A rapid and effective nonsurgical artificial insemination protocol using the NSET TM device for sperm transfer in mice without anesthesia
|
|
|
- Cameron Brown
- 8 years ago
- Views:
Transcription
1 Transgenic Res (2015) 24: DOI /s TECHNICAL REPORT A rapid and effective nonsurgical artificial insemination protocol using the NSET TM device for sperm transfer in mice without anesthesia Barbara J. Stone. Kendra H. Steele. Angelika Fath-Goodin Received: 27 January 2015 / Accepted: 3 June 2015 / Published online: 12 June 2015 The Author(s) This article is published with open access at Springerlink.com Abstract Artificial insemination (AI) is an assisted reproductive technique that is implemented successfully in humans as a fertility treatment, performed extensively for commercial breeding of livestock, and is also successful in laboratory rodents. AI in the mouse may be especially useful for breeding of transgenic or mutant mice with fertility problems, expansion of mouse colonies, and as an alternative to in vitro fertilization. Nonsurgical AI techniques for the mouse have been described previously but are not often implemented due to technical difficulties. Here we compare various protocols for preparation of CD1 recipients prior to AI for naïve (in estrus), ovulationinduced, and superovulated females. Timing of hormone administration relative to sperm delivery is also compared. An improved protocol for nonsurgical AI in mice is described, which incorporates a convenient hormone administration schedule for female recipients and rapid, non-stressful sperm transfer without the need for anesthesia or analgesia. Keywords Artificial insemination Nonsurgical NSET device In vitro fertilization Sperm Anesthesia B. J. Stone (&) K. H. Steele A. Fath-Goodin ParaTechs Corporation, 1122 Oak Hill Drive, Lexington, KY 40505, USA barbarastone@paratechs.com Introduction Artificial insemination (AI) is used to assist in reproduction by directly delivering motile sperm to the female reproductive tract. Artificial insemination in mice can be useful as an alternative to breeding for strains which are not easily bred due to physical or behavioral issues. The techniques of AI and in vitro fertilization (IVF) can both be used to generate viable embryos. Development after AI simply continues in the recipient female, while embryos generated from IVF must be transferred to a pseudopregnant recipient. AI or IVF can be used to generate embryos after cryopreservation of sperm, making them useful for cryorecovery of mouse strains. IVF requires collection of a large number of oocytes, fertilization is performed in vitro, and embryos are then transferred into a suitable recipient host for development. The process can be time consuming, technically challenging, and the resources expensive. AI is a convenient alternative to IVF for some applications. In theory, the AI technique is quite simple; sperm are directly added to the reproductive tract of the recipient female and all further development occurs in vivo. Historically, however, procedures for AI in mice have had variable rates of success, tend to be complicated, and as a result are underutilized. AI can be performed either surgically or nonsurgically. The surgical procedure has the benefit of using a small volume of sperm injected directly into the oviduct (Nakagata 1992; De Repentigny and Kothary
2 776 Transgenic Res (2015) 24: ) or bursa (Sato and Kimura 2001; Sato et al. 2002) to produce viable embryos. Surgical AI using previously cryopreserved sperm has also been used to generate live pups (Nakagata 1992). However, this procedure requires surgical training and use of anesthesia and analgesia. The surgical procedure requires opening of the abdominal cavity to reach the female reproductive tract, an invasive procedure requiring pain relief and post-operative supportive care. The use of various nonsurgical methods has also been previously reported. AI has been performed with varying degrees of success in laboratory mice (Mus musculus) to produce implanted fetuses (Dziuk and Runner 1960; Leckie et al. 1973). Dziuk and Runner (1960) describe a transcervical technique using a blunt needle and a glass speculum to transfer sperm to mice, which then receive a daily progesterone administration. Leckie et al. (1973) describe a procedure to transfer sperm transcervically with a blunt needle, with the use of an artificial penis and a vaginal tampon (as an alternative to pairing with a vasectomized male to induce a pseudopregnant state) to increase the efficiency of embryo implantation. More recently, nonsurgical transcervical AI has been performed under general anesthesia for production of embryos in superovulated deer mice (Peromyscus maniculatus) (Veres et al. 2012). While techniques for assisted reproduction in Peromyscus have generally lagged behind techniques available for Mus, attempts are being made to refine methods for embryo recovery and in vitro culture (Veres et al. 2012). To this end, a nonsurgical AI technique introducing sperm into the vagina without anesthesia has also been described for Peromyscus for use with females in estrus or with superovulated females (Behringer et al. 2014; Duselis and Vrana 2007). However, data from the nonsurgical AI procedures regarding the production of live pups is not available. The laboratory mouse community is in need of a simple user-friendly method for AI, one that also creates an ethical standard for treatment to mice. To develop this protocol, we first examined some variable aspects of the previously described surgical and nonsurgical AI techniques available for Mus. (1) Hormone administration can be used to induce timed estrus and ovulation to generate oocytes for fertilization. However, superovulation has been shown to have adverse effects on embryonic and fetal development (Beaumont and Smith 1975; Ertzeid and Storeng 1992; Van der Auwera and D Hooghe 2001; Van der Auwera et al. 1999). (2) Timing of hormone administration relative to sperm delivery needs to be considered. Given a natural estrus situation, ovulation and fertilization is thought to occur around midnight, during the dark cycle (Behringer et al. 2014). Oocytes generated after hormone induced ovulation are most efficiently fertilized from surgical AI 7 12 h after administration of hcg, corresponding to timing of ovulation (Sato et al. 2004). This timing of ovulation and fertilization is rather inconvenient for routine use of AI. (3) Induction of pseudopregnancy in the recipient before (Nakagata 1992; De Repentigny and Kothary 1996; Sato and Kimura 2001; Sato et al. 2002), during (Leckie et al. 1973), or after (this study) insemination is required for fetal development. These three variables have made use of techniques for AI in mice problematic. While surgical and nonsurgical techniques for AI have been developed for use in mice, adoption of these techniques for routine use has not occurred. Therefore, our goal was to develop a simple and convenient protocol for AI in mice that would result in the birth of healthy pups. To that end, we compared various protocols for preparation of female recipients prior to AI; naïve (in estrus), ovulation induced, and superovulated females. Sperm transfer was performed with the NSET device, typically used for nonsurgical embryo transfer in mice (Green et al. 2009; Bin Ali et al. 2014), because it has been shown to be less stressful to mice than a comparable surgical procedure (Steele et al. 2013). Timing of hormone administration relative to sperm delivery was also compared. As a result, an improved protocol for nonsurgical AI in mice is described. This protocol incorporates a convenient hormone administration schedule for female recipients and a rapid, non-stressful sperm transfer, without the need for anesthesia or analgesia. The protocol can be used to produce large litters of healthy pups. Methods Animals All animal research was approved by the ParaTechs Institutional Animal Care and Use Committee and conducted under the standards dictated by the Office of Laboratory Animal Welfare, National Institutes of
3 Transgenic Res (2015) 24: Health, Public Health Service, United States Department of Health and Human Services. All mice were obtained from Charles River Laboratories (Wilmington, MA, USA). Vasectomized male CD1 mice ([2 months old) were used to induce pseudopregnancy. Female CD1 mice between 2 and 5 months old were used as sperm recipients. Male C3H mice ([2 months old) with proven fertility were used as sperm donors. The vivarium was maintained at C with an average relative humidity of % under a 12:12 h light:dark (light from 8 am to 8 pm) cycle. Mice were housed in standardized ventilated microisolation caging (Innovive, San Diego, CA, USA). Mice had access to Teklad Global 19 % protein extruded rodent diet #2919 (Harlan Laboratories, Madison, WI, USA) and bottled sterile water (Innovive) ad libitum. Sperm isolation and preparation Human tubal fluid medium (HTF) (Irvine Scientific, Santa Ana, CA, USA, Cat #90126) was prepared by adding 4 mg/ml bovine serum albumin (BSA) (Sigma- Aldrich, St. Louis, MO, USA, Cat #A3311), and filter sterilizing. The HTF media was equilibrated prior to use for at least 30 min at 37 C in the presence of 5 % CO 2. Fresh sperm was collected from the cauda epididymis and vas deferens of C3H males. The sperm was capacitated in 500 ll freshly prepared HTF media under embryo culture grade mineral oil (Sigma- Aldrich, Cat #M8410) at 37 C in the presence of 5%CO 2 for 45 min. Hormone administration Pregnant mare serum gonadotropin (PMSG) and human chorionic gonadotropin (hcg, Cat# AFP8456A) were obtained from the National Hormone and Peptide Program, National Institute of Diabetes and Digestive and Kidney Diseases, (Torrance, CA, USA) and diluted in sterile phosphate buffered saline (PBS) (Butler-Schein, Dublin, OH, USA, Cat #002477) to 1000 IU/ml, and stored at -80 C until use. Female CD1 mice for this study were either naïve (chosen by outward appearance to be in estrus) or were injected with hormones for induction of estrus or superovulation. For naïve females, estrus cycle indicators were observed as previously described (Behringer et al. 2014). Superovulated females were prepared by intraperitoneal (i.p.) injection with 5 IU PMSG and hcg administered *47 h apart at various time schedules relative to the vivarium light dark cycle. To induce a more natural ovulation condition in CD1 females, the hormones were reduced to 1 IU per i.p. injection. Artificial insemination After optimization of the procedure, a total of 40 llof fresh sperm was transferred to the uterine horns of female CD1 mice. The transfer was performed without the use of anesthesia or analgesia and took\1 min per mouse. Sperm transfer was carried out using a commercially available device originally developed for nonsurgical embryo transfer in mice, the NSET TM device (ParaTechs, Lexington, KY, USA). The NSET device, a tapered Teflon catheter connected to a specially designed hub, was attached to a Rainin P20 pipette (Mettler-Toledo, Columbus, OH, USA). The device was loaded with 20 ll sperm from the edge of the sperm sample, avoiding clumps. The un-anesthetized recipient female was placed on a wire bar cage top and allowed to grasp the surface with its forefeet. The mouse was held at the base of the tail using a thumb and forefinger, and the rear angled slightly upward, while lightly pressing the base of the hind legs with two fingers from the same hand. The holding technique has been described previously for nonsurgical embryo transfer (Green et al. 2009) and a video of the procedure is available ( com/nset/). Either the small or large speculum included with the device was used to hold open the vagina. If the larger diameter speculum was used, the cervix could be visually located with the aid of a direct light source. The NSET tip was inserted through the cervix and into the uterus. Once the NSET hub contacted the speculum, the sperm was expelled by pressing the pipette plunger to the first stop. The procedure was then repeated to deliver a total sperm volume of 40 ll. The speculum was removed, and the mouse was immediately paired with a vasectomized male overnight. No post-procedure monitoring was required. Results Initial attempts to perform AI using the NSET device for sperm transfer followed a previously described
4 778 Transgenic Res (2015) 24: protocol for nonsurgical insemination (Nagy et al. 2003) with the exception that sperm transfer occurred at the time of hcg injection (instead of 13.5 h after hcg injection). The females were injected i.p. with 0.5 or 1.0 IU PMSG at 1 pm 2 days prior to sperm transfer and 0.5 or 1.0 IU hcg at noon the day of sperm transfer. The NSET device was used to transfer ll sperm just prior to hcg injection. The recipient was immediately housed overnight with a vasectomized male to mate, simulating normal breeding behavior. However, no implantation sites were observed (N = 17) and no pups were delivered (N = 13) following these initial pilot studies. To determine if fertilization of oocytes could occur with this method, the protocol was modified and the female recipients were injected with 5 IU of each hormone to induce ovulation as before. After sperm transfer and pairing overnight with a vasectomized male, female recipients were euthanized and oviducts were flushed with M2 medium. All AI procedures resulted in viable embryos, with an average of 9.7 blastocysts developed after in vitro culture from an average of 30.3 oocytes per ovulation (N = 20). However, using this same protocol for insemination, only three AI procedures resulted in implanted embryos (N = 21). These results indicated that the sperm transfer was successful in that oocytes were fertilized and viable embryos were being produced. However, implantation of embryos in the uterine horns was inefficient. Since the oocytes were being fertilized but implantation was restricted, it was thought that the amount of hormones administered for ovulation and/or the timing of administration needed to be altered. In confirmation, the first successful demonstration that the NSET device could be used for AI to produce live pups was performed using an alternate injection schedule (A. Harman, personal communication, Children s Hospital of Philadelphia, PA, USA). 5 IU PMSG was injected at 8:30 pm 3 days prior to sperm transfer and 5 IU hcg at 8:00 pm 1 day prior to sperm transfer. Sperm volume was increased to 40 ll; transferring this volume required two transcervical sperm deliveries per female. The AI procedure produced a litter of pups in a strain of obese mice (that was unable to copulate). In our hands, this protocol resulted in a pregnancy rate of 36 % (N = 19) (Table 1; Trial 9) in female CD1 mice, with litter sizes ranging from 2 to 9 pups, and an average of 4.5 pups per litter. However, pup mortality was high (57 %) with only 13 of 32 pups surviving to weaning. To increase pregnancy rates and to find a more convenient work schedule, other hormone injection and sperm transfer times were tested using PMSG and hcg at concentrations of 5 IU. No increase in pregnancy rate was achieved after administration of 5 IU of hormones prior to AI, although an increase in litter size (10 pups on average) and an improvement in survival rate (to ) were observed when the injection schedule was begun earlier in the day (Table 1; Trial 8). With the aim of potentially increasing embryo implantation and improving the health of the pups resulting from the AI protocol, a decrease was made in the amount of hormones delivered to the female prior to sperm transfer. First, females in estrus were selected as AI recipients (N = 20). No hormones were administered to induce ovulation. AI was performed at 5:00 pm or at 8:00 pm (Table 1; Trials 1 and 2) before females were paired with vasectomized males overnight. No pups were obtained. Control matings with stud males confirmed that the females were receptive. The pregnancy rate for control matings was 50 % with an average litter size of 12 pups (N = 6). Next, female AI recipients were given low levels of hormones to induce a more natural ovulation condition; PMSG and hcg were reduced to 1 IU per i.p. injection, given 47 h apart. With the administration of a lower hormone dose, a trend was observed in improvement in pregnancy rate and pup health. The protocol with the highest pregnancy rate (Trial 14) followed a convenient time schedule for hormone injections and AI. 1 IU of PMSG was administered at 5:30 pm 3 days prior to sperm transfer and 1 IU of hcg was administered at 5:00 pm 1 day prior to sperm transfer to ovulate AI recipients. The next morning, sperm was collected at 8:00 am, capacitated for 1 h, and 40 ll delivered at 9:00 am transcervically to AI recipients. Recipients were immediately paired with vasectomized males overnight. Full term pregnancy rates with fresh sperm reached 50 % (N = 20) (Table 1; Trial 14). Litter size averaged 5.0 pups with all pups surviving to weaning. Discussion A protocol refinement for nonsurgical AI in mice is described, which incorporates a convenient hormone administration schedule for female recipients and
5 Transgenic Res (2015) 24: Table 1 AI procedures Trial Hormone delivery Day 1 Day 3 Day 4 Pregnancy rate Average litter size (range) Survival rate 1 None AI 5:00 pm 2 None AI 8:00 pm 3 5 IU PMSG 8:30 am hcg 8:00 am AI 12:00 pm 4 5 IU PMSG 8:30 am hcg 8:00 am AI 5:00 pm 5 5 IU PMSG 8:30 am hcg 8:00 am AI 10:00 pm 6 5 IU PMSG 12:00 pm hcg 11:00 am AI 11:00 am 10 % 2.0 (2) 7 5 IU PMSG 12:00 pm hcg 11:00 am AI 11:00 am 10 % 1.0 (1) 8 5 IU PMSG 5:30 pm hcg 5:00 pm AI 9:00 am 30 % 10.0 (7 13) 9 5 IU PMSG 8:30 pm hcg 8:00 pm AI 9:30 am 36 % 4.5 (2 9) 10 5 IU PMSG 6:30 pm hcg 6:00 pm AI 5:30 pm 11 1 IU PMSG 8:30 am hcg 8:00 am AI 5:00 pm 12 1 IU PMSG 8:30 am hcg 8:00 am AI 10:00 pm 0% 20 % 2.0 (2) 40 % 5.0 (2 11) 13 1 IU PMSG 8:30 pm hcg 8:00 pm AI 9:30 am 10 % 1.0 (1) 14 1 IU PMSG 5:30 pm hcg 5:00 pm AI 9:00 am 50 % 5.0 (1 12) Hormone concentrations, hormone administration, and sperm delivery schedules are shown for individual AI trials. Pregnancy rates, average litter size, the range of litter size, and the survival rate of pups to weaning age (21 days) are indicated. Sperm was delivered using the NSET device for mice without anesthesia. Analysis of data by Fisher s exact test shows that a h time interval between the hcg hormone injection and AI has a significant positive effect on pregnancy rates (p \ 0.05) relative to other conditions tested. By the same test, administration of a hormone concentration of 1 IU has a significant positive effect on pup survival rate (p \ 0.05) over 5 IU. No significant effect of hormone concentration on litter size or pregnancy rate is seen. Significance cannot be determined between individual trials due to insufficient sample size 43 % rapid, non-stressful sperm transfer without the need for anesthesia or analgesia. Sperm transfer was performed using the NSET device for mice, originally designed for nonsurgical transfer of embryos into the uterine horns of pseudopregnant female mice. The device consists of a flexible Teflon catheter that is able to pass the cervix and deposit embryos or sperm into the uterine horn. Utilization of the device for embryo transfer is considered a refinement (Steele et al. 2013) according to Russell and Burch s 3Rs of animal research (Russell and Burch 1959) defining alternative procedures which either replace animals, reduce the number of animals used, or refine methodologies to minimize pain or distress. In the case of AI, use of the NSET device provides a convenient 3Rs refinement to a surgical procedure (Nakagata 1992; De Repentigny and Kothary 1996; Sato and Kimura 2001) and an alternative to a nonsurgical transcervical procedure using a blunt needle and glass speculum (Dziuk and Runner 1960), or a nonsurgical procedure requiring an artificial penis and vaginal tampon (Leckie et al. 1973; Takeshima and Toyoda 1977).
6 780 Transgenic Res (2015) 24: Nonsurgical AI is an assisted reproduction technique that has applications as a fertility treatment in humans and for commercial breeding of livestock. However, AI also has applications for use in laboratory mice. AI can be used for rapid expansion of mouse colonies as well as for generating age-matched cohorts for studies. Therefore, the use of AI may reduce the need for large in-house breeding colonies to provide mice for research projects. The ability to rapidly expand colonies on-site could reduce the cost of maintaining large breeding colonies. AI may be especially useful for breeding of transgenic or mutant mice with fertility problems and as an alternative to in vitro fertilization. Additionally, sperm can be shipped globally either cold (Takeo et al. 2012) or cryopreserved using one of two efficient protocols (Behringer et al. 2014), reducing the need to ship live animals. Nonsurgical AI could provide a means to rapidly recover a strain after shipment of sperm. Additionally, use of this nonsurgical AI protocol has potential to be coupled with thawed cryopreserved sperm. As reviewed in Takeo and Nakagata (2010), recent advances in cryopreservation of mouse strains have increased viability of thawed sperm. Therefore, transfer of thawed sperm with the NSET device has the potential to provide an alternative to IVF for strain cryorecovery. Acknowledgments We would like to thank Adele Harman at Children s Hospital of Philadelphia, PA, USA, for sharing her data regarding the first successful use of the NSET device for AI resulting in live pups. Research reported in this publication was funded by the Office of the Director, National Institutes of Health, United States Department of Health and Human Services, under Award Numbers 1R43RR A1, 2R44RR , and 8R44OD Conflict of interest Barbara Stone and Kendra Steele are employed by ParaTechs Corporation; Angelika Fath-Goodin is co-owner of ParaTechs Corporation. ParaTechs holds exclusive licensing rights to manufacture the NSET device for mice. Ethical standard All applicable international, national, and institutional guidelines for the care and use of animals were followed. All procedures performed in studies involving animals were in accordance with the ethical standards of the Institutional Animal Care and Use Committee at ParaTechs Corporation, where the studies were conducted. ParaTechs Corporation performed all studies involving animals under an Assurance of Compliance filed with the Office of Laboratory Animal Welfare, National Institutes of Health, Public Health Service, United States Department of Health and Human Services. Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License ( creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. References Beaumont HM, Smith AF (1975) Embryonic mortality during the pre-and post-implantation periods of pregnancy in mature mice after superovulation. J Reprod Fertil 45: Behringer R, Gertsenstein M, Nagy KV, Nagy A (2014) Manipulating the mouse embryo: a laboratory manual, 4th edn. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York Bin Ali R, van der Ahé F, Braumuller TM, Pritchard C, Krimpenfort P, Berns A, Huijbers IJ (2014) Improved pregnancy and birth rates with routine application of nonsurgical embryo transfer. Transgenic Res 23(4): De Repentigny Y, Kothary R (1996) An improved method for artificial insemination of mice: Oviduct transfer of spermatozoa. Trends Genet 12:44 45 Duselis AR, Vrana PB (2007) Harvesting sperm and artificial insemination of mice. J Vis Exp 3:184. doi: /184 Dziuk PJ, Runner MN (1960) Recovery of blastocysts and induction of implantation following artificial insemination of immature mice. J Reprod Fertil 1: Ertzeid G, Storeng R (1992) Adverse effects of gonadotropin treatment on pre- and post-implantation development in mice. J Reprod Fertil 96: Green MA, Bass S, Spear BT (2009) A device for the simple and rapid transcervical transfer of mouse embryos eliminates the need for surgery and potential post-operative complications. Biotechniques 47: Leckie PA, Watson JG, Chaykin S (1973) An improved method for the artificial insemination of the mouse (Mus musculus). Biol Reprod 9: Nagy A, Gertsenstein M, Vintersten K, Behringer R (2003) Manipulating the mouse embryo, 3rd edn. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY Nakagata N (1992) Production of normal young following insemination of frozen-thawed mouse spermatozoa into Fallopian tubes of pseudopregnant females. Jikken Dobutsu 41: Russell WMS, Burch RL (1959) The principles of humane experimental technique. Methuen & Co., Ltd, London Sato M, Kimura M (2001) Intrabursal transfer of spermatozoa (ITS): a new route for artificial insemination in mice. Theriogenology 55: Sato M, Nagashima A, Watanabe T, Kimura M (2002) Comparison of intrabursal transfer of spermatozoa, a new method for artificial insemination in mice, with intraoviductal transfer of spermatozoa. J Assist Reprod Genet 19(11):
7 Transgenic Res (2015) 24: Sato M, Tanigawa M, Watanabe T (2004) Effect of time of ovulation on fertilization after intrabursal transfer of spermatozoa (ITS): improvement of a new method for artificial insemination in mice. Theriogenology 62: Steele KH, Hester JM, Stone BJ, Carrico KM, Spear BT, Fath- Goodin A (2013) Nonsurgical embryo transfer device (NSET) is less stressful than surgery for embryo transfer in mice. J Am Assoc Lab Anim Sci 52(1):17 21 Takeo T, Nakagata N (2010) Mouse sperm cryopreservation and effective embryo production using cryopreserved C57BL/6 mouse sperm. J Mamm Ova Res 27:70 78 Takeo T, Tsutsumi A, Omaru T, Fukumoto K, Haruguchi Y, Kondo T, Nakamuta Y, Takeshita Y, Matsunaga H, Tsuchiyama S, Sakoh K, Nakao S, Yoshimoto H, Shimizu N, Nakagata N (2012) Establishment of a transport system for mouse epididymal sperm at refrigerated temperatures. Cryobiology 65(3): Takeshima T, Toyoda Y (1977) Artificial insemination in the mouse with special reference to the effect of sperm numbers on the conception rate and litter size. Jikken Dobutsu 26(4): Van der Auwera I, D Hooghe T (2001) Superovulation of female mice delays embryonic and fetal development. Human Reprod 16(6): Van der Auwera I, Pijnenborg R, Koninckx PR (1999) The influence of in vitro culture versus stimulated and untreated oviductal environment on mouse embryo development and implantation. Human Reprod 14(10): Veres M, Duselis A, Graft A, Pryor W, Crossland J, Vrana PB, Szalai G (2012) The biology and methodology of assisted reproduction in deer mice (Peromyscus maniculatus). Theriogenology 77(2):
Assisted Reproductive Technologies at IGO
 9339 Genesee Avenue, Suite 220 San Diego, CA 92121 858 455 7520 Assisted Reproductive Technologies at IGO Although IGO no longer operates an IVF laboratory or program as such, we work closely with area
9339 Genesee Avenue, Suite 220 San Diego, CA 92121 858 455 7520 Assisted Reproductive Technologies at IGO Although IGO no longer operates an IVF laboratory or program as such, we work closely with area
LONDON REGIONAL TRANSGENIC AND GENE TARGETING FACILITY
 LONDON REGIONAL TRANSGENIC AND GENE TARGETING FACILITY SPERM CRYOPRESERVATION AND IN VITRO FERTILIZATION RESOURCE PACKAGE For information, contact: Dr. Christopher Pin, PhD, Scientific Director London
LONDON REGIONAL TRANSGENIC AND GENE TARGETING FACILITY SPERM CRYOPRESERVATION AND IN VITRO FERTILIZATION RESOURCE PACKAGE For information, contact: Dr. Christopher Pin, PhD, Scientific Director London
A POWERFUL IN VITRO FERTILIZATION
 A POWERFUL During the past 50 years technological advances in the field of bovine reproduction have led to some dramatic changes in the way cattle look, reproduce, perform, and even taste. Artificial Insemination
A POWERFUL During the past 50 years technological advances in the field of bovine reproduction have led to some dramatic changes in the way cattle look, reproduce, perform, and even taste. Artificial Insemination
Artificial insemination
 Artificial insemination What is involved? Artificial insemination is an assisted reproduction technique that consists of inserting laboratory-treated spermatozoa into the woman s uterus or cervical canal.
Artificial insemination What is involved? Artificial insemination is an assisted reproduction technique that consists of inserting laboratory-treated spermatozoa into the woman s uterus or cervical canal.
Recommended Resources: The following resources may be useful in teaching this
 Unit B: Anatomy and Physiology of Poultry Lesson 4: Artificial Poultry Reproduction Student Learning Objectives: Instruction in this lesson should result in students achieving the following objectives:
Unit B: Anatomy and Physiology of Poultry Lesson 4: Artificial Poultry Reproduction Student Learning Objectives: Instruction in this lesson should result in students achieving the following objectives:
In - Vitro Fertilization Handbook
 In - Vitro Fertilization Handbook William F. Ziegler, D.O. Medical Director Scott Kratka, ELD, TS Embryology Laboratory Director Lauren F. Lucas, P.A.-C, M.S. Physician Assistant Frances Cerniak, R.N.
In - Vitro Fertilization Handbook William F. Ziegler, D.O. Medical Director Scott Kratka, ELD, TS Embryology Laboratory Director Lauren F. Lucas, P.A.-C, M.S. Physician Assistant Frances Cerniak, R.N.
Infertility Services Medical Policy For University of Vermont Medical Center and Central Vermont Medical Center employer groups
 Infertility Services Medical Policy For University of Vermont Medical Center and Central Vermont Medical Center employer groups File name: Infertility Services File code: UM.REPRO.01 Last Review: 02/2016
Infertility Services Medical Policy For University of Vermont Medical Center and Central Vermont Medical Center employer groups File name: Infertility Services File code: UM.REPRO.01 Last Review: 02/2016
Artificial Insemination. Advanced Reproductive Techniques in Small Ruminants. Success of AI Programs 9/20/2013
 Artificial Insemination Advanced Reproductive Techniques in Small Ruminants Jason W. Johnson, DVM, MS, Dip ACT Medical Director Large Animal Teaching and Research Center Assistant Professor of Theriogenology
Artificial Insemination Advanced Reproductive Techniques in Small Ruminants Jason W. Johnson, DVM, MS, Dip ACT Medical Director Large Animal Teaching and Research Center Assistant Professor of Theriogenology
Symposium on RECENT ADVANCES IN ASSISTED REPRODUCTIVE TECHNOLOGY
 Symposium on RECENT ADVANCES IN ASSISTED REPRODUCTIVE TECHNOLOGY Dr Niel Senewirathne Senior Consultant of Obstetrician & Gynaecologist De zoyza Maternity Hospita 1 ART - IVF & ICSI 2 Infertility No pregnancy
Symposium on RECENT ADVANCES IN ASSISTED REPRODUCTIVE TECHNOLOGY Dr Niel Senewirathne Senior Consultant of Obstetrician & Gynaecologist De zoyza Maternity Hospita 1 ART - IVF & ICSI 2 Infertility No pregnancy
Artificial insemination with donor sperm
 Artificial insemination with donor sperm Ref. 123 / 2009 Reproductive Medicine Unit Servicio de Medicina de la Reproducción Gran Vía Carlos III 71-75 08028 Barcelona Tel. (+34) 93 227 47 00 Fax. (+34)
Artificial insemination with donor sperm Ref. 123 / 2009 Reproductive Medicine Unit Servicio de Medicina de la Reproducción Gran Vía Carlos III 71-75 08028 Barcelona Tel. (+34) 93 227 47 00 Fax. (+34)
EVERY LIVING THING has a number of
 Anatomy and Physiology of Animal Reproductive Systems EVERY LIVING THING has a number of organ systems operating to perform specific functions. If you were to examine one of these systems, you would observe
Anatomy and Physiology of Animal Reproductive Systems EVERY LIVING THING has a number of organ systems operating to perform specific functions. If you were to examine one of these systems, you would observe
BOER GOAT EMBRYO TRANSFER
 BOER GOAT EMBRYO TRANSFER Good management No shortcuts PLAN AHEAD AVOID STRESS Some examples of how stress is induced are: Mixing groups or individual animals together that have not previously been together.
BOER GOAT EMBRYO TRANSFER Good management No shortcuts PLAN AHEAD AVOID STRESS Some examples of how stress is induced are: Mixing groups or individual animals together that have not previously been together.
(Received 29th July 1963)
 EGG TRANSFER IN SHEEP EFFECT OF DEGREE OF SYNCHRONIZATION BETWEEN DONOR AND RECIPIENT, AGE OF EGG, AND SITE OF TRANSFER ON THE SURVIVAL OF TRANSFERRED EGGS N. W. MOORE and J. N. SHELTON Jf.S. W. and The
EGG TRANSFER IN SHEEP EFFECT OF DEGREE OF SYNCHRONIZATION BETWEEN DONOR AND RECIPIENT, AGE OF EGG, AND SITE OF TRANSFER ON THE SURVIVAL OF TRANSFERRED EGGS N. W. MOORE and J. N. SHELTON Jf.S. W. and The
The IUI procedure Who should consider an IUI IUI success rates IUI cost What to consider if IUI is unsuccessful. The IUI procedure:
 A Complete Guide to understanding IUI (intrauterine insemination) and artificial insemination (Eric Daiter, MD Board Certified in Reproductive Endocrinology and Infertility) The IUI procedure Who should
A Complete Guide to understanding IUI (intrauterine insemination) and artificial insemination (Eric Daiter, MD Board Certified in Reproductive Endocrinology and Infertility) The IUI procedure Who should
Welcome to chapter 8. The following chapter is called "Monitoring IVF Cycle & Oocyte Retrieval". The author is Professor Jie Qiao.
 Welcome to chapter 8. The following chapter is called "Monitoring IVF Cycle & Oocyte Retrieval". The author is Professor Jie Qiao. The learning objectives of this chapter are 2 fold. The first section
Welcome to chapter 8. The following chapter is called "Monitoring IVF Cycle & Oocyte Retrieval". The author is Professor Jie Qiao. The learning objectives of this chapter are 2 fold. The first section
Illinois Insurance Facts Illinois Department of Financial and Professional Regulation Division of Insurance
 Illinois Insurance Facts Illinois Department of Financial and Professional Regulation Division of Insurance Insurance Coverage for Infertility Treatment Revised November 2004 Infertility is a condition
Illinois Insurance Facts Illinois Department of Financial and Professional Regulation Division of Insurance Insurance Coverage for Infertility Treatment Revised November 2004 Infertility is a condition
REPRODUCTION IN DONKEYS
 REPRODUCTION IN DONKEYS Stephen R. Purdy, DVM Department of Veterinary and Animal Science University of Massachusetts, Amherst, MA INTRODUCTION This is an overview of the practical aspects of male and
REPRODUCTION IN DONKEYS Stephen R. Purdy, DVM Department of Veterinary and Animal Science University of Massachusetts, Amherst, MA INTRODUCTION This is an overview of the practical aspects of male and
Center for Women s Reproductive Care at Columbia University
 Center for Women s Reproductive Care at Columbia University Oocyte Recipients Greetings, Thank you for your interest in the Center for Women s Reproductive Care at Columbia University. We hope that the
Center for Women s Reproductive Care at Columbia University Oocyte Recipients Greetings, Thank you for your interest in the Center for Women s Reproductive Care at Columbia University. We hope that the
FACTORS THAT AFFECT IN VITRO FERTILIZATION USING CRYOPRESERVED MOUSE SPERM
 FACTORS THAT AFFECT IN VITRO FERTILIZATION USING CRYOPRESERVED MOUSE SPERM Sue Bath Melbourne, Australia EMMA Cryopreservation Workshop Madrid, Spain May 2012 1 Outline Intrinsic Factors Strain of mouse
FACTORS THAT AFFECT IN VITRO FERTILIZATION USING CRYOPRESERVED MOUSE SPERM Sue Bath Melbourne, Australia EMMA Cryopreservation Workshop Madrid, Spain May 2012 1 Outline Intrinsic Factors Strain of mouse
Understanding Animal Reproduction Technology
 Lesson 251c Understanding Animal Reproduction Technology Core Area. Animal Science Unit 250. Genetics and Breeding Topic 251. Fertilization California Academic Standard. Science Grades 9 through 12 Biology/Life
Lesson 251c Understanding Animal Reproduction Technology Core Area. Animal Science Unit 250. Genetics and Breeding Topic 251. Fertilization California Academic Standard. Science Grades 9 through 12 Biology/Life
Artificial Insemination in Cattle
 Artificial Insemination in Cattle Introduction This slide show is designed to introduce students to artificial insemination in cattle. However, it is only a brief overview and further training is necessary
Artificial Insemination in Cattle Introduction This slide show is designed to introduce students to artificial insemination in cattle. However, it is only a brief overview and further training is necessary
Risks and complications of assisted conception
 Risks and complications of assisted conception August 005 Richard Kennedy British Fertility Society Factsheet www.fertility.org.uk No medical treatment is entirely free from risk and infertility treatment
Risks and complications of assisted conception August 005 Richard Kennedy British Fertility Society Factsheet www.fertility.org.uk No medical treatment is entirely free from risk and infertility treatment
An Introduction to Artificial Insemination
 A New Way to Your Dream Dog An Introduction to Artificial Insemination Contents of the brochure What does artificial insemination mean?... 4 Advantages of artificial insemination... 5 What does artificial
A New Way to Your Dream Dog An Introduction to Artificial Insemination Contents of the brochure What does artificial insemination mean?... 4 Advantages of artificial insemination... 5 What does artificial
Rationale for replacing IVIG with Intralipid (IL) for immunological pregnancy loss
 Rationale for replacing IVIG with Intralipid (IL) for immunological pregnancy loss Recurrent Pregnancy Loss The reason that an embryo may not implant successfully is either because there is something intrinsically
Rationale for replacing IVIG with Intralipid (IL) for immunological pregnancy loss Recurrent Pregnancy Loss The reason that an embryo may not implant successfully is either because there is something intrinsically
Reproduction Multiple Choice questions
 Reproduction Multiple Choice questions 1. In mammals that are seasonal breeders, females are receptive only once a year. This is called A) a follicular cycle B) an estrous cycle C) a menstrual cycle D)
Reproduction Multiple Choice questions 1. In mammals that are seasonal breeders, females are receptive only once a year. This is called A) a follicular cycle B) an estrous cycle C) a menstrual cycle D)
Authorized By: Holly C. Bakke, Commissioner, Department of Banking and Insurance.
 INSURANCE DIVISION OF INSURANCE Actuarial Services Benefit Standards for Infertility Coverage Proposed New Rules: N.J.A.C. 11:4-54 Authorized By: Holly C. Bakke, Commissioner, Department of Banking and
INSURANCE DIVISION OF INSURANCE Actuarial Services Benefit Standards for Infertility Coverage Proposed New Rules: N.J.A.C. 11:4-54 Authorized By: Holly C. Bakke, Commissioner, Department of Banking and
REPRODUCTIVE MEDICINE AND INFERTILITY ASSOCIATES Woodbury Medical Arts Building 2101 Woodwinds Drive Woodbury, MN 55125 (651) 222-6050
 REPRODUCTIVE MEDICINE AND INFERTILITY ASSOCIATES Woodbury Medical Arts Building 2101 Woodwinds Drive Woodbury, MN 55125 (651) 222-6050 RECIPIENT COUPLE INFORMED CONSENT AND AUTHORIZATION FOR IN VITRO FERTILIZATION
REPRODUCTIVE MEDICINE AND INFERTILITY ASSOCIATES Woodbury Medical Arts Building 2101 Woodwinds Drive Woodbury, MN 55125 (651) 222-6050 RECIPIENT COUPLE INFORMED CONSENT AND AUTHORIZATION FOR IN VITRO FERTILIZATION
Managing the Heat-Stressed Cow to Improve Reproduction
 Managing the Heat-Stressed Cow to Improve Reproduction Peter J. Hansen Department of Animal Sciences University of Florida, Gainesville Florida 32611-0910 Ph: 352-392-5590 Fax: 352-392-5595 The Growing
Managing the Heat-Stressed Cow to Improve Reproduction Peter J. Hansen Department of Animal Sciences University of Florida, Gainesville Florida 32611-0910 Ph: 352-392-5590 Fax: 352-392-5595 The Growing
Clinical Policy Committee
 Northern, Eastern and Western Devon Clinical Commissioning Group South Devon and Torbay Clinical Commissioning Group Clinical Policy Committee Commissioning policy: Assisted Conception Fertility assessment
Northern, Eastern and Western Devon Clinical Commissioning Group South Devon and Torbay Clinical Commissioning Group Clinical Policy Committee Commissioning policy: Assisted Conception Fertility assessment
IVF OVERVIEW. Tracy Telles, M.D.
 IVF OVERVIEW By Tracy Telles, M.D. Dr. Hendler: Hello and welcome to KP Healthcast. I m your host Dr. Peter Hendler and today our guest is Dr. Tracy Telles. Dr. Telles is an IVF physician in Kaiser Walnut
IVF OVERVIEW By Tracy Telles, M.D. Dr. Hendler: Hello and welcome to KP Healthcast. I m your host Dr. Peter Hendler and today our guest is Dr. Tracy Telles. Dr. Telles is an IVF physician in Kaiser Walnut
In Vitro Fertilization (IVF) Page 1 of 11
 In Vitro Fertilization (IVF) Page 1 of 11 This document is a part of your informed consent process. Both partners should read the entire document carefully. In vitro fertilization (IVF) is a treatment
In Vitro Fertilization (IVF) Page 1 of 11 This document is a part of your informed consent process. Both partners should read the entire document carefully. In vitro fertilization (IVF) is a treatment
CONSENT TO PARTICIPATE IN THE IN VITRO FERTILIZATION-EMBRYO TRANSFER PROGRAM
 CONSENT TO PARTICIPATE IN THE IN VITRO FERTILIZATION-EMBRYO TRANSFER PROGRAM I, after consultation with my physician, request to participate in the In Vitro Fertilization (IVF)-Embryo Transfer (ET) procedures
CONSENT TO PARTICIPATE IN THE IN VITRO FERTILIZATION-EMBRYO TRANSFER PROGRAM I, after consultation with my physician, request to participate in the In Vitro Fertilization (IVF)-Embryo Transfer (ET) procedures
Lesbian Pregnancy: Donor Insemination
 Lesbian Pregnancy: Donor Insemination (Based on an article originally published in the American Fertility Association 2010 National Fertility and Adoption Directory. Much of this information will also
Lesbian Pregnancy: Donor Insemination (Based on an article originally published in the American Fertility Association 2010 National Fertility and Adoption Directory. Much of this information will also
AGE & FERTILITY: Effective Evaluation & Treatment I. LANE WONG, MD, FACOG. www.hopefertilitycenter.com www.hopeivf.com
 Page 1 of 6 AGE & FERTILITY: Effective Evaluation & Treatment I. LANE WONG, MD, FACOG. www.hopefertilitycenter.com www.hopeivf.com Age has a profound effect on female fertility. This is common knowledge,
Page 1 of 6 AGE & FERTILITY: Effective Evaluation & Treatment I. LANE WONG, MD, FACOG. www.hopefertilitycenter.com www.hopeivf.com Age has a profound effect on female fertility. This is common knowledge,
ANS 431 - Reproductive Physiology of Domestic Animals (Spring 2015)
 1 ANS 431 - Reproductive Physiology of Domestic Animals (Spring 2015) Instructor: Dr. Eduardo L. Gastal, DVM, MS, PhD Room: AG 129; Phone: 453-1774; E-mail: egastal@siu.edu Office hours: MWF 11-12 a.m.;
1 ANS 431 - Reproductive Physiology of Domestic Animals (Spring 2015) Instructor: Dr. Eduardo L. Gastal, DVM, MS, PhD Room: AG 129; Phone: 453-1774; E-mail: egastal@siu.edu Office hours: MWF 11-12 a.m.;
Consent to Perform Preimplantation Genetic Screening (PGS) using. Comparative Genomic Hybridization (acgh) or Next Generation Sequencing (NGS)
 Consent to Perform Preimplantation Genetic Screening (PGS) using Array Comparative Genomic Hybridization (acgh ) or Next Generation Sequencing (NGS) Purpose The purpose of Preimplantation Genetic Screening
Consent to Perform Preimplantation Genetic Screening (PGS) using Array Comparative Genomic Hybridization (acgh ) or Next Generation Sequencing (NGS) Purpose The purpose of Preimplantation Genetic Screening
Effects of in-vitro fertilization, culture, freezing and transfer on the ability of mouse embryos to implant and survive
 Effects of in-vitro fertilization, culture, freezing and transfer on the ability of mouse embryos to implant and survive A. Massip, P. Van der Zwalmen, F. Puissant, M. Camus and F. Leroy Department of
Effects of in-vitro fertilization, culture, freezing and transfer on the ability of mouse embryos to implant and survive A. Massip, P. Van der Zwalmen, F. Puissant, M. Camus and F. Leroy Department of
Reproductive Technology. Chapter 21
 Reproductive Technology Chapter 21 Assisted Reproduction When a couple is sub-fertile or infertile they may need Assisted Reproduction to become pregnant: Replace source of gametes Sperm, oocyte or zygote
Reproductive Technology Chapter 21 Assisted Reproduction When a couple is sub-fertile or infertile they may need Assisted Reproduction to become pregnant: Replace source of gametes Sperm, oocyte or zygote
Patient Information: Endometriosis Disease Process and Treatment
 1 William N. Burns, M. D. Associate Professor Department of Obstetrics & Gynecology Joan C. Edwards School of Medicine Marshall University Huntington, West Virginia, USA Patient Information: Endometriosis
1 William N. Burns, M. D. Associate Professor Department of Obstetrics & Gynecology Joan C. Edwards School of Medicine Marshall University Huntington, West Virginia, USA Patient Information: Endometriosis
Assignment Discovery Online Curriculum
 Assignment Discovery Online Curriculum Lesson title: In Vitro Fertilization Grade level: 9-12, with adaptation for younger students Subject area: Life Science Duration: Two class periods Objectives: Students
Assignment Discovery Online Curriculum Lesson title: In Vitro Fertilization Grade level: 9-12, with adaptation for younger students Subject area: Life Science Duration: Two class periods Objectives: Students
ANS 3319C Reproductive Physiology and Endocrinology Artificial Insemination in Cattle. Objectives. What are the advantages and disadvantages of AI?
 ANS 3319C Reproductive Physiology and Endocrinology Artificial Insemination in Cattle Objectives 1) To provide an overview of the process of artificial insemination (AI) in cattle. 2) To gain an understanding
ANS 3319C Reproductive Physiology and Endocrinology Artificial Insemination in Cattle Objectives 1) To provide an overview of the process of artificial insemination (AI) in cattle. 2) To gain an understanding
In Vitro Fertilization
 Patient Education In Vitro Fertilization What to expect This handout describes how to prepare for and what to expect when you have in vitro fertilization. It provides written information about this process,
Patient Education In Vitro Fertilization What to expect This handout describes how to prepare for and what to expect when you have in vitro fertilization. It provides written information about this process,
ART - - - - - IVF - NO.1 IVF - IVF
 The success of any ART laboratory depends on its IVF laboratory. The primary function of an ART laboratory is to provide an optimal environment for gametes and embryos. To set up an ART laboratory, the
The success of any ART laboratory depends on its IVF laboratory. The primary function of an ART laboratory is to provide an optimal environment for gametes and embryos. To set up an ART laboratory, the
Areas of Concern. Reproductive Ethics: Issues &
 Reproductive Ethics: Issues & Areas of Concern Conception Control: under what conditions is conception control in harmony with a Christian ethic? Genetic Screening & Counseling: under what conditions should
Reproductive Ethics: Issues & Areas of Concern Conception Control: under what conditions is conception control in harmony with a Christian ethic? Genetic Screening & Counseling: under what conditions should
ASSISTED REPRODUCTIVE TECHNOLOGIES (ART)
 ASSISTED REPRODUCTIVE TECHNOLOGIES (ART) Dr. Herve Lucas, MD, PhD, Biologist, Andrologist Dr. Taher Elbarbary, MD Gynecologist-Obstetrician Definitions of Assisted Reproductive Technologies Techniques
ASSISTED REPRODUCTIVE TECHNOLOGIES (ART) Dr. Herve Lucas, MD, PhD, Biologist, Andrologist Dr. Taher Elbarbary, MD Gynecologist-Obstetrician Definitions of Assisted Reproductive Technologies Techniques
How To Get A Refund On An Ivf Cycle
 100% IVF Refund Program Community Hospital North Clearvista Dr. N Dr. David Carnovale and Dr. Jeffrey Boldt, along with everyone at Community Reproductive Endocrinology, are committed to providing you
100% IVF Refund Program Community Hospital North Clearvista Dr. N Dr. David Carnovale and Dr. Jeffrey Boldt, along with everyone at Community Reproductive Endocrinology, are committed to providing you
Page 1. 1. The production of monoploid cells by spermatogenesis occurs in (1) zygotes (3) ovaries (2) testes (4) meristems
 1. The production of monoploid cells by spermatogenesis occurs in (1) zygotes (3) ovaries (2) testes (4) meristems Base your answers to questions 2 and 3 on the diagram below of the female reproductive
1. The production of monoploid cells by spermatogenesis occurs in (1) zygotes (3) ovaries (2) testes (4) meristems Base your answers to questions 2 and 3 on the diagram below of the female reproductive
Artificial Insemination in Goats: An Update Sukanya Leethongdeei' Suppawiwat Ponglowhapan^
 Review Article Artificial Insemination in Goats: An Update Sukanya Leethongdeei' Suppawiwat Ponglowhapan^ Abstract ««oriated with the sperm collection, storage and use. The success of the artificial insemination
Review Article Artificial Insemination in Goats: An Update Sukanya Leethongdeei' Suppawiwat Ponglowhapan^ Abstract ««oriated with the sperm collection, storage and use. The success of the artificial insemination
Training farmers to perform artificial insemination in sheep
 Training farmers to perform artificial insemination in sheep FNC13-901 Project Type: Farmer/Rancher Project Projected End Date: 2015 Funds Awarded: $19,980 Region: North Central State: Ohio Coordinators:
Training farmers to perform artificial insemination in sheep FNC13-901 Project Type: Farmer/Rancher Project Projected End Date: 2015 Funds Awarded: $19,980 Region: North Central State: Ohio Coordinators:
INFORMED CONSENT AND AUTHORIZATION FOR IN VITRO FERTILIZATION OF PREVIOUSLY CRYOPRESERVED OOCYTES
 INFORMED CONSENT AND AUTHORIZATION FOR IN VITRO FERTILIZATION OF PREVIOUSLY CRYOPRESERVED OOCYTES We, the undersigned, as patient and partner understand that we will be undergoing one or more procedures
INFORMED CONSENT AND AUTHORIZATION FOR IN VITRO FERTILIZATION OF PREVIOUSLY CRYOPRESERVED OOCYTES We, the undersigned, as patient and partner understand that we will be undergoing one or more procedures
Revised minimum standards for in vitro fertilization, gamete intrafallopian transfer, and related procedures
 Revised minimum standards for in vitro fertilization, gamete intrafallopian transfer, and related procedures A PRACTICE COMMITTEE REPORT Guidelines and Minimum Standards I. Introduction Treatment of the
Revised minimum standards for in vitro fertilization, gamete intrafallopian transfer, and related procedures A PRACTICE COMMITTEE REPORT Guidelines and Minimum Standards I. Introduction Treatment of the
Consent for Frozen Donor Oocyte In Vitro Fertilization and Embryo Transfer (Recipient)
 Name of Patient: Name of Partner: We, the Patient and Partner (if applicable) named above, are each over the age of twenty-one (21) years. By our signatures below, I/we request and authorize the performance
Name of Patient: Name of Partner: We, the Patient and Partner (if applicable) named above, are each over the age of twenty-one (21) years. By our signatures below, I/we request and authorize the performance
Recent Progress in In Vitro Fertilization and Intracytoplasmic Sperm Injection Technologies in Japan
 Research and Reviews Recent Progress in In Vitro Fertilization and Intracytoplasmic Sperm Injection Technologies in Japan JMAJ 52(1): 29 33, 2009 Kaoru YANAGIDA* 1 Abstract The three basic pillars of fertility
Research and Reviews Recent Progress in In Vitro Fertilization and Intracytoplasmic Sperm Injection Technologies in Japan JMAJ 52(1): 29 33, 2009 Kaoru YANAGIDA* 1 Abstract The three basic pillars of fertility
Welcome to chapter 2. The following chapter is called "Indications For IVF". The author is Dr Kamini A. Rao.
 Welcome to chapter 2. The following chapter is called "Indications For IVF". The author is Dr Kamini A. Rao. The indications for an IVF treatment have increased since the birth of the first IVF baby. The
Welcome to chapter 2. The following chapter is called "Indications For IVF". The author is Dr Kamini A. Rao. The indications for an IVF treatment have increased since the birth of the first IVF baby. The
Artificial insemination with frozen semen in dogs: A retrospective study of 10 years using a non-surgical approach
 Theriogenology 66 (2006) 1645 1650 www.journals.elsevierhealth.com/periodicals/the Artificial insemination with frozen semen in dogs: A retrospective study of 10 years using a non-surgical approach R.
Theriogenology 66 (2006) 1645 1650 www.journals.elsevierhealth.com/periodicals/the Artificial insemination with frozen semen in dogs: A retrospective study of 10 years using a non-surgical approach R.
PRODUCERS can choose to use natural
 Artificial Insemination PRODUCERS can choose to use natural or artificial means of breeding their animals. Technology has advanced in the last 30 to 40 years to allow animal producers to use means other
Artificial Insemination PRODUCERS can choose to use natural or artificial means of breeding their animals. Technology has advanced in the last 30 to 40 years to allow animal producers to use means other
Guide to IVF Laboratory Results
 Guide to IVF Laboratory Results PACIFIC CENTRE FOR REPRODUCTIVE MEDICINE 500-4601 Canada Way, Burnaby BC V5G 4X7 LAB-416-20140122-1 The following information will guide you through what results to expect
Guide to IVF Laboratory Results PACIFIC CENTRE FOR REPRODUCTIVE MEDICINE 500-4601 Canada Way, Burnaby BC V5G 4X7 LAB-416-20140122-1 The following information will guide you through what results to expect
Anatomy and Physiology of Human Reproduction. Module 10a
 This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike License. Your use of this material constitutes acceptance of that license and the conditions of use of materials on this
This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike License. Your use of this material constitutes acceptance of that license and the conditions of use of materials on this
PICSI Sperm Selection Device Instructions for Use
 PICSI Sperm Selection Device Instructions for Use Manufacturer: Biocoat, Inc., 211 Witmer Rd., Horsham, PA 19044 USA Distributor: MidAtlantic Diagnostics, Inc., Telephone: 856-762-2000, www.midatlanticdiagnostics.com
PICSI Sperm Selection Device Instructions for Use Manufacturer: Biocoat, Inc., 211 Witmer Rd., Horsham, PA 19044 USA Distributor: MidAtlantic Diagnostics, Inc., Telephone: 856-762-2000, www.midatlanticdiagnostics.com
Clinical Reference Group Quality & Safety Committee Governing Body. Policy Screened
 Fertility Policy 1 SUMMARY This policy is intended to support individuals and couples who want to become parents but who have a possible pathological problem (physical or psychological) leading to them
Fertility Policy 1 SUMMARY This policy is intended to support individuals and couples who want to become parents but who have a possible pathological problem (physical or psychological) leading to them
DARTMOUTH-HITCHCOCK MEDICAL CENTER Lebanon, New Hampshire IN VITRO FERTILIZATION PROCEDURE DESCRIPTION
 DARTMOUTH-HITCHCOCK MEDICAL CENTER Lebanon, New Hampshire IN VITRO FERTILIZATION PROCEDURE DESCRIPTION I. INTRODUCTION A. The Assisted Reproductive Technology (ART) Program. The ART Program is operated
DARTMOUTH-HITCHCOCK MEDICAL CENTER Lebanon, New Hampshire IN VITRO FERTILIZATION PROCEDURE DESCRIPTION I. INTRODUCTION A. The Assisted Reproductive Technology (ART) Program. The ART Program is operated
Reproductive System & Development: Practice Questions #1
 Reproductive System & Development: Practice Questions #1 1. Which two glands in the diagram produce gametes? A. glands A and B B. glands B and E C. glands C and F D. glands E and F 2. Base your answer
Reproductive System & Development: Practice Questions #1 1. Which two glands in the diagram produce gametes? A. glands A and B B. glands B and E C. glands C and F D. glands E and F 2. Base your answer
Director, IVF Program, Division of Reproductive Endocrinology & Infertility
 Director, IVF Program, Division of Reproductive Endocrinology & Infertility Date: January 17, 2006 To: From: RE: All IVF candidates Chief, Reproductive Endocrinology & Infertility Criteria for IVF program
Director, IVF Program, Division of Reproductive Endocrinology & Infertility Date: January 17, 2006 To: From: RE: All IVF candidates Chief, Reproductive Endocrinology & Infertility Criteria for IVF program
COVENTRY HEALTH CARE OF ILLINOIS, INC. COVENTRY HEALTH CARE OF MISSOURI, INC. Medical Management Policy and Procedure PROPRIETARY
 COVENTRY HEALTH CARE OF ILLINOIS, INC. COVENTRY HEALTH CARE OF MISSOURI, INC. Medical Management Policy and Procedure PROPRIETARY Policy: Infertility Evaluation and Treatment Number: MM 1306 Date Effective:
COVENTRY HEALTH CARE OF ILLINOIS, INC. COVENTRY HEALTH CARE OF MISSOURI, INC. Medical Management Policy and Procedure PROPRIETARY Policy: Infertility Evaluation and Treatment Number: MM 1306 Date Effective:
How to choose an IVF clinic and understand success rates: Questions to ask when choosing an IVF clinic.
 Australia s National Infertility Network How to choose an IVF clinic and understand success rates: Questions to ask when choosing an IVF clinic. updated 26 05 2015 20 The information contained here is
Australia s National Infertility Network How to choose an IVF clinic and understand success rates: Questions to ask when choosing an IVF clinic. updated 26 05 2015 20 The information contained here is
Reproduction and its Hormonal Control
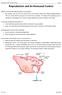 Reproduction and its Hormonal Control Page 1 Reproduction and its Hormonal Control Different mammals have different patterns of reproduction Eg mammals, rats and mice can breed all year round, whereas
Reproduction and its Hormonal Control Page 1 Reproduction and its Hormonal Control Different mammals have different patterns of reproduction Eg mammals, rats and mice can breed all year round, whereas
Headquarters in Sioux Center, IA 1
 Nicholas Lemmel Cornfields, soybeans, and cows, I had finally arrived at Trans Ova Genetics Headquarters in Sioux Center, Iowa. I pulled into the drive of the intern house located on the corner of the
Nicholas Lemmel Cornfields, soybeans, and cows, I had finally arrived at Trans Ova Genetics Headquarters in Sioux Center, Iowa. I pulled into the drive of the intern house located on the corner of the
Artificial Reproductive Technologies I: insemination
 Artificial Reproductive Technologies I: insemination Cinzia Allegrucci LO s List the main advantages and disadvantages of artificial insemination (AI) Explain methods for evaluating volume and concentration,
Artificial Reproductive Technologies I: insemination Cinzia Allegrucci LO s List the main advantages and disadvantages of artificial insemination (AI) Explain methods for evaluating volume and concentration,
TIMING THE FERTILE PERIOD OF THE BITCH: BRIEF REVIEW
 TIMING THE FERTILE PERIOD OF THE BITCH: BRIEF REVIEW Canine Reproduction Seminar Dr. Scarlette Gotwals Stages of the Estrous Cycle Proestrus: Estrus: Diestrus: Anestrus: Vulvar swelling and bloody vaginal
TIMING THE FERTILE PERIOD OF THE BITCH: BRIEF REVIEW Canine Reproduction Seminar Dr. Scarlette Gotwals Stages of the Estrous Cycle Proestrus: Estrus: Diestrus: Anestrus: Vulvar swelling and bloody vaginal
FERTILITY AND AGE. Introduction. Fertility in the later 30's and 40's. Am I fertile?
 FERTILITY AND AGE Introduction Delaying pregnancy is a common choice for women in today's society. The number of women in their late 30s and 40s attempting pregnancy and having babies has increased in
FERTILITY AND AGE Introduction Delaying pregnancy is a common choice for women in today's society. The number of women in their late 30s and 40s attempting pregnancy and having babies has increased in
THE CENTER FOR ADVANCED REPRODUCTIVE SERVICES (CARS) (The Center) CONSENT FOR IN VITRO FERTILIZATION AND EMBRYO TRANSFER
 THE CENTER FOR ADVANCED REPRODUCTIVE SERVICES (CARS) (The Center) CONSENT FOR IN VITRO FERTILIZATION AND EMBRYO TRANSFER Partner #1 Last Name (Surname): Partner #1 First Name: Partner #1 Last 5 Digits
THE CENTER FOR ADVANCED REPRODUCTIVE SERVICES (CARS) (The Center) CONSENT FOR IN VITRO FERTILIZATION AND EMBRYO TRANSFER Partner #1 Last Name (Surname): Partner #1 First Name: Partner #1 Last 5 Digits
Methods for improvement of the success rate of artificial insemination with donor semen
 INTERNATIONAL JOURNAL OFANDROLOGY 9 (1986) 14-2 Departments of Internul Medicine and Obstetrics, State University Hospital, Gent, Belgium Methods for improvement of the success rate of artificial insemination
INTERNATIONAL JOURNAL OFANDROLOGY 9 (1986) 14-2 Departments of Internul Medicine and Obstetrics, State University Hospital, Gent, Belgium Methods for improvement of the success rate of artificial insemination
Overview of Artificial Insemination of Kentucky Meat and Dairy Goats Terry Hutchens, Extension Associate University of Kentucky (G10307)
 Overview of Artificial Insemination of Kentucky Meat and Dairy Goats Terry Hutchens, Extension Associate University of Kentucky (G10307) General Prospective Kentucky goat producers can make great strides
Overview of Artificial Insemination of Kentucky Meat and Dairy Goats Terry Hutchens, Extension Associate University of Kentucky (G10307) General Prospective Kentucky goat producers can make great strides
Course: AG 534 Zoology - Science of Animal Reproduction
 Course: AG 53 Zoology - Science of Animal Reproduction Unit Objective CAERT Lesson Plan Library Unit Problem Area Les son Animal. Plant & Soil Science 1 1,2, 3 Introduction to Animal Science Match terms
Course: AG 53 Zoology - Science of Animal Reproduction Unit Objective CAERT Lesson Plan Library Unit Problem Area Les son Animal. Plant & Soil Science 1 1,2, 3 Introduction to Animal Science Match terms
Unit 3 REPRODUCTIVE SYSTEMS AND THE MENSTRUAL CYCLE
 Unit 3 REPRODUCTIVE SYSTEMS AND THE MENSTRUAL CYCLE Learning Objectives By the end of this unit, the learner should be able to: Explain the importance of understanding the male and female reproductive
Unit 3 REPRODUCTIVE SYSTEMS AND THE MENSTRUAL CYCLE Learning Objectives By the end of this unit, the learner should be able to: Explain the importance of understanding the male and female reproductive
Uterine fibroids (Leiomyoma)
 Uterine fibroids (Leiomyoma) What are uterine fibroids? Uterine fibroids are fairly common benign (not cancer) growths in the uterus. They occur in about 25 50% of all women. Many women who have fibroids
Uterine fibroids (Leiomyoma) What are uterine fibroids? Uterine fibroids are fairly common benign (not cancer) growths in the uterus. They occur in about 25 50% of all women. Many women who have fibroids
Abnormal Uterine Bleeding
 Abnormal Uterine Bleeding WOMENCARE A Healthy Woman is a Powerful Woman (407) 898-1500 Abnormal uterine bleeding is one of the most common reasons women see their doctors. It can occur at any age and has
Abnormal Uterine Bleeding WOMENCARE A Healthy Woman is a Powerful Woman (407) 898-1500 Abnormal uterine bleeding is one of the most common reasons women see their doctors. It can occur at any age and has
Reproduction in Mammals
 Reproduction in Mammals A. Introduction 1. The organs of the male and female reproductive systems ensure the continuation of the species. 2. They do this by producing gametes and by providing a method
Reproduction in Mammals A. Introduction 1. The organs of the male and female reproductive systems ensure the continuation of the species. 2. They do this by producing gametes and by providing a method
INTRODUCTION TO ARTIFICIAL INSEMINATION APICHAI POONCHAI D.V.M.
 INTRODUCTION TO ARTIFICIAL INSEMINATION APICHAI POONCHAI D.V.M. In natural mating a bull mounts a cow & ejuculates his semen in her vagina INTRODUCTION In A.I. Semen is collected from a bull in an artificial
INTRODUCTION TO ARTIFICIAL INSEMINATION APICHAI POONCHAI D.V.M. In natural mating a bull mounts a cow & ejuculates his semen in her vagina INTRODUCTION In A.I. Semen is collected from a bull in an artificial
The following chapter is called "Follow-ups with a Positive or a Negative Pregnancy Test".
 Slide 1 Welcome to chapter 7. The following chapter is called "Follow-ups with a Positive or a Negative Pregnancy Test". The author is Professor Pasquale Patrizio. Slide 2 This chapter has the following
Slide 1 Welcome to chapter 7. The following chapter is called "Follow-ups with a Positive or a Negative Pregnancy Test". The author is Professor Pasquale Patrizio. Slide 2 This chapter has the following
Hysterosalpingography
 Scan for mobile link. Hysterosalpingography Hysterosalpingography uses a real-time form of x-ray called fluoroscopy to examine the uterus and fallopian tubes of a woman who is having difficulty becoming
Scan for mobile link. Hysterosalpingography Hysterosalpingography uses a real-time form of x-ray called fluoroscopy to examine the uterus and fallopian tubes of a woman who is having difficulty becoming
Proceedings, Applied Reproductive Strategies in Beef Cattle November 1 and 2, 2005, Lexington, Kentucky TIMING OF INSEMINATION IN BEEF HEIFERS
 Proceedings, Applied Reproductive Strategies in Beef Cattle November 1 and 2, 2005, Lexington, Kentucky TIMING OF INSEMINATION IN BEEF HEIFERS John B. Hall and Benjamin R. Dorsey Department of Animal &
Proceedings, Applied Reproductive Strategies in Beef Cattle November 1 and 2, 2005, Lexington, Kentucky TIMING OF INSEMINATION IN BEEF HEIFERS John B. Hall and Benjamin R. Dorsey Department of Animal &
Informed Consent Packet - In Vitro Fertilization (IVF)
 Center for Reproductive Medicine (CRM) Informed Consent Packet - In Vitro Fertilization (IVF) This packet contains the required IVF treatment consent documents. Please read, consider and, if you agree,
Center for Reproductive Medicine (CRM) Informed Consent Packet - In Vitro Fertilization (IVF) This packet contains the required IVF treatment consent documents. Please read, consider and, if you agree,
Understanding Fertility
 Understanding Fertility 6 Introduction The word fertile means the ability to become pregnant or to cause pregnancy. Basic knowledge of both the male and female reproductive systems is important for understanding
Understanding Fertility 6 Introduction The word fertile means the ability to become pregnant or to cause pregnancy. Basic knowledge of both the male and female reproductive systems is important for understanding
University Hospitals Coventry and Warwickshire NHS Trust. Centre for Reproductive Medicine. We Care. We Achieve. We Innovate.
 University Hospitals Coventry and Warwickshire NHS Trust Centre for Reproductive Medicine We Care. We Achieve. We Innovate. Introduction We were the first NHS Hospital in the West Midlands to set up a
University Hospitals Coventry and Warwickshire NHS Trust Centre for Reproductive Medicine We Care. We Achieve. We Innovate. Introduction We were the first NHS Hospital in the West Midlands to set up a
Von Mäusen und Menschen E - 1
 Von Mäusen und Menschen E - 1 Mus musculus: Genetic Portrait of the House Mouse E - 3 Outline Mouse genome Mouse life cycle Transgenic protocols Addition of genes by nuclear injection Removal of genes
Von Mäusen und Menschen E - 1 Mus musculus: Genetic Portrait of the House Mouse E - 3 Outline Mouse genome Mouse life cycle Transgenic protocols Addition of genes by nuclear injection Removal of genes
Stem Cell Quick Guide: Stem Cell Basics
 Stem Cell Quick Guide: Stem Cell Basics What is a Stem Cell? Stem cells are the starting point from which the rest of the body grows. The adult human body is made up of hundreds of millions of different
Stem Cell Quick Guide: Stem Cell Basics What is a Stem Cell? Stem cells are the starting point from which the rest of the body grows. The adult human body is made up of hundreds of millions of different
IN VITRO FERTILISATION IVF and ICSI
 IN VITRO FERTILISATION IVF and ICSI Page 1 of 7 WHAT ARE IVF and ICSI? IVF is short for in vitro fertilisation which means fertilisation outside the body. It usually involves stimulation of the ovaries
IN VITRO FERTILISATION IVF and ICSI Page 1 of 7 WHAT ARE IVF and ICSI? IVF is short for in vitro fertilisation which means fertilisation outside the body. It usually involves stimulation of the ovaries
Effect of honeybee royal jelly on the nuclear maturation of bovine oocytes in vitro
 CPh6.8 Effect of honeybee royal jelly on the nuclear maturation of bovine oocytes in vitro M Kuran 1*, E Sirin 1,E Soydan 1, AG Onal 2 Department of Animal Science, Universities of 1 Ondokuz Mayis, Samsun,
CPh6.8 Effect of honeybee royal jelly on the nuclear maturation of bovine oocytes in vitro M Kuran 1*, E Sirin 1,E Soydan 1, AG Onal 2 Department of Animal Science, Universities of 1 Ondokuz Mayis, Samsun,
Tower Hamlets CCG Fertility policy
 Tower Hamlets CCG Fertility policy Approved December 2014 Introduction Tower Hamlets CCG is responsible for commissioning a range of health services including hospital, mental health and community services
Tower Hamlets CCG Fertility policy Approved December 2014 Introduction Tower Hamlets CCG is responsible for commissioning a range of health services including hospital, mental health and community services
Unit B: Understanding Animal Reproduction. Lesson 3: Understanding Animal Reproduction Technology
 Unit B: Understanding Animal Reproduction Lesson 3: Understanding Animal Reproduction Technology Student Learning Objectives: Instruction in this lesson should result in students achieving the following
Unit B: Understanding Animal Reproduction Lesson 3: Understanding Animal Reproduction Technology Student Learning Objectives: Instruction in this lesson should result in students achieving the following
OUR IVF/ICSI PROGRAMME
 OUR IVF/ICSI PROGRAMME The Manzanera Fertility Clinic has designed a simple fertility programme that aims to be convenient for couples living outside of Spain while maximising your chances of success.
OUR IVF/ICSI PROGRAMME The Manzanera Fertility Clinic has designed a simple fertility programme that aims to be convenient for couples living outside of Spain while maximising your chances of success.
it right? activity (page 4) to highlight ethical issues associated with IVF
 IN VITRO FERTILIZATION I V F In some cases, a sperm is directly injected into an egg IVF: THE MEETING OF SPERM AND EGG IN GLASS Louise Brown, the first test tube baby was born in 1978. Since then, there
IN VITRO FERTILIZATION I V F In some cases, a sperm is directly injected into an egg IVF: THE MEETING OF SPERM AND EGG IN GLASS Louise Brown, the first test tube baby was born in 1978. Since then, there
485 Adopted: 23 Oct 1986
 OECD GUIDELINE FOR TESTING OF CHEMICALS 485 Adopted: 23 Oct 1986 "Genetic Toxicology: Mouse Heritable 1. I N T R O D U C T O R Y I N F O R M A T I O N P r e r e q u i s i t e s Solid, liquid, vapour or
OECD GUIDELINE FOR TESTING OF CHEMICALS 485 Adopted: 23 Oct 1986 "Genetic Toxicology: Mouse Heritable 1. I N T R O D U C T O R Y I N F O R M A T I O N P r e r e q u i s i t e s Solid, liquid, vapour or
Treating heavy menstrual bleeding caused by fibroids or polyps
 Treating heavy menstrual bleeding caused by fibroids or polyps With today s medical advances the outlook for successful treatment of fibroids and polyps has never been better. You don t have to live with
Treating heavy menstrual bleeding caused by fibroids or polyps With today s medical advances the outlook for successful treatment of fibroids and polyps has never been better. You don t have to live with
Final Version Two (Sept 2014) Eastern Cheshire Clinical Commissioning Group NHS Funded Treatment for Subfertility Policy
 Final Version Two (Sept 2014) Eastern Cheshire Clinical Commissioning Group NHS Funded Treatment for Subfertility Policy NHS FUNDED TREATMENT FOR SUBFERTILITY ELIGIBILITY CRITERIA Table of Contents 1.
Final Version Two (Sept 2014) Eastern Cheshire Clinical Commissioning Group NHS Funded Treatment for Subfertility Policy NHS FUNDED TREATMENT FOR SUBFERTILITY ELIGIBILITY CRITERIA Table of Contents 1.
The relevant NICE Clinical Guidance 156, Fertility can be accessed here: http://www.nice.org.uk/guidance/cg156
 City and Hackney CCG Fertility policy Approved January 2015 Introduction City and Hackney CCG is responsible for commissioning a range of health services including hospital, mental health and community
City and Hackney CCG Fertility policy Approved January 2015 Introduction City and Hackney CCG is responsible for commissioning a range of health services including hospital, mental health and community
Consent for In Vitro Fertilization
 Consent for In Vitro Fertilization Print Patient s Name Print Partner s Name We (I), the undersigned, request, authorize and consent to the procedure of In Vitro Fertilization (IVF) and Embryo Transfer
Consent for In Vitro Fertilization Print Patient s Name Print Partner s Name We (I), the undersigned, request, authorize and consent to the procedure of In Vitro Fertilization (IVF) and Embryo Transfer
NON MEDICARE FEES CANBERRA FERTILITY CENTRE VERSION JANUARY 2015 AM QWB 295
 NON MEDICARE FEES CANBERRA FERTILITY CENTRE VERSION JANUARY 2015 AM QWB 295 1 CANBERRA FERTILITY CENTRE Suite 9, Level 2, Peter Yorke Building, Calvary John James Hospital, 173 Strickland Crescent, DEAKIN
NON MEDICARE FEES CANBERRA FERTILITY CENTRE VERSION JANUARY 2015 AM QWB 295 1 CANBERRA FERTILITY CENTRE Suite 9, Level 2, Peter Yorke Building, Calvary John James Hospital, 173 Strickland Crescent, DEAKIN
EARLY PREGNANCY LOSS A Patient Guide to Treatment
 EARLY PREGNANCY LOSS A Patient Guide to Treatment You have a pregnancy that has stopped growing, or you have started to miscarry and the process has not completed. If so, there are four ways to manage
EARLY PREGNANCY LOSS A Patient Guide to Treatment You have a pregnancy that has stopped growing, or you have started to miscarry and the process has not completed. If so, there are four ways to manage
