RESEARCHERS REPORT NEW WAYS TO PREDICT THE DEVELOPMENT OF ALZHEIMER S DISEASE
|
|
|
- Kory Hart
- 8 years ago
- Views:
Transcription
1 CONTACT: Alzheimer s Association AAIC newsroom, , media@alz.org Niles Frantz, Alzheimer s Association, , niles.frantz@alz.org RESEARCHERS REPORT NEW WAYS TO PREDICT THE DEVELOPMENT OF ALZHEIMER S DISEASE Possible New Saliva Test Reported at Alzheimer s Association International Conference 2015 WASHINGTON, DC, July 19, 2015 Brain scans, memory tests and body fluids such as saliva may hold the keys to understanding a person s likelihood of developing Alzheimer s, even among those who don t yet have memory and thinking problems associated with the disease, suggest four studies reported at the Alzheimer s Association International Conference 2015 (AAIC 2015) in Washington, D.C. Two of the studies suggest that an excess of certain proteins in cerebrospinal fluid (CSF) are strong predictors of Alzheimer s, and that the accuracy of these predictions is stronger when they are considered together with other diagnostic tools, such as memory tests or MRI brain scans. A third report suggests that new ways of creating images of inflammation in the brain with PET scans could one day be used to identify treatments that protect the brain. Finally, a small but intriguing study suggests it could be possible to detect Alzheimer s-like changes in saliva, which is easily obtained, safe and affordable, but there is a lot of work still to be done. There is now consensus that Alzheimer s disease begins with changes in the brain that are happening while people are still cognitively normal, decades before memory and thinking problems begin, which then accelerate as the disease progresses, said Maria Carrillo, PhD, Alzheimer s Association Chief Science Officer. Still, diagnosis of Alzheimer s usually happens fairly late in the progression of the disease, typically not until symptoms are severe enough to prompt a visit to the doctor. Earlier diagnosis or, better still, the ability to predict the onset of Alzheimer s, would significantly increase the window of opportunity a person with Alzheimer s has to formulate an informed response to the news and empower them to be an active participant in decision-making while they still have the ability. It also would help researchers choose participants for treatment research, especially people at the earliest, presymptomatic stages of the disease who are needed for prevention trials, Carrillo said. The Alzheimer s Association 2015 Alzheimer s Disease Facts and Figures report found that only 45 percent of people diagnosed with Alzheimer s or their caregivers say they were told the diagnosis by their doctor. Research suggests that one of the reasons doctors do not disclose a diagnosis of Alzheimer's is they have insufficient time and resources to provide support to patients and caregivers at the time of the diagnosis. Extending the time between diagnosis and the onset of symptoms could change that situation. Six Factors May Identify People Most Likely to Get Alzheimer s Disease Research suggests that certain biological changes take place over time that signal the future onset of Alzheimer s disease symptoms in people with otherwise normal memory and thinking skills. Understanding which changes are most predictive of Alzheimer s may help in the selection of candidates for clinical trials and help monitor an individual s response to treatment or prevention strategies.
2 Most Alzheimer s clinical trials have been conducted among individuals in the dementia phase of the disease. More recently, trials have been initiated during an earlier phase, known as mild cognitive impairment (MCI), where people have a significantly increased risk of progressing to Alzheimer s disease dementia. For more than a decade, these trials have been unsuccessful in identifying new Alzheimer s medications. A few clinical trials have recently been initiated among individuals in the preclinical phase of the disease. However, there is limited data on which to base the selection of individuals for a clinical trial in the preclinical phase; there is a great need for better tools for earlier detection and cognitive assessment to identify these individuals and track their progress. To assess the usefulness of different predictive tests, Marilyn Albert, PhD, Director of the Division of Cognitive Neuroscience in the Department of Neurology at Johns Hopkins University School of Medicine and Director of the Johns Hopkins Alzheimer's Disease Research Center and colleagues repeatedly evaluated the mental status of 189 participants from the BIOCARD Study who were cognitively normal at the beginning of the study, They used a variety of measurement tools and identified those tools that best predicted the onset of MCI five years later. They found that the combined results of six measures were particularly useful: Two memory and thinking tests (the Digit Symbol and Paired Associates Immediate Recall tests). Levels of two different proteins in CSF (amyloid beta and p-tau). Two MRI brain scans one to assess the thickness of the right entorhinal cortex and another to measure the volume of the hippocampus, both of which are important for memory. Several statistical measures were used to characterize the accuracy of prediction, including the Area Under the Curve (AUC), sensitivity and specificity (AUC=0.886, sensitivity = 0.85, specificity = 0.70). Our study shows that up to five years before any Alzheimer s symptoms appear a small set of factors can tell us, with significant accuracy, which cognitively normal individuals will develop mild cognitive impairment due to Alzheimer s, said Albert. We hope that this information will be useful for designing clinical trials aimed at delaying the onset of symptoms among cognitively normal individuals. An approach such as ours could be used for determining which people might be most likely to benefit. Saliva Test May Identify Normal Aging, MCI, and Alzheimer s Early Results Early detection of Alzheimer s related symptoms is critically important for individuals with the disease and for clinical studies seeking to slow or stop disease progression. However, many diagnosis techniques can be costly or invasive. Saliva is simple to obtain, easily transportable, and has been successfully used in a variety of diseases and conditions. Since multiple samples can be readily obtained, saliva testing is particularly useful for performing repeated assessments that span days, weeks, months or longer. Knowing that Alzheimer s typically co-exists with certain metabolic disorders, Shraddha Sapkota, MSc, a neuroscience graduate student at the University of Alberta, Canada, and colleagues reported success at AAIC 2015 in identifying substances in saliva that differentiated among people with Alzheimer s disease (n=22), MCI (n=25) or normal aging (NA; n=35). A validation sample included 10 NA, 10 MCI, and 7 participants with Alzheimer s. The researchers conducted their study using saliva samples, clinical diagnoses and cognitive data from the Victoria Longitudinal Study (VLS), a long-term, large-scale investigation of human aging. Protein analysis technology, called liquid chromatography-mass spectrometry (LCMS), was used to analyze the saliva samples and identify which substances were predominant in the saliva of each of the three types of individuals. Linking that data back to each participant s clinical diagnosis, researchers reported strong associations between certain substances and a person s cognitive abilities. For example, higher levels of one substance in the MCI group and another in the Alzheimer s group were observed. When these were examined in NA, higher levels of both predicted worse episodic memory performance. Another substance with higher levels in the Alzheimer s group predicted slower speed in processing information. 2
3 Saliva is easily obtained, safe and affordable, and has promising potential for predicting and tracking cognitive decline, but we re in the very early stages of this work and much more research is needed, said Sapkota. Equally important is the possibility of using saliva to find targets for treatment to address the metabolic component of Alzheimer s, which is still not well understood. This study brings us closer to solving that mystery. Neurogranin, a CSF Biomarker for Synaptic Loss, Predicts Decline to Alzheimer s Dementia Cerebrospinal fluid (CSF) buffers and protects the brain and spinal cord; when the brain is injured or damaged, certain proteins are released into the CSF. Examining CSF has led to the discovery of proteins that are strongly linked to Alzheimer s and can be useful in assessing an individual s health status. One such protein is neurogranin. Neurogranin is a protein found only in the brain and is involved in the communication pathways between brain cells, known as synaptic signaling pathways. Synapse damage and loss is a common, early feature of Alzheimer's and there is a strong correlation between the extent of synapse loss and the severity of dementia. Recent research shows that neurogranin levels in the CSF are elevated in people with Alzheimer s. At AAIC 2015, Maartje Kester, MD, PhD of VU University Medical Center, Amsterdam, Netherlands and colleagues presented findings from a study of 162 individuals from the Amsterdam Dementia Cohort who were either cognitively normal, had MCI or Alzheimer s. All of the participants had two cerebral spinal fluid samples taken over the course of two years, allowing researchers to compare the protein content over time. Participants also had cognitive exams about four years apart, giving researchers a picture of how participants memory and thinking status changed over time. The research team found that baseline levels of neurogranin were significantly higher in individuals with Alzheimer s than in cognitively normal individuals. Initial neurogranin levels were also significantly higher in MCI individuals who progressed to Alzheimer s compared to those with stable MCI, and they were predictive of progression from MCI to Alzheimer s. They found that neurogranin levels were strongly correlated with two other Alzheimer s-related proteins, tau and ptau-181, in all three groups. The researchers also observed that while neurogranin increased slightly over time in the cognitively normal group, it did not in those with MCI or Alzheimer s. This may indicate that neurogranin levels in CSF reflect very early synaptic loss in Alzheimer s and may be useful for early detection, Kester said. We found that neurogranin is a potentially useful marker for the diagnosis, prognosis and monitoring of Alzheimer s, said Kester. Imaging Inflammation in the Brain Is It In Our Future? PET imaging compounds for amyloid plaques in the brain have been FDA approved for use in people with suspected Alzheimer s disease but an unclear diagnosis or an unusual presentation, and, as reported at AAIC 2014, significant advances are being made in PET imaging of tau tangles. These are the two hallmark brain lesions of Alzheimer s disease and are thought to be involved in its cause and progression. Inflammation is another condition/pathway being investigated for its role in Alzheimer s; it can be deadly to brain cells and may be activated by the plaques and tangles. Microglial cells, as the brain s immune cells, constitute the active immune defense in the central nervous system (CNS). They have the potential to either protect or when activated destroy critical links in the brain. Attempts have been made to develop treatments that keep the cells in a protective state. Andreas H. Jacobs, MD, Professor at the European Institute for Molecular Imaging, Muenster, Germany, and Director of the Department of Geriatrics at the Johanniter Hospital, Bonn, Germany, presenting at AAIC 2015 on behalf of his collaborators from the INMIND consortium, gave an overview of the stateof-the-art in PET imaging of inflammation in the brain. 3
4 To monitor the effects of new treatments on microglial cells, researchers often use positron emission tomography (PET) scans to visualize active microglial cells by tracking a protein that is produced at higher levels when the cells are active/destructive. Use of this technique is limited, however, because the relationship between this protein and microglial function is not fully understood yet, making interpretation of imaging results challenging. Consequently, other imaging targets are now under investigation. The data reported at AAIC demonstrates the potential of new target structures such as cannabinoid type 2, P2X7 and COX2 receptors to more accurately reflect various stages or types of the microglial cells, telling us if they re being destructive or protective, said Jacobs. It is our hope that these new imaging tools can help us assess the effectiveness of treatments that lessen their destructive behavior. About AAIC The Alzheimer s Association International Conference (AAIC) is the world s largest gathering of researchers from around the world focused on Alzheimer s and other dementias. As a part of the Alzheimer s Association s research program, AAIC serves as a catalyst for generating new knowledge about dementia and fostering a vital, collegial research community. AAIC 2015 home page: AAIC 2015 newsroom: About the Alzheimer s Association The Alzheimer s Association is the leading voluntary health organization in Alzheimer's care, support and research. Our mission is to eliminate Alzheimer s disease through the advancement of research, to provide and enhance care and support for all affected, and to reduce the risk of dementia through the promotion of brain health. Our vision is a world without Alzheimer s. Visit alz.org or call # # # Marilyn Albert, PhD, et al. Using Combinations of Variables to Identify Individuals with Preclinical AD. (Funders: U.S. National Institute on Aging) Shraddha Sapkota, MSc, et al. Metabolomics Analyses of Salivary Samples Discriminate Normal Aging, Mild Cognitive Impairment, and Alzheimer s Disease Groups and Produce Biomarkers Predictive of Neurocognitive Performance. (Funders: Canadian Institutes of Health Research, U.S. National Institutes of Health) Maartje Kester, MD, PhD, et al. Neurogranin, a CSF Biomarker for Synaptic Loss, Predicts Decline to Dementia Due to Alzheimer's Disease. (Funders: Alzheimer s Nederland, U.S. National Institute on Aging) Andreas Jacobs, MD, et al. Imaging Neuroinflammation in Neurodegenerative Diseases (INMIND) Concepts and Future Directions. (Funders: European Commission Directorate-General for Research and Innovation, European Union's Seventh Framework Programme for Research and Technological Development) 4
5 Proposal ID: F Featured Research Session, Sunday, July 19, 8-9:30 am, Ballroom C Theme Selection: Diagnosis and Prognosis Using Combinations of Variables to Identify Individuals with Preclinical AD FRS Topic: Longitudinal Study of Pre-Symptomatic Alzheimer s Disease Biomarkers: A Foundation for Research on Prevention. Marilyn Albert, PhD Johns Hopkins University School of Medicine, Baltimore, MD, USA malbert9@jhmi.edu Background: Several lines of evidence indicate that a range of measures obtained in cognitively normal individuals are significantly related to time to onset of clinical symptoms of Alzheimer s disease (AD) that develop many years later. This suggests that combinations of biomarkers might be useful for selecting individuals to include in clinical trials and for assessing disease progression in response to treatment. Methods: Using data from the BIOCARD study, which includes serial assessment of CSF, MRI and cognitive measures, we used time-dependent ROC methods to examine combinations of measures that were significant predictors of which cognitively normal individuals had a high likelihood of progressing to mild cognitive impairment (MCI) due to AD over time. The best model was selected using the AIC criterion. Results: Using data from baseline in 189 participants, and predicting onset of clinical symptoms up to 5 years later, sets of biomarkers were examined using several approaches: (1) the least costly to the most costly, (2) the least invasive to the most invasive, and (3) the best fit model. The best fit model included 6 variables: two cognitive measures (Digit Symbol and Paired Associates Immediate Recall), two CSF measures (Abeta and ptau) and two MRI measures (right entorhinal cortex thickness and right hippocampal volume), adjusted for demographics (AUC=0.886, sensitivity = 0.85, specificity = 0.70). Moreover, the addition of each domain (cognitive + CSF + MRI) added significantly to the accuracy of prediction. Conclusions: Combinations of biomarkers obtained at least 5 years prior to symptom onset can be used to identify which cognitively normal individuals will progress to MCI due to AD. Additionally, these findings demonstrate that biomarkers useful for prediction later in the disease course are useful in preclinical AD. Clinical trials aimed at delaying the onset of symptoms could utilize such an approach for determining which individuals might be most likely to benefit from inclusion in a clinical trial aimed at individuals with preclinical AD and/or tracking response to treatment. 5
6 Proposal ID: P3-090 Poster, Thursday, July 21 Theme Selection: Diagnosis and Prognosis Metabolomics Analyses of Salivary Samples Discriminate Normal Aging, Mild Cognitive Impairment, and Alzheimer s Disease Groups and Produce Biomarkers Predictive of Neurocognitive Performance Shraddha Sapkota, MSc University of Alberta, Edmonton AB, Canada ssapkota@ualberta.ca Background: Metabolomics is a global approach to detecting perturbations in metabolic pathways that can reflect early and subtle disease-related changes in the central nervous system. The metabolome, the end product of gene-environment interactions, can characterize and discriminate metabolic signatures of Alzheimer s disease (AD) and Mild Cognitive Impairment (MCI). We previously established the technology for using salivary samples for discriminating normal cognitive aging (NA) from MCI groups. We report pairwise metabolomics comparisons among new NA, MCI, and AD groups. Three main goals were to (1) fully discriminate the groups based on metabolomics signatures, (2) identify top discriminative metabolites, and (3) test these metabolites as biomarkers predictive of neurocognitive performance. Methods: Salivary specimens, validated clinical classifications, and cognitive data were collected on two sets of three groups from the Victoria Longitudinal Study. The discovery sample (64% female) included NA (n=35; M age=69.94 years), MCI (n=25; M age=70.40 years), and AD (n=22; M age=77.09 years), whereas the validation sample included 10 (NA), 10 (MCI), and 7 (AD) participants (N=109). We used the established 13C/12C isotope dansylation labeling technique and liquid chromatography-mass spectrometry to identify metabolite biomarkers. The top pool of discriminant metabolites were identified using orthogonal partial least squares discriminant analyses (OPLS-DA) and Variable Importance in Projection indices (>1.5), Fold Change (>2.0), and goodness of fit (R2Y (cum)>0.9). Regression analyses (Mplus) determined biomarker-cognition associations for the top discriminative metabolites. Results: First, M=1515 metabolites was detected in each pairwise comparison. Second, OPLS-DA revealed clear discrimination between all groups, and volcano plots identified top discriminant metabolites between each pairs. Third, a pattern of predictably directional metabolite-cognition associations were identified. For example, one metabolite was upregulated in the MCI group (log2 (FC)=1.11;AUC=0.732) and another metabolite (Trp Glu;Glu Trp) (log2(fc)=1.87;auc=0.866) was upregulated in AD and, when examined as biomarkers in NA, higher levels of both predicted worse episodic memory performance. Another metabolite (Tyr-Asn-Ser) (log2(fc)=1.24;auc=0.826) was upregulated in AD and higher levels in predicted slower neurocognitive speed in the AD group. Conclusions: Metabolomics analyses can produce clinical discrimination, biological pathway information, and predict biomarker-cognition associations. Saliva is a non-invasive biofluid with promising biomarker translation implications in Alzheimer s research. 6
7 Proposal ID: O Oral Session, Thursday, July 23, 11:30 am 1 pm, #207 Theme Selection: Diagnosis and Prognosis Neurogranin, a CSF Biomarker for Synaptic Loss, Predicts Decline to Dementia Due to Alzheimer's Disease Maartje Kester, MD, PhD VU University Medical Center, Amsterdam, Netherlands m.kester@vumc.nl Background: We examined the utility of a novel cerebrospinal fluid (CSF) biomarker of synaptic loss, neurogranin, for diagnosis, prognosis, and longitudinal monitoring of Alzheimer s disease (AD). Methods: CSF levels of neurogranin were measured in 37 cognitively normal (64(2)y, f38%, MMSE 28(0.3)), 61 subjects with mild cognitive impairment (MCI; 68(1)y, f38%, MMSE 27(0.3)), and 65 ADpatients (65(1)y, f45%, MMSE 22(0.7)) from the Amsterdam Dementia Cohort who underwent two lumbar punctures (mean (SE) interval of 2.0 (0.1) years). Mean (SE) duration of cognitive follow-up was 3.8 (0.2) years. We used ANOVA (log-transformed CSF biomarker levels) to assess baseline differences, and Cox-regression (CSF biomarker levels in tertiles) to predict progression to AD in MCI. Linear Mixed Models were used to assess within-person annual change in neurogranin levels. All analyses were adjusted for sex and age. Results: Baseline levels of neurogranin in AD were higher than in cognitively normal individuals (median (IQR) 2381 ( )vs ( ), p=0.04). Baseline neurogranin levels were strongly correlated with tau and ptau-181 in all clinical groups (all: Spearman r>0.77, p<0.001), but not to Aβ42. Baseline neurogranin levels were also higher in MCI individuals who progressed to AD compared to those with stable MCI (median (IQR) 2842 ( ) vs ( ), p=0.004), and they were predictive of progression from MCI to AD (HR [95% CI] 1.8 [ ]). Linear Mixed Model analyses demonstrated that within-person levels of neurogranin increased in the cognitively normal group (mean (SE) 90 (45) pg/ml per year, p<0.05), but not in those with MCI or AD. Conclusions: Neurogranin is a promising biomarker for AD, as levels were elevated in AD compared to cognitively normal individuals and predicted progression from MCI to AD. Within-person increases in neurogranin in cognitively normal individuals, but not those with later stage MCI or AD, suggest that neurogranin reflects early synaptic dysfunction or loss. 7
8 Proposal ID: F Featured Research Session, Sunday, July 19, 8-9:30 am, Ballroom A Theme Selection: Basic and Translational Science Imaging Neuroinflammation in Neurodegenerative Diseases (INMIND) Concepts and Future Directions FRS Topic: Innate Immunity and Alzheimer's Disease Andreas Jacobs, MD European Institute for Molecular Imaging, Munster, Germany ahjacobs@uni-muenster.de Background: Positron Emission Tomography (PET) is one of the key molecular imaging technologies which allows the location and quantification of the expression of molecular target structures in vivo by employing specific radiopharmaceuticals. PET allows the translational and reverse-translational assessment of disease-specific molecular alterations over time, during the disease course and under therapeutic intervention. The aim of the lecture is to demonstrate (i) the current state-of-the-art of tracer technology for assessing microglial activation and (ii) implementation of old and new microglial imaging strategies in various disease models and human application. Methods: For 30 years, imaging the activation of microglia cells in vivoby PET is accomplished by employing the mitochondrial translocator protein 18kDa (TSPO) as molecular target. TSPO is overexpressed by activated microglial cells, and in some conditions also by astrocytes. Over the last decade, about 50 radiopharmaceuticals targeting TSPO have been developed and investigated, only some of them have reached clinical application so far. As the biology of TSPO in relation to microglial function is not fully understood yet, interpretation of TSPO-based imaging results is sometimes difficult. Therefore, new microglial target structures such as cannabinoid type 2, P2X7, and COX2 receptors, respectively, are all being explored for imaging. Results: All research data presented originate from joint efforts within the INMIND consortium. Conclusions: The ultimate goal is to differentiate various microglial phenotypes (such as M1 and M2) during the disease course for the overall aim to establish imaging-guided therapeutic modification of microglia function to enhance their neuroprotective and repress their neurodestructive function. # # # 8
GOING BEYOND RISK REDUCTION: PHYSICAL EXERCISE MAY BE AN EFFECTIVE TREATMENT FOR ALZHEIMER S DISEASE AND VASCULAR DEMENTIA
 CONTACT: Alzheimer s Association AAIC newsroom, 202-249-4002, media@alz.org Niles Frantz, Alzheimer s Association, 312-335-5777, niles.frantz@alz.org GOING BEYOND RISK REDUCTION: PHYSICAL EXERCISE MAY
CONTACT: Alzheimer s Association AAIC newsroom, 202-249-4002, media@alz.org Niles Frantz, Alzheimer s Association, 312-335-5777, niles.frantz@alz.org GOING BEYOND RISK REDUCTION: PHYSICAL EXERCISE MAY
Primary Endpoints in Alzheimer s Dementia
 Primary Endpoints in Alzheimer s Dementia Dr. Karl Broich Federal Institute for Drugs and Medical Devices (BfArM) Kurt-Georg-Kiesinger-Allee 38, D-53175 Bonn Germany Critique on Regulatory Decisions in
Primary Endpoints in Alzheimer s Dementia Dr. Karl Broich Federal Institute for Drugs and Medical Devices (BfArM) Kurt-Georg-Kiesinger-Allee 38, D-53175 Bonn Germany Critique on Regulatory Decisions in
Local Clinical Trials
 Local Clinical Trials The Alzheimer s Association, Connecticut Chapter does not officially endorse any specific research study. The following information regarding clinical trials is provided as a service
Local Clinical Trials The Alzheimer s Association, Connecticut Chapter does not officially endorse any specific research study. The following information regarding clinical trials is provided as a service
PDF hosted at the Radboud Repository of the Radboud University Nijmegen
 PDF hosted at the Radboud Repository of the Radboud University Nijmegen The following full text is an author's version which may differ from the publisher's version. For additional information about this
PDF hosted at the Radboud Repository of the Radboud University Nijmegen The following full text is an author's version which may differ from the publisher's version. For additional information about this
A Support System for Diagnosis of Dementia, Alzheimer or Mild Cognitive Impairment
 Toronto, November 4, 2013 04:00 pm 05:30 pm-4th Oral Session A Support System for Diagnosis of Dementia, Alzheimer or Mild Cognitive Impairment Flávio L. Seixas Aura Conci Débora C. Muchaluat Saade Bianca
Toronto, November 4, 2013 04:00 pm 05:30 pm-4th Oral Session A Support System for Diagnosis of Dementia, Alzheimer or Mild Cognitive Impairment Flávio L. Seixas Aura Conci Débora C. Muchaluat Saade Bianca
Biomarkers for Alzheimer's Disease in Down Syndrome
 Biomarkers for Alzheimer's Disease in Down Syndrome Brad Christian, Ph.D. Waisman Laboratory for Brain Imaging Outline Rationale for Studying AD in Down Syndrome Background of Alzheimer s Disease Biomarkers
Biomarkers for Alzheimer's Disease in Down Syndrome Brad Christian, Ph.D. Waisman Laboratory for Brain Imaging Outline Rationale for Studying AD in Down Syndrome Background of Alzheimer s Disease Biomarkers
Diseases of the Nervous System. Neal G. Simon, Ph.D. Professor, Dept of Biological Sciences Lehigh University
 Diseases of the Nervous System Neal G. Simon, Ph.D. Professor, Dept of Biological Sciences Lehigh University Outline A. Stress-related Disorders 1. Emotional Circuitry: Key Components 2. The Hypothalamic
Diseases of the Nervous System Neal G. Simon, Ph.D. Professor, Dept of Biological Sciences Lehigh University Outline A. Stress-related Disorders 1. Emotional Circuitry: Key Components 2. The Hypothalamic
MCDB 4777/5777 Molecular Neurobiology Lecture 38 Alzheimer s Disease
 MCDB 4777/5777 Molecular Neurobiology Lecture 38 Alzheimer s Disease Outline of Today s Lecture Why is Alzheimer s disease a problem? What is Alzheimer s Disease? What causes Alzheimer s disease? How can
MCDB 4777/5777 Molecular Neurobiology Lecture 38 Alzheimer s Disease Outline of Today s Lecture Why is Alzheimer s disease a problem? What is Alzheimer s Disease? What causes Alzheimer s disease? How can
Is there a Distinct Phenotype to Memory Loss in Alzheimer's Disease?
 Is there a Distinct Phenotype to Memory Loss in Alzheimer's Disease? David A. Wolk, M.D. Assistant Director Penn Memory Center Assistant Professor of Neurology University of Pennsylvania 5 Million Clinical
Is there a Distinct Phenotype to Memory Loss in Alzheimer's Disease? David A. Wolk, M.D. Assistant Director Penn Memory Center Assistant Professor of Neurology University of Pennsylvania 5 Million Clinical
FSH Society s 2014 Biennial FSHD Connect Meeting: Natural History Studies
 FSH Society s 2014 Biennial FSHD Connect Meeting: Natural History Studies Raymond A. Huml, MS, DVM, RAC Executive Director, Head, Global Biosimilars Business Development and Strategic Planning, Quintiles
FSH Society s 2014 Biennial FSHD Connect Meeting: Natural History Studies Raymond A. Huml, MS, DVM, RAC Executive Director, Head, Global Biosimilars Business Development and Strategic Planning, Quintiles
Running Head: THE DETECTION OF ALZHEIMER S DISEASE: A LITERATURE 1. The Detection of Alzheimer s Disease: A Literature Review and Case Study
 Running Head: THE DETECTION OF ALZHEIMER S DISEASE: A LITERATURE 1 The Detection of Alzheimer s Disease: A Literature Review and Case Study November 15, 2012 Detection of Alzheimer s Disease 2 Abstract
Running Head: THE DETECTION OF ALZHEIMER S DISEASE: A LITERATURE 1 The Detection of Alzheimer s Disease: A Literature Review and Case Study November 15, 2012 Detection of Alzheimer s Disease 2 Abstract
Retinal Imaging Biomarkers for Early Diagnosis of Alzheimer s Disease
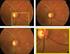 Retinal Imaging Biomarkers for Early Diagnosis of Alzheimer s Disease Eleonora (Nora) Lad, MD, PhD Assistant Professor of Ophthalmology, Vitreoretinal diseases Duke Center for Macular Diseases Duke University
Retinal Imaging Biomarkers for Early Diagnosis of Alzheimer s Disease Eleonora (Nora) Lad, MD, PhD Assistant Professor of Ophthalmology, Vitreoretinal diseases Duke Center for Macular Diseases Duke University
Alzheimer s disease. What is Alzheimer s disease?
 Alzheimer s disease What is Alzheimer s disease? What we know about dementia and Alzheimer s disease Alzheimer s disease is the most common of a large group of disorders known as dementias. It is an irreversible
Alzheimer s disease What is Alzheimer s disease? What we know about dementia and Alzheimer s disease Alzheimer s disease is the most common of a large group of disorders known as dementias. It is an irreversible
How To Change Medicine
 P4 Medicine: Personalized, Predictive, Preventive, Participatory A Change of View that Changes Everything Leroy E. Hood Institute for Systems Biology David J. Galas Battelle Memorial Institute Version
P4 Medicine: Personalized, Predictive, Preventive, Participatory A Change of View that Changes Everything Leroy E. Hood Institute for Systems Biology David J. Galas Battelle Memorial Institute Version
Optic Neuritis. The optic nerve fibers are coated with myelin to help them conduct the electrical signals back to your brain.
 Optic Neuritis Your doctor thinks that you have had an episode of optic neuritis. This is the most common cause of sudden visual loss in a young patient. It is often associated with discomfort in or around
Optic Neuritis Your doctor thinks that you have had an episode of optic neuritis. This is the most common cause of sudden visual loss in a young patient. It is often associated with discomfort in or around
Chapter 10. Summary & Future perspectives
 Summary & Future perspectives 123 Multiple sclerosis is a chronic disorder of the central nervous system, characterized by inflammation and axonal degeneration. All current therapies modulate the peripheral
Summary & Future perspectives 123 Multiple sclerosis is a chronic disorder of the central nervous system, characterized by inflammation and axonal degeneration. All current therapies modulate the peripheral
2012 Medical School for Actuaries Nov. 6-7, 2012 Session #1: Alzheimer s Disease
 2012 Medical School for Actuaries Nov. 6-7, 2012 Session #1: Alzheimer s Disease Dylan Wint, M.D. ALZHEIMER DISEASE Dylan Wint, M.D. Lou Ruvo Center for Brain Health DEFINITIONS Cognitive related to thinking,
2012 Medical School for Actuaries Nov. 6-7, 2012 Session #1: Alzheimer s Disease Dylan Wint, M.D. ALZHEIMER DISEASE Dylan Wint, M.D. Lou Ruvo Center for Brain Health DEFINITIONS Cognitive related to thinking,
Scandinavian Bio-Business Seminar, Zürich. Presentation 10 February 2016 Håkon Sæterøy, CEO
 Scandinavian Bio-Business Seminar, Zürich Presentation 10 February 2016 Håkon Sæterøy, CEO Clinical need 75 m patients WW A global estimate of 75m patients with Mild Cognitive Impairment (MCI) who may
Scandinavian Bio-Business Seminar, Zürich Presentation 10 February 2016 Håkon Sæterøy, CEO Clinical need 75 m patients WW A global estimate of 75m patients with Mild Cognitive Impairment (MCI) who may
GE Global Research. The Future of Brain Health
 GE Global Research The Future of Brain Health mission statement We will know the brain as well as we know the body. Future generations won t have to face Alzheimer s, TBI and other neurological diseases.
GE Global Research The Future of Brain Health mission statement We will know the brain as well as we know the body. Future generations won t have to face Alzheimer s, TBI and other neurological diseases.
ANXIETY & COGNITIVE IMPAIRMENT
 ANXIETY & COGNITIVE IMPAIRMENT Dr. Sherri Hayden, Ph.D., R. Psych. Neuropsychologist, UBC Hospital Clinic for Alzheimer Disease & Related Disorders Clinical Assistant Professor, UBC Department of Medicine,
ANXIETY & COGNITIVE IMPAIRMENT Dr. Sherri Hayden, Ph.D., R. Psych. Neuropsychologist, UBC Hospital Clinic for Alzheimer Disease & Related Disorders Clinical Assistant Professor, UBC Department of Medicine,
Asymptomatic HIV-associated Neurocognitive Disorder (ANI) Increases Risk for Future Symptomatic Decline: A CHARTER Longitudinal Study
 Asymptomatic HIV-associated Neurocognitive Disorder (ANI) Increases Risk for Future Symptomatic Decline: A CHARTER Longitudinal Study Robert Heaton, PhD 1, Donald Franklin, BS 1, Steven Woods, PsyD 1,
Asymptomatic HIV-associated Neurocognitive Disorder (ANI) Increases Risk for Future Symptomatic Decline: A CHARTER Longitudinal Study Robert Heaton, PhD 1, Donald Franklin, BS 1, Steven Woods, PsyD 1,
J D R F R E Q U E S T S L E T T E R S O F I N T E N T F O R : B I O M AR K E R S O F P AN C R E A T I C B E T A C E L L S T R E S S AN D H E AL T H
 J D R F R E Q U E S T S L E T T E R S O F I N T E N T F O R : B I O M AR K E R S O F P AN C R E A T I C B E T A C E L L S T R E S S AN D H E AL T H PURPOSE JDRF, the world s leading non-profit organization
J D R F R E Q U E S T S L E T T E R S O F I N T E N T F O R : B I O M AR K E R S O F P AN C R E A T I C B E T A C E L L S T R E S S AN D H E AL T H PURPOSE JDRF, the world s leading non-profit organization
Steps to getting a diagnosis: Finding out if it s Alzheimer s Disease.
 Steps to getting a diagnosis: Finding out if it s Alzheimer s Disease. Memory loss and changes in mood and behavior are some signs that you or a family member may have Alzheimer s disease. If you have
Steps to getting a diagnosis: Finding out if it s Alzheimer s Disease. Memory loss and changes in mood and behavior are some signs that you or a family member may have Alzheimer s disease. If you have
2.1 Who first described NMO?
 History & Discovery 54 2 History & Discovery 2.1 Who first described NMO? 2.2 What is the difference between NMO and Multiple Sclerosis? 2.3 How common is NMO? 2.4 Who is affected by NMO? 2.1 Who first
History & Discovery 54 2 History & Discovery 2.1 Who first described NMO? 2.2 What is the difference between NMO and Multiple Sclerosis? 2.3 How common is NMO? 2.4 Who is affected by NMO? 2.1 Who first
Participating in Alzheimer s Disease Clinical Trials and Studies
 Participating in Alzheimer s Disease Clinical Trials and Studies FACT SHEET When Margaret was diagnosed with earlystage Alzheimer s disease at age 68, she wanted to do everything possible to combat the
Participating in Alzheimer s Disease Clinical Trials and Studies FACT SHEET When Margaret was diagnosed with earlystage Alzheimer s disease at age 68, she wanted to do everything possible to combat the
Primary Care Update January 28 & 29, 2016 Alzheimer s Disease and Mild Cognitive Impairment
 Primary Care Update January 28 & 29, 2016 Alzheimer s Disease and Mild Cognitive Impairment Kinga Szigeti, MD Associate Professor UBMD Neurology UB Department of Neurology Questions How do we differentiate
Primary Care Update January 28 & 29, 2016 Alzheimer s Disease and Mild Cognitive Impairment Kinga Szigeti, MD Associate Professor UBMD Neurology UB Department of Neurology Questions How do we differentiate
PROMISING NEW TREATMENTS FOR ALZHEIMER S TARGET MULTIPLE DISEASE-RELATED PROTEINS, MAY HAVE BENEFITS FOR SEVERAL BRAIN DISEASES
 CONTACT: Alzheimer s Association AAIC newsroom, 202-249-4002, media@alz.org Niles Frantz, Alzheimer s Association, 312-363-8782, niles.frantz@alz.org PROMISING NEW TREATMENTS FOR ALZHEIMER S TARGET MULTIPLE
CONTACT: Alzheimer s Association AAIC newsroom, 202-249-4002, media@alz.org Niles Frantz, Alzheimer s Association, 312-363-8782, niles.frantz@alz.org PROMISING NEW TREATMENTS FOR ALZHEIMER S TARGET MULTIPLE
Frequently Asked Questions
 Frequently Asked Questions Business Office: 598 Airport Boulevard Suite 1400 Morrisville NC 27560 Contact: support@cognitrax.com Phone: 888.750.6941 Fax: 888.650.6795 www.cognitrax.com Diseases of the
Frequently Asked Questions Business Office: 598 Airport Boulevard Suite 1400 Morrisville NC 27560 Contact: support@cognitrax.com Phone: 888.750.6941 Fax: 888.650.6795 www.cognitrax.com Diseases of the
Surgery in Individuals Age 65+ Possible Risks. Possible Benefits. Potential Causes of POCD 11/24/2014. What is POCD?
 Surgery in Individuals Age 65+ Postoperative Cognitive Dysfunction in Older Adults Ryan W. Schroeder, Psy.D., LP, ABPP-CN Neuropsychologist & Assistant Professor University of Kansas School of Medicine
Surgery in Individuals Age 65+ Postoperative Cognitive Dysfunction in Older Adults Ryan W. Schroeder, Psy.D., LP, ABPP-CN Neuropsychologist & Assistant Professor University of Kansas School of Medicine
ß-interferon and. ABN Guidelines for 2007 Treatment of Multiple Sclerosis with. Glatiramer Acetate
 ABN Guidelines for 2007 Treatment of Multiple Sclerosis with ß-interferon and Glatiramer Acetate Published by the Association of British Neurologists Ormond House, 27 Boswell Street, London WC1N 3JZ Contents
ABN Guidelines for 2007 Treatment of Multiple Sclerosis with ß-interferon and Glatiramer Acetate Published by the Association of British Neurologists Ormond House, 27 Boswell Street, London WC1N 3JZ Contents
American Society of Addiction Medicine
 American Society of Addiction Medicine Public Policy Statement On Drug Testing as a Component of Addiction Treatment and Monitoring Programs and in other Clinical Settings [Note: ASAM also has a Public
American Society of Addiction Medicine Public Policy Statement On Drug Testing as a Component of Addiction Treatment and Monitoring Programs and in other Clinical Settings [Note: ASAM also has a Public
Social Security Disability Insurance and young onset dementia: A guide for employers and employees
 Social Security Disability Insurance and young onset dementia: A guide for employers and employees What is Social Security Disability Insurance? Social Security Disability Insurance (SSDI) is a payroll
Social Security Disability Insurance and young onset dementia: A guide for employers and employees What is Social Security Disability Insurance? Social Security Disability Insurance (SSDI) is a payroll
Changing the Trajectory of Alzheimer s Disease: How a Treatment by 2025 Saves Lives and Dollars
 Changing the Trajectory of Alzheimer s Disease: How a Treatment by 2025 Saves Lives and Dollars The appendices for this report including a detailed description of the model developed by The Lewin Group;
Changing the Trajectory of Alzheimer s Disease: How a Treatment by 2025 Saves Lives and Dollars The appendices for this report including a detailed description of the model developed by The Lewin Group;
Normal and Abnormal Aging and the Brain. Joel Kramer, PsyD Saul Villeda, PhD Kristine Yaffe, MD
 Normal and Abnormal Aging and the Brain Joel Kramer, PsyD Saul Villeda, PhD Kristine Yaffe, MD The myth of cognitive decline The myth of cognitive decline Individual change varies Individual change varies
Normal and Abnormal Aging and the Brain Joel Kramer, PsyD Saul Villeda, PhD Kristine Yaffe, MD The myth of cognitive decline The myth of cognitive decline Individual change varies Individual change varies
1695 N.W. 9th Avenue, Suite 3302H Miami, FL. 33136. Days and Hours: Monday Friday 8:30a.m. 6:00p.m. (305) 355 9028 (JMH, Downtown)
 UNIVERSITY OF MIAMI, LEONARD M. MILLER SCHOOL OF MEDICINE CLINICAL NEUROPSYCHOLOGY UHEALTH PSYCHIATRY AT MENTAL HEALTH HOSPITAL CENTER 1695 N.W. 9th Avenue, Suite 3302H Miami, FL. 33136 Days and Hours:
UNIVERSITY OF MIAMI, LEONARD M. MILLER SCHOOL OF MEDICINE CLINICAL NEUROPSYCHOLOGY UHEALTH PSYCHIATRY AT MENTAL HEALTH HOSPITAL CENTER 1695 N.W. 9th Avenue, Suite 3302H Miami, FL. 33136 Days and Hours:
Translational research facilitating experimental medicine in dementia in the UK
 Translational research facilitating experimental medicine in dementia in the UK Simon Lovestone Director Biomedical Research Unit for dementia at Maudsley and King s Route Map for Dementia Research June
Translational research facilitating experimental medicine in dementia in the UK Simon Lovestone Director Biomedical Research Unit for dementia at Maudsley and King s Route Map for Dementia Research June
The Nuts and Bolts of Multiple Sclerosis. Rebecca Milholland, M.D., Ph.D. Center for Neurosciences
 The Nuts and Bolts of Multiple Sclerosis Rebecca Milholland, M.D., Ph.D. Center for Neurosciences Objectives Discuss which patients are at risk for Multiple Sclerosis Discuss the diagnostic criteria for
The Nuts and Bolts of Multiple Sclerosis Rebecca Milholland, M.D., Ph.D. Center for Neurosciences Objectives Discuss which patients are at risk for Multiple Sclerosis Discuss the diagnostic criteria for
NEW HYBRID IMAGING TECHNOLOGY MAY HAVE BIG POTENTIAL FOR IMPROVING DIAGNOSIS OF PROSTATE CANCER
 Media Release April 7, 2009 For Immediate Release NEW HYBRID IMAGING TECHNOLOGY MAY HAVE BIG POTENTIAL FOR IMPROVING DIAGNOSIS OF PROSTATE CANCER London, Ontario Improved hybrid imaging techniques developed
Media Release April 7, 2009 For Immediate Release NEW HYBRID IMAGING TECHNOLOGY MAY HAVE BIG POTENTIAL FOR IMPROVING DIAGNOSIS OF PROSTATE CANCER London, Ontario Improved hybrid imaging techniques developed
Cognitive Rehabilitation for Executive Dysfunction in Parkinson s Disease
 Calleo, J., Burrows, C., Levin, H., Marsh, L., Lai, E., York, M. (2012). Cognitive rehabilitation for executive dysfunction in Parkinson s disease: application and current directions., vol. 2012, Article
Calleo, J., Burrows, C., Levin, H., Marsh, L., Lai, E., York, M. (2012). Cognitive rehabilitation for executive dysfunction in Parkinson s disease: application and current directions., vol. 2012, Article
A Definition of Multiple Sclerosis
 English 182 READING PRACTICE by Alyx Meltzer, Spring 2009 Vocabulary Preview (see bolded, underlined words) gait: (n) a particular way of walking transient: (adj) temporary; synonym = transitory remission:
English 182 READING PRACTICE by Alyx Meltzer, Spring 2009 Vocabulary Preview (see bolded, underlined words) gait: (n) a particular way of walking transient: (adj) temporary; synonym = transitory remission:
CHAPTER- 6. Okadaic acid induced neurotoxicity leads to central cholinergic dysfunction in rats. 1. Introduction. 2. Methods
 CHAPTER- 6 Okadaic acid induced neurotoxicity leads to central cholinergic dysfunction in rats 1. Introduction Neurodegenerative disorders, such as AD are often characterized by the degeneration of the
CHAPTER- 6 Okadaic acid induced neurotoxicity leads to central cholinergic dysfunction in rats 1. Introduction Neurodegenerative disorders, such as AD are often characterized by the degeneration of the
GYMR A4 Study Long Form Film
 VIDEO INTRO GRAPHICS GRAPHIC DR. SPERLING AUDIO 0:00 [Music & Graphics Only] 0:05 The A4 Study is a landmark clinical trial to prevent the memory loss associated with Alzheimer s disease. 0:10 Thank you
VIDEO INTRO GRAPHICS GRAPHIC DR. SPERLING AUDIO 0:00 [Music & Graphics Only] 0:05 The A4 Study is a landmark clinical trial to prevent the memory loss associated with Alzheimer s disease. 0:10 Thank you
Summary chapter 2 chapter 2
 Summary Multiple Sclerosis (MS) is a chronic disease of the brain and the spinal cord. The cause of MS is unknown. MS usually starts in young adulthood. In the course of the disease progression of neurological
Summary Multiple Sclerosis (MS) is a chronic disease of the brain and the spinal cord. The cause of MS is unknown. MS usually starts in young adulthood. In the course of the disease progression of neurological
CBT IN THE CITY. adjusted to the news of being with MS? April 2013. Experts at your fingertips call now. Check out our new services in you local area
 April 2013 Experts at your fingertips call now CBT IN THE CITY Check out our new services in you local area contents. A message from Susie, Information Multiple Sclerosis CBT can make a difference on the
April 2013 Experts at your fingertips call now CBT IN THE CITY Check out our new services in you local area contents. A message from Susie, Information Multiple Sclerosis CBT can make a difference on the
Enabling Discovery, Development, and translation of treatments for Cognitive Dysfunctions in Depression: A Workshop Session IV
 Enabling Discovery, Development, and translation of treatments for Cognitive Dysfunctions in Depression: A Workshop Session IV Maria Isaac, MASc, MD, PhD, MFPM, Psychiatrist Senior Scientific Officer Institute
Enabling Discovery, Development, and translation of treatments for Cognitive Dysfunctions in Depression: A Workshop Session IV Maria Isaac, MASc, MD, PhD, MFPM, Psychiatrist Senior Scientific Officer Institute
Alzheimer s & Dementia 7 (2011) 280 292
 Alzheimer s & Dementia 7 (2011) 280 292 Toward defining the preclinical stages of Alzheimer s disease: Recommendations from the National Institute on Aging-Alzheimer s Association workgroups on diagnostic
Alzheimer s & Dementia 7 (2011) 280 292 Toward defining the preclinical stages of Alzheimer s disease: Recommendations from the National Institute on Aging-Alzheimer s Association workgroups on diagnostic
Seminar/Talk Calendar
 Seminar/Talk Calendar Tuesday, February 3rd Dr. John Neumaier, Professor of Psychiatry and Pharmacology, University of Washington DREADDing Addiction Dr. Neumaier s laboratory is studying stress and addiction
Seminar/Talk Calendar Tuesday, February 3rd Dr. John Neumaier, Professor of Psychiatry and Pharmacology, University of Washington DREADDing Addiction Dr. Neumaier s laboratory is studying stress and addiction
Testimony of Timothy Coetzee, Ph.D. Representative of a Patient Advocacy Organization President, Fast Forward LLC
 Testimony of Timothy Coetzee, Ph.D. Representative of a Patient Advocacy Organization President, Fast Forward LLC Committee on Oversight and Government Reform Domestic Policy Subcommittee U.S. House of
Testimony of Timothy Coetzee, Ph.D. Representative of a Patient Advocacy Organization President, Fast Forward LLC Committee on Oversight and Government Reform Domestic Policy Subcommittee U.S. House of
CRITERIA FOR AD DEMENTIA June 11, 2010
 CRITERIA F AD DEMENTIA June 11, 2010 Alzheimer s Disease Dementia Workgroup Guy McKhann, Johns Hopkins University (Chair) Bradley Hyman, Massachusetts General Hospital Clifford Jack, Mayo Clinic Rochester
CRITERIA F AD DEMENTIA June 11, 2010 Alzheimer s Disease Dementia Workgroup Guy McKhann, Johns Hopkins University (Chair) Bradley Hyman, Massachusetts General Hospital Clifford Jack, Mayo Clinic Rochester
Clinical Trials of Disease Modifying Treatments
 MS CENTER CLINICAL RESEARCH The UCSF MS Center is an internationally recognized leader in multiple sclerosis clinical research. We conduct clinical trials involving the use of experimental treatments,
MS CENTER CLINICAL RESEARCH The UCSF MS Center is an internationally recognized leader in multiple sclerosis clinical research. We conduct clinical trials involving the use of experimental treatments,
Preventing Dementia: The Depression-Diabetes Nexus
 Preventing Dementia: The Depression-Diabetes Nexus Roger S McIntyre Assoc. Professor of Psychiatry and Pharmacology, University of Toronto Head, Mood Disorders Psychopharmacology Unit, University Health
Preventing Dementia: The Depression-Diabetes Nexus Roger S McIntyre Assoc. Professor of Psychiatry and Pharmacology, University of Toronto Head, Mood Disorders Psychopharmacology Unit, University Health
Depression in Older Persons
 Depression in Older Persons How common is depression in later life? Depression affects more than 6.5 million of the 35 million Americans aged 65 or older. Most people in this stage of life with depression
Depression in Older Persons How common is depression in later life? Depression affects more than 6.5 million of the 35 million Americans aged 65 or older. Most people in this stage of life with depression
Alzheimer s and Depression: What is the Connection?
 Alzheimer s and Depression: What is the Connection? Ladson Hinton MD Professor and Director of Geriatric Psychiatry Department of Psychiatry and Behavioral Sciences Director, Education Core, Alzheimer
Alzheimer s and Depression: What is the Connection? Ladson Hinton MD Professor and Director of Geriatric Psychiatry Department of Psychiatry and Behavioral Sciences Director, Education Core, Alzheimer
Clinically isolated syndrome (CIS)
 Clinically isolated syndrome (CIS) Spirella Building, Letchworth, SG6 4ET 01462 476700 www.mstrust.org.uk reg charity no. 1088353 We hope you find the information in this factsheet helpful. If you would
Clinically isolated syndrome (CIS) Spirella Building, Letchworth, SG6 4ET 01462 476700 www.mstrust.org.uk reg charity no. 1088353 We hope you find the information in this factsheet helpful. If you would
Multiple Sclerosis & MS Ireland Media Fact Sheet
 Multiple Sclerosis & MS Ireland Media Fact Sheet This fact sheets gives a summary of the main facts and issues relating to Multiple Sclerosis and gives an overview of the services offered by MS Ireland.
Multiple Sclerosis & MS Ireland Media Fact Sheet This fact sheets gives a summary of the main facts and issues relating to Multiple Sclerosis and gives an overview of the services offered by MS Ireland.
PHYSICAL ACTIVITY FOR BRAIN HEALTH AND FIGHTING DEMENTIA
 PHYSICAL ACTIVITY FOR BRAIN HEALTH AND FIGHTING DEMENTIA PAPER 36 SEPTEMBER 2013 DR MAREE FARROW DR KATHRYN ELLIS ISBN 978-1-921570-31-5 2013 Alzheimer s Australia Inc ABN 79 625 582 771 ARBN 066 779 557
PHYSICAL ACTIVITY FOR BRAIN HEALTH AND FIGHTING DEMENTIA PAPER 36 SEPTEMBER 2013 DR MAREE FARROW DR KATHRYN ELLIS ISBN 978-1-921570-31-5 2013 Alzheimer s Australia Inc ABN 79 625 582 771 ARBN 066 779 557
Cholinesterase inhibitors and memantine use for Alzheimer s disease TOPIC REVIEW
 Cholinesterase inhibitors and memantine use for Alzheimer s disease TOPIC REVIEW Diagnosis of Dementia : DSM-IV criteria Loss of memory and one or more other cognitive abilities Aphasia Apraxia Agnosia
Cholinesterase inhibitors and memantine use for Alzheimer s disease TOPIC REVIEW Diagnosis of Dementia : DSM-IV criteria Loss of memory and one or more other cognitive abilities Aphasia Apraxia Agnosia
Unmet Needs for Parkinson s Disease Therapeutics
 Unmet Needs for Parkinson s Disease Therapeutics Coalition Against Major Diseases & FDA Workshop October 20, 2014 Caroline M Tanner MD, PhD Director, Parkinson's Disease Research Education and Clinical
Unmet Needs for Parkinson s Disease Therapeutics Coalition Against Major Diseases & FDA Workshop October 20, 2014 Caroline M Tanner MD, PhD Director, Parkinson's Disease Research Education and Clinical
Clinician Portal: Enabling a Continuity of Concussion Care
 Clinician Portal: Enabling a Continuity of Concussion Care www.concussionvitalsigns.com Clinician Portal: Enabling a Continuity of Concussion CARE The Clinician Portal advances sports concussion care by
Clinician Portal: Enabling a Continuity of Concussion Care www.concussionvitalsigns.com Clinician Portal: Enabling a Continuity of Concussion CARE The Clinician Portal advances sports concussion care by
Ontario Reimburses CIS Indication for REBIF, a First-Line Treatment for Multiple Sclerosis
 May 25, 2015 Contact: Shikha Virdi 905-919-0200 ext. 5504 Ontario Reimburses CIS Indication for REBIF, a First-Line Treatment for Multiple Sclerosis Rebif now reimbursed under Ontario Drug Benefit Program
May 25, 2015 Contact: Shikha Virdi 905-919-0200 ext. 5504 Ontario Reimburses CIS Indication for REBIF, a First-Line Treatment for Multiple Sclerosis Rebif now reimbursed under Ontario Drug Benefit Program
Targeting Specific Cell Signaling Pathways for the Treatment of Malignant Peritoneal Mesothelioma
 The Use of Kinase Inhibitors: Translational Lab Results Targeting Specific Cell Signaling Pathways for the Treatment of Malignant Peritoneal Mesothelioma Sheelu Varghese, Ph.D. H. Richard Alexander, M.D.
The Use of Kinase Inhibitors: Translational Lab Results Targeting Specific Cell Signaling Pathways for the Treatment of Malignant Peritoneal Mesothelioma Sheelu Varghese, Ph.D. H. Richard Alexander, M.D.
How To Write Long Term Care Insurance
 By Lori Boyce, AVP Risk Management and R&D Underwriting long term care insurance: a primer Every day Canadians die, are diagnosed with cancer, have heart attacks and become disabled and our insurance solutions
By Lori Boyce, AVP Risk Management and R&D Underwriting long term care insurance: a primer Every day Canadians die, are diagnosed with cancer, have heart attacks and become disabled and our insurance solutions
Detection and staging of recurrent prostate cancer is still one of the important clinical problems in prostate cancer. A rise in PSA or biochemical
 Summary. 111 Detection and staging of recurrent prostate cancer is still one of the important clinical problems in prostate cancer. A rise in PSA or biochemical recurrence (BCR) is the first sign of recurrent
Summary. 111 Detection and staging of recurrent prostate cancer is still one of the important clinical problems in prostate cancer. A rise in PSA or biochemical recurrence (BCR) is the first sign of recurrent
Dementia: Delivering the Diagnosis
 Dementia: Delivering the Diagnosis Daniel D. Christensen, M.D. Clinical Professor of Psychiatry Clinical Professor of Neurology Adjunct Professor of Pharmacology University of Utah Diagnosing Dementia
Dementia: Delivering the Diagnosis Daniel D. Christensen, M.D. Clinical Professor of Psychiatry Clinical Professor of Neurology Adjunct Professor of Pharmacology University of Utah Diagnosing Dementia
NEW CRITERIA AND GUIDELINES FOR THE DIAGNOSIS OF ALZHEIMER S DISEASE PUBLISHED FOR FIRST TIME IN 27 YEARS
 FOR IMMEDIATE RELEASE CONTACT: Niles Frantz, Alzheimer s Association, 312-335-5777, niles.frantz@alz.org Alzheimer s Association media line, 312-335-4078, media@alz.org NEW CRITERIA AND GUIDELINES FOR
FOR IMMEDIATE RELEASE CONTACT: Niles Frantz, Alzheimer s Association, 312-335-5777, niles.frantz@alz.org Alzheimer s Association media line, 312-335-4078, media@alz.org NEW CRITERIA AND GUIDELINES FOR
The NeurOmics team at a recent project meeting
 Introduction Welcome to the NeurOmics project newsletter. This is the second edition and comes after the project has been underway for just over a year. This means that whilst we still have lots of work
Introduction Welcome to the NeurOmics project newsletter. This is the second edition and comes after the project has been underway for just over a year. This means that whilst we still have lots of work
Johns Hopkins Memory & Alzheimer s Treatment Center
 Memory is The Memory Center has not only provided great care to my husband, but also has helped me learn to care for myself while caring for him. - Sylvia Mackey Have you forgotten someone s name you should
Memory is The Memory Center has not only provided great care to my husband, but also has helped me learn to care for myself while caring for him. - Sylvia Mackey Have you forgotten someone s name you should
Understanding FTD Clinical Trials Continuing Education Training FTD Support Group Facilitators May 13, 2013
 Understanding FTD Clinical Trials Continuing Education Training FTD Support Group Facilitators May 13, 2013 Our Shared Vision We envision a world where frontotemporal degeneration is understood, effectively
Understanding FTD Clinical Trials Continuing Education Training FTD Support Group Facilitators May 13, 2013 Our Shared Vision We envision a world where frontotemporal degeneration is understood, effectively
Cognitive Neuroscience. Questions. Multiple Methods. Electrophysiology. Multiple Methods. Approaches to Thinking about the Mind
 Cognitive Neuroscience Approaches to Thinking about the Mind Cognitive Neuroscience Evolutionary Approach Sept 20-22, 2004 Interdisciplinary approach Rapidly changing How does the brain enable cognition?
Cognitive Neuroscience Approaches to Thinking about the Mind Cognitive Neuroscience Evolutionary Approach Sept 20-22, 2004 Interdisciplinary approach Rapidly changing How does the brain enable cognition?
Understanding. Multiple Sclerosis. Tim, diagnosed in 2004.
 Understanding Multiple Sclerosis Tim, diagnosed in 2004. What Is Multiple Sclerosis? Multiple sclerosis (MS) is a neurologic disorder that affects the central nervous system (CNS). The CNS includes the
Understanding Multiple Sclerosis Tim, diagnosed in 2004. What Is Multiple Sclerosis? Multiple sclerosis (MS) is a neurologic disorder that affects the central nervous system (CNS). The CNS includes the
Advances towards Remote Assessment of Disease and Relapse in Multiple Sclerosis
 Advances towards Remote Assessment of Disease and Relapse in Multiple Sclerosis Jane Rhodes March 31 st 2016 CAMD Digital Biomarkers Conference Baltimore, MD Emerging technology for the enhancement of
Advances towards Remote Assessment of Disease and Relapse in Multiple Sclerosis Jane Rhodes March 31 st 2016 CAMD Digital Biomarkers Conference Baltimore, MD Emerging technology for the enhancement of
Supporting MS-Related Disability Claims to Private Insurers: The Physician s Role
 Supporting MS-Related Disability Claims to Private Insurers: The Physician s Role What Is This Guide? This guide was compiled by the National Multiple Sclerosis Society as an aid to health care professionals
Supporting MS-Related Disability Claims to Private Insurers: The Physician s Role What Is This Guide? This guide was compiled by the National Multiple Sclerosis Society as an aid to health care professionals
Continuous Performance Test 3 rd Edition. C. Keith Conners, Ph.D.
 Continuous Performance Test 3 rd Edition C. Keith Conners, Ph.D. Assessment Report Name/ID: Alexandra Sample Age: 16 Gender: Female Birth Date: February 16, 1998 Grade: 11 Administration Date: February
Continuous Performance Test 3 rd Edition C. Keith Conners, Ph.D. Assessment Report Name/ID: Alexandra Sample Age: 16 Gender: Female Birth Date: February 16, 1998 Grade: 11 Administration Date: February
Diagnostik der Zukunft: Wissen wir mit Proteomik und Genomik wirklich mehr?
 Diagnostik der Zukunft: Wissen wir mit Proteomik und Genomik wirklich mehr? Prof. Dr. J. Wiltfang Klinik für Psychiatrie und Psychotherapie (Direktor: Prof. Dr. J. Wiltfang), Rheinische Kliniken, Universität
Diagnostik der Zukunft: Wissen wir mit Proteomik und Genomik wirklich mehr? Prof. Dr. J. Wiltfang Klinik für Psychiatrie und Psychotherapie (Direktor: Prof. Dr. J. Wiltfang), Rheinische Kliniken, Universität
Ageing Research in Trinity College
 Ageing Research in Trinity College 1. Trinity College Institute of Neuroscience & partner hospitals and centres Trinity s ageing research is carried out primarily by researchers affiliated to the Trinity
Ageing Research in Trinity College 1. Trinity College Institute of Neuroscience & partner hospitals and centres Trinity s ageing research is carried out primarily by researchers affiliated to the Trinity
PREDICTION AND PREVENTION OF DEMENTIA: NEW HOPE SECOND INTERNATIONAL MEETING OF THE MILAN CENTER (NEUROMI) MILAN, JULY 6-8, 2016 FOR NEUROSCIENCE
 www.neuromi.it SECOND INTERNATIONAL MEETING OF THE MILAN CENTER FOR NEUROSCIENCE (NEUROMI) PREDICTION AND PREVENTION OF DEMENTIA: NEW HOPE MILAN, JULY 6-8, 2016 Consiglio Nazionale delle Ricerche A new
www.neuromi.it SECOND INTERNATIONAL MEETING OF THE MILAN CENTER FOR NEUROSCIENCE (NEUROMI) PREDICTION AND PREVENTION OF DEMENTIA: NEW HOPE MILAN, JULY 6-8, 2016 Consiglio Nazionale delle Ricerche A new
Compositional Changes of B and T Cell Subtypes during Fingolimod Treatment in Multiple Sclerosis Patients: A 12-Month Follow-Up Study
 Compositional Changes of B and T Cell Subtypes during Fingolimod Treatment in Multiple Sclerosis Patients: A 12-Month Follow-Up Study Nele Claes 1 *; Tessa Dhaeze 1 *; Judith Fraussen PhD 1 ; Bieke Broux
Compositional Changes of B and T Cell Subtypes during Fingolimod Treatment in Multiple Sclerosis Patients: A 12-Month Follow-Up Study Nele Claes 1 *; Tessa Dhaeze 1 *; Judith Fraussen PhD 1 ; Bieke Broux
OHTAC Recommendation
 OHTAC Recommendation Multiple Sclerosis and Chronic Cerebrospinal Venous Insufficiency Presented to the Ontario Health Technology Advisory Committee in May 2010 May 2010 Issue Background A review on the
OHTAC Recommendation Multiple Sclerosis and Chronic Cerebrospinal Venous Insufficiency Presented to the Ontario Health Technology Advisory Committee in May 2010 May 2010 Issue Background A review on the
1 in 3 seniors dies with Alzheimer s or another dementia.
 2013 Alzheimer s disease facts and figures Includes a Special Report on long-distance caregivers 1 in 3 seniors dies with Alzheimer s or another dementia. Out-of-pocket expenses for long-distance caregivers
2013 Alzheimer s disease facts and figures Includes a Special Report on long-distance caregivers 1 in 3 seniors dies with Alzheimer s or another dementia. Out-of-pocket expenses for long-distance caregivers
2014 Alzheimer s Disease Facts and Figures
 2014 Alzheimer s Disease Facts and Figures Includes a Special Report on Women and Alzheimer s Disease Almost two-thirds of Americans with Alzheimer s disease are women. Alzheimer s Disease is the six eading
2014 Alzheimer s Disease Facts and Figures Includes a Special Report on Women and Alzheimer s Disease Almost two-thirds of Americans with Alzheimer s disease are women. Alzheimer s Disease is the six eading
placebo-controlledcontrolled double-blind, blind,
 Clinical Potential of Minocycline for Depression with Psychotic Features Tsuyoshi Miyaoka Department of Psychiatry Shimane University School of Medicine Minocycline 1. Second-generation tetracycline which
Clinical Potential of Minocycline for Depression with Psychotic Features Tsuyoshi Miyaoka Department of Psychiatry Shimane University School of Medicine Minocycline 1. Second-generation tetracycline which
The Role of Neuropsychological Testing in Guiding Decision- Making Related to Dementia
 The Role of Neuropsychological Testing in Guiding Decision- Making Related to Dementia By Scott Knight, Director, SLR Diagnostics & Assessments, a division of Sibley & Associates Inc., and Konstantine
The Role of Neuropsychological Testing in Guiding Decision- Making Related to Dementia By Scott Knight, Director, SLR Diagnostics & Assessments, a division of Sibley & Associates Inc., and Konstantine
Guidance for Industry Alzheimer s Disease: Developing Drugs for the Treatment of Early Stage Disease
 Guidance for Industry Alzheimer s Disease: Developing Drugs for the Treatment of Early Stage Disease DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments and suggestions
Guidance for Industry Alzheimer s Disease: Developing Drugs for the Treatment of Early Stage Disease DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments and suggestions
09/05/2014. Painting pictures of the brain with numbers. Overview
 Painting pictures of the brain with numbers Neurology for Insurers Dr Ian Cox & Adele Groyer (Gen Re) Overview Critical Illness Product Background Why should we be interested in neurology? Consult our
Painting pictures of the brain with numbers Neurology for Insurers Dr Ian Cox & Adele Groyer (Gen Re) Overview Critical Illness Product Background Why should we be interested in neurology? Consult our
Automatic Morphological Analysis of the Medial Temporal Lobe
 Automatic Morphological Analysis of the Medial Temporal Lobe Neuroimaging analysis applied to the diagnosis and prognosis of the Alzheimer s disease. Andrea Chincarini, INFN Genova Clinical aspects of
Automatic Morphological Analysis of the Medial Temporal Lobe Neuroimaging analysis applied to the diagnosis and prognosis of the Alzheimer s disease. Andrea Chincarini, INFN Genova Clinical aspects of
Case Studies, MCI, Clinical Trials and Care Management Support
 Case Studies, MCI, Clinical Trials and Care Management Support These slides form part of a Clinical Education programme. To benefit fully and earn CME credits, please contact clinician@re-cognitionhealth.com
Case Studies, MCI, Clinical Trials and Care Management Support These slides form part of a Clinical Education programme. To benefit fully and earn CME credits, please contact clinician@re-cognitionhealth.com
Financial advisors and Alzheimer s disease: What you need to know
 Financial advisors and Alzheimer s disease: What you need to know In today s aging society, with people living longer lives, chances are high that you ll be called upon to assist clients who are caring
Financial advisors and Alzheimer s disease: What you need to know In today s aging society, with people living longer lives, chances are high that you ll be called upon to assist clients who are caring
Predicting the future progression of dementia. Ashish Raj, PhD
 Business Presentation Predicting the future progression of dementia Ashish Raj, PhD Founder and CEO Associate Professor of Computer Science in Radiology Director, IDEAL Laboratory Associate Professor of
Business Presentation Predicting the future progression of dementia Ashish Raj, PhD Founder and CEO Associate Professor of Computer Science in Radiology Director, IDEAL Laboratory Associate Professor of
Functional neuroinflammatoryand serotonergic imaging in Alzheimer s disease: validation and preliminary clinical findings
 RIJKSUNIVERSITEIT GRONINGEN Functional neuroinflammatoryand serotonergic imaging in Alzheimer s disease: validation and preliminary clinical findings Jan Versijpt Josef Sudek ( 1896-1976) was a famous
RIJKSUNIVERSITEIT GRONINGEN Functional neuroinflammatoryand serotonergic imaging in Alzheimer s disease: validation and preliminary clinical findings Jan Versijpt Josef Sudek ( 1896-1976) was a famous
2015 Alzheimer s Disease Facts and Figures
 2015 Alzheimer s Disease Facts and Figures Includes a Special Report on Disclosing a Diagnosis of Alzheimer s Disease Alzheimer s Disease is the sixth-leading cause of death in the United States. more
2015 Alzheimer s Disease Facts and Figures Includes a Special Report on Disclosing a Diagnosis of Alzheimer s Disease Alzheimer s Disease is the sixth-leading cause of death in the United States. more
Community Network for Dementia and Critical Path in Japan
 Research and Reviews Community Network for Dementia and Critical Path in Japan JMAJ 54(5): 305 309, 2011 Satoshi ORIMO* 1 Abstract In Setagaya City of Tokyo, regional hospitals and medical associations
Research and Reviews Community Network for Dementia and Critical Path in Japan JMAJ 54(5): 305 309, 2011 Satoshi ORIMO* 1 Abstract In Setagaya City of Tokyo, regional hospitals and medical associations
Financial Advisors and Alzheimer s Disease: What You Need to Know
 Financial Advisors and Alzheimer s Disease: What You Need to Know In today s aging society, with people living longer lives, chances are good that you ll be called upon to assist clients who have Alzheimer
Financial Advisors and Alzheimer s Disease: What You Need to Know In today s aging society, with people living longer lives, chances are good that you ll be called upon to assist clients who have Alzheimer
CHAPTER 2: CLASSIFICATION AND ASSESSMENT IN CLINICAL PSYCHOLOGY KEY TERMS
 CHAPTER 2: CLASSIFICATION AND ASSESSMENT IN CLINICAL PSYCHOLOGY KEY TERMS ABC chart An observation method that requires the observer to note what happens before the target behaviour occurs (A), what the
CHAPTER 2: CLASSIFICATION AND ASSESSMENT IN CLINICAL PSYCHOLOGY KEY TERMS ABC chart An observation method that requires the observer to note what happens before the target behaviour occurs (A), what the
Understanding your Tecfidera treatment
 Understanding your Tecfidera treatment Information for patients who have been prescribed treatment with Tecfidera. (dimethyl fumarate) Contents About Multiple Sclerosis (MS) What is MS? Symptoms of MS
Understanding your Tecfidera treatment Information for patients who have been prescribed treatment with Tecfidera. (dimethyl fumarate) Contents About Multiple Sclerosis (MS) What is MS? Symptoms of MS
Understanding your Tecfidera treatment
 Understanding your Tecfidera treatment Information for patients who have been prescribed treatment with Tecfidera. (dimethyl fumarate) Contents About Multiple Sclerosis (MS) What is MS? Symptoms of MS
Understanding your Tecfidera treatment Information for patients who have been prescribed treatment with Tecfidera. (dimethyl fumarate) Contents About Multiple Sclerosis (MS) What is MS? Symptoms of MS
Objectives. Evaluation of Memory Loss. Cognitive Impairment. Clinical Questions. Medicare Wellness Visit
 Evaluation of Memory Loss and Mild Cognitive Impairment Skotti Church, MD Geriatrics Grand Rounds 4/3/2014 Objectives 1. Describe recommendations and tools for evaluation of cognitive impairment 2. Define
Evaluation of Memory Loss and Mild Cognitive Impairment Skotti Church, MD Geriatrics Grand Rounds 4/3/2014 Objectives 1. Describe recommendations and tools for evaluation of cognitive impairment 2. Define
How To Find Out If The Gulf War And Health Effects Of Serving In The Gulf Wars Are Linked To Health Outcomes
 Gulf War and Health: Update of Health Effects of Serving in the Gulf War Statement of Stephen L. Hauser, M.D. Professor and Chair of Neurology University of California, San Francisco, School of Medicine
Gulf War and Health: Update of Health Effects of Serving in the Gulf War Statement of Stephen L. Hauser, M.D. Professor and Chair of Neurology University of California, San Francisco, School of Medicine
PHARMACOMETABOLOMICS IN BIPOLAR DISORDER
 PHARMACOMETABOLOMICS IN BIPOLAR DISORDER V I C K I L. E L L I N G R O D, P H A R M. D., F C C P J O H N G I D E O N S E A R L E P R O F E S S O R O F C L I N I C A L A N D T R A N S L AT I O N A L P H
PHARMACOMETABOLOMICS IN BIPOLAR DISORDER V I C K I L. E L L I N G R O D, P H A R M. D., F C C P J O H N G I D E O N S E A R L E P R O F E S S O R O F C L I N I C A L A N D T R A N S L AT I O N A L P H
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE. Proposed Health Technology Appraisal
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Proposed Health Technology Appraisal Daclizumab for treating relapsing-remitting multiple Draft scope (pre-referral) Draft remit/appraisal objective To
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Proposed Health Technology Appraisal Daclizumab for treating relapsing-remitting multiple Draft scope (pre-referral) Draft remit/appraisal objective To
Spinal Muscular Atrophy
 Maryam Oskoui, MD, MSc, FRCPC Pediatric Neurologist Spinal Muscular Atrophy Elise Historical Timeline In vitro and animal studies Werdnig and Hoffmann describe SMA 1 (Arch Psych Nervenkrankheiten) Disease
Maryam Oskoui, MD, MSc, FRCPC Pediatric Neurologist Spinal Muscular Atrophy Elise Historical Timeline In vitro and animal studies Werdnig and Hoffmann describe SMA 1 (Arch Psych Nervenkrankheiten) Disease
