Leaf photosynthetic characteristics in eight shaded Malaysian filmy ferns
|
|
|
- April Wells
- 8 years ago
- Views:
Transcription
1 Características fotosintéticas foliares en ocho himenofiláceas de ambientes sombreados en Malasia Nurul Hafiza MR, KT Yong, N Osman, A Nasrulhaq-Boyce Abstract. The photosynthetic characteristics of eight Malaysian Hymenophyllaceae filmy ferns from shady habitats were investigated in this study. Chlorophyll content was highest in Trichomanes meifolium, followed by Cephalomanes obscurum, Hymenophyllum serrulatum, H. denticulatum, H. javanicum, H. acanthoides, H. exsertum and H. blandum, with values ranging from 3.3 to. mg/g fresh weight. Soluble protein content was remarkably high in H. serrulatum, with values of 53 ± 3.5 mg/g, followed by H. denticulatum, H. acanthoides, T. meifolium and the other species. Protein to chlorophyll ratios in the filmy ferns were low as expected, except for in H. serrulatum, H. acanthoides and H. denticulatum. Chloroplast number and size ranged between 3 to 13 per cell profile, and between. to.5 µm in diameter, in the Hymenophyllaceae. Quantum efficiency measurements in four Hymenophyllaceae spp. exhibited Fv/Fm values ranging between.73 to.1. The Hymenophyllaceae spp. also showed low in vivo CO assimilatory rates and light saturation points, ranging between 5 to 15 µmol CO /m /s and below 15 µmol/m /s, respectively. The findings add further to our understanding on how the filmy ferns adapt and thrive in their humid and shady habitats. Keywords: Plant science; Photosynthesis; Filmy ferns; Hymenophyllaceae; Shaded habitats. Resumen. Las características fotosintéticas de ocho Malasia Hymenophyllaceae helechos membranosos de hábitats sombreados fueron investigados en este estudio. El contenido de clorofila fue mayor en Trichomanes meifolium, seguido por Cephalomanes obscurum, Hymenophyllum serrulatum, H. denticulatum, H. javanicum, H. acanthoides, H. y H. exsertum blandum con valores que van desde 3,3 hasta, mg/g peso fresco. El contenido de proteína soluble fue notablemente alta en H. serrulatum, con valores de 53 ± 3,5 mg/g, seguido por H. denticulatum, H. acanthoides, T. meifolium y las otras especies. La relación proteína:clorofila en los helechos fue baja como se esperaba, excepto en H. serrulatum, H. acanthoides y H. denticulatum. Número y tamaño de cloroplasto oscilaron entre las 3 y 13 por perfil de célula y entre, a,5 μm de diámetro, en el Hymenophyllaceae. Mediciones de la eficiencia cuántica de cuatro Hymenophyllaceae spp. mostraron valores de Fv / Fm que oscilan entre,73 a,1. El Hymenophyllaceae spp. también mostró baja en las tasas de asimilación de CO in vivo y los puntos de saturación, que oscila entre 5 a 15 µmol CO /m /s y por debajo de 15 µmol/m /s, respectivamente. Los hallazgos se añaden a nuestra comprensión de cómo los helechos membranosos se adaptan y prosperan en sus hábitats húmedos y sombríos. Palabras clave: Ciencia de las plantas; Fotosíntesis; Helechos vaporosos; Hymenophyllaceae; Hábitats sombreados. Institute of Biological Sciences, Faculty of Science, University of Malaya, 53 Kuala Lumpur, Malaysia. Address Correspondence to: Prof. Amru Nasrulhaq Boyce, amru@um.edu.my Recibido / Received.VIII.13. Aceptado / Accepted 1.XII.13.
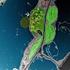
2 35 Nurul Hafiza MR et al., FYTON 3 (1) INTRODUCTION Filmy ferns from the Hymenophyllaceae family are aesthetically attractive ferns that live abundantly in the humid tropical rainforest. They show an amazing diversity in terms of morphology and the habitats they occupy, making Hymenophyllaceae a great model for studying evolutionary ecology and related adaptive survival strategies in pteridophytes (Dubuisson et al., 3). They are small in size and some of them appear as very dark green or even black clumps and can be characterized by a single-cell thick lamina with lack of cuticle, giving them direct contact with air in their surroundings and are able to carry out rapid gas exchange. Their physiology, however, has received little attention despite the large amount of literature on their morphology and taxonomy. Studies on sun and shade plants, particularly ferns, have been carried out for many years, since the early 19s albeit infrequently. It has been well documented that shaded plants are incapable of achieving high photosynthetic rates although they perform more efficiently at low light irradiances (Boardman, 1977; Givnish, 19). Sun and shade plants have been shown to exhibit obvious differences in their morphology, ultrastructure, physiology and biochemistry. Shade plants generally possess fewer, larger chloroplasts with larger grana, higher pigment content and lower photosynthetic rates. On the other hand, most plants that grow in high light intensities in their natural habitats show the opposite characteristics of shaded plants, with higher rates of photosynthesis at saturating light intensity. Similar studies have been reported, particularly on ferns and bryophytes (Nasrulhaq-Boyce & Mohamed, 197; Nasrulhaq-Boyce & Duckett, 1991; Proctor, 3; Marschall & Proctor, ). More recently, Wong et al. (1) studied the photosynthetic rates of woody and fern species from different light regimes. They reported that the shade adapted plants showed low gross photosynthetic rate when the dark-adapted leaves were exposed to 5 or µmol/m /s photosynthetic photon flux (PPF). Proctor (3) studied and compared the ecophysiological characteristics, such as light response, water relations and dessication tolerance between the filmy ferns, Hymenophyllum peltatum (Poir.) Desv. [syn. H. wilsonii] and H. tunbrigense (L.) Sm. They reported that the CO uptake in the filmy ferns studied were low at low light irradiances. Several authors have reported similar findings with other ferns species in terms of their structure and ecological adaptation (Hebant & Lee, 19; Nasrulhaq-Boyce & Duckett, 1991; Johnson et al., ; Proctor, 5; Proctor, 1). The aim of the present study was to investigate the chlorophyll and protein content, and chloroplast anatomy in selected Hymenophyllaceae species as well as their chlorophyll fluorescence and photosynthetic activity in an attempt to understand further photosynthesis in the shaded filmy ferns. MATERIALS AND METHODS Plant material. The filmy ferns were collected from Gunung (Mount) Ulu Kali, Pahang which is 3 miles northeast of Kuala Lumpur. Gunung Ulu Kali (177 m.a.s.l.) is the southernmost tallest mountain in the Titiwangsa Range in Peninsular Malaysia, not far from the summit area where the renown hill resort, Genting Highlands is situated (Fig. 1). Although there are heavy anthropogenic activities in the resort area, the surrounding region is made up of large tracts of montane forests that remain untouched and house rich biological diversity. The plants investigated in the present study were obtained from these forests, with an elevation ranging between m.a.s.l. and annual rainfall ranging be- Fig. 1. Map of Gunung Ulu Kali, Genting Highlands, Pahang, Malaysia, where the ferns were collected from. Fig. 1. Mapa de Gunung Ulu Kali, Genting Highlands, Pahang, Malasia, donde se recogieron los helechos.
3 355 tween -37 mm. Irradiance, surrounding temperature and relative humidity measured ranged between µmol/ m /s, C and 51% - 1%, respectively. The mean temperature was 17.9 C, while the mean RH was 95.%. The plants were collected from the ground and tree trunks in moist and shady areas, together with their underlying soil, placed in plastic bags, and brought back to the laboratory for work on the same day. Chlorophyll and protein analysis. Chlorophyll was extracted in % acetone from freshly collected leaves and its content was determined using the method of Arnon (199) as outlined in Nasrulhaq-Boyce et al. (11). Total protein content was estimated using the Lowry et al. (1951) method. Measurement of light intensity, relative humidity and temperature. Light intensity was taken using a Lux-meter (Field scout quantum meter, USA) at each site where the fern species under study occur naturally. Readings were taken between 11. am to. pm in the early afternoon when it was certain that no dark clouds were present in the sky. Readings were taken between ten to fifteen times. The same procedure was followed for relative humidity and temperature. Chlorophyll-fluorescence measurements. Chlorophyll fluorescence measurements were made using a modulated chlorophyll fluorometer (Hansatech Ltd. England). The leaves of the ferns were placed in a standard Hansatech leaf clip and chlorophyll fluorescence measured after dark adaptation for 1 minutes. Photosynthetic activity and light response determination. Gas exchange measurements were made with a portable LICOR photosynthesis system (LI-XT, USA). Light response curve measurements were taken within a PAR range of - µmol/m /s. Microscopy. Fresh leaves were cut into small pieces and then mounted on a slide using distilled water as medium. The slides were examined and photographed using a Leica DFC9 digital camera that attached to a compound microscope. Chloroplast size and number per cell were calculated from different cells, with four cells each of five different leaves. Statistical analysis. The data obtained were pooled and analysed using Minitab Pro v1.1.. and SPSS statistical software. A one-way ANOVA was applied to evaluate significant differences in the studied parameters. Least significant difference was calculated following a significance at p=.5. RESULTS The Hymenophyllaceae species collected in the present study were generally from constantly moist, shaded highland habitats. Table 1 shows that the irradiances of their habitats were considerably low for all the eight species of Hymenophyllaceae, where the light intensities received by the plants were below 1 µmol/ m /s. Light intensity increased in the morning as a result of sunrise but remained low and fairly constant until early afternoon when it started to decrease. The temperature and relative humidity readings of the habitats of the Hymenophyllaceae species ranged between C and %, respectively. Table and Table 3 show the results of the determination of the chlorophyll and protein contents in the leaves of the eight shaded Hymenophyllaceae species. Chlorophyll content expressed on a fresh weight basis was notably very high in Trichomanes meifolium Bory (. mg/g), followed by the other species ranging between mg/g fresh weight (Table ). Cephalomanes obscurum (Blume) K. Iwats., Hymenophyllum serrulatum C.Chr. and H. denticulatum Sw. showed chlorophyll values of.,.3 and 5. mg/g fresh weight, respectively, whilst the other species exhibited chlorophyll values of.7 (H. Table 1. Summary of some environmental measurements of the habitats from which the species were collected. Tabla 1. Resumen de algunas de las mediciones ambientales de los hábitats de los que se recogieron las especies. Species Irradiances (µmol/m /s] Temperature ( C) Relative Humidity (%) Cephalomanes obscurum 33.3 ± ±.5 9. ±.5 Hymenophyllum acanthoides 9. ± 1.1. ±.9.9 ± 1.3 H. blandum. ±.3 1. ± ±. H. denticulatum 9.9 ±.9.5 ±. 9. ±. H. exsertum 5.7 ± ± ±.3 H. javanicum 93.3 ±.1 3. ±.7. ±.35 H. serrulatum.1 ± ±.7. ±.91 Trichomanes meifolium 5. ± ± ±. All values are expressed as mean ± S.E. Todos los valores se expresan como promedio ± error estándar (E.E.).
4 35 Nurul Hafiza MR et al., FYTON 3 (1) A B Light irradiance, µmole/m /s Light irradiance, µmole/m /s C Light irradiance, µmole/m /s D Light irradiance, µmole/m /s E Light irradiance, µmole/m /s Fig.. Photosynthetic light response curves of shaded Hymenophyllaceae species (A) Trichomanes meiofolium; (B) Hymenophyllum javanicum; (C) H. denticulatum; (D) H. exsertum, (E) H. serrulatum, at varying light intensities. Measurements were taken in the field using a portable infrared gas analyzer (LICOR, LI-XT). Data are expressed in terms of projected leaf area. Each point is an average of three readings. Fig.. Curvas de respuesta de la fotosíntesis a la luz de las especies sombreadas Hymenophyllaceae (A) Trichomanes meiofolium; (B) Hymenophyllum javanicum; (C) H. denticulatum; (D) H. exsertum, (E) H. serrulatum, a diferentes intensidades de luz. Las mediciones se realizaron en el campo usando un analizador de gases infrarrojo portátil (LICOR, LI-XT). Los datos se expresan en términos de área foliar proyectada. Cada punto es un promedio de tres lecturas.
5 357 javanicum Spreng.), 3. [H. acanthoides (Bosch) Rosenst.], 3.5 (H. exsertum Wall. ex Hook.), and 3.3 mg/g fresh weight (H. blandum Racib.). However, the opposite was observed with regard to the chlorophyll a/b ratio, where the Hymenophyllaceae species exhibited lower ratios, ranging between.9~1.9. Contrary to the level of chlorophyll, the soluble protein content determined using the Lowry method, was generally lower in all Hymenophyllaceae species, with the exception of H. serrulatum, (Table 3). Soluble protein content in H. serrulatum, however, was remarkably high, recording values of 53 mg/g fresh weight. H. denticulatum, H. acanthoides, T. meifolium, C. obscurum, H. javanicum, H. exsertum and H. blandum exhibited protein content ranging between.3 to 9.7 mg/g fresh weight. In addition to this, the protein/chlorophyll ratio in H. serrulatum was also higher (Table 3). It has been well documented that shaded ferns have larger and fewer chloroplasts but are richer in chlorophyll content when compared to sun ferns (Boardman, 1977; Nasrulhaq- Boyce and Mohamed, 197; Nasrulhaq-Boyce & Duckett, 1991). Light microscopy observations of the leaves of all the Hymenophyllaceae species collected showed that C. obscurum possessed the highest number of chloroplast per cell profile, numbering 13, followed by H. acanthoides (3), H. serrulatum (5), H. exsertum (), H. javanicum (3), H. denticulatum (), and H. blandum (3) (Table ). However, the chloroplasts were significantly larger in H. blandum (.5 µm) and H. denticulatum (. µm) leaves which together had the fewest chloroplast number per cell than the other species, namely C. obscurum, H. acanthoides, H. serrulatum, H. exsertum and H. javanicum. These results were not suprising as the species with the larger number of chloroplasts also had smaller chloroplasts. Nevertheless the chloroplast numbers were relatively low and the leaf parenchyma cells were closely packed with chloroplasts. Table. Leaf chlorophyll content of Hymenophyllaceae species. Tabla. Contenido foliar de clorofila de las especies Hymenophyllaceae. Species Total chlorophyll (mg/g fresh weight) Chlorophyll a (mg/g fresh weight) Chlorophyll b (mg/g fresh weight) Chlorophyll a/b ratio Shade Cephalomanes obscurum. ab ±. 3.3 bc ±. 3. a ±..9 c ±. Hymenophyllum acanthoides 3. bcd ±..5 bc ± c ± ab ±.1 H. blandum 3.3 cd ±.5.1 c ±.3 1. c ± b ±.13 H. denticulatum 5. abc ±. 3. abc ±.1. c ± a ±.1 H. exsertum 3.5 cd ±.. bc ± c ±.1 1. ab ±.9 H. javanicum.7 bcd ±.5.9 bc ± c ±. 1.7 ab ±. H. serrulatum.3 abc ± 1..1 ab ± 1.1. bc ±. 1.9 ab ±.3 Trichomanes meifolium. a ±.3 5. a ±. 3.1 ab ±.1 1. ab ±. All values are expressed as mean ± SE. Means that do not share a letter are significantly different at α=.5. Todos los valores se expresan como promedio ± error estándar (E.E.). Los promedios que no comparten una letra son significativamente diferentes a α=,5. Table 3. Leaf soluble protein content of Hymenophyllaceae species determined via the Lowry method. Tabla 3. Contenido foliar de proteína soluble de especies Hymenophyllaceae determinado mediante el método de Lowry. Species Protein content (mg/g) Protein/chl ratio Shade Cephalomanes obscurum 1.3 c ±.. c ±.1 Hymenophyllum acanthoides. b ±.7.9 bc ±.79 H. blandum.3 c ±.3.1 c ±. H. denticulatum 9.7 b ± c ±.1 H. exsertum. c ±.5.1 c ±.1 H. javanicum 1. c ±..3 c ±.3 H. serrulatum 53 a ± a ±. Trichomanes meifolium. bc ±..5 c ±.3 All values are expressed as mean ± S.E. Means that do not share a letter are significantly different at α=.5. Todos los valores se expresan como promedio ± error estándar (E.E.). Los promedios que no comparten una letra son significativamente diferentes a α=,5.
6 35 Nurul Hafiza MR et al., FYTON 3 (1) Table. Mesophyll chloroplast number per profile in the leaves of the Hymenophyllaceae species. Tabla. Número de cloroplastos del mesófilo por perfil en las hojas de las especies de Hymenophyllaceae. Species Chloroplast number per cell profile Chloroplast size (µm) Cephalomanes obscurum 13 a ± 3. c ±. Hymenophyllum acanthoides 3 b ± 3 5. b ±.1 H. blandum 3 e ± 1.5 a ±.15 H. denticulatum de ± 1. a ±.11 H. exsertum cd ± 5.7 b ±.11 H. javanicum 3 de ± 5.7 b ±.9 H. serrulatum 5 bc ±.1 bc ±.1 All values are expressed as mean ± SE. Means that do not share a letter are significantly different at α=.5. Todos los valores se expresan como promedio ± error estándar (E.E.). Los promedios que no comparten una letra son significativamente diferentes a α=,5. Table 5. Chlorophyll fluorescence in the leaves of the Hymenophyllaceae species collected. Tabla 5. Fluorescencia de la clorofila en las hojas de las especies de Hymenophyllaceae recogidas. Species Fv Fm Fv/Fm Hymenophyllum denticulatum 379 b ± a ± 15.1 a ±.9 H. javanicum 59 a ± 5 a ± 75.7 b ±.1 H. serrulatum 51 ab ± a ± b ±. Trichomanes meifolium 51 ab ± a ± ab ±.1 All values are expressed as mean ± SE. Means that do not share a letter are significantly different at α=.5. Todos los valores se expresan como promedio ± error estándar (E.E.). Los promedios que no comparten una letra son significativamente diferentes a α=,5. Chlorophyll fluorescence in H. denticulatum, T. meifolium, H. javanicum and H. serrulatum exhibited Fv/Fm ratios or photosynthetic quantum yield values, ranging between.71 to.1 (Table 5) which are values within the range for a normal healthy leaf. Figure shows the in vivo light saturation curve for photosynthesic rates in the Hymenophyllaceae species determined in their natural habitats. Initial in vivo light saturation studies with a LICOR infrared gas analyzer on five Hymenophyllaceae species, namely, H. serrulatum, T. meiofolium, H. exsertum, H. denticulatum and H. javanicum showed that CO assimilatory rates for all these filmy ferns were low ranging between 5 to 15 µmol CO /m /s. Saturation of CO uptake occurred at low irradiances, between 1 µmol/m /s to 15 µmol/m /s. Photosynthetic activity increased with increasing light intensity until light saturation was reached, throughout the duration of the experiments. The diurnal photosynthetic rates fluctuated throughout the day depending on the light intensity that came through the canopy to the forest floor. The filmy ferns showed optimal photosynthesis at light intensities lower than 15 µmol/m /s, a light intensity to which they are normally exposed to in their native environment. DISCUSSION As it is well known and documented, the eight species selected for this study, thrived in cool, moist habitats at high elevation with low light irradiances, as shown in Table 1. Similar readings were reported previously in the same area of study (Nasrulhaq-Boyce et al., 11). There was relatively little difference in irradiance, temperature and relative humidity between the different habitats where the ferns were collected as mentioned above and in Table 1. As a result of this it was not possible to see any significant correlation between habitat light irradiance and protein content or habitat light irradiance and chloroplast number or the rate of photosynthetic activity, even though the two ferns with the highest chlorophyll content were collected from habitats with the lowest irradiances. As shown in Table, the chlorophyll content expressed on a fresh weight basis was high in all the species studied. It was notably very high in the needle-like dark green leaves of Trichomanes meifolium. High chlorophyll values have been reported previously in Selaginella willdenowii (Desv. ex Poir.) Baker (Hebant & Lee, 19), Cyclosorus multilineatus (Wall. ex Hook.) Tardieu & C. Chr. [syn. Abacopteris multilineata],
7 359 Christensenia aesculifolia (Blume) Maxon, Tectaria singaporiana (Wall. ex Hook. & Grev.) Ching. [syn. Tectaria singaporeana], T. vasta (Blume) Copel. (Nasrulhaq-Boyce & Mohamed, 197), and Teratophyllum rotundifoliatum (Bonap.) Holttum (Nasrulhaq-Boyce & Duckett, 1991), with values ranging between. ~ 5. mg/g fresh weight. However, the values between.3 to. mg/g fresh weight observed for Trichomanes meifolium, Cephalomanes obscurum and Hymenophyllum serrulatum were remarkably high. More recently, studies on the shaded Malaysian Pogonatum cirratum subsp. macrophyllum (Dozy & Molk.) Hyvönen moss species showed chlorophyll values of.79 mg/g fresh weight (Nasrulhaq-Boyce et al., 11). The high chlorophyll content associated with the low irradiance level in the habitats in which the filmy ferns lived reflects the adaptation to living in a shady environment. Another observation made was the generally low chlorophyll a/b ratios in the Hymenophyllaceae leaves of the plants compared to the two ferns living in sunlit habitats, namely Dicranopteris linearis and Nephrolepis biserrata (Table ). This is indicative of shady plants possessing a larger proportion of chl a/b-binding light harvesting complexes associated with photosystem II (PSII) and also of chloroplasts with more granal thylakoids where PSII resides. It can be regarded as an adaptation for plants adapted to living in low light environments (Anderson et al., 19; Chow et al., 19; Brach et al., 1993; Björkman & Demmig-Adams, 1995; Johnson et al., ; Marschall & Proctor. ; Nasrulhaq-Boyce et al., 11; Proctor, 1; Wong et al., 1). A previous study on Trichomanes speciosum Willd. reported that the low chlorophyll a:b ratio observed was accompanied by an increase in the proportion of stacked thylakoids within the chloroplasts ( Johnson et al., ). This was also reported for the deep shade fern Teratophyllum rotundifoliatum, which possesses large grana stacks with thylakoid membranes packing the stroma of the chloroplasts (Nasrulhaq-Boyce & Duckett, 1991). However, (generally) plants found in higher light environments tend to produce a higher chlorophyll a:b ratio (Ludlow & Wolf, 1975). More recently, Mathew et al. (5) reported high chlorophyll a:b ratios in the sunny fern Onoclea sensibilis L. indicating a smaller light harvesting system with less stacking of thylakoid membranes. Generally, plants living in shade environments have larger chloroplasts but lower chloroplast number per cell than plants thriving in an exposed area (Boardman, 1977). This was observed in Teratophyllum rotundifoliatum and the other ferns species mentioned above (Hebant & Lee, 19; Givnish, 19; Nasrulhaq-Boyce & Duckett, 1991; Proctor, ). In this study, the chloroplast numbers per profile observed in the leaves of the shaded H. acanthoides, H. serrulatum, H. exsertum, H. javanicum, H. denticulatum and H. blandum were relatively low, between 3 and 3, with significantly large chloroplast diameters ranging between 5. to.1 µm. This is consistent with previous research findings, with the exception of C. obscurum, which had a high numbers of chloroplast (13) and the smallest chloroplast size (. µm). Not all shade plants exhibit similar anatomical structures. For instance, Fatsia japonica (Thunb.) Decne. & Planch. and Alocasia macrorrhiza (L.) Schott have been reported to exhibit low numbers of chloroplasts per cell whilst living in environments of high light irradiance (Chow et al., 19). Trichomanes meifolium, which grows in shady habitats, possess lamina cells with 1- chloroplasts per cell, with diameter ranging between 3- µm, despite their needle-like structure (Nasrulhaq-Boyce & Duckett, unpublished data). Recently, Sheue et al. (7) reported a single giant cup-shaped chloroplast, termed a bizonoplast found in the deep-shade spike moss Selaginella erythropus (Mart.) Spring. They suggested that the chloroplast structure may have an evolutionary significance in photosynthetic functionality in adaptation to low-light environments. It is well documented that the amount of soluble protein content or ratio of soluble protein to chlorophyll content in shade plants is considerably lower than that in the sun species (Boardman, 1977; Givnish, 19; Nasrulhaq-Boyce & Mohamed, 197; Nasrulhaq-Boyce et al., 11). It has been suggested that sun plants have more protein, in particular the major protein in the leaf, the photosynthetic enzyme ribulose bisphosphate carboxylase (rubisco), to enable them to photosynthesize more rapidly and efficiently. As shown in Table 3 the protein content determined by the Lowry method was low in all the shaded ferns, with values ranging between mg/g fresh weight, compared to the sun ferns which registered values ranging between 1- mg/g. The exception was H. serrulatum which posted a remarkably high value, above 5 mg/g fresh weight. The latter observation runs contrary to the general rule for sun and shade plants. Nevertheless, all but one of the Hymenophyllaceae species studied exhibited a lower soluble protein to chlorophyll ratio (Table 3) than do the sun ferns Dicranopteris linearis and Nephrolepis biserrata. Chlorophyll fluorescence in H. denticulatum, Trichomanes meifolium and H. serrulatum exhibited maximal fluorescence (Fv/Fm) ratios or photosynthetic quantum yield values, ranging between.73 to.1 (Table 5). These values are generally slightly lower than what has been reported for a normal healthy plant (. ~.3) (Peter Horton, personal communication). The low light intensities in which these plants thrive makes it difficult to get an accurate estimate of the maximum quantum yield using fluorescence. Our data suggest the maximum value of the ratio of variable to maximal fluorescence (Fv/Fm) to be ~.. Previous studies on T. speciosum showed Fv/Fm ratios of around.75, lower than the Fv/Fm ratios of T. meifolium and H. denticulatum studied ( Johnson et al., ). They suggested that T. speciosum possessed a limited ability to quench chlorophyll fluorescence. However, Proctor (3) has reported Fv/Fm ratios of about. in H. peltatum and H. tunbrigense, which is close to the values obtained for the Malaysian Hymenophyllaceae ferns and T. speciosum. Recent studies by Wong et al. (1) similarly reported Fv/Fm
8 3 Nurul Hafiza MR et al., FYTON 3 (1) for the dark adapted-fern leaves of Pyrrosia lingua (Thunb.) Farw., Asplenium nidus L. [syn. A. antiquum], Diplazium donianum (Mett.) Tardieu and Angiopteris somae (Hayata) Makino & Nemoto [syn. Archangiopteris somai] to be in the region of ~.. The lower chlorophyll fluorescence values observed in H. denticulatum, T. meifolium, H. javanicum and H. serrulatum probably indicate their limited ability to quench chlorophyll fluorescence. It has been reported in higher vascular plants that plants grown under high light intensities show a greater photosynthetic capacity at light saturation than shade plants, but lower rates at low light intensities compared to shade plants (Boardman, 1977; Nasrulhaq-Boyce & Mohamed, 197; Givnish, 19; Johnson et al. ; Proctor, 1; Wong et al., 1). Previous studies on the filmy ferns Hymenophyllum wilsonii and Hymenophyllum tunbrigense have shown that saturation of photosynthetic electron flow and CO uptake were lower at low light irradiances (Proctor, 3). It has been reported that isolated chloroplasts from sun Malaysian ferns showed greater in vitro photochemical activity at saturating irradiance than chloroplasts from shade ferns (Nasrulhaq-Boyce & Mohamed, 197). They suggested the greater capacity for electron transport might be attributed to the observed higher level of electron transport carriers (photosynthetic cytochromes f, b 559, b 53 ) found in the sun ferns. Marschall and Proctor () studied 39 species of mosses and 1 liverworts for their ratios of chlorophyll and total carotenoids and light saturation of photosynthetic electron flow. These authors concluded that total chlorophyll, chlorophyll a:b and chlorophyll:carotenoids ratios correlated significantly with photosynthetic photon flux density. The in vivo CO assimilation activities in the leaves of the filmy ferns in this study revealed low rates consistent with those observed in other ferns and bryophytes. Similar recent studies by Proctor (1) showed that the photosynthetic rates of the Hymenophyllaceae species from Trinidad, Venezuela and New Zealand also saturated at low light intensities, where all four of the shade-adapted species had PPFD 95% 51 µmol/m /s. Another study on four fern species, Pyrrosia lingus, Asplenium antiquum, Diplazium donianum and Archangiopteris somai, by Wong et al. (1), showed low gross photosynthetic rate when the dark-adapted leaves were exposed to 5- µmol/m /s of light irradiance. The shadeadapted H. javanicum recorded the lowest photo-assimilatory rates [~5 µmol CO /m /s] followed by H. serrulatum [~9 µmol CO /m /s], H. denticulatum [~1 µmol CO /m /s], T. meiofolium [~11 µmol CO /m /s] and H. exsertum [~15 µmol CO / m /s]. These Hymenophyllaceae species showed optimal photosynthesis at light intensities below 1 µmol/m /s, the highest light intensity to which they are normally exposed to in their natural environment. Proctor (1) proposed that the filmy ferns (Hymenophyllaceae) are a rare example of an evolutionary shift of adaptive strategy from typical vascular plant adaptation to an integrated package adapted to more or less constantly shade humid environments, where its photosynthesis saturates at low irradiance and generally low levels of desiccation tolerance. The results on the filmy ferns are consistent with previous reports on studies with sun and shaded living ferns and show their ability to adapt to their habitats, particularly their need to utilize periodic sun flecks (Boardman, 1977; Givnish, 19; Alfredo et al., 1; Huang et al., 11; Johnson et al., ; Proctor, 3; Proctor, 1; Wong et al., 1). As expected one cannot draw any correlation between the differences in photosynthetic parameters between the species studied with their habitat light irradiances as the latter was relatively similar and all the species were from shaded habitats. Nevertheless, the findings of this study showed that the Hymenophyllaceae species were able to adapt well to their shaded, cool and moist environment, and possessed the ability to make an efficient use of what little light is available to them for photosynthesis and growth. ACKNOWLEDGEMENTS This work was financially supported by the University of Malaya Research University Grant scheme no. RG/11BIO and the Fundamental Research Grant Scheme, Ministry of Higher Education, Malaysia no. FP/1A. REFERENCES Anderson, J.M., W.S. Chow & D.J. Goodchild (19). Thylakoid membrane organisation in sun/shade acclimation. Australian Journal of Plant Physiology 15: 11. Alfredo, O.S., C. Hernandez, E.C. Rafael, A.B. Leon & L.J. Corcuera (1). Differences in light usage among three fern species of genus Blechnum of contrasting ecological breadth in a forest light gradient. Ecological Research 5: Arnon, D.L (199). A copper enzyme is isolated chloroplast polyphenol oxidase in B. vulgaries. Plant Physiology : Björkman, O. & B. Demmig-Adams (199). Regulation of photosynthetic light energy capture, conversion and dissipation in leaves of higher plants. In: Schulze E-D, Caldwell MM (Eds). Ecophysiology of Photosynthesis Vol. 1. Berlin, Germany: Springer Verlag, pp Boardman, N.K. (1977). Comparative photosynthesis of sun and shade plants. Annual Review of Plant Physiology : Brach, A.R., S.J. McNaughton & D.J. Raynal (1993). Photosynthetic adaptibility of two fern species of a Northern Hardwood Forest. American Fern Journal 3: Chow, W.S., L. Qian, D.J. Goodchild & J.M. Anderson (19). Photosynthetic acclimation of Alocasia macrorrhiza (L.) G. Don to growth irradiance: Structure, function and composition of chloroplast. Australian Journal of Plant Physiology 15: Dubuisson, J.Y., S. Hennequin, F. Rakotondrainibe & H. Schneider (3). Ecological diversity and adaptive tendencies in the tropical fern Trichomanes L. (Hymenophyllaceae) with special reference to climbing and epiphytic habits. Botanical Journal of the Linnean Society 1: 1 3.
9 31 Givnish, T.J. (19). Adaptation to sun and shade:a whole-plant perspective. Plant Physiology 15: 3-9. Hebant, C. & D.W. Lee (19). Ultrastructure basis and development control of blue iridescence in Sellaginella leaves. American Journal of Botany 71: Huang, D., L. Wu, J.R. Chen & L. Dong (11). Morphological plasticity, photosynthesis and chlorophyll fluorescence of Athyrium pachyphlebium at different shade levels. Photosynthetica 9: Johnson, G.N., F.J. Rumsey, A.D. Headley & E. Sheffield (). Adaptations to extreme low light in the fern Trichomanes speciosum. New Phytologist 1: Lowry, O.H., N.J. Rosebrough, A.L. Farr & R.J. Randall (1951). Protein measurement with the Folin reagents. Journal of Biological Chemistry 193: Ludlow, C.J. & F.T. Wolf (1975). Photosynthesis and respiration rates of ferns. American Fern Journal 5: 3-. Mathew, W.R., P.S. Joshua, B. Xu, A. Cunkelman & A.B. Ronald (5). A comparison of physiological and morphological properties of deciduous and wintergreen ferns in Sounthern Pennsylvania. American Fern Journal 95: 5-5. Marschall, M. & M.C.F. Proctor (). Are Bryophytes Shade Plants? Photosynthetic Light Responses and Proportions of Chlorophyll a, Chlorophyll b and Total Carotenoids. Annals of Botany 9: Nasrulhaq-Boyce, A. & M.A.H. Mohamed (197). Photosynthetic and respiratory characteristics of Malayan sun and shade ferns. New Phytologist 15: 1 -. Nasrulhaq-Boyce, A. & J.G. Duckett (1991). Dimorphic epidermal cell chloroplasts in the mesophyll less leaves of an extreme shade tropical fern, Teratophyllum rotundifoliatum (R.Bonap.) Holtt.: a light and electron microscope study. New Phytologist 119: 33. Nasrulhaq-Boyce, A., M.A.H. Mohamed, A.L. Lim, S.S. Barakbah, K.T. Yong & D.M. Nor (11). Comparative morphological and photosynthetic studies on three Malaysian species of Pogonatum from habitats of varying light irradiance. Journal of Bryology 33: Proctor, M.C.F. (3). Comparative ecophysiological measurements on the light responses, water relations and dessiccation tolerance of the filmy ferns Hymenophyllum wilsonii Hook. and H. Tunbrigense (L.) Smith. Annals of Botany 91: Proctor, M.C.F. (5). Why do Polytrichaceae have lamellae? Journal of Bryology 7: 1-9. Proctor, M.C.F. (1). Light and desiccation responses of some Hymenophyllaceae (filmy ferns) from Trinidad, Venezuela and New Zealand: poikilohydry in a light-limited but low evaporation ecological niche. Annals of Botany 19: Sheue, C.R., V. Sarafis, S. Kiew, H.Y. Liu, A. Salino, L.L. Kuo- Huang, Y.P. Yang, C.C. Tsai, C.H. Lin, J.W.H. Yong & M.S.B. Ku (7). Bizonoplast, a unique chloroplast in the epidermal cells of microphylis in the shade plant Selaginella erythropu (Selaginellaceae). American Journal of Botany 9: Wong, S.L., C.W. Chen, H.W. Huang & J.H. Weng (1). Using combined measurements for comparison of light induction of stomatal conductance, electron transport rate and CO fixation in woody and fern species adapted to different light regimes. Tree Physiology 3:
Which regions of the electromagnetic spectrum do plants use to drive photosynthesis?
 Which regions of the electromagnetic spectrum do plants use to drive photosynthesis? Green Light: The Forgotten Region of the Spectrum. In the past, plant physiologists used green light as a safe light
Which regions of the electromagnetic spectrum do plants use to drive photosynthesis? Green Light: The Forgotten Region of the Spectrum. In the past, plant physiologists used green light as a safe light
Like The Guy From Krypton Photosynthesis: Energy from Sunlight What Is Photosynthesis?
 Like The Guy From Krypton Photosynthesis: Energy from Sunlight What Is Photosynthesis? Photosynthesis: synthesis from light The broad outline: Plants take in CO 2 and release water and O 2 Light is required
Like The Guy From Krypton Photosynthesis: Energy from Sunlight What Is Photosynthesis? Photosynthesis: synthesis from light The broad outline: Plants take in CO 2 and release water and O 2 Light is required
Photosynthesis (Life from Light)
 Photosynthesis Photosynthesis (Life from Light) Energy needs of life All life needs a constant input of energy o Heterotrophs (consumers) Animals, fungi, most bacteria Get their energy from other organisms
Photosynthesis Photosynthesis (Life from Light) Energy needs of life All life needs a constant input of energy o Heterotrophs (consumers) Animals, fungi, most bacteria Get their energy from other organisms
- 1 - BISC 367 Plant Physiology Laboratory SFU
 - 1 - BISC 367 Plant Physiology Laboratory SFU CO 2 exchange in plants The effect of light intensity and quality on photosynthetic CO 2 fixation and respiration in C3 and C4 plants Using light energy and
- 1 - BISC 367 Plant Physiology Laboratory SFU CO 2 exchange in plants The effect of light intensity and quality on photosynthetic CO 2 fixation and respiration in C3 and C4 plants Using light energy and
Jan Baptisa van Helmont (1648)
 Instructions To help you navigate these slides, you should set your viewer to display thumbnails of these slides. On many viewers, this can be done by pressing the F4 key. The slides should be viewed in
Instructions To help you navigate these slides, you should set your viewer to display thumbnails of these slides. On many viewers, this can be done by pressing the F4 key. The slides should be viewed in
Zhang YG & HY Dai. Keywords: Columnar apple; Photosynthetic rate; Morphological characteristics; Microstructure.
 Comparison of photosynthetic and morphological characteristics, and microstructure of roots and shoots, between columnar apple and standard apple trees of hybrid seedlings Comparación de características
Comparison of photosynthetic and morphological characteristics, and microstructure of roots and shoots, between columnar apple and standard apple trees of hybrid seedlings Comparación de características
Lecture 7 Outline (Ch. 10)
 Lecture 7 Outline (Ch. 10) I. Photosynthesis overview A. Purpose B. Location II. The light vs. the dark reaction III. Chloroplasts pigments A. Light absorption B. Types IV. Light reactions A. Photosystems
Lecture 7 Outline (Ch. 10) I. Photosynthesis overview A. Purpose B. Location II. The light vs. the dark reaction III. Chloroplasts pigments A. Light absorption B. Types IV. Light reactions A. Photosystems
Photosynthesis and (Aerobic) Respiration. Photosynthesis
 Photosynthesis and (Aerobic) Respiration These two processes have many things in common. 1. occur in organelles that seem to be descended from bacteria (endosymbiont theory): chloroplasts and mitochondria
Photosynthesis and (Aerobic) Respiration These two processes have many things in common. 1. occur in organelles that seem to be descended from bacteria (endosymbiont theory): chloroplasts and mitochondria
VII. NARRATION FOR PHOTOSYNTHESIS: TRANSFORMING LIGHT TO LIFE
 7. Why do leaves turn color in the fall? 8. How are photosystems I and II different? How are they related? 9. What is the source of energy for dark reactions? 10. Describe the C3 cycle. 11. What is the
7. Why do leaves turn color in the fall? 8. How are photosystems I and II different? How are they related? 9. What is the source of energy for dark reactions? 10. Describe the C3 cycle. 11. What is the
Photosynthesis January 23 Feb 1, 2013 WARM-UP JAN 23/24. Mr. Stephens, IB Biology III 1
 WARM-UP JAN 23/24 Mr. Stephens, IB Biology III 1 Photosynthesis & Cellular Respiration What is the connection between Photosynthesis and Cellular Respiration? Energy Production Inorganic Molecules Specialized
WARM-UP JAN 23/24 Mr. Stephens, IB Biology III 1 Photosynthesis & Cellular Respiration What is the connection between Photosynthesis and Cellular Respiration? Energy Production Inorganic Molecules Specialized
Biology Slide 1 of 51
 Biology 1 of 51 8-3 The Reactions of Photosynthesis 2 of 51 Inside a Chloroplast 1. In plants, photosynthesis takes place inside chloroplasts. Plant Chloroplast Plant cells 3 of 51 Inside a Chloroplast
Biology 1 of 51 8-3 The Reactions of Photosynthesis 2 of 51 Inside a Chloroplast 1. In plants, photosynthesis takes place inside chloroplasts. Plant Chloroplast Plant cells 3 of 51 Inside a Chloroplast
Photosynthesis. Photosynthesis: Converting light energy into chemical energy. Photoautotrophs capture sunlight and convert it to chemical energy
 Photosynthesis: Converting light energy into chemical energy Photosynthesis 6 + 12H 2 O + light energy Summary Formula: C 6 H 12 O 6 + 6O 2 + 6H 2 O 6 + 6H 2 O C 6 H 12 O 6 + 6O 2 Photosythesis provides
Photosynthesis: Converting light energy into chemical energy Photosynthesis 6 + 12H 2 O + light energy Summary Formula: C 6 H 12 O 6 + 6O 2 + 6H 2 O 6 + 6H 2 O C 6 H 12 O 6 + 6O 2 Photosythesis provides
Photosynthetic Solar constant
 hotosynthetic Solar constant D. C. Agrawal Department of Farm Engineering, Banaras Hindu University, Varanasi 100, India. E-mail: dca_bhu@yahoo.com (Received 3 November 009; accepted 11 January 010) Abstract
hotosynthetic Solar constant D. C. Agrawal Department of Farm Engineering, Banaras Hindu University, Varanasi 100, India. E-mail: dca_bhu@yahoo.com (Received 3 November 009; accepted 11 January 010) Abstract
2. PHOTOSYNTHESIS. The general equation describing photosynthesis is light + 6 H 2 O + 6 CO 2 C 6 H 12 O 6 + 6 O 2
 2. PHOTOSYNTHESIS Photosynthesis is the process by which light energy is converted to chemical energy whereby carbon dioxide and water are converted into organic molecules. The process occurs in most algae,
2. PHOTOSYNTHESIS Photosynthesis is the process by which light energy is converted to chemical energy whereby carbon dioxide and water are converted into organic molecules. The process occurs in most algae,
Photosynthesis and Cellular Respiration. Stored Energy
 Photosynthesis and Cellular Respiration Stored Energy What is Photosynthesis? plants convert the energy of sunlight into the energy in the chemical bonds of carbohydrates sugars and starches. SUMMARY EQUATION:
Photosynthesis and Cellular Respiration Stored Energy What is Photosynthesis? plants convert the energy of sunlight into the energy in the chemical bonds of carbohydrates sugars and starches. SUMMARY EQUATION:
Photosynthesis Part I: Overview & The Light-Dependent Reactions
 Photosynthesis Part I: Overview & The Light-Dependent Reactions Photosynthesis: The BIG Picture Photosynthesis is the process by which PHOTOAUTOTROPHS convert the energy in SUNLIGHT into the energy stored
Photosynthesis Part I: Overview & The Light-Dependent Reactions Photosynthesis: The BIG Picture Photosynthesis is the process by which PHOTOAUTOTROPHS convert the energy in SUNLIGHT into the energy stored
Question. Which of the following are necessary in order for photosynthesis to occur? A. water B. light energy C. carbon dioxide D.
 Photosynthesis is the process through which plants convert light energy to chemical energy in order to produce food The energy involved in photosynthesis is eventually stored in the chemical bonds of molecules
Photosynthesis is the process through which plants convert light energy to chemical energy in order to produce food The energy involved in photosynthesis is eventually stored in the chemical bonds of molecules
Photosynthesis 6CO 2 + 6H 2 O C 6 H 12 O 6 + 6O 2. An anabolic, endergonic, carbon dioxide (CO 2
 PHOTOSYNTHESIS Photosynthesis An anabolic, endergonic, carbon dioxide (CO 2 ) requiring process that uses light energy (photons) and water (H 2 O) to produce organic macromolecules (glucose). photons SUN
PHOTOSYNTHESIS Photosynthesis An anabolic, endergonic, carbon dioxide (CO 2 ) requiring process that uses light energy (photons) and water (H 2 O) to produce organic macromolecules (glucose). photons SUN
Review Questions Photosynthesis
 Review Questions Photosynthesis 1. Describe a metabolic pathway. In a factory, labor is divided into small individual jobs. A carmaker, for example, will have one worker install the front windshield, another
Review Questions Photosynthesis 1. Describe a metabolic pathway. In a factory, labor is divided into small individual jobs. A carmaker, for example, will have one worker install the front windshield, another
Photosynthesis: Harvesting Light Energy
 Photosynthesis: Harvesting Light Energy Importance of Photosynthesis A. Ultimate source of energy for all life on Earth 1. All producers are photosynthesizers 2. All consumers and decomposers are dependent
Photosynthesis: Harvesting Light Energy Importance of Photosynthesis A. Ultimate source of energy for all life on Earth 1. All producers are photosynthesizers 2. All consumers and decomposers are dependent
Photosynthesis. Name. Light reactions Calvin cycle Oxidation Reduction Electronegativity Photosystem Electron carrier NADP+ Concentration gradient
 Vocabulary Terms Photoautotroph Chemoautotroph Electromagnetic spectrum Wavelength Chloroplast Thylakoid Stroma Chlorophyll Absorption spectrum Photosynthesis Light reactions Calvin cycle Oxidation Reduction
Vocabulary Terms Photoautotroph Chemoautotroph Electromagnetic spectrum Wavelength Chloroplast Thylakoid Stroma Chlorophyll Absorption spectrum Photosynthesis Light reactions Calvin cycle Oxidation Reduction
Photosynthesis Chapter 8 E N E R G Y T O M A K E F O O D?
 Photosynthesis Chapter 8 H O W D O E S T H E P L A N T U S E T H E S U N S E N E R G Y T O M A K E F O O D? http://www.youtube.com/watch?v=pe82qtkssh4 Autotroph vs. Heterotroph Autotrophs/Producers-organisms
Photosynthesis Chapter 8 H O W D O E S T H E P L A N T U S E T H E S U N S E N E R G Y T O M A K E F O O D? http://www.youtube.com/watch?v=pe82qtkssh4 Autotroph vs. Heterotroph Autotrophs/Producers-organisms
Electron Transport Generates a Proton Gradient Across the Membrane
 Electron Transport Generates a Proton Gradient Across the Membrane Each of respiratory enzyme complexes couples the energy released by electron transfer across it to an uptake of protons from water in
Electron Transport Generates a Proton Gradient Across the Membrane Each of respiratory enzyme complexes couples the energy released by electron transfer across it to an uptake of protons from water in
4.1 Chemical Energy and ATP. KEY CONCEPT All cells need chemical energy.
 4.1 Chemical Energy and ATP KEY CONCEPT All cells need chemical energy. 4.1 Chemical Energy and ATP Molecules in food store chemical energy in their bonds. Starch molecule Glucose molecule The chemical
4.1 Chemical Energy and ATP KEY CONCEPT All cells need chemical energy. 4.1 Chemical Energy and ATP Molecules in food store chemical energy in their bonds. Starch molecule Glucose molecule The chemical
Equation for Photosynthesis
 Photosynthesis Definition The process by which cells harvest light energy to make sugars (glucose). -Sugar is used to power the process of cellular respiration, which produces the ATP that cells utilize
Photosynthesis Definition The process by which cells harvest light energy to make sugars (glucose). -Sugar is used to power the process of cellular respiration, which produces the ATP that cells utilize
> C 6 H 12 O 6 + 6O 2
 Photosynthesis- is the process that converts light energy into chemical energy. This chemical energy is usually a carbohydrate. Only photoautrotrops can do photosynthesis. Heterotrophs must obtain their
Photosynthesis- is the process that converts light energy into chemical energy. This chemical energy is usually a carbohydrate. Only photoautrotrops can do photosynthesis. Heterotrophs must obtain their
b. What is/are the overall function(s) of photosystem II?
 Use your model and the information in Chapter 10 of Biology, 7th edition, to answer the questions. 1. The various reactions in photosynthesis are spatially segregated from each other within the chloroplast.
Use your model and the information in Chapter 10 of Biology, 7th edition, to answer the questions. 1. The various reactions in photosynthesis are spatially segregated from each other within the chloroplast.
1. f. Students know usable energy is captured from sunlight by chloroplasts and is stored through the synthesis of sugar from carbon dioxide.
 1. The fundamental life processes of plants and animals depend on a variety of chemical reactions that occur in specialized areas of the organism s cells. As a basis for understanding this concept: 1.
1. The fundamental life processes of plants and animals depend on a variety of chemical reactions that occur in specialized areas of the organism s cells. As a basis for understanding this concept: 1.
Plants, like all other living organisms have basic needs: a source of nutrition (food),
 LEARNING FROM LEAVES: A LOOK AT LEAF SIZE Grades 3 6 I. Introduction Plants, like all other living organisms have basic needs: a source of nutrition (food), water, space in which to live, air, and optimal
LEARNING FROM LEAVES: A LOOK AT LEAF SIZE Grades 3 6 I. Introduction Plants, like all other living organisms have basic needs: a source of nutrition (food), water, space in which to live, air, and optimal
Biology. Slide 1of 51. End Show. Copyright Pearson Prentice Hall
 Biology 1of 51 8-3 The Reactions of Photosynthesis 2of 51 Inside a Chloroplast Inside a Chloroplast In plants, photosynthesis takes place inside chloroplasts. Plant Chloroplast Plant cells 3of 51 Inside
Biology 1of 51 8-3 The Reactions of Photosynthesis 2of 51 Inside a Chloroplast Inside a Chloroplast In plants, photosynthesis takes place inside chloroplasts. Plant Chloroplast Plant cells 3of 51 Inside
Anatomy and Physiology of Leaves
 I. Leaf Structure and Anatomy Anatomy and Physiology of Leaves A. Structural Features of the Leaf Question: How do plants respire? Plants must take in CO 2 from the atmosphere in order to photosynthesize.
I. Leaf Structure and Anatomy Anatomy and Physiology of Leaves A. Structural Features of the Leaf Question: How do plants respire? Plants must take in CO 2 from the atmosphere in order to photosynthesize.
Introduction: Growth analysis and crop dry matter accumulation
 PBIO*3110 Crop Physiology Lecture #2 Fall Semester 2008 Lecture Notes for Tuesday 9 September How is plant productivity measured? Introduction: Growth analysis and crop dry matter accumulation Learning
PBIO*3110 Crop Physiology Lecture #2 Fall Semester 2008 Lecture Notes for Tuesday 9 September How is plant productivity measured? Introduction: Growth analysis and crop dry matter accumulation Learning
CHAPTER 6: PHOTOSYNTHESIS CAPTURING & CONVERTING ENERGY
 CHAPTER 6: PHOTOSYNTHESIS CAPTURING & CONVERTING ENERGY 2 PROCESSES OF PHOTOSYNTHESIS Photosynthesis is actually 2 processes: light reactions - convert solar energy (sunlight) to chemical energy (ATP &
CHAPTER 6: PHOTOSYNTHESIS CAPTURING & CONVERTING ENERGY 2 PROCESSES OF PHOTOSYNTHESIS Photosynthesis is actually 2 processes: light reactions - convert solar energy (sunlight) to chemical energy (ATP &
A. Incorrect! No, while this statement is correct, it is not the best answer to the question.
 Biochemistry - Problem Drill 18: Photosynthesis No. 1 of 10 1. What is photosynthesis? Select the best answer. (A) Photosynthesis happens in the chloroplasts. (B) Light absorption by chlorophyll induces
Biochemistry - Problem Drill 18: Photosynthesis No. 1 of 10 1. What is photosynthesis? Select the best answer. (A) Photosynthesis happens in the chloroplasts. (B) Light absorption by chlorophyll induces
Chloroplasts and Mitochondria
 Name: KEY Period: Chloroplasts and Mitochondria Plant cells and some Algae contain an organelle called the chloroplast. The chloroplast allows plants to harvest energy from sunlight to carry on a process
Name: KEY Period: Chloroplasts and Mitochondria Plant cells and some Algae contain an organelle called the chloroplast. The chloroplast allows plants to harvest energy from sunlight to carry on a process
Dynamics of Typha latifolia population in a free water surface flow constructed wetland in Estonia
 TÍTULO DEL TRABAJO Dynamics of Typha latifolia population in a free water surface flow constructed wetland in Estonia NOMBRE DE AUTORES Martin Maddison* and Ülo Mander NOMBRE Y DIRECCIÓN DE LAS INSTITUCIONES
TÍTULO DEL TRABAJO Dynamics of Typha latifolia population in a free water surface flow constructed wetland in Estonia NOMBRE DE AUTORES Martin Maddison* and Ülo Mander NOMBRE Y DIRECCIÓN DE LAS INSTITUCIONES
 Quantum Photosynthesis N n n n 1 Exciton delocalization and energy flow in the Fenna-Matthews-Olson (FMO) complex Quantum Photosynthesis Even so, it's a source of recent excitement amongst physicists,
Quantum Photosynthesis N n n n 1 Exciton delocalization and energy flow in the Fenna-Matthews-Olson (FMO) complex Quantum Photosynthesis Even so, it's a source of recent excitement amongst physicists,
Elite Scientific Instruments LI-6400XTR Portable Photosynthesis System Training Courses
 Elite Scientific Instruments LI-6400XTR Portable Photosynthesis System Training Courses ESI offers LI-6400 and LI-6400XTR training at its Head Quarters Office or any location convenient to you. You will
Elite Scientific Instruments LI-6400XTR Portable Photosynthesis System Training Courses ESI offers LI-6400 and LI-6400XTR training at its Head Quarters Office or any location convenient to you. You will
CHAPTER 9 - LEAVES AND PLANTS... 1 9.1 INTRODUCTION...2 9.2 CARBON ASSIMILATION... 2 9.2.1 Light reactions... 3 9.2.2 Dark reactions... 5 9.2.
 Chapter 9 - Leaves and Plants CHAPTER 9 - LEAVES AND PLANTS... 1 9.1 INTRODUCTION...2 9.2 CARBON ASSIMILATION... 2 9.2.1 Light reactions... 3 9.2.2 Dark reactions... 5 9.2.3 Stomata... 7 9.2.4 Net photosynthesis...
Chapter 9 - Leaves and Plants CHAPTER 9 - LEAVES AND PLANTS... 1 9.1 INTRODUCTION...2 9.2 CARBON ASSIMILATION... 2 9.2.1 Light reactions... 3 9.2.2 Dark reactions... 5 9.2.3 Stomata... 7 9.2.4 Net photosynthesis...
CSS 560 Principles of Ecology for Environmental Educators
 CSS 560 Principles of Ecology for Environmental Educators Journaling task (15:00 min/each) Draw a diagram that shows the major components (boxes) and interactions (arrows) of a terrestrial ecosystem Conceptual
CSS 560 Principles of Ecology for Environmental Educators Journaling task (15:00 min/each) Draw a diagram that shows the major components (boxes) and interactions (arrows) of a terrestrial ecosystem Conceptual
MAIN SOURCE OF ENERGY FOR LIFE ON EARTH? THE SUN!!
 MAIN SOURCE OF ENERGY FOR LIFE ON EARTH? THE SUN!! THE BASICS OF PHOTOSYNTHESIS Almost all plants are photosynthetic autotrophs, as are some bacteria and protists Autotrophs generate their own organic
MAIN SOURCE OF ENERGY FOR LIFE ON EARTH? THE SUN!! THE BASICS OF PHOTOSYNTHESIS Almost all plants are photosynthetic autotrophs, as are some bacteria and protists Autotrophs generate their own organic
AP BIOLOGY 2007 SCORING GUIDELINES
 AP BIOLOGY 2007 SCORING GUIDELINES Question 3 Compared with other terrestrial biomes, deserts have extremely low productivity. (a) Discuss how temperature, soil composition, and annual precipitation limit
AP BIOLOGY 2007 SCORING GUIDELINES Question 3 Compared with other terrestrial biomes, deserts have extremely low productivity. (a) Discuss how temperature, soil composition, and annual precipitation limit
2. 1. What are the three parts of an ATP molecule? (100 points)
 Photosynthesis Date Created: 12/8/14, 11:22:50 AM Questions: 34 Date Modified: 12/17/14, 8:27:08 AM 1. ATP & Photosynthesis Review Game 30 Multiple Choice Questions Final Question Correct Answers = +$100
Photosynthesis Date Created: 12/8/14, 11:22:50 AM Questions: 34 Date Modified: 12/17/14, 8:27:08 AM 1. ATP & Photosynthesis Review Game 30 Multiple Choice Questions Final Question Correct Answers = +$100
ecture 16 Oct 7, 2005
 Lecture utline ecture 16 ct 7, 005 hotosynthesis 1 I. Reactions 1. Importance of Photosynthesis to all life on earth - primary producer, generates oxygen, ancient. What needs to be accomplished in photosynthesis
Lecture utline ecture 16 ct 7, 005 hotosynthesis 1 I. Reactions 1. Importance of Photosynthesis to all life on earth - primary producer, generates oxygen, ancient. What needs to be accomplished in photosynthesis
8-3 The Reactions of Photosynthesis Slide 1 of 51
 8-3 The of Photosynthesis 1 of 51 Inside a Chloroplast Inside a Chloroplast In plants, photosynthesis takes place inside chloroplasts. Plant Chloroplast Plant cells 2 of 51 Inside a Chloroplast Chloroplasts
8-3 The of Photosynthesis 1 of 51 Inside a Chloroplast Inside a Chloroplast In plants, photosynthesis takes place inside chloroplasts. Plant Chloroplast Plant cells 2 of 51 Inside a Chloroplast Chloroplasts
-Loss of energy -Loss of hydrogen from carbons. -Gain of energy -Gain of hydrogen to carbons
 Cellular Respiration- Equation C6H12O6 + 6O2 6CO2 +6H20 and energy -The energy is released from the chemical bonds in the complex organic molecules -The catabolic process of releasing energy from food
Cellular Respiration- Equation C6H12O6 + 6O2 6CO2 +6H20 and energy -The energy is released from the chemical bonds in the complex organic molecules -The catabolic process of releasing energy from food
Name Class Date. Figure 8-1
 Chapter 8 Photosynthesis Chapter Test A Multiple Choice Write the letter that best answers the question or completes the statement on the line provided. 1. Which of the following is an autotroph? a. mushroom
Chapter 8 Photosynthesis Chapter Test A Multiple Choice Write the letter that best answers the question or completes the statement on the line provided. 1. Which of the following is an autotroph? a. mushroom
Bioenergetics Module A Anchor 3
 Bioenergetics Module A Anchor 3 Key Concepts: - ATP can easily release and store energy by breaking and re-forming the bonds between its phosphate groups. This characteristic of ATP makes it exceptionally
Bioenergetics Module A Anchor 3 Key Concepts: - ATP can easily release and store energy by breaking and re-forming the bonds between its phosphate groups. This characteristic of ATP makes it exceptionally
ECOSISTEMAS. Luis A. Rivera M.A. Biological Education, M.A. Forestry Tropical Vegetation Specialist
 ECOSISTEMAS Luis A. Rivera M.A. Biological Education, M.A. Forestry Tropical Vegetation Specialist Ecosistemas en el Bosque Nacional El Yunque 1. Mature Tabanuco montane rain forest Ecosistemas Preliminarmente
ECOSISTEMAS Luis A. Rivera M.A. Biological Education, M.A. Forestry Tropical Vegetation Specialist Ecosistemas en el Bosque Nacional El Yunque 1. Mature Tabanuco montane rain forest Ecosistemas Preliminarmente
Photosynthesis Practice. 2. Chlorophyll a and b absorb _B -_V and _R wavelengths of light best.
 Photosynthesis Practice Fill in the blanks. Name Date Period 1. Molecules that collect light energy are called _P. 2. Chlorophyll a and b absorb _B -_V and _R wavelengths of light best. 3. _C is the main
Photosynthesis Practice Fill in the blanks. Name Date Period 1. Molecules that collect light energy are called _P. 2. Chlorophyll a and b absorb _B -_V and _R wavelengths of light best. 3. _C is the main
Photosynthesis-Review. Pigments. Chloroplasts. Chloroplasts 5. Pigments are located in the thylakoid membranes. An Overview of Photosynthesis
 An Overview of Photosynthesis Photosynthesis-Review 1. Photosynthesis uses the energy of sunlight to convert water and carbon dioxide into high-energy sugars and oxygen. 6 CO 2 + 6 H 2 O C 6 H 12 O 6 +
An Overview of Photosynthesis Photosynthesis-Review 1. Photosynthesis uses the energy of sunlight to convert water and carbon dioxide into high-energy sugars and oxygen. 6 CO 2 + 6 H 2 O C 6 H 12 O 6 +
Plant adaptations, Chlorophyll & Photosynthesis. Surviving in hot environments and making the most of light
 Plant adaptations, Chlorophyll & Photosynthesis Surviving in hot environments and making the most of light Photosynthesis is the basis of life of earth. It provides the energy that drives virtually all
Plant adaptations, Chlorophyll & Photosynthesis Surviving in hot environments and making the most of light Photosynthesis is the basis of life of earth. It provides the energy that drives virtually all
Green pigment that absorbs solar energy and is important in photosynthesis
 PHOTOSYNTHESIS REVIEW SHEET FOR TEST Part A: Match the terms below with the correct description Chlorophyll Chloroplast Electromagnetic spectrum Electron transport chain Grana Light-dependant reactions
PHOTOSYNTHESIS REVIEW SHEET FOR TEST Part A: Match the terms below with the correct description Chlorophyll Chloroplast Electromagnetic spectrum Electron transport chain Grana Light-dependant reactions
A B C D. Name Class Date
 Chapter 8 Photosynthesis Chapter Test A Multiple Choice Write the letter that best answers the question or completes the statement on the line provided. 1. Which of the following is an autotroph? a. mushroom
Chapter 8 Photosynthesis Chapter Test A Multiple Choice Write the letter that best answers the question or completes the statement on the line provided. 1. Which of the following is an autotroph? a. mushroom
AP Bio Photosynthesis & Respiration
 AP Bio Photosynthesis & Respiration Multiple Choice Identify the letter of the choice that best completes the statement or answers the question. 1. What is the term used for the metabolic pathway in which
AP Bio Photosynthesis & Respiration Multiple Choice Identify the letter of the choice that best completes the statement or answers the question. 1. What is the term used for the metabolic pathway in which
Trace Gas Exchange Measurements with Standard Infrared Analyzers
 Practical Environmental Measurement Methods Trace Gas Exchange Measurements with Standard Infrared Analyzers Last change of document: February 23, 2007 Supervisor: Charles Robert Room no: S 4381 ph: 4352
Practical Environmental Measurement Methods Trace Gas Exchange Measurements with Standard Infrared Analyzers Last change of document: February 23, 2007 Supervisor: Charles Robert Room no: S 4381 ph: 4352
COTTON WATER RELATIONS
 COTTON WATER RELATIONS Dan R. Krieg 1 INTRODUCTION Water is the most abundant substance on the Earth s surface and yet is the most limiting to maximum productivity of nearly all crop plants. Land plants,
COTTON WATER RELATIONS Dan R. Krieg 1 INTRODUCTION Water is the most abundant substance on the Earth s surface and yet is the most limiting to maximum productivity of nearly all crop plants. Land plants,
Water Relations, Root Growth Potential and Plant Survival of Cold Stored Pinus radiata D. Don Seedlings
 Phyton (Austria) Special issue: "Root-soil interactions" Vol. 40 Fasc. 4 (143)-(148) 25.7.2000 Water Relations, Root Growth Potential and Plant Survival of Cold Stored Pinus radiata D. Don Seedlings By
Phyton (Austria) Special issue: "Root-soil interactions" Vol. 40 Fasc. 4 (143)-(148) 25.7.2000 Water Relations, Root Growth Potential and Plant Survival of Cold Stored Pinus radiata D. Don Seedlings By
Flowers; Seeds enclosed in fruit
 Name Class Date Chapter 22 Plant Diversity Section Review 22-1 Reviewing Key Concepts Short Answer On the lines provided, answer the following questions. 1. Describe the main characteristics of plants.
Name Class Date Chapter 22 Plant Diversity Section Review 22-1 Reviewing Key Concepts Short Answer On the lines provided, answer the following questions. 1. Describe the main characteristics of plants.
While reading these chapters, constantly ask yourself, How is this information helping me to understand how cells get energy from food?
 Biology 160 Reading Guide 07: Photosynthesis NAME: This is DUE: Come prepared to share your findings with your group. ** Fill this reading guide out as you are reading the chapters. This will help you
Biology 160 Reading Guide 07: Photosynthesis NAME: This is DUE: Come prepared to share your findings with your group. ** Fill this reading guide out as you are reading the chapters. This will help you
Unit 5 Photosynthesis and Cellular Respiration
 Unit 5 Photosynthesis and Cellular Respiration Advanced Concepts What is the abbreviated name of this molecule? What is its purpose? What are the three parts of this molecule? Label each part with the
Unit 5 Photosynthesis and Cellular Respiration Advanced Concepts What is the abbreviated name of this molecule? What is its purpose? What are the three parts of this molecule? Label each part with the
Instrumentation for the Plant & Soil Sciences Short Form Catalog
 Instrumentation for the Plant & Soil Sciences Short Form Catalog www.ppsystems.com Trusted and tested technology CIRAS-2 Portable Photosynthesis System PLC6 (U) Automatic Universal Leaf Cuvette For High
Instrumentation for the Plant & Soil Sciences Short Form Catalog www.ppsystems.com Trusted and tested technology CIRAS-2 Portable Photosynthesis System PLC6 (U) Automatic Universal Leaf Cuvette For High
Hands on an infrared gas analyzer (IRGA) to measure photosynthesis
 University of Toledo - Dept of Envir. Sci., EEES 6660/8960: Biophysical Processes of Ecosystems, Spring 2010 Hands on an infrared gas analyzer (IRGA) to measure photosynthesis Burkhard Wilske for Jiquan
University of Toledo - Dept of Envir. Sci., EEES 6660/8960: Biophysical Processes of Ecosystems, Spring 2010 Hands on an infrared gas analyzer (IRGA) to measure photosynthesis Burkhard Wilske for Jiquan
Plants, like all living organisms have basic needs: a source of nutrition (food), water,
 WHAT PLANTS NEED IN ORDER TO SURVIVE AND GROW: LIGHT Grades 3 6 I. Introduction Plants, like all living organisms have basic needs: a source of nutrition (food), water, space in which to live, air, and
WHAT PLANTS NEED IN ORDER TO SURVIVE AND GROW: LIGHT Grades 3 6 I. Introduction Plants, like all living organisms have basic needs: a source of nutrition (food), water, space in which to live, air, and
Name Date Period PHOTOSYNTHESIS HW REVIEW ENERGY AND LIFE
 1 Name Date Period PHOTOSYNTHESIS HW REVIEW ENERGY AND LIFE MULTIPLE CHOICE: CIRCLE ALL THE ANSWERS THAT ARE TRUE. THERE MAY BE MORE THAN ONE CORRECT ANSWER! 1. Which molecule stores more than 90 times
1 Name Date Period PHOTOSYNTHESIS HW REVIEW ENERGY AND LIFE MULTIPLE CHOICE: CIRCLE ALL THE ANSWERS THAT ARE TRUE. THERE MAY BE MORE THAN ONE CORRECT ANSWER! 1. Which molecule stores more than 90 times
Water movement in the xylem Water moves from roots to leaves through the xylem. But how? Hypotheses: 1. Capillary action - water will move upward in
 Transport in Plants Two Transport Processes Occur in Plants 1. Carbohydrates carried from leaves (or storage organs) to where they are needed (from sources to sinks) 2. Water transported from roots to
Transport in Plants Two Transport Processes Occur in Plants 1. Carbohydrates carried from leaves (or storage organs) to where they are needed (from sources to sinks) 2. Water transported from roots to
APh/BE161: Physical Biology of the Cell Winter 2009 Recap on Photosynthesis Rob Phillips
 APh/BE161: Physical Biology of the Cell Winter 2009 Recap on Photosynthesis Rob Phillips Big picture: why are we doing this? A) photosynthesis will explain shortly, b) more generally, interaction of light
APh/BE161: Physical Biology of the Cell Winter 2009 Recap on Photosynthesis Rob Phillips Big picture: why are we doing this? A) photosynthesis will explain shortly, b) more generally, interaction of light
Seasonal responses of photosynthesis and growth of a bioenergy crop (Phalaris arundinacea L.) to climate change under varying water regimes
 Dissertationes Forestales 127 Seasonal responses of photosynthesis and growth of a bioenergy crop (Phalaris arundinacea L.) to climate change under varying water regimes Xiao Zhou Faculty of Science and
Dissertationes Forestales 127 Seasonal responses of photosynthesis and growth of a bioenergy crop (Phalaris arundinacea L.) to climate change under varying water regimes Xiao Zhou Faculty of Science and
Overview. Suggested Lesson Please see the Greenlinks Module description.
 Overview Plants interact with their environment in many ways that we cannot see. Children often enjoy learning about these hidden secrets of plant life. In this lesson, children will learn about role of
Overview Plants interact with their environment in many ways that we cannot see. Children often enjoy learning about these hidden secrets of plant life. In this lesson, children will learn about role of
Impressions of a Stoma
 Huntington Library, Art Collections, and Botanical Gardens Impressions of a Stoma Overview Students use two different methods to view stomata on the underside of leaves. Introduction Plants exchange the
Huntington Library, Art Collections, and Botanical Gardens Impressions of a Stoma Overview Students use two different methods to view stomata on the underside of leaves. Introduction Plants exchange the
Schema XML_PGE.xsd. element GrupoInformes. attribute GrupoInformes/@version. XML_PGE.xsd unqualified qualified http://sgpfc.igae.minhap.
 Schema XML_PGE.xsd schema location: attribute form default: element form default: targetnamespace: XML_PGE.xsd unqualified qualified http://sgpfc.igae.minhap.es/xmlpge element GrupoInformes children Informe
Schema XML_PGE.xsd schema location: attribute form default: element form default: targetnamespace: XML_PGE.xsd unqualified qualified http://sgpfc.igae.minhap.es/xmlpge element GrupoInformes children Informe
An approach on the climate effects due to biomass burning aerosols
 ÓPTICA PURA Y APLICADA Vol. 37, núm. 3-2004 An approach on the climate effects due to biomass burning aerosols Un método sobre los efectos climáticos de los aerosoles por quema de biomasa Martín José Montero-Martínez
ÓPTICA PURA Y APLICADA Vol. 37, núm. 3-2004 An approach on the climate effects due to biomass burning aerosols Un método sobre los efectos climáticos de los aerosoles por quema de biomasa Martín José Montero-Martínez
LIST OF RECENT PLANTSCREEN TM INSTALLATIONS
 LIST OF RECENT PLANTSCREEN TM INSTALLATIONS The below-listed overview is example of recent PlantScreen TM high-throughput automated Systems manufactured and installed by the Photon Systems Instruments,
LIST OF RECENT PLANTSCREEN TM INSTALLATIONS The below-listed overview is example of recent PlantScreen TM high-throughput automated Systems manufactured and installed by the Photon Systems Instruments,
Tropical rainforests grow in areas of high rainfall, they are found between the Tropic of Cancer and the Tropic of Capricorn.
 Tracking KS3 the Tropics In our Tropical Rainforest Biome you will discover how plants, animals and people of the Tropical Rainforest fit together. Let s Start! Look point 1 (Map on Left as you enter the
Tracking KS3 the Tropics In our Tropical Rainforest Biome you will discover how plants, animals and people of the Tropical Rainforest fit together. Let s Start! Look point 1 (Map on Left as you enter the
DIFFERENTIATIONS OF OBJECTS IN DIFFUSE DATABASES DIFERENCIACIONES DE OBJETOS EN BASES DE DATOS DIFUSAS
 Recibido: 01 de agosto de 2012 Aceptado: 23 de octubre de 2012 DIFFERENTIATIONS OF OBJECTS IN DIFFUSE DATABASES DIFERENCIACIONES DE OBJETOS EN BASES DE DATOS DIFUSAS PhD. Amaury Caballero*; PhD. Gabriel
Recibido: 01 de agosto de 2012 Aceptado: 23 de octubre de 2012 DIFFERENTIATIONS OF OBJECTS IN DIFFUSE DATABASES DIFERENCIACIONES DE OBJETOS EN BASES DE DATOS DIFUSAS PhD. Amaury Caballero*; PhD. Gabriel
Metabolism Poster Questions
 Metabolism Poster Questions Answer the following questions concerning respiration. 1. Consider the mitochondrial electron transport chain. a. How many hydrogen ions can be pumped for every NADH? b. How
Metabolism Poster Questions Answer the following questions concerning respiration. 1. Consider the mitochondrial electron transport chain. a. How many hydrogen ions can be pumped for every NADH? b. How
Ch. 4 ATP & Photosynthesis
 Name: Biology G Vocabulary Section 4.1 Ch. 4 ATP & Photosynthesis Period: ADP Adenosine Diphosphate ATP Adenosine Triphosphate Chemosynthesis Vocabulary Section 4.2 Photosynthesis Chlorophyll Thylakoid
Name: Biology G Vocabulary Section 4.1 Ch. 4 ATP & Photosynthesis Period: ADP Adenosine Diphosphate ATP Adenosine Triphosphate Chemosynthesis Vocabulary Section 4.2 Photosynthesis Chlorophyll Thylakoid
Name Date Class. energy phosphate adenine charged ATP chemical bonds work ribose
 Energy in a Cell Reinforcement and Study Guide Section.1 The Need for Energy In your textbook, read about cell energy. Use each of the terms below just once to complete the passage. energy phosphate adenine
Energy in a Cell Reinforcement and Study Guide Section.1 The Need for Energy In your textbook, read about cell energy. Use each of the terms below just once to complete the passage. energy phosphate adenine
(e) i. 22. (a) ii (b) iv (c) v (d) iii
 Chapter 5 Review, pages 246 251 Knowledge 1. a 2. b 3. b 4. d 5. d 6. b 7. b 8. b 9. b 10. a 11. True 12. False. Chlorophyll a, when excited, becomes oxidized as it passes an electron to a primary electron
Chapter 5 Review, pages 246 251 Knowledge 1. a 2. b 3. b 4. d 5. d 6. b 7. b 8. b 9. b 10. a 11. True 12. False. Chlorophyll a, when excited, becomes oxidized as it passes an electron to a primary electron
Chapter 10: Photosynthesis
 Name Period Chapter 10: Photosynthesis This chapter is as challenging as the one you just finished on cellular respiration. However, conceptually it will be a little easier because the concepts learned
Name Period Chapter 10: Photosynthesis This chapter is as challenging as the one you just finished on cellular respiration. However, conceptually it will be a little easier because the concepts learned
PLANT PHYSIOLOGY. Az Agrármérnöki MSc szak tananyagfejlesztése TÁMOP-4.1.2-08/1/A-2009-0010
 PLANT PHYSIOLOGY Az Agrármérnöki MSc szak tananyagfejlesztése TÁMOP-4.1.2-08/1/A-2009-0010 The light reactions of the photosynthesis Photosynthesis inhibiting herbicides Overview 1. Photosynthesis, general
PLANT PHYSIOLOGY Az Agrármérnöki MSc szak tananyagfejlesztése TÁMOP-4.1.2-08/1/A-2009-0010 The light reactions of the photosynthesis Photosynthesis inhibiting herbicides Overview 1. Photosynthesis, general
Photosynthesis Reactions. Photosynthesis Reactions
 Photosynthesis Reactions Photosynthesis occurs in two stages linked by ATP and NADPH NADPH is similar to NADH seen in mitochondria; it is an electron/hydrogen carrier The complete process of photosynthesis
Photosynthesis Reactions Photosynthesis occurs in two stages linked by ATP and NADPH NADPH is similar to NADH seen in mitochondria; it is an electron/hydrogen carrier The complete process of photosynthesis
8.3 The Process of Photosynthesis
 8.3 The Process of Photosynthesis Lesson Objectives Describe what happens during the light-dependent reactions. Describe what happens during the light-independent reactions. Identify factors that affect
8.3 The Process of Photosynthesis Lesson Objectives Describe what happens during the light-dependent reactions. Describe what happens during the light-independent reactions. Identify factors that affect
Biology I. Chapter 8/9
 Biology I Chapter 8/9 NOTEBOOK #1 Interest Grabber Suppose you earned extra money by having a part-time job. At first, you might be tempted to spend all of the money, but then you decide to open a bank
Biology I Chapter 8/9 NOTEBOOK #1 Interest Grabber Suppose you earned extra money by having a part-time job. At first, you might be tempted to spend all of the money, but then you decide to open a bank
R.J. Strasser, A. Srivastava and M. Tsimilli-Michael
 Chapter The fluorescence 25 transient as a tool to characterize and screen photosynthetic samples 445 The fluorescence transient as a tool to characterize and screen photosynthetic samples R.J. Strasser,
Chapter The fluorescence 25 transient as a tool to characterize and screen photosynthetic samples 445 The fluorescence transient as a tool to characterize and screen photosynthetic samples R.J. Strasser,
Reactive oxygen species in leaves and how to catch these
 Reactive oxygen species in leaves and how to catch these Éva Hideg Molecular Stress- & Photobiology Group, BRC Szeged EPPN Summer School 2013 Outline How to prove that ROS are involved? The central role
Reactive oxygen species in leaves and how to catch these Éva Hideg Molecular Stress- & Photobiology Group, BRC Szeged EPPN Summer School 2013 Outline How to prove that ROS are involved? The central role
Transpiration. C should equal D.BUT SOMETIMES. 1. Loss in mass is greater than volume of water added.
 Transpiration Transpiration is the loss of water by evaporation from the leaves through the stomata. The source of water for the plants is soil water. It is taken up by root hair cells by osmosis. Once
Transpiration Transpiration is the loss of water by evaporation from the leaves through the stomata. The source of water for the plants is soil water. It is taken up by root hair cells by osmosis. Once
SOME Important Points About Cellular Energetics by Dr. Ty C.M. Hoffman
 SOME Important Points About Cellular Energetics by Dr. Ty C.M. Hoffman An Introduction to Metabolism Most biochemical processes occur as biochemical pathways, each individual reaction of which is catalyzed
SOME Important Points About Cellular Energetics by Dr. Ty C.M. Hoffman An Introduction to Metabolism Most biochemical processes occur as biochemical pathways, each individual reaction of which is catalyzed
instruments for plant physiology plant & soil sciences scientific and meteorological instruments ingenieursbureau wittich & visser
 instruments for plant physiology plant & soil sciences ingenieursbureau wittich & visser scientific and meteorological instruments LEAF GAS EXCHANGE FOR RESEARCH The CIRAS-2 is considered by many scientists
instruments for plant physiology plant & soil sciences ingenieursbureau wittich & visser scientific and meteorological instruments LEAF GAS EXCHANGE FOR RESEARCH The CIRAS-2 is considered by many scientists
TREE STRUCTURE AND BIOLOGY
 TREE STRUCTURE AND BIOLOGY Introduction. Sarasota County lies along a transient tension zone line. In this region, climate cycles cause periods of warmer weather, when normally tropical trees find their
TREE STRUCTURE AND BIOLOGY Introduction. Sarasota County lies along a transient tension zone line. In this region, climate cycles cause periods of warmer weather, when normally tropical trees find their
Photosystems I and II
 Photosystems I and II March 17, 2003 Bryant Miles Within the thylakoid membranes of the chloroplast, are two photosystems. Photosystem I optimally absorbs photons of a wavelength of 700 nm. Photosystem
Photosystems I and II March 17, 2003 Bryant Miles Within the thylakoid membranes of the chloroplast, are two photosystems. Photosystem I optimally absorbs photons of a wavelength of 700 nm. Photosystem
ATP & Photosynthesis Honors Biology
 ATP & Photosynthesis Honors Biology ATP All cells need for life. Some things we use energy for are: Moving Thinking Sleeping Breathing Growing Reproducing ENERGY Labeled Sketch: The principal chemical
ATP & Photosynthesis Honors Biology ATP All cells need for life. Some things we use energy for are: Moving Thinking Sleeping Breathing Growing Reproducing ENERGY Labeled Sketch: The principal chemical
A level workbook. A2 level student guide. Brian Banks
 A level workbook Photosynthesis A2 level student guide Brian Banks A level guide. Photosynthesis 2 Using the workbook This workbook is designed to provide the student with notes, illustrations, questions
A level workbook Photosynthesis A2 level student guide Brian Banks A level guide. Photosynthesis 2 Using the workbook This workbook is designed to provide the student with notes, illustrations, questions
CIRAS-2 Portable Photosynthesis System For Research
 CIRAS-2 Portable Photosynthesis System For Research www.ppsystems.com CIRAS-2 Portable Photosynthesis System Trusted and tested technology Measurement Ranges CO 2 0-2,000 μmol mol -1 (optimal) CO 2 0-9,999
CIRAS-2 Portable Photosynthesis System For Research www.ppsystems.com CIRAS-2 Portable Photosynthesis System Trusted and tested technology Measurement Ranges CO 2 0-2,000 μmol mol -1 (optimal) CO 2 0-9,999
Studying Photosynthetic Electron Transport through the Hill Reaction
 Studying Photosynthetic Electron Transport through the Hill Reaction In this lab exercise you will learn techniques for studying electron transport. While our research system will be chloroplasts and photosynthetic
Studying Photosynthetic Electron Transport through the Hill Reaction In this lab exercise you will learn techniques for studying electron transport. While our research system will be chloroplasts and photosynthetic
VARIATION IN LIGHT INTENSITY AT DIFFERENT LATITUDES AND SEASONS, EFFECTS OF CLOUD COVER, AND THE AMOUNTS OF DIRECT AND DIFFUSED LIGHT
 VARIATION IN LIGHT INTENSITY AT DIFFERENT LATITUDES AND SEASONS, EFFECTS OF CLOUD COVER, AND THE AMOUNTS OF DIRECT AND DIFFUSED LIGHT Paul Burgess School of Applied Sciences, Cranfield University, Cranfield,
VARIATION IN LIGHT INTENSITY AT DIFFERENT LATITUDES AND SEASONS, EFFECTS OF CLOUD COVER, AND THE AMOUNTS OF DIRECT AND DIFFUSED LIGHT Paul Burgess School of Applied Sciences, Cranfield University, Cranfield,
AUTOTROPHES AND HETEROTROPHES
 WHAT IS CLIL??? Da alcuni anni nel Liceo Linguistico Da Vinci di Alba viene sperimentata la metodologia CLIL (Content and Language Integrated Learning), anticipando quanto previsto dalla recente riforma
WHAT IS CLIL??? Da alcuni anni nel Liceo Linguistico Da Vinci di Alba viene sperimentata la metodologia CLIL (Content and Language Integrated Learning), anticipando quanto previsto dalla recente riforma
REVIEW UNIT 10: ECOLOGY SAMPLE QUESTIONS
 Period Date REVIEW UNIT 10: ECOLOGY SAMPLE QUESTIONS A. Sample Multiple Choice Questions Complete the multiple choice questions to review this unit. 1. All of the following are density-dependent factors
Period Date REVIEW UNIT 10: ECOLOGY SAMPLE QUESTIONS A. Sample Multiple Choice Questions Complete the multiple choice questions to review this unit. 1. All of the following are density-dependent factors
Susana Sanduvete-Chaves, Salvador Chacón-Moscoso, Milagrosa Sánchez- Martín y José Antonio Pérez-Gil ( )
 ACCIÓN PSICOLÓGICA, junio 2014, vol. 10, n. o 2, 3-20. ISSN: 1578-908X 19 THE REVISED OSTERLIND INDEX. A COMPARATIVE ANALYSIS IN CONTENT VALIDITY STUDIES 1 EL ÍNDICE DE OSTERLIND REVISADO. UN ANÁLISIS
ACCIÓN PSICOLÓGICA, junio 2014, vol. 10, n. o 2, 3-20. ISSN: 1578-908X 19 THE REVISED OSTERLIND INDEX. A COMPARATIVE ANALYSIS IN CONTENT VALIDITY STUDIES 1 EL ÍNDICE DE OSTERLIND REVISADO. UN ANÁLISIS
