An update on Crucell s Corporate Social Responsibility program July Passion. for. protection.
|
|
|
- Jeffry Martin
- 8 years ago
- Views:
Transcription
1 An update on Crucell s Corporate Social Responsibility program July 2012 Passion for protection
2 2 Passion for Protection: Crucell s Corporate Social Responsibility Program
3 3 Contents About Crucell 6 Our CSR policy 8 Performance 12 People 25 Planet 32 Philanthropy 36 Images of hope 46 Appendix 51 Crucell welcomes feedback on this report and issues related to our CSR activities. Reactions can be sent to csr@crucell.com Our social responsibility Crucell has always been a company with a deep commitment to society. Our mission is to protect people from infectious diseases by bringing innovative vaccines and antibodies to all who need them. At the same time, we work in other ways to help make the world a better place. Crucell strives to be a good employer and business partner, to respect the planet and to reach out to communities with knowledge and expertise as well as material support. Our commitment Several years ago, Crucell set up a special global program focusing on good corporate citizenship. We call it our corporate social responsibility (CSR) program. Its goal is to maximize our contribution to society and minimize our impact on the environment. Employee engagement, accountability and transparent reporting are key elements of this initiative. In this report, we share highlights from our CSR program during 2011 and outline the way forward this year. An ongoing journey Those of you who have seen our previous annual CSR reports will notice a new approach this year. We have opted for a more informal style of reporting. As the center of excellence for vaccines within the Janssen Pharmaceutical Companies of Johnson & Johnson, we anticipate that the ways we measure CSR progress may change in the future. What will remain is the spirit that has inspired this initiative and, indeed, everything we do as a company dedicated to bringing innovation to global health.
4 4 Passion for Protection: Crucell s Corporate Social Responsibility Program
5 5 A meaningful difference We are proud to present this report on Crucell s CSR program during As relative newcomers to Crucell, we are struck by the breadth and depth of the program, and impressed by the way employees are bringing it to life. One example is Crucell s community outreach initiative called Footprint. This is a program that gives teams of Crucell volunteers the chance to visit disadvantaged communities where Crucell is helping to make a difference to people s lives. The photo reportage on pages shares the experience of one of these teams. In this reportage, we follow the volunteers to the remote areas of Bangladesh, where they learn what vaccines and clean water mean to people who had been living without these basic necessities. We see the same spirit at work in Korea, where teams of Crucell volunteers give up free time to create clean, safe homes for people living in urban squats not far from the Crucell factory in Incheon. Meanwhile, our Government Affairs & Public Health representatives are working with policy makers at the national, regional and global levels to improve access to essential healthcare. On page 43 you can read about what they have been doing to address the cholera crisis in Haiti, for example. Crucell s worldwide team of Environment, Health and Safety (EHS) managers is making good progress with raising safety awareness in our organization, as well as aligning with Johnson & Johnson standards. The appointment of a Crucell Health Care Compliance Officer, supported by Johnson & Johnson s extensive resources in this field, will help to safeguard good business conduct as we grow. During 2011, Crucell employees achieved important advances in discovery and product development, several of which are highlighted in this report. Significant investments in Crucell s research and development capabilities, quality assurance systems and production facilities were initiated. The full social significance of these investments will only be felt years down the track, as Crucell brings a new generation of vaccines to market. Crucell s CSR program is a natural extension of Crucell s mission to make a meaningful difference to global health. We dream of helping to eradicate polio. Of transforming the prevention and treatment of influenza. Of contributing to the prevention of HIV/AIDS. Of helping the world deal with tuberculosis and malaria. Joining the Johnson & Johnson Family of Companies in February last year has given us additional resources and support to try to realize at least some of these dreams. The seeds of this future are being sown now, day after day, by Crucell employees around the globe. This report testifies to their passion and commitment to make the world a better place. That is what Crucell s CSR program is all about. Johan Van Hoof and Bart van Zijll Langhout, Managing Directors of Crucell
6 6 Passion for Protection: Crucell s Corporate Social Responsibility Program Business with a warm Crucell is a global biopharmaceutical company heart dedicated to bringing meaningful innovation to global health. Our teams focus on the discovery, development, manufacture and marketing of vaccines and antibodies to protect people worldwide from major health threats. Crucell s specialty is fighting infectious diseases a growing healthcare challenge. Crucell strives to do business with a warm heart and a cool head, generating sustainable profit in order to continue to innovate and give back to society. Our corporate mission reflects a deep sense of responsibility to the people who live on our planet more than 7 billion today, and counting. Protecting the health and well-being of these billions of individuals is more important and difficult than ever before. That s because trends such as climate change, globalization, urbanization, wider travel and population aging are presenting new opportunities for viruses and bacteria to thrive and spread. Vaccines and antibodies Vaccines and antibody products are complementary approaches to immunization. A vaccine can induce lasting protection sometimes for a lifetime against an infectious threat, whereas an injection of antibodies provides immediate protection but only for a limited period of time (weeks or months). Different infectious diseases or different circumstances call for different approaches. For example, a universal influenza vaccine could protect a person against the flu for life, whereas an influenza antibody product would be invaluable in a pandemic situation. The antibody approach would also help the elderly, who are especially hard hit by flu and less well protected by the flu vaccine than younger people. That s because the immune system deteriorates with age. Immunization is one of the most promising strategies for tackling this problem. It comes in two main forms: vaccines that safely and effectively mobilize the body s own immune defenses against infections, and antibodies that provide ready-made immune protection against invading microbes. Each has its own special role (see box). Crucell s vaccine and antibody researchers are working on innovative immunization products to fight tuberculosis, malaria, HIV/AIDS, influenza, rabies, polio and several other diseases that are high on the list of global health problems. Meanwhile, we are already combating a dozen major infectious threats with our range of marketed vaccines against paediatric, respiratory, and travel or endemic infections. Though infectious diseases don t stop at borders, they home in on poverty and congregate in underdeveloped communities. So the tools to fight them must be afford able and accessible for all. After clean water, immunization is believed to be the most cost-effective health intervention there is, and its key role in promoting socioeconomic development is increasingly being documented. 1 Crucell wants to take this benefit further. That is one reason why we have invested in innovations such as our proprietary PER.C6 human cell-line technology, which promises to enable vaccines and therapeutic proteins (such as antibodies) to be produced in virtually unlimited quantities, much faster and more cheaply than traditional manufacturing methods allow. 1 See references cited on the GAVI Alliance website:
7 7 Our stakeholders Crucell s efforts to bring significant benefit to the lives of people worldwide involve constructive interaction with many different stakeholders. Here is a short list of examples.* Crucell employees: almost 1700 individuals worldwide, united by a shared dream for global health. Colleagues in other Janssen Pharmaceutical Companies. Legislators such as the US Food and Drug Administration (FDA), European Medicines Agency (EMA) and national regulatory authorities. Policy makers: World Health Organization (WHO), Pan American Health Organization (PAHO), GAVI Alliance, and many more. Business partners: DSM Biologics, Novartis, GlaxoSmithKline and Pfizer. Non-profit and academic organizations and government institutions such as Aeras, US National Institutes of Health, IAVI, Harvard University and PATH. Customers: Supranational purchasing organizations such as UNICEF (on behalf of developing countries); public and/or private health organizations in developed countries; physicians and pharmacists. Consumers: All the people worldwide who use our products and participate in clinical trials of our candidate products. *Status at the end of 2011
8 8 Passion for Protection: Crucell s Corporate Social Responsibility Program Crucell s CSR policy is a natural extension of our mission to bring innovation to global health in ways that make a meaningful difference to the lives of people worldwide.
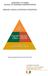
9 9 Taking responsibility Social responsibility is built into the DNA of our organization. But we are conscious of the need to nurture this spirit. As our business expands in line with our ambitions, our impact on the world will increase. Achieving the optimal balance between the benefits we bring to society and the resources we consume will be an ongoing challenge. Crucell s CSR program is a concerted effort to meet that challenge through proactive planning and review. Crucell recognizes four key responsibilities to society and has drawn up a global CSR policy to help all employees work together to meet these responsibilities. The policy is built around a structure we call the 4P framework. This defines the four main ways in which Crucell strives to be a good corporate citizen and contribute to society. Performance First and foremost, we see a responsibility to perform at our best as a business bringing innovation to global health. People We want to take care of the people we work with, inside and outside the company. Planet We strive to care for the planet by minimizing our environmental footprint while maximizing our beneficial impact on global health. Philanthropy We take on the responsibility to do more for society than we must do from a business point of view. The four categories of Performance, People, Planet and Philanthropy are each further subdivided into a number of related themes or responsibilities. These are shown in the figure on the next page.
10 10 Passion for Protection: Crucell s Corporate Social Responsibility Program Performance Saving lives Innovation Responsible supply chain Transparency People Responsible employer Health & safety Business conduct Planet Philanthropy Contributing to science Access to healthcare NGO partnerships Community outreach Emissions to air Emissions to water Consumption Recycling
11 11 A dynamic framework Themes can be added or subtracted over time, as this is a dynamic framework. For example, participating in CSR-related sector initiatives such as the Dow Jones Sustainability Index was a priority for Crucell when it was listed on the stock exchange. We are no longer participating in such initiatives independently but as the center of excellence for vaccines within the Janssen Pharmaceutical Companies of Johnson & Johnson. Intersections The 4P classification system is somewhat artificial, as there is considerable overlap and interdependence among the P-categories and their component themes. However, it serves as a useful reminder of the many different facets of our CSR program and provides a clear overview of what Crucell stands for, as a socially responsible company. Adding value A practical advantage of the 4P framework is that it breaks the task of CSR development and reporting into manageable portions. Early in 2010, one or more leaders ( P-owners ) were appointed for each P-category. They have taken real ownership for progress in their area, providing crucial inspiration and direction as well as coordinating the flow of information. This approach fosters transparency, clarity and focus, which helps the P-owners in consultation with top-level management to define realistic but challenging targets for the coming years. This step is vital for achieving the continuous progress we strive for and ensuring that our CSR strategy delivers tangible benefits for society. Adding real value is our goal. Social responsibility is built into Crucell s DNA. Our CSR program is designed to bring this to full expression. Measuring outcomes Key performance indicators (KPIs) have been defined for each of the four P-categories and their component themes. Coupled to targets, they serve as measures of how well we are living up to our corporate social responsibilities and goals. Like the 4P framework itself, the KPIs can change over time to reflect the evolution of the company and our approach to CSR. The KPIs and targets used in 2011 are shown in the Appendix. Organization A CSR Working Group comprising the P-owners and several Crucell employees with a special advisory role was established in the first quarter of It is a diverse group of senior managers from different functional areas and Crucell sites. Members of the CSR Working Group gather information from all levels and parts of the organization. They also initiate improvements with support and advice from Crucell s Management Committee. The CSR Working Group meets quarterly to discuss progress and formulate plans for the future. These meetings are chaired by Crucell s top management, reflecting the importance we place on good corporate citizenship.
12 12 Passion for Protection: Crucell s Corporate Social Responsibility Program Optimizing performance The P-category we call Performance embraces four social responsibilities that we see as being central to our core business and our mission: saving lives, innovation, responsible supply chain management and transparency, by which we mean the responsibility to evaluate and report on our CSR activities.
13 Performance 13 Saving lives Crucell s licensed vaccines protect people around the world from a range of serious infectious diseases. They prevent illness, suffering and lasting disability. They save lives. Marketed products in 2011 Quinvaxem Fully liquid vaccine for protection against five childhood diseases Hepavax-Gene Recombinant hepatitis B vaccine Epaxal Junior Low-dosage, aluminium-free hepatitis A vaccine (0.25ml) MoRu-Viraten 2 Vaccine for protection against measles and rubella Epaxal Aluminium-free hepatitis A vaccine Vivotif Oral typhoid vaccine Dukoral Oral vaccine against cholera Inflexal V Virosomal adjuvanted influenza vaccine Every year, tens of thousands of babies born in the developing world have a much better chance of a future because they receive our combination vaccine against five serious childhood infections: diphtheria, tetanus, pertussis (whooping cough), hepatitis B and Haemophilus influenzae type B (Hib), a leading cause of severe pneumonia and meningitis. Meanwhile, Crucell s vaccines against hepatitis A, hepatitis B, typhoid fever and cholera safeguard children and adults living in areas where these diseases are endemic, as well as travelers to these regions. And year after year, our influenza vaccine helps at-risk people of all ages from as young as 6 months to avoid a disease with potentially life-threatening respiratory complications. In 2011, Crucell sold well over 107 million vaccine doses to protect people in more than 100 countries around the world. 1 Allowing for the fact that some vaccines inevitably go to waste, we can estimate that a Crucell vaccine was 1 Based on 2011 sales of all Crucell s marketed products. 2 MoRu-Viraten is no longer marketed. Global distribution of Crucell s Quinvaxem pentavalent vaccine in 2011
14 14 Passion for Protection: Crucell s Corporate Social Responsibility Program given to individuals every minute during That s a thought that makes us proud though we are determined to reach even more people in the coming years. Expanding our reach Joining the Janssen Pharmaceutical Companies of Johnson & Johnson in February 2011 has opened up new opportunities for Crucell to deliver life-saving vaccines to all the people who need them. Janssen s commercial organization is truly global, with a presence in virtually every country around the world. To take advantage of this worldwide network, Crucell s local marketing and sales teams are progressively being integrated into the Janssen commercial organization. The transition began in Canada in September This marriage of Crucell s vaccine expertise with Janssen s commercial strength expands the reach of our vaccines within existing and new markets. This will enable us to save more lives and prevent more cases of serious disease. Increasing supply In order to meet growth in demand for Crucell vaccines, we need to build supply. Our goal is to produce more vaccine doses more quickly and efficiently, so that more people worldwide can receive the vaccines they need, when they need them. For several years, we have been working to expand 1 Number reflects sales volume of all Crucell s marketed products (minus 2% estimated wastage) divided by total minutes in 2011: 105,579,320 doses/526,600 minutes = 200 doses per minute mln doses of Crucell vaccines were distributed in more than 100 countries in production capacity, boost productivity, reduce scrap and tighten quality assurance. Becoming part of the Janssen group has given Crucell access to additional expertise and financial support to step up these efforts. Looking ahead, Janssen resources will also be directed towards helping our organization solve supply constraints relating to raw materials, build capabilities for high-scale manufacturing and perform clinical studies to support the development of new markets. New packaging line takes shape Crucell s oral typhoid fever vaccine Vivotif comes in capsules, which are sealed in blister packs, boxed and packed for shipping at one of our production sites in Switzerland. Throughout 2011, a project team has been working on an investment project that will replace the current blistering and packaging equipment and transfer these operations to another Swiss site by All three packaging steps will be combined in a single line, greatly increasing speed and efficiency. Stateof-the-art automation and control features will improve operational safety and quality.
15 Performance people received a Crucell vaccine every minute during New Korean facility fully commissioned Crucell s new manufacturing facility in Korea s Incheon Free Economic Zone started commercial manufacturing of Quinvaxem pentavalent vaccine on September 26, 2011, after the Korean Food and Drug Administration (KFDA) gave the green light. Later that month, the KFDA also approved the facility for production of Hepavax-Gene TF, Crucell s thiomersal-free hepatitis B vaccine. The new plant offers significantly greater production capacity and efficiency compared with its predecessor in Yongin City, Korea. The Incheon facility was approved by the regulatory authorities less than three years after construction work started a feat that testifies to the extraordinary commitment of Crucell employees.
16 16 Passion for Protection: Crucell s Corporate Social Responsibility Program What innovation means to us While our marketed vaccines are already making a significant difference to global health, we want to do much more. Ongoing innovation is the key to realizing this ambition. Crucell s broad research and development (R&D) pipeline currently includes 13 candidate vaccines and antibody products. All are based on Crucell s innovative proprietary technologies, which provide unique opportunities for combating major threats to human health. These threats include some of the world s most powerful and elusive killers, such as HIV/AIDS, tuberculosis and malaria. According to the latest statistics from the World Health Organization, 2.7 million people were newly infected with HIV and there were 1.8 million AIDS deaths in Tuberculosis was responsible for 1.4 million deaths and a staggering 8.8 million new cases of illness in that same year, while malaria afflicted 216 million people and caused 655,000 deaths mainly among children under five. 1 These enormous numbers highlight the importance and urgency of our vaccine research in these disease areas. Influenza is another field where we see a great need and potential for innovation. Every year, flu viruses make millions of people severely ill, claim between 250,000 and 500,000 lives, and burden society with considerable economic losses and healthcare costs. 2 The emergence of a lethal flu virus with pandemic potential could push the death toll into tens of millions. We are tackling these challenges by developing a new-generation seasonal flu vaccine based on our PER.C6 technology, which will overcome the many drawbacks of egg-based vaccine mln R&D investment in 2011, up from mln in production, while simultaneously working on revolutionary approaches to influenza prevention and treatment in the longer term. For the 70,000 people 3 (mostly children) who die of rabies each year, we are developing a monoclonal antibody (mab) product. A person bitten by a rabid animal can be saved by immediate injection of rabies antibodies and vaccine, both of which exist, but the blood-derived antibodies now available are in short supply and too expensive for most developing countries, where the need is greatest. Similarly, we are working with a great sense of urgency to develop a polio vaccine that will support the WHO s efforts to wipe out this ancient disease. Mass vaccination campaigns over the past 20 years have dramatically reduced the number of polio victims worldwide, but the poliovirus is fighting back. The WHO sounded the alarm in May 2012, warning that the world will see an explosive resurgence of polio cases undoing all the progress made unless the job of eradication can be finished. This cannot be achieved with the oral polio vaccine used in developing countries, which contains live-attenuated virus that can revert to a disease-causing form. Crucell s PER.C6 technology holds the key to delivering what the world needs: an inexpensive IPV (inactivated polio vaccine) in large quantities. Using this high-density cell culture process, we hope to be able to grow poliovirus cheaply enough to make IPV available to every child in the world. We don t stop innovating after bringing a new product to market. We look for opportunities to create further value, for example by simplifying administration. Our project to put one of our vaccines into Uniject is an example of this approach. (See page 21) 1 Source: World Health Organization reports for the year Influenza (Seasonal) Fact Sheet, World Health Organization, Global Alliance for Rabies Control, press release September 2011.
17 Performance patents were granted to Crucell in 2011, compared with 220 in 2010 and 146 in Animal welfare These are just some of the ways in which Crucell is working on medical innovations that really matter for individuals and for the world. We strive to lead rather than follow, directing our energies and expertise towards finding and delivering creative solutions for tough health challenges. More information is available on the R&D section of our website. Healthy investment Crucell s investment in R&D climbed to million euro in 2011, one-third higher than the million invested in The increase continues the sharp upward trend seen since 2009, when R&D expenditure was 70.2 million. This healthy injection of funds to drive discovery and product development reflects Crucell s focus on innovation as a key driver for the advancement of global health, as well as the commitment of our new parent organization, Janssen. Before any candidate medical product can be given to humans, it must be rigorously tested in preclinical (non-human) models, in compliance with the regulations governing the development of new pharmaceutical products. Crucell performs animal testing to the minimum extent that is required by law and in accordance with the highest international standards, which are designed to prevent suffering and minimize any discomfort in the animals tested. Crucell applies the 3R principles Reduce, Refine and Replace to preclinical studies involving animal testing. For example, our scientists have been working for many years to replace animal tests with cellbased assays and these efforts have resulted in a significant reduction of animal testing. Cholera vaccine Crucell researchers in Sweden have substantially improved our ability to deliver Dukoral, Crucell s oral cholera vaccine. Thanks to innovation and the introduction of robust manufacturing processes, average yields of the active ingredient rctb increased by 20% in The higher productivity is sustainable in the long term. 589 employees were involved in R&D in 2011 compared with 440 in 2010.
18 18 Passion for Protection: Crucell s Corporate Social Responsibility Program Innovation in action We are making progress towards our vision of a healthier world. Over the next few pages we share a small selection of innovation highlights from Promising results from our HIV vaccine program Crucell s HIV vaccine researchers and collaborators are encouraged by the results achieved by their novel vaccine regimens in recent preclinical tests. These findings provide strong support for starting trials in humans. In the first week of 2012, the prestigious journal Nature published promising results from a preclinical study of novel HIV vaccine regimens that are being developed by Crucell in collaboration with Harvard and the US Military HIV Research Program (MHRP). The study explored several prime-boost regimens designed to deliver the vaccine in two or three shots containing different immunity-stimulating components. Regimens including Crucell s adenovirus vectors (vaccine carriers) and the MHRP s MVA vector were shown to be highly immunogenic in an animal model. Further analysis of the stored samples provided evidence that the type of immunity elicited by these regimens could play a pivotal role in protecting people against HIV infection. They confirmed the presence of a specific immune response that has recently been identified as a potential correlate of protection against HIV in humans. We were delighted to learn that our best vaccine candidate induced this important immune response, said Maria Grazia Pau, leader of Crucell s HIV program. An HIV discovery team led by Hanneke Schuitemaker has started exploring ways to further boost this particular aspect of immunity. The study results pave the way for clinical testing of the novel HIV vaccine combinations. One of the planned clinical trial sites is the world-class CAPRISA center for AIDS prevention and research in Durban, South Africa. Crucell s Laura Digilio, clinical leader of the HIV program, and collaborating researcher Dan Barouch of Harvard visited the site together last November. They also visited a rural clinic west of Durban, in an area where 8 9% of teenage girls become infected with HIV. What we saw made us painfully aware of the devastation AIDS is causing in disadvantaged communities despite intervention efforts and made it very clear that a vaccine is needed to slow the spread of HIV, Digilio said. A step closer to saving people from rabies A phase II clinical trial of Crucell s rabies antibody combination started in India just before the end of This was good news for our clinical team, who have been working for several years to get the go-ahead from the Indian regulatory authorities. This takes us a step closer to our goal: saving around 70,000 people every year from a gruesome death, said Crucell s rabies program director Brad Duncan. An estimated 20,000 of these deaths occur in India, mainly among children. The trial will test our candidate antibody product (together with rabies vaccine) in a clinical setting that simulates its intended use: to provide immediate protection against rabies after exposure to the rabies virus. It will include 240 subjects and focus mainly on gathering a large amount of safety data. These findings will build on the data collected in smaller phase II trials in the USA and Philippines, and pave the way for a definitive phase III efficacy trial in patients exposed to rabies virus. Step by step, we are working to deliver life-saving innovations.
19 Performance 19 A cocktail of two antibodies like these may be sufficient to neutralize most influenza A subtypes says Robert Friesen, who heads Crucell s antibody discovery group. This paves the way for developing broad-spectrum antibody therapies that could save the thousands of people now dying from flu each year and help to stop a potential pandemic in its tracks. A revolutionary approach to flu Crucell is blazing a trail with its discovery and development of broad-spectrum antibodies against influenza. The ultimate goal is to deliver immunization products that will revolutionize the fight against seasonal and pandemic flu. Back in 2008, Crucell scientists were the first in the world to find antibodies that can neutralize a broad range of influenza viruses. The vast majority of antibodies our immune system makes in response to an invading flu virus (or vaccine) are quite specific: they won t protect against the next strain that comes along. So Crucell s discovery was a breakthrough that raised hopes of developing a completely new approach to combating flu: a one-shotfits-all antibody treatment or even a universal vaccine. Other research groups soon confirmed the importance of Crucell s finding by reporting the discovery of similar broadly neutralizing influenza antibodies. Like ours, these new antibodies were effective against a variety of influenza A group 1 viruses. Crucell researchers have since discovered broad-spectrum antibodies against influenza A group 2 viruses and type B viruses as well. This covers all the influenza viruses that make people ill. The novel broad-spectrum antibody against influenza A group 2 viruses was handed over to our development teams last year, after performing strongly in preclinical tests. These encouraging results were published in Science journal in August Similar results for Crucell s leading broadly neutralizing antibody against group 1 viruses were published in Science journal in February Protecting vulnerable groups The need for such a therapy is very clear. Current flu vaccination does not generate adequate protection in the people who need it most: those whose bodies are unable to mount a strong immune response. This includes the elderly (because the immune system deteriorates with age) and immunocompromized patients. More and more influenza strains are showing resistance to antiviral drugs such as oseltamivir, which are currently the only treatment option for influenza. And antivirals are useless once patients develop serious symptoms. Towards a universal flu vaccine Crucell also dreams of designing a vaccine that will protect people for life against virtually every future influenza virus those causing ordinary seasonal flu and those with pandemic potential. Crystal structure studies have provided molecular snapshots of these antibodies attached to influenza viruses, enabling researchers to locate and study their binding sites on the viral surface. We now know that ordinary influenza antibodies bind to parts of the virus that rapidly mutate and change shape, whereas the broadly neutralizing antibodies bind to stable sites that are not subject to mutation and change. Armed with this knowledge, Crucell researchers are trying to rebuild these stable binding domains in a form that can induce the production of broadly neutralizing antibodies. It s a very complex and difficult task, says Crucell s Katarina Radosevic, VP Immunology & Proof of Concept. But if we can make this work in preclinical models, we will be a step closer to a revolutionary flu vaccine. The significance of this work was highlighted in a Nature special on influenza in December 2011.
20 20 Passion for Protection: Crucell s Corporate Social Responsibility Program A new team at the vaccinology frontier Crucell launched an Innovation & Discovery Laboratory in La Jolla, San Diego, in July The new team is in the right place at the right time to help tackle some of the largest issues in global health. Vaccinologists have gone as far as they can go with the traditional approach to vaccine design, says Anthony Williamson, Senior Director of the La Jolla lab. The low-hanging fruit has already been picked and we re left with the difficult diseases. We can t develop the new vaccines we need using empirical ( trial and error ) methods. One example of rational vaccine design is the epitope-based vaccine. Researchers start with a library of monoclonal antibodies targeting specific pathogens, screen them to identify the best ones, define the structural epitopes (binding sites) these antibodies attach to, and then try to turn these molecular structures into vaccines. The problem is that these structural epitopes are often quite poorly immunogenic they don t trigger a strong immune response. We need to understand why. One of the key questions for the future of vaccine design is how to translate epitope structures into immunogenicity, says Williamson. He hopes his team will make a meaningful contribu tion to finding answers, and expects to attract high-quality collaborators. That won t be hard, because Crucell is looking to solve some of the largest issues in global health and is willing to invest in very high-quality basic science to underpin development, he said at the time of launch. The lab s location in San Diego s concentrated bioscience community will facilitate such collaboration. Acclaimed research centers like the Scripps Institute, University of California San Diego and La Jolla Institute for Allergy & Immunology are all virtually within walking distance, and Crucell already has excellent relationships with scientists at these institutes. Expanding horizons Jan Poolman (left) joined Crucell in August 2011 to build Crucell s capabilities for bacterial vaccine innovation. His appointment signals an upcoming expansion of our discovery and pipeline programs, which currently focus on vaccines and antibodies against viral diseases, and vaccines based on adenovirus-vector technology. These were established under the leadership of virologist Jaap Goudsmit (right). In the past, Crucell didn t have the money to do everything a big player can do, so we focused on our capabilities in the viral field. Now, with the support of Janssen, we can afford to widen our focus, says Goudsmit. To realize our ambition for global health, we need to deliver innovative vaccines against viral and bacterial diseases and ultimately noninfectious diseases as well. Goudsmit is already working towards that third level of expansion as director of the Crucell Vaccine Institute. The institute was established towards the end of 2011 to make Crucell s innovative vaccine technology platforms available to Janssen researchers in noninfectious disease areas, like cancer and neurology. Now, with the support of Janssen, we can afford to widen our focus.
Crucell Operational Excellence: Healthy Ambition
 Crucell Operational Excellence: Healthy Ambition Cees de Jong Chief Operating Officer March 12 th, 2008 London Agenda Introduction COO s perspective on Crucell Marketed products Excellent scope for growth
Crucell Operational Excellence: Healthy Ambition Cees de Jong Chief Operating Officer March 12 th, 2008 London Agenda Introduction COO s perspective on Crucell Marketed products Excellent scope for growth
HSA Consumer Guide. Understanding Vaccines, Vaccine Development and Production. www.hsa.gov.sg November 2009. How a Vaccine Works.
 November 2009 Understanding Vaccines, Vaccine Development and Production Vaccines, in general, help protect people from harmful infections before they come in contact with the disease. Vaccines may also
November 2009 Understanding Vaccines, Vaccine Development and Production Vaccines, in general, help protect people from harmful infections before they come in contact with the disease. Vaccines may also
CANADA S VACCINE INDUSTRY COMMITTEE
 WWW.BIOTECH.CA CANADA S VACCINE INDUSTRY COMMITTEE LEADERSHIP IN GLOBAL HEALTH The Vaccine Industry Committee (VIC), Canada s Voice for Vaccines The VIC is comprised of Canada s major vaccine developers
WWW.BIOTECH.CA CANADA S VACCINE INDUSTRY COMMITTEE LEADERSHIP IN GLOBAL HEALTH The Vaccine Industry Committee (VIC), Canada s Voice for Vaccines The VIC is comprised of Canada s major vaccine developers
Bringing innovation to global health. Crucell 2010
 Bringing innovation to global health Crucell 2010 www.crucell.com Contents Our mission Crucell s mission is to protect human lives from infectious diseases by bringing meaningful innovation to global health.
Bringing innovation to global health Crucell 2010 www.crucell.com Contents Our mission Crucell s mission is to protect human lives from infectious diseases by bringing meaningful innovation to global health.
Plan early - get your vaccinations in time for full protection. To prepare for your trip, schedule an appointment: (910) 347-2154, option #2.
 The Onslow County Health Department Travel Clinic offers a complete line of immunizations and prescriptions to protect you while traveling abroad. The most appropriate immunizations and travel medications
The Onslow County Health Department Travel Clinic offers a complete line of immunizations and prescriptions to protect you while traveling abroad. The most appropriate immunizations and travel medications
Vaccination: It s what your child would choose. Your guide to childhood vaccinations.
 Vaccination: It s what your child would choose No-one likes an injection, but your child would never choose to suffer from serious conditions like measles, mumps or whooping cough. Your guide to childhood
Vaccination: It s what your child would choose No-one likes an injection, but your child would never choose to suffer from serious conditions like measles, mumps or whooping cough. Your guide to childhood
AB Volvo, 405 08 Göteborg, Sweden. Ref No 953810003, August 2009. The Volvo Way
 AB Volvo, 405 08 Göteborg, Sweden Ref No 953810003, August 2009 The Volvo Way index Preface Our mission Customers first Customer focus Clear objectives Quality, safety, environmental care Continuous improvements
AB Volvo, 405 08 Göteborg, Sweden Ref No 953810003, August 2009 The Volvo Way index Preface Our mission Customers first Customer focus Clear objectives Quality, safety, environmental care Continuous improvements
Tuberculosis OUR MISSION THE OPPORTUNITY
 Tuberculosis OUR MISSION Guided by the belief that every life has equal value, the Bill & Melinda Gates Foundation works to help all people lead healthy, productive lives. Our Global Health Program is
Tuberculosis OUR MISSION Guided by the belief that every life has equal value, the Bill & Melinda Gates Foundation works to help all people lead healthy, productive lives. Our Global Health Program is
exactly. The need for efficiency in developing effective new therapeutics has never been greater.
 exactly. The need for efficiency in developing effective new therapeutics has never been greater. As demands on the global healthcare system increase and treating disease becomes more complex, the research,
exactly. The need for efficiency in developing effective new therapeutics has never been greater. As demands on the global healthcare system increase and treating disease becomes more complex, the research,
DEVELOPING WORLD HEALTH PARTNERSHIPS DIRECTORY
 DEVELOPING WORLD HEALTH PARTNERSHIPS DIRECTORY OVER 250 HEALTH PARTNERSHIPS THAT MAKE A DIFFERENCE TO PEOPLE S LIVES The global health community needs to overcome both new and current challenges in the
DEVELOPING WORLD HEALTH PARTNERSHIPS DIRECTORY OVER 250 HEALTH PARTNERSHIPS THAT MAKE A DIFFERENCE TO PEOPLE S LIVES The global health community needs to overcome both new and current challenges in the
University Health Services Information Guide 2014-2015. Student Services Building 512-471-4955 healthyhorns.utexas.edu
 University Health Services Information Guide 2014-2015 Student Services Building 512-471-4955 healthyhorns.utexas.edu Message from the Director University Health Services (UHS) supports the academic mission
University Health Services Information Guide 2014-2015 Student Services Building 512-471-4955 healthyhorns.utexas.edu Message from the Director University Health Services (UHS) supports the academic mission
Preparing for and responding to influenza pandemics: Roles and responsibilities of Roche. Revised August 2014
 Preparing for and responding to influenza pandemics: Roles and responsibilities of Roche Revised August 2014 Table of Contents Introduction... 3 Executive summary... 4 Overview... 4 Roche's role... 4 Roche's
Preparing for and responding to influenza pandemics: Roles and responsibilities of Roche Revised August 2014 Table of Contents Introduction... 3 Executive summary... 4 Overview... 4 Roche's role... 4 Roche's
GSK Vaccines: Easing Compliance with SAP Process Control
 2014 SAP AG or an SAP affiliate company. All rights reserved. GSK Vaccines: Easing Compliance with SAP Process Control GlaxoSmithKline Vaccines Industry Life sciences pharmaceuticals Products and Services
2014 SAP AG or an SAP affiliate company. All rights reserved. GSK Vaccines: Easing Compliance with SAP Process Control GlaxoSmithKline Vaccines Industry Life sciences pharmaceuticals Products and Services
Caring, one person at a time.
 JOHNSON & JOHNSON STRATEGIC FRAMEWORK Caring, one person at a time. STRATEG IC FRAM EWOR K 1 Nothing is more important than the health and well-being of those we love. That s why the Johnson & Johnson
JOHNSON & JOHNSON STRATEGIC FRAMEWORK Caring, one person at a time. STRATEG IC FRAM EWOR K 1 Nothing is more important than the health and well-being of those we love. That s why the Johnson & Johnson
Corporate Social Responsibility: Implications for Human Resources and Talent Engagement
 Corporate Social Responsibility: Implications for Human Resources and Talent Engagement Winnie Kwan and Emily Tuuk Center for Advanced Human Resource Studies May 2012 I. Introduction Over the past few
Corporate Social Responsibility: Implications for Human Resources and Talent Engagement Winnie Kwan and Emily Tuuk Center for Advanced Human Resource Studies May 2012 I. Introduction Over the past few
We d like to do the same for you. Owen J. Sullivan CEO, Right Management President, Specialty Brands ManpowerGroup
 Business & Talent. Aligned. Regardless of the economic environment, your industry or geography, your size or your earnings, your most valuable asset is your workforce. How you manage this asset spells
Business & Talent. Aligned. Regardless of the economic environment, your industry or geography, your size or your earnings, your most valuable asset is your workforce. How you manage this asset spells
Optimizing vaccine supply chains
 Optimizing vaccine supply chains More and more vaccines are becoming available Since immunization programs were launched worldwide in the mid-1970s, most countries have been using the same standard package
Optimizing vaccine supply chains More and more vaccines are becoming available Since immunization programs were launched worldwide in the mid-1970s, most countries have been using the same standard package
SECURITIES AND EXCHANGE COMMISSION WASHINGTON, D.C. 20549 FORM 8-K. GEOVAX LABS, INC. (Exact name of registrant as specified in its charter)
 SECURITIES AND EXCHANGE COMMISSION WASHINGTON, D.C. 20549 FORM 8-K CURRENT REPORT Pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934 Date of report (Date of earliest event reported):
SECURITIES AND EXCHANGE COMMISSION WASHINGTON, D.C. 20549 FORM 8-K CURRENT REPORT Pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934 Date of report (Date of earliest event reported):
Adult Vaccination Frequently Asked Questions: The Basics
 The Basics Why should I get vaccinated? Vaccination is the best way to protect against infections that can make you sick and be passed on to those around you. 1 What kinds of side effects will I get from
The Basics Why should I get vaccinated? Vaccination is the best way to protect against infections that can make you sick and be passed on to those around you. 1 What kinds of side effects will I get from
Human Infl uenza Pandemic. What your organisation needs to do
 Human Infl uenza Pandemic What your organisation needs to do 21 Human Influenza Pandemic: What your organisation needs to do It s time to get ready In 2007 the Victorian Government released the Victorian
Human Infl uenza Pandemic What your organisation needs to do 21 Human Influenza Pandemic: What your organisation needs to do It s time to get ready In 2007 the Victorian Government released the Victorian
Since achieving independence from Great Britain in 1963, Kenya has worked to improve its healthcare system.
 Medical Management Plan Kenya OVERVIEW Company Mission Our mission is to encourage young people to volunteer for worthwhile work in developing countries. We expect that doing this kind of voluntary work
Medical Management Plan Kenya OVERVIEW Company Mission Our mission is to encourage young people to volunteer for worthwhile work in developing countries. We expect that doing this kind of voluntary work
Seven Key Reasons Why immunization must remain a priority in the WHO European Region
 Seven Key Reasons Why immunization must remain a priority in the WHO European Region A dramatic decrease in cases of vaccine-preventable diseases has made many infectious diseases seen as a thing of the
Seven Key Reasons Why immunization must remain a priority in the WHO European Region A dramatic decrease in cases of vaccine-preventable diseases has made many infectious diseases seen as a thing of the
Pentavalent Vaccine. Guide for Health Workers. with Answers to Frequently Asked Questions
 Pentavalent Vaccine Guide for Health Workers with Answers to Frequently Asked Questions Ministry of Health and Family Welfare Government of India 2012 Immunization is one of the most well-known and effective
Pentavalent Vaccine Guide for Health Workers with Answers to Frequently Asked Questions Ministry of Health and Family Welfare Government of India 2012 Immunization is one of the most well-known and effective
2011-2016 Strategic Plan. Creating a healthier world through bold innovation
 2011-2016 Strategic Plan Creating a healthier world through bold innovation 2011-2016 STRATEGIC PLAN Table of contents I. Global direction 1 Mission and vision statements 2 Guiding principles 3 Organizational
2011-2016 Strategic Plan Creating a healthier world through bold innovation 2011-2016 STRATEGIC PLAN Table of contents I. Global direction 1 Mission and vision statements 2 Guiding principles 3 Organizational
Strategic Executive Coaching: An Integrated Approach to Executive Development
 Strategic Executive Coaching: An Integrated Approach to Executive Development A new concept in executive coaching is leading companies to fully utilize the talent they have to drive organizational strategy
Strategic Executive Coaching: An Integrated Approach to Executive Development A new concept in executive coaching is leading companies to fully utilize the talent they have to drive organizational strategy
Policy on Philanthropic Donations and non-commercial Sponsorship
 Policy on Philanthropic Donations and non-commercial Sponsorship 1/10 Content 1. THE ROCHE APPROACH... 3 2. SCOPE... 3 3. GUIDING PRINCIPLES FOR PHILANTHROPIC DONATIONS AND NON-COMMERCIAL SPONSORSHIP...
Policy on Philanthropic Donations and non-commercial Sponsorship 1/10 Content 1. THE ROCHE APPROACH... 3 2. SCOPE... 3 3. GUIDING PRINCIPLES FOR PHILANTHROPIC DONATIONS AND NON-COMMERCIAL SPONSORSHIP...
5/11/2015. Seven Benchmarks of a Successful Worksite Wellness Program. Benchmark #1 Capturing Senior Level Support
 Healthcare costs are an issue of significant concern. Much of the illness in the U.S. is directly preventable. With over 60% of Americans in the workforce, it is an is an ideal setting to address health
Healthcare costs are an issue of significant concern. Much of the illness in the U.S. is directly preventable. With over 60% of Americans in the workforce, it is an is an ideal setting to address health
The. for DUKE MEDICINE. Duke University School of Medicine. People
 The for DUKE MEDICINE Decades of hard work by dedicated physicians and scientists, along with capable and often inspired leadership, have placed the Duke School of Medicine among the nation s best. Now,
The for DUKE MEDICINE Decades of hard work by dedicated physicians and scientists, along with capable and often inspired leadership, have placed the Duke School of Medicine among the nation s best. Now,
Manufacturing CUSTOM CHEMICALS AND SERVICES, SUPPORTING SCIENTIFIC ADVANCES FOR HUMAN HEALTH
 Manufacturing CUSTOM CHEMICALS AND SERVICES, SUPPORTING SCIENTIFIC ADVANCES FOR HUMAN HEALTH VWR enables the advancement of science by providing high-quality chemicals and services, customized to your
Manufacturing CUSTOM CHEMICALS AND SERVICES, SUPPORTING SCIENTIFIC ADVANCES FOR HUMAN HEALTH VWR enables the advancement of science by providing high-quality chemicals and services, customized to your
2 P age. Babies from Birth to Age 2
 Contents Babies from Birth to Age 2... 2 Vaccines give parents the power... 2 Vaccines are recommended throughout our lives... 3 Talk to your doctor... 3 Vaccines are very safe... 3 Whooping Cough (Pertussis)...
Contents Babies from Birth to Age 2... 2 Vaccines give parents the power... 2 Vaccines are recommended throughout our lives... 3 Talk to your doctor... 3 Vaccines are very safe... 3 Whooping Cough (Pertussis)...
THE UNIVERSITY ON NAIROBI STRATEGIC PLAN, 2008-2013
 THE UNIVERSITY ON NAIROBI STRATEGIC PLAN, 2008-2013 INTRODUCTION University of Nairobi is the oldest university in Kenya and has its origins in the Royal Technical College of East Africa (RTCA), which
THE UNIVERSITY ON NAIROBI STRATEGIC PLAN, 2008-2013 INTRODUCTION University of Nairobi is the oldest university in Kenya and has its origins in the Royal Technical College of East Africa (RTCA), which
Multiple Choice Questions
 C hapter 13 WHY DO WE FALL ILL Multiple Choice Questions 1. Which one of the following is not a viral disease? (a) Dengue (b) AIDS (c) Typhoid (d) Influenza 2. Which one of the following is not a bacterial
C hapter 13 WHY DO WE FALL ILL Multiple Choice Questions 1. Which one of the following is not a viral disease? (a) Dengue (b) AIDS (c) Typhoid (d) Influenza 2. Which one of the following is not a bacterial
Frequently asked questions about whooping cough (pertussis)
 Frequently asked questions about whooping cough (pertussis) About whooping cough What is whooping cough? Whooping cough is a highly contagious illness caused by bacteria. It mainly affects the respiratory
Frequently asked questions about whooping cough (pertussis) About whooping cough What is whooping cough? Whooping cough is a highly contagious illness caused by bacteria. It mainly affects the respiratory
Join our scientific talent community
 Join our scientific talent community There has never been a better time to be a part of Janssen Research & Development. We are at the forefront of healthcare leading, evolving and transforming it into
Join our scientific talent community There has never been a better time to be a part of Janssen Research & Development. We are at the forefront of healthcare leading, evolving and transforming it into
An Enterprise Resource Planning Solution (ERP) for Mining Companies Driving Operational Excellence and Sustainable Growth
 SAP for Mining Solutions An Enterprise Resource Planning Solution (ERP) for Mining Companies Driving Operational Excellence and Sustainable Growth 2013 SAP AG or an SAP affi iate company. All rights reserved.
SAP for Mining Solutions An Enterprise Resource Planning Solution (ERP) for Mining Companies Driving Operational Excellence and Sustainable Growth 2013 SAP AG or an SAP affi iate company. All rights reserved.
Uncover the Best Ideas
 Uncover the Best Ideas Table of Contents: overview 2-4 [INNOVATION] overview 5 picture this 6 roi metrics 7-8 [EMPLOYEE NETWORKS] overview 9 picture this 10 roi metrics 11-12 [CITIZEN RELATIONSHIPS] overview
Uncover the Best Ideas Table of Contents: overview 2-4 [INNOVATION] overview 5 picture this 6 roi metrics 7-8 [EMPLOYEE NETWORKS] overview 9 picture this 10 roi metrics 11-12 [CITIZEN RELATIONSHIPS] overview
2 months Diptheria; Tetanus; Whooping Cough; Hib & Polio 1st dose Pneumococcal Conjugate Vaccination
 IMMUNISATIONS You may want to know if your child should have routine immunisations and whether there could be an increased risk of complications because of the heart condition. We have sought the opinions
IMMUNISATIONS You may want to know if your child should have routine immunisations and whether there could be an increased risk of complications because of the heart condition. We have sought the opinions
Vaccinations and Spina Bifida and Hydrocephalus
 shinecharity.org.uk info@shinecharity.org.uk 42 Park Road Peterborough PE1 2UQ 01733 555988 Vaccinations and Spina Bifida and Hydrocephalus Vaccines are made from parts of bacteria or viruses that cause
shinecharity.org.uk info@shinecharity.org.uk 42 Park Road Peterborough PE1 2UQ 01733 555988 Vaccinations and Spina Bifida and Hydrocephalus Vaccines are made from parts of bacteria or viruses that cause
Solving for the Future: Addressing Major Societal Challenges Through Innovative Technology and Cloud Computing
 Solving for the Future: Addressing Major Societal Challenges Through Innovative Technology and Cloud Computing As economic challenges persist in communities, nations, and regions around the world, the
Solving for the Future: Addressing Major Societal Challenges Through Innovative Technology and Cloud Computing As economic challenges persist in communities, nations, and regions around the world, the
Up to $402,000. Insight HIV. Drug Class. 1.2 million people in the United States were living with HIV at the end of 2011 (most recent data).
 HIV Background, new developments, key strategies Drug Class Insight INTRODUCTION Human Immunodeficiency Virus (HIV) is the virus that can lead to Acquired Immunodeficiency Syndrome, or AIDS. No safe and
HIV Background, new developments, key strategies Drug Class Insight INTRODUCTION Human Immunodeficiency Virus (HIV) is the virus that can lead to Acquired Immunodeficiency Syndrome, or AIDS. No safe and
SIXTY-SEVENTH WORLD HEALTH ASSEMBLY. Agenda item 12.3 24 May 2014. Hepatitis
 SIXTY-SEVENTH WORLD HEALTH ASSEMBLY WHA67.6 Agenda item 12.3 24 May 2014 Hepatitis The Sixty-seventh World Health Assembly, Having considered the report on hepatitis; 1 Reaffirming resolution WHA63.18,
SIXTY-SEVENTH WORLD HEALTH ASSEMBLY WHA67.6 Agenda item 12.3 24 May 2014 Hepatitis The Sixty-seventh World Health Assembly, Having considered the report on hepatitis; 1 Reaffirming resolution WHA63.18,
Frequently Asked Questions (FAQs)
 Frequently Asked Questions (FAQs) Research Rationale 1. What does PrEP stand for? There is scientific evidence that antiretroviral (anti-hiv) medications may be able to play an important role in reducing
Frequently Asked Questions (FAQs) Research Rationale 1. What does PrEP stand for? There is scientific evidence that antiretroviral (anti-hiv) medications may be able to play an important role in reducing
GENERAL INFORMATION. Hepatitis B Foundation - Korean Chapter Pg. 3 www.hepb.org
 GENERAL INFORMATION What is hepatitis B? Hepatitis B is the world's most common liver infection that can lead to cirrhosis and liver cancer. It is caused by the hepatitis B virus (HBV), which attacks and
GENERAL INFORMATION What is hepatitis B? Hepatitis B is the world's most common liver infection that can lead to cirrhosis and liver cancer. It is caused by the hepatitis B virus (HBV), which attacks and
Crucell 2007 Combating infectious diseases
 Crucell 2007 Combating infectious diseases Annual Report and Form 20-F www.crucell.com Mission: Combating infectious diseases Crucell s mission is to develop, produce and market vaccines and antibodies
Crucell 2007 Combating infectious diseases Annual Report and Form 20-F www.crucell.com Mission: Combating infectious diseases Crucell s mission is to develop, produce and market vaccines and antibodies
The Challenge of Whooping Cough:
 The Challenge of Whooping Cough: Canada s Role in the Development of Pertussis Vaccines AUTHORS: Luis Barreto, M.B.B.S., M.D., M.H.Sc. Rob Van Exan, Ph.D. Christopher Rutty, Ph.D. CREATED THROUGH AN EDUCATIONAL
The Challenge of Whooping Cough: Canada s Role in the Development of Pertussis Vaccines AUTHORS: Luis Barreto, M.B.B.S., M.D., M.H.Sc. Rob Van Exan, Ph.D. Christopher Rutty, Ph.D. CREATED THROUGH AN EDUCATIONAL
2015 Annual Convention
 2015 Annual Convention Date: Monday, October 12, 2015 Time: 8:00 am 9:30 am Location: Gaylord National Harbor Resort and Convention Center, National Harbor 10 Title: Activity Type: Speaker: Expanding Immunization
2015 Annual Convention Date: Monday, October 12, 2015 Time: 8:00 am 9:30 am Location: Gaylord National Harbor Resort and Convention Center, National Harbor 10 Title: Activity Type: Speaker: Expanding Immunization
Open Innovation: An Imperative for the Pharmaceutical Industry. Berkeley Innovation Forum
 Open Innovation: An Imperative for the Pharmaceutical Industry Berkeley Innovation Forum October 28-29, 2010 Minneapolis, MN Ted Torphy, Ph.D. Vice President & Head External Innovation Research Capabilities
Open Innovation: An Imperative for the Pharmaceutical Industry Berkeley Innovation Forum October 28-29, 2010 Minneapolis, MN Ted Torphy, Ph.D. Vice President & Head External Innovation Research Capabilities
Health, history and hard choices: Funding dilemmas in a fast-changing world
 Health, history and hard choices: Funding dilemmas in a fast-changing world Thomson Prentice Global Health Histories Health and Philanthropy: Leveraging change University of Indiana, August 2006 An embarrassment
Health, history and hard choices: Funding dilemmas in a fast-changing world Thomson Prentice Global Health Histories Health and Philanthropy: Leveraging change University of Indiana, August 2006 An embarrassment
cambodia Maternal, Newborn AND Child Health and Nutrition
 cambodia Maternal, Newborn AND Child Health and Nutrition situation Between 2000 and 2010, Cambodia has made significant progress in improving the health of its children. The infant mortality rate has
cambodia Maternal, Newborn AND Child Health and Nutrition situation Between 2000 and 2010, Cambodia has made significant progress in improving the health of its children. The infant mortality rate has
Our communities. We engage our people to meaningfully contribute to our communities.
 Our communities At Li & Fung we are committed to creating positive impact where we live and work and aligning our community initiatives with our supply chain activities. We focus on investing in the potential
Our communities At Li & Fung we are committed to creating positive impact where we live and work and aligning our community initiatives with our supply chain activities. We focus on investing in the potential
UNIVERSITY OF MIAMI SCHOOL OF BUSINESS ADMINISTRATION MISSION, VISION & STRATEGIC PRIORITIES. Approved by SBA General Faculty (April 2012)
 UNIVERSITY OF MIAMI SCHOOL OF BUSINESS ADMINISTRATION MISSION, VISION & STRATEGIC PRIORITIES Approved by SBA General Faculty (April 2012) Introduction In 1926, we embarked on a noble experiment the creation
UNIVERSITY OF MIAMI SCHOOL OF BUSINESS ADMINISTRATION MISSION, VISION & STRATEGIC PRIORITIES Approved by SBA General Faculty (April 2012) Introduction In 1926, we embarked on a noble experiment the creation
Building on Success: A National Strategy to Save Lives
 Remarks by Howard K. Koh, MD, MPH Assistant Secretary for Health, U.S. Department of Health and Human Services XIX International AIDS Conference Washington, DC Plenary Session Theme: Challenges and Solutions
Remarks by Howard K. Koh, MD, MPH Assistant Secretary for Health, U.S. Department of Health and Human Services XIX International AIDS Conference Washington, DC Plenary Session Theme: Challenges and Solutions
career in the Production
 The Syngenta Production & Supply Graduate Program Building your career in the Production & Supply organization while helping to meet the rising demand for food, feed and fuel * Our work matters The Syngenta
The Syngenta Production & Supply Graduate Program Building your career in the Production & Supply organization while helping to meet the rising demand for food, feed and fuel * Our work matters The Syngenta
NO MORE MISSED MDG4 OPPORTUNITIES: OPTIMIZING EXISTING HEALTH PLATFORMS FOR CHILD SURVIVAL. Polio Campaigns
 NO MORE MISSED MDG4 OPPORTUNITIES: OPTIMIZING EXISTING HEALTH PLATFORMS FOR CHILD SURVIVAL Polio Campaigns With fewer than 600 days remaining to the Millennium Development Goal (MDG) deadline, new strategies
NO MORE MISSED MDG4 OPPORTUNITIES: OPTIMIZING EXISTING HEALTH PLATFORMS FOR CHILD SURVIVAL Polio Campaigns With fewer than 600 days remaining to the Millennium Development Goal (MDG) deadline, new strategies
DRAFT. CUNY Pandemic Influenza Response Plan Incident Level Responsibilities
 CUNY Incident Criteria Situation Characteristics No current hazard to students, faculty and staff. Requires internal CUNY preparedness, but no outside agency assistance. Human infections with a new subtype,
CUNY Incident Criteria Situation Characteristics No current hazard to students, faculty and staff. Requires internal CUNY preparedness, but no outside agency assistance. Human infections with a new subtype,
Business model innovation as a response to increased competition in the energy sector
 ARTICLE BASED ON INTERVIEWS AND DEVELOPMENT PROJECTS WITH DONG ENERGY Business model innovation as a response to increased competition in the energy sector By Thomas Børve and Johan Lawaetz Increased competition
ARTICLE BASED ON INTERVIEWS AND DEVELOPMENT PROJECTS WITH DONG ENERGY Business model innovation as a response to increased competition in the energy sector By Thomas Børve and Johan Lawaetz Increased competition
Parent s Guide to CHILDHOOD IMMUNIZATIONS
 Part One Vaccine-Preventable Part Three More Diseases About Vaccines and Childhood Vaccines 30 Parent s Guide to CHILDHOOD IMMUNIZATIONS Part 3: More About Vaccines How do vaccines work? To understand
Part One Vaccine-Preventable Part Three More Diseases About Vaccines and Childhood Vaccines 30 Parent s Guide to CHILDHOOD IMMUNIZATIONS Part 3: More About Vaccines How do vaccines work? To understand
Link Sustainability to Corporate Strategy Using the Balanced Scorecard
 Link Sustainability to Corporate Strategy Using the Balanced Scorecard People and their managers are working so hard to be sure things are done right, that they hardly have time to decide if they are doing
Link Sustainability to Corporate Strategy Using the Balanced Scorecard People and their managers are working so hard to be sure things are done right, that they hardly have time to decide if they are doing
HUMAN SERVICES MANAGEMENT COMPETENCIES
 HUMAN SERVICES MANAGEMENT COMPETENCIES A Guide for Non-Profit and For Profit Agencies, Foundations and Academic Institutions Prepared by: Anthony Hassan, MSW, Ed.D. William Waldman, MSW Shelly Wimpfheimer,
HUMAN SERVICES MANAGEMENT COMPETENCIES A Guide for Non-Profit and For Profit Agencies, Foundations and Academic Institutions Prepared by: Anthony Hassan, MSW, Ed.D. William Waldman, MSW Shelly Wimpfheimer,
IKEA case study SWOT analysis and sustainable business planning
 IKEA case study SWOT analysis and sustainable business planning Page 1: Introduction IKEA is an internationally known home furnishing retailer. It has grown rapidly since it was founded in 1943. Today
IKEA case study SWOT analysis and sustainable business planning Page 1: Introduction IKEA is an internationally known home furnishing retailer. It has grown rapidly since it was founded in 1943. Today
Procurement must be more than just cost cutting
 Procurement must be more than just cost cutting In today s challenging environment, aggressive cost control has become a common theme in the pharmaceutical industry. It became fashionable later than in
Procurement must be more than just cost cutting In today s challenging environment, aggressive cost control has become a common theme in the pharmaceutical industry. It became fashionable later than in
The NIH Roadmap: Re-Engineering the Clinical Research Enterprise
 NIH BACKGROUNDER National Institutes of Health The NIH Roadmap: Re-Engineering the Clinical Research Enterprise Clinical research is the linchpin of the nation s biomedical research enterprise. Before
NIH BACKGROUNDER National Institutes of Health The NIH Roadmap: Re-Engineering the Clinical Research Enterprise Clinical research is the linchpin of the nation s biomedical research enterprise. Before
Influenza Vaccine Frequently Asked Questions. Influenza Control Program
 Influenza Vaccine Frequently Asked Questions Influenza Control Program Influenza or the flu can be a serious contagious disease, which is spread by droplet transmission through close contact with an infected
Influenza Vaccine Frequently Asked Questions Influenza Control Program Influenza or the flu can be a serious contagious disease, which is spread by droplet transmission through close contact with an infected
_experience the commitment TM. We deliver. responsibly
 _experience the commitment TM We deliver responsibly Corporate social responsibility roadmap 2011 2 Corporate Social Responsibility Our dream To create an environment in which we enjoy working together
_experience the commitment TM We deliver responsibly Corporate social responsibility roadmap 2011 2 Corporate Social Responsibility Our dream To create an environment in which we enjoy working together
Chapter 3. Immunity and how vaccines work
 Chapter 3 Immunity and how vaccines work 3.1 Objectives: To understand and describe the immune system and how vaccines produce immunity To understand the differences between Passive and Active immunity
Chapter 3 Immunity and how vaccines work 3.1 Objectives: To understand and describe the immune system and how vaccines produce immunity To understand the differences between Passive and Active immunity
Medical Management Plan Togo
 Medical Management Plan Togo OVERVIEW Company Mission Our mission is to encourage young people to volunteer for worthwhile work in developing countries. We expect that doing this kind of voluntary work
Medical Management Plan Togo OVERVIEW Company Mission Our mission is to encourage young people to volunteer for worthwhile work in developing countries. We expect that doing this kind of voluntary work
What is whooping cough. (pertussis)? Information and Prevention. Ocument dn
 What is whooping cough Ocument dn (pertussis)? Information and Prevention IMPORTANT Pertussis (or whooping cough) is a highly contagious infection that can cause uncontrollable, violent coughing. If you
What is whooping cough Ocument dn (pertussis)? Information and Prevention IMPORTANT Pertussis (or whooping cough) is a highly contagious infection that can cause uncontrollable, violent coughing. If you
Immunization Infrastructure: The Role of Section 317
 Immunization Infrastructure: The Role of Section 317 Immunization plays a vital role in the control and prevention of infectious disease. Current immunization recommendations target 17 vaccine-preventable
Immunization Infrastructure: The Role of Section 317 Immunization plays a vital role in the control and prevention of infectious disease. Current immunization recommendations target 17 vaccine-preventable
Information on Measles and Whooping Cough: Vaccination and Disease
 Information on Measles and Whooping Cough: Vaccination and Disease Vaccine s Mechanism of Action Vaccines expose the recipient to a small amount of an weakened organism. Through this exposure, the body
Information on Measles and Whooping Cough: Vaccination and Disease Vaccine s Mechanism of Action Vaccines expose the recipient to a small amount of an weakened organism. Through this exposure, the body
Case Study. Reduction Story. Clay Nesler Vice President, Global Energy & Sustainability. Steve Thomas Manager, Energy & Sustainability Communications
 Case Study The Johnson Controls Greenhouse Gas Reduction Story Clay Nesler Vice President, Global Energy & Sustainability Steve Thomas Manager, Energy & Sustainability Communications August 2010 Pressures
Case Study The Johnson Controls Greenhouse Gas Reduction Story Clay Nesler Vice President, Global Energy & Sustainability Steve Thomas Manager, Energy & Sustainability Communications August 2010 Pressures
The EU role in global health QUESTIONNAIRE: Question 1 Question 2 Question 3: Question 4: Question 5:
 The EU role in global health QUESTIONNAIRE: Question 1: In your opinion, does the proposed concept global health cover the most relevant dimensions? If not, which other essential factors would you suggest?
The EU role in global health QUESTIONNAIRE: Question 1: In your opinion, does the proposed concept global health cover the most relevant dimensions? If not, which other essential factors would you suggest?
Targeting Cancer: Innovation in the Treatment of Chronic Myelogenous Leukemia EXECUTIVE SUMMARY. New England Healthcare Institute
 Targeting Cancer: Innovation in the Treatment of Chronic Myelogenous Leukemia New England Healthcare Institute NEHI Innovation Series March 2004 Executive Summary From drugs and medical devices, to information
Targeting Cancer: Innovation in the Treatment of Chronic Myelogenous Leukemia New England Healthcare Institute NEHI Innovation Series March 2004 Executive Summary From drugs and medical devices, to information
MSD Information Technology Global Innovation Center. Digitization and Health Information Transparency
 MSD Information Technology Global Innovation Center Digitization and Health Information Transparency 10 February 2014 1 We are seeking energetic, forward thinking graduates to join our Information Technology
MSD Information Technology Global Innovation Center Digitization and Health Information Transparency 10 February 2014 1 We are seeking energetic, forward thinking graduates to join our Information Technology
Renault-Nissan CSR Guidelines for Suppliers
 Renault-Nissan CSR Guidelines for Suppliers May, 2010 Renault S.A.S. Nissan Motor Co., Ltd. Contents Introduction Highlights 1. Renault-Nissan Purchasing Way 2. CSR Guidelines for Suppliers (A) Overview
Renault-Nissan CSR Guidelines for Suppliers May, 2010 Renault S.A.S. Nissan Motor Co., Ltd. Contents Introduction Highlights 1. Renault-Nissan Purchasing Way 2. CSR Guidelines for Suppliers (A) Overview
Editorial. Yves Levy, Executive Director
 Editorial Yves Levy, Executive Director I am delighted to announce the creation of a new research centre: the Vaccine Research Institute (VRI). The VRI addresses the unprecedented challenge of developing
Editorial Yves Levy, Executive Director I am delighted to announce the creation of a new research centre: the Vaccine Research Institute (VRI). The VRI addresses the unprecedented challenge of developing
It takes a planet: global implications/consequences of local vaccine refusal
 It takes a planet: global implications/consequences of local vaccine refusal Meg Fisher, MD Medical Director Disclosures I have no disclosures. I may be mentioning off label uses of vaccines. Objectives/Outcomes
It takes a planet: global implications/consequences of local vaccine refusal Meg Fisher, MD Medical Director Disclosures I have no disclosures. I may be mentioning off label uses of vaccines. Objectives/Outcomes
UCSF Clinical Enterprise Strategic Plan 2014-2019
 UCSF HEALTH SYSTEM UCSF Clinical Enterprise Strategic Plan 2014-2019 Building the Health System 1 UCSF Health System Five Year Strategic Plan: FY 2014-2019 Advancing the Health System A revolution is underway
UCSF HEALTH SYSTEM UCSF Clinical Enterprise Strategic Plan 2014-2019 Building the Health System 1 UCSF Health System Five Year Strategic Plan: FY 2014-2019 Advancing the Health System A revolution is underway
PARTICIPATORY ANALYSIS
 PARTICIPATORY ANALYSIS Expanding Stakeholder Involvement in Evaluation Veena Pankaj Myia Welsh Laura Ostenso April 2011 2011 Innovation Network, Inc. You may remix, tweak, and build upon this work for
PARTICIPATORY ANALYSIS Expanding Stakeholder Involvement in Evaluation Veena Pankaj Myia Welsh Laura Ostenso April 2011 2011 Innovation Network, Inc. You may remix, tweak, and build upon this work for
Immunity and how vaccines work
 1 Introduction Immunity is the ability of the human body to protect itself from infectious disease. The defence mechanisms of the body are complex and include innate (non-specific, non-adaptive) mechanisms
1 Introduction Immunity is the ability of the human body to protect itself from infectious disease. The defence mechanisms of the body are complex and include innate (non-specific, non-adaptive) mechanisms
Health in the 2030 Agenda for Sustainable Development
 EXECUTIVE BOARD EB138/CONF./8 138th session 27 January 2016 Agenda item 7.2 Health in the 2030 Agenda for Sustainable Development Draft resolution proposed by Japan, Panama, South Africa, Thailand, United
EXECUTIVE BOARD EB138/CONF./8 138th session 27 January 2016 Agenda item 7.2 Health in the 2030 Agenda for Sustainable Development Draft resolution proposed by Japan, Panama, South Africa, Thailand, United
Key Facts about Influenza (Flu) & Flu Vaccine
 Key Facts about Influenza (Flu) & Flu Vaccine mouths or noses of people who are nearby. Less often, a person might also get flu by touching a surface or object that has flu virus on it and then touching
Key Facts about Influenza (Flu) & Flu Vaccine mouths or noses of people who are nearby. Less often, a person might also get flu by touching a surface or object that has flu virus on it and then touching
Viral hepatitis. Report by the Secretariat
 SIXTY-THIRD WORLD HEALTH ASSEMBLY A63/15 Provisional agenda item 11.12 25 March 2010 Viral hepatitis Report by the Secretariat THE DISEASES AND BURDEN 1. The group of viruses (hepatitis A, B, C, D and
SIXTY-THIRD WORLD HEALTH ASSEMBLY A63/15 Provisional agenda item 11.12 25 March 2010 Viral hepatitis Report by the Secretariat THE DISEASES AND BURDEN 1. The group of viruses (hepatitis A, B, C, D and
SCP Issues for Business and Industry
 SCP Issues for Business and Industry Introduction Business and industry are key players in the SCP agenda. They are at the core of production and are also key organizational consumers. As the most important
SCP Issues for Business and Industry Introduction Business and industry are key players in the SCP agenda. They are at the core of production and are also key organizational consumers. As the most important
NEA Leadership Competencies Guide
 NEA Leadership Competencies Guide NEA Leadership Competencies Guide Table of Contents NEA Vision, Mission, and Values 5 Setting the context for leadership competencies 7 Leadership competency framework
NEA Leadership Competencies Guide NEA Leadership Competencies Guide Table of Contents NEA Vision, Mission, and Values 5 Setting the context for leadership competencies 7 Leadership competency framework
Recruitment and Selection
 Recruitment and Selection The recruitment and selection belongs to value added HR Processes. The recruitment is about: the ability of the organization to source new employees, to keep the organization
Recruitment and Selection The recruitment and selection belongs to value added HR Processes. The recruitment is about: the ability of the organization to source new employees, to keep the organization
Valneva Reports Preliminary FY 2013 Revenues of EUR 36.0m and Cash Position of EUR 40.2m at End of 2013
 Valneva Reports Preliminary FY 2013 Revenues of EUR 36.0m and Cash Position of EUR 40.2m at End of 2013 Revenue Ahead of Guidance and Cash Target Met in First year Following Merger Total revenues and grants
Valneva Reports Preliminary FY 2013 Revenues of EUR 36.0m and Cash Position of EUR 40.2m at End of 2013 Revenue Ahead of Guidance and Cash Target Met in First year Following Merger Total revenues and grants
Vaccination against pertussis (Whooping cough) for pregnant women- 2014. Information for healthcare professionals
 Vaccination against pertussis (Whooping cough) for pregnant women- 2014 Information for healthcare professionals About Public Health England Public Health England s mission is to protect and improve the
Vaccination against pertussis (Whooping cough) for pregnant women- 2014 Information for healthcare professionals About Public Health England Public Health England s mission is to protect and improve the
Early Lessons learned from strong revenue cycle performers
 Healthcare Informatics June 2012 Accountable Care Organizations Early Lessons learned from strong revenue cycle performers Healthcare Informatics Accountable Care Organizations Early Lessons learned from
Healthcare Informatics June 2012 Accountable Care Organizations Early Lessons learned from strong revenue cycle performers Healthcare Informatics Accountable Care Organizations Early Lessons learned from
Polio and the Introduction of IPV
 Polio and the Introduction of IPV Poliomyelitis (polio) is a highly infectious disease that is caused when a person is infected by the polio virus that invades the nervous system. Poliomyelitis can cause
Polio and the Introduction of IPV Poliomyelitis (polio) is a highly infectious disease that is caused when a person is infected by the polio virus that invades the nervous system. Poliomyelitis can cause
THE L ORÉAL SUSTAINABILITY COMMITMENT
 THE L ORÉAL SUSTAINABILITY COMMITMENT OUR SHARING BEAUTY WITH ALL COMMITMENT TOGETHER WE WILL MAKE BEAUTY SUSTAINABLE. TOGETHER WE WILL MAKE SUSTAINABILITY BEAUTIFUL. We want to bring beauty to all people.
THE L ORÉAL SUSTAINABILITY COMMITMENT OUR SHARING BEAUTY WITH ALL COMMITMENT TOGETHER WE WILL MAKE BEAUTY SUSTAINABLE. TOGETHER WE WILL MAKE SUSTAINABILITY BEAUTIFUL. We want to bring beauty to all people.
Introduction. 1. General outline. 1) Position of the Healthcare Policy. 2) Basic principles of the Healthcare Policy (Article 2 of the Promotion Act)
 Overview of the Healthcare Policy Introduction For Japan, which is becoming an ultra-aging society ahead of other countries, it is important to realize the world s most advanced medical technologies and
Overview of the Healthcare Policy Introduction For Japan, which is becoming an ultra-aging society ahead of other countries, it is important to realize the world s most advanced medical technologies and
Attribute 1: COMMUNICATION
 The positive are intended for use as a guide only and are not exhaustive. Not ALL will be applicable to ALL roles within a grade and in some cases may be appropriate to a Attribute 1: COMMUNICATION Level
The positive are intended for use as a guide only and are not exhaustive. Not ALL will be applicable to ALL roles within a grade and in some cases may be appropriate to a Attribute 1: COMMUNICATION Level
HEALTH SYSTEM. Introduction. The. jurisdictions and we. Health Protection. Health Improvement. Health Services. Academic Public
 FUNCTIONS OF THE LOCAL PUBLIC HEALTH SYSTEM Introduction This document sets out the local PH function in England. It was originally drafted by a working group led by Maggie Rae, FPH Local Board Member
FUNCTIONS OF THE LOCAL PUBLIC HEALTH SYSTEM Introduction This document sets out the local PH function in England. It was originally drafted by a working group led by Maggie Rae, FPH Local Board Member
Max Foundation plan 2014-2016
 Max Foundation plan 2014-2016 Page1 Content 1. Introduction 3 2. Goal 4 3. Our values 5 4. How we work 6 5. Max-WASH approach 7 6. Were we work 9 7. Ambition 10 Page2 1. Introduction Max Foundation prevents
Max Foundation plan 2014-2016 Page1 Content 1. Introduction 3 2. Goal 4 3. Our values 5 4. How we work 6 5. Max-WASH approach 7 6. Were we work 9 7. Ambition 10 Page2 1. Introduction Max Foundation prevents
Helene D. Gayle, President and CEO, CARE USA
 The Atlanta Declaration U.S. Leadership in Improving the World s Health 21 May 2012 It is timely and important that we celebrate America s global health successes, and that we continue to lead investing
The Atlanta Declaration U.S. Leadership in Improving the World s Health 21 May 2012 It is timely and important that we celebrate America s global health successes, and that we continue to lead investing
Best Practices in Strategic Planning
 Best Practices in Strategic Planning Charles Aubrey V.P. Performance Excellence AndersonBrecon An AmerisourceBergen Company 1 Foundation Where do We Begin? 2 HOSHIN KANRI Hoshin Kanri Policy Deployment
Best Practices in Strategic Planning Charles Aubrey V.P. Performance Excellence AndersonBrecon An AmerisourceBergen Company 1 Foundation Where do We Begin? 2 HOSHIN KANRI Hoshin Kanri Policy Deployment
2019 Healthcare That Works for All
 2019 Healthcare That Works for All This paper is one of a series describing what a decade of successful change in healthcare could look like in 2019. Each paper focuses on one aspect of healthcare. To
2019 Healthcare That Works for All This paper is one of a series describing what a decade of successful change in healthcare could look like in 2019. Each paper focuses on one aspect of healthcare. To
INVESTORS IN PEOPLE REVIEW REPORT
 INVESTORS IN PEOPLE REVIEW REPORT Lower Farm Primary School Page: 1 of 13 CONTENTS Key Information 3 Assessor Decision 3 Milestone Dates 3 Introduction 4 Assessment Objectives 4 Feedback Against the Assessment
INVESTORS IN PEOPLE REVIEW REPORT Lower Farm Primary School Page: 1 of 13 CONTENTS Key Information 3 Assessor Decision 3 Milestone Dates 3 Introduction 4 Assessment Objectives 4 Feedback Against the Assessment
SYRACUSE UNIVERSITY: ACADEMIC STRATEGIC PLAN
 SYRACUSE UNIVERSITY: ACADEMIC STRATEGIC PLAN These are extraordinary times for higher education and for students aspiring to succeed in a rapidly changing world. Technology is affecting the way we learn
SYRACUSE UNIVERSITY: ACADEMIC STRATEGIC PLAN These are extraordinary times for higher education and for students aspiring to succeed in a rapidly changing world. Technology is affecting the way we learn
School of Project Management
 School of Project Management For Your Business Success Master Certifications in Project Management from Managing Projects Sponsor s Note Dear Colleague, Project management - the skills, knowledge and ability
School of Project Management For Your Business Success Master Certifications in Project Management from Managing Projects Sponsor s Note Dear Colleague, Project management - the skills, knowledge and ability
