CORPORATE PRESENTATION
|
|
|
- Berenice Bradley
- 8 years ago
- Views:
Transcription
1 CORPORATE PRESENTATION 1537 NW 65th Avenue Plantation, FL USA Phone: (954) Fax: (954)
2 OVERVIEW ReceptoPharm is an innovative biopharmaceutical drug discovery company developing proprietary therapeutic protein products and providing specialized contract services catering to small and start-up biotechnology companies.
3 Introduction ReceptoPharm is a biopharmaceutical drug discovery company based in Plantation, Florida. The Company is primarily engaged in the discovery and development of novel and off-patent peptide technologies for the prevention and treatment of disease in humans. In 2008, ReceptoPharm began offering a full range of contract services catering to small and start-up biotechnology companies. OVERVIEW Drug Development The emphasis of ReceptoPharm's approach to drug discovery and the development of new therapeutic agents is based upon specialized receptor-binding proteins found in nature. With its' large volume of research and preclinical data ReceptoPharm believes it has identified a number of therapeutic products that have the capability to provide significant benefits for patients with neurological, immunological, autoimmune and viral conditions. CRO Services ReceptoPharm offers a full range of contract services catering to start-up and small biotechnology companies. These services include pre-production studies, contract manufacturing services, US & EU regulatory support, and quality systems/gmp certification.
4 Management Team ReceptoPharm s management is comprised of a team of highly experienced businesspeople and scientists with over 100 years of combined industry expertise. OVERVIEW Name Rik J. Deitsch Paul F. Reid, Ph.D. Laurence N. Raymond, Ph.D. Harold Rumph Chairman and Chief Executive Officer, Nutra Pharma Corporation Chief Executive Officer, ReceptoPharm Chief Scientific Officer, ReceptoPharm Director, ReceptoPharm
5 Advisors ReceptoPharm maintains collaborative and research relationships with a number of noted academic researchers associated with the National Institutes of Health (NIH), Harvard University, Massachusetts General Hospital, Yale University, Baylor College of Medicine and Soochow University. OVERVIEW Name Dorothy Bray, Ph.D. Zouhair M. Atassi, Ph.D., D.Sc. Zheng-Hong Qin, MD, Ph.D. Thomas L. Lentz, MD Organization ImmunoClin Ltd. Baylor College of Medicine Harvard University / Massachusetts General Hospital (Boston, Massachusetts) and Soochow University (Suzhou, China) Yale University
6 DRUG DISCOVERY ReceptoPharm conducts research into a number of products with applications for neurological, immunological, autoimmune and viral conditions. The Company s leading drug candidates are RPI-MN and RPI-78M.
7 Drug Discovery Overview ReceptoPharm is an innovative biopharmaceutical drug discovery company developing proprietary therapeutic protein products for the billion-dollar biologics market. The Company has two leading products: RPI-MN and RPI-78M. RPI-MN RPI-MN inhibits the entry of several viruses that are known to cause severe neurological damage in such diseases as encephalitis and Human Immunodeficiency Virus (HIV). It is being developed first for the treatment of HIV. DRUG DISCOVERY RPI-78M RPI-78M is being developed for the treatment of Multiple Sclerosis (MS) and Adrenomyeloneuropathy (AMN). Other neurological disorders that may be served by RPI-78M include Myasthenia Gravis (MG) and Amyotrophic Lateral Sclerosis (ALS). RPI-78M and RPI-MN contain anticholinergic peptides that recognize the same receptors as nicotine (acetylcholine receptors) but have the opposite effect. In a specific chemical process unique to ReceptoPharm, the drugs are created through a process of chemical modification.
8 Benefits of RPI-MN and RPI-78M RPI-MN and RPI-78M possess several desirable properties as drugs: 1. They lack measurable toxicity but are still capable of attaching to and affecting the target site on the nerve cells. This means that patients cannot overdose. 2. They display no serious adverse side effects following years of investigations in humans and animals. DRUG DISCOVERY 3. They are extremely stable and resistant to heat, which gives the drugs a long shelf life. The drugs' stability has been determined to be over 4 years at room temperature. This is extremely unusual for a biologic drug. 4. They are easy to administer. 5. RPI-78M can be administered orally -- a first for a biologic MS drug. This will present MS patients with additional quality of life benefits by eliminating the requirement for routine injections.
9 Specific Benefits of RPI-MN Benefits of ReceptoPharm s HIV/AIDs Treatment RPI-MN Fuzeon by Roche/Trimeris Mutations are not Created Mutations are Created Relatively Inexpensive Very Expensive Twice Current Therapies DRUG DISCOVERY No Significant Adverse Side Effects Long Shelf Life Patient Does Not Develop Resistance Easy to Manufacture Adverse Side Effects Present Some Severe Limited Shelf Life Patient Develops Immune Resistance Difficult Manufacturing Leads to Limited Use
10 Specific Benefits of RPI-78M Benefits of ReceptoPharm s Multiple Sclerosis (MS) Treatment RPI-78M Competitive Drugs Disease Progress Stops Disease Progression Only Slows Patient Condition Improves No Claims Against Patient Improvement DRUG DISCOVERY No Significant Adverse Side Effects Long Shelf Life Resistant to Heat Patient Does Not Develop Resistance Improves Quality of Life Adverse Side Effects Present Some Severe Limited Shelf Life Requires Refrigeration Patient Develops Immune Resistance No Evidence for Quality of Life Improvement
11 Cobroxin Overview Cobroxin is the first over-the-counter pain reliever clinically proven to treat Stage 2 (moderate to severe) chronic pain. Cobroxin, which was developed by ReceptoPharm, was launched in August 2009 and will be available as an oral spray for treating lower back pain, migraines, neck aches, shoulder pain, cramps and neuralgia and as a topical gel for treating joint pain and pain associated with repetitive stress and arthritis. DRUG DISCOVERY Additional Benefits to Cobroxin Include: All Natural Non-Addictive Non-Narcotic Non-Opiate More Potent than Morphine Long Lasting
12 Cobroxin Market Potential Pain is the single most common reason patients seek medical care and accounts for half of all physician office visits in the United States. According to the American Pain Foundation, each year, more than 25 million people in the United States experience acute pain as a result of injury or surgery. Additionally, more than 50 million people in the United States are affected by ongoing chronic pain. DRUG DISCOVERY The Cobroxin Advantage: 1. Makes potent pain relief accessible to: People without healthcare insurance (42 million) People without prescription coverage People afraid of opiate side effects People subject to drug screening for work People for whom OTC NSAIDs didn t work and are just living with the pain Doctors afraid to prescribe opiates 2. Targets consumers not well-served by current products
13 Research & Development Pipeline ReceptoPharm's research and development pipeline consists of several novel therapies in various stages of development to prevent and/or treat Multiple Sclerosis (MS), Human Immunodeficiency Virus (HIV), Adrenomyeloneuropathy (AMN), Herpes, Rheumatoid Arthritis (RA) and Pain. DRUG DISCOVERY Abbreviations: Preclinical (PC), Phase 1 (P-I), Phase 2 (P-II), Phase 3 (P-III)
14 Published Research Cobratoxin Inhibits Pain-Evoked Discharge of Neurons in Thalamic Parafascicular Nucleus in Rats: Involvement of cholinergic and serotonergic systems // Toxicon (April 2009) Alpha Cobratoxin as a Possible Therapy for Multiple Sclerosis: A Review of the Literature leading to Its Development for This Application // Critical Reviews in Immunology (Volume 27 / Issue ) DRUG DISCOVERY Analgesic Effects of Receptin, a Chemically Modified Cobratoxin from Thailand Cobra Venom // Neuroscience Bulletin (September 2006) Amelioration of Acute and Relapsing Stages of the Experimental Allergic Encephalomyelitis by Cobra Toxins // Biomedical Sciences Instrumentation (June 2006) A Long-Form a-neurotoxin from Cobra Venom Produces Potent Opioid-Independent Analgesia // Acta Pharmacologica Sinica (April 2006) A Short-Chain a-neurotoxin from Naja naja atra Produces Potent Cholinergic-Dependent Analgesia // Neuroscience Bulletin (March 2006)
15 CONTRACT RESEARCH SERVICES ReceptoPharm has installed the pathways to develop, produce and supply clinical material for Phase I and II studies at the highest levels. In 2008, the Company began offering services catering to small and start-up biotechnology companies.
16 Partnering with ReceptoPharm As a clinical stage company specializing in biologics, ReceptoPharm has cleared many of the hurdles that face emerging biotech companies. Below are the top reasons that companies choose to partner with ReceptoPharm as its CRO: 1. Therapeutic experience encompassing infectious disease, autoimmune conditions, neurological disorders and oncology indications. 2. A renowned team of scientists, led by Dr. Paul Reid, specializing in human and veterinary regulatory affairs for the United States, Canada, and Europe, and drug applications (pre IND, IND and Orphan), drug production, new drug development, clinical trial design, patient monitoring, and experimental design. CONTRACT RESEARCH SERVICES 3. Significant investment in establishing a production facility to meet US and EU standards, including the installation of quality systems to meet GMP standards and ISO Class 5 certification.
17 A Global View on Drug Development ReceptoPharm prides itself on taking a global view on drug development; whether it s preparing submissions, protocols, formulations, or organizing clinical trials internationally. ReceptoPharm has invested heavily in establishing its own production facility to meet US and EU standards, including the installation of quality systems to meet GMP standards. Its commitment to maintaining the highest quality standards can now be used as a resource by other biopharmaceutical companies. CONTRACT RESEARCH SERVICES
18 Top-Rated Drug Production Facilities Establishing and maintaining a qualified drug production facility can represent a significant drain on a small company s resources. ReceptoPharm understands that its investment in manufacturing and its first-hand knowledge in drug discovery and development can be very valuable assets to small companies that do not have these resources or capabilities inhouse. Working with ReceptoPharm through its ISO class 5 and GMP certified facilities will allow these companies to avoid considerable clinical development expenses and allow them to focus on a more important task, expeditiously getting their products to the next stage. CONTRACT RESEARCH SERVICES
19 Pre-Production Studies Stability Studies Formulation Adventitious Viral Testing Preservative Efficacy Testing Assay Development/Potency Testing Methods Development US & EU Regulatory Support FDA Drug Applications, IND Preparation EU Submissions, IMPD Preparation Sterile Filling Capabilities ReceptoPharm can manually fill a variety of drug products for your clinical development projects in the US and EU. While ReceptoPharm specializes in biologics, including MAbs, natural, recombinant and synthetic proteins, it can also prepare and fill pharmaceutical products in its ISO class 5 cleanroom facilities. Contract Manufacturing Services Drug Formulation ISO 5 Sterile Filling Suite, Single and Multi-Dose Liquids for Injection Clinical Trial Supplies to FDA & EU Requirements Packaging and Shipment to Sites Release Testing and Certification Quality Systems/GMP Certification Installation of Quality Systems Using Our SOPs GMP Audits GMP Training QA Function IQ/OQ/PQ Validations (Equipment and Processes) CONTRACT RESEARCH SERVICES
20 CONTACT RECEPTOPHARM 1537 NW 65th Avenue Plantation, FL USA Phone: (954) Fax: (954)
NEW CHEMICAL ENTITIES
 NEW CHEMICAL ENTITIES PIONEERING PARTNER FOR PEPTIDES With more than 40 years of expertise in peptide synthesis, a track record in process development, large-scale manufacturing and outstanding product
NEW CHEMICAL ENTITIES PIONEERING PARTNER FOR PEPTIDES With more than 40 years of expertise in peptide synthesis, a track record in process development, large-scale manufacturing and outstanding product
NEUROSCIENCE. THE POWER OFxTM. Experts. Experience. Execution. Medpace Neuroscience Expertise. medpace.com
 THE POWER OFxTM Experts. Experience. Execution. Medpace Neuroscience Expertise As a best-in-class strategic partner for sponsors in neuroscience drug and device development, Medpace utilizes key industry
THE POWER OFxTM Experts. Experience. Execution. Medpace Neuroscience Expertise As a best-in-class strategic partner for sponsors in neuroscience drug and device development, Medpace utilizes key industry
exactly. The need for efficiency in developing effective new therapeutics has never been greater.
 exactly. The need for efficiency in developing effective new therapeutics has never been greater. As demands on the global healthcare system increase and treating disease becomes more complex, the research,
exactly. The need for efficiency in developing effective new therapeutics has never been greater. As demands on the global healthcare system increase and treating disease becomes more complex, the research,
Abt Associates Inc. Abt Associates Clinical Trials Comprehensive clinical research services
 Abt Associates Inc. Abt Associates Clinical Trials Comprehensive clinical research services Providing a wide range of clinical research services Abt Associates Clinical Trials (AACT) is dedicated to providing
Abt Associates Inc. Abt Associates Clinical Trials Comprehensive clinical research services Providing a wide range of clinical research services Abt Associates Clinical Trials (AACT) is dedicated to providing
CAN-FITE BIOPHARMA LTD.
 UNITED STATES SECURITIES AND EXCHANGE COMMISSION Washington, D.C. 20549 FORM 6-K Report of Foreign Private Issuer Pursuant to Rule 13a-16 or 15d-16 Under the Securities Exchange Act of 1934 For the Month
UNITED STATES SECURITIES AND EXCHANGE COMMISSION Washington, D.C. 20549 FORM 6-K Report of Foreign Private Issuer Pursuant to Rule 13a-16 or 15d-16 Under the Securities Exchange Act of 1934 For the Month
Clinical and Therapeutic Cannabis Information. Written by Cannabis Training University (CTU) All rights reserved
 Clinical and Therapeutic Cannabis Information Written by Cannabis Training University (CTU) All rights reserved Contents Introduction... 3 Chronic Pain... 6 Neuropathic Pain... 8 Movement Disorders...
Clinical and Therapeutic Cannabis Information Written by Cannabis Training University (CTU) All rights reserved Contents Introduction... 3 Chronic Pain... 6 Neuropathic Pain... 8 Movement Disorders...
BioBusiness Biopharmaceutical Ingredients Recombinant albumins & albumin fusion Dermot Pearson, Strategic Product Director, NZ Biopharma
 BioBusiness Biopharmaceutical Ingredients Recombinant albumins & albumin fusion Dermot Pearson, Strategic Product Director, NZ Biopharma 2 Novozymes solutions dedicated to the biopharmaceutical industry
BioBusiness Biopharmaceutical Ingredients Recombinant albumins & albumin fusion Dermot Pearson, Strategic Product Director, NZ Biopharma 2 Novozymes solutions dedicated to the biopharmaceutical industry
Achieving Regulatory Success: Areas of focus for biotechnology companies. Michael J. Schlosser, PhD, DABT April 21, 2013
 Achieving Regulatory Success: Areas of focus for biotechnology companies Michael J. Schlosser, PhD, DABT April 21, 2013 Regulatory Success Outline Regulatory Initiatives Regulatory Science Pre-Regulatory
Achieving Regulatory Success: Areas of focus for biotechnology companies Michael J. Schlosser, PhD, DABT April 21, 2013 Regulatory Success Outline Regulatory Initiatives Regulatory Science Pre-Regulatory
Annual Report on Form 20-F
 Annual Report on Form 20-F 2 Teva at a Glance Founded in 1901 by three young pharmacists, today Teva Pharmaceutical Industries Ltd. is a leader in the global pharmaceutical industry, providing medicines
Annual Report on Form 20-F 2 Teva at a Glance Founded in 1901 by three young pharmacists, today Teva Pharmaceutical Industries Ltd. is a leader in the global pharmaceutical industry, providing medicines
Monoclonal antibody (mab) products are currently a fast
 Antibody Monoclonal Therapeutics Janice M. Reichert, Ph.D. Monoclonal antibody (mab) products are currently a fast growing category of drugs. Like antibodies produced naturally by the human immune system,
Antibody Monoclonal Therapeutics Janice M. Reichert, Ph.D. Monoclonal antibody (mab) products are currently a fast growing category of drugs. Like antibodies produced naturally by the human immune system,
A vaccine for rheumatoid arthritis
 A vaccine for rheumatoid arthritis Professor Ranjeny Thomas Founder and Director of Dendright Pty Limited Head, Autoimmunity Division, Diamantina Institute, The University of Queensland About Dendright
A vaccine for rheumatoid arthritis Professor Ranjeny Thomas Founder and Director of Dendright Pty Limited Head, Autoimmunity Division, Diamantina Institute, The University of Queensland About Dendright
SERVICES FOR. Devices and Combination Products
 SERVICES FOR Devices and Combination Products How to Contact Us U.S. FACILITIES / CLIENT SERVICES St. Paul 2540 Executive Drive St. Paul, MN 55120 FAX 651.675.2005 Atlanta 1265 Kennestone Circle Marietta,
SERVICES FOR Devices and Combination Products How to Contact Us U.S. FACILITIES / CLIENT SERVICES St. Paul 2540 Executive Drive St. Paul, MN 55120 FAX 651.675.2005 Atlanta 1265 Kennestone Circle Marietta,
EMD Serono Presents New Data on Rebif (Interferon beta-1a) and Multiple Sclerosis Pipeline at Joint ACTRIMS-ECTRIMS Meeting in Boston
 Erin-Marie Beals Phone 1-781-681-2850 September 9, 2014 EMD Serono Presents New Data on Rebif (Interferon beta-1a) and Multiple Sclerosis Pipeline at Joint ACTRIMS-ECTRIMS Meeting in Boston Data include
Erin-Marie Beals Phone 1-781-681-2850 September 9, 2014 EMD Serono Presents New Data on Rebif (Interferon beta-1a) and Multiple Sclerosis Pipeline at Joint ACTRIMS-ECTRIMS Meeting in Boston Data include
Biologic Treatments for Rheumatoid Arthritis
 Biologic Treatments Rheumatoid Arthritis (also known as cytokine inhibitors, TNF inhibitors, IL 1 inhibitor, or Biologic Response Modifiers) Description Biologics are new class of drugs that have been
Biologic Treatments Rheumatoid Arthritis (also known as cytokine inhibitors, TNF inhibitors, IL 1 inhibitor, or Biologic Response Modifiers) Description Biologics are new class of drugs that have been
A Product and Pipeline Analysis of the Multiple Sclerosis Therapeutics Market
 A Product and Pipeline Analysis of the Multiple Sclerosis Therapeutics Market Launch of Several Pipeline Oral Products Could Diminish the Market for Injectable Therapies NEC3-52 November 2014 Contents
A Product and Pipeline Analysis of the Multiple Sclerosis Therapeutics Market Launch of Several Pipeline Oral Products Could Diminish the Market for Injectable Therapies NEC3-52 November 2014 Contents
A leader in the development and application of information technology to prevent and treat disease.
 A leader in the development and application of information technology to prevent and treat disease. About MOLECULAR HEALTH Molecular Health was founded in 2004 with the vision of changing healthcare. Today
A leader in the development and application of information technology to prevent and treat disease. About MOLECULAR HEALTH Molecular Health was founded in 2004 with the vision of changing healthcare. Today
New York Bio Conference 2016. Mark J. Alles Chief Executive Officer
 New York Bio Conference 2016 Mark J. Alles Chief Executive Officer Great Progress AGE 72 World Life Expectancy 2005-2014* 17 new drugs for rare diseases 71.5 71 Two new MS drugs First drug to target root
New York Bio Conference 2016 Mark J. Alles Chief Executive Officer Great Progress AGE 72 World Life Expectancy 2005-2014* 17 new drugs for rare diseases 71.5 71 Two new MS drugs First drug to target root
EMA and Progressive Multifocal Leukoencephalopathy.
 EMA and Progressive Multifocal Leukoencephalopathy. ENCePP Plenary, London 23 November 2011 Presented by: Henry Fitt Head of Coordination & Networking, Pharmacovigilance & Risk Management An agency of
EMA and Progressive Multifocal Leukoencephalopathy. ENCePP Plenary, London 23 November 2011 Presented by: Henry Fitt Head of Coordination & Networking, Pharmacovigilance & Risk Management An agency of
Presented at: Jefferies 2015 Global Healthcare Conference
 Presented at: Jefferies 2015 Global Healthcare Conference Agenda 1 Overview & Service Platforms 2 Adaptability, Scalability & Expansion Plans 3 Best Practices 4 Blue Chip Customer Base 5 Roadmap of Evolution
Presented at: Jefferies 2015 Global Healthcare Conference Agenda 1 Overview & Service Platforms 2 Adaptability, Scalability & Expansion Plans 3 Best Practices 4 Blue Chip Customer Base 5 Roadmap of Evolution
A Genetic Analysis of Rheumatoid Arthritis
 A Genetic Analysis of Rheumatoid Arthritis Introduction to Rheumatoid Arthritis: Classification and Diagnosis Rheumatoid arthritis is a chronic inflammatory disorder that affects mainly synovial joints.
A Genetic Analysis of Rheumatoid Arthritis Introduction to Rheumatoid Arthritis: Classification and Diagnosis Rheumatoid arthritis is a chronic inflammatory disorder that affects mainly synovial joints.
Many people with MS use some form of conventional medical treatment, and many people also use complementary and alternative medicine (CAM).
 Complementary and alternative medicine (CAM) CAM therapies can generally be divided into the following categories: Biologically based therapies (eg, dietary supplements, diets, bee venom therapy, hyperbaric
Complementary and alternative medicine (CAM) CAM therapies can generally be divided into the following categories: Biologically based therapies (eg, dietary supplements, diets, bee venom therapy, hyperbaric
Antares Pharma, Inc. Princeton South Corporate Center 100 Princeton South, Suite 300 Ewing, NJ 08628 NASDAQ : ATRS. Paul
 Antares Pharma, Inc. Princeton South Corporate Center 100 Princeton South, Suite 300 Ewing, NJ 08628 NASDAQ : ATRS Paul Antares Mission: To develop and commercialize self-administered parenteral medicines
Antares Pharma, Inc. Princeton South Corporate Center 100 Princeton South, Suite 300 Ewing, NJ 08628 NASDAQ : ATRS Paul Antares Mission: To develop and commercialize self-administered parenteral medicines
Specialty Drug Care: Case management services in Quebec
 Specialty Drug Care: Case management services in Quebec QUEBEC SUPPORTING HEALTHIER OUTCOMES Your health benefit plan is designed to provide you and your family with financial protection for a variety
Specialty Drug Care: Case management services in Quebec QUEBEC SUPPORTING HEALTHIER OUTCOMES Your health benefit plan is designed to provide you and your family with financial protection for a variety
Bachelor of Science in Pharmaceutical Sciences (BSPS) Program Overview and Internship Requirements
 Bachelor of Science in Pharmaceutical Sciences (BSPS) Program Overview and Internship Requirements Pharmaceutical Sciences Applied sciences that underlie the practice of pharmacy the development, manufacture,
Bachelor of Science in Pharmaceutical Sciences (BSPS) Program Overview and Internship Requirements Pharmaceutical Sciences Applied sciences that underlie the practice of pharmacy the development, manufacture,
Biopharmaceutical Process Evaluated for Viral Clearance
 Authored by S. Steve Zhou, Ph.D. Microbac Laboratories, Inc., Microbiotest Division The purpose of Viral Clearance evaluation is to assess the capability of a manufacturing production process to inactivate
Authored by S. Steve Zhou, Ph.D. Microbac Laboratories, Inc., Microbiotest Division The purpose of Viral Clearance evaluation is to assess the capability of a manufacturing production process to inactivate
National MS Society Information Sourcebook www.nationalmssociety.org/sourcebook
 National MS Society Information Sourcebook www.nationalmssociety.org/sourcebook Chemotherapy The literal meaning of the term chemotherapy is to treat with a chemical agent, but the term generally refers
National MS Society Information Sourcebook www.nationalmssociety.org/sourcebook Chemotherapy The literal meaning of the term chemotherapy is to treat with a chemical agent, but the term generally refers
TABLE OF CONTENTS. Introduction...1. Chapter1 AdvancesinTreatment...2. Chapter2 MedicinesinDevelopment...11. Chapter3 ValueandSpending...
 CANCER TABLE OF CONTENTS Introduction...1 Chapter1 AdvancesinTreatment...2 Chapter2 MedicinesinDevelopment......11 Chapter3 ValueandSpending......15 Chapter4 Conclusion...22 INTRODUCTION Researchers and
CANCER TABLE OF CONTENTS Introduction...1 Chapter1 AdvancesinTreatment...2 Chapter2 MedicinesinDevelopment......11 Chapter3 ValueandSpending......15 Chapter4 Conclusion...22 INTRODUCTION Researchers and
MRC Technology Centre for Therapeutics Discovery
 MRC Technology Centre for Therapeutics Discovery fast-tracking discovery and development of novel drugs from academia Dr Duncan Young Business Development Manager UbiFrance Symposium, October 2012 established
MRC Technology Centre for Therapeutics Discovery fast-tracking discovery and development of novel drugs from academia Dr Duncan Young Business Development Manager UbiFrance Symposium, October 2012 established
Background: Pathways for the Master s Degree (Thesis and Non Thesis)
 Differences between the Non Thesis (Project) and Thesis Options for the M.S. in Pharmacy with an emphasis in BioPharma Regulatory Affairs Offered by the University of Georgia College of Pharmacy Background:
Differences between the Non Thesis (Project) and Thesis Options for the M.S. in Pharmacy with an emphasis in BioPharma Regulatory Affairs Offered by the University of Georgia College of Pharmacy Background:
Performance Profile. Clinical Study Physician. www.celyad.com 1
 Performance Profile Clinical Study Physician www.celyad.com 1 Role: Provide medical guidance and leadership throughout the development and conduct of clinical trials. You will be responsible for overseeing
Performance Profile Clinical Study Physician www.celyad.com 1 Role: Provide medical guidance and leadership throughout the development and conduct of clinical trials. You will be responsible for overseeing
FEDERAL PAIN PORTFOLIO PREVIEW
 FEDERAL PAIN PORTFOLIO PREVIEW Federal Agencies: AHRQ, CDC, DoD, FDA, NIH, VA Interagency Pain Research Coordinating Committee March 27, 2012 IPRCC Charge I. Develop a summary of advances in pain care
FEDERAL PAIN PORTFOLIO PREVIEW Federal Agencies: AHRQ, CDC, DoD, FDA, NIH, VA Interagency Pain Research Coordinating Committee March 27, 2012 IPRCC Charge I. Develop a summary of advances in pain care
Guidance on Investigational Medicinal Products (IMPs) and other medicinal products used in Clinical Trials
 EUROPEAN COMMISSION ENTERPRISE AND INDUSTRY DIRECTORATE-GENERAL Consumer goods Pharmaceuticals Guidance on Investigational Medicinal Products (IMPs) and other medicinal products used in Clinical Trials
EUROPEAN COMMISSION ENTERPRISE AND INDUSTRY DIRECTORATE-GENERAL Consumer goods Pharmaceuticals Guidance on Investigational Medicinal Products (IMPs) and other medicinal products used in Clinical Trials
Winter 2013. Changing landscapes, pipeline products and plan sponsor impact
 Winter 2013 Changing landscapes, pipeline products and plan sponsor impact Changing landscapes, pipeline products and plan sponsor impact The pharmaceutical landscape is changing as is the profile of blockbuster
Winter 2013 Changing landscapes, pipeline products and plan sponsor impact Changing landscapes, pipeline products and plan sponsor impact The pharmaceutical landscape is changing as is the profile of blockbuster
Roche s marketing applications for review of OCREVUS (ocrelizumab) in two forms of multiple sclerosis accepted by EMA and FDA
 Media Release Basel, 28 June 2016 Roche s marketing applications for review of OCREVUS (ocrelizumab) in two forms of multiple sclerosis accepted by EMA and FDA OCREVUS is the first investigational medicine
Media Release Basel, 28 June 2016 Roche s marketing applications for review of OCREVUS (ocrelizumab) in two forms of multiple sclerosis accepted by EMA and FDA OCREVUS is the first investigational medicine
During the course of this presentation, data from certain clinical studies may be shown. These studies were sponsored independently by Generex.
 GENEREX BIOTECHNOLOGY 2009 Annual General Meeting July 30, 2009 Forward Looking Statements During the course of this presentation, it is possible that statements that could be construed as forward-looking
GENEREX BIOTECHNOLOGY 2009 Annual General Meeting July 30, 2009 Forward Looking Statements During the course of this presentation, it is possible that statements that could be construed as forward-looking
FORM 6-K. SECURITIES AND EXCHANGE COMMISSION Washington, D.C. 20549. Report of Foreign Private Issuer
 FORM 6-K SECURITIES AND EXCHANGE COMMISSION Washington, D.C. 20549 Report of Foreign Private Issuer Pursuant to Rule 13a-16 or 15d-16 under the Securities Exchange Act of 1934 For the month of September
FORM 6-K SECURITIES AND EXCHANGE COMMISSION Washington, D.C. 20549 Report of Foreign Private Issuer Pursuant to Rule 13a-16 or 15d-16 under the Securities Exchange Act of 1934 For the month of September
BN201: a paradigm-shift (neuroprotection) in the treatment of neurodegenerative diseases
 Programa Cooperación Farma-Biotech 9º encuentro (4 de julio de 2013) BN201: a paradigm-shift (neuroprotection) in the treatment of neurodegenerative diseases Barcelona, 4 de julio de 2013 Programa Cooperación
Programa Cooperación Farma-Biotech 9º encuentro (4 de julio de 2013) BN201: a paradigm-shift (neuroprotection) in the treatment of neurodegenerative diseases Barcelona, 4 de julio de 2013 Programa Cooperación
Data include post-hoc assessments of controlled studies in relapsing MS regarding evolution of
 September 10, 2014 Contact: Shikha Virdi 905-919-0200 ext. 5504 EMD Serono Presents New Data on Rebif (Interferon beta-1a) and Multiple Sclerosis Pipeline at Joint ACTRIMS-ECTRIMS Meeting in Boston Data
September 10, 2014 Contact: Shikha Virdi 905-919-0200 ext. 5504 EMD Serono Presents New Data on Rebif (Interferon beta-1a) and Multiple Sclerosis Pipeline at Joint ACTRIMS-ECTRIMS Meeting in Boston Data
THE BIOTECH & PHARMACEUTICAL INDUSTRY
 THE BIOTECH & PHARMACEUTICAL INDUSTRY ESSENTIAL CAREERS INFORMATION CALUM LECKIE KATIE BISARO CAREERS CONSULTANTS What we will cover Sector overview Types of role Graduate recruitment trends and issues
THE BIOTECH & PHARMACEUTICAL INDUSTRY ESSENTIAL CAREERS INFORMATION CALUM LECKIE KATIE BISARO CAREERS CONSULTANTS What we will cover Sector overview Types of role Graduate recruitment trends and issues
Valentina Gualato, Ph.D. Process Development Scientist
 COMPANY PRESENTATION Quality and Innovation Valentina Gualato, Ph.D. Process Development Scientist MISSION areta international is a biotech company dedicated to the contract development and manufacturing
COMPANY PRESENTATION Quality and Innovation Valentina Gualato, Ph.D. Process Development Scientist MISSION areta international is a biotech company dedicated to the contract development and manufacturing
Discover more, discover faster. High performance, flexible NLP-based text mining for life sciences
 Discover more, discover faster. High performance, flexible NLP-based text mining for life sciences It s not information overload, it s filter failure. Clay Shirky Life Sciences organizations face the challenge
Discover more, discover faster. High performance, flexible NLP-based text mining for life sciences It s not information overload, it s filter failure. Clay Shirky Life Sciences organizations face the challenge
Disclosures. Consultant and Speaker for Biogen Idec, TEVA Neuroscience, EMD Serrono, Mallinckrodt, Novartis, Genzyme, Accorda Therapeutics
 Mitzi Joi Williams, MD Neurologist MS Center of Atlanta, Atlanta, GA Disclosures Consultant and Speaker for Biogen Idec, TEVA Neuroscience, EMD Serrono, Mallinckrodt, Novartis, Genzyme, Accorda Therapeutics
Mitzi Joi Williams, MD Neurologist MS Center of Atlanta, Atlanta, GA Disclosures Consultant and Speaker for Biogen Idec, TEVA Neuroscience, EMD Serrono, Mallinckrodt, Novartis, Genzyme, Accorda Therapeutics
A NUSAGE-PharmEng Pharmaceutical and Biotechnology Training Program
 A NUSAGE-PharmEng Pharmaceutical and Biotechnology Training Program Shaping Human Capital for Challenges in the Pharmaceutical Industry CLEANING VALIDATION Instructors Loh Kean Chong Rick Ng Date and Time
A NUSAGE-PharmEng Pharmaceutical and Biotechnology Training Program Shaping Human Capital for Challenges in the Pharmaceutical Industry CLEANING VALIDATION Instructors Loh Kean Chong Rick Ng Date and Time
Non-clinical development of biologics
 Aurigon Life Science GmbH Non-clinical development of biologics Requirements, challenges and case studies Committed to Life. Sigrid Messemer vet. med. M4 Seminar March 10 th 2014 Aurigon - your full service
Aurigon Life Science GmbH Non-clinical development of biologics Requirements, challenges and case studies Committed to Life. Sigrid Messemer vet. med. M4 Seminar March 10 th 2014 Aurigon - your full service
Naltrexone Pellet Treatment for Opiate, Heroin, and Alcohol Addiction. Frequently Asked Questions
 Naltrexone Pellet Treatment for Opiate, Heroin, and Alcohol Addiction Frequently Asked Questions What is Naltrexone? Naltrexone is a prescription drug that effectively blocks the effects of heroin, alcohol,
Naltrexone Pellet Treatment for Opiate, Heroin, and Alcohol Addiction Frequently Asked Questions What is Naltrexone? Naltrexone is a prescription drug that effectively blocks the effects of heroin, alcohol,
Enabling Discovery, Development, and translation of treatments for Cognitive Dysfunctions in Depression: A Workshop Session IV
 Enabling Discovery, Development, and translation of treatments for Cognitive Dysfunctions in Depression: A Workshop Session IV Maria Isaac, MASc, MD, PhD, MFPM, Psychiatrist Senior Scientific Officer Institute
Enabling Discovery, Development, and translation of treatments for Cognitive Dysfunctions in Depression: A Workshop Session IV Maria Isaac, MASc, MD, PhD, MFPM, Psychiatrist Senior Scientific Officer Institute
Novel Targeted Immunotherapy Approach for Metastatic Melanoma Under Phase 3 Investigation
 Novel Targeted Immunotherapy Approach for Metastatic Melanoma Under Phase 3 Investigation Hans S. Keirstead President, NeoStem Oncology NeoStem Presented by GTC, a conference production company 626-256-6405
Novel Targeted Immunotherapy Approach for Metastatic Melanoma Under Phase 3 Investigation Hans S. Keirstead President, NeoStem Oncology NeoStem Presented by GTC, a conference production company 626-256-6405
MANAGEMENT OF CHRONIC NON MALIGNANT PAIN
 MANAGEMENT OF CHRONIC NON MALIGNANT PAIN Introduction The Manitoba Prescribing Practices Program (MPPP) recognizes the important role served by physicians in relieving pain and suffering and acknowledges
MANAGEMENT OF CHRONIC NON MALIGNANT PAIN Introduction The Manitoba Prescribing Practices Program (MPPP) recognizes the important role served by physicians in relieving pain and suffering and acknowledges
Emergence of Compassionate Use programmes
 Emergence of Compassionate Use programmes Rosanna Melchior, PharmD, MS IDRAC, Thomson Scientific Overview What is compassionate use (CU) Overview of CU in Europe Authorities and Sponsors Roles and Responsibilities
Emergence of Compassionate Use programmes Rosanna Melchior, PharmD, MS IDRAC, Thomson Scientific Overview What is compassionate use (CU) Overview of CU in Europe Authorities and Sponsors Roles and Responsibilities
Graduate and Postdoctoral Affairs School of Biomedical Sciences College of Medicine. Graduate Certificate. Metabolic & Nutritional Medicine
 Graduate and Postdoctoral Affairs School of Biomedical Sciences College of Medicine Graduate Certificate in Metabolic & Nutritional Medicine Graduate Certificate Metabolic & Nutritional Medicine Purpose
Graduate and Postdoctoral Affairs School of Biomedical Sciences College of Medicine Graduate Certificate in Metabolic & Nutritional Medicine Graduate Certificate Metabolic & Nutritional Medicine Purpose
DRUG APPROVAL PROCESS FOR THE TREATMENT OF ALZHEIMER S DISEASE
 Research DRUG APPROVAL PROCESS FOR THE TREATMENT OF ALZHEIMER S DISEASE There are certain principles that should be followed when involving people with Alzheimer s disease in research. For more information,
Research DRUG APPROVAL PROCESS FOR THE TREATMENT OF ALZHEIMER S DISEASE There are certain principles that should be followed when involving people with Alzheimer s disease in research. For more information,
www.iproteos.com Corporate Presentation November, 2013
 www.iproteos.com Corporate Presentation November, 2013 The company Iproteos is an early-stage drug development company founded in 2011: Spin-Out from Institute for Research in Biomedicine (IRB Barcelona)
www.iproteos.com Corporate Presentation November, 2013 The company Iproteos is an early-stage drug development company founded in 2011: Spin-Out from Institute for Research in Biomedicine (IRB Barcelona)
Introduction to Enteris BioPharma
 Introduction to Enteris BioPharma Enteris BioPharma Intelligent Solutions for Oral Drug Delivery Privately held, New Jersey based biotech company Owned solely by Victory Park Capital, a large Chicago based
Introduction to Enteris BioPharma Enteris BioPharma Intelligent Solutions for Oral Drug Delivery Privately held, New Jersey based biotech company Owned solely by Victory Park Capital, a large Chicago based
Investigational Drugs: Investigational Drugs and Biologics
 : I. PURPOSE The purpose of this policy is to establish procedures for the proper control, storage, use and handling of investigational drugs and biologics to ensure that adequate safeguards are in place
: I. PURPOSE The purpose of this policy is to establish procedures for the proper control, storage, use and handling of investigational drugs and biologics to ensure that adequate safeguards are in place
Guidance for Industry
 Guidance for Industry Container and Closure System Integrity Testing in Lieu of Sterility Testing as a Component of the Stability Protocol for Sterile Products For questions on the content of the guidance,
Guidance for Industry Container and Closure System Integrity Testing in Lieu of Sterility Testing as a Component of the Stability Protocol for Sterile Products For questions on the content of the guidance,
CTC Technology Readiness Levels
 CTC Technology Readiness Levels Readiness: Software Development (Adapted from CECOM s Software Technology Readiness Levels) Level 1: Basic principles observed and reported. Lowest level of software readiness.
CTC Technology Readiness Levels Readiness: Software Development (Adapted from CECOM s Software Technology Readiness Levels) Level 1: Basic principles observed and reported. Lowest level of software readiness.
U.S. Contract Research Outsourcing Market: Trends, Challenges and Competition in the New Decade. N8B7-52 December 2010
 U.S. Contract Research Outsourcing Market: Trends, Challenges and Competition in the New Decade December 2010 Table of Contents Notes on Methodology 8 Market Introduction and Segmentation Introduction
U.S. Contract Research Outsourcing Market: Trends, Challenges and Competition in the New Decade December 2010 Table of Contents Notes on Methodology 8 Market Introduction and Segmentation Introduction
Groundbreaking Collaborative Clinical Trial Launched
 Groundbreaking Collaborative Clinical Trial Launched For immediate release Media Contacts: June 16, 2014 Richard Folkers Alison Hendrie 9:00 a.m., EDT Foundation for the NIH Rubenstein Communications (301)
Groundbreaking Collaborative Clinical Trial Launched For immediate release Media Contacts: June 16, 2014 Richard Folkers Alison Hendrie 9:00 a.m., EDT Foundation for the NIH Rubenstein Communications (301)
From Research Services and Process Development to GMP Manufacturing
 From Research Services and Process Development to GMP Manufacturing P a r ag o n B i o s e r v i c e s, I n c. A contract research and GMP manufacturing organization (CMO) with a focus on the development
From Research Services and Process Development to GMP Manufacturing P a r ag o n B i o s e r v i c e s, I n c. A contract research and GMP manufacturing organization (CMO) with a focus on the development
A Letter from MabVax Therapeutics President and Chief Executive Officer
 A Letter from MabVax Therapeutics President and Chief Executive Officer Dear Fellow Stockholder: You have invested in MabVax Therapeutics because you share our passion for finding new therapies for the
A Letter from MabVax Therapeutics President and Chief Executive Officer Dear Fellow Stockholder: You have invested in MabVax Therapeutics because you share our passion for finding new therapies for the
BOTOX Treatment. for Chronic Migraine. Information for patients and their families. Botulinum Toxin Type A
 BOTOX Treatment Botulinum Toxin Type A for Chronic Migraine Information for patients and their families. Is Chronic Migraine the same as Migraine? Chronic Migraine is similar to migraine as sufferers experience
BOTOX Treatment Botulinum Toxin Type A for Chronic Migraine Information for patients and their families. Is Chronic Migraine the same as Migraine? Chronic Migraine is similar to migraine as sufferers experience
Course Curriculum for Master Degree in Clinical Pharmacy
 Course Curriculum for Master Degree in Clinical Pharmacy The Master Degree in Clinical Pharmacy is awarded by the Faculty of Graduate studies at Jordan University of Science and Technology (JUST) upon
Course Curriculum for Master Degree in Clinical Pharmacy The Master Degree in Clinical Pharmacy is awarded by the Faculty of Graduate studies at Jordan University of Science and Technology (JUST) upon
C 5. chemical development contract research custom synthesis cgmp API manufacturing commercial production. Welcome to
 C 5 chemical development contract research custom synthesis cgmp API manufacturing commercial production Welcome to ChemCon Company profile Company profile ChemCon offers outstanding chemical services
C 5 chemical development contract research custom synthesis cgmp API manufacturing commercial production Welcome to ChemCon Company profile Company profile ChemCon offers outstanding chemical services
BIOTECHNOLOGY OPERATIONS
 BIOTECHNOLOGY OPERATIONS Principles and Practices Michael J. Roy TECHNISCHE INFORMATION SBIBLIOTHEK UNIVERSITATSBIBLIOTHEK HANNOVER CRC Press TaylorStFrancis Croup Boca Raton London New York CRC Press
BIOTECHNOLOGY OPERATIONS Principles and Practices Michael J. Roy TECHNISCHE INFORMATION SBIBLIOTHEK UNIVERSITATSBIBLIOTHEK HANNOVER CRC Press TaylorStFrancis Croup Boca Raton London New York CRC Press
Strategic Consulting Services
 Services 1 Leadership Team Mark Levonyak President mlevonyak@davaonc.com Mobile: 214.460.5051 Martin W. Lee, MD EVP, Clinical Trial Services mlee@davaonc.com Mobile: 952.373.1405 John Eckardt, MD Chief
Services 1 Leadership Team Mark Levonyak President mlevonyak@davaonc.com Mobile: 214.460.5051 Martin W. Lee, MD EVP, Clinical Trial Services mlee@davaonc.com Mobile: 952.373.1405 John Eckardt, MD Chief
How Is OnabotulinumtoxinA Reimbursed For Chronic Migraine? Impact Of FDA Approval And The New CPT Code
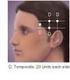 How Is OnabotulinumtoxinA Reimbursed For Chronic Migraine? Impact Of FDA Approval And The New CPT Code Effective January 1, 2013, physicians will be able to report the new CPT code 64615 when performing
How Is OnabotulinumtoxinA Reimbursed For Chronic Migraine? Impact Of FDA Approval And The New CPT Code Effective January 1, 2013, physicians will be able to report the new CPT code 64615 when performing
- Patients treated with alemtuzumab in CARE-MS II were more than twice as likely to experience disability improvement compared to Rebif -
 PRESS RELEASE Significant Improvement in Disability Scores Observed in Multiple Sclerosis Patients Who Received Lemtrada TM* (Alemtuzumab) Compared With Rebif in Phase lll Trial - Patients treated with
PRESS RELEASE Significant Improvement in Disability Scores Observed in Multiple Sclerosis Patients Who Received Lemtrada TM* (Alemtuzumab) Compared With Rebif in Phase lll Trial - Patients treated with
Objectives: Immunity Gone Wrong: Autoimmune Diseases in Dental Hygiene Practice
 Objectives: 1) Understand the concept of self- tolerance versus non- self- tolerance. 2) Recognize systemic and oral indicators of autoimmune diseases. 3) Identify various autoimmune diseases and their
Objectives: 1) Understand the concept of self- tolerance versus non- self- tolerance. 2) Recognize systemic and oral indicators of autoimmune diseases. 3) Identify various autoimmune diseases and their
The 505(b)(2) Drug Development Pathway:
 The 505(b)(2) Drug Development Pathway: When and How to Take Advantage of a Unique American Regulatory Pathway By Mukesh Kumar, PhD, RAC and Hemant Jethwani, MS The 505(b)(2) regulation offers a less expensive
The 505(b)(2) Drug Development Pathway: When and How to Take Advantage of a Unique American Regulatory Pathway By Mukesh Kumar, PhD, RAC and Hemant Jethwani, MS The 505(b)(2) regulation offers a less expensive
To be taught at the Ausia Biotech Aseptic Training Center in Hangzhou China
 Come learn from our 3-day course! Introduction to Aseptic Processing To be taught at the Ausia Biotech Aseptic Training Center in Hangzhou China March 23-25, 2016 1. Introduction to TPA-IT: China s only
Come learn from our 3-day course! Introduction to Aseptic Processing To be taught at the Ausia Biotech Aseptic Training Center in Hangzhou China March 23-25, 2016 1. Introduction to TPA-IT: China s only
Catalent Biologics & Clinical Supplies The SMART Solution
 Catalent Biologics & Clinical Supplies The SMART Solution Advanced Technology and Integrated Solutions From DNA to Clinical Supply & Cold Chain Distribution Dr. Florian Schwaak Account Manager Germany
Catalent Biologics & Clinical Supplies The SMART Solution Advanced Technology and Integrated Solutions From DNA to Clinical Supply & Cold Chain Distribution Dr. Florian Schwaak Account Manager Germany
Guidelines for the Use of Controlled Substances in the Treatment of Pain Adopted by the New Hampshire Medical Society, July 1998
 Guidelines for the Use of Controlled Substances in the Treatment of Pain Adopted by the New Hampshire Medical Society, July 1998 Section I: Preamble The New Hampshire Medical Society believes that principles
Guidelines for the Use of Controlled Substances in the Treatment of Pain Adopted by the New Hampshire Medical Society, July 1998 Section I: Preamble The New Hampshire Medical Society believes that principles
PharmD Postdoctoral Fellowship Program
 Novartis Institutes for Biomedical Research and Massachusetts College of Pharmacy and Health Sciences, Worcester/Manchester PharmD Postdoctoral Fellowship Program Introduction Fellowship training in the
Novartis Institutes for Biomedical Research and Massachusetts College of Pharmacy and Health Sciences, Worcester/Manchester PharmD Postdoctoral Fellowship Program Introduction Fellowship training in the
MULTIPLE SCLEROSIS AUSTRALIA MULTIPLE SCLEROSIS RESEARCH AUSTRALIA
 MULTIPLE SCLEROSIS AUSTRALIA MULTIPLE SCLEROSIS RESEARCH AUSTRALIA Submission to the ACT Legislative Assembly Health, Ageing, Community and Social Services Inquiry into the exposure draft of the Drugs
MULTIPLE SCLEROSIS AUSTRALIA MULTIPLE SCLEROSIS RESEARCH AUSTRALIA Submission to the ACT Legislative Assembly Health, Ageing, Community and Social Services Inquiry into the exposure draft of the Drugs
PX Therapeutics : the partner for early stage biotherapeutics development Biotuesday, May 5 2009
 PX Therapeutics : the partner for early stage biotherapeutics development Biotuesday, May 5 2009 Christelle Dagoneau, PhD Business Development Director Company Profile Protein expert incorporated in 2000
PX Therapeutics : the partner for early stage biotherapeutics development Biotuesday, May 5 2009 Christelle Dagoneau, PhD Business Development Director Company Profile Protein expert incorporated in 2000
LFB GROUP & SANOFI combine their bioproduction capabilities to provide integrated CMO services for biopharmaceuticals
 LFB GROUP & SANOFI combine their bioproduction capabilities to provide integrated CMO services for biopharmaceuticals MAbLaunch TM is a joint bioproduction platform combining LFB Biomanufacturing (LFB
LFB GROUP & SANOFI combine their bioproduction capabilities to provide integrated CMO services for biopharmaceuticals MAbLaunch TM is a joint bioproduction platform combining LFB Biomanufacturing (LFB
THE VIRAL HEPATITIS CONGRESS 2015. 10 12 September 2015, Kap Europa, Frankfurt, Germany THE VIRAL HEPATITIS CONGRESS. Scientific Programme
 THE VIRAL HEPATITIS CONGRESS THE VIRAL HEPATITIS CONGRESS 2015 10 12 September 2015, Kap Europa, Frankfurt, Germany Scientific Programme PRE-CONGRESS WORKSHOP SPONSOR ACHILLION Achillion is seeking to
THE VIRAL HEPATITIS CONGRESS THE VIRAL HEPATITIS CONGRESS 2015 10 12 September 2015, Kap Europa, Frankfurt, Germany Scientific Programme PRE-CONGRESS WORKSHOP SPONSOR ACHILLION Achillion is seeking to
Job Profile Clinical Research Associate III (CRA)
 PART 1 - PROFILE You are an experienced CRA with a strong background in Clinical Research who is passionate about drug development and are seeking a challenging and rewarding career in a quality focussed
PART 1 - PROFILE You are an experienced CRA with a strong background in Clinical Research who is passionate about drug development and are seeking a challenging and rewarding career in a quality focussed
Guidance for Industry
 Guidance for Industry IND Exemptions for Studies of Lawfully Marketed Drug or Biological Products for the Treatment of Cancer U.S. Department of Health and Human Services Food and Drug Administration Center
Guidance for Industry IND Exemptions for Studies of Lawfully Marketed Drug or Biological Products for the Treatment of Cancer U.S. Department of Health and Human Services Food and Drug Administration Center
FORM 6-K. SECURITIES AND EXCHANGE COMMISSION Washington, D.C. 20549. Report of Foreign Private Issuer
 FORM 6-K SECURITIES AND EXCHANGE COMMISSION Washington, D.C. 20549 Report of Foreign Private Issuer Pursuant to Rule 13a-16 or 15d-16 under the Securities Exchange Act of 1934 For the month of August 2011
FORM 6-K SECURITIES AND EXCHANGE COMMISSION Washington, D.C. 20549 Report of Foreign Private Issuer Pursuant to Rule 13a-16 or 15d-16 under the Securities Exchange Act of 1934 For the month of August 2011
Two-Year Phase III Data Presented at AAN 61st Annual Meeting Show Positive Outcome of Cladribine Tablets in Patients with Multiple Sclerosis
 Your contact News Release Barbara Fry Phone +1 905 919 0163 April 29/30, 2009 Two-Year Phase III Data Presented at AAN 61st Annual Meeting Show Positive Outcome of Cladribine Tablets in Patients with Multiple
Your contact News Release Barbara Fry Phone +1 905 919 0163 April 29/30, 2009 Two-Year Phase III Data Presented at AAN 61st Annual Meeting Show Positive Outcome of Cladribine Tablets in Patients with Multiple
US Perspective on the Regulatory Assessment of Benefit-Risk of Vaccines
 US Perspective on the Regulatory Assessment of Benefit-Risk of Vaccines Norman W. Baylor Biologics Consulting Group, Inc. 23-25 June 2014 Foundation Merieux Conference Center OUTLINE OF PRESENTATION Introduction
US Perspective on the Regulatory Assessment of Benefit-Risk of Vaccines Norman W. Baylor Biologics Consulting Group, Inc. 23-25 June 2014 Foundation Merieux Conference Center OUTLINE OF PRESENTATION Introduction
CDRD and the MSSC Seek Collaborative Projects to Discover and Develop New Treatments for Progressive Multiple Sclerosis
 CDRD and the MSSC Seek Collaborative Projects to Discover and Develop New Treatments for Progressive Multiple Sclerosis The Centre for Drug Research and Development (CDRD) and the Multiple Sclerosis Society
CDRD and the MSSC Seek Collaborative Projects to Discover and Develop New Treatments for Progressive Multiple Sclerosis The Centre for Drug Research and Development (CDRD) and the Multiple Sclerosis Society
9. WestLB Deutschland Conference. Frankfurt November 16, 2011
 9. WestLB Deutschland Conference Frankfurt November 16, 2011 Safe Harbour This presentation includes forward-looking statements. Actual results could differ materially from those included in the forward-looking
9. WestLB Deutschland Conference Frankfurt November 16, 2011 Safe Harbour This presentation includes forward-looking statements. Actual results could differ materially from those included in the forward-looking
Technology Transfer of CMC Activities for MAb Manufacturing. 2010 ge healthcare (www.gelifesciences.com)
 M a n u f a c t u r i n g OPERATIONS Technology Transfer of CMC Activities for MAb Manufacturing by Patricia Seymour, Susan Dana Jones, Howard L. Levine With combined 2009 revenues estimated to be over
M a n u f a c t u r i n g OPERATIONS Technology Transfer of CMC Activities for MAb Manufacturing by Patricia Seymour, Susan Dana Jones, Howard L. Levine With combined 2009 revenues estimated to be over
Like cocaine, heroin is a drug that is illegal in some areas of the world. Heroin is highly addictive.
 Heroin Introduction Heroin is a powerful drug that affects the brain. People who use it can form a strong addiction. Addiction is when a drug user can t stop taking a drug, even when he or she wants to.
Heroin Introduction Heroin is a powerful drug that affects the brain. People who use it can form a strong addiction. Addiction is when a drug user can t stop taking a drug, even when he or she wants to.
What You Need to Know About Lung Cancer Immunotherapy
 What You Need to Know About Lung Cancer Immunotherapy Lung.org/immunotherapy What is immunotherapy? Immunotherapy for cancer, sometimes called immune-oncology, is a type of medicine that treats cancer
What You Need to Know About Lung Cancer Immunotherapy Lung.org/immunotherapy What is immunotherapy? Immunotherapy for cancer, sometimes called immune-oncology, is a type of medicine that treats cancer
FACT SHEET TESTETROL, A NOVEL ORALLY BIOACTIVE ANDROGEN
 FACT SHEET TESTETROL, A NOVEL ORALLY BIOACTIVE ANDROGEN General Pantarhei Bioscience B.V. is an emerging specialty pharmaceutical company with a creative approach towards drug development. The Company
FACT SHEET TESTETROL, A NOVEL ORALLY BIOACTIVE ANDROGEN General Pantarhei Bioscience B.V. is an emerging specialty pharmaceutical company with a creative approach towards drug development. The Company
2009 Foley & Lardner LLP Attorney Advertising Prior results do not guarantee a similar outcome Models used are not clients but may be representative
 Attorney Advertising Prior results do not guarantee a similar outcome Models used are not clients but may be representative of clients 321 N. Clark Street, Suite 2800, Chicago, IL 60654 312.832.4500 Life
Attorney Advertising Prior results do not guarantee a similar outcome Models used are not clients but may be representative of clients 321 N. Clark Street, Suite 2800, Chicago, IL 60654 312.832.4500 Life
Recruitment Start date: April 2010 End date: Recruitment will continue until enrolment is fully completed
 Apitope study The study drug (ATX-MS-1467) is a new investigational drug being tested as a potential treatment for relapsing forms of multiple sclerosis (RMS). The term investigational drug means it has
Apitope study The study drug (ATX-MS-1467) is a new investigational drug being tested as a potential treatment for relapsing forms of multiple sclerosis (RMS). The term investigational drug means it has
University of Nebraska Prescription Drug Program 2014
 University of Nebraska Prescription Drug Program 2014 The University of Nebraska s prescription benefit program is administered by CVS Caremark, a leading national provider of prescription drug benefit
University of Nebraska Prescription Drug Program 2014 The University of Nebraska s prescription benefit program is administered by CVS Caremark, a leading national provider of prescription drug benefit
Company Presentation
 Company Presentation DISCLAIMER This document contains forward-looking statements on overall economic development as well as on the business, earnings, financial and asset situation of amp biosimilars
Company Presentation DISCLAIMER This document contains forward-looking statements on overall economic development as well as on the business, earnings, financial and asset situation of amp biosimilars
Providing Trusted and Innovative Solutions t o the Life Science Communities
 Providing Trusted and Innovative Solutions t o the Life Science Communities Eurogentec Eurogentec is a leading supplier of trusted and innovative reagents, kits, specialty products and custom research/development
Providing Trusted and Innovative Solutions t o the Life Science Communities Eurogentec Eurogentec is a leading supplier of trusted and innovative reagents, kits, specialty products and custom research/development
The Clinical Trials Process an educated patient s guide
 The Clinical Trials Process an educated patient s guide Gwen L. Nichols, MD Site Head, Oncology Roche TCRC, Translational and Clinical Research Center New York DISCLAIMER I am an employee of Hoffmann-
The Clinical Trials Process an educated patient s guide Gwen L. Nichols, MD Site Head, Oncology Roche TCRC, Translational and Clinical Research Center New York DISCLAIMER I am an employee of Hoffmann-
GLP vs GMP vs GCP Dominique Pifat, Ph.D., MBA The Biologics Consulting Group dpifat@bcg-usa.com
 GLP vs GMP vs GCP Dominique Pifat, Ph.D., MBA The Biologics Consulting Group dpifat@bcg-usa.com Common Misconception Good Laboratory Practices 1) A quality system concerned with the organizational process
GLP vs GMP vs GCP Dominique Pifat, Ph.D., MBA The Biologics Consulting Group dpifat@bcg-usa.com Common Misconception Good Laboratory Practices 1) A quality system concerned with the organizational process
Drug Development Process
 Drug Development Process Original Arthur: Addie D. Anderson CRB Consulting Engineers, Inc. Overview Important milestones establishing our current system of regulations Step-by-step overview of the drug
Drug Development Process Original Arthur: Addie D. Anderson CRB Consulting Engineers, Inc. Overview Important milestones establishing our current system of regulations Step-by-step overview of the drug
Targeting Cancer: Innovation in the Treatment of Chronic Myelogenous Leukemia EXECUTIVE SUMMARY. New England Healthcare Institute
 Targeting Cancer: Innovation in the Treatment of Chronic Myelogenous Leukemia New England Healthcare Institute NEHI Innovation Series March 2004 Executive Summary From drugs and medical devices, to information
Targeting Cancer: Innovation in the Treatment of Chronic Myelogenous Leukemia New England Healthcare Institute NEHI Innovation Series March 2004 Executive Summary From drugs and medical devices, to information
Decision to Continue the Development of Tecemotide (L-BLP25) in Non-Small Cell Lung Cancer to be Announced
 September 27, 2013 ONO PHARMACEUTICAL CO., LTD. Corporate Communications Phone: +81-6-6263-5670 Decision to Continue the Development of Tecemotide (L-BLP25) in Non-Small Cell Lung Cancer to be Announced
September 27, 2013 ONO PHARMACEUTICAL CO., LTD. Corporate Communications Phone: +81-6-6263-5670 Decision to Continue the Development of Tecemotide (L-BLP25) in Non-Small Cell Lung Cancer to be Announced
Not All Clinical Trials Are Created Equal Understanding the Different Phases
 Not All Clinical Trials Are Created Equal Understanding the Different Phases This chapter will help you understand the differences between the various clinical trial phases and how these differences impact
Not All Clinical Trials Are Created Equal Understanding the Different Phases This chapter will help you understand the differences between the various clinical trial phases and how these differences impact
