Thermohahe Convection with Two Stable Regimes of Flow. (Manuscript received January 21, 1961) Abstract
|
|
|
- Crystal Philippa Cooper
- 9 years ago
- Views:
Transcription
1 Thermohahe Convection with Two Stable Regimes of Flow By HENRY STOMMEL, Pierce Hd, Harvard University, Massachusetts (Manuscript received January 21, 1961) Abstract Free convection between two interconnected reservoirs, due to density differences maintained by heat and salt transfer to the reservoirs, is shown to occur sometimes in two different stable regimes, and may possibly be analogous to certain features of the oseanic circulation. The density of seawater is modified while at the surface by two distinct processes: heating and cooling, which change the temperature, and precipitation and evaporation, which change the salinity. n many important oceanic regions these two density modifying processes work contrarily to one another. For exam le, the water in and above the main thermoc P,,. e in all subtropical oceans is able to float on top of the denser deep water (we neglect here all consideration of the saltfountain effect) because its density is depressed through surface heating but the excessive evaporation in these same latitudes is able to increase the salinity of the surface waters to such an extent that in general their density contrast with deep waters is only one half of what it would be on the basis of temperature alone. n some large semienclosed seas (e.g. Mediterranean and Red Seas) subject to similar surface heating and evaporation, the salinity can have a predominant influence on the density field. By qualitative comparison of these cases we form an impression that the heat transfer mechanism has a more rapid effect on the density of a newly arriving parcel of water, but that in the long run, given sufficient time, evaporation can reverse the thermal influence. We do not know enough about the details of oceanic circulation to pursue these questions in their full complexity in nature but we can explore their implications in simple idealized systems. That is all we propose to do here. First, let us consider a very simple system: a vessel of water stirred so as to maintain uniform tem erature T and salinity S. (fig. ). The walls o P the vessel are made of a porous substance which permits transfer of heat and salt in a simple linear fashion: dt =c(t T) ds =d(s S) T and S are the temperature and salinity outside the wall and are regarded as fixed. The equations may be made nondimen 4orous wa1sl Fig.. The idealized experiment, consisting of a well stirred vessel of water with temperature T and salinity S (in general variable in time) separated by porous walls from and outside vessel whose temperature 7 and salinity S are maintained at constant values. Tellus X11 (1961), 2
2 THERMOHALNE CONVECTON WTH TWO STABLE REGMES OF FLOW 225 dx =d(1 x) The quantity 6 is considered to be a small quantity, that is we are most interested in situations where the salinity transfer coefficient $ d is less than the temperature transfer coefficient p t, or 6 <. No matter what the initial values a of x and say xo and yo at t = o the initial final equjbrium state as t + 00 is x =, y =, c the solutions of the above equations being, in general y = + (yo ) e? x = + (xo ) edr Moreover, the temperature approaches its as mptotic value more quickly than the salty. Now let us consider a simple form of equation of state e =eo ( at+bs) which, when expressed in terms of the nondimensional quantities x and y is e=eo t(+t) (r+wl where R = B S/aT, a measure of the ratio of the effect of salinity and temperature on the density in the final equilibrium state x =, y =. We are interested primarily in cases where R >, because this corresponds to the case where the density at x =, y = is greater than that at x = 0, y = 0. The time rate of change of density at any time is dq = e 0 T[ + y + Rd ( x)] f in the special case xo=y0=o, the quantity R6 <, but R> then in the beginning at t=o the density at first decreases, but eventually increases again until at t = m it is greater than at the beginning. The entire process can easily be visualized on an S,T diagram or in nondimensional terms, the x, y plane. n figure 2, the lines of constant density anomaly /(at) are drawn for the case R =z. The density anomaly is o at the initial condition x = 0, y = 0, and at the asymptotic limit t +m, Tellus X11 (1961). 2 X C Salinity Fig. 2. Nondimensional salinitytemperature diagram show lines of constant density (u is density anomaly) for the case R = 2. The points marked T = 0,, 2, 3, 5, 00 illustrate values of temperature and salinity at successive instants of time for 6 = /6. the density anomaly is +, therefore the fluid in the time dependent process in the case where 6 = s are plotted as points along a curve for increasing z. During the early stages the density decreases, until at Z= it begins to increase again. At about t=4 the density is back at its initial value and henceforth increases to its asymptotic limit as z+m. Another way of visualizing the different rates of the salt and temperature transfer processes in controlling the density is to consider a simple steady state process, in which water at T= 0, S = o flows into the vessel at rate q, and the mixture is withdrawn at the same rate. The arrangement for this experiment is shown in figure 3. The equations describing this state are dt = o= c (7 T) qt ds =o=d(ss)qs and reduced to nondimensional form as before ( +f )y= 0 6 (6+f )x = 0
3 226 HENRY STOMMEL inflow c outflow Fig. 3. The idealized steady state experiment. where f = q/c denotes a dimensionless flow rate. The equilibrium values of x, y which obtain in the vessel are Y = q The water which enters the vessel has zero density anomaly. When it leaves its density may be greater or less than that entering depending upon the flow ratef.. The points x, y corresponding to the salinity and temperature of the outflow lie along the curve: Y S(4 Y depicted in figure 4 for the value 6 = X The actual points x, y for certain choices of flow ratef. The shape of the curve depends only upon 6. The position of the points x, y on this curve depends only on f. f we now choose R = z as before we can draw the same lines of density anomaly as in figure 2. Hence the most dense outflow occurs or very small discharge rates f + 0, the outflowing water being denser than that flowing into the vessel. On the other hand, the position of points for whch /4 <: f sm indicates that in this range of discharge the outflow is less dense than the inflow. The purpose of Fig. 4. Values of nondimensional temperature y and salinity x for steady state experiment (fig. 3) for different rates of flow, f. exploring these extremely simple models has been to lay a groundwork for the remarks made in the introduction where it was stated that the very different density structure in the Atlantic and Mediterranean might be simply due to different rates of flow, but the models are obviously too idealized for direct application to natural conditions. They are, however, suitable to use as a basis for exploring further consequences of the density transforming processes : in particular the fact that the flow rates in free convective systems depend upon the density differences themselves. There is a kmd of feedback of the density difference produced upon the flow rate which produces it, that now must be introduced into the system in a simple way. The easiest way to accomplish ths is to introduce another stirred vessel surrounded by a resevoir at T, and S, as shown in figure 5. The two vessels are connected by a large overflow at the top so that free surface of water is level across the top of each vessel. Connecting them at the bottom is a capillary tube whose resistance k is such that the flow q in the tube is directed from the hgh pressure (hgh density) vessel toward the low pressure (low density) vessel by a simple linear law kq=e1e2 X Tellus X11 (1961). 2
4 THERMOHALNE CONVECTON WTH TWO STABLE REGMES OF FLOW 227 Vessel vessel 2 copillory c 9 Fig. 5. Two vesselexperiment, with rate of flow, q. through capillary determined by the density difference between the two vessels. The upper overflow is provided so that the surface level in each vessel remains the same. The density difference between the two vessels depends on the flow rate as well as the nature of the transfer through the walls. The flow 4 is positive if directed from vessel to vessel 2, and negative otherwise. There is, of course, a counterflow of the same amount returning through the overflow, so that the volume of water in each vessel remains the same. We are concerned with solution of symmetric form, so we can define a single temperature T= T= T, and a single salinity S = S, = S,, so that the laws for conservation of temperature and salinity are simply dt = c(t T) 1241 T ds =d(s S) 1241s t is important to note that in the second term on the right hand side the flux enters with an absolute value sign. This means that the exchange of properties is insensitive to the direction of the circulation. ntroducing the previous notation, and defrning dx =6(1x)flx A..=(y+Rx) The last equation implies that the flow in the capillary is in a series of quasiequilibrium statessince no time derivatives occur in it. Substitution of the last equation into the first two, to eliminate f; yields two nonlinear equations $ 4 y ; y+rxl dx The points of equilibrium correspond to those values of x and y for which dy/ and dx/ vanish, thus leading to a cubic for y in terms of x. There will, therefore, sometimes be three real solutions, or three sets of values of x, y which are equilibrium points. A simple graphical construction enables us to see under what conditions several equilibria are admitted. Solving the first two equations for x and y, in stationary state, we obtain and X Y= +lfl n figure 6 the function + (f; R, 6) has been plotted as a function of ffor R=2, and for two choices of 6, 6 =, and 6 ='s. 2 +(faal T = (L) k 4 eoat the a propriate nondimensional forms of the re P ations describing the systems are Tellur X11 (1961). 2 Fig. 6. Graph for determining the equilibria of the two vessel convection experiment (see text).
5 228 HENRY STOMMEL The solution (or real roots of the cubic) occur where this curve intersects the line AJ Two lines are drawn, one for 1 = and one for 1 = 's. 2 is defined as positive. n the case R=2, S=l/,, A=l/b there are three intersections, located at points a, b and c; the corres onding approximate values of the roots are = 1.1, 0.30, These re resent three P different ways in which t R e g simple convection can occur between the e coupled vessels without change in time. With a a somewhat larger value of 1 the line cuts the function 4 (fi 2, '/J in ody one point in the case 1 =, it cuts at d only. For certain choices of the parameters R and S there are forms of 4 (j R, 6) for which no choice of 1 can produce three real roots. For example, the choice R = 2, 6 = gives ody one intersection (e or g) for any one choice of 1. t can be seen that this is always true when 4 (f; R, S) has no zeros. To explore the limitation on zeros of the 4 function, we note that if 4 = o then or =R 1 + f1 ( R) 6 = (R6 ) f1 1 + flp Thus the necessary condition for three intersections is RS< ifr> or R ~ > ifo<r< To be a sufficient condition A must also be small enough. Proceeding now to the x, y sionless, S, T diagram) we can the lines of equal density. These, of course, coincide with the lines of equal flow f in the capillary. n figure 7, the three equilibrium points a, b, c, are located for the particular case R=z, a='/,, The locations are computed from the values of fluxfas determined in figure 6. The paths which temperature and salinity follow in the course of ap roaching equilibrium points can be plottecf by the method of isoclines as given in STOKER (gso), a few are sketched in figure 7. Both a and c are stable equilibrium points. U on detailed examination by the method o P PoincarC it can be shown that point a ' Salinity Fig. 7. The three equilibria a, b, and c for the two vessel convection experiment with R = 2, 6 = 116, 1=1/s. A few sample integral curves are sketched to show the stable node (, the saddle 6, and the stable spiral c. is a stable node, whereas point c is a stable spiral. Point b on the other hand is a saddle point, so that the system would not stay in that state if perturbed ever so slightly. A similar sketch for the system where only one equllibrium point (g in figure 6) in the system where R=z, 6=1, A=1/5 is shown in figure 8. t is a single stable node. The fact that even in a very simple convective system, such as here described, two distinct stable regimes can occur (as in figure 7)one (point a) where temperature differences dominate the deiai differences and the flow through the capilary 7 is from the cold to the warm vessel, and the other where salinity dominates the density difference so that the flow in the capillary is opposite, from warm to coldsuggests that a similar situation may exist somewhere in nature. One wonders whether other quite different states of flow are permissible in the ocean or some estuaries and if such a system might jump into on of these with a sufficient perturbation. f so, the system is inherently frought with possibilities for speculation about climatic change. Such a perturbation could be in the momentary state of the systemwith all parameters remaining constant, or it could Tellus X11 (1961). 2
6 THERMOHALNE CONVECTON WTH TWO STABLE REGMES OF FLOW 229 be a slight change in the parameters. Refer Moreover it is necessary to distinguish ring to figure 6 to see the effect of a Slight between two cases: () where the values 7 > increase in we see that if 3, were to become Rz, where < Rz. Equilibrium in SW Y greater than /4 9 the two intersections case (1) have negative J in case (2) positive 5 and would vanish. in the One must allow, in this way for the absolute in figure a light change in value sip Forming the quotient of the two linearized perturbation equations, leads to the general form could cause the temperature dominated circdation at u to jump over to the reverse salinity circulation at c, and it would then sta there even when 1 was restored to its origina r value. dy ux + by dx cx + dy Appendix The question of the stab&ty of the equili,., where the coefficients a, b, c, d are defined rium points is difficult to decide by graphical separately for the as fo1lows construction. We let x=x+x, y=y+y where X and Y are values at a particular 1 Case (1) Y> RX Case (2) F< RX 1 e uilibrium point; hence they satisfy the re f ation o=vt~y RX a f < O f>o b X 0 = 6 ( x 1 Y RJil The quantities x and y are regarded as small departures from and Y and we form a linearized expression for dxn/ and dy / which is valid in the neighborhood of X, Y by substitution and dropping all products of x y. t Y c d RF/ (2u RX)/ 8 +(2RX y)/ x/ RF/ + (z y RE)/3, 6 (z RX Y)/ %/ Fig. 8. The single stable node g for the case RZ, 61, Tellus X11 (1961). 2 A=1/s.
7 230 HENRY STOMMEL Table. Numerical Calculations on Stability of Equilibrium Points. System R=z 6 = ni = 's P=2 6=1 A='/, f Case X Y 1.10 (1) (1) (2) (2) a b C d (bc)' 4 ad (bc)' + 4aJ ad bc adbc b + c E e 3. 8 a" The Poincard conditions on the form of the equilibrium points are (STOKER, 1950) Type (b c)2+4ad>o Type 1 (b c), + 4 ad < 0 (A) Node if ad bc < o (B) Saddle if ad bc > o (A) Center if b + c = o (B) Spiral if b + c # o Stable if b + c < o Unstable if b + c > o Stable if b + c < o Unstable if b + c > o Type 111 (b~)~+4ad=o Node Stable if b + c < o Unstable if b + c > o The numerical values of all quantities are calculated in Table for the systems shown in figures 7 and 8. REFERENCE STOKER, J. J., 1950: Nonfinear Vibrations, nterscience Publisher, New York, 273 pp. Tellus X11 (1961), 2
Chapter 18 Temperature, Heat, and the First Law of Thermodynamics. Problems: 8, 11, 13, 17, 21, 27, 29, 37, 39, 41, 47, 51, 57
 Chapter 18 Temperature, Heat, and the First Law of Thermodynamics Problems: 8, 11, 13, 17, 21, 27, 29, 37, 39, 41, 47, 51, 57 Thermodynamics study and application of thermal energy temperature quantity
Chapter 18 Temperature, Heat, and the First Law of Thermodynamics Problems: 8, 11, 13, 17, 21, 27, 29, 37, 39, 41, 47, 51, 57 Thermodynamics study and application of thermal energy temperature quantity
Lecture 2. Marginal Functions, Average Functions, Elasticity, the Marginal Principle, and Constrained Optimization
 Lecture 2. Marginal Functions, Average Functions, Elasticity, the Marginal Principle, and Constrained Optimization 2.1. Introduction Suppose that an economic relationship can be described by a real-valued
Lecture 2. Marginal Functions, Average Functions, Elasticity, the Marginal Principle, and Constrained Optimization 2.1. Introduction Suppose that an economic relationship can be described by a real-valued
3.2 Sources, Sinks, Saddles, and Spirals
 3.2. Sources, Sinks, Saddles, and Spirals 6 3.2 Sources, Sinks, Saddles, and Spirals The pictures in this section show solutions to Ay 00 C By 0 C Cy D 0. These are linear equations with constant coefficients
3.2. Sources, Sinks, Saddles, and Spirals 6 3.2 Sources, Sinks, Saddles, and Spirals The pictures in this section show solutions to Ay 00 C By 0 C Cy D 0. These are linear equations with constant coefficients
12.307. 1 Convection in water (an almost-incompressible fluid)
 12.307 Convection in water (an almost-incompressible fluid) John Marshall, Lodovica Illari and Alan Plumb March, 2004 1 Convection in water (an almost-incompressible fluid) 1.1 Buoyancy Objects that are
12.307 Convection in water (an almost-incompressible fluid) John Marshall, Lodovica Illari and Alan Plumb March, 2004 1 Convection in water (an almost-incompressible fluid) 1.1 Buoyancy Objects that are
Carbon Cable. Sergio Rubio Carles Paul Albert Monte
 Carbon Cable Sergio Rubio Carles Paul Albert Monte Carbon, Copper and Manganine PhYsical PropERTieS CARBON PROPERTIES Carbon physical Properties Temperature Coefficient α -0,0005 ºC-1 Density D 2260 kg/m3
Carbon Cable Sergio Rubio Carles Paul Albert Monte Carbon, Copper and Manganine PhYsical PropERTieS CARBON PROPERTIES Carbon physical Properties Temperature Coefficient α -0,0005 ºC-1 Density D 2260 kg/m3
3. Reaction Diffusion Equations Consider the following ODE model for population growth
 3. Reaction Diffusion Equations Consider the following ODE model for population growth u t a u t u t, u 0 u 0 where u t denotes the population size at time t, and a u plays the role of the population dependent
3. Reaction Diffusion Equations Consider the following ODE model for population growth u t a u t u t, u 0 u 0 where u t denotes the population size at time t, and a u plays the role of the population dependent
Review of Fundamental Mathematics
 Review of Fundamental Mathematics As explained in the Preface and in Chapter 1 of your textbook, managerial economics applies microeconomic theory to business decision making. The decision-making tools
Review of Fundamental Mathematics As explained in the Preface and in Chapter 1 of your textbook, managerial economics applies microeconomic theory to business decision making. The decision-making tools
Understanding Poles and Zeros
 MASSACHUSETTS INSTITUTE OF TECHNOLOGY DEPARTMENT OF MECHANICAL ENGINEERING 2.14 Analysis and Design of Feedback Control Systems Understanding Poles and Zeros 1 System Poles and Zeros The transfer function
MASSACHUSETTS INSTITUTE OF TECHNOLOGY DEPARTMENT OF MECHANICAL ENGINEERING 2.14 Analysis and Design of Feedback Control Systems Understanding Poles and Zeros 1 System Poles and Zeros The transfer function
Module 1 : Conduction. Lecture 5 : 1D conduction example problems. 2D conduction
 Module 1 : Conduction Lecture 5 : 1D conduction example problems. 2D conduction Objectives In this class: An example of optimization for insulation thickness is solved. The 1D conduction is considered
Module 1 : Conduction Lecture 5 : 1D conduction example problems. 2D conduction Objectives In this class: An example of optimization for insulation thickness is solved. The 1D conduction is considered
Differential Relations for Fluid Flow. Acceleration field of a fluid. The differential equation of mass conservation
 Differential Relations for Fluid Flow In this approach, we apply our four basic conservation laws to an infinitesimally small control volume. The differential approach provides point by point details of
Differential Relations for Fluid Flow In this approach, we apply our four basic conservation laws to an infinitesimally small control volume. The differential approach provides point by point details of
CBE 6333, R. Levicky 1 Differential Balance Equations
 CBE 6333, R. Levicky 1 Differential Balance Equations We have previously derived integral balances for mass, momentum, and energy for a control volume. The control volume was assumed to be some large object,
CBE 6333, R. Levicky 1 Differential Balance Equations We have previously derived integral balances for mass, momentum, and energy for a control volume. The control volume was assumed to be some large object,
3.3. Solving Polynomial Equations. Introduction. Prerequisites. Learning Outcomes
 Solving Polynomial Equations 3.3 Introduction Linear and quadratic equations, dealt within Sections 3.1 and 3.2, are members of a class of equations, called polynomial equations. These have the general
Solving Polynomial Equations 3.3 Introduction Linear and quadratic equations, dealt within Sections 3.1 and 3.2, are members of a class of equations, called polynomial equations. These have the general
Natural Convection. Buoyancy force
 Natural Convection In natural convection, the fluid motion occurs by natural means such as buoyancy. Since the fluid velocity associated with natural convection is relatively low, the heat transfer coefficient
Natural Convection In natural convection, the fluid motion occurs by natural means such as buoyancy. Since the fluid velocity associated with natural convection is relatively low, the heat transfer coefficient
ME 315 - Heat Transfer Laboratory. Experiment No. 7 ANALYSIS OF ENHANCED CONCENTRIC TUBE AND SHELL AND TUBE HEAT EXCHANGERS
 ME 315 - Heat Transfer Laboratory Nomenclature Experiment No. 7 ANALYSIS OF ENHANCED CONCENTRIC TUBE AND SHELL AND TUBE HEAT EXCHANGERS A heat exchange area, m 2 C max maximum specific heat rate, J/(s
ME 315 - Heat Transfer Laboratory Nomenclature Experiment No. 7 ANALYSIS OF ENHANCED CONCENTRIC TUBE AND SHELL AND TUBE HEAT EXCHANGERS A heat exchange area, m 2 C max maximum specific heat rate, J/(s
Comparison of Heat Transfer between a Helical and Straight Tube Heat Exchanger
 International Journal of Engineering Research and Technology. ISSN 0974-3154 Volume 6, Number 1 (2013), pp. 33-40 International Research Publication House http://www.irphouse.com Comparison of Heat Transfer
International Journal of Engineering Research and Technology. ISSN 0974-3154 Volume 6, Number 1 (2013), pp. 33-40 International Research Publication House http://www.irphouse.com Comparison of Heat Transfer
Figure 2.1: Center of mass of four points.
 Chapter 2 Bézier curves are named after their inventor, Dr. Pierre Bézier. Bézier was an engineer with the Renault car company and set out in the early 196 s to develop a curve formulation which would
Chapter 2 Bézier curves are named after their inventor, Dr. Pierre Bézier. Bézier was an engineer with the Renault car company and set out in the early 196 s to develop a curve formulation which would
CFD SIMULATION OF SDHW STORAGE TANK WITH AND WITHOUT HEATER
 International Journal of Advancements in Research & Technology, Volume 1, Issue2, July-2012 1 CFD SIMULATION OF SDHW STORAGE TANK WITH AND WITHOUT HEATER ABSTRACT (1) Mr. Mainak Bhaumik M.E. (Thermal Engg.)
International Journal of Advancements in Research & Technology, Volume 1, Issue2, July-2012 1 CFD SIMULATION OF SDHW STORAGE TANK WITH AND WITHOUT HEATER ABSTRACT (1) Mr. Mainak Bhaumik M.E. (Thermal Engg.)
ES 106 Laboratory # 3 INTRODUCTION TO OCEANOGRAPHY. Introduction The global ocean covers nearly 75% of Earth s surface and plays a vital role in
 ES 106 Laboratory # 3 INTRODUCTION TO OCEANOGRAPHY 3-1 Introduction The global ocean covers nearly 75% of Earth s surface and plays a vital role in the physical environment of Earth. For these reasons,
ES 106 Laboratory # 3 INTRODUCTION TO OCEANOGRAPHY 3-1 Introduction The global ocean covers nearly 75% of Earth s surface and plays a vital role in the physical environment of Earth. For these reasons,
Ocean in Motion 2: What Causes Ocean Currents and How Do We Measure Them?
 Ocean in Motion 2: What Causes Ocean Currents and How Do We Measure Them? A. Overview 1. The Ocean in Motion -- Circulation In this program, students will learn about the driving forces responsible for
Ocean in Motion 2: What Causes Ocean Currents and How Do We Measure Them? A. Overview 1. The Ocean in Motion -- Circulation In this program, students will learn about the driving forces responsible for
CONSERVATION LAWS. See Figures 2 and 1.
 CONSERVATION LAWS 1. Multivariable calculus 1.1. Divergence theorem (of Gauss). This states that the volume integral in of the divergence of the vector-valued function F is equal to the total flux of F
CONSERVATION LAWS 1. Multivariable calculus 1.1. Divergence theorem (of Gauss). This states that the volume integral in of the divergence of the vector-valued function F is equal to the total flux of F
Lecture 6 - Boundary Conditions. Applied Computational Fluid Dynamics
 Lecture 6 - Boundary Conditions Applied Computational Fluid Dynamics Instructor: André Bakker http://www.bakker.org André Bakker (2002-2006) Fluent Inc. (2002) 1 Outline Overview. Inlet and outlet boundaries.
Lecture 6 - Boundary Conditions Applied Computational Fluid Dynamics Instructor: André Bakker http://www.bakker.org André Bakker (2002-2006) Fluent Inc. (2002) 1 Outline Overview. Inlet and outlet boundaries.
Lecture 5 Hemodynamics. Description of fluid flow. The equation of continuity
 1 Lecture 5 Hemodynamics Description of fluid flow Hydrodynamics is the part of physics, which studies the motion of fluids. It is based on the laws of mechanics. Hemodynamics studies the motion of blood
1 Lecture 5 Hemodynamics Description of fluid flow Hydrodynamics is the part of physics, which studies the motion of fluids. It is based on the laws of mechanics. Hemodynamics studies the motion of blood
Fluid Mechanics: Static s Kinematics Dynamics Fluid
 Fluid Mechanics: Fluid mechanics may be defined as that branch of engineering science that deals with the behavior of fluid under the condition of rest and motion Fluid mechanics may be divided into three
Fluid Mechanics: Fluid mechanics may be defined as that branch of engineering science that deals with the behavior of fluid under the condition of rest and motion Fluid mechanics may be divided into three
Density: Sea Water Mixing and Sinking
 Density: Sea Water Mixing and Sinking Unit: Salinity Patterr~s & the Water Cycle I Grade Level: Middle or High I Time Required: two 45 minute class periods I Content Standard: NSES Physical Science, properties
Density: Sea Water Mixing and Sinking Unit: Salinity Patterr~s & the Water Cycle I Grade Level: Middle or High I Time Required: two 45 minute class periods I Content Standard: NSES Physical Science, properties
The final numerical answer given is correct but the math shown does not give that answer.
 Note added to Homework set 7: The solution to Problem 16 has an error in it. The specific heat of water is listed as c 1 J/g K but should be c 4.186 J/g K The final numerical answer given is correct but
Note added to Homework set 7: The solution to Problem 16 has an error in it. The specific heat of water is listed as c 1 J/g K but should be c 4.186 J/g K The final numerical answer given is correct but
High Speed Aerodynamics Prof. K. P. Sinhamahapatra Department of Aerospace Engineering Indian Institute of Technology, Kharagpur
 High Speed Aerodynamics Prof. K. P. Sinhamahapatra Department of Aerospace Engineering Indian Institute of Technology, Kharagpur Module No. # 01 Lecture No. # 06 One-dimensional Gas Dynamics (Contd.) We
High Speed Aerodynamics Prof. K. P. Sinhamahapatra Department of Aerospace Engineering Indian Institute of Technology, Kharagpur Module No. # 01 Lecture No. # 06 One-dimensional Gas Dynamics (Contd.) We
Stability and Cloud Development. Stability in the atmosphere AT350. Why did this cloud form, whereas the sky was clear 4 hours ago?
 Stability and Cloud Development AT350 Why did this cloud form, whereas the sky was clear 4 hours ago? Stability in the atmosphere An Initial Perturbation Stable Unstable Neutral If an air parcel is displaced
Stability and Cloud Development AT350 Why did this cloud form, whereas the sky was clear 4 hours ago? Stability in the atmosphere An Initial Perturbation Stable Unstable Neutral If an air parcel is displaced
Steady Heat Conduction
 Steady Heat Conduction In thermodynamics, we considered the amount of heat transfer as a system undergoes a process from one equilibrium state to another. hermodynamics gives no indication of how long
Steady Heat Conduction In thermodynamics, we considered the amount of heat transfer as a system undergoes a process from one equilibrium state to another. hermodynamics gives no indication of how long
Essay 5 Tutorial for a Three-Dimensional Heat Conduction Problem Using ANSYS Workbench
 Essay 5 Tutorial for a Three-Dimensional Heat Conduction Problem Using ANSYS Workbench 5.1 Introduction The problem selected to illustrate the use of ANSYS software for a three-dimensional steadystate
Essay 5 Tutorial for a Three-Dimensional Heat Conduction Problem Using ANSYS Workbench 5.1 Introduction The problem selected to illustrate the use of ANSYS software for a three-dimensional steadystate
Ocean Tracers. From Particles to sediment Thermohaline Circulation Past present and future ocean and climate. Only 4 hours left.
 Ocean Tracers Basic facts and principles (Size, depth, S, T,, f, water masses, surface circulation, deep circulation, observing tools, ) Seawater not just water (Salt composition, Sources, sinks,, mixing
Ocean Tracers Basic facts and principles (Size, depth, S, T,, f, water masses, surface circulation, deep circulation, observing tools, ) Seawater not just water (Salt composition, Sources, sinks,, mixing
ES250: Electrical Science. HW7: Energy Storage Elements
 ES250: Electrical Science HW7: Energy Storage Elements Introduction This chapter introduces two more circuit elements, the capacitor and the inductor whose elements laws involve integration or differentiation;
ES250: Electrical Science HW7: Energy Storage Elements Introduction This chapter introduces two more circuit elements, the capacitor and the inductor whose elements laws involve integration or differentiation;
Nonlinear Systems of Ordinary Differential Equations
 Differential Equations Massoud Malek Nonlinear Systems of Ordinary Differential Equations Dynamical System. A dynamical system has a state determined by a collection of real numbers, or more generally
Differential Equations Massoud Malek Nonlinear Systems of Ordinary Differential Equations Dynamical System. A dynamical system has a state determined by a collection of real numbers, or more generally
CONVECTION CURRENTS AND ANOMALOUS BEHAVIOUR OF WATER
 CONVECTION CURRENTS AND ANOMALOUS BEHAVIOUR OF WATER Objective: To compare the thermal behaviour of water with that of other liquids, specifically alcohol and edible oil. To point out the anomaly of water
CONVECTION CURRENTS AND ANOMALOUS BEHAVIOUR OF WATER Objective: To compare the thermal behaviour of water with that of other liquids, specifically alcohol and edible oil. To point out the anomaly of water
Lecture 3 Fluid Dynamics and Balance Equa6ons for Reac6ng Flows
 Lecture 3 Fluid Dynamics and Balance Equa6ons for Reac6ng Flows 3.- 1 Basics: equations of continuum mechanics - balance equations for mass and momentum - balance equations for the energy and the chemical
Lecture 3 Fluid Dynamics and Balance Equa6ons for Reac6ng Flows 3.- 1 Basics: equations of continuum mechanics - balance equations for mass and momentum - balance equations for the energy and the chemical
When the fluid velocity is zero, called the hydrostatic condition, the pressure variation is due only to the weight of the fluid.
 Fluid Statics When the fluid velocity is zero, called the hydrostatic condition, the pressure variation is due only to the weight of the fluid. Consider a small wedge of fluid at rest of size Δx, Δz, Δs
Fluid Statics When the fluid velocity is zero, called the hydrostatic condition, the pressure variation is due only to the weight of the fluid. Consider a small wedge of fluid at rest of size Δx, Δz, Δs
How To Calculate Thermal Resistance On A Pb (Plastipo)
 VISHAY BEYSCHLAG Resistive Products 1. INTRODUCTION Thermal management is becoming more important as the density of electronic components in modern printed circuit boards (PCBs), as well as the applied
VISHAY BEYSCHLAG Resistive Products 1. INTRODUCTION Thermal management is becoming more important as the density of electronic components in modern printed circuit boards (PCBs), as well as the applied
Circuits 1 M H Miller
 Introduction to Graph Theory Introduction These notes are primarily a digression to provide general background remarks. The subject is an efficient procedure for the determination of voltages and currents
Introduction to Graph Theory Introduction These notes are primarily a digression to provide general background remarks. The subject is an efficient procedure for the determination of voltages and currents
Module 2. Analysis of Statically Indeterminate Structures by the Matrix Force Method. Version 2 CE IIT, Kharagpur
 Module Analysis of Statically Indeterminate Structures by the Matrix Force Method esson 11 The Force Method of Analysis: Frames Instructional Objectives After reading this chapter the student will be able
Module Analysis of Statically Indeterminate Structures by the Matrix Force Method esson 11 The Force Method of Analysis: Frames Instructional Objectives After reading this chapter the student will be able
Experiment 3 Pipe Friction
 EML 316L Experiment 3 Pipe Friction Laboratory Manual Mechanical and Materials Engineering Department College of Engineering FLORIDA INTERNATIONAL UNIVERSITY Nomenclature Symbol Description Unit A cross-sectional
EML 316L Experiment 3 Pipe Friction Laboratory Manual Mechanical and Materials Engineering Department College of Engineering FLORIDA INTERNATIONAL UNIVERSITY Nomenclature Symbol Description Unit A cross-sectional
FREESTUDY HEAT TRANSFER TUTORIAL 3 ADVANCED STUDIES
 FREESTUDY HEAT TRANSFER TUTORIAL ADVANCED STUDIES This is the third tutorial in the series on heat transfer and covers some of the advanced theory of convection. The tutorials are designed to bring the
FREESTUDY HEAT TRANSFER TUTORIAL ADVANCED STUDIES This is the third tutorial in the series on heat transfer and covers some of the advanced theory of convection. The tutorials are designed to bring the
INJECTION MOLDING COOLING TIME REDUCTION AND THERMAL STRESS ANALYSIS
 INJECTION MOLDING COOLING TIME REDUCTION AND THERMAL STRESS ANALYSIS Tom Kimerling University of Massachusetts, Amherst MIE 605 Finite Element Analysis Spring 2002 ABSTRACT A FEA transient thermal structural
INJECTION MOLDING COOLING TIME REDUCTION AND THERMAL STRESS ANALYSIS Tom Kimerling University of Massachusetts, Amherst MIE 605 Finite Element Analysis Spring 2002 ABSTRACT A FEA transient thermal structural
FLUID FLOW STREAMLINE LAMINAR FLOW TURBULENT FLOW REYNOLDS NUMBER
 VISUAL PHYSICS School of Physics University of Sydney Australia FLUID FLOW STREAMLINE LAMINAR FLOW TURBULENT FLOW REYNOLDS NUMBER? What type of fluid flow is observed? The above pictures show how the effect
VISUAL PHYSICS School of Physics University of Sydney Australia FLUID FLOW STREAMLINE LAMINAR FLOW TURBULENT FLOW REYNOLDS NUMBER? What type of fluid flow is observed? The above pictures show how the effect
On using numerical algebraic geometry to find Lyapunov functions of polynomial dynamical systems
 Dynamics at the Horsetooth Volume 2, 2010. On using numerical algebraic geometry to find Lyapunov functions of polynomial dynamical systems Eric Hanson Department of Mathematics Colorado State University
Dynamics at the Horsetooth Volume 2, 2010. On using numerical algebraic geometry to find Lyapunov functions of polynomial dynamical systems Eric Hanson Department of Mathematics Colorado State University
Network Traffic Modelling
 University of York Dissertation submitted for the MSc in Mathematics with Modern Applications, Department of Mathematics, University of York, UK. August 009 Network Traffic Modelling Author: David Slade
University of York Dissertation submitted for the MSc in Mathematics with Modern Applications, Department of Mathematics, University of York, UK. August 009 Network Traffic Modelling Author: David Slade
Atmospheric Stability & Cloud Development
 Atmospheric Stability & Cloud Development Stable situations a small change is resisted and the system returns to its previous state Neutral situations a small change is neither resisted nor enlarged Unstable
Atmospheric Stability & Cloud Development Stable situations a small change is resisted and the system returns to its previous state Neutral situations a small change is neither resisted nor enlarged Unstable
Scholars Research Library
 Available online at www.scholarsresearchlibrary.com Scholars Research Library Archives of Physics Research, 2010, 1 (2):103-111 (http://scholarsresearchlibrary.com/archive.html) ISSN 0976-0970 Lubricating
Available online at www.scholarsresearchlibrary.com Scholars Research Library Archives of Physics Research, 2010, 1 (2):103-111 (http://scholarsresearchlibrary.com/archive.html) ISSN 0976-0970 Lubricating
EXIT TIME PROBLEMS AND ESCAPE FROM A POTENTIAL WELL
 EXIT TIME PROBLEMS AND ESCAPE FROM A POTENTIAL WELL Exit Time problems and Escape from a Potential Well Escape From a Potential Well There are many systems in physics, chemistry and biology that exist
EXIT TIME PROBLEMS AND ESCAPE FROM A POTENTIAL WELL Exit Time problems and Escape from a Potential Well Escape From a Potential Well There are many systems in physics, chemistry and biology that exist
Exergy: the quality of energy N. Woudstra
 Exergy: the quality of energy N. Woudstra Introduction Characteristic for our society is a massive consumption of goods and energy. Continuation of this way of life in the long term is only possible if
Exergy: the quality of energy N. Woudstra Introduction Characteristic for our society is a massive consumption of goods and energy. Continuation of this way of life in the long term is only possible if
Document extract. AAMT supporting and enhancing the work of teachers. A Classroom Investigation into the Catenary. Ed Staples
 Document extract Title of chapter/article A Classroom Investigation into the Catenary Author(s) Ed Staples Copyright owner The Australian Association of Mathematics Teachers (AAMT) Inc. Published in Australian
Document extract Title of chapter/article A Classroom Investigation into the Catenary Author(s) Ed Staples Copyright owner The Australian Association of Mathematics Teachers (AAMT) Inc. Published in Australian
Coupling Forced Convection in Air Gaps with Heat and Moisture Transfer inside Constructions
 Coupling Forced Convection in Air Gaps with Heat and Moisture Transfer inside Constructions M. Bianchi Janetti 1, F. Ochs 1 and R. Pfluger 1 1 University of Innsbruck, Unit for Energy Efficient Buildings,
Coupling Forced Convection in Air Gaps with Heat and Moisture Transfer inside Constructions M. Bianchi Janetti 1, F. Ochs 1 and R. Pfluger 1 1 University of Innsbruck, Unit for Energy Efficient Buildings,
Measuring Optical and Thermal Properties of High Temperature Receivers
 www.dlr.de Folie 1 Measuring Optical and Thermal Properties of High Temperature Receivers Johannes Pernpeintner, Thomas Fend 4 th SFERA Summerschool, May 15-16, 2013, Burg Hornberg www.dlr.de Folie 2 Part
www.dlr.de Folie 1 Measuring Optical and Thermal Properties of High Temperature Receivers Johannes Pernpeintner, Thomas Fend 4 th SFERA Summerschool, May 15-16, 2013, Burg Hornberg www.dlr.de Folie 2 Part
A LAMINAR FLOW ELEMENT WITH A LINEAR PRESSURE DROP VERSUS VOLUMETRIC FLOW. 1998 ASME Fluids Engineering Division Summer Meeting
 TELEDYNE HASTINGS TECHNICAL PAPERS INSTRUMENTS A LAMINAR FLOW ELEMENT WITH A LINEAR PRESSURE DROP VERSUS VOLUMETRIC FLOW Proceedings of FEDSM 98: June -5, 998, Washington, DC FEDSM98 49 ABSTRACT The pressure
TELEDYNE HASTINGS TECHNICAL PAPERS INSTRUMENTS A LAMINAR FLOW ELEMENT WITH A LINEAR PRESSURE DROP VERSUS VOLUMETRIC FLOW Proceedings of FEDSM 98: June -5, 998, Washington, DC FEDSM98 49 ABSTRACT The pressure
The Oceans Role in Climate
 The Oceans Role in Climate Martin H. Visbeck A Numerical Portrait of the Oceans The oceans of the world cover nearly seventy percent of its surface. The largest is the Pacific, which contains fifty percent
The Oceans Role in Climate Martin H. Visbeck A Numerical Portrait of the Oceans The oceans of the world cover nearly seventy percent of its surface. The largest is the Pacific, which contains fifty percent
APPLICATION OF TRANSIENT WELLBORE SIMULATOR TO EVALUATE DELIVERABILITY CURVE ON HYPOTHETICAL WELL-X
 PROCEEDINGS, Thirty-Third Workshop on Geothermal Reservoir Engineering Stanford University, Stanford, California, January 8-30, 008 SGP-TR-185 APPLICATION OF TRANSIENT WELLBORE SIMULATOR TO EVALUATE DELIVERABILITY
PROCEEDINGS, Thirty-Third Workshop on Geothermal Reservoir Engineering Stanford University, Stanford, California, January 8-30, 008 SGP-TR-185 APPLICATION OF TRANSIENT WELLBORE SIMULATOR TO EVALUATE DELIVERABILITY
Integration of a fin experiment into the undergraduate heat transfer laboratory
 Integration of a fin experiment into the undergraduate heat transfer laboratory H. I. Abu-Mulaweh Mechanical Engineering Department, Purdue University at Fort Wayne, Fort Wayne, IN 46805, USA E-mail: mulaweh@engr.ipfw.edu
Integration of a fin experiment into the undergraduate heat transfer laboratory H. I. Abu-Mulaweh Mechanical Engineering Department, Purdue University at Fort Wayne, Fort Wayne, IN 46805, USA E-mail: mulaweh@engr.ipfw.edu
The aerodynamic center
 The aerodynamic center In this chapter, we re going to focus on the aerodynamic center, and its effect on the moment coefficient C m. 1 Force and moment coefficients 1.1 Aerodynamic forces Let s investigate
The aerodynamic center In this chapter, we re going to focus on the aerodynamic center, and its effect on the moment coefficient C m. 1 Force and moment coefficients 1.1 Aerodynamic forces Let s investigate
FREE CONVECTION FROM OPTIMUM SINUSOIDAL SURFACE EXPOSED TO VERTICAL VIBRATIONS
 International Journal of Mechanical Engineering and Technology (IJMET) Volume 7, Issue 1, Jan-Feb 2016, pp. 214-224, Article ID: IJMET_07_01_022 Available online at http://www.iaeme.com/ijmet/issues.asp?jtype=ijmet&vtype=7&itype=1
International Journal of Mechanical Engineering and Technology (IJMET) Volume 7, Issue 1, Jan-Feb 2016, pp. 214-224, Article ID: IJMET_07_01_022 Available online at http://www.iaeme.com/ijmet/issues.asp?jtype=ijmet&vtype=7&itype=1
Lesson. 11 Vapour Compression Refrigeration Systems: Performance Aspects And Cycle Modifications. Version 1 ME, IIT Kharagpur 1
 Lesson Vapour Compression Refrigeration Systems: Performance Aspects And Cycle Modifications Version ME, IIT Kharagpur The objectives of this lecture are to discuss. Performance aspects of SSS cycle and
Lesson Vapour Compression Refrigeration Systems: Performance Aspects And Cycle Modifications Version ME, IIT Kharagpur The objectives of this lecture are to discuss. Performance aspects of SSS cycle and
FUNDAMENTALS OF ENGINEERING THERMODYNAMICS
 FUNDAMENTALS OF ENGINEERING THERMODYNAMICS System: Quantity of matter (constant mass) or region in space (constant volume) chosen for study. Closed system: Can exchange energy but not mass; mass is constant
FUNDAMENTALS OF ENGINEERING THERMODYNAMICS System: Quantity of matter (constant mass) or region in space (constant volume) chosen for study. Closed system: Can exchange energy but not mass; mass is constant
Density Lab. If you get stuck or are uncertain, please ask questions and/or refer to the hints at the end of the lab. Name: Section: Due Date:
 Name: Section: Due Date: Lab 01B-1 If you get stuck or are uncertain, please ask questions and/or refer to the hints at the end of the lab. Density Lab Density is an important concept in oceanography,
Name: Section: Due Date: Lab 01B-1 If you get stuck or are uncertain, please ask questions and/or refer to the hints at the end of the lab. Density Lab Density is an important concept in oceanography,
Solving Simultaneous Equations and Matrices
 Solving Simultaneous Equations and Matrices The following represents a systematic investigation for the steps used to solve two simultaneous linear equations in two unknowns. The motivation for considering
Solving Simultaneous Equations and Matrices The following represents a systematic investigation for the steps used to solve two simultaneous linear equations in two unknowns. The motivation for considering
Orbits of the Lennard-Jones Potential
 Orbits of the Lennard-Jones Potential Prashanth S. Venkataram July 28, 2012 1 Introduction The Lennard-Jones potential describes weak interactions between neutral atoms and molecules. Unlike the potentials
Orbits of the Lennard-Jones Potential Prashanth S. Venkataram July 28, 2012 1 Introduction The Lennard-Jones potential describes weak interactions between neutral atoms and molecules. Unlike the potentials
Convective Clouds. Convective clouds 1
 Convective clouds 1 Convective Clouds Introduction Convective clouds are formed in vertical motions that result from the instability of the atmosphere. This instability can be caused by: a. heating at
Convective clouds 1 Convective Clouds Introduction Convective clouds are formed in vertical motions that result from the instability of the atmosphere. This instability can be caused by: a. heating at
Gas Well Deliverability Testing
 Gas Well Deliverability Testing What is deliverability testing? The "deliverability" of a gas well can be defined as the well's capacity to produce against the restrictions of the well bore and the system
Gas Well Deliverability Testing What is deliverability testing? The "deliverability" of a gas well can be defined as the well's capacity to produce against the restrictions of the well bore and the system
We will try to get familiar with a heat pump, and try to determine its performance coefficient under different circumstances.
 C4. Heat Pump I. OBJECTIVE OF THE EXPERIMENT We will try to get familiar with a heat pump, and try to determine its performance coefficient under different circumstances. II. INTRODUCTION II.1. Thermodynamic
C4. Heat Pump I. OBJECTIVE OF THE EXPERIMENT We will try to get familiar with a heat pump, and try to determine its performance coefficient under different circumstances. II. INTRODUCTION II.1. Thermodynamic
BIOE 370 1. Lotka-Volterra Model L-V model with density-dependent prey population growth
 BIOE 370 1 Populus Simulations of Predator-Prey Population Dynamics. Lotka-Volterra Model L-V model with density-dependent prey population growth Theta-Logistic Model Effects on dynamics of different functional
BIOE 370 1 Populus Simulations of Predator-Prey Population Dynamics. Lotka-Volterra Model L-V model with density-dependent prey population growth Theta-Logistic Model Effects on dynamics of different functional
Comparing Air Cooler Ratings Part 1: Not All Rating Methods are Created Equal
 Technical Bulletin By Bruce I. Nelson, P.E., President, Colmac Coil Manufacturing, Inc. Comparing Air Cooler Ratings Part 1: Not All Rating Methods are Created Equal SUMMARY Refrigeration air coolers (evaporators)
Technical Bulletin By Bruce I. Nelson, P.E., President, Colmac Coil Manufacturing, Inc. Comparing Air Cooler Ratings Part 1: Not All Rating Methods are Created Equal SUMMARY Refrigeration air coolers (evaporators)
Heat Transfer Prof. Dr. Aloke Kumar Ghosal Department of Chemical Engineering Indian Institute of Technology, Guwahati
 Heat Transfer Prof. Dr. Aloke Kumar Ghosal Department of Chemical Engineering Indian Institute of Technology, Guwahati Module No. # 02 One Dimensional Steady State Heat Transfer Lecture No. # 05 Extended
Heat Transfer Prof. Dr. Aloke Kumar Ghosal Department of Chemical Engineering Indian Institute of Technology, Guwahati Module No. # 02 One Dimensional Steady State Heat Transfer Lecture No. # 05 Extended
PERFORMANCE ANALYSIS OF VAPOUR COMPRESSION REFRIGERATION SYSTEM WITH R404A, R407C AND R410A
 Int. J. Mech. Eng. & Rob. Res. 213 Jyoti Soni and R C Gupta, 213 Research Paper ISSN 2278 149 www.ijmerr.com Vol. 2, No. 1, January 213 213 IJMERR. All Rights Reserved PERFORMANCE ANALYSIS OF VAPOUR COMPRESSION
Int. J. Mech. Eng. & Rob. Res. 213 Jyoti Soni and R C Gupta, 213 Research Paper ISSN 2278 149 www.ijmerr.com Vol. 2, No. 1, January 213 213 IJMERR. All Rights Reserved PERFORMANCE ANALYSIS OF VAPOUR COMPRESSION
CHAPTER 9 CHANNELS APPENDIX A. Hydraulic Design Equations for Open Channel Flow
 CHAPTER 9 CHANNELS APPENDIX A Hydraulic Design Equations for Open Channel Flow SEPTEMBER 2009 CHAPTER 9 APPENDIX A Hydraulic Design Equations for Open Channel Flow Introduction The Equations presented
CHAPTER 9 CHANNELS APPENDIX A Hydraulic Design Equations for Open Channel Flow SEPTEMBER 2009 CHAPTER 9 APPENDIX A Hydraulic Design Equations for Open Channel Flow Introduction The Equations presented
Procon Engineering. Technical Document PELR 1002. TERMS and DEFINITIONS
 Procon Engineering Technical Document PELR 1002 TERMS and DEFINITIONS The following terms are widely used in the weighing industry. Informal comment on terms is in italics and is not part of the formal
Procon Engineering Technical Document PELR 1002 TERMS and DEFINITIONS The following terms are widely used in the weighing industry. Informal comment on terms is in italics and is not part of the formal
8.5 Comparing Canadian Climates (Lab)
 These 3 climate graphs and tables of data show average temperatures and precipitation for each month in Victoria, Winnipeg and Whitehorse: Figure 1.1 Month J F M A M J J A S O N D Year Precipitation 139
These 3 climate graphs and tables of data show average temperatures and precipitation for each month in Victoria, Winnipeg and Whitehorse: Figure 1.1 Month J F M A M J J A S O N D Year Precipitation 139
G(s) = Y (s)/u(s) In this representation, the output is always the Transfer function times the input. Y (s) = G(s)U(s).
 Transfer Functions The transfer function of a linear system is the ratio of the Laplace Transform of the output to the Laplace Transform of the input, i.e., Y (s)/u(s). Denoting this ratio by G(s), i.e.,
Transfer Functions The transfer function of a linear system is the ratio of the Laplace Transform of the output to the Laplace Transform of the input, i.e., Y (s)/u(s). Denoting this ratio by G(s), i.e.,
A Dynamic Analysis of Price Determination Under Joint Profit Maximization in Bilateral Monopoly
 A Dynamic Analysis of Price Determination Under Joint Profit Maximization in Bilateral Monopoly by Stephen Devadoss Department of Agricultural Economics University of Idaho Moscow, Idaho 83844-2334 Phone:
A Dynamic Analysis of Price Determination Under Joint Profit Maximization in Bilateral Monopoly by Stephen Devadoss Department of Agricultural Economics University of Idaho Moscow, Idaho 83844-2334 Phone:
Energy savings in commercial refrigeration. Low pressure control
 Energy savings in commercial refrigeration equipment : Low pressure control August 2011/White paper by Christophe Borlein AFF and l IIF-IIR member Make the most of your energy Summary Executive summary
Energy savings in commercial refrigeration equipment : Low pressure control August 2011/White paper by Christophe Borlein AFF and l IIF-IIR member Make the most of your energy Summary Executive summary
Modern Construction Materials Prof. Ravindra Gettu Department of Civil Engineering Indian Institute of Technology, Madras
 Modern Construction Materials Prof. Ravindra Gettu Department of Civil Engineering Indian Institute of Technology, Madras Module - 2 Lecture - 2 Part 2 of 2 Review of Atomic Bonding II We will continue
Modern Construction Materials Prof. Ravindra Gettu Department of Civil Engineering Indian Institute of Technology, Madras Module - 2 Lecture - 2 Part 2 of 2 Review of Atomic Bonding II We will continue
Heat equation examples
 Heat equation examples The Heat equation is discussed in depth in http://tutorial.math.lamar.edu/classes/de/intropde.aspx, starting on page 6. You may recall Newton s Law of Cooling from Calculus. Just
Heat equation examples The Heat equation is discussed in depth in http://tutorial.math.lamar.edu/classes/de/intropde.aspx, starting on page 6. You may recall Newton s Law of Cooling from Calculus. Just
How Do Oceans Affect Weather and Climate?
 How Do Oceans Affect Weather and Climate? In Learning Set 2, you explored how water heats up more slowly than land and also cools off more slowly than land. Weather is caused by events in the atmosphere.
How Do Oceans Affect Weather and Climate? In Learning Set 2, you explored how water heats up more slowly than land and also cools off more slowly than land. Weather is caused by events in the atmosphere.
Dimensional analysis is a method for reducing the number and complexity of experimental variables that affect a given physical phenomena.
 Dimensional Analysis and Similarity Dimensional analysis is very useful for planning, presentation, and interpretation of experimental data. As discussed previously, most practical fluid mechanics problems
Dimensional Analysis and Similarity Dimensional analysis is very useful for planning, presentation, and interpretation of experimental data. As discussed previously, most practical fluid mechanics problems
A Comparison of Analytical and Finite Element Solutions for Laminar Flow Conditions Near Gaussian Constrictions
 A Comparison of Analytical and Finite Element Solutions for Laminar Flow Conditions Near Gaussian Constrictions by Laura Noelle Race An Engineering Project Submitted to the Graduate Faculty of Rensselaer
A Comparison of Analytical and Finite Element Solutions for Laminar Flow Conditions Near Gaussian Constrictions by Laura Noelle Race An Engineering Project Submitted to the Graduate Faculty of Rensselaer
5.2. Vaporizers - Types and Usage
 5.2. Vaporizers - Types and Usage 5.2.1. General Vaporizers are constructed in numerous designs and operated in many modes. Depending upon the service application the design, construction, inspection,
5.2. Vaporizers - Types and Usage 5.2.1. General Vaporizers are constructed in numerous designs and operated in many modes. Depending upon the service application the design, construction, inspection,
The simulation of machine tools can be divided into two stages. In the first stage the mechanical behavior of a machine tool is simulated with FEM
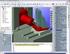 1 The simulation of machine tools can be divided into two stages. In the first stage the mechanical behavior of a machine tool is simulated with FEM tools. The approach to this simulation is different
1 The simulation of machine tools can be divided into two stages. In the first stage the mechanical behavior of a machine tool is simulated with FEM tools. The approach to this simulation is different
Performance of networks containing both MaxNet and SumNet links
 Performance of networks containing both MaxNet and SumNet links Lachlan L. H. Andrew and Bartek P. Wydrowski Abstract Both MaxNet and SumNet are distributed congestion control architectures suitable for
Performance of networks containing both MaxNet and SumNet links Lachlan L. H. Andrew and Bartek P. Wydrowski Abstract Both MaxNet and SumNet are distributed congestion control architectures suitable for
Experiment 1: Colligative Properties
 Experiment 1: Colligative Properties Determination of the Molar Mass of a Compound by Freezing Point Depression. Objective: The objective of this experiment is to determine the molar mass of an unknown
Experiment 1: Colligative Properties Determination of the Molar Mass of a Compound by Freezing Point Depression. Objective: The objective of this experiment is to determine the molar mass of an unknown
Diffusion and Fluid Flow
 Diffusion and Fluid Flow What determines the diffusion coefficient? What determines fluid flow? 1. Diffusion: Diffusion refers to the transport of substance against a concentration gradient. ΔS>0 Mass
Diffusion and Fluid Flow What determines the diffusion coefficient? What determines fluid flow? 1. Diffusion: Diffusion refers to the transport of substance against a concentration gradient. ΔS>0 Mass
Review D: Potential Energy and the Conservation of Mechanical Energy
 MSSCHUSETTS INSTITUTE OF TECHNOLOGY Department of Physics 8.01 Fall 2005 Review D: Potential Energy and the Conservation of Mechanical Energy D.1 Conservative and Non-conservative Force... 2 D.1.1 Introduction...
MSSCHUSETTS INSTITUTE OF TECHNOLOGY Department of Physics 8.01 Fall 2005 Review D: Potential Energy and the Conservation of Mechanical Energy D.1 Conservative and Non-conservative Force... 2 D.1.1 Introduction...
Heating & Cooling in Molecular Clouds
 Lecture 8: Cloud Stability Heating & Cooling in Molecular Clouds Balance of heating and cooling processes helps to set the temperature in the gas. This then sets the minimum internal pressure in a core
Lecture 8: Cloud Stability Heating & Cooling in Molecular Clouds Balance of heating and cooling processes helps to set the temperature in the gas. This then sets the minimum internal pressure in a core
Selection and Determination of Tubing and Production Casing Sizes
 CHAPTER 3 Selection and Determination of Tubing and Production Casing Sizes OUTLINE 3.1 Overview 3.2 Overview of Nodal Analysis 3.3 Selection and Determination of Tubing and Production Casing Sizes for
CHAPTER 3 Selection and Determination of Tubing and Production Casing Sizes OUTLINE 3.1 Overview 3.2 Overview of Nodal Analysis 3.3 Selection and Determination of Tubing and Production Casing Sizes for
Fluids and Solids: Fundamentals
 Fluids and Solids: Fundamentals We normally recognize three states of matter: solid; liquid and gas. However, liquid and gas are both fluids: in contrast to solids they lack the ability to resist deformation.
Fluids and Solids: Fundamentals We normally recognize three states of matter: solid; liquid and gas. However, liquid and gas are both fluids: in contrast to solids they lack the ability to resist deformation.
Physics 9e/Cutnell. correlated to the. College Board AP Physics 1 Course Objectives
 Physics 9e/Cutnell correlated to the College Board AP Physics 1 Course Objectives Big Idea 1: Objects and systems have properties such as mass and charge. Systems may have internal structure. Enduring
Physics 9e/Cutnell correlated to the College Board AP Physics 1 Course Objectives Big Idea 1: Objects and systems have properties such as mass and charge. Systems may have internal structure. Enduring
Heat Transfer and Energy
 What is Heat? Heat Transfer and Energy Heat is Energy in Transit. Recall the First law from Thermodynamics. U = Q - W What did we mean by all the terms? What is U? What is Q? What is W? What is Heat Transfer?
What is Heat? Heat Transfer and Energy Heat is Energy in Transit. Recall the First law from Thermodynamics. U = Q - W What did we mean by all the terms? What is U? What is Q? What is W? What is Heat Transfer?
11.1. Objectives. Component Form of a Vector. Component Form of a Vector. Component Form of a Vector. Vectors and the Geometry of Space
 11 Vectors and the Geometry of Space 11.1 Vectors in the Plane Copyright Cengage Learning. All rights reserved. Copyright Cengage Learning. All rights reserved. 2 Objectives! Write the component form of
11 Vectors and the Geometry of Space 11.1 Vectors in the Plane Copyright Cengage Learning. All rights reserved. Copyright Cengage Learning. All rights reserved. 2 Objectives! Write the component form of
7. DYNAMIC LIGHT SCATTERING 7.1 First order temporal autocorrelation function.
 7. DYNAMIC LIGHT SCATTERING 7. First order temporal autocorrelation function. Dynamic light scattering (DLS) studies the properties of inhomogeneous and dynamic media. A generic situation is illustrated
7. DYNAMIC LIGHT SCATTERING 7. First order temporal autocorrelation function. Dynamic light scattering (DLS) studies the properties of inhomogeneous and dynamic media. A generic situation is illustrated
The Ideal Gas Law. Gas Constant. Applications of the Gas law. P = ρ R T. Lecture 2: Atmospheric Thermodynamics
 Lecture 2: Atmospheric Thermodynamics Ideal Gas Law (Equation of State) Hydrostatic Balance Heat and Temperature Conduction, Convection, Radiation Latent Heating Adiabatic Process Lapse Rate and Stability
Lecture 2: Atmospheric Thermodynamics Ideal Gas Law (Equation of State) Hydrostatic Balance Heat and Temperature Conduction, Convection, Radiation Latent Heating Adiabatic Process Lapse Rate and Stability
Common Core Unit Summary Grades 6 to 8
 Common Core Unit Summary Grades 6 to 8 Grade 8: Unit 1: Congruence and Similarity- 8G1-8G5 rotations reflections and translations,( RRT=congruence) understand congruence of 2 d figures after RRT Dilations
Common Core Unit Summary Grades 6 to 8 Grade 8: Unit 1: Congruence and Similarity- 8G1-8G5 rotations reflections and translations,( RRT=congruence) understand congruence of 2 d figures after RRT Dilations
Part IV. Conclusions
 Part IV Conclusions 189 Chapter 9 Conclusions and Future Work CFD studies of premixed laminar and turbulent combustion dynamics have been conducted. These studies were aimed at explaining physical phenomena
Part IV Conclusions 189 Chapter 9 Conclusions and Future Work CFD studies of premixed laminar and turbulent combustion dynamics have been conducted. These studies were aimed at explaining physical phenomena
Chapter 28 Fluid Dynamics
 Chapter 28 Fluid Dynamics 28.1 Ideal Fluids... 1 28.2 Velocity Vector Field... 1 28.3 Mass Continuity Equation... 3 28.4 Bernoulli s Principle... 4 28.5 Worked Examples: Bernoulli s Equation... 7 Example
Chapter 28 Fluid Dynamics 28.1 Ideal Fluids... 1 28.2 Velocity Vector Field... 1 28.3 Mass Continuity Equation... 3 28.4 Bernoulli s Principle... 4 28.5 Worked Examples: Bernoulli s Equation... 7 Example
Physics 5D - Nov 18, 2013
 Physics 5D - Nov 18, 2013 30 Midterm Scores B } Number of Scores 25 20 15 10 5 F D C } A- A A + 0 0-59.9 60-64.9 65-69.9 70-74.9 75-79.9 80-84.9 Percent Range (%) The two problems with the fewest correct
Physics 5D - Nov 18, 2013 30 Midterm Scores B } Number of Scores 25 20 15 10 5 F D C } A- A A + 0 0-59.9 60-64.9 65-69.9 70-74.9 75-79.9 80-84.9 Percent Range (%) The two problems with the fewest correct
4. Introduction to Heat & Mass Transfer
 4. Introduction to Heat & Mass Transfer This section will cover the following concepts: A rudimentary introduction to mass transfer. Mass transfer from a molecular point of view. Fundamental similarity
4. Introduction to Heat & Mass Transfer This section will cover the following concepts: A rudimentary introduction to mass transfer. Mass transfer from a molecular point of view. Fundamental similarity
Numerical Simulation of Temperature and Stress Fields in the Rock Heating Experiment
 Numerical Simulation of Temperature and Stress Fields in the Rock Heating Experiment Author P. Rálek 1*, M. Hokr 1 1 Technical University of Liberec, Liberec, Czech Republic *Corresponding author: petr.ralek@tul.cz
Numerical Simulation of Temperature and Stress Fields in the Rock Heating Experiment Author P. Rálek 1*, M. Hokr 1 1 Technical University of Liberec, Liberec, Czech Republic *Corresponding author: petr.ralek@tul.cz
