguí Palmer
 Miquel Àngel Seguí Palmer HER2+ Breast Cancer is characterized by overexpression of HER2 receptors HER2+ Breast Cancer is characterized by overexpression of HER2 receptors HER2+ status is associated with
Miquel Àngel Seguí Palmer HER2+ Breast Cancer is characterized by overexpression of HER2 receptors HER2+ Breast Cancer is characterized by overexpression of HER2 receptors HER2+ status is associated with
Inteligentaj decidoj... Intelligente Entscheide bei der adjuvanten Therapie des Mammakarzinoms. Intelligent Questions?
 Intelligente Entscheide bei der adjuvanten Therapie des Mammakarzinoms Stefan Aebi Universitätsspital Bern, Inselspital Klinik für Medizinische Onkologie und Brust /Tumorzentrum der Frauenklinik Inteligentaj
Intelligente Entscheide bei der adjuvanten Therapie des Mammakarzinoms Stefan Aebi Universitätsspital Bern, Inselspital Klinik für Medizinische Onkologie und Brust /Tumorzentrum der Frauenklinik Inteligentaj
GUIDELINES ADJUVANT SYSTEMIC BREAST CANCER
 GUIDELINES ADJUVANT SYSTEMIC BREAST CANCER Author: Dr Susan O Reilly On behalf of the Breast CNG Written: December 2008 Agreed at CNG: June 2009 & June 2010 Review due: June 2011 Guidelines Adjuvant Systemic
GUIDELINES ADJUVANT SYSTEMIC BREAST CANCER Author: Dr Susan O Reilly On behalf of the Breast CNG Written: December 2008 Agreed at CNG: June 2009 & June 2010 Review due: June 2011 Guidelines Adjuvant Systemic
Avastin in breast cancer: Summary of clinical data
 Avastin in breast cancer: Summary of clinical data Worldwide, over one million people are diagnosed with breast cancer every year 1. It is the most frequently diagnosed cancer in women 1,2, and the leading
Avastin in breast cancer: Summary of clinical data Worldwide, over one million people are diagnosed with breast cancer every year 1. It is the most frequently diagnosed cancer in women 1,2, and the leading
U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research (CDER)
 Guidance for Industry Pathological Complete Response in Neoadjuvant Treatment of High-Risk Early-Stage Breast Cancer: Use as an Endpoint to Support Accelerated Approval U.S. Department of Health and Human
Guidance for Industry Pathological Complete Response in Neoadjuvant Treatment of High-Risk Early-Stage Breast Cancer: Use as an Endpoint to Support Accelerated Approval U.S. Department of Health and Human
BREAST CANCER UPDATE C H R I S S Z Y A R T O, D O G E N E S E E H E M A T O L O G Y O N C O L O G Y F L I N T, M I
 BREAST CANCER UPDATE C H R I S S Z Y A R T O, D O G E N E S E E H E M A T O L O G Y O N C O L O G Y F L I N T, M I Overview Why is it important to understand breast cancer? Choosing wisely Appropriateness
BREAST CANCER UPDATE C H R I S S Z Y A R T O, D O G E N E S E E H E M A T O L O G Y O N C O L O G Y F L I N T, M I Overview Why is it important to understand breast cancer? Choosing wisely Appropriateness
The Impact of Taxotere on Adjuvant Breast Cancer
 The Impact of Taxotere on Adjuvant Breast Cancer a report by Pierre Fumoleau and Henri Roché Centre Georges François Leclerc, Dijon, and Institut Claudius Regaud, Toulouse, France DOI: 10.17925/EOH.2005.0.0.1l
The Impact of Taxotere on Adjuvant Breast Cancer a report by Pierre Fumoleau and Henri Roché Centre Georges François Leclerc, Dijon, and Institut Claudius Regaud, Toulouse, France DOI: 10.17925/EOH.2005.0.0.1l
Positività per HER-2 nei carcinomi subcentimetrici
 Positività per HER-2 nei carcinomi Antonella Ferro U.O. Oncologia Medica Trento Small Tumors Small tumors are becoming increasingly common with the use of mammography > screening Some of these tumors,
Positività per HER-2 nei carcinomi Antonella Ferro U.O. Oncologia Medica Trento Small Tumors Small tumors are becoming increasingly common with the use of mammography > screening Some of these tumors,
Management of stage III A-B of NSCLC. Hamed ALHusaini Medical Oncologist
 Management of stage III A-B of NSCLC Hamed ALHusaini Medical Oncologist Global incidence, CA cancer J Clin 2011;61:69-90 Stage III NSCLC Includes heterogeneous group of patients with differences in the
Management of stage III A-B of NSCLC Hamed ALHusaini Medical Oncologist Global incidence, CA cancer J Clin 2011;61:69-90 Stage III NSCLC Includes heterogeneous group of patients with differences in the
Annals of Oncology Advance Access published May 4, 2015
 Annals of Oncology Advance Access published May 4, 2015 1 Tailoring therapies - improving the management of early breast cancer: St GallenInternational Expert Consensus on the Primary Therapy of Early
Annals of Oncology Advance Access published May 4, 2015 1 Tailoring therapies - improving the management of early breast cancer: St GallenInternational Expert Consensus on the Primary Therapy of Early
Progress in Treating Advanced Triple Negative Breast Cancer
 Progress in Treating Advanced Triple Negative Breast Cancer Lisa A. Carey, M.D. University of North Carolina at Chapel Hill Lineberger Comprehensive Cancer Center Triple Negative Breast Cancer by Subtype
Progress in Treating Advanced Triple Negative Breast Cancer Lisa A. Carey, M.D. University of North Carolina at Chapel Hill Lineberger Comprehensive Cancer Center Triple Negative Breast Cancer by Subtype
What is breast cancer?
 Breast Cancer What is breast cancer? Let us explain it to you. www.anticancerfund.org www.esmo.org ESMO/ACF Patient Guide Series based on the ESMO Clinical Practice Guidelines BREAST CANCER: A GUIDE FOR
Breast Cancer What is breast cancer? Let us explain it to you. www.anticancerfund.org www.esmo.org ESMO/ACF Patient Guide Series based on the ESMO Clinical Practice Guidelines BREAST CANCER: A GUIDE FOR
Adjuvant Endocrine Therapy in Breast Cancer: 2015 Update
 Adjuvant Endocrine Therapy in Breast Cancer: 2015 Update Shannon Puhalla, MD Director, Breast Cancer Clinical Research Program Magee Womens Cancer Program University of Pittsburgh Cancer Institute Questions
Adjuvant Endocrine Therapy in Breast Cancer: 2015 Update Shannon Puhalla, MD Director, Breast Cancer Clinical Research Program Magee Womens Cancer Program University of Pittsburgh Cancer Institute Questions
Cellular, Molecular, and Biochemical Targets in Breast Cancer
 Cellular, Molecular, and Biochemical Targets in Breast Cancer Kristy Kummerow Ingrid Meszoely December 12, 2012 VUMC Resident Bonus Conference One size fits all surgical treatment of breast cancer Wilhelm
Cellular, Molecular, and Biochemical Targets in Breast Cancer Kristy Kummerow Ingrid Meszoely December 12, 2012 VUMC Resident Bonus Conference One size fits all surgical treatment of breast cancer Wilhelm
Management of Postmenopausal Women with T1 ER+ Tumors: Options and Tradeoffs. Case Study. Surgery. Lumpectomy and Radiation
 Management of Postmenopausal Women with T1 ER+ Tumors: Options and Tradeoffs Michael Alvarado, MD Associate Professor of Surgery University of California San Francisco Case Study 59 yo woman with new palpable
Management of Postmenopausal Women with T1 ER+ Tumors: Options and Tradeoffs Michael Alvarado, MD Associate Professor of Surgery University of California San Francisco Case Study 59 yo woman with new palpable
Metastatic Breast Cancer: The Art and Science of Systemic Therapy. Vallerie Gordon MD, FRCPC Medical Oncologist CancerCare Manitoba
 Metastatic Breast Cancer: The Art and Science of Systemic Therapy Vallerie Gordon MD, FRCPC Medical Oncologist CancerCare Manitoba Presenter Disclosure Faculty: Dr. Vallerie Gordon Relationships with commercial
Metastatic Breast Cancer: The Art and Science of Systemic Therapy Vallerie Gordon MD, FRCPC Medical Oncologist CancerCare Manitoba Presenter Disclosure Faculty: Dr. Vallerie Gordon Relationships with commercial
Qu avons-nous appris du développement des anti-her2? Ahmad Awada MD, PhD Medical Oncology Clinic Institut Jules Bordet Université Libre de Bruxelles
 Qu avons-nous appris du développement des anti-her2? Ahmad Awada MD, PhD Medical Oncology Clinic Institut Jules Bordet Université Libre de Bruxelles FOM Lille 2013 1 Her2 breast cancer expression = Poor
Qu avons-nous appris du développement des anti-her2? Ahmad Awada MD, PhD Medical Oncology Clinic Institut Jules Bordet Université Libre de Bruxelles FOM Lille 2013 1 Her2 breast cancer expression = Poor
Van Cutsem E et al. Proc ASCO 2009;Abstract LBA4509.
 Efficacy Results from the ToGA Trial: A Phase III Study of Trastuzumab Added to Standard Chemotherapy in First-Line HER2- Positive Advanced Gastric Cancer Van Cutsem E et al. Proc ASCO 2009;Abstract LBA4509.
Efficacy Results from the ToGA Trial: A Phase III Study of Trastuzumab Added to Standard Chemotherapy in First-Line HER2- Positive Advanced Gastric Cancer Van Cutsem E et al. Proc ASCO 2009;Abstract LBA4509.
One of the most mature trials that examined PROCEEDINGS. Hormone Therapy in Postmenopausal Women With Breast Cancer * William J.
 Hormone Therapy in Postmenopausal Women With Breast Cancer * William J. Gradishar, MD ABSTRACT *Based on a presentation given by Dr Gradishar at a roundtable symposium held in Baltimore on June 28, 25.
Hormone Therapy in Postmenopausal Women With Breast Cancer * William J. Gradishar, MD ABSTRACT *Based on a presentation given by Dr Gradishar at a roundtable symposium held in Baltimore on June 28, 25.
In treating triple negative breast cancer,
 Treatment of triple negative breast cancer Triple negative breast cancers, as a subgroup, are associated with a poor prognosis. But different subtypes within triple negative disease are associated with
Treatment of triple negative breast cancer Triple negative breast cancers, as a subgroup, are associated with a poor prognosis. But different subtypes within triple negative disease are associated with
Prognostic Factors for Triple-Negative Breast Cancer Patients Receiving Preoperative Systemic Chemotherapy
 Original Study Prognostic Factors for Triple-Negative Breast Cancer Patients Receiving Preoperative Systemic Chemotherapy Sota Asaga, Takayuki Kinoshita, Takashi Hojo, Junko Suzuki, Kenjiro Jimbo, Hitoshi
Original Study Prognostic Factors for Triple-Negative Breast Cancer Patients Receiving Preoperative Systemic Chemotherapy Sota Asaga, Takayuki Kinoshita, Takashi Hojo, Junko Suzuki, Kenjiro Jimbo, Hitoshi
Drug/Drug Combination: Bevacizumab in combination with chemotherapy
 AHFS Final Determination of Medical Acceptance: Off-label Use of Bevacizumab in Combination with Chemotherapy for the Treatment of Metastatic Breast Cancer Previously Treated with Cytotoxic Chemotherapy
AHFS Final Determination of Medical Acceptance: Off-label Use of Bevacizumab in Combination with Chemotherapy for the Treatment of Metastatic Breast Cancer Previously Treated with Cytotoxic Chemotherapy
San Antonio Breast Cancer Symposium Cancer Therapy and Research Center at UT Health Science Center December 10 14, 2013
 Final Analysis of a Phase II, 3-Arm, Randomized Trial of Neoadjuvant Trastuzumab or Lapatinib or the Combination of Trastuzumab and Lapatinib, Followed by 6 cycles of Docetaxel and Carboplatin with Trastuzumab
Final Analysis of a Phase II, 3-Arm, Randomized Trial of Neoadjuvant Trastuzumab or Lapatinib or the Combination of Trastuzumab and Lapatinib, Followed by 6 cycles of Docetaxel and Carboplatin with Trastuzumab
Treating Patients with Hormone Receptor Positive, HER2 Positive Operable or Locally Advanced Breast Cancer
 Breast Studies Adjuvant therapy after surgery Her 2 positive Breast Cancer B 52 Docetaxel, Carboplatin, Trastuzumab, and Pertuzumab With or Without Estrogen Deprivation in Treating Patients with Hormone
Breast Studies Adjuvant therapy after surgery Her 2 positive Breast Cancer B 52 Docetaxel, Carboplatin, Trastuzumab, and Pertuzumab With or Without Estrogen Deprivation in Treating Patients with Hormone
Everolimus plus exemestane for second-line endocrine treatment of oestrogen receptor positive metastatic breast cancer
 LONDON CANCER NEWS DRUGS GROUP RAPID REVIEW Everolimus plus exemestane for second-line endocrine treatment of oestrogen receptor positive metastatic breast cancer Everolimus plus exemestane for second-line
LONDON CANCER NEWS DRUGS GROUP RAPID REVIEW Everolimus plus exemestane for second-line endocrine treatment of oestrogen receptor positive metastatic breast cancer Everolimus plus exemestane for second-line
Metastatic breast cancer, HER2 overexpression, first-line therapy in combination with a taxane and trastuzumab
 COMPENDIA TRANSPARENCY TRACKING FORM DRUG: Carboplatin INDICATION: Metastatic breast cancer, HER2 overexpression, first-line therapy in combination with a taxane and trastuzumab COMPENDIA TRANSPARENCY
COMPENDIA TRANSPARENCY TRACKING FORM DRUG: Carboplatin INDICATION: Metastatic breast cancer, HER2 overexpression, first-line therapy in combination with a taxane and trastuzumab COMPENDIA TRANSPARENCY
Mechanism Of Action of Palbociclib & PFS Benefit
 A Phase II Randomized Controlled Trial of Palbociclib & Tamoxifen/Fulvestrant in Postmenopausal Women and Men With Hormone-Receptor Positive, HER2- Negative Metastatic Breast Cancer (MBC) Protocol Chair:
A Phase II Randomized Controlled Trial of Palbociclib & Tamoxifen/Fulvestrant in Postmenopausal Women and Men With Hormone-Receptor Positive, HER2- Negative Metastatic Breast Cancer (MBC) Protocol Chair:
Metastatic Breast Cancer...
 DIAGNOSIS: Metastatic Breast Cancer... What Does It Mean For You? A diagnosis of metastatic breast cancer can be frightening. It raises many questions and reminds us of days past when cancer was such a
DIAGNOSIS: Metastatic Breast Cancer... What Does It Mean For You? A diagnosis of metastatic breast cancer can be frightening. It raises many questions and reminds us of days past when cancer was such a
Breast cancer research and a changing treatment pathway
 Breast cancer research and a changing treatment pathway Stuart McIntosh Clinical Senior Lecturer in Surgical Oncology, QUB Consultant Breast Surgeon, BCH What is the breast surgeon s role in 2016? Surgery
Breast cancer research and a changing treatment pathway Stuart McIntosh Clinical Senior Lecturer in Surgical Oncology, QUB Consultant Breast Surgeon, BCH What is the breast surgeon s role in 2016? Surgery
Breast cancer: Diagnosis and complex treatment. Ibolya Czegle MD PhD Semmelweis University 3rd Department of Internal Medicine
 Breast cancer: Diagnosis and complex treatment Ibolya Czegle MD PhD Semmelweis University 3rd Department of Internal Medicine Epidemiology Worldwide, breast cancer is the most frequently diagnosed life-threatening
Breast cancer: Diagnosis and complex treatment Ibolya Czegle MD PhD Semmelweis University 3rd Department of Internal Medicine Epidemiology Worldwide, breast cancer is the most frequently diagnosed life-threatening
Review of Breast Cancer Clinical Trials Conducted by the National Surgical Adjuvant Breast Project
 Surg Clin N Am 87 (2007) 279 305 Review of Breast Cancer Clinical Trials Conducted by the National Surgical Adjuvant Breast Project Lisa A. Newman, MD, MPH, FACS a, *, Eleftherios P. Mamounas, MD, MPH,
Surg Clin N Am 87 (2007) 279 305 Review of Breast Cancer Clinical Trials Conducted by the National Surgical Adjuvant Breast Project Lisa A. Newman, MD, MPH, FACS a, *, Eleftherios P. Mamounas, MD, MPH,
Principles of Radiation Therapy A Bapsi Chakravarthy, MD Associate e P rofessor Professor Radiation Oncology
 Principles of Radiation Therapy A Bapsi Chakravarthy, MD Associate Professor Radiation Oncology Disclosure Information I have no financial relationships to disclose relevant to the conten of this presentation.
Principles of Radiation Therapy A Bapsi Chakravarthy, MD Associate Professor Radiation Oncology Disclosure Information I have no financial relationships to disclose relevant to the conten of this presentation.
Early and Locally Advanced Breast
 Early and Locally Advanced Breast Cancer Audrea H. Szabatura, Pharm.D., BCOP; and Amy Hatfield Seung, Pharm.D., BCOP Reviewed by Jared M. Freml, Pharm.D., BCOP; Clarence Chant, Pharm.D., BCPS, FCSHP; and
Early and Locally Advanced Breast Cancer Audrea H. Szabatura, Pharm.D., BCOP; and Amy Hatfield Seung, Pharm.D., BCOP Reviewed by Jared M. Freml, Pharm.D., BCOP; Clarence Chant, Pharm.D., BCPS, FCSHP; and
Triple negative Breast Cancer Patient
 Triple negative Breast Cancer Patient Alison L Jones November 2013 Mrs Trisha Negative Aged 52) Diagnosed November 2001 T2 N1 (2/11)M0 Left breast. No family history WLE/ANC then FEC/T + RT Relapsed 2013
Triple negative Breast Cancer Patient Alison L Jones November 2013 Mrs Trisha Negative Aged 52) Diagnosed November 2001 T2 N1 (2/11)M0 Left breast. No family history WLE/ANC then FEC/T + RT Relapsed 2013
Inflammatory Breast Cancer
 Inflammatory Breast Cancer This document briefly describes inflammatory breast cancer. For more comprehensive information on this and other types of breast cancer, please refer to our document called Breast
Inflammatory Breast Cancer This document briefly describes inflammatory breast cancer. For more comprehensive information on this and other types of breast cancer, please refer to our document called Breast
Guideline for the Non Surgical Treatment of Breast Cancer
 Guideline for the Non Surgical Treatment of Breast Cancer incorporating former guidelines for systemic treatment, radiotherapy and aromatase inhibitors. Version History Version Date Comments 2.0 20.02.08
Guideline for the Non Surgical Treatment of Breast Cancer incorporating former guidelines for systemic treatment, radiotherapy and aromatase inhibitors. Version History Version Date Comments 2.0 20.02.08
The evolution of rectal cancer therapy. Objectives
 The evolution of rectal cancer therapy Hagen Kennecke MD MHA FRCPC Western Canada Consensus Conference September 5, 2014 Objectives Identify standard therapy: stage II/III rectal cancer Update recent adjuvant
The evolution of rectal cancer therapy Hagen Kennecke MD MHA FRCPC Western Canada Consensus Conference September 5, 2014 Objectives Identify standard therapy: stage II/III rectal cancer Update recent adjuvant
PI3K signaling pathway a new target for breast cancer treatment
 PI3K signaling pathway a new target for breast cancer treatment Introduction At the 37 th annual San Antonio Breast Cancer Symposium, SABCS, a number of interesting research trends, novelties as well as
PI3K signaling pathway a new target for breast cancer treatment Introduction At the 37 th annual San Antonio Breast Cancer Symposium, SABCS, a number of interesting research trends, novelties as well as
CANCER PULMON: ESTADIOS INICIALES POSTMUNDIAL PULMON DENVER 2015. 8-10-2015.Manuel Cobo Dols S. Oncología Médica HU Málaga Regional y VV
 CANCER PULMON: ESTADIOS INICIALES POSTMUNDIAL PULMON DENVER 2015 8-10-2015.Manuel Cobo Dols S. Oncología Médica HU Málaga Regional y VV Meta-analisis LACE: adyuvancia vs no adyuvancia Pignon JP, et al.
CANCER PULMON: ESTADIOS INICIALES POSTMUNDIAL PULMON DENVER 2015 8-10-2015.Manuel Cobo Dols S. Oncología Médica HU Málaga Regional y VV Meta-analisis LACE: adyuvancia vs no adyuvancia Pignon JP, et al.
Adjuvant Therapy Non Small Cell Lung Cancer. Sunil Nagpal MD Director, Thoracic Oncology Jan 30, 2015
 Adjuvant Therapy Non Small Cell Lung Cancer Sunil Nagpal MD Director, Thoracic Oncology Jan 30, 2015 No Disclosures Number of studies Studies Per Month 12 10 8 6 4 2 0 1 2 3 4 5 6 7 8 9 10 11 12 1 2 3
Adjuvant Therapy Non Small Cell Lung Cancer Sunil Nagpal MD Director, Thoracic Oncology Jan 30, 2015 No Disclosures Number of studies Studies Per Month 12 10 8 6 4 2 0 1 2 3 4 5 6 7 8 9 10 11 12 1 2 3
Seconda linea di trattamento
 XVIII Congresso Nazionale CIPOMO Roma, Giugno 2013 Nuovo paradigma terapeutico nel trattamento del carcinoma mammario HER2+ metastatico: dagli studi alla pratica clinica Seconda linea di trattamento Giorgio
XVIII Congresso Nazionale CIPOMO Roma, Giugno 2013 Nuovo paradigma terapeutico nel trattamento del carcinoma mammario HER2+ metastatico: dagli studi alla pratica clinica Seconda linea di trattamento Giorgio
New developments and controversies in breast cancer treatment: PARP Inhibitors: a breakthrough?
 New developments and controversies in breast cancer treatment: PARP Inhibitors: a breakthrough? F. Cardoso, MD Champalimaud Cancer Center Lisbon, Portugal BBM 2010 Thank you to A Tutt & PRIME Oncology
New developments and controversies in breast cancer treatment: PARP Inhibitors: a breakthrough? F. Cardoso, MD Champalimaud Cancer Center Lisbon, Portugal BBM 2010 Thank you to A Tutt & PRIME Oncology
The NCPE has issued a recommendation regarding the use of pertuzumab for this indication. The NCPE does not recommend reimbursement of pertuzumab.
 Cost Effectiveness of Pertuzumab (Perjeta ) in Combination with Trastuzumab and Docetaxel in Adults with HER2-Positive Metastatic or Locally Recurrent Unresectable Breast Cancer Who Have Not Received Previous
Cost Effectiveness of Pertuzumab (Perjeta ) in Combination with Trastuzumab and Docetaxel in Adults with HER2-Positive Metastatic or Locally Recurrent Unresectable Breast Cancer Who Have Not Received Previous
Corporate Medical Policy
 Corporate Medical Policy Ado-Trastuzumab Emtansine (Trastuzumab-DM1) for Treatment of File Name: Origination: Last CAP Review: Next CAP Review: Last Review: ado_trastuzumab_emtansine_(trastuzumab-dm1)_for_treatment_of_her-2_positivemalignancies
Corporate Medical Policy Ado-Trastuzumab Emtansine (Trastuzumab-DM1) for Treatment of File Name: Origination: Last CAP Review: Next CAP Review: Last Review: ado_trastuzumab_emtansine_(trastuzumab-dm1)_for_treatment_of_her-2_positivemalignancies
REPORT ASCO 2002 ORLANDO : LUNG CANCER Johan F. Vansteenkiste, MD, PhD, Univ. Hospital and Leuven Lung Cancer Group
 REPORT ASCO 2002 ORLANDO : LUNG CANCER Johan F. Vansteenkiste, MD, PhD, Univ. Hospital and Leuven Lung Cancer Group In the 2002 edition of the ASCO meeting, a total of 315 abstracts in the field of respiratory
REPORT ASCO 2002 ORLANDO : LUNG CANCER Johan F. Vansteenkiste, MD, PhD, Univ. Hospital and Leuven Lung Cancer Group In the 2002 edition of the ASCO meeting, a total of 315 abstracts in the field of respiratory
Gentest für DCIS? SENOLOGIE update 2015 21. Mai 2015 Kongress-Eventzentrum LAKE SIDE Prof. Bernhard Pestalozzi, Klinik für Onkologie, USZ
 Gentest für DCIS? SENOLOGIE update 2015 21. Mai 2015 Kongress-Eventzentrum LAKE SIDE Prof. Bernhard Pestalozzi, Klinik für Onkologie, USZ Gliederung Einführung: Multigen Profile OncotypeDX (21-gene score)
Gentest für DCIS? SENOLOGIE update 2015 21. Mai 2015 Kongress-Eventzentrum LAKE SIDE Prof. Bernhard Pestalozzi, Klinik für Onkologie, USZ Gliederung Einführung: Multigen Profile OncotypeDX (21-gene score)
Prognostic and Predictive Factors in Oncology. Mustafa Benekli, M.D.
 Prognostic and Predictive Factors in Oncology Mustafa Benekli, M.D. NCI Definitions ESMO Course -Essentials of Medical Oncology -Istanbul 2 Prognostic factor: NCI Definition A situation or condition, or
Prognostic and Predictive Factors in Oncology Mustafa Benekli, M.D. NCI Definitions ESMO Course -Essentials of Medical Oncology -Istanbul 2 Prognostic factor: NCI Definition A situation or condition, or
AIDS IN IDENTIFYING CANDIDATES FOR HER2-TARGETED THERAPY
 AIDS IN IDENTIFYING CANDIDATES FOR HER2-TARGETED THERAPY THE HERMARK BREAST CANCER ASSAY HERmark is based on our proprietary VeraTag technology that precisely quantifies HER2 proteins and protein complexes
AIDS IN IDENTIFYING CANDIDATES FOR HER2-TARGETED THERAPY THE HERMARK BREAST CANCER ASSAY HERmark is based on our proprietary VeraTag technology that precisely quantifies HER2 proteins and protein complexes
La biologia molecolare «driver» delle scelte terapeutiche: k mammario HER2+
 La biologia molecolare «driver» delle scelte terapeutiche: k mammario HER2+ Dr.ssa Lucia Del Mastro U.O. Sviluppo Terapie Innovative IRCCS AOU San Martino-IST Verona, 18 settembre 2015 Copyright 2014 -
La biologia molecolare «driver» delle scelte terapeutiche: k mammario HER2+ Dr.ssa Lucia Del Mastro U.O. Sviluppo Terapie Innovative IRCCS AOU San Martino-IST Verona, 18 settembre 2015 Copyright 2014 -
Breast Cancer: Background
 Disclaimer This slide deck in its original and unaltered format is for educational purposes and is current as of August 2014. All materials contained herein reflect the views of the faculty, and not those
Disclaimer This slide deck in its original and unaltered format is for educational purposes and is current as of August 2014. All materials contained herein reflect the views of the faculty, and not those
Abstract Introduction. Aim of the study. Conclusion. Patients and methods. Keywords. Results. R. Abo El Hassan 1, M. Moneer 2
 Original Study Outcome of HER2 positive luminal operable breast cancer in comparison with outcome of other operable luminal breast cancer patients: Long follow-up of single center randomized study R. Abo
Original Study Outcome of HER2 positive luminal operable breast cancer in comparison with outcome of other operable luminal breast cancer patients: Long follow-up of single center randomized study R. Abo
Pharmacogenetic Activities in SWOG Breast Cancer
 Pharmacogenetic Activities in SWOG Breast Cancer Pharmacogenomics: Future Plans S8897 Adjuvant CMF vs. CAF/ no Treatment Ambrosone RO1: Other genes (TBCI approved, analyses ongoing) S0221 Adjuvant Dose
Pharmacogenetic Activities in SWOG Breast Cancer Pharmacogenomics: Future Plans S8897 Adjuvant CMF vs. CAF/ no Treatment Ambrosone RO1: Other genes (TBCI approved, analyses ongoing) S0221 Adjuvant Dose
Appendix One. HER2-positive early breast cancer, its treatment and prognosis
 Appendix One. HER2-positive early breast cancer, its treatment and prognosis Breast cancer and HER2/neu over-expression Health need is one of PHARMAC s nine decision criteria (http://www.pharmac.govt.nz/pdf/231205.pdf
Appendix One. HER2-positive early breast cancer, its treatment and prognosis Breast cancer and HER2/neu over-expression Health need is one of PHARMAC s nine decision criteria (http://www.pharmac.govt.nz/pdf/231205.pdf
GUIDELINES FOR THE MANAGEMENT OF LUNG CANCER
 GUIDELINES FOR THE MANAGEMENT OF LUNG CANCER BY Ali Shamseddine, MD (Coordinator); as04@aub.edu.lb Fady Geara, MD Bassem Shabb, MD Ghassan Jamaleddine, MD CLINICAL PRACTICE GUIDELINES FOR THE TREATMENT
GUIDELINES FOR THE MANAGEMENT OF LUNG CANCER BY Ali Shamseddine, MD (Coordinator); as04@aub.edu.lb Fady Geara, MD Bassem Shabb, MD Ghassan Jamaleddine, MD CLINICAL PRACTICE GUIDELINES FOR THE TREATMENT
Inflammatory Breast Cancer: A Unique Pathologic Entity?
 Inflammatory Breast Cancer: A Unique Pathologic Entity? Sandra M. Swain, M.D. Director, Washington Cancer Institute Washington Hospital Center Washington DC Outline Overview Therapy High dose chemotherapy
Inflammatory Breast Cancer: A Unique Pathologic Entity? Sandra M. Swain, M.D. Director, Washington Cancer Institute Washington Hospital Center Washington DC Outline Overview Therapy High dose chemotherapy
Recommendations for the management of early breast cancer
 Recommendations for the management of early breast cancer in women with an identified BRCA1 or BRCA2 gene mutation or at high risk of a gene mutation FEBRUARY 2014 Incorporates published evidence to August
Recommendations for the management of early breast cancer in women with an identified BRCA1 or BRCA2 gene mutation or at high risk of a gene mutation FEBRUARY 2014 Incorporates published evidence to August
PRIMARY GLIOMA (oligodendroglioma, astrocytoma, oligodendroglioma, oligoastrocytoma, including anaplastic, gliosarcoma and glioblastoma multiforme)
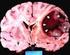 Protocol for Planning and Treatment The process to be followed when a course of chemotherapy is required to treat: PRIMARY GLIOMA (oligodendroglioma, astrocytoma, oligodendroglioma, oligoastrocytoma, including
Protocol for Planning and Treatment The process to be followed when a course of chemotherapy is required to treat: PRIMARY GLIOMA (oligodendroglioma, astrocytoma, oligodendroglioma, oligoastrocytoma, including
5.04.20. Perjeta. Perjeta (pertuzumab) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.04.20 Subject: Perjeta Page: 1 of 5 Last Review Date: June 19, 2015 Perjeta Description Perjeta (pertuzumab)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.04.20 Subject: Perjeta Page: 1 of 5 Last Review Date: June 19, 2015 Perjeta Description Perjeta (pertuzumab)
Sequential adjuvant docetaxel and anthracycline chemotherapy for node positive breast cancers: a retrospective study
 JBUON 2013; 18(2): 314-320 ISSN: 1107-0625 www.jbuon.com E-mail: info@jbuon.com ORIGINAL ARTICLE Sequential adjuvant docetaxel and anthracycline chemotherapy for node positive breast cancers: a retrospective
JBUON 2013; 18(2): 314-320 ISSN: 1107-0625 www.jbuon.com E-mail: info@jbuon.com ORIGINAL ARTICLE Sequential adjuvant docetaxel and anthracycline chemotherapy for node positive breast cancers: a retrospective
Definition and Impact of Pathologic Complete Response on Prognosis After Neoadjuvant Chemotherapy in Various Intrinsic Breast Cancer Subtypes
 Published Ahead of Print on April 16, 2012 as 10.1200/JCO.2011.38.8595 The latest version is at http://jco.ascopubs.org/cgi/doi/10.1200/jco.2011.38.8595 JOURNAL OF CLINICAL ONCOLOGY O R I G I N A L R E
Published Ahead of Print on April 16, 2012 as 10.1200/JCO.2011.38.8595 The latest version is at http://jco.ascopubs.org/cgi/doi/10.1200/jco.2011.38.8595 JOURNAL OF CLINICAL ONCOLOGY O R I G I N A L R E
Avastin in Metastatic Breast Cancer
 Non-interventional study Avastin in Metastatic Breast Cancer ML 21165 / 2007 Clinical Study Report Synopsis ROCHE ML21165 / WiSP Project RH09 / V. 1.0 / 24.06.2013 ROCHE ML21165-2 - Name of Sponsor Roche
Non-interventional study Avastin in Metastatic Breast Cancer ML 21165 / 2007 Clinical Study Report Synopsis ROCHE ML21165 / WiSP Project RH09 / V. 1.0 / 24.06.2013 ROCHE ML21165-2 - Name of Sponsor Roche
NCCN Non-Small Cell Lung Cancer V.1.2011 Update Meeting 07/09/10
 Guideline Page and Request NSCL-3 Stage IA, margins positive delete the recommendation for chemoradiation. Stage IB, IIA, margins positive delete the recommendation for chemoradiation + Stage IIA, Stage
Guideline Page and Request NSCL-3 Stage IA, margins positive delete the recommendation for chemoradiation. Stage IB, IIA, margins positive delete the recommendation for chemoradiation + Stage IIA, Stage
Komorbide brystkræftpatienter kan de tåle behandling? Et registerstudie baseret på Danish Breast Cancer Cooperative Group
 Komorbide brystkræftpatienter kan de tåle behandling? Et registerstudie baseret på Danish Breast Cancer Cooperative Group Lotte Holm Land MD, ph.d. Onkologisk Afd. R. OUH Kræft og komorbiditet - alle skal
Komorbide brystkræftpatienter kan de tåle behandling? Et registerstudie baseret på Danish Breast Cancer Cooperative Group Lotte Holm Land MD, ph.d. Onkologisk Afd. R. OUH Kræft og komorbiditet - alle skal
DECISION AND SUMMARY OF RATIONALE
 DECISION AND SUMMARY OF RATIONALE Indication under consideration Clinical evidence Everolimus in combination with exemestane hormone therapy for oestrogen receptor positive locally advanced or metastatic
DECISION AND SUMMARY OF RATIONALE Indication under consideration Clinical evidence Everolimus in combination with exemestane hormone therapy for oestrogen receptor positive locally advanced or metastatic
Hereditary Multifocal Breast Cancer. Farin Amersi M.D., F.A.C.S Division of Surgical Oncology Department of Surgery Cedar Sinai Medical Center
 Hereditary Multifocal Breast Cancer Farin Amersi M.D., F.A.C.S Division of Surgical Oncology Department of Surgery Cedar Sinai Medical Center CASE STUDY 30 year old Ashkenazi Jewish woman Nulliparous Felt
Hereditary Multifocal Breast Cancer Farin Amersi M.D., F.A.C.S Division of Surgical Oncology Department of Surgery Cedar Sinai Medical Center CASE STUDY 30 year old Ashkenazi Jewish woman Nulliparous Felt
Lung Cancer Treatment Guidelines
 Updated June 2014 Derived and updated by consensus of members of the Providence Thoracic Oncology Program with the aid of evidence-based National Comprehensive Cancer Network (NCCN) national guidelines,
Updated June 2014 Derived and updated by consensus of members of the Providence Thoracic Oncology Program with the aid of evidence-based National Comprehensive Cancer Network (NCCN) national guidelines,
Dose-Dense Chemotherapy in High-Risk Breast Cancer: Treatment Outcome and Toxicity
 original article < Dose-Dense Chemotherapy in High-Risk Breast Cancer: Treatment Outcome and Toxicity Salah El-Mesidy, MD, Mohsen Mokhtar, MD, Amr El-Kashif, MD, Loay Kasem, M.Sc Department of Clinical
original article < Dose-Dense Chemotherapy in High-Risk Breast Cancer: Treatment Outcome and Toxicity Salah El-Mesidy, MD, Mohsen Mokhtar, MD, Amr El-Kashif, MD, Loay Kasem, M.Sc Department of Clinical
New Agents as Options in the Treatment of Breast Cancer in 2012 2013. Realities and Possibilities
 New Agents as Options in the Treatment of Breast Cancer in 2012 2013. Realities and Possibilities Peter M. Ravdin, MD, PhD UT Health Science Center San Antonio San Antonio, TX 2012 2013 New Options for
New Agents as Options in the Treatment of Breast Cancer in 2012 2013. Realities and Possibilities Peter M. Ravdin, MD, PhD UT Health Science Center San Antonio San Antonio, TX 2012 2013 New Options for
Management of low grade glioma s: update on recent trials
 Management of low grade glioma s: update on recent trials M.J. van den Bent The Brain Tumor Center at Erasmus MC Cancer Center Rotterdam, the Netherlands Low grades Female, born 1976 1 st seizure 2005,
Management of low grade glioma s: update on recent trials M.J. van den Bent The Brain Tumor Center at Erasmus MC Cancer Center Rotterdam, the Netherlands Low grades Female, born 1976 1 st seizure 2005,
