Prof.M.Perucca CORSO DI APPROFONDIMENTO DI FISICA ATOMICA: (III-INCONTRO) RISONANZA MAGNETICA NUCLEARE
|
|
|
- Merry Gaines
- 8 years ago
- Views:
Transcription
1 Prof.M.Perucca CORSO DI APPROFONDIMENTO DI FISICA ATOMICA: (III-INCONTRO) RISONANZA MAGNETICA NUCLEARE
2 SUMMARY (I/II) Angular momentum and the spinning gyroscope stationary state equation Magnetic dipole and Bohr Magnetron Torque and energy of magnetic dipole(s) immersed in a stationary magnetic field Larmor precession Space quantisation From spectral triplets to doublets: the need of the spin quantum number
3 SUMMARY (II/II) The Landè splitting factor Bloch equations and resonance The electron spin resonance (ESR) apparatus The ESR experience with DPPH free radical Down to the nucleus: NMR Applications of NMR & one of the cutting edge research topics The NMR experiment with Glycerine, PTFE, PS
4 ANGULAR MOMENTUM
5 MAGNETIC MOMENTUM m = Ip r 2 =IA
6 GYROSCOPE PRECESSION sin L P
7 The orbital angular momentum for an atomic electron can be visualized in terms of a vector model where the angular momentum vector is seen as precessing (wobbling) about a direction in space. While the angular momentum vector has the magnitude shown, only a maximum of l units can be measured along a given direction, where l is the orbital quantum number. THE ORBITAL QUANTUM ANGULAR MOMENTUM
8 The orbital angular momentum is a "vector", actually it is a special kind of vector because it's projection along a direction in space is quantized to values one unit of angular momentum apart. The diagram shows that the possible values for the "magnetic quantum number" m l for l=2 can take the values m l = -2, -1, 0, 1, 2 or, in general m l = -l, -l+1,..., l-1, l. THE ANGULAR MOMENTUM AND THE MAGNETIC MOMENTUM
9 A magnetic moment is associated with the orbital angular momentum, the precession can be compared to the precession of a classical magnetic moment caused by the torque exerted by a magnetic field. This precession is called Larmor precession and has a characteristic frequency called the Larmor frequency Potential energy and energy separation between parallel and antiparallel LARMOR PRECESSION AND ASSOCIATED ENERGY
10 LARMOR (RESONANCE) FREQUENCY w = g B ; g giromagnetic ratio
11 THE TOTAL ANGULAR MOMENTUM
12 ORBITAL ANGULAR AND MAGNETIC MOMENTA m = Ip r 2 =IA
13 A magnetic moment experiences a torque in a magnetic field B. The energy of the interaction can be expressed as The interaction energy of an atomic electron can then be written MAGNETIC MOMENT AND INTERACTION WITH STATIONARY MAGNETIC FIELD
14 MAGNETIC MOMENTS
15 BOHR MAGNETRON AND LANDÈ SPLITTING FACTOR
16 ENERGY INVOLVED IN STATES TRANSITION evaluated in terms of the relevant quantum numbers takes the form g L = Landè splitting factor m B =Bohor magnetron m j = magnetic quantum number
17 THE TOTAL ANGULAR MOMENTUM J Once combined orbital and spin angular momenta according to the vector model, the resulting total angular momentum can be visuallized as precessing about any externally applied magnetic field.
18 BLOCH EQUATIONS y M n 1 m i
19 RESONANCE PEAKS
20 RESONANCE INTENSITY FUNCTION
21 BOLTZMAN DISTRIBUTION the Boltzmann distribution is a certain distribution function or probability measure for the distribution of the states of a system. It underpins the concept of the canonical ensemble, providing its underlying distribution. In more general mathematical settings, the Boltzmann distribution is also known as the Gibbs measure. The Boltzmann distribution for the fractional number of particles N i / N occupying a set of states i possessing energy E i is:
22 TWO STATES POPULATIONS
23 THE (REGULAR) ZEEMAN EFFECT IN H-ATOM When an external magnetic field is applied, sharp spectral lines like the n=3 2 transition of hydrogen split into multiple closely spaced lines. First observed by Pieter Zeeman, this splitting is attributed to the interaction between the magnetic field and the magnetic dipole moment associated with the orbital angular momentum.
24 THE ATOM INTERACTS WITH ITSELF. Normal" Zeeman effect This type of splitting is observed with hydrogen and the Zinc singlet. But another anomalous Zeeman Effect was detected in the Na doublet
25 SODIUM DOUBLET (SPIN-ORBITS INTERACTION)
26 SELECTION RULES In spectral phenomena such as the Zeeman effect it becomes evident that transitions are not observed between all pairs of energy levels. Some transitions are "forbidden" ( i.e., highly improbable) while others are "allowed" by a set of selection rules. The number of split components observed in the Zeeman effect is consistent with the selection rules: any electron transition which involves the emission of a photon must involve a change of 1 in the angular momentum. The photon is said to have an intrinsic angular momentum or "spin" of one, so that conservation of angular momentum in photon emission requires a change of 1 in the atom's angular momentum. The electron spin quantum number does not change in such transitions, so an additional selection rule is: The total angular momentum may change be either zero or one: (An exception to this last selection rule it that you cannot have a transition from j=0 to j=0; i.e., since the vector angular momentum must change by one unit in a electronic transition, j=0 -> 0 can't happen because there is no total angular momentum to re-orient to get a change of 1).
27 ORBITAL AND SPIN MAGNETIC MOMENTS
28 OSCILLATING CIRCUITS PRODUCTING RF SIGNAL
29 \
30 CW NMR One of the earliest and simplest applications of nuclear magnetic resonanceis the continuous wave or CW NMR. In this kind of application, both the magnetic field and the RF excitation signal are continuously on. Either the magnetic field is kept constant and the frequency of the RF excitation is varied, or the magnetic field is varied while keeping the RF input frequency constant. Either approach allows you to sweep through the resonance of the sample under investigation.
31 ESR NMR CONSOLE
32
33 DPPH (ESR) DPPH is a common abbreviation for an organic chemical compound 2,2-diphenyl-1- picrylhydrazyl. It is a dark-colored crystalline powder composed of stable free-radicalmolecules. DPPH has two major applications, both in laboratory research: one is a monitor of chemical reactions involving radicals and another is a standard of the position and intensity of electron paramagnetic resonance signals
34 POLYSTIRENE (NMR) Polystyrene is a long chain hydrocarbon with every other carbon connected to a phenyl group (the name given to the aromatic ring benzene, when bonded to complex carbon substituents). Polystyrene's chemical formula is (C 8 H 8 ) n ; it contains the chemical elements carbon and hydrogen. In NMR hydrogen spin nuclei are excited
35 GLYCERINE (NMR) Glycerol (or glycerin, glycerine) is a simple polyol compound. It is a colorless, odorless, viscous liquid that is widely used in pharmaceutical formulations. Glycerol has three hydrophilic hydroxyl groups that are responsible for its solubility in water and itshygroscopic nature. The glycerol backbone is central to all lipids known as triglycerides. In NMR hydrogen spin nuclei are excited
36 POLYETHYLENETEREPHTALATE - PET (NMR) Polyethylene terephthalate (sometimes written poly(ethylene terephthalate)), commonly abbreviated PET, PETE, or the obsolete PETP or PET-P, is a thermoplastic polymer resin of the polyester family and is used insynthetic fibers; beverage, food and other liquid containers; thermoforming applications; and engineering resins often in combination with glass fiber. In NMR hydrogen spin nuclei are excited
37 MRI Proton nuclear magnetic resonance (NMR) detects the presence of hydrogens (protons) by subjecting them to a large magnetic field to partially polarize the nuclear spins, then exciting the spins with properly tuned radio frequency (RF) radiation, and then detecting weak radio frequency radiation from them as they "relax" from this magnetic interaction. The frequency of this proton "signal" is proportional to the magnetic field to which they are subjected during this relaxation process.
38 MRI In the medical application known as Magnetic Resonance Imaging (MRI), an image of a cross-section of tissue can be made by producing a well-calibrated magnetic field gradient across the tissue so that a certain value of magnetic field can be associated with a given location in the tissue. Since the proton signal frequency is proportional to that magnetic field, a given proton signal frequency can be assigned to a location in the tissue. This provides the information to map the tissue in terms of the protons present there. Since the proton density varies with the type of tissue, a certain amount of contrast is achieved to image the organs and other tissue variations in the subject tissue.
39 Since the MRI uses proton NMR, it images the concentration of protons. Many of those protons are the protons in water, so MRI is particularly well suited for the imaging of soft tissue, like the brain, eyes, and other soft tissue structures in the head as shown at left. The bone of the skull doesn't have many protons, so it shows up dark. Also the sinus cavities image as a dark region. MAGNETIC RESONANCE IMAGING (NMR-> MRI)
40 LOCATING PROTONS THROUGH GRAD(B)
41 LOCATING H ATOMS AND TISSUES there are two regions of the sample which contain enough hydrogen atoms to produce a strong NMR signal. The top sketch visualizes an NMR process with a constant magnetic field applied to the entire sample. The hydrogen spin-flip frequency is then the same for all parts of the sample. Once excited by the RF signal, the hydrogens will tend to return to their lower state in a process called "relaxation" and will reemit RF radiation at their Larmor frequency. This signal is detected as a function of time, and then is converted to signal strength as a function of frequency by means of a Fourier transformation. Since the protons in each of the active areas of the sample are subjected to the same magnetic field, they will produce the same frequency of radiation and the Fourier transform of the detected signal will have only one peak. This one peak demonstrates the presence of hydrogen atoms, but gives no information to locate them in the sample
42 ROTATING B When a rotating field gradient is used, linear positioning information is collected along a number of different directions. That information can be combined to produce a two-dimensional map of the proton densities. The proton NMR signals are quite sensitive to differences in proton content that are characteristic of different kinds of tissue. Even though the spatial resolution of MRI is not as great as a conventional x-ray film, its contrast resolution is much better for tissue. Rapid scanning and computer reconstruction give wellresolved images of organs.
43 THANK YOU FOR YOUR INTEREST AND ATTENTION!
NMR SPECTROSCOPY. Basic Principles, Concepts, and Applications in Chemistry. Harald Günther University of Siegen, Siegen, Germany.
 NMR SPECTROSCOPY Basic Principles, Concepts, and Applications in Chemistry Harald Günther University of Siegen, Siegen, Germany Second Edition Translated by Harald Günther JOHN WILEY & SONS Chichester
NMR SPECTROSCOPY Basic Principles, Concepts, and Applications in Chemistry Harald Günther University of Siegen, Siegen, Germany Second Edition Translated by Harald Günther JOHN WILEY & SONS Chichester
Basic Principles of Magnetic Resonance
 Basic Principles of Magnetic Resonance Contents: Jorge Jovicich jovicich@mit.edu I) Historical Background II) An MR experiment - Overview - Can we scan the subject? - The subject goes into the magnet -
Basic Principles of Magnetic Resonance Contents: Jorge Jovicich jovicich@mit.edu I) Historical Background II) An MR experiment - Overview - Can we scan the subject? - The subject goes into the magnet -
NMR - Basic principles
 NMR - Basic principles Subatomic particles like electrons, protons and neutrons are associated with spin - a fundamental property like charge or mass. In the case of nuclei with even number of protons
NMR - Basic principles Subatomic particles like electrons, protons and neutrons are associated with spin - a fundamental property like charge or mass. In the case of nuclei with even number of protons
Precession of spin and Precession of a top
 6. Classical Precession of the Angular Momentum Vector A classical bar magnet (Figure 11) may lie motionless at a certain orientation in a magnetic field. However, if the bar magnet possesses angular momentum,
6. Classical Precession of the Angular Momentum Vector A classical bar magnet (Figure 11) may lie motionless at a certain orientation in a magnetic field. However, if the bar magnet possesses angular momentum,
How To Understand The Measurement Process
 April 24, 2015 Exam #3: Solution Key online now! Graded exams by Monday! Final Exam Monday, May 4 th, 10:30 a.m. Room: Perkins 107 1 A Classical Perspective A classical view will help us understand the
April 24, 2015 Exam #3: Solution Key online now! Graded exams by Monday! Final Exam Monday, May 4 th, 10:30 a.m. Room: Perkins 107 1 A Classical Perspective A classical view will help us understand the
Proton Nuclear Magnetic Resonance Spectroscopy
 Proton Nuclear Magnetic Resonance Spectroscopy Introduction: The NMR Spectrum serves as a great resource in determining the structure of an organic compound by revealing the hydrogen and carbon skeleton.
Proton Nuclear Magnetic Resonance Spectroscopy Introduction: The NMR Spectrum serves as a great resource in determining the structure of an organic compound by revealing the hydrogen and carbon skeleton.
Nuclear Magnetic Resonance (NMR) Spectroscopy
 April 28, 2016 Exam #3: Graded exams on Tuesday! Final Exam Tuesday, May 10 th, 10:30 a.m. Room: Votey 207 (tentative) Review Session: Sunday, May 8 th, 4 pm, Kalkin 325 (tentative) Office Hours Next week:
April 28, 2016 Exam #3: Graded exams on Tuesday! Final Exam Tuesday, May 10 th, 10:30 a.m. Room: Votey 207 (tentative) Review Session: Sunday, May 8 th, 4 pm, Kalkin 325 (tentative) Office Hours Next week:
2. Spin Chemistry and the Vector Model
 2. Spin Chemistry and the Vector Model The story of magnetic resonance spectroscopy and intersystem crossing is essentially a choreography of the twisting motion which causes reorientation or rephasing
2. Spin Chemistry and the Vector Model The story of magnetic resonance spectroscopy and intersystem crossing is essentially a choreography of the twisting motion which causes reorientation or rephasing
13C NMR Spectroscopy
 13 C NMR Spectroscopy Introduction Nuclear magnetic resonance spectroscopy (NMR) is the most powerful tool available for structural determination. A nucleus with an odd number of protons, an odd number
13 C NMR Spectroscopy Introduction Nuclear magnetic resonance spectroscopy (NMR) is the most powerful tool available for structural determination. A nucleus with an odd number of protons, an odd number
Chapter 13 Spectroscopy NMR, IR, MS, UV-Vis
 Chapter 13 Spectroscopy NMR, IR, MS, UV-Vis Main points of the chapter 1. Hydrogen Nuclear Magnetic Resonance a. Splitting or coupling (what s next to what) b. Chemical shifts (what type is it) c. Integration
Chapter 13 Spectroscopy NMR, IR, MS, UV-Vis Main points of the chapter 1. Hydrogen Nuclear Magnetic Resonance a. Splitting or coupling (what s next to what) b. Chemical shifts (what type is it) c. Integration
Used to determine relative location of atoms within a molecule Most helpful spectroscopic technique in organic chemistry Related to MRI in medicine
 Structure Determination: Nuclear Magnetic Resonance CHEM 241 UNIT 5C 1 The Use of NMR Spectroscopy Used to determine relative location of atoms within a molecule Most helpful spectroscopic technique in
Structure Determination: Nuclear Magnetic Resonance CHEM 241 UNIT 5C 1 The Use of NMR Spectroscopy Used to determine relative location of atoms within a molecule Most helpful spectroscopic technique in
The Hydrogen Atom Is a Magnet. http://www.seed.slb.com/en/scictr/watch/gashydrates/detecting.htm
 The Hydrogen Atom Is a Magnet Nuclear Magnetic Resonance Spectroscopy (NMR) Proton NMR A hydrogen nucleus can be viewed as a proton, which can be viewed as a spinning charge. As with any spinning charge,
The Hydrogen Atom Is a Magnet Nuclear Magnetic Resonance Spectroscopy (NMR) Proton NMR A hydrogen nucleus can be viewed as a proton, which can be viewed as a spinning charge. As with any spinning charge,
NMR SPECTROSCOPY A N I N T R O D U C T I O N T O... Self-study booklet NUCLEAR MAGNETIC RESONANCE. 4 3 2 1 0 δ PUBLISHING
 A N I N T R O D U T I O N T O... NMR SPETROSOPY NULEAR MAGNETI RESONANE 4 3 1 0 δ Self-study booklet PUBLISING NMR Spectroscopy NULEAR MAGNETI RESONANE SPETROSOPY Origin of Spectra Theory All nuclei possess
A N I N T R O D U T I O N T O... NMR SPETROSOPY NULEAR MAGNETI RESONANE 4 3 1 0 δ Self-study booklet PUBLISING NMR Spectroscopy NULEAR MAGNETI RESONANE SPETROSOPY Origin of Spectra Theory All nuclei possess
Nuclear Magnetic Resonance
 Nuclear Magnetic Resonance NMR is probably the most useful and powerful technique for identifying and characterizing organic compounds. Felix Bloch and Edward Mills Purcell were awarded the 1952 Nobel
Nuclear Magnetic Resonance NMR is probably the most useful and powerful technique for identifying and characterizing organic compounds. Felix Bloch and Edward Mills Purcell were awarded the 1952 Nobel
Nuclear Magnetic Resonance and the Measurement of Relaxation Times of Acetone with Gadolinium
 Nuclear Magnetic Resonance and the Measurement of Relaxation Times of Acetone with Gadolinium Xia Lee and Albert Tsai June 15, 2006 1 1 Introduction Nuclear magnetic resonance (NMR) is a spectroscopic
Nuclear Magnetic Resonance and the Measurement of Relaxation Times of Acetone with Gadolinium Xia Lee and Albert Tsai June 15, 2006 1 1 Introduction Nuclear magnetic resonance (NMR) is a spectroscopic
Proton Nuclear Magnetic Resonance ( 1 H-NMR) Spectroscopy
 Proton Nuclear Magnetic Resonance ( 1 H-NMR) Spectroscopy Theory behind NMR: In the late 1940 s, physical chemists originally developed NMR spectroscopy to study different properties of atomic nuclei,
Proton Nuclear Magnetic Resonance ( 1 H-NMR) Spectroscopy Theory behind NMR: In the late 1940 s, physical chemists originally developed NMR spectroscopy to study different properties of atomic nuclei,
4. It is possible to excite, or flip the nuclear magnetic vector from the α-state to the β-state by bridging the energy gap between the two. This is a
 BASIC PRINCIPLES INTRODUCTION TO NUCLEAR MAGNETIC RESONANCE (NMR) 1. The nuclei of certain atoms with odd atomic number, and/or odd mass behave as spinning charges. The nucleus is the center of positive
BASIC PRINCIPLES INTRODUCTION TO NUCLEAR MAGNETIC RESONANCE (NMR) 1. The nuclei of certain atoms with odd atomic number, and/or odd mass behave as spinning charges. The nucleus is the center of positive
Indiana's Academic Standards 2010 ICP Indiana's Academic Standards 2016 ICP. map) that describe the relationship acceleration, velocity and distance.
 .1.1 Measure the motion of objects to understand.1.1 Develop graphical, the relationships among distance, velocity and mathematical, and pictorial acceleration. Develop deeper understanding through representations
.1.1 Measure the motion of objects to understand.1.1 Develop graphical, the relationships among distance, velocity and mathematical, and pictorial acceleration. Develop deeper understanding through representations
Chapter 18: The Structure of the Atom
 Chapter 18: The Structure of the Atom 1. For most elements, an atom has A. no neutrons in the nucleus. B. more protons than electrons. C. less neutrons than electrons. D. just as many electrons as protons.
Chapter 18: The Structure of the Atom 1. For most elements, an atom has A. no neutrons in the nucleus. B. more protons than electrons. C. less neutrons than electrons. D. just as many electrons as protons.
Nuclear Magnetic Resonance notes
 Reminder: These notes are meant to supplement, not replace, the laboratory manual. Nuclear Magnetic Resonance notes Nuclear Magnetic Resonance (NMR) is a spectrometric technique which provides information
Reminder: These notes are meant to supplement, not replace, the laboratory manual. Nuclear Magnetic Resonance notes Nuclear Magnetic Resonance (NMR) is a spectrometric technique which provides information
Proton Nuclear Magnetic Resonance Spectroscopy
 CHEM 334L Organic Chemistry Laboratory Revision 2.0 Proton Nuclear Magnetic Resonance Spectroscopy In this laboratory exercise we will learn how to use the Chemistry Department's Nuclear Magnetic Resonance
CHEM 334L Organic Chemistry Laboratory Revision 2.0 Proton Nuclear Magnetic Resonance Spectroscopy In this laboratory exercise we will learn how to use the Chemistry Department's Nuclear Magnetic Resonance
Nuclear Magnetic Resonance Spectroscopy
 Nuclear Magnetic Resonance Spectroscopy Introduction NMR is the most powerful tool available for organic structure determination. It is used to study a wide variety of nuclei: 1 H 13 C 15 N 19 F 31 P 2
Nuclear Magnetic Resonance Spectroscopy Introduction NMR is the most powerful tool available for organic structure determination. It is used to study a wide variety of nuclei: 1 H 13 C 15 N 19 F 31 P 2
Organic Chemistry Tenth Edition
 Organic Chemistry Tenth Edition T. W. Graham Solomons Craig B. Fryhle Welcome to CHM 22 Organic Chemisty II Chapters 2 (IR), 9, 3-20. Chapter 2 and Chapter 9 Spectroscopy (interaction of molecule with
Organic Chemistry Tenth Edition T. W. Graham Solomons Craig B. Fryhle Welcome to CHM 22 Organic Chemisty II Chapters 2 (IR), 9, 3-20. Chapter 2 and Chapter 9 Spectroscopy (interaction of molecule with
Classical Angular Momentum. The Physics of Rotational Motion.
 3. Angular Momentum States. We now employ the vector model to enumerate the possible number of spin angular momentum states for several commonly encountered situations in photochemistry. We shall give
3. Angular Momentum States. We now employ the vector model to enumerate the possible number of spin angular momentum states for several commonly encountered situations in photochemistry. We shall give
NMR for Physical and Biological Scientists Thomas C. Pochapsky and Susan Sondej Pochapsky Table of Contents
 Preface Symbols and fundamental constants 1. What is spectroscopy? A semiclassical description of spectroscopy Damped harmonics Quantum oscillators The spectroscopic experiment Ensembles and coherence
Preface Symbols and fundamental constants 1. What is spectroscopy? A semiclassical description of spectroscopy Damped harmonics Quantum oscillators The spectroscopic experiment Ensembles and coherence
2, 8, 20, 28, 50, 82, 126.
 Chapter 5 Nuclear Shell Model 5.1 Magic Numbers The binding energies predicted by the Liquid Drop Model underestimate the actual binding energies of magic nuclei for which either the number of neutrons
Chapter 5 Nuclear Shell Model 5.1 Magic Numbers The binding energies predicted by the Liquid Drop Model underestimate the actual binding energies of magic nuclei for which either the number of neutrons
Nuclear Magnetic Resonance Spectroscopy
 Nuclear Magnetic Resonance Spectroscopy Nuclear magnetic resonance spectroscopy is a powerful analytical technique used to characterize organic molecules by identifying carbonhydrogen frameworks within
Nuclear Magnetic Resonance Spectroscopy Nuclear magnetic resonance spectroscopy is a powerful analytical technique used to characterize organic molecules by identifying carbonhydrogen frameworks within
Objectives. PAM1014 Introduction to Radiation Physics. Constituents of Atoms. Atoms. Atoms. Atoms. Basic Atomic Theory
 PAM1014 Introduction to Radiation Physics Basic Atomic Theory Objectives Introduce and Molecules The periodic Table Electronic Energy Levels Atomic excitation & de-excitation Ionisation Molecules Constituents
PAM1014 Introduction to Radiation Physics Basic Atomic Theory Objectives Introduce and Molecules The periodic Table Electronic Energy Levels Atomic excitation & de-excitation Ionisation Molecules Constituents
PROTON NUCLEAR MAGNETIC RESONANCE SPECTROSCOPY (H-NMR)
 PROTON NUCLEAR MAGNETIC RESONANCE SPECTROSCOPY (H-NMR) WHAT IS H-NMR SPECTROSCOPY? References: Bruice 14.1, 14.2 Introduction NMR or nuclear magnetic resonance spectroscopy is a technique used to determine
PROTON NUCLEAR MAGNETIC RESONANCE SPECTROSCOPY (H-NMR) WHAT IS H-NMR SPECTROSCOPY? References: Bruice 14.1, 14.2 Introduction NMR or nuclear magnetic resonance spectroscopy is a technique used to determine
DO PHYSICS ONLINE FROM QUANTA TO QUARKS QUANTUM (WAVE) MECHANICS
 DO PHYSICS ONLINE FROM QUANTA TO QUARKS QUANTUM (WAVE) MECHANICS Quantum Mechanics or wave mechanics is the best mathematical theory used today to describe and predict the behaviour of particles and waves.
DO PHYSICS ONLINE FROM QUANTA TO QUARKS QUANTUM (WAVE) MECHANICS Quantum Mechanics or wave mechanics is the best mathematical theory used today to describe and predict the behaviour of particles and waves.
Nuclear Magnetic Resonance (NMR) Spectroscopy cont... Recommended Reading:
 Applied Spectroscopy Nuclear Magnetic Resonance (NMR) Spectroscopy cont... Recommended Reading: Banwell and McCash Chapter 7 Skoog, Holler Nieman Chapter 19 Atkins, Chapter 18 Relaxation processes We need
Applied Spectroscopy Nuclear Magnetic Resonance (NMR) Spectroscopy cont... Recommended Reading: Banwell and McCash Chapter 7 Skoog, Holler Nieman Chapter 19 Atkins, Chapter 18 Relaxation processes We need
Introduction to Nuclear Magnetic Resonance Spectroscopy
 Introduction to Nuclear Magnetic Resonance Spectroscopy Dr. Dean L. Olson, NMR Lab Director School of Chemical Sciences University of Illinois Called figures, equations, and tables are from Principles
Introduction to Nuclear Magnetic Resonance Spectroscopy Dr. Dean L. Olson, NMR Lab Director School of Chemical Sciences University of Illinois Called figures, equations, and tables are from Principles
Application of Nuclear Magnetic Resonance in Petroleum Exploration
 Application of Nuclear Magnetic Resonance in Petroleum Exploration Introduction Darko Tufekcic, consultant email: darkotufekcic@hotmail.com Electro-magnetic resonance method (GEO-EMR) is emerging as the
Application of Nuclear Magnetic Resonance in Petroleum Exploration Introduction Darko Tufekcic, consultant email: darkotufekcic@hotmail.com Electro-magnetic resonance method (GEO-EMR) is emerging as the
Nuclear Magnetic Resonance Spectroscopy
 Most spinning nuclei behave like magnets. Nuclear Magnetic Resonance Spectroscopy asics owever, as opposed to the behavior of a classical magnet the nuclear spin magnetic moment does not always align with
Most spinning nuclei behave like magnets. Nuclear Magnetic Resonance Spectroscopy asics owever, as opposed to the behavior of a classical magnet the nuclear spin magnetic moment does not always align with
Theory of spin magnetic resonance: derivations of energy spacing and chemical shifts in NMR spectroscopy.
 William McFadden Physics 352 Quantum Mechanics Final Project Theory of spin magnetic resonance: derivations of energy spacing and chemical shifts in NMR spectroscopy. Introduction The simplicity of a spin
William McFadden Physics 352 Quantum Mechanics Final Project Theory of spin magnetic resonance: derivations of energy spacing and chemical shifts in NMR spectroscopy. Introduction The simplicity of a spin
How To Understand Electron Spin Resonance
 HB 10-24-08 Electron Spin Resonance Lab 1 Electron Spin Resonance Equipment Electron Spin Resonance apparatus, leads, BK oscilloscope, 15 cm ruler for setting coil separation Reading Review the Oscilloscope
HB 10-24-08 Electron Spin Resonance Lab 1 Electron Spin Resonance Equipment Electron Spin Resonance apparatus, leads, BK oscilloscope, 15 cm ruler for setting coil separation Reading Review the Oscilloscope
Multi-electron atoms
 Multi-electron atoms Today: Using hydrogen as a model. The Periodic Table HWK 13 available online. Please fill out the online participation survey. Worth 10points on HWK 13. Final Exam is Monday, Dec.
Multi-electron atoms Today: Using hydrogen as a model. The Periodic Table HWK 13 available online. Please fill out the online participation survey. Worth 10points on HWK 13. Final Exam is Monday, Dec.
Physics of Imaging Systems Basic Principles of Magnetic Resonance Imaging II
 1 10/30/2015 Page 1 Master s Program in Medical Physics Physics of Imaging Systems Basic Principles of Magnetic Resonance Imaging II Chair in Faculty of Medicine Mannheim University of Heidelberg Theodor-Kutzer-Ufer
1 10/30/2015 Page 1 Master s Program in Medical Physics Physics of Imaging Systems Basic Principles of Magnetic Resonance Imaging II Chair in Faculty of Medicine Mannheim University of Heidelberg Theodor-Kutzer-Ufer
Molecular-Orbital Theory
 Molecular-Orbital Theory 1 Introduction Orbitals in molecules are not necessarily localized on atoms or between atoms as suggested in the valence bond theory. Molecular orbitals can also be formed the
Molecular-Orbital Theory 1 Introduction Orbitals in molecules are not necessarily localized on atoms or between atoms as suggested in the valence bond theory. Molecular orbitals can also be formed the
NMR Nuclear Magnetic Resonance
 NMR Nuclear Magnetic Resonance Nuclear magnetic resonance (NMR) is an effect whereby magnetic nuclei in a magnetic field absorb and re-emit electromagnetic (EM) energy. This energy is at a specific resonance
NMR Nuclear Magnetic Resonance Nuclear magnetic resonance (NMR) is an effect whereby magnetic nuclei in a magnetic field absorb and re-emit electromagnetic (EM) energy. This energy is at a specific resonance
Electron Orbits. Binding Energy. centrifugal force: electrostatic force: stability criterion: kinetic energy of the electron on its orbit:
 Electron Orbits In an atom model in which negatively charged electrons move around a small positively charged nucleus stable orbits are possible. Consider the simple example of an atom with a nucleus of
Electron Orbits In an atom model in which negatively charged electrons move around a small positively charged nucleus stable orbits are possible. Consider the simple example of an atom with a nucleus of
Nuclear Magnetic Resonance
 Nuclear Magnetic Resonance Practical Course M I. Physikalisches Institut Universität zu Köln May 15, 2014 Abstract Nuclear magnetic resonance (NMR) techniques are widely used in physics, chemistry, and
Nuclear Magnetic Resonance Practical Course M I. Physikalisches Institut Universität zu Köln May 15, 2014 Abstract Nuclear magnetic resonance (NMR) techniques are widely used in physics, chemistry, and
Background A nucleus with an odd atomic number or an odd mass number has a nuclear spin that can be observed by NMR spectrometers.
 NMR Spectroscopy I Reading: Wade chapter, sections -- -7 Study Problems: -, -7 Key oncepts and Skills: Given an structure, determine which protons are equivalent and which are nonequivalent, predict the
NMR Spectroscopy I Reading: Wade chapter, sections -- -7 Study Problems: -, -7 Key oncepts and Skills: Given an structure, determine which protons are equivalent and which are nonequivalent, predict the
Generation and Detection of NMR Signals
 Generation and Detection of NMR Signals Hanudatta S. Atreya NMR Research Centre Indian Institute of Science NMR Spectroscopy Spin (I)=1/2h B 0 Energy 0 = B 0 Classical picture (B 0 ) Quantum Mechanical
Generation and Detection of NMR Signals Hanudatta S. Atreya NMR Research Centre Indian Institute of Science NMR Spectroscopy Spin (I)=1/2h B 0 Energy 0 = B 0 Classical picture (B 0 ) Quantum Mechanical
Pulsed Fourier Transform NMR The rotating frame of reference. The NMR Experiment. The Rotating Frame of Reference.
 Pulsed Fourier Transform NR The rotating frame of reference The NR Eperiment. The Rotating Frame of Reference. When we perform a NR eperiment we disturb the equilibrium state of the sstem and then monitor
Pulsed Fourier Transform NR The rotating frame of reference The NR Eperiment. The Rotating Frame of Reference. When we perform a NR eperiment we disturb the equilibrium state of the sstem and then monitor
Nuclear Physics. Nuclear Physics comprises the study of:
 Nuclear Physics Nuclear Physics comprises the study of: The general properties of nuclei The particles contained in the nucleus The interaction between these particles Radioactivity and nuclear reactions
Nuclear Physics Nuclear Physics comprises the study of: The general properties of nuclei The particles contained in the nucleus The interaction between these particles Radioactivity and nuclear reactions
E. K. A. ADVANCED PHYSICS LABORATORY PHYSICS 3081, 4051 NUCLEAR MAGNETIC RESONANCE
 E. K. A. ADVANCED PHYSICS LABORATORY PHYSICS 3081, 4051 NUCLEAR MAGNETIC RESONANCE References for Nuclear Magnetic Resonance 1. Slichter, Principles of Magnetic Resonance, Harper and Row, 1963. chapter
E. K. A. ADVANCED PHYSICS LABORATORY PHYSICS 3081, 4051 NUCLEAR MAGNETIC RESONANCE References for Nuclear Magnetic Resonance 1. Slichter, Principles of Magnetic Resonance, Harper and Row, 1963. chapter
Electron Paramagnetic (Spin) Resonance
 Electron Paramagnetic (Spin) Resonance References: Jardetzky & Jardetzky, Meth. Biochem. Anal. 9, 235. Wertz & Bolton, Electron Spin Resonance Poole, Electron Spin Resonance... Abragam & Bleaney, EPR of
Electron Paramagnetic (Spin) Resonance References: Jardetzky & Jardetzky, Meth. Biochem. Anal. 9, 235. Wertz & Bolton, Electron Spin Resonance Poole, Electron Spin Resonance... Abragam & Bleaney, EPR of
Determining the Structure of an Organic Compound
 Determining the Structure of an Organic Compound The analysis of the outcome of a reaction requires that we know the full structure of the products as well as the reactants In the 19 th and early 20 th
Determining the Structure of an Organic Compound The analysis of the outcome of a reaction requires that we know the full structure of the products as well as the reactants In the 19 th and early 20 th
Ph 3504 Nuclear Magnetic Resonance and Electron Spin Resonance
 Ph 3504 Nuclear Magnetic Resonance and Electron Spin Resonance Required background reading Tipler, Llewellyn, section 12-3 (you only need to read the part labeled Nuclear Magnetic Resonance on pages 596-597
Ph 3504 Nuclear Magnetic Resonance and Electron Spin Resonance Required background reading Tipler, Llewellyn, section 12-3 (you only need to read the part labeled Nuclear Magnetic Resonance on pages 596-597
NMR Spectroscopy of Aromatic Compounds (#1e)
 NMR Spectroscopy of Aromatic Compounds (#1e) 1 H NMR Spectroscopy of Aromatic Compounds Erich Hückel s study of aromaticity in the 1930s produced a set of rules for determining whether a compound is aromatic.
NMR Spectroscopy of Aromatic Compounds (#1e) 1 H NMR Spectroscopy of Aromatic Compounds Erich Hückel s study of aromaticity in the 1930s produced a set of rules for determining whether a compound is aromatic.
An Adaptive Signal Processing Approach to Dynamic Magnetic Resonance Imaging
 An Adaptive Signal Processing Approach to Dynamic Magnetic Resonance Imaging A Thesis Presented by William Scott Hoge to The Department of Electrical and Computer Engineering in partial fulfillment of
An Adaptive Signal Processing Approach to Dynamic Magnetic Resonance Imaging A Thesis Presented by William Scott Hoge to The Department of Electrical and Computer Engineering in partial fulfillment of
COURSE#1022: Biochemical Applications of NMR Spectroscopy. http://www.bioc.aecom.yu.edu/labs/girvlab/nmr/course/ Basic Principles
 COURSE#1022: Biochemical Applications of NMR Spectroscopy http://www.bioc.aecom.yu.edu/labs/girvlab/nmr/course/ Basic Principles LAST UPDATE: 1/11/2012 Reading Selected Readings for Basic Principles of
COURSE#1022: Biochemical Applications of NMR Spectroscopy http://www.bioc.aecom.yu.edu/labs/girvlab/nmr/course/ Basic Principles LAST UPDATE: 1/11/2012 Reading Selected Readings for Basic Principles of
Nuclear Magnetic Resonance Spectroscopy
 Chapter 8 Nuclear Magnetic Resonance Spectroscopy http://www.yteach.co.uk/page.php/resources/view_all?id=nuclear_magnetic _resonance_nmr_spectroscopy_spin_spectrometer_spectrum_proton_t_pag e_5&from=search
Chapter 8 Nuclear Magnetic Resonance Spectroscopy http://www.yteach.co.uk/page.php/resources/view_all?id=nuclear_magnetic _resonance_nmr_spectroscopy_spin_spectrometer_spectrum_proton_t_pag e_5&from=search
ELECTRON SPIN RESONANCE Last Revised: July 2007
 QUESTION TO BE INVESTIGATED ELECTRON SPIN RESONANCE Last Revised: July 2007 How can we measure the Landé g factor for the free electron in DPPH as predicted by quantum mechanics? INTRODUCTION Electron
QUESTION TO BE INVESTIGATED ELECTRON SPIN RESONANCE Last Revised: July 2007 How can we measure the Landé g factor for the free electron in DPPH as predicted by quantum mechanics? INTRODUCTION Electron
Chapter 11 Structure Determination: Nuclear Magnetic Resonance Spectroscopy. Nuclear Magnetic Resonance Spectroscopy. 11.1 Nuclear Magnetic Resonance
 John E. McMurry http://www.cengage.com/chemistry/mcmurry Chapter 11 Structure Determination: Nuclear Magnetic Resonance Spectroscopy 11.1 Nuclear Magnetic Resonance Spectroscopy Many atomic nuclei behave
John E. McMurry http://www.cengage.com/chemistry/mcmurry Chapter 11 Structure Determination: Nuclear Magnetic Resonance Spectroscopy 11.1 Nuclear Magnetic Resonance Spectroscopy Many atomic nuclei behave
WAVES AND ELECTROMAGNETIC RADIATION
 WAVES AND ELECTROMAGNETIC RADIATION All waves are characterized by their wavelength, frequency and speed. Wavelength (lambda, ): the distance between any 2 successive crests or troughs. Frequency (nu,):
WAVES AND ELECTROMAGNETIC RADIATION All waves are characterized by their wavelength, frequency and speed. Wavelength (lambda, ): the distance between any 2 successive crests or troughs. Frequency (nu,):
Light as a Wave. The Nature of Light. EM Radiation Spectrum. EM Radiation Spectrum. Electromagnetic Radiation
 The Nature of Light Light and other forms of radiation carry information to us from distance astronomical objects Visible light is a subset of a huge spectrum of electromagnetic radiation Maxwell pioneered
The Nature of Light Light and other forms of radiation carry information to us from distance astronomical objects Visible light is a subset of a huge spectrum of electromagnetic radiation Maxwell pioneered
Assessment Plan for Learning Outcomes for BA/BS in Physics
 Department of Physics and Astronomy Goals and Learning Outcomes 1. Students know basic physics principles [BS, BA, MS] 1.1 Students can demonstrate an understanding of Newton s laws 1.2 Students can demonstrate
Department of Physics and Astronomy Goals and Learning Outcomes 1. Students know basic physics principles [BS, BA, MS] 1.1 Students can demonstrate an understanding of Newton s laws 1.2 Students can demonstrate
Solving Spectroscopy Problems
 Solving Spectroscopy Problems The following is a detailed summary on how to solve spectroscopy problems, key terms are highlighted in bold and the definitions are from the illustrated glossary on Dr. Hardinger
Solving Spectroscopy Problems The following is a detailed summary on how to solve spectroscopy problems, key terms are highlighted in bold and the definitions are from the illustrated glossary on Dr. Hardinger
NUCLEAR MAGNETIC RESONANCE. Advanced Laboratory, Physics 407, University of Wisconsin Madison, Wisconsin 53706
 (revised 4/21/03) NUCLEAR MAGNETIC RESONANCE Advanced Laboratory, Physics 407, University of Wisconsin Madison, Wisconsin 53706 Abstract This experiment studies the Nuclear Magnetic Resonance of protons
(revised 4/21/03) NUCLEAR MAGNETIC RESONANCE Advanced Laboratory, Physics 407, University of Wisconsin Madison, Wisconsin 53706 Abstract This experiment studies the Nuclear Magnetic Resonance of protons
DIAGNOSTIC MEDICAL IMAGING 1st Part --Introduction. Ing. Tommaso Rossi tommaso.rossi@uniroma2.it
 DIAGNOSTIC MEDICAL IMAGING 1st Part --Introduction Ing. Tommaso Rossi tommaso.rossi@uniroma2.it Tommaso Rossi - Modulo di SEGNALI, a.a. 2013/2014 Overview 2 How we can look on the inside of human body?
DIAGNOSTIC MEDICAL IMAGING 1st Part --Introduction Ing. Tommaso Rossi tommaso.rossi@uniroma2.it Tommaso Rossi - Modulo di SEGNALI, a.a. 2013/2014 Overview 2 How we can look on the inside of human body?
Nuclear Magnetic Resonance
 Nuclear Magnetic Resonance Author: James Dragan Lab Partner: Stefan Evans Physics Department, Stony Brook University, Stony Brook, NY 794. (Dated: December 5, 23) We study the principles behind Nuclear
Nuclear Magnetic Resonance Author: James Dragan Lab Partner: Stefan Evans Physics Department, Stony Brook University, Stony Brook, NY 794. (Dated: December 5, 23) We study the principles behind Nuclear
Determination of Equilibrium Constants using NMR Spectrscopy
 CHEM 331L Physical Chemistry Laboratory Revision 1.0 Determination of Equilibrium Constants using NMR Spectrscopy In this laboratory exercise we will measure a chemical equilibrium constant using key proton
CHEM 331L Physical Chemistry Laboratory Revision 1.0 Determination of Equilibrium Constants using NMR Spectrscopy In this laboratory exercise we will measure a chemical equilibrium constant using key proton
Application Note AN4
 TAKING INVENTIVE STEPS IN INFRARED. MINIATURE INFRARED GAS SENSORS GOLD SERIES UK Patent App. No. 2372099A USA Patent App. No. 09/783,711 World Patents Pending INFRARED SPECTROSCOPY Application Note AN4
TAKING INVENTIVE STEPS IN INFRARED. MINIATURE INFRARED GAS SENSORS GOLD SERIES UK Patent App. No. 2372099A USA Patent App. No. 09/783,711 World Patents Pending INFRARED SPECTROSCOPY Application Note AN4
Wave Function, ψ. Chapter 28 Atomic Physics. The Heisenberg Uncertainty Principle. Line Spectrum
 Wave Function, ψ Chapter 28 Atomic Physics The Hydrogen Atom The Bohr Model Electron Waves in the Atom The value of Ψ 2 for a particular object at a certain place and time is proportional to the probability
Wave Function, ψ Chapter 28 Atomic Physics The Hydrogen Atom The Bohr Model Electron Waves in the Atom The value of Ψ 2 for a particular object at a certain place and time is proportional to the probability
Nuclear Magnetic Resonance (NMR) Wade Textbook
 Nuclear Magnetic Resonance (NMR) Wade Textbook Background Is a nondestructive structural analysis technique Has the same theoretical basis as magnetic resonance imaging (MRI) Referring to MRI as nuclear
Nuclear Magnetic Resonance (NMR) Wade Textbook Background Is a nondestructive structural analysis technique Has the same theoretical basis as magnetic resonance imaging (MRI) Referring to MRI as nuclear
Physics 9e/Cutnell. correlated to the. College Board AP Physics 1 Course Objectives
 Physics 9e/Cutnell correlated to the College Board AP Physics 1 Course Objectives Big Idea 1: Objects and systems have properties such as mass and charge. Systems may have internal structure. Enduring
Physics 9e/Cutnell correlated to the College Board AP Physics 1 Course Objectives Big Idea 1: Objects and systems have properties such as mass and charge. Systems may have internal structure. Enduring
GE Medical Systems Training in Partnership. Module 8: IQ: Acquisition Time
 Module 8: IQ: Acquisition Time IQ : Acquisition Time Objectives...Describe types of data acquisition modes....compute acquisition times for 2D and 3D scans. 2D Acquisitions The 2D mode acquires and reconstructs
Module 8: IQ: Acquisition Time IQ : Acquisition Time Objectives...Describe types of data acquisition modes....compute acquisition times for 2D and 3D scans. 2D Acquisitions The 2D mode acquires and reconstructs
MASTER OF SCIENCE IN PHYSICS MASTER OF SCIENCES IN PHYSICS (MS PHYS) (LIST OF COURSES BY SEMESTER, THESIS OPTION)
 MASTER OF SCIENCE IN PHYSICS Admission Requirements 1. Possession of a BS degree from a reputable institution or, for non-physics majors, a GPA of 2.5 or better in at least 15 units in the following advanced
MASTER OF SCIENCE IN PHYSICS Admission Requirements 1. Possession of a BS degree from a reputable institution or, for non-physics majors, a GPA of 2.5 or better in at least 15 units in the following advanced
Physical Principle of Formation and Essence of Radio Waves
 Physical Principle of Formation and Essence of Radio Waves Anatoli Bedritsky Abstract. This article opens physical phenomena which occur at the formation of the radio waves, and opens the essence of the
Physical Principle of Formation and Essence of Radio Waves Anatoli Bedritsky Abstract. This article opens physical phenomena which occur at the formation of the radio waves, and opens the essence of the
Determination of Molecular Structure by MOLECULAR SPECTROSCOPY
 Determination of Molecular Structure by MOLEULAR SPETROSOPY hemistry 3 B.Z. Shakhashiri Fall 29 Much of what we know about molecular structure has been learned by observing and analyzing how electromagnetic
Determination of Molecular Structure by MOLEULAR SPETROSOPY hemistry 3 B.Z. Shakhashiri Fall 29 Much of what we know about molecular structure has been learned by observing and analyzing how electromagnetic
Chemistry 102 Summary June 24 th. Properties of Light
 Chemistry 102 Summary June 24 th Properties of Light - Energy travels through space in the form of electromagnetic radiation (EMR). - Examples of types of EMR: radio waves, x-rays, microwaves, visible
Chemistry 102 Summary June 24 th Properties of Light - Energy travels through space in the form of electromagnetic radiation (EMR). - Examples of types of EMR: radio waves, x-rays, microwaves, visible
AP* Atomic Structure & Periodicity Free Response Questions KEY page 1
 AP* Atomic Structure & Periodicity ree Response Questions KEY page 1 1980 a) points 1s s p 6 3s 3p 6 4s 3d 10 4p 3 b) points for the two electrons in the 4s: 4, 0, 0, +1/ and 4, 0, 0, - 1/ for the three
AP* Atomic Structure & Periodicity ree Response Questions KEY page 1 1980 a) points 1s s p 6 3s 3p 6 4s 3d 10 4p 3 b) points for the two electrons in the 4s: 4, 0, 0, +1/ and 4, 0, 0, - 1/ for the three
Contents. Goldstone Bosons in 3He-A Soft Modes Dynamics and Lie Algebra of Group G:
 ... Vlll Contents 3. Textures and Supercurrents in Superfluid Phases of 3He 3.1. Textures, Gradient Energy and Rigidity 3.2. Why Superfuids are Superfluid 3.3. Superfluidity and Response to a Transverse
... Vlll Contents 3. Textures and Supercurrents in Superfluid Phases of 3He 3.1. Textures, Gradient Energy and Rigidity 3.2. Why Superfuids are Superfluid 3.3. Superfluidity and Response to a Transverse
5. The Nature of Light. Does Light Travel Infinitely Fast? EMR Travels At Finite Speed. EMR: Electric & Magnetic Waves
 5. The Nature of Light Light travels in vacuum at 3.0. 10 8 m/s Light is one form of electromagnetic radiation Continuous radiation: Based on temperature Wien s Law & the Stefan-Boltzmann Law Light has
5. The Nature of Light Light travels in vacuum at 3.0. 10 8 m/s Light is one form of electromagnetic radiation Continuous radiation: Based on temperature Wien s Law & the Stefan-Boltzmann Law Light has
The Phenomenon of Photoelectric Emission:
 The Photoelectric Effect. The Wave particle duality of light Light, like any other E.M.R (electromagnetic radiation) has got a dual nature. That is there are experiments that prove that it is made up of
The Photoelectric Effect. The Wave particle duality of light Light, like any other E.M.R (electromagnetic radiation) has got a dual nature. That is there are experiments that prove that it is made up of
Atomic Structure: Chapter Problems
 Atomic Structure: Chapter Problems Bohr Model Class Work 1. Describe the nuclear model of the atom. 2. Explain the problems with the nuclear model of the atom. 3. According to Niels Bohr, what does n stand
Atomic Structure: Chapter Problems Bohr Model Class Work 1. Describe the nuclear model of the atom. 2. Explain the problems with the nuclear model of the atom. 3. According to Niels Bohr, what does n stand
TIME OF COMPLETION NAME SOLUTION DEPARTMENT OF NATURAL SCIENCES. PHYS 3650, Exam 2 Section 1 Version 1 October 31, 2005 Total Weight: 100 points
 TIME OF COMPLETION NAME SOLUTION DEPARTMENT OF NATURAL SCIENCES PHYS 3650, Exam 2 Section 1 Version 1 October 31, 2005 Total Weight: 100 points 1. Check your examination for completeness prior to starting.
TIME OF COMPLETION NAME SOLUTION DEPARTMENT OF NATURAL SCIENCES PHYS 3650, Exam 2 Section 1 Version 1 October 31, 2005 Total Weight: 100 points 1. Check your examination for completeness prior to starting.
The Four Questions to Ask While Interpreting Spectra. 1. How many different environments are there?
 1 H NMR Spectroscopy (#1c) The technique of 1 H NMR spectroscopy is central to organic chemistry and other fields involving analysis of organic chemicals, such as forensics and environmental science. It
1 H NMR Spectroscopy (#1c) The technique of 1 H NMR spectroscopy is central to organic chemistry and other fields involving analysis of organic chemicals, such as forensics and environmental science. It
Lecture #7 (2D NMR) Utility of Resonance Assignments
 Lecture #7 (2D NMR) Basics of multidimensional NMR (2D NMR) 2D NOESY, COSY and TOCSY 2/23/15 Utility of Resonance Assignments Resonance Assignments: Assignment of frequency positions of resonances (peaks)
Lecture #7 (2D NMR) Basics of multidimensional NMR (2D NMR) 2D NOESY, COSY and TOCSY 2/23/15 Utility of Resonance Assignments Resonance Assignments: Assignment of frequency positions of resonances (peaks)
"in recognition of the services he rendered to the advancement of Physics by his discovery of energy quanta". h is the Planck constant he called it
 1 2 "in recognition of the services he rendered to the advancement of Physics by his discovery of energy quanta". h is the Planck constant he called it the quantum of action 3 Newton believed in the corpuscular
1 2 "in recognition of the services he rendered to the advancement of Physics by his discovery of energy quanta". h is the Planck constant he called it the quantum of action 3 Newton believed in the corpuscular
Magnetic Resonance Imaging
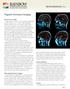 Magnetic Resonance Imaging What are the uses of MRI? To begin, not only are there a variety of scanning methodologies available, but there are also a variety of MRI methodologies available which provide
Magnetic Resonance Imaging What are the uses of MRI? To begin, not only are there a variety of scanning methodologies available, but there are also a variety of MRI methodologies available which provide
Chemistry 307 Chapter 10 Nuclear Magnetic Resonance
 Chemistry 307 Chapter 10 Nuclear Magnetic Resonance Nuclear magnetic resonance (NMR) spectroscopy is one of three spectroscopic techniques that are useful tools for determining the structures of organic
Chemistry 307 Chapter 10 Nuclear Magnetic Resonance Nuclear magnetic resonance (NMR) spectroscopy is one of three spectroscopic techniques that are useful tools for determining the structures of organic
Signal Manipulation. time domain NMR signal in MHz range is converted to khz (audio) range by mixing with the reference ( carrier ) frequency
 NMR Spectroscopy: 3 Signal Manipulation time domain NMR signal in MHz range is converted to khz (audio) range by mixing with the reference ( carrier ) frequency Ref in (MHz) mixer Signal in (MHz) Signal
NMR Spectroscopy: 3 Signal Manipulation time domain NMR signal in MHz range is converted to khz (audio) range by mixing with the reference ( carrier ) frequency Ref in (MHz) mixer Signal in (MHz) Signal
Quantum control of individual electron and nuclear spins in diamond lattice
 Quantum control of individual electron and nuclear spins in diamond lattice Mikhail Lukin Physics Department, Harvard University Collaborators: L.Childress, M.Gurudev Dutt, J.Taylor, D.Chang, L.Jiang,A.Zibrov
Quantum control of individual electron and nuclear spins in diamond lattice Mikhail Lukin Physics Department, Harvard University Collaborators: L.Childress, M.Gurudev Dutt, J.Taylor, D.Chang, L.Jiang,A.Zibrov
Spin-lattice and spin-spin relaxation
 Spin-lattice and spin-spin relaation Sequence of events in the NMR eperiment: (i) application of a 90 pulse alters the population ratios, and creates transverse magnetic field components (M () ); (ii)
Spin-lattice and spin-spin relaation Sequence of events in the NMR eperiment: (i) application of a 90 pulse alters the population ratios, and creates transverse magnetic field components (M () ); (ii)
Masses in Atomic Units
 Nuclear Composition - the forces binding protons and neutrons in the nucleus are much stronger (binding energy of MeV) than the forces binding electrons to the atom (binding energy of ev) - the constituents
Nuclear Composition - the forces binding protons and neutrons in the nucleus are much stronger (binding energy of MeV) than the forces binding electrons to the atom (binding energy of ev) - the constituents
NMR-the basic principles and its use in studies of water/ethanol/mixture
 NMR-the basic principles and its use in studies of water/ethanol/mixture Ayhan DEMİR, Bachelor Degree Project in Chemistry, 15 ECTS, April 2012, Sweden. Supervisor: Prof. Per Olof WESTLUND, Dr.Tobias SPARRMAN
NMR-the basic principles and its use in studies of water/ethanol/mixture Ayhan DEMİR, Bachelor Degree Project in Chemistry, 15 ECTS, April 2012, Sweden. Supervisor: Prof. Per Olof WESTLUND, Dr.Tobias SPARRMAN
Chapter 19 Nuclear Magnetic Resonance Spectroscopy (NMR)
 Chapter 19 Nuclear Magnetic Resonance Spectroscopy (NMR) 23 pages 2 weeks worth! Problems : 1, 2, 3, 4, 7, 10, 11, 19, 20, 22, 24, 27, 30, 34, 35 Absorption of radio-frequency E from 4-900 MHz (wavelengths
Chapter 19 Nuclear Magnetic Resonance Spectroscopy (NMR) 23 pages 2 weeks worth! Problems : 1, 2, 3, 4, 7, 10, 11, 19, 20, 22, 24, 27, 30, 34, 35 Absorption of radio-frequency E from 4-900 MHz (wavelengths
Atomic Structure Ron Robertson
 Atomic Structure Ron Robertson r2 n:\files\courses\1110-20\2010 possible slides for web\atomicstructuretrans.doc I. What is Light? Debate in 1600's: Since waves or particles can transfer energy, what is
Atomic Structure Ron Robertson r2 n:\files\courses\1110-20\2010 possible slides for web\atomicstructuretrans.doc I. What is Light? Debate in 1600's: Since waves or particles can transfer energy, what is
Nuclear Magnetic Resonance and Its Application in Condensed Matter Physics
 Nuclear Magnetic Resonance and Its Application in Condensed Matter Physics Kangbo Hao 1. Introduction Nuclear Magnetic Resonance (NMR) is a physics phenomenon first observed by Isidor Rabi in 1938. [1]
Nuclear Magnetic Resonance and Its Application in Condensed Matter Physics Kangbo Hao 1. Introduction Nuclear Magnetic Resonance (NMR) is a physics phenomenon first observed by Isidor Rabi in 1938. [1]
Time out states and transitions
 Time out states and transitions Spectroscopy transitions between energy states of a molecule excited by absorption or emission of a photon hn = DE = E i - E f Energy levels due to interactions between
Time out states and transitions Spectroscopy transitions between energy states of a molecule excited by absorption or emission of a photon hn = DE = E i - E f Energy levels due to interactions between
PHYS 1624 University Physics I. PHYS 2644 University Physics II
 PHYS 1624 Physics I An introduction to mechanics, heat, and wave motion. This is a calculus- based course for Scientists and Engineers. 4 hours (3 lecture/3 lab) Prerequisites: Credit for MATH 2413 (Calculus
PHYS 1624 Physics I An introduction to mechanics, heat, and wave motion. This is a calculus- based course for Scientists and Engineers. 4 hours (3 lecture/3 lab) Prerequisites: Credit for MATH 2413 (Calculus
Chem 1A Exam 2 Review Problems
 Chem 1A Exam 2 Review Problems 1. At 0.967 atm, the height of mercury in a barometer is 0.735 m. If the mercury were replaced with water, what height of water (in meters) would be supported at this pressure?
Chem 1A Exam 2 Review Problems 1. At 0.967 atm, the height of mercury in a barometer is 0.735 m. If the mercury were replaced with water, what height of water (in meters) would be supported at this pressure?
Atomic and Nuclear Physics
 Atomic and Nuclear Physics Nuclear Physics Nuclear Magnetic Resonance LD Physics Leaflets P6.5.3.1 Nuclear magnetic resonance in polystyrene, glycerine and teflon Objects g Nuclear Magnetic Resonance on
Atomic and Nuclear Physics Nuclear Physics Nuclear Magnetic Resonance LD Physics Leaflets P6.5.3.1 Nuclear magnetic resonance in polystyrene, glycerine and teflon Objects g Nuclear Magnetic Resonance on
NMR practice times. Mo 2---8 pm Jim 2-4:30 Ning 4:30-7. Tues 2--- 8 pm Jianing 2-4:30 Ting 4:30-7. Fri 10- --- 7 Donia 10-12:00 Ilya 2-4
 NMR practice times 1 Mo 2---8 pm Jim 2-4:30 Ning 4:30-7 Tues 2--- 8 pm Jianing 2-4:30 Ting 4:30-7 Wed 4:30---8 John 5:00-7:30 Fri 10- --- 7 Donia 10-12:00 Ilya 2-4 Samples are listed in the homework along
NMR practice times 1 Mo 2---8 pm Jim 2-4:30 Ning 4:30-7 Tues 2--- 8 pm Jianing 2-4:30 Ting 4:30-7 Wed 4:30---8 John 5:00-7:30 Fri 10- --- 7 Donia 10-12:00 Ilya 2-4 Samples are listed in the homework along
7-5.5. Translate chemical symbols and the chemical formulas of common substances to show the component parts of the substances including:
 7-5.5 Translate chemical symbols and the chemical formulas of common substances to show the component parts of the substances including: NaCl [salt], H 2 O [water], C 6 H 12 O 6 [simple sugar], O 2 [oxygen
7-5.5 Translate chemical symbols and the chemical formulas of common substances to show the component parts of the substances including: NaCl [salt], H 2 O [water], C 6 H 12 O 6 [simple sugar], O 2 [oxygen
For example: (Example is from page 50 of the Thinkbook)
 SOLVING COMBINED SPECTROSCOPY PROBLEMS: Lecture Supplement: page 50-53 in Thinkbook CFQ s and PP s: page 216 241 in Thinkbook Introduction: The structure of an unknown molecule can be determined using
SOLVING COMBINED SPECTROSCOPY PROBLEMS: Lecture Supplement: page 50-53 in Thinkbook CFQ s and PP s: page 216 241 in Thinkbook Introduction: The structure of an unknown molecule can be determined using
Atomic Calculations. 2.1 Composition of the Atom. number of protons + number of neutrons = mass number
 2.1 Composition of the Atom Atomic Calculations number of protons + number of neutrons = mass number number of neutrons = mass number - number of protons number of protons = number of electrons IF positive
2.1 Composition of the Atom Atomic Calculations number of protons + number of neutrons = mass number number of neutrons = mass number - number of protons number of protons = number of electrons IF positive
