How To Participate In A Bladder Cancer Study
|
|
|
- Bernice Taylor
- 3 years ago
- Views:
Transcription
1 SCOTTSDALE HEALTHCARE INSTITUTIONAL REVIEW BOARD Consent to Participate in Research Protocol Name: A Study to Compare the Study Drug, Erlotinib or Polyphenon E, to a Placebo (a substance that looks like a study drug but is not active) in Patients with Bladder Cancer That Has Been Removed. Principal Investigator: Donald Lamm, MD Telephone #: N 40 th St., Suite E Phoenix, AZ Sponsor: National Cancer Institute Introduction You are invited to consider taking part in this research study. We will be testing two medicines that we believe will reduce the risk of bladder tumor recurrence, Erlotinib (Tarceva ) and Polyphenon E. This form will describe the purpose and nature of the study, its possible risks and benefits, other options available to you, and your rights as a participant in the study. Please take whatever time you need to discuss the study with your physicians, hospital personnel and your family and friends. The decision to take part or not is yours. If you decide to take part, please initial each page, and sign and date the last line of this form. Background and Purpose of the Study We will be testing two new oral drugs for bladder cancer, and if you consent you will be taking either one of two experimental drugs; Erlotinib or Polyphenon E (green tea extract), or you will be taking a placebo (substance that looks like the study drug but is not active). Erlotinib is known to work by stopping an enzyme that helps tumors grow. Polyphenon E (green tea extract) may work by a number of ways, including stopping an enzyme that helps bladder tumors grow. This is a 5-year study. The administration of the study drugs will last for 9 months and the next four years will involve follow-up visits. Depending on when you enter the study, the maximum number of visits will be 15 visits including the screening and study completion visits. The main purpose of this study is to determine if either Erlotinib or Polyphenon E (green tea extract) will reduce the risk of bladder cancer recurrence and/or progression, and to find out the side effects (good or bad) that these drugs cause. Researchers will look at the tissue samples from your tumor and in the urine to find out what effect these drugs have on individual cancer cells. We will also look at your tissue samples to see if we can find a genetic link. If a genetic link can be found it may mean that your family members have an increased risk to develop bladder cancer. This information may be helpful or hurtful to your family members. You will not be given any genetic information unless you specifically request it on the last page of this form. Page 1 of 1
2 Erlotinib and Polyphenon E are investigational drugs that have been shown to shrink tumors in animals. Polyphenon E and other green tea extracts have been shown to prevent the development of several types of cancer, including bladder cancer, in animals. Erlotinib has been shown to shrink tumors in animals and has been studied in people with other cancers and seems promising, but it is not clear if it can improve results of standard treatment in those cancers. The effects of Erlotinib or Polyphenon E in patients with bladder cancer are unknown. Total Number of Participants About 330 people will take part in this study nationally. People in the study are referred to as "participants." About 30 participants will enroll at this site. General Plan of This Study If you sign the consent form and agree to participate, we will ask you to do the following: Screening Procedures Before starting the study you will have tests and procedures performed within 3 weeks to decide if you are eligible for the study. The tests and procedures will include the following: A medical history, and medication history. A complete physical examination. A chest x-ray. A pulmonary (lung) function test (PFT) will be performed. You may have to go to Scottsdale Healthcare Shea to have this test done. PFT s measure your lung capacity. You blow into a machine, and it records how much air you take in and how fast you exhale it. A pulmonary test called a CT scan of the lungs may be performed if the lung test shows abnormal at screening and a lung specialist (pulmonologist) deems it necessary to rule out the presence of interstitial lung disease. Interstitial lung disease (ILD) is a broad category of lung diseases that includes disorders which are characterized by scarring of the lungs. Vital signs, including pulse, oral temperature, weight, height, and blood pressure Routine blood work, about 2-3 teaspoons of blood will be taken for this testing. A urine pregnancy test. Cystoscopy (a procedure done on an out patient basis, in which a doctor inserts a small tube-like instrument to look inside the bladder). This standard of care procedure includes bladder washings and urine collection. When we insert the cystoscope, we will gently wash the bladder with saline (water with a dash of salt), remove that saline and send it to the lab where it will be examined under a microscope for abnormalities. You will complete a risk factor questionnaire asking about your diet, life style, and behavior. You will need approximately 20 minutes to complete the questionnaire. The blood samples (about 2 teaspoons) we collect from you will be tested to evaluate genetic (inherited) factors that may be related to risk of cancer recurrence. Page 2 of 2
3 Monitor Procedures If at any time during the course of the study you experience symptoms such as fever, cough or shortness of breath, it is important that you see your doctor as soon as possible. In order to diagnose the cause of your symptoms, Doctor Lamm may order or do the following procedures and tests: A medical history, and medication history. A complete physical examination. A pulmonary (lung) function test will be performed at 3, 6, 9 and 12 months. A pulmonary test called a CT scan of the lungs will be performed. At each study visit, you will be evaluated for any eye-related adverse events. If the study doctor finds any abnormal condition in your eyes, you will be referred for an eye examination. Research Procedure Randomization: This is a randomized study. Randomization is a process similar to a flip of the coin. Randomization means that you will be assigned to one of three study groups by chance. This means that you will have a 1 in 3 chance of being assigned to one of the following three groups: 1) 800mg of real Polyphenon E (green tea extract) plus (a substance that looks like the study drug but is not active) 2) (a substance that looks like the study drug but is not active) plus 50mg 100mg of real Erlotinib 3) plus (both placebos will look like the study drugs but will not be active). Experience from a large number of clinical studies with Erlotinib in patients with cancer suggests that some side effects such as skin rash and diarrhea are common with higher doses, e.g., 150 mg per day and usually occur within the first 4 weeks of treatment (see Risks and Discomforts of Erlotinib below). To reduce both the frequency and possibly the severity of these side effects, Erlotinib will be given at a much lower dose, e.g., 50 mg per day for the first 6 weeks. This dose is only 1/3 of the usual starting dose of 150 mg per day used in current clinical trials in patients with cancer. If there are no significant side effects during the first 6 weeks, the dose of Erlotinib will be increased to 100 mg per day and continued for the 9 month duration of the treatment phase of the trial (as shown in the table below). Since neither you nor the study doctors and personnel will know to which group you have been assigned, participants in each study group will take part in increasing the dose of real Erlotinib or (a substance that looks like the study drug but is not active). Page 3 of 3
4 Below is a chart of the dose schedule for each study group: Study Group Assignment Weeks 1-6 Weeks 7-39 Real Polyphenon E Plus Real Polyphenon E 800 mg (4 pills) Real Polyphenon E 800 mg (4 pills) Plus Real Erlotinib Plus 50 mg (2 pills) 800 mg. (4 pills) Real Erlotinib 50 mg (2 pills) 800 mg (4 pills) 50 mg (2 pills) 100 mg (1 pill) 800 mg (4 pills) Real Erlotinib 100 mg (1 pill) 800 mg (4 pills) 100 mg (1 pill) If you experience significant side effects (e.g., rash) resulting from the study drug during weeks 1-6, study agent will be held until the side effects resolve. If you experience significant side effects resulting from the study drug during weeks 7-39, the Erlotinib dose will be decreased to 75mg. You will need to take your study drug, by mouth, at the same time each day for 9 months. The study drug should be taken on an empty stomach at least 1 hour before breakfast, with at least 6-7 oz of water. You will have one teaspoon of blood drawn every three months for the first year to monitor the effects of the study drug, and subsequently as determined by your treating physician to follow these possible side effects. You will need to have standard cystoscopies (a procedure to look inside the bladder) every 3 months for 2 years. This is a procedure to view the inside of your bladder and urethra in great detail using a specialized endoscope (a tube with a small camera used to perform tests and surgeries). The procedure usually takes between 5 and 20 minutes, and you should be able to go home with the same means of transportation. This is standard care for patients with your type of bladder cancer. You will need to stop taking study drug if your bladder cancer returns or if you experience side effects or if new scientific developments occur that indicate that this study drug is not in your best interest. Your doctor may also stop your study treatment if he/she feels that it is no longer in your best interest to continue. Visits 2 to 15 (Every 3 Months for the First 2 Years and Every 6 Months for the Next 3 Years) At each visit, you will be asked about any side effects that you may be experiencing. If you develop any new tumors in your bladder while you are on study, you will need to have them removed and you will be taken off the study drug. A urine pregnancy test will be performed on every visit for the first 2 years, and the cost will be covered by the sponsor. You will have two teaspoon of blood drawn every three months for the first year to monitor the effects of the study drug, and subsequently as determined by your treating physician to follow these possible toxicities. Genetic testing will be also performed on your blood samples to determine whether any genes associated with cancer can be identified. You will need to have standard cystoscopies every 3 months for 2 years. This is standard care for patients with your type of bladder cancer. Each Page 4 of 4
5 visit will last approximately 15 to 30 minutes. A lung function test will be performed at 3, 6, and 12 months to monitor any changes in your lung. If changes occur, you will have a CT scan (a specialized X-ray that looks at organs in the body for abnormalities) of the chest for assessment. If a lung disease is confirmed, study drug will be discontinued. If you experience side effects such as fever, coughing, short of breath, and it causes you to stop taking study drug for more than 2 weeks, you will be taken off of the study. Your blood sample(s) will be used for the purpose of this research study only and will not be shared with any other investigators. Upon completion of the study, your sample will be destroyed. How Your Treatment Will be Determined in This Study You will be assigned to one of 3 research treatment groups. A computer will determine which group you will be in through a process that is much like picking a number out of a hat. Neither the researcher nor any of the participants will know who is in which group until the study ends. Your chance of being in any group is one in 3. In the event of an emergency, Dr. Lamm can obtain information about which group you are in and the treatment you are receiving. This process is called randomization. Length of the Study for Each Participant We expect that you will be in the study for 5 years. Possible Benefits of Participating in the Study You might lower the chance of bladder tumor recurrence. However, we cannot guarantee that you will experience medical benefits from participating in this study. Others may benefit in the future from the information we obtain while you are in this study. Possible Side Effects and Other Risks of Participating in the Study You may experience some side effects as a result of the experimental medicines and treatments you receive in this study. Erlotinib: The risks and side effects you might have with Erlotinib include those listed below that are reported with the higher dose of 150mg: Common (greater than 21% of patients: skin rash, itching and dry skin (this may be treated with anti-acne antibiotics), diarrhea (this may be treated with anti-diarrhea medication). Rarely, diarrhea can be severe and cause dehydration, low blood pressure, impaired kidney function, and problems with blood chemistry requiring treatment in hospital. Tiredness. Page 5 of 5
6 Less common (5 to 20% of patients): headache, nausea and vomiting, loss of appetite, sore mouth, painful swallowing, anxiety, nose bleeds, cough, dry eyes, low red blood level (anemia), liver enzyme abnormality, dry mouth constipation, other infections. Infrequent (fewer than 5% of patients) eye changes with blurred vision and corneal (front surface of the eye) damage, inflammation or infection of the skin, inflammation of the uterus (womb) with vaginal bleeding, fever, kidney damage, irritation of the stomach or bowel that may lead to ulcers or bleeding, dehydration, fall in blood counts that can lead to bleeding, and irritation of the chest/lungs that may lead to fever, cough, and shortness of breath. This may be severe and life threatening. Please tell your doctor if you have any problems breathing, shortness of breath, cough, or fever. Erlotinib will be stopped while the cause of these problems are investigated and will be permanently stopped if these problems are due to inflammation of the lungs caused by Erlotinib. Most of the participants who developed this serious pulmonary side effect had primary lung cancer or the cancer had spread to the lungs from a tumor that originated outside of the lungs. With and investigational drug such as Erlotinib additional unexpected and sometimes serious side effects are possible. If you are taking or are prescribed a blood thinner at any time during the study, it is essential that you inform your doctor immediately. Being treated with the study drug, Erlotinib, at the same time as you are taking a blood thinner called coumadin or warfarin may increase the risk of bleeding. Until more information is known about this, your physician will follow you closely with blood tests to monitor the situation. If ou are seeing another doctor at another clinic who prescribes your blood-thinners, it is important that you inform him/her as well. Abnormal elevation of values in liver function tests have bee seen in patients receiving Erlotinib. If you are taking another medication that has the potential to cause liver damage, you should have liver function tests. Page 6 of 6
7 Your doctor will watch you closely to see if you have side effects. When possible, other drugs will be given to you to make these side effects less serious and uncomfortable. Many side effects go away shortly after treatment is stopped but in some cases effects can be serious, long-lasting or permanent. If you experience serious side effects that require treatment between regular clinic/hospital visits, it is important that you make every effort to return to the clinic/hospital where Erlotinib was given. If you need immediate treatment and are unable to return to the clinic/hospital where Erlotinib was given, your study doctor should be contacted as soon as possible. Pregnancy Women who are pregnant or breast feeding should not receive Erlotinib. It is recommended that a negative pregnancy test be confirmed before administration of study drug to those who may possibly become pregnant. Both sexually active men and women should use effective contraceptive methods to prevent the possibility of fathering a child or becoming pregnant while receiving study drug. If you are a female of childbearing age, you must have a urine pregnancy test to determine if you are pregnant or not. This test will be paid for by the study sponsor. Should you become pregnant or planning to have a child during the course of this study, you must immediately notify your study physician. Polyphenon E: There is no safety information available on Polyphenon E (Green Tea Extracts) from large, controlled clinical studies. There has been a single case report where a powdered green tea started an asthma episode in an asthma patient who worked at a green tea factory. There have been reported side effects of nausea and bloating when taking Polyphenon E in participants in a small clinical study sponsored by the National Cancer Institute. That study showed that 800 mg. of Polyphenon E per day is safe and well tolerated. Cystoscopy: You may feel slight discomfort as the cystoscope is passed through the urethra into the bladder. You will feel uncomfortable (a strong need to urinate) when the fluid has filled the bladder. You may notice a small amount of blood in your urine following this procedure. If the bleeding continues after three voids (urinations), contact your health care provider. There is also a chance of infection. If you develop pain, chills, fever, or reduced urine output, contact the study doctor immediately. Blood draws There are possible side effects, which can occur from having blood drawn. You could possibly develop a bruise, redness, or soreness at the site where the needle was put in to draw your blood. You may briefly feel the prick of the needle when it is inserted into your vein. You may feel dizzy or faint when your blood is drawn. IF YOU HAVE EVER FAINTED WHILE HAVING YOUR BLOOD DRAWN YOU SHOULD INFORM THE PERSON DRAWING YOUR BLOOD BEFORE THE PROCEDURE STARTS. We will take reasonable safeguards to minimize known and potential risks but unknown Page 7 of 7
8 and/or unanticipated side effects might occur. Most side effects go away when the drug is stopped, but lung inflammation could be long lasting when the study drugs are stopped. Who Can Participate This study is designed for former smokers with bladder cancer that has not invaded the muscle wall of the bladder. Participants must have their bladder tumor removed but be at a high risk (estimated to be at least 50%) for tumor recurrence. Your suitability for this study will be determined by medical tests and by medical history, physical exam, chest x-ray, and lung function tests. Who Cannot Participate The medicines and procedures used in this study may be unsafe for an unborn baby, a nursing infant, sperm, and eggs. Therefore, women and men who participate in this study must agree to use an appropriate and effective method of birth control throughout the study period and for at least 3 months after the last day of the study. These methods should be discussed with Dr. Lamm. If you do become pregnant during the study or if you father a child during the study, you should immediately notify Dr. Lamm at In addition if you are already pregnant or are breast-feeding, you cannot participate in this study. You cannot participate in this study if you: had prior chemotherapy or radiation treatments, have other cancers, have other significant medical or psychiatric condition, a history of asthma or interstitial lung disease, normally drink more than 5 cups of green tea daily take medications that may interfere with the study agents (your doctor will describe these) a history of exposure to metal dust or wood dust in your work or home environment, have a history of a connective tissue disease such as scleroderma, or rheumatoid arthritis, have significant eye abnormality, or if you are using contact lenses. Other Treatment Options If you do not participate in this study, the following options are available to treat your illness/condition: Standard treatment, which would include removal of any tumors, as well as treatment with chemotherapy or immunotherapy such as BCG given into the bladder. BCG is a vaccination to prevent tuberculosis. You could also choose to have no treatment, or periodic follow-up cystoscopies. Green Tea, but not the study preparation, is available without participating in the study. Confidentiality of the Data Collected During the Study Every effort will be made to keep your medical records confidential, as well as other Page 8 of 8
9 personal information that we gather during this study. Please see the attached Authorization to Share Protected (personal) Health Information (PHI) in Research. Whenever data from this study are published, your name will not be used. Individuals from the Scottsdale Healthcare IRB, Scottsdale Healthcare, the U.S. Food and Drug Administration, the investigators at UCLA, the National Cancer Institute, and the companies that make Erlotinib and polyphenon may look at medical and research records related to this study, both to assure quality control and to analyze data. We will disclose personal information about you to others as required by law. Every tissue or fluid sample contains genetic information. The genetic information we obtain from your blood sample may identify you, and give us information about your parents and ancestors. We have learned from past research that we are not always able to predict problems that may arise because of the research. You should be aware that possible problems related to genetic testing include insurance or employment discrimination, but we will take every precaution to maintain your confidentiality now and in the future. We may learn that there is a genetic link to your cancer, and this may be helpful or hurtful to members of your family. We will not give you any information we learn about your parents and ancestors, but it may be helpful for us to study members of your family. We will not contact your relatives without your permission. You will not be given any genetic information we may find unless you specifically request it on the last page of this form. Electronic Data Security Information about your participation in this study is stored in a computer and we will take the following precautions to protect it from unauthorized disclosure, tampering, or damage: Only authorized users will have access. New Findings Throughout the study, we will tell you about new information we receive about treatments that may be appropriate for you, about the treatments under research in this study, and any information that may affect your interest in remaining in the study. Costs to You for Participating Erlotinib and Polyphenon E (green tea extract) are investigational drugs and will be supplied to you at no cost. Routine blood tests will be billed to your insurer; other blood tests done specifically for this study, the pulmonary function test, the possible CT chest scan, and a possible eye examination will be paid for by the study. Your periodic, routine cystoscopies (a procedure done on an out patient basis, in which a doctor inserts a small tube-like instrument to look inside the bladder) will also be billed to your insurer. (Standard, routine procedures are those that would be billed to your insurer for the medical care you would receive if you were not on the study). I t is possible that your insurance will not pay for all the standard treatments and tests you will receive if you participate in this research. That is because many insurance companies, HMOs, and health benefit plans do not cover the cost of standard treatments that are provided as part of a research study. If that happens, you will be Page 9 of 9
10 responsible for all charges relating to standard of care which includes laboratory charges, radiological studies, cystoscopy procedure, and hospitalization charges (if necessary). The study doctor will provide you with an estimate of the cost of participation in the research. Payments to You for Participating Study participants will not be paid for participating in this study. Compensation in Case of Injury We will make every effort to prevent study-related injuries and illnesses. If you are injured or become ill while you are in the study and the illness or injury is due to your participation in this study, you will receive emergency medical care. The costs of this care will be charged to you or to your health insurer. No funds are available from Scottsdale Healthcare or the federal government to compensate you for a study-related injury or illness. This does not mean that you are giving up any of your legal rights. Your Rights as a Participant in The Study Participation in this study is entirely voluntary. You have the right to leave the study at any time. Leaving the study will not result in any penalty or loss of benefits to which you are entitled. Should you decide to leave the study, please call Dr. Lamm at Should you decide not to participate or to withdraw, your medical care will not be affected nor will your relations with your physicians, other personnel, and the hospital. Your care, however, may be managed by different researchers or physicians. Problems and Questions Call Dr. Lamm at day or night if you have questions about the study, any problems, unexpected physical or psychological discomforts, any injuries, or think that something unusual or unexpected is happening. The Scottsdale Healthcare Institutional Review Board (IRB) has reviewed this document for compliance with federal guidelines, and ethics. If you have questions about your rights as a research participant, you may call or write: Amy Wood, IRB coordinator or Robert Marlow, MD, Chair, IRB, 9003 E. Shea Blvd., Scottsdale, AZ 85260, Withdrawal by Investigator, Physician, or Sponsor The investigator (doctor involved with the study) may withdraw you from participating in this research study if circumstances arise which warrant doing so. Your participation in this study will continue as long as the following conditions are met: 1. You agree to take part in the study. 2. Your disease does not return. If this occurs, your treatment will be stopped and alternative care will be offered. You will continue to be followed by the investigator. 3. You follow the instructions for participation in this study. 4. The principal investigator (doctor in charge of the study) and sponsor agree to continue to conduct the study. Page 10 of 10
11 5. You do not experience side effects or are able to tolerate the side effects you do experience. Investigator's Statement I have fully explained this study to the participant. I have discussed the procedures and treatments, the possible risks and benefits, the standard and research aspects of the study, and have answered all of the questions that the participant and the participant's family members have asked. Signature of Investigator or Investigator s Designee Date Participant's Consent I have read the information provided in this Informed Consent Form (or it was read to me by ). All my questions were answered to my satisfaction. I voluntarily agree to participate in this study. [Upon signing, you will receive a copy of this form, and the original will become part of your medical record.] Your signature Date INFORMATION ABOUT YOUR GENETIC SAMPLE Please indicate by checking and initialing the category below whether or not you want to receive genetic information we learn about you. It is your responsibility to let the investigator know if your address and/or telephone number changes. Dr. Lamm s address is on the first page of this consent. I want to receive: ρ Genetic information about myself. ρ No genetic information about myself. The risks associated with genetic testing occur only if the information is provided to you or others. Genetic information can be associated with the risk of developing cancer. Knowing such information can cause worry and increased medical testing. The information will be closely guarded so the risk of being denied medical insurance or being subjected to discrimination based on this information should be negligible. The specific genetic information to be searched for will be genes that are associated with the development and progress of bladder and other cancers as well as genes that regulate hormones or other molecules that cancer cells use to their advantage. Such molecules may tell us which patient is likely to respond to drug treatment or be targets for future drug development. Page 11 of 11
X-Plain Chemotherapy for Breast Cancer - Adriamycin, Cytoxan, and Tamoxifen Reference Summary
 X-Plain Chemotherapy for Breast Cancer - Adriamycin, Cytoxan, and Tamoxifen Reference Summary Introduction Breast cancer is a common condition that affects one out of every 11 women. Your doctor has recommended
X-Plain Chemotherapy for Breast Cancer - Adriamycin, Cytoxan, and Tamoxifen Reference Summary Introduction Breast cancer is a common condition that affects one out of every 11 women. Your doctor has recommended
CONSENT FORM 12/19/08
 12/19/08 1001 University Place Evanston, Illinois 60201 www.northshore.org CONSENT FORM Phone (224) 364-7100 Fax (847) 570-8011 Intravesical Alkalized Lidocaine for the Treatment of Overactive Bladder
12/19/08 1001 University Place Evanston, Illinois 60201 www.northshore.org CONSENT FORM Phone (224) 364-7100 Fax (847) 570-8011 Intravesical Alkalized Lidocaine for the Treatment of Overactive Bladder
Liver Disease & Hepatitis Program Providers: Brian McMahon, MD, Steve Livingston, MD, Lisa Townshend, ANP. Primary Care Provider:
 Liver Disease & Hepatitis Program Providers: Brian McMahon, MD, Steve Livingston, MD, Lisa Townshend, ANP Primary Care Provider: If you are considering hepatitis C treatment, please read this treatment
Liver Disease & Hepatitis Program Providers: Brian McMahon, MD, Steve Livingston, MD, Lisa Townshend, ANP Primary Care Provider: If you are considering hepatitis C treatment, please read this treatment
Tuberculosis and You A Guide to Tuberculosis Treatment and Services
 Tuberculosis and You A Guide to Tuberculosis Treatment and Services Tuberculosis (TB) is a serious disease that can damage the lungs or other parts of the body like the brain, kidneys or spine. There are
Tuberculosis and You A Guide to Tuberculosis Treatment and Services Tuberculosis (TB) is a serious disease that can damage the lungs or other parts of the body like the brain, kidneys or spine. There are
If you are signing for a minor child, you refers to your child throughout the consent document.
 CONSENT TO PARTICIPATE IN A CLINICAL RESEARCH STUDY Adult Patient or Parent, for Minor Patient INSTITUTE: National Cancer Institute PRINCIPAL INVESTIGATOR: Raffit Hassan, M.D. STUDY TITLE: Tissue Procurement
CONSENT TO PARTICIPATE IN A CLINICAL RESEARCH STUDY Adult Patient or Parent, for Minor Patient INSTITUTE: National Cancer Institute PRINCIPAL INVESTIGATOR: Raffit Hassan, M.D. STUDY TITLE: Tissue Procurement
UW MEDICINE PATIENT EDUCATION. Xofigo Therapy. For metastatic prostate cancer. What is Xofigo? How does it work?
 UW MEDICINE PATIENT EDUCATION Xofigo Therapy For metastatic prostate cancer This handout explains how the drug Xofigo is used to treat metastatic prostate cancer. What is Xofigo? Xofigo is a radioactive
UW MEDICINE PATIENT EDUCATION Xofigo Therapy For metastatic prostate cancer This handout explains how the drug Xofigo is used to treat metastatic prostate cancer. What is Xofigo? Xofigo is a radioactive
Radiation Therapy for Prostate Cancer
 Radiation Therapy for Prostate Cancer Introduction Cancer of the prostate is the most common form of cancer that affects men. About 240,000 American men are diagnosed with prostate cancer every year. Your
Radiation Therapy for Prostate Cancer Introduction Cancer of the prostate is the most common form of cancer that affects men. About 240,000 American men are diagnosed with prostate cancer every year. Your
National Emphysema Treatment Trial (NETT) Consent for Screening and Patient Registry
 National Emphysema Treatment Trial (NETT) Consent for Screening and Patient Registry Instructions: This consent statement is to be signed and dated by the patient in the presence of a certified study staff
National Emphysema Treatment Trial (NETT) Consent for Screening and Patient Registry Instructions: This consent statement is to be signed and dated by the patient in the presence of a certified study staff
Patient Information Leaflet: Part 1 select-d
 Patient Information Leaflet: Part 1 select-d Anticoagulation Therapy in SELECTeD Cancer Patients at Risk of Recurrence of Venous Thromboembolism Introduction This
Patient Information Leaflet: Part 1 select-d Anticoagulation Therapy in SELECTeD Cancer Patients at Risk of Recurrence of Venous Thromboembolism Introduction This
What Cancer Patients Need To Know
 Taking Part in Clinical Trials What Cancer Patients Need To Know NATIONAL INSTITUTES OF HEALTH National Cancer Institute Generous support for this publication was provided by Novartis Oncology. Taking
Taking Part in Clinical Trials What Cancer Patients Need To Know NATIONAL INSTITUTES OF HEALTH National Cancer Institute Generous support for this publication was provided by Novartis Oncology. Taking
RESEARCH SUBJECT INFORMATION AND CONSENT FORM
 1 1 1 1 1 1 1 0 1 0 1 0 RESEARCH SUBJECT INFORMATION AND CONSENT FORM TITLE: PROTOCOL NR: SPONSOR: INVESTIGATOR: WIRB VCU tracking number This template is based on a drug or device research study. The
1 1 1 1 1 1 1 0 1 0 1 0 RESEARCH SUBJECT INFORMATION AND CONSENT FORM TITLE: PROTOCOL NR: SPONSOR: INVESTIGATOR: WIRB VCU tracking number This template is based on a drug or device research study. The
Medication Guide TASIGNA (ta-sig-na) (nilotinib) Capsules
 Medication Guide TASIGNA (ta-sig-na) (nilotinib) Capsules Read this Medication Guide before you start taking Tasigna and each time you get a refill. There may be new information. This information does
Medication Guide TASIGNA (ta-sig-na) (nilotinib) Capsules Read this Medication Guide before you start taking Tasigna and each time you get a refill. There may be new information. This information does
Colon and Rectal Cancer
 Colon and Rectal Cancer What is colon or rectal cancer? Colon or rectal cancer is the growth of abnormal cells in your large intestine, which is also called the large bowel. The colon is the last 5 feet
Colon and Rectal Cancer What is colon or rectal cancer? Colon or rectal cancer is the growth of abnormal cells in your large intestine, which is also called the large bowel. The colon is the last 5 feet
For the Patient: Paclitaxel injection Other names: TAXOL
 For the Patient: Paclitaxel injection Other names: TAXOL Paclitaxel (pak'' li tax' el) is a drug that is used to treat many types of cancer. It is a clear liquid that is injected into a vein. Tell your
For the Patient: Paclitaxel injection Other names: TAXOL Paclitaxel (pak'' li tax' el) is a drug that is used to treat many types of cancer. It is a clear liquid that is injected into a vein. Tell your
Palm Beach Obstetrics & Gynecology, PA
 Palm Beach Obstetrics & Gynecology, PA 4671 S. Congress Avenue, Lake Worth, FL 33461 561.434.0111 4631 N. Congress Avenue, Suite 102, West Palm Beach, FL 33407 Urinary Tract Infection About one of every
Palm Beach Obstetrics & Gynecology, PA 4671 S. Congress Avenue, Lake Worth, FL 33461 561.434.0111 4631 N. Congress Avenue, Suite 102, West Palm Beach, FL 33407 Urinary Tract Infection About one of every
Patient identifier/label: Page 1 of 6 PATIENT AGREEMENT TO SYSTEMIC THERAPY: CONSENT FORM FEC-T. Patient s first names. Date of birth.
 Patient identifier/label: Page 1 of 6 PATIENT AGREEMENT TO SYSTEMIC THERAPY: CONSENT FORM FEC-T Patient s surname/family name Patient s first names Date of birth Hospital Name: Guy s Hospital St. Thomas
Patient identifier/label: Page 1 of 6 PATIENT AGREEMENT TO SYSTEMIC THERAPY: CONSENT FORM FEC-T Patient s surname/family name Patient s first names Date of birth Hospital Name: Guy s Hospital St. Thomas
Presenting the SUTENT Patient Call Center.
 Presenting the SUTENT Patient Call Center. Please see patient Medication Guide and full prescribing information attached. We re here to support you. Dealing with cancer is a journey. Along the way, you
Presenting the SUTENT Patient Call Center. Please see patient Medication Guide and full prescribing information attached. We re here to support you. Dealing with cancer is a journey. Along the way, you
Thymus Cancer. This reference summary will help you better understand what thymus cancer is and what treatment options are available.
 Thymus Cancer Introduction Thymus cancer is a rare cancer. It starts in the small organ that lies in the upper chest under the breastbone. The thymus makes white blood cells that protect the body against
Thymus Cancer Introduction Thymus cancer is a rare cancer. It starts in the small organ that lies in the upper chest under the breastbone. The thymus makes white blood cells that protect the body against
Notice of Privacy Practices
 Notice of Privacy Practices This notice describes how medical information about you may be used and disclosed, and how you may obtain access to this information. Please review it carefully. OMAC respects
Notice of Privacy Practices This notice describes how medical information about you may be used and disclosed, and how you may obtain access to this information. Please review it carefully. OMAC respects
VAD Chemotherapy Regimen for Multiple Myeloma Information for Patients
 VAD Chemotherapy Regimen for Multiple Myeloma Information for Patients The Regimen contains: V = vincristine (Oncovin ) A = Adriamycin (doxorubicin) D = Decadron (dexamethasone) How Is This Regimen Given?
VAD Chemotherapy Regimen for Multiple Myeloma Information for Patients The Regimen contains: V = vincristine (Oncovin ) A = Adriamycin (doxorubicin) D = Decadron (dexamethasone) How Is This Regimen Given?
PATIENT INFORMATION INSURANCE INFORMATION
 (mm/dd/yyyy): Have you been to Physicians Urgent Care before? Yes No Arrival Time: If yes, when? Is this a follow-up to a previous visit: Yes No PATIENT INFORMATION Patient s First Name: Middle Name: Last
(mm/dd/yyyy): Have you been to Physicians Urgent Care before? Yes No Arrival Time: If yes, when? Is this a follow-up to a previous visit: Yes No PATIENT INFORMATION Patient s First Name: Middle Name: Last
Patient Medication Guide Brochure
 Patient Medication Guide Brochure 1 MEDICATION GUIDE TASIGNA (ta-sig-na) (nilotinib) Capsules Read this Medication Guide before you start taking TASIGNA and each time you get a refill. There may be new
Patient Medication Guide Brochure 1 MEDICATION GUIDE TASIGNA (ta-sig-na) (nilotinib) Capsules Read this Medication Guide before you start taking TASIGNA and each time you get a refill. There may be new
Clinical Trials Network
 National Drug Abuse Treatment Clinical Trials Network STOP SMOKING STUDY Should I Join? NATIONAL INSTITUTES OF HEALTH U.S. DEPARTMENT OF HEALTH AND HUMAN SERVICES Introduction Many people who abuse drugs
National Drug Abuse Treatment Clinical Trials Network STOP SMOKING STUDY Should I Join? NATIONAL INSTITUTES OF HEALTH U.S. DEPARTMENT OF HEALTH AND HUMAN SERVICES Introduction Many people who abuse drugs
Southwest General Surgical Associates General & Vascular Surgery 8230 Walnut Hill Lane Suite 408 Dallas, TX 75231 Phone-214)369-5432 Fax-214)369-5591
 Southwest General Surgical Associates General & Vascular Surgery 8230 Walnut Hill Lane Suite 408 Dallas, TX 75231 Phone-214)369-5432 Fax-214)369-5591 Andres U. Katz, M.D. Richard S. Anderson, M.D. G. Thomas
Southwest General Surgical Associates General & Vascular Surgery 8230 Walnut Hill Lane Suite 408 Dallas, TX 75231 Phone-214)369-5432 Fax-214)369-5591 Andres U. Katz, M.D. Richard S. Anderson, M.D. G. Thomas
MEDICATION GUIDE POMALYST (POM-uh-list) (pomalidomide) capsules. What is the most important information I should know about POMALYST?
 MEDICATION GUIDE POMALYST (POM-uh-list) (pomalidomide) capsules What is the most important information I should know about POMALYST? Before you begin taking POMALYST, you must read and agree to all of
MEDICATION GUIDE POMALYST (POM-uh-list) (pomalidomide) capsules What is the most important information I should know about POMALYST? Before you begin taking POMALYST, you must read and agree to all of
Women s Continence and Pelvic Health Center
 Women s Continence and Pelvic Health Center Committed to Caring 580-590 Court Street Keene, New Hampshire 03431 (603) 354-5454 Ext. 6643 URINARY INCONTINENCE QUESTIONNAIRE The purpose of this questionnaire
Women s Continence and Pelvic Health Center Committed to Caring 580-590 Court Street Keene, New Hampshire 03431 (603) 354-5454 Ext. 6643 URINARY INCONTINENCE QUESTIONNAIRE The purpose of this questionnaire
PERIPHERAL STEM CELL TRANSPLANT INTRODUCTION
 PERIPHERAL STEM CELL TRANSPLANT INTRODUCTION This booklet was designed to help you and the important people in your life understand the treatment of high dose chemotherapy with stem cell support: a procedure
PERIPHERAL STEM CELL TRANSPLANT INTRODUCTION This booklet was designed to help you and the important people in your life understand the treatment of high dose chemotherapy with stem cell support: a procedure
ORAL ANTICOAGULANTS RIVAROXABAN (XARELTO) FOR PULMONARY EMBOLISM (PE)
 ORAL ANTICOAGULANTS RIVAROXABAN (XARELTO) FOR PULMONARY EMBOLISM (PE) Information Leaflet Your Health. Our Priority. Page 2 of 6 What Are Anticoagulants And What Do They Do? This information leaflet has
ORAL ANTICOAGULANTS RIVAROXABAN (XARELTO) FOR PULMONARY EMBOLISM (PE) Information Leaflet Your Health. Our Priority. Page 2 of 6 What Are Anticoagulants And What Do They Do? This information leaflet has
Chemotherapy Side Effects Worksheet
 Page 1 of 6 Chemotherapy Side Effects Worksheet Medicines or drugs that destroy cancer cells are called cancer chemotherapy. It is sometimes the first choice for treating many cancers. Chemotherapy differs
Page 1 of 6 Chemotherapy Side Effects Worksheet Medicines or drugs that destroy cancer cells are called cancer chemotherapy. It is sometimes the first choice for treating many cancers. Chemotherapy differs
CHOP Chemotherapy Regimen for Lymphoma Information for Patients
 CHOP Chemotherapy Regimen for Lymphoma Information for Patients The Regimen Contains: C: Cytoxan (cyclophosphamide) H: Adriamycin (hydroxy doxorubicin) O: vincristine (Oncovin ) P: Prednisone How Is This
CHOP Chemotherapy Regimen for Lymphoma Information for Patients The Regimen Contains: C: Cytoxan (cyclophosphamide) H: Adriamycin (hydroxy doxorubicin) O: vincristine (Oncovin ) P: Prednisone How Is This
You. guide to tuberculosis treatment and services
 Adapted from TB and You: A Guide to Tuberculosis Treatment and Services with permission from Division of Public Health TB Control Program State of North Carolina Department of Health and Human Services
Adapted from TB and You: A Guide to Tuberculosis Treatment and Services with permission from Division of Public Health TB Control Program State of North Carolina Department of Health and Human Services
Form ### Transgender Hormone Therapy - Estrogen Informed Consent SAMPLE
 What are the different medications that can help to feminize me? Estrogen - Different types of the hormone estrogen can help you appear more feminine. Estrogen is the female sex hormone. Androgen blocker
What are the different medications that can help to feminize me? Estrogen - Different types of the hormone estrogen can help you appear more feminine. Estrogen is the female sex hormone. Androgen blocker
Exposure. What Healthcare Personnel Need to Know
 Information from the Centers for Disease Control and Prevention National Center for Infectious Diseases Divison of Healthcare Quality Promotion and Division of Viral Hepatitis For additional brochures
Information from the Centers for Disease Control and Prevention National Center for Infectious Diseases Divison of Healthcare Quality Promotion and Division of Viral Hepatitis For additional brochures
Treating Chronic Hepatitis C. A Review of the Research for Adults
 Treating Chronic Hepatitis C A Review of the Research for Adults Is This Information Right for Me? Yes, this information is right for you if: Your doctor* has told you that you have chronic hepatitis C.
Treating Chronic Hepatitis C A Review of the Research for Adults Is This Information Right for Me? Yes, this information is right for you if: Your doctor* has told you that you have chronic hepatitis C.
MEDICATION GUIDE. PROCRIT (PRO KRIT) (epoetin alfa)
 MEDICATION GUIDE PROCRIT (PROKRIT) (epoetin alfa) Read this Medication Guide: before you start PROCRIT. if you are told by your healthcare provider that there is new information about PROCRIT. if you are
MEDICATION GUIDE PROCRIT (PROKRIT) (epoetin alfa) Read this Medication Guide: before you start PROCRIT. if you are told by your healthcare provider that there is new information about PROCRIT. if you are
Breast Cancer. Breast Cancer Page 1
 Breast Cancer Summary Breast cancers which are detected early are curable by local treatments. The initial surgery will give the most information about the cancer; such as size or whether the glands (or
Breast Cancer Summary Breast cancers which are detected early are curable by local treatments. The initial surgery will give the most information about the cancer; such as size or whether the glands (or
Mitoxantrone. For multiple sclerosis. InfoNEURO INFORMATION FOR PATIENTS. Montreal Neurological Hospital
 i InfoNEURO Montreal Neurological Hospital INFORMATION FOR PATIENTS Mitoxantrone For multiple sclerosis Centre universitaire de santé McGill McGill University Health Centre Collaborators: D. Lowden Clinical
i InfoNEURO Montreal Neurological Hospital INFORMATION FOR PATIENTS Mitoxantrone For multiple sclerosis Centre universitaire de santé McGill McGill University Health Centre Collaborators: D. Lowden Clinical
Streptococcal Infections
 Streptococcal Infections Introduction Streptococcal, or strep, infections cause a variety of health problems. These infections can cause a mild skin infection or sore throat. But they can also cause severe,
Streptococcal Infections Introduction Streptococcal, or strep, infections cause a variety of health problems. These infections can cause a mild skin infection or sore throat. But they can also cause severe,
Mesothelioma. 1995-2013, The Patient Education Institute, Inc. www.x-plain.com ocft0101 Last reviewed: 03/21/2013 1
 Mesothelioma Introduction Mesothelioma is a type of cancer. It starts in the tissue that lines your lungs, stomach, heart, and other organs. This tissue is called mesothelium. Most people who get this
Mesothelioma Introduction Mesothelioma is a type of cancer. It starts in the tissue that lines your lungs, stomach, heart, and other organs. This tissue is called mesothelium. Most people who get this
Long term observational study for multiple sclerosis patients using fingolimod
 Investigator: Dr Matthew Jackson (Consultant Neurologist) Centre number: 814 Contact for queries If you have any questions about this study, you can contact: Daytime: Elspeth Wolfenden on 01494 734312
Investigator: Dr Matthew Jackson (Consultant Neurologist) Centre number: 814 Contact for queries If you have any questions about this study, you can contact: Daytime: Elspeth Wolfenden on 01494 734312
CMF: Cyclophosphamide, Methotrexate and Fluorouracil
 PATIENT EDUCATION patienteducation.osumc.edu CMF: Cyclophosphamide, Methotrexate and Fluorouracil What is CMF? This is the short name for the drugs used for this chemotherapy treatment. The three drugs
PATIENT EDUCATION patienteducation.osumc.edu CMF: Cyclophosphamide, Methotrexate and Fluorouracil What is CMF? This is the short name for the drugs used for this chemotherapy treatment. The three drugs
NEW PATIENT HISTORY QUESTIONNAIRE. Physician Initials Date PATIENT INFORMATION
 NEW PATIENT HISTORY QUESTIONNAIRE Physician Initials Date PATIENT INFORMATION JHH# DOB# AGE HOME PH CELL PH DAY PH EMAIL Who is your REFERRING PHYSICIAN? (The doctor who referred you to Johns Hopkins Neurology.)
NEW PATIENT HISTORY QUESTIONNAIRE Physician Initials Date PATIENT INFORMATION JHH# DOB# AGE HOME PH CELL PH DAY PH EMAIL Who is your REFERRING PHYSICIAN? (The doctor who referred you to Johns Hopkins Neurology.)
What You Need to Know About LEMTRADA (alemtuzumab) Treatment: A Patient Guide
 For Patients What You Need to Know About LEMTRADA (alemtuzumab) Treatment: A Patient Guide Patients: Your doctor or nurse will go over this patient guide with you. It is important to ask any questions
For Patients What You Need to Know About LEMTRADA (alemtuzumab) Treatment: A Patient Guide Patients: Your doctor or nurse will go over this patient guide with you. It is important to ask any questions
University of Pennsylvania RESEARCH Subject Informed Consent Form AND RESEARCH SUBJECT HIPAA AUTHORIZATION
 University of Pennsylvania RESEARCH Subject Informed Consent Form AND RESEARCH SUBJECT HIPAA AUTHORIZATION Protocol Title: Evaluating the impact of cocaine use and HIV infection in arterial wall inflammation
University of Pennsylvania RESEARCH Subject Informed Consent Form AND RESEARCH SUBJECT HIPAA AUTHORIZATION Protocol Title: Evaluating the impact of cocaine use and HIV infection in arterial wall inflammation
For the Patient: GDP Other names: LYGDP
 1 For the Patient: GDP Other names: LYGDP G D P Gemcitabine Dexamethasone (taken by mouth) CISPlatin Uses: GDP is a drug treatment given for Non-Hodgkins and Hodgkin Lymphoma with the expectation of destroying
1 For the Patient: GDP Other names: LYGDP G D P Gemcitabine Dexamethasone (taken by mouth) CISPlatin Uses: GDP is a drug treatment given for Non-Hodgkins and Hodgkin Lymphoma with the expectation of destroying
Bile Duct Diseases and Problems
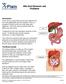 Bile Duct Diseases and Problems Introduction A bile duct is a tube that carries bile between the liver and gallbladder and the intestine. Bile is a substance made by the liver that helps with digestion.
Bile Duct Diseases and Problems Introduction A bile duct is a tube that carries bile between the liver and gallbladder and the intestine. Bile is a substance made by the liver that helps with digestion.
AC Chemotherapy Regimen (Doxorubicin + Cyclophosphamide)
 AC Chemotherapy Regimen (Doxorubicin + Cyclophosphamide) AC is a regimen or treatment plan that includes a combination of chemotherapy drugs that your doctor prescribed for the treatment of your cancer.
AC Chemotherapy Regimen (Doxorubicin + Cyclophosphamide) AC is a regimen or treatment plan that includes a combination of chemotherapy drugs that your doctor prescribed for the treatment of your cancer.
Safety Information Card for Xarelto Patients
 Safety Information Card for Xarelto Patients 15mg Simply Protecting More Patients 20mg Simply Protecting More Patients Keep this card with you at all times Present this card to every physician or dentist
Safety Information Card for Xarelto Patients 15mg Simply Protecting More Patients 20mg Simply Protecting More Patients Keep this card with you at all times Present this card to every physician or dentist
CVP Chemotherapy Regimen for Lymphoma Information for Patients
 CVP Chemotherapy Regimen for Lymphoma Information for Patients The Regimen Contains: C: Cyclophosphamide (Cytoxan ) V: Vincristine (Oncovin ) P: Prednisone How Is This Regimen Given? CVP is given every
CVP Chemotherapy Regimen for Lymphoma Information for Patients The Regimen Contains: C: Cyclophosphamide (Cytoxan ) V: Vincristine (Oncovin ) P: Prednisone How Is This Regimen Given? CVP is given every
AC: Doxorubicin and Cyclophosphamide
 PATIENT EDUCATION patienteducation.osumc.edu What is AC? It is the short name for the drugs used for this chemotherapy treatment. The two drugs you will receive during this treatment are Doxorubicin (Adriamycin
PATIENT EDUCATION patienteducation.osumc.edu What is AC? It is the short name for the drugs used for this chemotherapy treatment. The two drugs you will receive during this treatment are Doxorubicin (Adriamycin
Disease/Illness GUIDE TO ASBESTOS LUNG CANCER. What Is Asbestos Lung Cancer? www.simpsonmillar.co.uk Telephone 0844 858 3200
 GUIDE TO ASBESTOS LUNG CANCER What Is Asbestos Lung Cancer? Like tobacco smoking, exposure to asbestos can result in the development of lung cancer. Similarly, the risk of developing asbestos induced lung
GUIDE TO ASBESTOS LUNG CANCER What Is Asbestos Lung Cancer? Like tobacco smoking, exposure to asbestos can result in the development of lung cancer. Similarly, the risk of developing asbestos induced lung
MEDICAL HISTORY AND SCREENING FORM
 MEDICAL HISTORY AND SCREENING FORM The purpose of preventive exams is to screen for potential health problems and provide education to promote optimal health. It is best practice for chronic health problems
MEDICAL HISTORY AND SCREENING FORM The purpose of preventive exams is to screen for potential health problems and provide education to promote optimal health. It is best practice for chronic health problems
Gemcitabine and Cisplatin
 PATIENT EDUCATION patienteducation.osumc.edu What is Gemcitabine (jem-site-a been)? Gemcitabine is a chemotherapy medicine known as an anti-metabolite. Another name for this drug is Gemzar. This drug is
PATIENT EDUCATION patienteducation.osumc.edu What is Gemcitabine (jem-site-a been)? Gemcitabine is a chemotherapy medicine known as an anti-metabolite. Another name for this drug is Gemzar. This drug is
Lung Cancer. This reference summary will help you better understand lung cancer and the treatment options that are available.
 Lung Cancer Introduction Lung cancer is the number one cancer killer of men and women. Over 165,000 people die of lung cancer every year in the United States. Most cases of lung cancer are related to cigarette
Lung Cancer Introduction Lung cancer is the number one cancer killer of men and women. Over 165,000 people die of lung cancer every year in the United States. Most cases of lung cancer are related to cigarette
Temozolomide (oral) with concurrent radiotherapy to the brain
 Temozolomide (oral) with concurrent radiotherapy to the brain Temozolomide (oral) with concurrent radiotherapy to the brain This leaflet is offered as a guide to you and your family. You will find it useful
Temozolomide (oral) with concurrent radiotherapy to the brain Temozolomide (oral) with concurrent radiotherapy to the brain This leaflet is offered as a guide to you and your family. You will find it useful
HOSPITAL OF THE UNIVERSITY OF PENNSYLVANIA CONSENT TO PARTICIPATE IN A RESEARCH STUDY AND RESEARCH SUBJECT HIPAA AUTHORIZATION
 CONSENT TO PARTICIPATE IN A RESEARCH STUDY AND RESEARCH SUBJECT HIPAA AUTHORIZATION Protocol Title: A5322, Version 2.0, 01/28/15 Long-term Follow-up of Older HIV-infected Adults in the ACTG: Addressing
CONSENT TO PARTICIPATE IN A RESEARCH STUDY AND RESEARCH SUBJECT HIPAA AUTHORIZATION Protocol Title: A5322, Version 2.0, 01/28/15 Long-term Follow-up of Older HIV-infected Adults in the ACTG: Addressing
BASIC INFORMATION ABOUT HIV, HEPATITIS B and C, and TUBERCULOSIS Adapted from the CDC
 BASIC INFORMATION ABOUT HIV, HEPATITIS B and C, and TUBERCULOSIS Adapted from the CDC HIV What are HIV and AIDS? HIV stands for Human Immunodeficiency Virus. This is the virus that causes AIDS. HIV is
BASIC INFORMATION ABOUT HIV, HEPATITIS B and C, and TUBERCULOSIS Adapted from the CDC HIV What are HIV and AIDS? HIV stands for Human Immunodeficiency Virus. This is the virus that causes AIDS. HIV is
Medicines To Treat Alcohol Use Disorder A Review of the Research for Adults
 Medicines To Treat Alcohol Use Disorder A Review of the Research for Adults Is This Information Right for Me? Yes, this information is right for you if: Your doctor* said you have alcohol use disorder
Medicines To Treat Alcohol Use Disorder A Review of the Research for Adults Is This Information Right for Me? Yes, this information is right for you if: Your doctor* said you have alcohol use disorder
Brain Cancer. This reference summary will help you understand how brain tumors are diagnosed and what options are available to treat them.
 Brain Cancer Introduction Brain tumors are not rare. Thousands of people are diagnosed every year with tumors of the brain and the rest of the nervous system. The diagnosis and treatment of brain tumors
Brain Cancer Introduction Brain tumors are not rare. Thousands of people are diagnosed every year with tumors of the brain and the rest of the nervous system. The diagnosis and treatment of brain tumors
St. Luke s MS Center New Patient Questionnaire. Name: Date: Birth date: Right or Left handed? Who is your Primary Doctor?
 St. Luke s MS Center New Patient Questionnaire Name: Date: Birth date: Right or Left handed? Who is your Primary Doctor? Who referred you to the MS Center? List any other doctors you see: Reason you have
St. Luke s MS Center New Patient Questionnaire Name: Date: Birth date: Right or Left handed? Who is your Primary Doctor? Who referred you to the MS Center? List any other doctors you see: Reason you have
INITIATING ORAL AUBAGIO (teriflunomide) THERAPY
 FOR YOUR PATIENTS WITH RELAPSING FORMS OF MS INITIATING ORAL AUBAGIO (teriflunomide) THERAPY WARNING: HEPATOTOXICITY AND RISK OF TERATOGENICITY Severe liver injury including fatal liver failure has been
FOR YOUR PATIENTS WITH RELAPSING FORMS OF MS INITIATING ORAL AUBAGIO (teriflunomide) THERAPY WARNING: HEPATOTOXICITY AND RISK OF TERATOGENICITY Severe liver injury including fatal liver failure has been
Southwestern Foot & Ankle Associates, P.C. 3880 Parkwood Blvd, Suite 602 Frisco, TX 75034 Phone: 972-335-9071 Fax: 972-335-8920 Dr. Thomas H.
 Phone: 972-335-9071 Fax: 972-335-8920 Date: Home Phone ( ) Patient Information (Please Print) Email: Name: SS/Patient ID # Last Name First Name Middle Initial Address Cell Phone ( ) City State Zip Sex
Phone: 972-335-9071 Fax: 972-335-8920 Date: Home Phone ( ) Patient Information (Please Print) Email: Name: SS/Patient ID # Last Name First Name Middle Initial Address Cell Phone ( ) City State Zip Sex
1MFBTF GJMM PVU GPSNT BOE GBY 'PSNT XJMM CF TJHOFE BU ZPVS BQQPJOUNFOU
 CELL PHONE: PATIENT HISTORY FORM - CONFIDENTIAL DATE: PATIENT: (LAST NAME) (FIRST NAME) (Ml) (NICKNAME) DOB: Primary Physician/ Family Doctor: Phone: Past Medical History (Click all that apply) High blood
CELL PHONE: PATIENT HISTORY FORM - CONFIDENTIAL DATE: PATIENT: (LAST NAME) (FIRST NAME) (Ml) (NICKNAME) DOB: Primary Physician/ Family Doctor: Phone: Past Medical History (Click all that apply) High blood
PATIENT HEALTH QUESTIONNAIRE Radiation Oncology (Patient Label)
 REVIEWED DATE / INITIALS SAFETY: Are you at risk for falls? Do you have a Pacemaker? Females; Is there a possibility you may be pregnant? ALLERGIES: Do you have any allergies to medications? If, please
REVIEWED DATE / INITIALS SAFETY: Are you at risk for falls? Do you have a Pacemaker? Females; Is there a possibility you may be pregnant? ALLERGIES: Do you have any allergies to medications? If, please
Copayment Is Due At Time Of Visit. Self-pay (payment due at time of service)
 REGISTRATION FORM Please present your insurance card and photo ID at time of check-in. Settlement of patient financial responsibility is expected at time of service. Copayment Is Due At Time Of Visit.
REGISTRATION FORM Please present your insurance card and photo ID at time of check-in. Settlement of patient financial responsibility is expected at time of service. Copayment Is Due At Time Of Visit.
Paclitaxel and Carboplatin
 PATIENT EDUCATION patienteducation.osumc.edu What is Paclitaxel (pak-li-tax-el) and how does it work? Paclitaxel is a chemotherapy drug known as an anti-microtubule inhibitor. Another name for this drug
PATIENT EDUCATION patienteducation.osumc.edu What is Paclitaxel (pak-li-tax-el) and how does it work? Paclitaxel is a chemotherapy drug known as an anti-microtubule inhibitor. Another name for this drug
Patient Guide. Important information for patients starting therapy with LEMTRADA (alemtuzumab)
 Patient Guide Important information for patients starting therapy with LEMTRADA (alemtuzumab) This medicinal product is subject to additional monitoring. This will allow quick identification of new safety
Patient Guide Important information for patients starting therapy with LEMTRADA (alemtuzumab) This medicinal product is subject to additional monitoring. This will allow quick identification of new safety
Medication Guide EQUETRO (ē-kwĕ-trō) (carbamazepine) Extended-Release Capsules
 Medication Guide EQUETRO (ē-kwĕ-trō) (carbamazepine) Extended-Release Capsules Read this Medication Guide before you start taking EQUETRO and each time you get a refill. There may be new information. This
Medication Guide EQUETRO (ē-kwĕ-trō) (carbamazepine) Extended-Release Capsules Read this Medication Guide before you start taking EQUETRO and each time you get a refill. There may be new information. This
MS Treatments Aubagio TM
 1 MSology Essentials Series Aubagio TM (teriflunomide) Developed by MSology with the invaluable assistance of multiple sclerosis nurse advisors: Bonnie Blain Central Alberta MS Clinic, Red Deer, Alberta
1 MSology Essentials Series Aubagio TM (teriflunomide) Developed by MSology with the invaluable assistance of multiple sclerosis nurse advisors: Bonnie Blain Central Alberta MS Clinic, Red Deer, Alberta
Eastern Health MS Service. Tysabri Therapy. Information for People with MS and their Families
 Eastern Health MS Service Tysabri Therapy Information for People with MS and their Families The Eastern Health MS Service has developed this information for you as a guide through what will happen to you
Eastern Health MS Service Tysabri Therapy Information for People with MS and their Families The Eastern Health MS Service has developed this information for you as a guide through what will happen to you
Guy s, King s and St Thomas Cancer Centre The Cancer Outpatient Clinic Maintenance BCG for nonmuscle invasive bladder cancer
 Guy s, King s and St Thomas Cancer Centre The Cancer Outpatient Clinic Maintenance BCG for nonmuscle invasive bladder cancer This information sheet has been given to you to explain the use of maintenance
Guy s, King s and St Thomas Cancer Centre The Cancer Outpatient Clinic Maintenance BCG for nonmuscle invasive bladder cancer This information sheet has been given to you to explain the use of maintenance
TC Chemotherapy Regimen (Docetaxel + Cyclophosphamide)
 TC Chemotherapy Regimen (Docetaxel + Cyclophosphamide) TC is a regimen or treatment plan that includes a combination of chemotherapy drugs that your doctor prescribed for the treatment of your cancer.
TC Chemotherapy Regimen (Docetaxel + Cyclophosphamide) TC is a regimen or treatment plan that includes a combination of chemotherapy drugs that your doctor prescribed for the treatment of your cancer.
The degree of liver inflammation or damage (grade) Presence and extent of fatty liver or other metabolic liver diseases
 ilearning about your health Liver Biopsy www.cpmc.org/learning What is a Liver Biopsy? A liver biopsy is a procedure where a specially trained doctor (typically a hepatologist, radiologist, or gastroenterologist)
ilearning about your health Liver Biopsy www.cpmc.org/learning What is a Liver Biopsy? A liver biopsy is a procedure where a specially trained doctor (typically a hepatologist, radiologist, or gastroenterologist)
Inferior Vena Cava filter and removal
 Inferior Vena Cava filter and removal What is Inferior Vena Cava Filter Placement and Removal? An inferior vena cava filter placement procedure involves an interventional radiologist (a specialist doctor)
Inferior Vena Cava filter and removal What is Inferior Vena Cava Filter Placement and Removal? An inferior vena cava filter placement procedure involves an interventional radiologist (a specialist doctor)
Full name DOB Age Address Email Phone numbers (H) (W) (C) Emergency contact Phone
 DEMOGRAPHIC INFORMATION Full name DOB Age Address Email Phone numbers (H) (W) (C) Emergency contact Phone CARE INFORMATION Primary care physician: Address Phone Fax Referring physician: Specialty Address
DEMOGRAPHIC INFORMATION Full name DOB Age Address Email Phone numbers (H) (W) (C) Emergency contact Phone CARE INFORMATION Primary care physician: Address Phone Fax Referring physician: Specialty Address
What is cervical cancer?
 What is cervical cancer? Cervical cancer is a term used for a tumour that starts in the cervix where the uterus (womb) meets the vagina (birth canal). The cells of the cervix don t suddenly turn into cancer;
What is cervical cancer? Cervical cancer is a term used for a tumour that starts in the cervix where the uterus (womb) meets the vagina (birth canal). The cells of the cervix don t suddenly turn into cancer;
Excision of Vaginal Mesh
 What is excision of vaginal mesh? This procedure is done to remove mesh from the vagina. When is this surgery used? If mesh has eroded into the vagina, bladder, urethra, or bowel If there is pain associated
What is excision of vaginal mesh? This procedure is done to remove mesh from the vagina. When is this surgery used? If mesh has eroded into the vagina, bladder, urethra, or bowel If there is pain associated
Patient Information. If Patient is child, Parent s Name. City State Zip Cell# SS# of Patient Driver s License #
 Patient Information Patient Name Date of Birth If Patient is child, Parent s Name Street Address Male or Female City State Zip Cell# Home# Work# Name of Employer Email Address SS# of Patient Driver s License
Patient Information Patient Name Date of Birth If Patient is child, Parent s Name Street Address Male or Female City State Zip Cell# Home# Work# Name of Employer Email Address SS# of Patient Driver s License
Positron Emission Tomography - For Patients
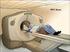 Positron Emission Tomography - For Patients A physician s written order is required for any PET-CT tests. How should I prepare for my PET-CT? PET-CT is more complicated than most other tests you may be
Positron Emission Tomography - For Patients A physician s written order is required for any PET-CT tests. How should I prepare for my PET-CT? PET-CT is more complicated than most other tests you may be
TAKING PART IN CANCER TREATMENT RESEARCH STUDIES
 For more infomation about Cancer Clinical Trials at Upstate Cancer Center please call Upstate Connect 1.800.464.8668 TAKING PART IN CANCER TREATMENT RESEARCH STUDIES Information provided by: National Cancer
For more infomation about Cancer Clinical Trials at Upstate Cancer Center please call Upstate Connect 1.800.464.8668 TAKING PART IN CANCER TREATMENT RESEARCH STUDIES Information provided by: National Cancer
Smoking and misuse of certain pain medicines can affect the risk of developing renal cell cancer.
 Renal cell cancer Renal cell cancer is a disease in which malignant (cancer) cells form in tubules of the kidney. Renal cell cancer (also called kidney cancer or renal adenocarcinoma) is a disease in which
Renal cell cancer Renal cell cancer is a disease in which malignant (cancer) cells form in tubules of the kidney. Renal cell cancer (also called kidney cancer or renal adenocarcinoma) is a disease in which
Life Insurance Application Form
 Life Insurance Application Form INSTRUCTION To be completed by all applicants PERSONAL DETAILS Surname First name Middle name Sex Female Male Marital status (please tick) Single Married Other Current residential
Life Insurance Application Form INSTRUCTION To be completed by all applicants PERSONAL DETAILS Surname First name Middle name Sex Female Male Marital status (please tick) Single Married Other Current residential
IMPORTANT: PLEASE READ
 PART III: CONSUMER INFORMATION combined hepatitis A (inactivated) and hepatitis B (recombinant) vaccine This leaflet is part III of a three-part "Product Monograph" published when was approved for sale
PART III: CONSUMER INFORMATION combined hepatitis A (inactivated) and hepatitis B (recombinant) vaccine This leaflet is part III of a three-part "Product Monograph" published when was approved for sale
Leflunomide Leflunomide
 Drug information Leflunomide Leflunomide This leaflet provides information on leflunomide and will answer any questions you have about the treatment. Arthritis Research UK produce and print our booklets
Drug information Leflunomide Leflunomide This leaflet provides information on leflunomide and will answer any questions you have about the treatment. Arthritis Research UK produce and print our booklets
Research Ethics Review Committee (WHO ERC)
 Research Ethics Review Committee (WHO ERC) 20, AVENUE APPIA CH-1211 GENEVA 27 SWITZERLAND HTTP://INTRANET.WHO.INT/HOMES/RPC/ERC HTTP://WWW.WHO.INT/RPC/RESEARCH_ETHICS Informed Consent Template for Clinical
Research Ethics Review Committee (WHO ERC) 20, AVENUE APPIA CH-1211 GENEVA 27 SWITZERLAND HTTP://INTRANET.WHO.INT/HOMES/RPC/ERC HTTP://WWW.WHO.INT/RPC/RESEARCH_ETHICS Informed Consent Template for Clinical
For the Patient: Dasatinib Other names: SPRYCEL
 For the Patient: Dasatinib Other names: SPRYCEL Dasatinib (da sa' ti nib) is a drug that is used to treat many types of cancer. It is a tablet that you take by mouth. Tell your doctor if you have ever
For the Patient: Dasatinib Other names: SPRYCEL Dasatinib (da sa' ti nib) is a drug that is used to treat many types of cancer. It is a tablet that you take by mouth. Tell your doctor if you have ever
Bone Marrow or Blood Stem Cell Transplants in Children With Severe Forms of Autoimmune Disorders or Certain Types of Cancer
 Bone Marrow or Blood Stem Cell Transplants in Children With Severe Forms of Autoimmune Disorders or Certain Types of Cancer A Review of the Research for Parents and Caregivers Is This Information Right
Bone Marrow or Blood Stem Cell Transplants in Children With Severe Forms of Autoimmune Disorders or Certain Types of Cancer A Review of the Research for Parents and Caregivers Is This Information Right
Galerie Dental Care. Patient Information. Emergency Contact Relationship: Phone:
 Galerie Dental Care Patient Information Date: Patient Name: Last First Middle Initial (Preferred Name) Gender: Birth Date: Marital/Family Status Address: Street Apartment # City Province Postal Code Phone
Galerie Dental Care Patient Information Date: Patient Name: Last First Middle Initial (Preferred Name) Gender: Birth Date: Marital/Family Status Address: Street Apartment # City Province Postal Code Phone
TC: Docetaxel and Cyclophosphamide
 PATIENT EDUCATION patienteducation.osumc.edu What is TC? It is the short name for the drugs used for this chemotherapy treatment. The two drugs you will receive during this treatment are Docetaxel (Taxotere
PATIENT EDUCATION patienteducation.osumc.edu What is TC? It is the short name for the drugs used for this chemotherapy treatment. The two drugs you will receive during this treatment are Docetaxel (Taxotere
MEDICATION GUIDE COUMADIN (COU-ma-din) (warfarin sodium)
 MEDICATION GUIDE COUMADIN (COU-ma-din) (warfarin sodium) Read this Medication Guide before you start taking COUMADIN (warfarin sodium) and each time you get a refill. There may be new information. This
MEDICATION GUIDE COUMADIN (COU-ma-din) (warfarin sodium) Read this Medication Guide before you start taking COUMADIN (warfarin sodium) and each time you get a refill. There may be new information. This
Prostate Cancer Screening. A Decision Guide for African Americans
 Prostate Cancer Screening A Decision Guide for African Americans This booklet was developed by the U.S. Department of Health and Human Services, Centers for Disease Control and Prevention (CDC). Published
Prostate Cancer Screening A Decision Guide for African Americans This booklet was developed by the U.S. Department of Health and Human Services, Centers for Disease Control and Prevention (CDC). Published
PREMIER PAIN CARE PA Carlos J Garcia MD 2435 W. Oak Street # 103 Denton, TX 76201 Phone 940-323-9404 Fax 940-323-9422 PATIENT REGISTRATION
 PREMIER PAIN CARE PA Carlos J Garcia MD 2435 W. Oak Street # 103 Denton, TX 76201 Phone 940-323-9404 Fax 940-323-9422 PATIENT REGISTRATION Last Name First Name MI Mailing Address City Zip code Home Phone
PREMIER PAIN CARE PA Carlos J Garcia MD 2435 W. Oak Street # 103 Denton, TX 76201 Phone 940-323-9404 Fax 940-323-9422 PATIENT REGISTRATION Last Name First Name MI Mailing Address City Zip code Home Phone
RALPH R. GARRAMONE, MD, FACS (239) 482-1900
 Information as of (enter today s date) (Please Print Legibly & Fill In or Correct All Fields) s Name Address First Middle Last Street & Apt # City State Zip Home Phone Cell Phone Other Phone Any restrictions
Information as of (enter today s date) (Please Print Legibly & Fill In or Correct All Fields) s Name Address First Middle Last Street & Apt # City State Zip Home Phone Cell Phone Other Phone Any restrictions
External Radiation Side Effects Worksheet
 Page 1 of 6 External Radiation Side Effects Worksheet Radiation therapy uses special equipment to deliver high doses of radiation to cancerous tumors, killing or damaging them so they cannot grow, multiply,
Page 1 of 6 External Radiation Side Effects Worksheet Radiation therapy uses special equipment to deliver high doses of radiation to cancerous tumors, killing or damaging them so they cannot grow, multiply,
Emory Eye Center New Patient Questionnaire
 Patient Name: Date: Current Address: Current Phone: Date of Birth: Primary Care Physician: Referring Physician: (First & Last Name) (First & Last Name) Pharmacy Name: Phone #: ( ) Please answer all questions
Patient Name: Date: Current Address: Current Phone: Date of Birth: Primary Care Physician: Referring Physician: (First & Last Name) (First & Last Name) Pharmacy Name: Phone #: ( ) Please answer all questions
YTTRIUM 90 MICROSPHERES THERAPY OF LIVER TUMORS
 YTTRIUM 90 MICROSPHERES THERAPY OF LIVER TUMORS The information regarding placement of Yttrium 90 microsphres for the management of liver tumors on the next several pages includes questions commonly asked
YTTRIUM 90 MICROSPHERES THERAPY OF LIVER TUMORS The information regarding placement of Yttrium 90 microsphres for the management of liver tumors on the next several pages includes questions commonly asked
Chemotherapy What It Is, How It Helps
 Chemotherapy What It Is, How It Helps What s in this guide If your doctor has told you that you have cancer, you may have a lot of questions. Can I be cured? What kinds of treatment would be best for me?
Chemotherapy What It Is, How It Helps What s in this guide If your doctor has told you that you have cancer, you may have a lot of questions. Can I be cured? What kinds of treatment would be best for me?
Understanding. Pancreatic Cancer
 Understanding Pancreatic Cancer Understanding Pancreatic Cancer The Pancreas The pancreas is an organ that is about 6 inches long. It s located deep in your belly between your stomach and backbone. Your
Understanding Pancreatic Cancer Understanding Pancreatic Cancer The Pancreas The pancreas is an organ that is about 6 inches long. It s located deep in your belly between your stomach and backbone. Your
Multiple Myeloma. This reference summary will help you understand multiple myeloma and its treatment options.
 Multiple Myeloma Introduction Multiple myeloma is a type of cancer that affects white blood cells. Each year, thousands of people find out that they have multiple myeloma. This reference summary will help
Multiple Myeloma Introduction Multiple myeloma is a type of cancer that affects white blood cells. Each year, thousands of people find out that they have multiple myeloma. This reference summary will help
INFLUENZA (FLU) Flu and You
 Influenza (Flu) What is the flu? The flu is an illness caused by flu viruses. The flu may make people cough and have a sore throat and fever. They may also have a runny or stuffy nose, feel tired, have
Influenza (Flu) What is the flu? The flu is an illness caused by flu viruses. The flu may make people cough and have a sore throat and fever. They may also have a runny or stuffy nose, feel tired, have
