World class academic leadership for your clinical trials
|
|
|
- Bruce Spencer
- 7 years ago
- Views:
Transcription
1 World class academic leadership for your clinical trials
2 Clinical Sites 1-45 Countries >150,000 Patients Enrolled
3 What is C5Research? C5Research is an academic research organization (ARO) which provides clinical research services to the biotechnology, medical device and pharmaceutical industries, as well as providing support for Cleveland Clinic investigator-initiated research trials. We bring clinical and academic expertise to research. We strive to promote innovation and to impact the clinical care of patients through the dissemination of trial results in top-tier journals, at scientific meetings and through education in our premier teaching hospital. We provide comprehensive, academically oriented planning, coordination, and management of multicenter clinical trials. We provide independent oversight of clinical trial committees (Executive, Steering and Data Monitoring). We facilitate efficient start-up and operation of clinical trials with the goal of publishing results that will have a major impact on the medical care of patients. Our Mission: To design and conduct innovative clinical research that enhances scientific knowledge about the treatment of diseases and improves medical care of patients.
4 What is C5Research to Sponsors? We are a non-profit organization dedicated to helping sponsors successfully collect relevant efficacy and safety data within agreed upon timelines, with the highest standards, complying with FDA regulations, and satisfying the demanding peer-reviewed scrutiny of the academic and scientific community. We bring world class academic leadership to clinical trials and customize our services to meet trial needs. Our Expertise For over a decade, C5Research has combined scientific and clinical leadership in cardiovascular disease with operational excellence in Phase II-IV multicenter, clinical trial coordination. This strong foundation in cardiovascular trial coordination has been expanded to include other therapeutic areas, capitalizing on the academic, scientific and medical expertise of Cleveland Clinic. When choosing C5Research, a sponsor can expect independent, accurate and detailed investigations that are respected in the academic and scientific communities, will strongly influence the care of patients, and will provide credibility and shorter time-to-market in the product development cycle. Detailed trial descriptions can be found on our website at:
5 Why Work With C5Research? We have expertise in a variety of therapeutic areas that is recognized worldwide. Cleveland Clinic Heart and Vascular Institute has been ranked the #1 program for cardiac care in the US for over a decade by US News and World Report, and Cleveland Clinic ranks among the top 5 hospitals overall. We have access to the expertise of leading scientists and physicians in all branches of medicine within Cleveland Clinic. We have provided scientific leadership in the conduct of clinical trials from small, single-center Phase II trials to large, global, multi-center Phases III and IV trials. Our physicians have served on FDA regulatory panels and thus have insight into agency thinking. We disseminate results quickly at national and international meetings and in top-tier journals. Complexity of Clinical trials A clinical trial requires expertise in the areas of Project Management, Data Management, Site Management, Core Laboratories, Regulatory / Safety, Randomization, Statistics, and Drug/ Device Supply. We have worked with a broad spectrum of different research organizations, including Contract Research Organizations (CROs) and other AROs, in the various aspects of trial conduct. We coordinate with all parties involved in a trial to minimize overlap and duplication of effort by determining who is best suited for each task. We believe partnering, shared responsibility and communication are the keys to success.
6 C5Research Physician Leadership A. Michael Lincoff, MD, Director of the Center for Clinical Research and Vice Chairman for Clinical Research of the Lerner Research Institute is also the C5Research Director and provides clinical trial leadership. In addition, Stephen J. Nicholls, MD, PhD, is Cardiovascular Director of C5Research and provides clinical trial leadership. There are seven Associate Directors, physicians with expertise in cardiovascular sub-specialties, additional Core Lab Directors and access to over 500 Cleveland Clinic physician experts in all areas of medicine, located on the campus of Cleveland Clinic. Steven E. Nissen, MD, is Chairman of the Robert and Suzanne Tomsich Department of Cardiovascular Medicine at Cleveland Clinic and former C5Research Director. Overview We are an academic group led by Marlene Goormastic, MPH, C5Research Director of Operations, that believes in doing the highest quality work following International Conference on Harmonization/Good Clinical Practice guidelines. We bring an independent, academic viewpoint that provides study credibility for the academic and scientific community. We provide world class academic leadership for your clinical trials by having a Cleveland Clinic physician lead the trial as the head of the Steering or Executive Committee. The academic lead is a physician who is both experienced in the conduct of clinical trials and who has expertise in the disease area under investigation. Our ultimate objective is to bridge our basic and translational scientific expertise to bring new therapies from the laboratory bench to the bedside. Many other physicians within the Cleveland Clinic collaborate and support C5Research The C5Research team works closely with the trial academic leadership to develop and finalize a study protocol that is both feasible and answers an important medical question. We assist with site selection and management. We organize and manage the clinical trial committees (Executive, Steering and Data Monitoring). The C5Research academic lead contributes his/ her expertise in meetings with the FDA, peer-topeer interactions at investigator meetings and presentation of trial results at major medical conferences. The benefit in working with an Academic Research Organization (ARO) cannot be overemphasized in an era of public and government wariness and increased scrutiny in trial conduct and results. The top journals require the authors to provide a statement of independent data access and validation of trial results. As an ARO, we require the complete trial dataset after database lock, and we validate the primary and secondary results before publication.
7 We have Core Labs that provide high quality and timely imaging analysis. We have an adjudication process that provides highly reliable and timely adjudication of clinical events. We collaborate with the Sponsor, often working with a Contract Research Organization (CRO) or other AROs to assure that all services required are done in the most efficient manner by the organization best equipped to provide that service. We facilitate efficient start-up and operation of clinical trials with the goal of publishing results in top tier medical journal that will have a major impact on the practice of medicine. Partnerships C5Research has collaborated with over 60 device and pharmaceutical companies during the past two decades, as diverse as small start-ups to Fortune 500 corporations. We develop a customized model of operational and communication requirements with each partnership. Often these models include CRO and/or ARO partners. This unique blend of C5Research, the sponsor, CRO and/or ARO provides an effective and efficient means to manage clinical studies and meet pre-specified timelines. Sponsors may find additional information on our website at:
8 C5Research Services Project and Site Management Core Laboratories Clinical Data Management Statistics Contracts and Finance Regulatory and Quality Assurance Project and Site Management Project leadership and coordination Physician, project manager and research coordinator expertise to enhance protocol development, implementation and trial conduct Site database and site selection Clinical resources for study sites including 24 hour clinical on-call service CRO / Sponsor liaison Site and Monitor education Enrollment and Communication expertise Steering Committee and Data Safety Monitoring Committee selection / management
9 Clinical Events Committee (CEC) The Clinical Events Committee Core Lab provides blinded, unbiased, expert adjudication of endpoint events. Endpoints adjudicated include Death Myocardial infarction Stroke Revascularization Bleeding (TIMI, GUSTO, CURE, and other definitions) Hospitalizations for acute coronary syndrome Hospitalizations for heart failure Peripheral vascular events Gastroenterological events
10 Imaging Core Laboratories Our imaging core laboratories provide unbiased interpretation of imaging examinations for device and pharmacologic clinical studies while decreasing inter-observer variability and increasing accuracy and precision. We establish and oversee compliance of imaging protocols and guidelines and interact with sites on an ongoing basis to maintain quality of images. We provide standardized image acquisition training to investigators to ensure consistent high quality imaging across all enrolling centers. In addition, our analysis processes incorporate quality control and assurance methods throughout the course of each study.
11 Our imaging core laboratories provide unbiased interpretation of imaging examinations for device and pharmacologic clinical studies while decreasing inter-observer variability and increasing accuracy and precision
12 Atherosclerosis Imaging Core Laboratory Angiography More than 20,000 angiographic films analyzed in over 50 clinical studies International experience in the US, Canada, Europe, South America and Asia Trial experience includes CAVEAT, TAMI-9, EPIC, EPISTENT, APEX-AMI, CATSCAN and IMPACT-II to name a few. Analysis provided for clinical trials in: Thrombolytic therapies Percutaneous Intervention (PCI) New coronary devices Interventions with concomitant drug therapy Progression / Regression LV function Angiographic assessments include: Quantitative Coronary and Vascular Angiography (QCA & QVA) TIMI grade TIMI frame count (ctfc) Myocardial Perfusion / blush (TMP) AHA/ACC lesion morphology assessments Left-ventricular ejection fraction (LVEF) Thrombus assessments
13 Intravascular Ultrasound (IVUS) More than 12,000 ultrasound imaging exams have been completed in over 20 clinical studies. International experience in the US, Canada, Europe, South America and Australia Trial experience includes REVERSAL, NORMALISE, ACTIVATE, ASTEROID, Apo A1 Milano, ILLUSTRATE and PERISCOPE. Analysis provided for clinical trials in: Atheroma Progression/Regression Coronary and Vascular Percutaneous Intervention (PCI) Transplant Vasculopathy IVUS assessments include: Volumetric analysis Restenosis analysis Plaque characterization assessment Carotid Intimal Medial Thickness (CIMT) Core Laboratory Initiated in 2006, the CIMT Core Lab has completed several projects ranging from 20 to more than 200 subjects. Trial experience includes PPAR and several internal studies. The CIMT Core Lab measures the thickness of the two inner layers of the carotid arteries with non-invasive ultrasound to predict risk of cardiovascular events.
14 Cardiovascular Imaging Core Laboratory Echocardiography Initiated in 1996, the Echo Core Lab has completed over 40 clinical studies and analyzed more than 20,000 echocardiograms. International experience in the US, Canada, Europe, Central America and Asia Trial experience includes VECTOR, RETHINQ, PREMIER, Perimount FenPhen, Fen-Phen and the ACUTE studies. Analysis provided for clinical trials in: Electrophysiology devices Heart failure/heart transplantation Intra-operative trials involving ventricular assist devices Aortic atheroma Prosthetic valves Valve repair/replacement Neurology, oncology and hypertension studies Echo assessments include: Left ventricular mass and function Wall motion scoring and analysis Transpulmonary echo contrast grading and analysis Atrial Fibrillation
15 Nuclear Cardiology The Nuclear Cardiology Core Laboratory at the Cleveland Clinic Heart and Vascular Institute analyzes Nuclear image data collected as part of clinical trials. Our physicians at Cleveland Clinic: are recognized nationally and internationally as leaders in their fields; are qualified to quantitatively analyze nuclear research exams; use a consistent set of definitions required to perform nuclear image analysis. Nuclear exams are analyzed using American Society of Nuclear Cardiology (ASNC) guidelines and standards as well as core lab standard operating procedures (SOPs). Cardiac Computed Tomography and Magnetic Resonance Imaging The Nuclear Core Lab has experience in providing analysis for clinical trials since Our nuclear experience includes SPECT, PET and MUGA modalities with image volume between 32 and 5000 exams per modality. Modality specific protocols are developed and customized for each trial. These protocols are also used to instruct the collaborating centers on the minimum imaging requirements for subjects enrolled in the trial. The Nuclear Core Lab has dedicated resources for processing, analyzing, transferring and storing image data associated with clinical trials. Initiated in 2006, the CT/MR core facilities can assess: Myocardial ischemia Improved characterization of myocardial function Coronary artery assessment (e.g., plaque-load determination) Response of the failing left ventricle to various interventions Pre-cardiac-surgery planning Arterial-wall imaging
16 Peripheral Vascular Core Laboratory Initiated in 1996, the PV Core Lab has completed over 10 studies and analyzed data for more than 2,500 subjects with typical study durations of 5 years. Trial experience includes Zenith, ENACT, SECURITY and Excluder. Analysis provided for clinical trials in: Abdominal and Thoracic Aortic Aneurysm devices Peripheral devices Carotid devices Imaging modalities reviewed include: Computed Tomography (CT) Magnetic Resonance Imaging (MR) Angiography X-ray Ultrasound (Duplex and IVUS) Assessments include: Centerline vessel analysis Volume measurements 3D/volume rendering Curved Planar Reformatting 4D analysis of cardiac gated studies Vascular stenosis, thrombosis, and calcification Detailed device integrity
17 Biomarker Core Laboratory An array of clinically validated mass spectometry assays is also available A CAP and CLIA Reference Lab measures a full suite of traditional laboratory diagnostics (both chemistry and immunodiagnostics), multiple novel markers of inflammation and cardiovascular risk that predict heart disease. An array of clinically validated mass spectrometry assays is also available. Assay development and/or validation for applying research grade assays to large scale clinical studies is available.
18 Clinical Data Management CDM provides data management services primarily for Core Labs. These services include implementation of standardized processes, development of standardized data forms, frequent communication / interaction with the Core Labs resulting in quick resolution of queries, and scheduled data transfers of cleaned / locked cases to sponsor and/or CRO. Statistics We provide trial design and sample size determination, protocol review, review and input into the statistical analysis plan (SAP), review and input on case report forms and edit checks, independent validation of trial results and statistical support for manuscripts and scientific presentations (primary and post study).
19 Contracts and Finance Our Contracts and Finance team facilitates confidentiality agreements with investigators / sites; handles third party contracts and payment including Data Monitoring Committee (DMC) / Steering Committee, Clinical Events Committee reviewers, meeting planners and other trial participants; facilitates sponsor / site contract negotiation and management, manages and administers site payments including detailed tracking and reporting to sponsor; and administrative accounting for each clinical study. Regulatory and Quality Assurance Regulatory and Quality Assurance for C5Research are provided by Compliance Specialists experienced with the regulatory requirements of both the Sponsor and Investigator. Regulatory and Quality Assurance offers the following services: Independent Auditing Good Clinical Practice (GCP) Training The Regulatory and Quality Assurance team provides C5Research with regulatory consultation and a comprehensive, multifaceted Quality Assurance Program. Independent auditing services are also available and provided to Sponsors. The primary goal of the Regulatory and Quality Assurance team is to promote data integrity and protection of human subjects by ensuring compliance with Federal Regulations, GCPs, and sponsoring agencies policies and procedures governing the use of human participants in research.
20 C5Research Cleveland Clinic JJ Euclid Avenue Cleveland, OH Tim Crowe Project Development Tel: Fax: Revised 9/2010
Diagnostic and Therapeutic Procedures
 Diagnostic and Therapeutic Procedures Diagnostic and therapeutic cardiovascular s are central to the evaluation and management of patients with cardiovascular disease. Consistent with the other sections,
Diagnostic and Therapeutic Procedures Diagnostic and therapeutic cardiovascular s are central to the evaluation and management of patients with cardiovascular disease. Consistent with the other sections,
Specific Basic Standards for Osteopathic Fellowship Training in Cardiology
 Specific Basic Standards for Osteopathic Fellowship Training in Cardiology American Osteopathic Association and American College of Osteopathic Internists BOT 07/2006 Rev. BOT 03/2009 Rev. BOT 07/2011
Specific Basic Standards for Osteopathic Fellowship Training in Cardiology American Osteopathic Association and American College of Osteopathic Internists BOT 07/2006 Rev. BOT 03/2009 Rev. BOT 07/2011
AI CPT Codes. x x. 70336 MRI Magnetic resonance (eg, proton) imaging, temporomandibular joint(s)
 Code Category Description Auth Required Medicaid Medicare 0126T IMT Testing Common carotid intima-media thickness (IMT) study for evaluation of atherosclerotic burden or coronary heart disease risk factor
Code Category Description Auth Required Medicaid Medicare 0126T IMT Testing Common carotid intima-media thickness (IMT) study for evaluation of atherosclerotic burden or coronary heart disease risk factor
FFR CT : Clinical studies
 FFR CT : Clinical studies Bjarne Nørgaard Department Cardiology B Aarhus University Hospital Skejby, Denmark Disclosures: Research grants: Edwards and Siemens Coronary CTA: High diagnostic sensitivity
FFR CT : Clinical studies Bjarne Nørgaard Department Cardiology B Aarhus University Hospital Skejby, Denmark Disclosures: Research grants: Edwards and Siemens Coronary CTA: High diagnostic sensitivity
There are 2 types of clinical trials that are of interest to the. The Clinical Trials Network of the Society of Nuclear Medicine
 The Clinical Trials Network of the Society of Nuclear Medicine Michael M. Graham, PhD, MD The Clinical Trials Network of the Society of Nuclear Medicine was formed to provide quality assurance of both
The Clinical Trials Network of the Society of Nuclear Medicine Michael M. Graham, PhD, MD The Clinical Trials Network of the Society of Nuclear Medicine was formed to provide quality assurance of both
BASIC STANDARDS FOR RESIDENCY TRAINING IN CARDIOLOGY
 BASIC STANDARDS FOR RESIDENCY TRAINING IN CARDIOLOGY American Osteopathic Association and the American College of Osteopathic Internists Specific Requirements For Osteopathic Subspecialty Training In Cardiology
BASIC STANDARDS FOR RESIDENCY TRAINING IN CARDIOLOGY American Osteopathic Association and the American College of Osteopathic Internists Specific Requirements For Osteopathic Subspecialty Training In Cardiology
Diagnostic Imaging Prior Review Code List 3 rd Quarter 2016
 Computerized Tomography (CT) Abdomen 6 Abdomen/Pelvis Combination 101 Service 74150 CT abdomen; w/o 74160 CT abdomen; with 74170 CT abdomen; w/o followed by 74176 Computed tomography, abdomen and pelvis;
Computerized Tomography (CT) Abdomen 6 Abdomen/Pelvis Combination 101 Service 74150 CT abdomen; w/o 74160 CT abdomen; with 74170 CT abdomen; w/o followed by 74176 Computed tomography, abdomen and pelvis;
CPT * Codes Included in AIM Preauthorization Program for 2013 With Grouper Numbers
 CPT * Codes Included in AIM Preauthorization Program for 2013 With Grouper Numbers Computerized Tomography (CT) CPT Description Abdomen 74150 CT abdomen; w/o contrast 6 74160 CT abdomen; with contrast
CPT * Codes Included in AIM Preauthorization Program for 2013 With Grouper Numbers Computerized Tomography (CT) CPT Description Abdomen 74150 CT abdomen; w/o contrast 6 74160 CT abdomen; with contrast
ST. DAVID S MEDICAL CENTER CARDIOLOGY - Special, Invasive, Diagnostic, or High-Risk Procedure Requirements
 ST. DAVID S MEDICAL CENTER CARDIOLOGY - Special, Invasive, Diagnostic, or High-Risk Procedure Requirements Cardiac Catheterization & Peripheral Angiography Completion of a fellowship in Cardiovascular
ST. DAVID S MEDICAL CENTER CARDIOLOGY - Special, Invasive, Diagnostic, or High-Risk Procedure Requirements Cardiac Catheterization & Peripheral Angiography Completion of a fellowship in Cardiovascular
The largest clinical study of Bayer's Xarelto (rivaroxaban) Wednesday, 14 November 2012 07:38
 Bayer HealthCare has announced the initiation of the COMPASS study, the largest clinical study of its oral anticoagulant Xarelto (rivaroxaban) to date, investigating the prevention of major adverse cardiac
Bayer HealthCare has announced the initiation of the COMPASS study, the largest clinical study of its oral anticoagulant Xarelto (rivaroxaban) to date, investigating the prevention of major adverse cardiac
CARDIOLOGY PROCEDURES REQUIRING PRECERTIFICATION
 CLINICAL POLICY CARDIOLOGY PROCEDURES REQUIRING PRECERTIFICATION Policy Number: CARDIOLOGY 026.6 T2 Effective Date: May 1, 2015 Table of Contents CONDITIONS OF COVERAGE... COVERAGE RATIONALE... BENEFIT
CLINICAL POLICY CARDIOLOGY PROCEDURES REQUIRING PRECERTIFICATION Policy Number: CARDIOLOGY 026.6 T2 Effective Date: May 1, 2015 Table of Contents CONDITIONS OF COVERAGE... COVERAGE RATIONALE... BENEFIT
THE ACADEMY OF MEDICINE OF MALAYSIA January 2005 CREDENTIALING REQUIREMENTS FOR THE SPECIALITY OF CARDIOLOGY
 THE ACADEMY OF MEDICINE OF MALAYSIA January 2005 CREDENTIALING REQUIREMENTS FOR THE SPECIALITY OF CARDIOLOGY 1 Contents 1. 2. 3. Introduction, Aims, Objectives and Terms of Reference Cardiology Advisory
THE ACADEMY OF MEDICINE OF MALAYSIA January 2005 CREDENTIALING REQUIREMENTS FOR THE SPECIALITY OF CARDIOLOGY 1 Contents 1. 2. 3. Introduction, Aims, Objectives and Terms of Reference Cardiology Advisory
CARDIOLOGY Delineation of Privileges
 CARDIOLOGY Delineation of Privileges APPLICANT: INITIAL APPOINTMENT REQUIREMENTS: BASIC EDUCATION: M.D. or D.O. from an accredited school of medicine or osteopathy. Successful completion of an ACGME or
CARDIOLOGY Delineation of Privileges APPLICANT: INITIAL APPOINTMENT REQUIREMENTS: BASIC EDUCATION: M.D. or D.O. from an accredited school of medicine or osteopathy. Successful completion of an ACGME or
Computed Tomography, Head Or Brain; Without Contrast Material, Followed By Contrast Material(S) And Further Sections
 1199SEIU BENEFIT AND PENSION FUNDS High Tech Diagnostic Radiology and s # 1 70336 Magnetic Resonance (Eg, Proton) Imaging, Temporomandibular Joint(S) 2 70450 Computed Tomography, Head Or Brain; Without
1199SEIU BENEFIT AND PENSION FUNDS High Tech Diagnostic Radiology and s # 1 70336 Magnetic Resonance (Eg, Proton) Imaging, Temporomandibular Joint(S) 2 70450 Computed Tomography, Head Or Brain; Without
A Proposal for an Advanced Cardiovascular Imaging Training Track
 Journal of the American College of Cardiology Vol. 48, No. 7, 2006 2006 by the American College of Cardiology Foundation ISSN 0735-1097/06/$32.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2006.04.096
Journal of the American College of Cardiology Vol. 48, No. 7, 2006 2006 by the American College of Cardiology Foundation ISSN 0735-1097/06/$32.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2006.04.096
Needs, Providing Solutions
 Identifying Needs, Providing Solutions 1 I n d u s t r y The growth of medical research and the countless innovations coming from the pharmaceutical, biotechnology and medical device industry, has improved
Identifying Needs, Providing Solutions 1 I n d u s t r y The growth of medical research and the countless innovations coming from the pharmaceutical, biotechnology and medical device industry, has improved
2015 European Cardiovascular Image Management Technology Leadership Award
 2015 European Cardiovascular Image Management Technology Leadership Award 2015 Contents Background and Company Performance... 3 Industry Challenges... 3 Technology Leverage and Business Impact... 4 Conclusion...
2015 European Cardiovascular Image Management Technology Leadership Award 2015 Contents Background and Company Performance... 3 Industry Challenges... 3 Technology Leverage and Business Impact... 4 Conclusion...
California Health and Safety Code, Section 1256.01
 California Health and Safety Code, Section 1256.01 1256.01. (a) The Elective Percutaneous Coronary Intervention (PCI) Pilot Program is hereby established in the department. The purpose of the pilot program
California Health and Safety Code, Section 1256.01 1256.01. (a) The Elective Percutaneous Coronary Intervention (PCI) Pilot Program is hereby established in the department. The purpose of the pilot program
Listen to your heart: Good Cardiovascular Health for Life
 Listen to your heart: Good Cardiovascular Health for Life Luis R. Castellanos MD, MPH Assistant Clinical Professor of Medicine University of California San Diego School of Medicine Sulpizio Family Cardiovascular
Listen to your heart: Good Cardiovascular Health for Life Luis R. Castellanos MD, MPH Assistant Clinical Professor of Medicine University of California San Diego School of Medicine Sulpizio Family Cardiovascular
For the NXT Investigators
 Diagnostic performance of non-invasive fractional flow reserve derived from coronary CT angiography in suspected coronary artery disease: The NXT trial Bjarne L. Nørgaard, Jonathon Leipsic, Sara Gaur,
Diagnostic performance of non-invasive fractional flow reserve derived from coronary CT angiography in suspected coronary artery disease: The NXT trial Bjarne L. Nørgaard, Jonathon Leipsic, Sara Gaur,
LEADING-EDGE Cardiovascular Care
 LEADING-Edge Cardiovascular Care Coral Gables Hospital North Shore Medical Center Hialeah Hospital Delray Medical Center Good Samaritan Medical Center Palm Beach Gardens Medical Center St. Mary s Medical
LEADING-Edge Cardiovascular Care Coral Gables Hospital North Shore Medical Center Hialeah Hospital Delray Medical Center Good Samaritan Medical Center Palm Beach Gardens Medical Center St. Mary s Medical
Main Effect of Screening for Coronary Artery Disease Using CT
 Main Effect of Screening for Coronary Artery Disease Using CT Angiography on Mortality and Cardiac Events in High risk Patients with Diabetes: The FACTOR-64 Randomized Clinical Trial Joseph B. Muhlestein,
Main Effect of Screening for Coronary Artery Disease Using CT Angiography on Mortality and Cardiac Events in High risk Patients with Diabetes: The FACTOR-64 Randomized Clinical Trial Joseph B. Muhlestein,
2014 Emerging Faculty Participants
 Nazem Akoum, MD, FACC Assistant Professor of Medicine, Clinical Cardiac Electrophysiology EPIC EMR Cardiology Liaison University of Utah Health Sciences nazem.akoum@hsc.utah.edu Areas of Interest: Cardiac
Nazem Akoum, MD, FACC Assistant Professor of Medicine, Clinical Cardiac Electrophysiology EPIC EMR Cardiology Liaison University of Utah Health Sciences nazem.akoum@hsc.utah.edu Areas of Interest: Cardiac
Purpose Members of the Department of Cardiology will provide cardiology services to patients of McLaren Greater Lansing.
 Purpose Members of the Department of Cardiology will provide cardiology services to patients of McLaren Greater Lansing. Qualifications To be eligible for core privileges in the Department of Cardiology,
Purpose Members of the Department of Cardiology will provide cardiology services to patients of McLaren Greater Lansing. Qualifications To be eligible for core privileges in the Department of Cardiology,
Regions Hospital Delineation of Privileges Cardiology
 Regions Hospital Delineation of s Cardiology Applicant s Name: Last First M. Instructions: Place a check-mark where indicated for each core group you are requesting. Review education and basic formal training
Regions Hospital Delineation of s Cardiology Applicant s Name: Last First M. Instructions: Place a check-mark where indicated for each core group you are requesting. Review education and basic formal training
Multidisciplinary Core Services
 Multidisciplinary Core Services A functionally integrated resource of expertise, instrumentation and technology designed to support investigators in the pursuit of their research interests University Town
Multidisciplinary Core Services A functionally integrated resource of expertise, instrumentation and technology designed to support investigators in the pursuit of their research interests University Town
CPT Radiology Codes Requiring Review by AIM Effective 01/01/2016
 CPT Radiology Codes Requiring Review by AIM Effective 01/01/2016 When a service is authorized only one test per group is payable. *Secondary codes or add-on codes do not require preauthorization or separate
CPT Radiology Codes Requiring Review by AIM Effective 01/01/2016 When a service is authorized only one test per group is payable. *Secondary codes or add-on codes do not require preauthorization or separate
AND SYLLABUS FOR INTERVENTIONAL CARDIOLOGY SUBSPECIALITY TRAINING IN EUROPE
 CURRICULUM AND SYLLABUS FOR INTERVENTIONAL CARDIOLOGY SUBSPECIALITY TRAINING IN EUROPE Document prepared by: Carlo Di Mario, FESC Germano Di Sciascio, FESC Jean-Luc Dubois-Rande,FESC Rolf Michels, FESC
CURRICULUM AND SYLLABUS FOR INTERVENTIONAL CARDIOLOGY SUBSPECIALITY TRAINING IN EUROPE Document prepared by: Carlo Di Mario, FESC Germano Di Sciascio, FESC Jean-Luc Dubois-Rande,FESC Rolf Michels, FESC
PRECOMBAT Trial. Seung-Whan Lee, MD, PhD On behalf of the PRECOMBAT Investigators
 Premier of Randomized Comparison of Bypass Surgery versus Angioplasty Using Sirolimus-Eluting Stent in Patients with Left Main Coronary Artery Disease PRECOMBAT Trial Seung-Whan Lee, MD, PhD On behalf
Premier of Randomized Comparison of Bypass Surgery versus Angioplasty Using Sirolimus-Eluting Stent in Patients with Left Main Coronary Artery Disease PRECOMBAT Trial Seung-Whan Lee, MD, PhD On behalf
Journal Club: Niacin in Patients with Low HDL Cholesterol Levels Receiving Intensive Statin Therapy by the AIM-HIGH Investigators
 Journal Club: Niacin in Patients with Low HDL Cholesterol Levels Receiving Intensive Statin Therapy by the AIM-HIGH Investigators Shaikha Al Naimi Doctor of Pharmacy Student College of Pharmacy Qatar University
Journal Club: Niacin in Patients with Low HDL Cholesterol Levels Receiving Intensive Statin Therapy by the AIM-HIGH Investigators Shaikha Al Naimi Doctor of Pharmacy Student College of Pharmacy Qatar University
Duration of Dual Antiplatelet Therapy After Coronary Stenting
 Duration of Dual Antiplatelet Therapy After Coronary Stenting C. DEAN KATSAMAKIS, DO, FACC, FSCAI INTERVENTIONAL CARDIOLOGIST ADVOCATE LUTHERAN GENERAL HOSPITAL INTRODUCTION Coronary artery stents are
Duration of Dual Antiplatelet Therapy After Coronary Stenting C. DEAN KATSAMAKIS, DO, FACC, FSCAI INTERVENTIONAL CARDIOLOGIST ADVOCATE LUTHERAN GENERAL HOSPITAL INTRODUCTION Coronary artery stents are
University of Missouri Kansas City School of Medicine and the Mid America Heart Institute of Saint Luke s Hospital
 PROGRAM DIRECTOR: Dr. Steven Laster University of Missouri Kansas City School of Medicine and the Mid America Heart Institute of Saint Luke s Hospital 1 Interventional Cardiology Fellowship Training Program
PROGRAM DIRECTOR: Dr. Steven Laster University of Missouri Kansas City School of Medicine and the Mid America Heart Institute of Saint Luke s Hospital 1 Interventional Cardiology Fellowship Training Program
Cilostazol versus Clopidogrel after Coronary Stenting
 Cilostazol versus Clopidogrel after Coronary Stenting Seong-Wook Park, MD, PhD, FACC Division of Cardiology, Asan Medical Center University of Ulsan College of Medicine Seoul, Korea AMC, 2004 Background
Cilostazol versus Clopidogrel after Coronary Stenting Seong-Wook Park, MD, PhD, FACC Division of Cardiology, Asan Medical Center University of Ulsan College of Medicine Seoul, Korea AMC, 2004 Background
Efficient Evaluation of Chest Pain
 Efficient Evaluation of Chest Pain Vikranth Gongidi, DO FACC FACOI Indian River Medical Center Vero Beach, FL No Disclosures Outline Background Chest pain pathway Indications for stress test Stress test
Efficient Evaluation of Chest Pain Vikranth Gongidi, DO FACC FACOI Indian River Medical Center Vero Beach, FL No Disclosures Outline Background Chest pain pathway Indications for stress test Stress test
Full-time or part-time upto 1.0 FTE, 4 year fix term Principal Investigator Department of Cardiology Westmead Hospital
 Position Description POSITION TITLE Project Manager TYPE OF EMPLOYMENT Full-time or part-time upto 1.0 FTE, 4 year fix term REPORTING TO a) Principal Investigator DIVISION Department of Cardiology COMPANY
Position Description POSITION TITLE Project Manager TYPE OF EMPLOYMENT Full-time or part-time upto 1.0 FTE, 4 year fix term REPORTING TO a) Principal Investigator DIVISION Department of Cardiology COMPANY
Fort Hamilton Hospital Specialty: Cardiology Department of Medicine Delineation of Privileges
 NAME Fort Hamilton Hospital Specialty: Cardiology Department of Medicine Delineation of Privileges GENERAL CARDIOLOGY Required Qualifications for General Cardiology Education/Training/Experience Must have
NAME Fort Hamilton Hospital Specialty: Cardiology Department of Medicine Delineation of Privileges GENERAL CARDIOLOGY Required Qualifications for General Cardiology Education/Training/Experience Must have
Heart Failure: From Prevention to Intervention
 Sharp HealthCare Presents Heart Failure: From Prevention to Intervention Saturday, May 16, 2015 DoubleTree by Hilton Hotel San Diego - Mission Valley San Diego, Calif. As medical professionals, we know
Sharp HealthCare Presents Heart Failure: From Prevention to Intervention Saturday, May 16, 2015 DoubleTree by Hilton Hotel San Diego - Mission Valley San Diego, Calif. As medical professionals, we know
Michigan Heart & Vascular Institute ON THE ST. JOSEPH MERCY HOSPITAL CAMPUS, ANN ARBOR, MICHIGAN
 ON THE ST. JOSEPH MERCY HOSPITAL CAMPUS, ANN ARBOR, MICHIGAN Dear Colleague: Cardiovascular medicine has marked an important milestone in the battle against heart disease. The latest available data indicates
ON THE ST. JOSEPH MERCY HOSPITAL CAMPUS, ANN ARBOR, MICHIGAN Dear Colleague: Cardiovascular medicine has marked an important milestone in the battle against heart disease. The latest available data indicates
Ch. 138 CARDIAC CATHETERIZATION SERVICES 28 138.1 CHAPTER 138. CARDIAC CATHETERIZATION SERVICES GENERAL PROVISIONS
 Ch. 138 CARDIAC CATHETERIZATION SERVICES 28 138.1 CHAPTER 138. CARDIAC CATHETERIZATION SERVICES Sec. 138.1 Principle. 138.2. Definitions. GENERAL PROVISIONS PROGRAM, SERVICE, PERSONNEL AND AGREEMENT REQUIREMENTS
Ch. 138 CARDIAC CATHETERIZATION SERVICES 28 138.1 CHAPTER 138. CARDIAC CATHETERIZATION SERVICES Sec. 138.1 Principle. 138.2. Definitions. GENERAL PROVISIONS PROGRAM, SERVICE, PERSONNEL AND AGREEMENT REQUIREMENTS
SECTION I: Request. SECTION II: Need. Program Description
 SECTION I: Request This is a formal request for the Utah CARMA Center to be formally recognized by the University of Utah as a large, collaborative medical and academic center whose focus is on comprehensive
SECTION I: Request This is a formal request for the Utah CARMA Center to be formally recognized by the University of Utah as a large, collaborative medical and academic center whose focus is on comprehensive
CARDIAC CARE. Giving you every advantage
 CARDIAC CARE Giving you every advantage Getting to the heart of the matter The Cardiovascular Program at Northwest Hospital & Medical Center is dedicated to the management of cardiovascular disease. The
CARDIAC CARE Giving you every advantage Getting to the heart of the matter The Cardiovascular Program at Northwest Hospital & Medical Center is dedicated to the management of cardiovascular disease. The
SUTTER MEDICAL CENTER, SACRAMENTO Department of Cardiovascular Disease Cardiology - Delineation of Privileges
 INITIAL: [ ] RENEWED: [ ] DATE: ADDITIONAL: [ ] Privileges are granted for Sutter General Hospital, Sutter Memorial Hospital, Sutter Center for Psychiatry, Sutter Oaks Midtown and the Capitol Pavilion
INITIAL: [ ] RENEWED: [ ] DATE: ADDITIONAL: [ ] Privileges are granted for Sutter General Hospital, Sutter Memorial Hospital, Sutter Center for Psychiatry, Sutter Oaks Midtown and the Capitol Pavilion
Educational Goals & Objectives
 Educational Goals & Objectives The Cardiology rotation will provide the resident with an understanding of cardiovascular physiology and its broad systemic manifestations. The resident will have the opportunity
Educational Goals & Objectives The Cardiology rotation will provide the resident with an understanding of cardiovascular physiology and its broad systemic manifestations. The resident will have the opportunity
University of Ulsan College of Medicine, Asan Medical Center on behalf of the REAL-LATE and the ZEST-LATE trial
 Duration of Dual Antiplatelet Therapy After Drug-Eluting Stent Implantation A Pooled Analysis of the REAL-LATE and the ZEST-LATE Trial Seung-Jung Park MD PhD Seung-Jung Park, MD, PhD, University of Ulsan
Duration of Dual Antiplatelet Therapy After Drug-Eluting Stent Implantation A Pooled Analysis of the REAL-LATE and the ZEST-LATE Trial Seung-Jung Park MD PhD Seung-Jung Park, MD, PhD, University of Ulsan
Cardiology Fellowship Manual. Goals & Objectives -Cardiac Imaging- 1 Page
 Cardiology Fellowship Manual Goals & Objectives -Cardiac Imaging- 1 Page 2015-2016 UNIV. OF NEBRASKA CHILDREN S HOSPITAL & MEDICAL CENTER DIVISION OF CARDIOLOGY FELLOWSHIP PROGRAM CARDIAC IMAGING ROTATION
Cardiology Fellowship Manual Goals & Objectives -Cardiac Imaging- 1 Page 2015-2016 UNIV. OF NEBRASKA CHILDREN S HOSPITAL & MEDICAL CENTER DIVISION OF CARDIOLOGY FELLOWSHIP PROGRAM CARDIAC IMAGING ROTATION
Bayer Extends Clinical Investigation of Rivaroxaban into Important Areas of Unmet Medical Need in Arterial Thromboembolism
 Investor News Not intended for U.S. and UK Media Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com Bayer Extends Clinical Investigation of Rivaroxaban into Important Areas of
Investor News Not intended for U.S. and UK Media Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com Bayer Extends Clinical Investigation of Rivaroxaban into Important Areas of
Press. Siemens solutions support diagnosis and treatment of cardiovascular diseases
 Press Healthcare Erlangen, August 29, 2015 ESC 2015: ExCel London Exhibition and Convention Center, Booth #G700 Siemens solutions support diagnosis and treatment of cardiovascular diseases New cardiovascular
Press Healthcare Erlangen, August 29, 2015 ESC 2015: ExCel London Exhibition and Convention Center, Booth #G700 Siemens solutions support diagnosis and treatment of cardiovascular diseases New cardiovascular
Cardiology ARCP Decision Aid August 2014
 Cardiology ARCP Decision Aid August 2014 The table that follows includes a column for each training year which documents the targets that have to be achieved for a satisfactory ARCP outcome at the end
Cardiology ARCP Decision Aid August 2014 The table that follows includes a column for each training year which documents the targets that have to be achieved for a satisfactory ARCP outcome at the end
Atherosclerosis of the aorta. Artur Evangelista
 Atherosclerosis of the aorta Artur Evangelista Atherosclerosis of the aorta Diagnosis Classification Prevalence Risk factors Marker of generalized atherosclerosis Risk of embolism Therapy Diagnosis Atherosclerosis
Atherosclerosis of the aorta Artur Evangelista Atherosclerosis of the aorta Diagnosis Classification Prevalence Risk factors Marker of generalized atherosclerosis Risk of embolism Therapy Diagnosis Atherosclerosis
DISCLOSURES RISK ASSESSMENT. Stroke and Heart Disease -Is there a Link Beyond Risk Factors? Daniel Lackland, MD
 STROKE AND HEART DISEASE IS THERE A LINK BEYOND RISK FACTORS? D AN IE L T. L AC K L AN D DISCLOSURES Member of NHLBI Risk Assessment Workgroup RISK ASSESSMENT Count major risk factors For patients with
STROKE AND HEART DISEASE IS THERE A LINK BEYOND RISK FACTORS? D AN IE L T. L AC K L AN D DISCLOSURES Member of NHLBI Risk Assessment Workgroup RISK ASSESSMENT Count major risk factors For patients with
on behalf of the AUGMENT-HF Investigators
 One Year Follow-Up Results from AUGMENT-HF: A Multicenter Randomized Controlled Clinical Trial of the Efficacy of Left Ventricular Augmentation with Algisyl-LVR in the Treatment of Heart Failure* Douglas
One Year Follow-Up Results from AUGMENT-HF: A Multicenter Randomized Controlled Clinical Trial of the Efficacy of Left Ventricular Augmentation with Algisyl-LVR in the Treatment of Heart Failure* Douglas
www.bioclinica.com Imaging Core Lab Solutions Electronic Data Capture Clinical Trial Supply Planning IWR/IVRS
 Global clinical trial solutions. Real-world results. Imaging Core Lab Solutions Electronic Data Capture Clinical Trial Supply Planning IWR/IVRS Clinical Trial Management Clinical Data Management www.bioclinica.com
Global clinical trial solutions. Real-world results. Imaging Core Lab Solutions Electronic Data Capture Clinical Trial Supply Planning IWR/IVRS Clinical Trial Management Clinical Data Management www.bioclinica.com
Christopher M. Wright, MD, MBA Pioneer Cardiovascular Consultants Tempe, Arizona
 Christopher M. Wright, MD, MBA Pioneer Cardiovascular Consultants Tempe, Arizona Areas to be covered Historical, current, and future treatments for various cardiovascular disease: Atherosclerosis (Coronary
Christopher M. Wright, MD, MBA Pioneer Cardiovascular Consultants Tempe, Arizona Areas to be covered Historical, current, and future treatments for various cardiovascular disease: Atherosclerosis (Coronary
Treating AF: The Newest Recommendations. CardioCase presentation. Ethel s Case. Wayne Warnica, MD, FACC, FACP, FRCPC
 Treating AF: The Newest Recommendations Wayne Warnica, MD, FACC, FACP, FRCPC CardioCase presentation Ethel s Case Ethel, 73, presents with rapid heart beating and mild chest discomfort. In the ED, ECG
Treating AF: The Newest Recommendations Wayne Warnica, MD, FACC, FACP, FRCPC CardioCase presentation Ethel s Case Ethel, 73, presents with rapid heart beating and mild chest discomfort. In the ED, ECG
CARDIAC NURSING. Graduate Diploma in Nursing Science. Overview. Entry Requirements. Fees. Contact. Teaching Methods.
 Graduate Diploma in Nursing Science CARDIAC NURSING Overview The Graduate Diploma in Nursing Science (Cardiac Nursing) is designed to develop advanced theoretical knowledge and specialist skills essential
Graduate Diploma in Nursing Science CARDIAC NURSING Overview The Graduate Diploma in Nursing Science (Cardiac Nursing) is designed to develop advanced theoretical knowledge and specialist skills essential
Magnetic Resonance Quantitative Analysis. MRV MR Flow. Reliable analysis of heart and peripheral arteries in the clinical workflow
 Magnetic Resonance Quantitative Analysis MRV MR Flow Reliable analysis of heart and peripheral arteries in the clinical workflow CAAS MRV Functional Workflow Designed for imaging specialists, CAAS MRV
Magnetic Resonance Quantitative Analysis MRV MR Flow Reliable analysis of heart and peripheral arteries in the clinical workflow CAAS MRV Functional Workflow Designed for imaging specialists, CAAS MRV
COMMITTEE FOR MEDICINAL PRODUCTS FOR HUMAN USE (CHMP) GUIDELINE ON THE EVALUATION OF MEDICINAL PRODUCTS FOR CARDIOVASCULAR DISEASE PREVENTION
 European Medicines Agency Pre-Authorisation Evaluation of Medicines for Human Use London, 25 September 2008 Doc. Ref. EMEA/CHMP/EWP/311890/2007 COMMITTEE FOR MEDICINAL PRODUCTS FOR HUMAN USE (CHMP) GUIDELINE
European Medicines Agency Pre-Authorisation Evaluation of Medicines for Human Use London, 25 September 2008 Doc. Ref. EMEA/CHMP/EWP/311890/2007 COMMITTEE FOR MEDICINAL PRODUCTS FOR HUMAN USE (CHMP) GUIDELINE
Medical images such as X-rays, computed tomography (CT) images,
 Performance Metrics in Clinical trials To be confident about making decisions based on medical images for individuals and for clinical trials, medical professionals can use metrics to develop adequate
Performance Metrics in Clinical trials To be confident about making decisions based on medical images for individuals and for clinical trials, medical professionals can use metrics to develop adequate
Bayer Pharma AG 13342 Berlin Germany Tel. +49 30 468-1111 www.bayerpharma.com. News Release. Not intended for U.S. and UK Media
 News Release Not intended for U.S. and UK Media Bayer Pharma AG 13342 Berlin Germany Tel. +49 30 468-1111 www.bayerpharma.com Bayer Forms Collaboration with Academic and Governmental Institutions for Rivaroxaban
News Release Not intended for U.S. and UK Media Bayer Pharma AG 13342 Berlin Germany Tel. +49 30 468-1111 www.bayerpharma.com Bayer Forms Collaboration with Academic and Governmental Institutions for Rivaroxaban
History and Principles of Good Clinical Practice
 History and Principles of Good Clinical Practice Cristina E. Torres, Ph.D. Social Science Professor, UPM-NIH FERCAP Coordinator ICH: International Conference on Harmonization GCP: Good Clinical Practices
History and Principles of Good Clinical Practice Cristina E. Torres, Ph.D. Social Science Professor, UPM-NIH FERCAP Coordinator ICH: International Conference on Harmonization GCP: Good Clinical Practices
What We Are..! www.ardent-cro.com
 Your Trusted CRO! Regus, Level-2, Connaught Place, Bund Garden Road, Pune-411001, MH, India. Phone: 020-65-31-31-71 Email: ardent@ardent-cro.com Web: What We Are..! Ardent Clinical Research Services is
Your Trusted CRO! Regus, Level-2, Connaught Place, Bund Garden Road, Pune-411001, MH, India. Phone: 020-65-31-31-71 Email: ardent@ardent-cro.com Web: What We Are..! Ardent Clinical Research Services is
Atrial Fibrillation, Chronic - Antithrombotic Treatment - OBSOLETE
 Atrial Fibrillation, Chronic - Antithrombotic Treatment - OBSOLETE Clinical practice guidelines serve as an educational reference, and do not supersede the clinical judgment of the treating physician with
Atrial Fibrillation, Chronic - Antithrombotic Treatment - OBSOLETE Clinical practice guidelines serve as an educational reference, and do not supersede the clinical judgment of the treating physician with
PHARMACOLOGICAL Stroke Prevention in Atrial Fibrillation STROKE RISK ASSESSMENT SCORES Vs. BLEEDING RISK ASSESSMENT SCORES.
 PHARMACOLOGICAL Stroke Prevention in Atrial Fibrillation STROKE RISK ASSESSMENT SCORES Vs. BLEEDING RISK ASSESSMENT SCORES. Hossam Bahy, MD (1992 2012), 19 tools have been identified 11 stroke scores 1
PHARMACOLOGICAL Stroke Prevention in Atrial Fibrillation STROKE RISK ASSESSMENT SCORES Vs. BLEEDING RISK ASSESSMENT SCORES. Hossam Bahy, MD (1992 2012), 19 tools have been identified 11 stroke scores 1
Center of Excellence in Naturopathic Cardiovascular Medicine
 Introducing the Center of Excellence in Naturopathic Cardiovascular Medicine The Heart & Lung Wellness program at Center for Natural Medicine and National College of Natural Medicine WHO WE ARE A Center
Introducing the Center of Excellence in Naturopathic Cardiovascular Medicine The Heart & Lung Wellness program at Center for Natural Medicine and National College of Natural Medicine WHO WE ARE A Center
Presenter: Marco Valgimigli, MD PhD, FESC Erasmus MC, Thoraxcenter Rotterdam The Netherlands
 Comparing zotarolimus-eluting and bare-metal stent efficacy in selected high bleeding risk patients treated with a short dual antiplatelet therapy duration. A pre-specified analysis from the The Zotarolimuseluting
Comparing zotarolimus-eluting and bare-metal stent efficacy in selected high bleeding risk patients treated with a short dual antiplatelet therapy duration. A pre-specified analysis from the The Zotarolimuseluting
Coronary Artery Disease leading cause of morbidity & mortality in industrialised nations.
 INTRODUCTION Coronary Artery Disease leading cause of morbidity & mortality in industrialised nations. Although decrease in cardiovascular mortality still major cause of morbidity & burden of disease.
INTRODUCTION Coronary Artery Disease leading cause of morbidity & mortality in industrialised nations. Although decrease in cardiovascular mortality still major cause of morbidity & burden of disease.
MYOCARDIAL PERFUSION COMPUTED TOMOGRAPHY PhD course in Medical Imaging. Anne Günther Department of Radiology OUS Rikshospitalet
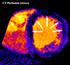 MYOCARDIAL PERFUSION COMPUTED TOMOGRAPHY PhD course in Medical Imaging Anne Günther Department of Radiology OUS Rikshospitalet CORONARY CT ANGIOGRAPHY (CTA) Accurate method in the assessment of possible
MYOCARDIAL PERFUSION COMPUTED TOMOGRAPHY PhD course in Medical Imaging Anne Günther Department of Radiology OUS Rikshospitalet CORONARY CT ANGIOGRAPHY (CTA) Accurate method in the assessment of possible
Steven J. Yakubov, MD FACC For the CoreValve US Clinical Investigators
 Long-Term Outcomes Using a Self- Expanding Bioprosthesis in Patients With Severe Aortic Stenosis Deemed Extreme Risk for Surgery: Two-Year Results From the CoreValve US Pivotal Trial Steven J. Yakubov,
Long-Term Outcomes Using a Self- Expanding Bioprosthesis in Patients With Severe Aortic Stenosis Deemed Extreme Risk for Surgery: Two-Year Results From the CoreValve US Pivotal Trial Steven J. Yakubov,
ABOUT XARELTO CLINICAL STUDIES
 ABOUT XARELTO CLINICAL STUDIES FAST FACTS Xarelto (rivaroxaban) is a novel, oral direct Factor Xa inhibitor. On September 30, 2008, the European Commission granted marketing approval for Xarelto for the
ABOUT XARELTO CLINICAL STUDIES FAST FACTS Xarelto (rivaroxaban) is a novel, oral direct Factor Xa inhibitor. On September 30, 2008, the European Commission granted marketing approval for Xarelto for the
Subject: No. Page PROTOCOL AND CASE REPORT FORM DEVELOPMENT AND REVIEW Standard Operating Procedure
 703 1 of 11 POLICY The Beaumont Research Coordinating Center (BRCC) will provide advice to clinical trial investigators on protocol development, content and format. Upon request, the BRCC will review a
703 1 of 11 POLICY The Beaumont Research Coordinating Center (BRCC) will provide advice to clinical trial investigators on protocol development, content and format. Upon request, the BRCC will review a
Provider Checklist-Outpatient Imaging. Checklist: Nuclear Stress Test, Thallium/Technetium/Sestamibi (CPT Code 78451-78454 78469)
 Provider Checklist-Outpatient Imaging Checklist: Nuclear Stress Test, Thallium/Technetium/Sestamibi (CPT Code 78451-78454 78469) Medical Review Note: Per InterQual, if any of the following are present,
Provider Checklist-Outpatient Imaging Checklist: Nuclear Stress Test, Thallium/Technetium/Sestamibi (CPT Code 78451-78454 78469) Medical Review Note: Per InterQual, if any of the following are present,
The Cardiac Society of Australia and New Zealand
 The Cardiac Society of Australia and New Zealand Guidelines on Support Facilities for Coronary Angiography and Percutaneous Coronary Intervention (PCI) including Guidelines on the Performance of Procedures
The Cardiac Society of Australia and New Zealand Guidelines on Support Facilities for Coronary Angiography and Percutaneous Coronary Intervention (PCI) including Guidelines on the Performance of Procedures
CHAPTER 4 QUALITY ASSURANCE AND TEST VALIDATION
 CHAPTER 4 QUALITY ASSURANCE AND TEST VALIDATION CINDY WEILAND AND SANDRA L. KATANICK Continued innovations in noninvasive testing equipment provide skilled sonographers and physicians with the technology
CHAPTER 4 QUALITY ASSURANCE AND TEST VALIDATION CINDY WEILAND AND SANDRA L. KATANICK Continued innovations in noninvasive testing equipment provide skilled sonographers and physicians with the technology
PIHRATE Trial. Polish-Italian-Hungarian Randomized ThrombEctomy Trial. Dariusz Dudek MD, PhD. On behalf PIHRATE investigators
 Polish-Italian-Hungarian Randomized ThrombEctomy Trial PIHRATE Trial On behalf PIHRATE investigators Dariusz Dudek MD, PhD Institute of Cardiology, Krakow, Poland Impact of distal embolization Distal embolization
Polish-Italian-Hungarian Randomized ThrombEctomy Trial PIHRATE Trial On behalf PIHRATE investigators Dariusz Dudek MD, PhD Institute of Cardiology, Krakow, Poland Impact of distal embolization Distal embolization
Jeffrey M. Carlton Heart & Vascular Institute Presents
 Jeffrey M. Carlton Heart & Vascular Institute Presents 6 th ANNUAL CARDIOLOGY UPDATE: NEW DEVELOPMENTS Program Director: Robert and Georgia Roth Endowed Chair for Excellence in Cardiac Care Saturday, September
Jeffrey M. Carlton Heart & Vascular Institute Presents 6 th ANNUAL CARDIOLOGY UPDATE: NEW DEVELOPMENTS Program Director: Robert and Georgia Roth Endowed Chair for Excellence in Cardiac Care Saturday, September
Cardiovascular diseases. pathology
 Cardiovascular diseases pathology Atherosclerosis Vascular diseases A disease that results in arterial wall thickens as a result of build- up of fatty materials such cholesterol, resulting in acute and
Cardiovascular diseases pathology Atherosclerosis Vascular diseases A disease that results in arterial wall thickens as a result of build- up of fatty materials such cholesterol, resulting in acute and
CPT CODE PROCEDURE DESCRIPTION. CT Scans 70450 CT HEAD/BRAIN W/O CONTRAST 70460 CT HEAD/BRAIN W/ CONTRAST 70470 CT HEAD/BRAIN W/O & W/ CONTRAST
 CPT CODE PROCEDURE DESCRIPTION CT Scans 70450 CT HEAD/BRAIN W/O CONTRAST 70460 CT HEAD/BRAIN W/ CONTRAST 70470 CT HEAD/BRAIN W/O & W/ CONTRAST 70480 CT ORBIT W/O CONTRAST 70481 CT ORBIT W/ CONTRAST 70482
CPT CODE PROCEDURE DESCRIPTION CT Scans 70450 CT HEAD/BRAIN W/O CONTRAST 70460 CT HEAD/BRAIN W/ CONTRAST 70470 CT HEAD/BRAIN W/O & W/ CONTRAST 70480 CT ORBIT W/O CONTRAST 70481 CT ORBIT W/ CONTRAST 70482
SAMPLE. Asia-Pacific Interventional Cardiology Procedures Outlook to 2020. Reference Code: GDMECR0061PDB. Publication Date: May 2014
 Asia-Pacific Interventional Cardiology Procedures Outlook to 2020 Reference Code: GDMECR0061PDB Publication Date: May 2014 Page 1 1 Table of Contents 1 Table of Contents... 2 1.1 List of Tables... 4 1.2
Asia-Pacific Interventional Cardiology Procedures Outlook to 2020 Reference Code: GDMECR0061PDB Publication Date: May 2014 Page 1 1 Table of Contents 1 Table of Contents... 2 1.1 List of Tables... 4 1.2
2/20/2015. Cardiac Evaluation of Potential Solid Organ Transplant Recipients. Issues Specific to Transplantation. Kidney Transplantation.
 DISCLOSURES I have no relevant financial relationships to disclose. Cardiac Evaluation of Potential Solid Organ Transplant Recipients Michele Hamilton, MD Director, Heart Failure Program Cedars Sinai Heart
DISCLOSURES I have no relevant financial relationships to disclose. Cardiac Evaluation of Potential Solid Organ Transplant Recipients Michele Hamilton, MD Director, Heart Failure Program Cedars Sinai Heart
Cardiac Clinical Advisory Group Cardiology Services
 Cardiac Clinical Advisory Group Response to Green Paper The Cardiac Clinical Advisory Group (CAG) is pleased to have this opportunity to provide this response to the Government s Green Paper for. There
Cardiac Clinical Advisory Group Response to Green Paper The Cardiac Clinical Advisory Group (CAG) is pleased to have this opportunity to provide this response to the Government s Green Paper for. There
What Does it Mean When Patients Choose Wisely? David Ansley Senior Analyst, Health Product Development Consumer Reports
 What Does it Mean When Patients Choose Wisely? David Ansley Senior Analyst, Health Product Development Consumer Reports People are realizing that more medicine is not necessarily better. And that we re
What Does it Mean When Patients Choose Wisely? David Ansley Senior Analyst, Health Product Development Consumer Reports People are realizing that more medicine is not necessarily better. And that we re
PG Certificate / PG Diploma / MSc in Clinical Pharmacy
 PG Certificate / PG Diploma / MSc in Clinical Pharmacy Programme Information September 2014 Entry School of Pharmacy Queen s University Belfast Queen s University Belfast - Clinical Pharmacy programme
PG Certificate / PG Diploma / MSc in Clinical Pharmacy Programme Information September 2014 Entry School of Pharmacy Queen s University Belfast Queen s University Belfast - Clinical Pharmacy programme
NEW HYBRID IMAGING TECHNOLOGY MAY HAVE BIG POTENTIAL FOR IMPROVING DIAGNOSIS OF PROSTATE CANCER
 Media Release April 7, 2009 For Immediate Release NEW HYBRID IMAGING TECHNOLOGY MAY HAVE BIG POTENTIAL FOR IMPROVING DIAGNOSIS OF PROSTATE CANCER London, Ontario Improved hybrid imaging techniques developed
Media Release April 7, 2009 For Immediate Release NEW HYBRID IMAGING TECHNOLOGY MAY HAVE BIG POTENTIAL FOR IMPROVING DIAGNOSIS OF PROSTATE CANCER London, Ontario Improved hybrid imaging techniques developed
Task Force 1: Training in Clinical Cardiology
 Journal of the American College of Cardiology Vol. 51, No., 008 008 by the American College of Cardiology Foundation ISSN 075-1097/08/$4.00 Published by Elsevier Inc. Task Forces Task Force 1: Training
Journal of the American College of Cardiology Vol. 51, No., 008 008 by the American College of Cardiology Foundation ISSN 075-1097/08/$4.00 Published by Elsevier Inc. Task Forces Task Force 1: Training
Contemporary Management of Cardiovascular Disease
 Contemporary Management of Cardiovascular Disease NOVEMBER 20 21, 2015 Hilton Anatole Hotel Register Today! ccfcme.org/gocvdtx Contemporary Management of Cardiovascular Disease Join us on November 20-21
Contemporary Management of Cardiovascular Disease NOVEMBER 20 21, 2015 Hilton Anatole Hotel Register Today! ccfcme.org/gocvdtx Contemporary Management of Cardiovascular Disease Join us on November 20-21
OHTAC Recommendation
 OHTAC Recommendation Multiple Sclerosis and Chronic Cerebrospinal Venous Insufficiency Presented to the Ontario Health Technology Advisory Committee in May 2010 May 2010 Issue Background A review on the
OHTAC Recommendation Multiple Sclerosis and Chronic Cerebrospinal Venous Insufficiency Presented to the Ontario Health Technology Advisory Committee in May 2010 May 2010 Issue Background A review on the
Hot Line Session at European Society of Cardiology (ESC) Congress 2014:
 Investor News Not intended for U.S. and UK Media Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com Hot Line Session at European Society of Cardiology (ESC) Congress 2014: Once-Daily
Investor News Not intended for U.S. and UK Media Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com Hot Line Session at European Society of Cardiology (ESC) Congress 2014: Once-Daily
Cardiac Catheterization Curriculum for Fellows in Cardiology Dartmouth-Hitchcock Medical Center Level 1 and Level 2 Training 2008-2009
 Cardiac Catheterization Curriculum for Fellows in Cardiology Dartmouth-Hitchcock Medical Center Level 1 and Level 2 Training 2008-2009 I. Overview of Training in Cardiac Catheterization Cardiac catheterization
Cardiac Catheterization Curriculum for Fellows in Cardiology Dartmouth-Hitchcock Medical Center Level 1 and Level 2 Training 2008-2009 I. Overview of Training in Cardiac Catheterization Cardiac catheterization
Antiplatelet and anticoagulation treatment of patients undergoing carotid and peripheral artery angioplasty
 Round Table: Antithrombotic therapy beyond ACS Antiplatelet and anticoagulation treatment of patients undergoing carotid and peripheral artery angioplasty M. Matsagkas, MD, PhD, EBSQ-Vasc Associate Professor
Round Table: Antithrombotic therapy beyond ACS Antiplatelet and anticoagulation treatment of patients undergoing carotid and peripheral artery angioplasty M. Matsagkas, MD, PhD, EBSQ-Vasc Associate Professor
Ultrasound in Vascular Surgery. Torbjørn Dahl
 Ultrasound in Vascular Surgery Torbjørn Dahl 1 The field of vascular surgery Veins dilatation and obstruction (varicose veins and valve dysfunction) Arteries dilatation and narrowing (aneurysms and atherosclerosis)
Ultrasound in Vascular Surgery Torbjørn Dahl 1 The field of vascular surgery Veins dilatation and obstruction (varicose veins and valve dysfunction) Arteries dilatation and narrowing (aneurysms and atherosclerosis)
ESC/EASD Pocket Guidelines Diabetes, pre-diabetes and cardiovascular disease
 Diabetes, prediabetes and cardiovascular disease Classes of recommendations Levels of evidence Recommended treatment targets for patients with diabetes and CAD Definition, classification and screening
Diabetes, prediabetes and cardiovascular disease Classes of recommendations Levels of evidence Recommended treatment targets for patients with diabetes and CAD Definition, classification and screening
STROKE PREVENTION IN ATRIAL FIBRILLATION
 STROKE PREVENTION IN ATRIAL FIBRILLATION OBJECTIVE: To guide clinicians in the selection of antithrombotic therapy for the secondary prevention of ischemic stroke and arterial thromboembolism in patients
STROKE PREVENTION IN ATRIAL FIBRILLATION OBJECTIVE: To guide clinicians in the selection of antithrombotic therapy for the secondary prevention of ischemic stroke and arterial thromboembolism in patients
CLINICAL TRIALS SHOULD YOU PARTICIPATE? by Gwen L. Nichols, MD
 CLINICAL TRIALS SHOULD YOU PARTICIPATE? by Gwen L. Nichols, MD Gwen L. Nichols, M.D., is currently the Oncology Site Head of the Roche Translational Clinical Research Center at Hoffman- LaRoche. In this
CLINICAL TRIALS SHOULD YOU PARTICIPATE? by Gwen L. Nichols, MD Gwen L. Nichols, M.D., is currently the Oncology Site Head of the Roche Translational Clinical Research Center at Hoffman- LaRoche. In this
HEART CENTER. Touching Lives
 HEART CENTER Touching Lives with INNOVATIVE TOOLS and an EXPERIENCED TEAM THE HEART MATTERS If you or someone you love is faced with a heart problem, you want to put your trust in experienced professionals
HEART CENTER Touching Lives with INNOVATIVE TOOLS and an EXPERIENCED TEAM THE HEART MATTERS If you or someone you love is faced with a heart problem, you want to put your trust in experienced professionals
RITMIR024 - STEM CELL RESEARCH IN CARDIOLOGY
 1. SCOPE AND METHODOLOGY 2. REPORT SYNOPSIS 2.1 General Definitions Myoblasts Pluripotent Cells Multipotent Cells Progenitor Cells Role of Stem Cells in Repairing the Heart Stem Cell Therapy Stem Cells
1. SCOPE AND METHODOLOGY 2. REPORT SYNOPSIS 2.1 General Definitions Myoblasts Pluripotent Cells Multipotent Cells Progenitor Cells Role of Stem Cells in Repairing the Heart Stem Cell Therapy Stem Cells
Medical management of CHF: A New Class of Medication. Al Timothy, M.D. Cardiovascular Institute of the South
 Medical management of CHF: A New Class of Medication Al Timothy, M.D. Cardiovascular Institute of the South Disclosures Speakers Bureau for Amgen Background Chronic systolic congestive heart failure remains
Medical management of CHF: A New Class of Medication Al Timothy, M.D. Cardiovascular Institute of the South Disclosures Speakers Bureau for Amgen Background Chronic systolic congestive heart failure remains
Majestic Trial 12 Month Results
 Majestic Trial 12 Month Results S.Müller-Hülsbeck, MD, EBIR, FCIRSE, FICA ACADEMIC HOSPITALS Flensburg of Kiel University Ev.-Luth. Diakonissenanstalt zu Flensburg Knuthstraße 1, 24939 FLENSBURG Dept.
Majestic Trial 12 Month Results S.Müller-Hülsbeck, MD, EBIR, FCIRSE, FICA ACADEMIC HOSPITALS Flensburg of Kiel University Ev.-Luth. Diakonissenanstalt zu Flensburg Knuthstraße 1, 24939 FLENSBURG Dept.
GE Healthcare. Centricity* PACS and PACS-IW with Universal Viewer. Universal Viewer. Where it all comes together.
 GE Healthcare Centricity* PACS and PACS-IW with Universal Viewer Universal Viewer. Where it all comes together. Where it all comes together Centricity PACS and Centricity PACS-IW with Universal Viewer
GE Healthcare Centricity* PACS and PACS-IW with Universal Viewer Universal Viewer. Where it all comes together. Where it all comes together Centricity PACS and Centricity PACS-IW with Universal Viewer
4D Magnetic Resonance Analysis. MR 4D Flow. Visualization and Quantification of Aortic Blood Flow
 4D Magnetic Resonance Analysis MR 4D Flow Visualization and Quantification of Aortic Blood Flow 4D Magnetic Resonance Analysis Pie Medical Imaging, manufacturer of Quantitative Analysis software for cardiology
4D Magnetic Resonance Analysis MR 4D Flow Visualization and Quantification of Aortic Blood Flow 4D Magnetic Resonance Analysis Pie Medical Imaging, manufacturer of Quantitative Analysis software for cardiology
Specific Standards of Accreditation for Residency Programs in General Surgery
 Specific Standards of Accreditation for Residency Programs in General Surgery 2010 INTRODUCTION The purpose of this document is to provide program directors and surveyors with an interpretation of the
Specific Standards of Accreditation for Residency Programs in General Surgery 2010 INTRODUCTION The purpose of this document is to provide program directors and surveyors with an interpretation of the
