Charles C Wykoff, David M Brown, Maria E Maldonado, Daniel E Croft. Clinical science
|
|
|
- Lydia Madeline Payne
- 8 years ago
- Views:
Transcription
1 Aflibercept treatment for patients with exudative age-related macular degeneration who were incomplete responders to multiple ranibizumab injections (TURF trial) Charles C Wykoff, David M Brown, Maria E Maldonado, Daniel E Croft Clinical science Retina Consultants of Houston, Weill Cornell Medical College, Houston Methodist Hospital, Houston, Texas, USA Correspondence to Dr Charles C Wykoff, Retina Consultants of Houston, Weill Cornell Medical College, Houston Methodist Hospital, 6560 Fannin, Suite 750, Houston, Texas 77030, USA; ccwmd@houstonretina.com Preliminary data from this study was presented at ARVO 2013, Seattle, WA. Received 3 December 2013 Revised 26 December 2013 Accepted 18 January 2014 Published Online First 11 February 2014 Open Access Scan to access more free content To cite: Wykoff CC, Brown DM, Maldonado ME, et al. Br J Ophthalmol 2014;98: ABSTRACT Aim To determine the efficacy of 2.0 mg aflibercept in the management of patients with recalcitrant exudative age-related macular degeneration (AMD). Methods In this prospective, open-label, single-arm clinical trial, patients were seen monthly and given mandatory 2.0 mg aflibercept at baseline, months 1, 2 and 4. Pro re nata (PRN) retreatment at months 3 and 5 was performed upon evidence of disease on spectral domain-optical coherence tomography (SD-OCT). End point at month 6: mean change in Early Treatment Diabetic Retinopathy Study best corrected visual acuity (ETDRS BCVA) and central subfield thickness (CST), mean number of aflibercept injections, percentage of PRN injections required, patients with no fluid on SD- OCT and patients losing >15 letters. Results At baseline, 46 patients with a mean of 42 prior antivascular endothelial growth factor-a (anti- VEGF) intravitreal treatments had a mean of 74.2 letters (Snellen equivalent 20/32) and mean CST of 347 mm. ETDRS letters remained stable throughout the trial; at month 6, mean BCVA change was +0.2 letters (range 10 to +13, p=0.71). Anatomically, mean CST improved significantly from baseline at each study visit including 23.6 mm at month 1 and 27.3 mm at month 6 ( p=0.018). Seventy-one of 90 (79%) possible PRN injections were required and a mean of 5.6 aflibercept injections out of the maximum six were administered. Ten of 45 (22%) patients had no retinal fluid on SD-OCT at month 6. No patient lost >15 letters. Conclusions Aflibercept 2.0 mg treatment maintained mean visual acuity improvements previously achieved with high-dose 2.0-mg ranibizumab injections in recalcitrant wet AMD patients. Aflibercept 2.0 mg treatment led to significant anatomic improvement and was required monthly in most patients. Clinical Trials Registration FDA IND# NCT Trial Details IND 12462, NCT clinicaltrials.gov/show/nct INTRODUCTION Neovascular age-related macular degeneration (AMD) is a leading cause of vision loss around the world. 1 Pharmaceutical agents that block vascular endothelial growth factor-a (VEGF) have revolutionised the management of neovascular AMD and most exudative retinal diseases. Nevertheless, many eyes treated with monthly dosing of ranibizumab (Lucentis, Genentech, South San Francisco, California, USA), 23 bevacizumab (Avastin, Genentech, South San Francisco, California, USA), 4 or aflibercept (Eylea, Regeneron, Tarrytown, New Jersey) 5 manifest recalcitrant fluid. For example, in the CATT trial (Comparison of AMD Treatment Trial), despite monthly treatment with anti-vegf agents for 2 years, 51.5% of patients treated with ranibuzumab and 67.4% of patients given bevacizumab showed evidence of persistent fluidontime-domain optical coherence tomography (OCT). 4 Similarly, in the VIEW1 and VIEW2 trials (VEGF Trap-Eye: Investigation of Efficacy and Safety in Wet AMD), between 27.6% and 32.3% of patients had evidence of persistent intraretinal or subretinal fluid at the primary end-point of 1 year despite 2.0 mg aflibercept treatment. 5 Such residual intraretinal or subretinal fluid likely limits optimal visual improvement, 6 and patients with recalcitrant wet AMD represent a substantial clinical burden. Evidence suggests that some of these patients may benefit from a higher dose of anti-vegf medication or switching to a different pharmacologic agent. The SAVE trial (Super-dose Anti-VEGF (SAVE) Trial: 2.0 mg Intravitreal Ranibizumab for Recalcitrant Neovascular Age-Related Macular Degeneration) demonstrated significant visual and anatomic gains in recalcitrant wet AMD eyes at both 1 and 2 years of treatment. 7 8 However, pro re nata (PRN) retreatments were required at almost every monthly visit in these aggressive wet AMD eyes, and retinal fluid was still present in 70% (45/ 64) of patients at the end of 2 years. 8 The current study aimed to determine if 2.0 mg aflibercept could maintain or even improve upon the visual acuity and anatomic gains of the SAVE trial for these well-characterised recalcitrant exudative AMD eyes. MATERIALS AND METHODS This study was a prospective, multicentre, openlabel, single-arm clinical trial (aflibercept for subjects with exudative AMD who were incomplete responders to multiple Ranibizumab anti-vegf injections (TURF trial); Food and Drug Administration (FDA) Investigational New Drug #12462). Inclusion criteria were that only patients who completed the 2-year, prospective SAVE trial (NCT ) in which recalcitrant wet AMD eyes were treated with 2.0 mg ranibizumab 7 9 were eligible. TURF sample size was determined by enrolling all consenting patients who enrolled directly into TURF immediately following completion of the SAVE trial. There was a protocol-mandated 28-day minimum wash-out before enrolment in Wykoff CC, et al. Br J Ophthalmol 2014;98: doi: /bjophthalmol
2 the TURF study in which patients could not have received any anti-vegf medication. No patient had received prior aflibercept treatment. Exclusion criteria included significant subretinal fibrosis or geographic atrophy involving the fovea. After obtaining institutional review board (IRB) approval of the study protocol and consent, appropriate patients seen at Retina Consultants of Houston were identified, provided with informed consent documents, and enrolled. Data was collected at Retina Consultants of Houston in the Texas Medical Center (6560 Fannin, Suite 750, Houston, Texas) and in The Woodlands, Texas (17350 St Luke s Way, Suite 120). At all study visits, subjects underwent best corrected Early Treatment Diabetic Retinopathy Study best corrected visual acuity (ETDRS BCVA) and comprehensive ophthalmic examination including applanation tonometry, slit-lamp evaluation and dilated binocular indirect ophthalmoscopy. Fundus photography and fluorescein angiography were performed at baseline, month 3 and month 6. Spectral domain OCT (SD-OCT) was performed at each visit using the Heidelberg Spectralis HRA+OCT (Spectralis; Heidelberg Engineering, Heidelberg, Germany). All patients received 0.05 ml open-label intravitreal injections of 2.0 mg aflibercept administered every 28 days for the first 3 months (baseline, month 1 and month 2), one mandatory dose at month 4 and, as needed, doses at months 3 and 5. PRN retreatments at months 3 and 5 were performed in the presence of intraretinal or subretinal fluid on SD-OCT or if BCVA decreased >5 letters from the previous visit. Sterile surgical technique was applied for each intravitreal injection. Patients self-administered topical antimicrobials four times daily for 3 days prior to treatment. After topical anaesthesia, the periocular skin, eyelid and eyelashes were treated with 10% povidone iodine swabs, and 5% povidone iodine ophthalmic solution was applied to the conjunctival surface. Following intravitreal injection, finger-counting testing was performed and indirect ophthalmoscopy was performed to confirm central retinal artery perfusion. Key outcome measures included mean change in ETDRS BCVA from baseline, mean change in central subfield thickness (CST), mean number of 2.0 mg intravitreal aflibercept injections administered, percentage of PRN injections required, percentage of patients with no intraretinal, subretinal, or subretinal pigment epithelial (RPE) fluid on SD-OCT at month 6 and percentage of patients who lost >15 letters BCVA. All SD-OCT segmentations were manually confirmed and corrected as needed to follow the internal limiting membrane and Bruch s membrane prior to computation of SD-OCT change maps and CST (MEM, CCW). Statistical comparisons were performed with paired Student t tests where appropriate. RESULTS Patient characteristics Forty-six patients enrolled directly from the SAVE trial into the TURF trial between April and September Baseline demographics and clinical findings are described in table 1. Forty-five of 46 (98%) patients completed the 6-month trial. At baseline, patients had received a mean of 42 (range 19 67) prior anti-vegf intravitreal treatments including a mean of 21 (range 13 24) prior 2.0 mg ranibizumab treatments out of a maximum 24 during the immediately preceding 24 months of the SAVE trial. Median time from prior anti-vegf treatment to the first aflibercept treatment was 28 days (mean 33.3, range 28 to 68 days). Table 1 Baseline demographics Baseline patient demographics Patients 46 Sex (male/female) 22/24 Age m(r) 77.8 (55 95) Prior anti-vegf injections m(r) 42 (19 67) 2.0 mg ranibizumab injections m(r) 21 (13 24) Washout period in days m(r) 33 (28 68) ETDRS BCVA m(r) 74.2 (41 92) Snellen equivalent (r) (20/100 20/16) SD OCT CST μm m(r) 347 ( ) Intraretinal fluid n(%) 17 (37) Subretinal fluid n(%) 21 (46) Sub-RPE fluid n(%) 18 (39) No intraretinal, subretinal, or Sub-RPE fluid n(%) 8 (17) anti-vegf, anti-vascular endothelial growth factor-a; CST, central subfield thickness; ETDRS BCVA, Early Treatment Diabetic Retinopathy Study best corrected visual acuity; m, mean; OCT, optical coherence tomography; r, range; sub-rpe, subretinal pigment epithelial. Visual outcomes At enrolment, mean BCVA was 74.2 ETDRS letters (Snellen equivalent 20/32, range 41 91, 20/100 20/16). BCVA remained stable throughout the 6-month trial with no significant fluctuation; at month 6, mean change was +0.2 ETDRS letters (range 10 to +13, p=0.71) (figure 1). At 6 months, 4 eyes (9%) improved by 5 ETDRS letters and 1 eye (2%) improved by 10 ETDRS letters, gaining 13 letters within the first month of treatment; PRN retreatment was required at both PRN visits in 3 of 4 of these patients, while 1 patient received neither PRN retreatment. At month 6, 4 eyes (9%) lost 5 letters or more with 1 eye (2%) receiving neither PRN retreatment and losing 10 letters associated with collapse of a pigment epithelial detachment (PED) and progression of geographic atrophy (figure 2A); PRN retreatment was required at both PRN visits in 2 of 4 of these patients, and 2 patients received neither PRN retreatment. No patient lost >15 letters BCVA. Anatomic outcomes At enrolment, mean CST was 347 μm (range mm). Mean CST improved significantly from baseline at all study visits including 23.6 mm at month 1 ( 5%, range 384 to +32 mm), and 27.3 mm at month 6 ( 6%, range 381 to +59 mm, p=0.018) (figure 3). CST decreased >10% in 9 of 46 patients (19.6%) at month 3, and in 7 of 45 patients (15.6%) at month 6. For these eyes, the mean decrease in CST at month 6 was 149 mm ( 31%, range 48 to 381 mm). Of the 7 patients with CST decrease >10% at month 6, 4 had complete, or near complete, resolution of PED (figure 2B); 94% of the decrease in CST in these patients was realised within the first 2 months of aflibercept treatment. At month 3, no patient gained >10% CST, but at month 6, 2 patients gained >10% CST, gaining either significant subretinal (60 mm) or intraretinal (92 mm) fluid after not receiving a PRN injection. In total, 71 of 90 (79%) possible PRN injections were administered and a mean of 5.6 aflibercept injections out of the minimum 4 and maximum 6 total injections were administered. PRN retreatment was required at both PRN visits in 33 of 46 (72%) patients due to SD-OCT findings, and neither of the PRN injections were given in 8 of 46 (17%) patients. At months 952 Wykoff CC, et al. Br J Ophthalmol 2014;98: doi: /bjophthalmol
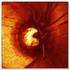
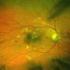
3 Figure 1 Change in mean best-corrected visual acuity (best corrected visual acuity (BCVA); Early Treatment Diabetic Retinopathy Study (ETDRS) letters) over 6 months with standard error (SE) bars. 3 and 6, 17 (37%) and 10 (22%) patients had no intraretinal, subretinal, or sub-rpe fluid, respectively. Adverse events Ocular and systemic adverse events are reported in table 2. There were no cases of endophthalmitis, intraocular inflammation, new subretinal haemorrhage, or traumatic cataract. In total, three patients were noted to have increased geographic atrophy (3/46, 7%). No systemic arterial thromboembolic events were identified. One (1/46, 2%) patient died at month 5 related to complications of acute onset leukaemia. DISCUSSION Eyes with recalcitrant exudative AMD are a substantial clinical burden, representing approximately a quarter to one-third of all new wet AMD eyes. In such patients, 2.0 mg ranibizumab, a fourfold higher dose than the FDA approved 0.5 mg dose for wet AMD management, appears capable of additional anatomic and visual benefit based on the SAVE trial outcomes at month 3, year 1 and year 2, 7 9 as well as the LAST study (evaluation of high-dose ranibizumab (2.0 mg) in the management of AMD in patients with persistent/recurrent macular fluid). 10 In the HARBOR study, 2.0 mg ranibizumab demonstrated no clinical advantage over the 0.5 mg ranibizumab dosing in treatment naive eyes 11 and Genentech discontinued further development and clinical trials of the 2.0 mg dose. As the SAVE cohort had demonstrated sustained clinical benefit from the higher dose, we were reticent to switch these patients back to 0.5 mg ranibizumab. With FDA approval of aflibercept for therapeutic use, the TURF trial was designed to assess if the anatomic and BCVA Figure 2 Case examples. Baseline late-phase fluorescein angiograms (FA) with associated spectral domain optical coherence tomography (SD-OCT) orientations (white dashed line) followed by sequential SD-OCT images at baseline (left), 1 month (centre) and 6 months (right). (A) flattening of pigment epithelial detachment (PED) from baseline associated with retinal pigment epithelial and outer retinal atrophy. (B) flattening of PED with residual subretinal fluid under the fovea at month 6. Wykoff CC, et al. Br J Ophthalmol 2014;98: doi: /bjophthalmol
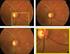
4 Figure 3 Change in central subfield thickness (spectral domain optical coherence tomography (SD-OCT)) over 6 months with standard error (SE) bars. Central subfield thickness was evaluated monthly as well as 7 days after the first aflibercept treatment. gains could be maintained with this alternative pharmacologic agent. The TURF trial consisted entirely of patients exiting the SAVE trial, and demonstrates that commercially available 2.0 mg aflibercept treatment maintained the mean visual gains attained with 2.0 mg ranibizumab treatment and led to significant mean anatomic improvement. These anatomic gains were primarily driven by a minority of patients (20%, 9 of 45), many with dramatic flattening of PED. Despite no mean change in BCVA including all TURF patients, one patient gained 13 ETDRS letters within 1 month of aflibercept initiation, a patient who required both PRN retreatments. The FDA approved labelling for aflibercept use in the management of wet AMD includes two options: 2 mg every 8 weeks after 3 initial monthly doses or 2 mg monthly. While both treatment regimens led to clinically equivalent mean visual outcomes in treatment-naive patients, there is evidence that continued monthly therapy after the first 3 monthly doses translates into better visual outcomes in some patients, particularly those with recalcitrant exudative disease activity. For example, when eyes with persistent macular oedema during each of the first 4 visits in VIEW1 and VIEW2 were considered, continued monthly aflibercept achieved superior outcomes compared with every 8-week dosing. 12 The current TURF trial employed every other month dosing after the first 3-monthly doses, but was designed to permit PRN dosing at the intervening months. Supporting Table 2 Ocular and serious systemic adverse events (AEs) Ocular adverse event (%) Patients with ocular AEs 6 (13) Cataract progression 3 (7) Geographic atrophy progression 3 (7) Serious adverse events (%) Patients with SAEs 9 (20) Total SAEs 11 (24) Death 1 (2) Atrial fibrillation 1 (2) Squamous cell carcinoma 2 (4) Upper respiratory tract infection 5 (11) Urinary tract infection 2 (4) the observation that aflibercept does not maintain maximal retinal deturgesence for 2 months in many patients, and indicative of the recalcitrant exudative nature of the study eyes in TURF, 72% of eyes required retreatment at both PRN visits, and 79% of PRN retreatments were required. Furthermore, the only two patients who experienced a >10% gain in CST during the TURF trial both did not receive a PRN treatment the month prior. In the treatment-naive eyes included in the VIEW1 and VIEW2 trials, aflibercept and ranibizumab treatment resulted in clinically equivalent visual outcomes. 5 While both pharmacologic agents neutralise VEGF-A, aflibercept has a distinct mechanism of action and also inhibits VEGF-B and placental growth factor. 13 As such, eyes with recalcitrant exudative AMD may respond differently to these medications. Indeed, many analyses have recently reported positive anatomic benefit, and in some cases visual benefit, with aflibercept in recalcitrant wet AMD eyes previously treated with ranbizumab or bevacizumab The strength of this study is its prospective design involving a well-defined patient cohort who had received rigorous previous treatment with anti-vegf therapies, as well as its standardised treatment and follow-up intervals. The limitations of this trial include the lack of a control group, relatively small sample size, and the limited duration of 6 months. This study provides evidence that aflibercept treatment may be anatomically valuable in some recalcitrant exudative AMD eyes, while maintaining prior visual gains. Such value may be apparent relatively quickly after initiation of aflibercept treatment. However, a substantial majority of the patients in this prospective trial required monthly aflibercept dosing to achieve maximal anatomic and visual benefit. Longer-term data will be valuable to guide the ongoing management of patients with recalcitrant wet AMD. Contributors CCW: concept, design, PI, writing of the manuscript (guarantor); DMB: concept, design, revision of the manuscript; MEM: data collection, analysis, writing of the manuscript. DEC: data collection, analysis, writing of the manuscript. Funding Research grant from Regeneron Pharmaceuticals. The funding organisation had no role in the design or conduct of this research. Competing interests Yes. ICMJE conflicts of interest forms have been attached. Ethics approval Sterling IRB. Provenance and peer review Not commissioned; externally peer reviewed. 954 Wykoff CC, et al. Br J Ophthalmol 2014;98: doi: /bjophthalmol
5 Open Access This is an Open Access article distributed in accordance with the Creative Commons Attribution Non Commercial (CC BY-NC 3.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: licenses/by-nc/3.0/ REFERENCES 1 Jager RD, Mieler WF, Miller JW. Age-related macular degeneration. N Engl J Med 2008;358: Brown DM, Kaiser PK, Michels M, et al. Ranibizumab versus verteporfin for neovascular age-related macular degeneration. N Engl J Med 2006;355: Rosenfeld PJ, Brown DM, Heier JS, et al. Ranibizumab for neovascular age-related macular degeneration. N Engl J Med 2006;355: Martin DF, Maguire MG, Fine SL, et al. Ranibizumab and Bevacizumab for treatment of Neovascular age-related macular degeneration: two-year results. Ophthalmology 2012;119: Heier JS, Brown DM, Chong V, et al. Intravitreal Aflibercept (VEGF Trap-Eye) in Wet Age-related Macular Degeneration. Ophthalmology 2012;119: Brown DM, Tuomi L, Shapiro H. Anatomical measures as predictors of visual outcomes in ranibizumab-treated eyes with neovascular age-related macular degeneration. Retina 2013;33: Wykoff CC, Brown DM, Chen E, et al. SAVE (Super-dose Anti-VEGF) trial: 2.0 mg Ranibizumab for recalcitrant Neovascular age-related macular degeneration: 1-Year results. Ophthalmic Surg Lasers Imaging Retin 2013;44: Wykoff CC, Brown DM, Croft DE, et al. Two year SAVE outcomes: 2.0 mg ranibizumab for recalcitrant neovascular AMD. Ophthalmology 2013;120: e Brown DM, Chen E, Mariani A, et al. Super-dose anti-vegf (SAVE) trial: 2.0 mg intravitreal ranibizumab for recalcitrant neovascular macular degeneration-primary end point. Ophthalmology 2013;120: Fung AT, Kumar N, Vance SK, et al. Pilot study to evaluate the role of high-dose ranibizumab 2.0 mg in the management of neovascular age-related macular degeneration in patients with persistent/recurrent macular fluid <30 days following treatment with intravitreal anti-vegf therapy (the LAST Study). Eye (Lond) 2012;26: Busbee BG, Ho AC, Brown DM, et al. Twelve-month efficacy and safety of 0.5 mg or 2.0 mg ranibizumab in patients with subfoveal neovascular age-related macular degeneration. Ophthalmology 2013;120: Jaffe G. The Effect of Early, Persistent Fluid on Subsequent Visual Acuity in the VIEW 1 and VIEW 2 Studies of Neovascular AMD. American Societey of Retina Specialists Toronto, Canada, August 25th Nguyen QD, Shah SM, Browning DJ, et al. A phase I study of intravitreal vascular endothelial growth factor trap-eye in patients with neovascular age-related macular degeneration. Ophthalmology 2009;116: e Chang AA, Li H, Broadhead GK, et al. Intravitreal aflibercept for treatment-resistant neovascular age-related macular degeneration. Ophthalmology 2014;121: Bakall B, Folk JC, Boldt HC, et al. Aflibercept therapy for exudative age-related macular degeneration resistant to bevacizumab and ranibizumab. Am J Ophthalmol 2013;156: Yonekawa Y, Andreoli C, Miller JB, et al. Conversion to aflibercept for chronic refractory or recurrent neovacular age-related macular degeneration. Am J Ophthalmol 2013;156: Cho H, Shah CP, Weber M, et al. Aflibercept for exudative AMD with persistent fluid on ranibizumab and/or bevacizumab. Br J Ophthalmol 2013;97: Schachat AP. Switching anti-vascular endothelial growth factor therapy for neovascular age-related macular degeneration. American J Ophthalmol 2013;156:1 2 e1. Wykoff CC, et al. Br J Ophthalmol 2014;98: doi: /bjophthalmol
IR Conference Call on EYLEA (aflibercept) Injection in Diabetic Macular Edema. September 28, 2013
 IR Conference Call on EYLEA (aflibercept) Injection in Diabetic Macular Edema September 28, 2013 Safe Harbor Statement This presentation includes forward-looking statements that involve risks and uncertainties
IR Conference Call on EYLEA (aflibercept) Injection in Diabetic Macular Edema September 28, 2013 Safe Harbor Statement This presentation includes forward-looking statements that involve risks and uncertainties
12-month results of the standardised combination therapy for diabetic macular oedema: intravitreal bevacizumab and navigated retinal photocoagulation
 BJO Online First, published on April 10, 2014 as 10.1136/bjophthalmol-2013-304488 Clinical science 12-month results of the standardised combination therapy for diabetic macular oedema: intravitreal bevacizumab
BJO Online First, published on April 10, 2014 as 10.1136/bjophthalmol-2013-304488 Clinical science 12-month results of the standardised combination therapy for diabetic macular oedema: intravitreal bevacizumab
FDA approves Lucentis (ranibizumab injection) for treatment of diabetic macular edema
 Media Release Basel, 13 August 2012 FDA approves Lucentis (ranibizumab injection) for treatment of diabetic macular edema First major treatment advance in more than 25 years for sight-threatening condition
Media Release Basel, 13 August 2012 FDA approves Lucentis (ranibizumab injection) for treatment of diabetic macular edema First major treatment advance in more than 25 years for sight-threatening condition
An Informational Guide to CENTRAL RETINAL VEIN OCCLUSION
 Science of CRVO www.scienceofcrvo.org An Informational Guide to CENTRAL RETINAL VEIN OCCLUSION This brochure will guide you in understanding CRVO and the treatment options available to prevent vision loss.
Science of CRVO www.scienceofcrvo.org An Informational Guide to CENTRAL RETINAL VEIN OCCLUSION This brochure will guide you in understanding CRVO and the treatment options available to prevent vision loss.
ACTVE INTERVENTIONAL CLINICAL TRIALS - 03/01/2015. Principal Investigator Study Title and Sponsor Study Description
 Cornea and External Disease Service Li, Jennifer A Prospective, Multicenter Post-Approval Study (PAS) of VisionCare s Implantable Miniature Telescope (by Dr. Isaac Lipshitz) in Patients with Bilateral
Cornea and External Disease Service Li, Jennifer A Prospective, Multicenter Post-Approval Study (PAS) of VisionCare s Implantable Miniature Telescope (by Dr. Isaac Lipshitz) in Patients with Bilateral
Completed Clinical Research Trials Prema Abraham, M.D.
 Completed Clinical Research Trials Prema Abraham, M.D. AMD Studies 2010 Present Principal Investigator: A Phase 2, randomized, double-masked, controlled trial to establish the safety and efficacy of intravitreous
Completed Clinical Research Trials Prema Abraham, M.D. AMD Studies 2010 Present Principal Investigator: A Phase 2, randomized, double-masked, controlled trial to establish the safety and efficacy of intravitreous
ICG IMAGING FOR EXUDATIVE ARMD
 ICG IMAGING FOR EXUDATIVE ARMD MARK H. NELSON, MD MBA WINSTON-SALEM, NC ASRS INSTRUCTION COURSE AUGUST 24, 2011 FINANCIAL DISCLOSURE Novartis Pharmaceutics, Incorporated Consultant, Grant Recipient Heidelberg
ICG IMAGING FOR EXUDATIVE ARMD MARK H. NELSON, MD MBA WINSTON-SALEM, NC ASRS INSTRUCTION COURSE AUGUST 24, 2011 FINANCIAL DISCLOSURE Novartis Pharmaceutics, Incorporated Consultant, Grant Recipient Heidelberg
NAVILAS Laser System Focal Laser Treatment for Diabetic Macular Edema - One Year Results of a Case Series
 Send Orders for Reprints to reprints@benthamscience.net 48 The Open Ophthalmology Journal, 2013, 7, 48-53 Open Access NAVILAS Laser System Focal Laser Treatment for Diabetic Macular Edema - One Year Results
Send Orders for Reprints to reprints@benthamscience.net 48 The Open Ophthalmology Journal, 2013, 7, 48-53 Open Access NAVILAS Laser System Focal Laser Treatment for Diabetic Macular Edema - One Year Results
1981-1982 Ophthalmic Consultants Northwest, Seattle, Washington Retinal and vitreous consultation and surgery
 EDUCATION Richard S. Munsen Jr., MD University of Washington Eye Institute HMC Box 359608, 325 Ninth Ave, Seattle WA 98104-2499 (206) 744-2020 for patient appointments, Fax (206) 897-4320 Academic phone
EDUCATION Richard S. Munsen Jr., MD University of Washington Eye Institute HMC Box 359608, 325 Ninth Ave, Seattle WA 98104-2499 (206) 744-2020 for patient appointments, Fax (206) 897-4320 Academic phone
Retina InSight Volume 1 Issue 1 Fall 2014
 Retina InSight Volume 1 Issue 1 Fall 2014 Welcome to our GHRRF Newsletter! We are passionate about fighting blindness through cutting edge research. The purpose of this newsletter is to share information
Retina InSight Volume 1 Issue 1 Fall 2014 Welcome to our GHRRF Newsletter! We are passionate about fighting blindness through cutting edge research. The purpose of this newsletter is to share information
Dexamethasone intravitreal implant for the treatment of macular oedema secondary to retinal vein occlusion
 Dexamethasone intravitreal implant for the treatment of macular oedema secondary to retinal vein occlusion Issued: July 2011 guidance.nice.org.uk/ta229 NICE has accredited the process used by the Centre
Dexamethasone intravitreal implant for the treatment of macular oedema secondary to retinal vein occlusion Issued: July 2011 guidance.nice.org.uk/ta229 NICE has accredited the process used by the Centre
Trends in Ophthalmology a Market Perspective. Matthias Karl, Michael Kempe
 Trends in Ophthalmology a Market Perspective Matthias Karl, Michael Kempe 30.04.2013 Agenda 1 2 3 4 Role of Optical Technologies in Ophthalmology Demands of the Market - Trends in the Application Implications
Trends in Ophthalmology a Market Perspective Matthias Karl, Michael Kempe 30.04.2013 Agenda 1 2 3 4 Role of Optical Technologies in Ophthalmology Demands of the Market - Trends in the Application Implications
Why Does Anti VEGF Treatment Fail in Age Related Macular Degeneration (AMD)
 Original Article Why Does Anti VEGF Treatment Fail in Age Related Macular Degeneration (AMD) Manoj S. MS Aim: To evaluate eyes with AMD that responded poorly to anti VEGF therapy and study reasons for
Original Article Why Does Anti VEGF Treatment Fail in Age Related Macular Degeneration (AMD) Manoj S. MS Aim: To evaluate eyes with AMD that responded poorly to anti VEGF therapy and study reasons for
Long-term Outcomes of Ranibizumab Therapy for Diabetic Macular Edema: The 36-Month Results from Two Phase III Trials
 Long-term Outcomes of Therapy for Diabetic Macular Edema: The 36-Month Results from Two Phase III Trials RISE and RIDE David M. Brown, MD, 1,x Quan Dong Nguyen, MD, MSc, 2,x Dennis M. Marcus, MD, 3 David
Long-term Outcomes of Therapy for Diabetic Macular Edema: The 36-Month Results from Two Phase III Trials RISE and RIDE David M. Brown, MD, 1,x Quan Dong Nguyen, MD, MSc, 2,x Dennis M. Marcus, MD, 3 David
Avastin (Bevacizumab) Intravitreal Injection
 Avastin (Bevacizumab) Intravitreal Injection This handout describes how Avastin may be used to treat wet age related macular degeneration (AMD) or macular edema due to retinal vascular disease such as
Avastin (Bevacizumab) Intravitreal Injection This handout describes how Avastin may be used to treat wet age related macular degeneration (AMD) or macular edema due to retinal vascular disease such as
Commissioning better eye care
 Commissioning better eye care Clinical commissioning guidance from The College of Optometrists and The Royal College of Ophthalmologists Age-related macular degeneration Version: 1 Published: 25 Nov 2013
Commissioning better eye care Clinical commissioning guidance from The College of Optometrists and The Royal College of Ophthalmologists Age-related macular degeneration Version: 1 Published: 25 Nov 2013
Age- Related Macular Degeneration
 Age- Related Macular Degeneration Age-Related Macular Degeneration (AMD) is the leading cause of blindness in the United States. It is caused by damage to a localized area of the central retina called
Age- Related Macular Degeneration Age-Related Macular Degeneration (AMD) is the leading cause of blindness in the United States. It is caused by damage to a localized area of the central retina called
Angiogenesis, Exudation, and Degeneration 2016. February 6, 2016 Mandarin Oriental, Miami, FL
 Angiogenesis, Exudation, and Degeneration 2016 February 6, 2016 Mandarin Oriental, Miami, FL Program Directors: Philip J. Rosenfeld, MD, PhD and Harry W. Flynn, Jr., MD Course Associate Director: Carmen
Angiogenesis, Exudation, and Degeneration 2016 February 6, 2016 Mandarin Oriental, Miami, FL Program Directors: Philip J. Rosenfeld, MD, PhD and Harry W. Flynn, Jr., MD Course Associate Director: Carmen
Ranibizumab for Macular Edema following Central Retinal Vein Occlusion
 for Macular Edema following Central Retinal Vein Occlusion Six-Month Primary End Point Results of a Phase III Study David M. Brown, MD, FACS, 1 Peter A. Campochiaro, MD, 2 Rishi P. Singh, MD, 3 Zhengrong
for Macular Edema following Central Retinal Vein Occlusion Six-Month Primary End Point Results of a Phase III Study David M. Brown, MD, FACS, 1 Peter A. Campochiaro, MD, 2 Rishi P. Singh, MD, 3 Zhengrong
Current Management of Retinal Vein Occlusion Robert A. Mittra, MD VRS Course 2015
 Central Retinal Vein Occlusion Hemi-Central Vein Occlusion Current Management of Retinal Vein Occlusion Robert A. Mittra, MD VRS Course 2015 Branch Retinal Vein Occlusion Ischemic vs. non-ischemic BRVO
Central Retinal Vein Occlusion Hemi-Central Vein Occlusion Current Management of Retinal Vein Occlusion Robert A. Mittra, MD VRS Course 2015 Branch Retinal Vein Occlusion Ischemic vs. non-ischemic BRVO
Ohr Pharmaceutical Reports Fiscal Year 2015 Financial and Business Results
 December 10, 2015 Ohr Pharmaceutical Reports Fiscal Year 2015 Financial and Business Results OHR-102 Phase 3 Program in Wet-AMD to be Initiated Upon Completion of SPA; Enroll Patients in Q1 2016 Conference
December 10, 2015 Ohr Pharmaceutical Reports Fiscal Year 2015 Financial and Business Results OHR-102 Phase 3 Program in Wet-AMD to be Initiated Upon Completion of SPA; Enroll Patients in Q1 2016 Conference
INTERNATIONAL CLINICAL DIABETIC RETINOPATHY DISEASE SEVERITY SCALE DETAILED TABLE
 Proposed Disease Severity Level No apparent Mild Non- Proliferative Moderate Nonproliferative Severe Non- Proliferative Proliferative INTERNATIONAL CLINICAL DIABETIC RETINOPATHY DISEASE SEVERITY SCALE
Proposed Disease Severity Level No apparent Mild Non- Proliferative Moderate Nonproliferative Severe Non- Proliferative Proliferative INTERNATIONAL CLINICAL DIABETIC RETINOPATHY DISEASE SEVERITY SCALE
Introduction. Stephan Michels, MD, MBA, is a professor at the University of Zurich and vice chair at Triemli Hospital in Zurich, Switzerland.
 Introduction A panel of experts gathered during the 2015 annual meeting of the American Academy of Ophthalmology to participate in a roundtable discussion regarding protocols for the treatment of diabetic
Introduction A panel of experts gathered during the 2015 annual meeting of the American Academy of Ophthalmology to participate in a roundtable discussion regarding protocols for the treatment of diabetic
Angiogenesis, Exudation, and Degeneration 2014. February 8, 2014 Mandarin Oriental, Miami, Florida
 Angiogenesis, Exudation, and Degeneration 2014 February 8, 2014 Mandarin Oriental, Miami, Florida Program Directors: Philip J. Rosenfeld, MD, PhD and Harry W. Flynn, Jr., MD Course Associate Director:
Angiogenesis, Exudation, and Degeneration 2014 February 8, 2014 Mandarin Oriental, Miami, Florida Program Directors: Philip J. Rosenfeld, MD, PhD and Harry W. Flynn, Jr., MD Course Associate Director:
Visual Disorders in Middle-Age and Elderly Patients with Diabetic Retinopathy
 Medical Care for the Elderly Visual Disorders in Middle-Age and Elderly Patients with Diabetic Retinopathy JMAJ 46(1): 27 32, 2003 Shigehiko KITANO Professor, Department of Ophthalmology, Diabetes Center,
Medical Care for the Elderly Visual Disorders in Middle-Age and Elderly Patients with Diabetic Retinopathy JMAJ 46(1): 27 32, 2003 Shigehiko KITANO Professor, Department of Ophthalmology, Diabetes Center,
Diabetes & blindness. due to DME BLINDNESS IN EUROPE
 Diabetes & blindness due to DME BLINDNESS IN EUROPE Blindness is a life-changing disability which puts a heavy strain on the daily lives of sufferers, their families, and society at large. Today, 284 million
Diabetes & blindness due to DME BLINDNESS IN EUROPE Blindness is a life-changing disability which puts a heavy strain on the daily lives of sufferers, their families, and society at large. Today, 284 million
White Paper. Ophthalmology Site Certification
 White Paper Ophthalmology Site Certification Table of Contents 1. Introduction... 3 2. What is Site Certification?... 3 3. An Example of Certification for Ophthalmology Studies... 3 4. Visual Acuity Certification...
White Paper Ophthalmology Site Certification Table of Contents 1. Introduction... 3 2. What is Site Certification?... 3 3. An Example of Certification for Ophthalmology Studies... 3 4. Visual Acuity Certification...
APRIL 8th 2016 Therapy
 APRIL 8th 2016 Therapy 09.00-10.00 SESSION 1: Age-related Macular Degeneration Moderators: S. Sadda, D. Sarraf, K. Bailey Freund, E. Souied 09.00-09.15 Prospective SD OCT Analysis of PED Associated with
APRIL 8th 2016 Therapy 09.00-10.00 SESSION 1: Age-related Macular Degeneration Moderators: S. Sadda, D. Sarraf, K. Bailey Freund, E. Souied 09.00-09.15 Prospective SD OCT Analysis of PED Associated with
Which eye conditions can avastin injections be used for?
 What is avastin? Avastin is a drug that is licensed for the treatment of a certain type of colorectal cancer but can also be used to treat certain eye conditions by being injected into the eye. Although
What is avastin? Avastin is a drug that is licensed for the treatment of a certain type of colorectal cancer but can also be used to treat certain eye conditions by being injected into the eye. Although
Photographic Assessment of Baseline Fundus Morphologic Features in the Comparison of Age-Related Macular Degeneration Treatments Trials
 Photographic Assessment of Baseline Fundus Morphologic Features in the Comparison of Age-Related Macular Degeneration Treatments Trials Juan E. Grunwald, MD, 1,2 Ebenezer Daniel, MBBS, PhD, 1,2 Gui-shuang
Photographic Assessment of Baseline Fundus Morphologic Features in the Comparison of Age-Related Macular Degeneration Treatments Trials Juan E. Grunwald, MD, 1,2 Ebenezer Daniel, MBBS, PhD, 1,2 Gui-shuang
LUCENTIS ranibizumab (rbe)
 1 LUCENTIS ranibizumab (rbe) NAME OF THE MEDICINE Active ingredient: Ranibizumab Chemical name: Immunoglobulin G1, anti-(human vascular endothelial growth factor) Fab fragment (human-mouse monoclonal rhufab
1 LUCENTIS ranibizumab (rbe) NAME OF THE MEDICINE Active ingredient: Ranibizumab Chemical name: Immunoglobulin G1, anti-(human vascular endothelial growth factor) Fab fragment (human-mouse monoclonal rhufab
Retinal Update: November 2009 - March 2010
 Retinal Update: November 2009 - March 2010 Diabetic Retinopathy Macular Degeneration: Age-related macular degeneration Macular Degeneration: Choroidal neovascularization Macular Disease: Central serous
Retinal Update: November 2009 - March 2010 Diabetic Retinopathy Macular Degeneration: Age-related macular degeneration Macular Degeneration: Choroidal neovascularization Macular Disease: Central serous
Facts about diabetic macular oedema
 Patient information medical retina services Facts about diabetic macular oedema What is diabetic macular oedema? Diabetic eye disease is a leading cause of blindness registration among working age adults
Patient information medical retina services Facts about diabetic macular oedema What is diabetic macular oedema? Diabetic eye disease is a leading cause of blindness registration among working age adults
Spectral domain OCT used to view and quantify choroidal vascular congestion in new subretinal fluid following scleral buckling.
 Spectral domain OCT used to view and quantify choroidal vascular congestion in new subretinal fluid following scleral buckling. Robert Gizicki MD, Mohamed Haji, Flavio Rezende Retina Service, Hôpital Maisonneuve-Rosemont,
Spectral domain OCT used to view and quantify choroidal vascular congestion in new subretinal fluid following scleral buckling. Robert Gizicki MD, Mohamed Haji, Flavio Rezende Retina Service, Hôpital Maisonneuve-Rosemont,
Angiogenesis, Exudation, and Degeneration 2015. February 7, 2015 Mandarin Oriental, Miami, Florida
 Angiogenesis, Exudation, and Degeneration 2015 February 7, 2015 Mandarin Oriental, Miami, Florida Program Directors: Philip J. Rosenfeld, MD, PhD and Harry W. Flynn, Jr., MD Course Associate Director:
Angiogenesis, Exudation, and Degeneration 2015 February 7, 2015 Mandarin Oriental, Miami, Florida Program Directors: Philip J. Rosenfeld, MD, PhD and Harry W. Flynn, Jr., MD Course Associate Director:
A Study of Encapsulated Cell Technology (ECT) Implant for Patients With Late Stage Retinitis Pigmentosa
 A Study of Encapsulated Cell Technology (ECT) Implant for Patients With Late Stage Retinitis Pigmentosa Purpose This study is currently patients. Verified by Neurotech Pharmaceuticals March 2007 Sponsored
A Study of Encapsulated Cell Technology (ECT) Implant for Patients With Late Stage Retinitis Pigmentosa Purpose This study is currently patients. Verified by Neurotech Pharmaceuticals March 2007 Sponsored
Affiliation : ASSOCIATION FOR RESEARCH IN VISION AND OPHTHALMOLOGY (ARVO). AMERICAN ACADEMY OF OPHTHALMOLOGY (AAO)
 Curriculum Vitae Federico Ricci, MD Name: Federico Ricci, MD. Titolo Accademico /Title: Professore Aggregato di Oftalmologia / Aggregate Professor of Ophthalmology Direttore UOSD Patologie Retiniche [Centro
Curriculum Vitae Federico Ricci, MD Name: Federico Ricci, MD. Titolo Accademico /Title: Professore Aggregato di Oftalmologia / Aggregate Professor of Ophthalmology Direttore UOSD Patologie Retiniche [Centro
Optical Coherence Tomography Imaging and Quantitative Assessment for Monitoring Dry Age-related Macular Degeneration.
 Imaging Optical Coherence Tomography Imaging and Quantitative Assessment for Monitoring Dry Age-related Macular Degeneration Albert J Augustin Head, Department of Ophthalmology, Staedtisches Klinikum Karlsruhe
Imaging Optical Coherence Tomography Imaging and Quantitative Assessment for Monitoring Dry Age-related Macular Degeneration Albert J Augustin Head, Department of Ophthalmology, Staedtisches Klinikum Karlsruhe
NC DIVISION OF SERVICES FOR THE BLIND POLICIES AND PROCEDURES VOCATIONAL REHABILITATION
 NC DIVISION OF SERVICES FOR THE BLIND POLICIES AND PROCEDURES VOCATIONAL REHABILITATION Section: E Revision History Revised 01/97; 05/03; 02/08; 04/08; 03/09; 05/09; 12/09; 01/11; 12/14 An individual is
NC DIVISION OF SERVICES FOR THE BLIND POLICIES AND PROCEDURES VOCATIONAL REHABILITATION Section: E Revision History Revised 01/97; 05/03; 02/08; 04/08; 03/09; 05/09; 12/09; 01/11; 12/14 An individual is
Peptide and Sustained Release Technology to Treat Ocular Diseases and Cancer
 Peptide and Sustained Release Technology to Treat Ocular Diseases and Cancer Jordan Green, PhD, Co-Founder and CEO Wendy Perrow, MBA, Senior Business Advisor February 11, 2015 www.asclepix.com Advantages
Peptide and Sustained Release Technology to Treat Ocular Diseases and Cancer Jordan Green, PhD, Co-Founder and CEO Wendy Perrow, MBA, Senior Business Advisor February 11, 2015 www.asclepix.com Advantages
Geographic Atrophy: The Advanced Form of Dry AMD
 Age-related macular degeneration (AMD) is a disease associated with aging that gradually destroys sharp, central vision. Central vision is needed for seeing objects clearly and for common daily tasks such
Age-related macular degeneration (AMD) is a disease associated with aging that gradually destroys sharp, central vision. Central vision is needed for seeing objects clearly and for common daily tasks such
Age-Related Macular Degeneration. K. Bailey Freund, M.D. James M. Klancnik, Jr., M.D. Lawrence A. Yannuzzi, M.D. Bruce Rosenthal, O.D.
 Age-Related Macular Degeneration K. Bailey Freund, M.D. James M. Klancnik, Jr., M.D. Lawrence A. Yannuzzi, M.D. Bruce Rosenthal, O.D. Contents Introduction 5 Anatomy of the Eye 6 The Eye 7 The Retina 9
Age-Related Macular Degeneration K. Bailey Freund, M.D. James M. Klancnik, Jr., M.D. Lawrence A. Yannuzzi, M.D. Bruce Rosenthal, O.D. Contents Introduction 5 Anatomy of the Eye 6 The Eye 7 The Retina 9
American Society of Retina Specialists Retinal Innovation Forum Abstracts
 Genentech Genentech Research in Ophthalmology Gary Sternberg, MD At Genentech we focus our research and development practice on discovering and developing new medicines for unmet medical needs. The Genentech
Genentech Genentech Research in Ophthalmology Gary Sternberg, MD At Genentech we focus our research and development practice on discovering and developing new medicines for unmet medical needs. The Genentech
Diabetic Eye Screening Revised Grading Definitions
 Diabetic Eye Screening Revised Grading Definitions Version 1.3, 1 November 2012 To provide guidance on revised grading definitions for the NHS Diabetic Eye Screening Programme Project/Category Document
Diabetic Eye Screening Revised Grading Definitions Version 1.3, 1 November 2012 To provide guidance on revised grading definitions for the NHS Diabetic Eye Screening Programme Project/Category Document
Diabetic macular edema (DME) is the primary cause of
 Retina Fully Software for Retinal Thickness in Eyes With Diabetic Macular Edema From Images Acquired by Cirrus and Spectralis Systems Joo Yong Lee, 1,2 Stephanie J. Chiu, 3 Pratul P. Srinivasan, 3 Joseph
Retina Fully Software for Retinal Thickness in Eyes With Diabetic Macular Edema From Images Acquired by Cirrus and Spectralis Systems Joo Yong Lee, 1,2 Stephanie J. Chiu, 3 Pratul P. Srinivasan, 3 Joseph
Explanation of the Procedure
 Informed Consent Cataract Surgery with Intraocular Lens Implant Please initial below indicating that you have read and understand each section Introduction The internal lens of the eye can become cloudy
Informed Consent Cataract Surgery with Intraocular Lens Implant Please initial below indicating that you have read and understand each section Introduction The internal lens of the eye can become cloudy
DIABETIC RETINOPATHY. Diabetes mellitus is an abnormality of the blood glucose metabolism due to altered
 DIABETIC RETINOPATHY Diabetes mellitus Diabetes mellitus is an abnormality of the blood glucose metabolism due to altered insulin production or activity. There are two types diabetes mellitus- Insulin
DIABETIC RETINOPATHY Diabetes mellitus Diabetes mellitus is an abnormality of the blood glucose metabolism due to altered insulin production or activity. There are two types diabetes mellitus- Insulin
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS
 ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Lucentis 10 mg/ml solution for injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION One ml contains 10 mg ranibizumab*. Each
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Lucentis 10 mg/ml solution for injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION One ml contains 10 mg ranibizumab*. Each
EFFECT OF MYOPIC LASIK ON RETINAL NERVE FIBER LAYER THICKNESS- IS IT SAFE OR UNSAFE?
 24. Glaucoma: Imaging EFFECT OF MYOPIC LASIK ON RETINAL NERVE FIBER LAYER THICKNESS- IS IT SAFE OR UNSAFE? Chief Author: Dr. Amit porwal 1 Co-Authors: Dr. Kavita Porwal 2, Dr. Puja Rai 1 1. Choithram Netralaya,
24. Glaucoma: Imaging EFFECT OF MYOPIC LASIK ON RETINAL NERVE FIBER LAYER THICKNESS- IS IT SAFE OR UNSAFE? Chief Author: Dr. Amit porwal 1 Co-Authors: Dr. Kavita Porwal 2, Dr. Puja Rai 1 1. Choithram Netralaya,
Optic Nerve Imaging. Management of Glaucoma
 Optic Nerve Imaging in the Diagnosis and Management of Glaucoma Scott D. Smith, MD, MPH Associate Professor of Clinical Ophthalmology Director, Glaucoma Service Columbia University, New York, NY Glaucoma
Optic Nerve Imaging in the Diagnosis and Management of Glaucoma Scott D. Smith, MD, MPH Associate Professor of Clinical Ophthalmology Director, Glaucoma Service Columbia University, New York, NY Glaucoma
Principles of Ophthalmic Nursing Unit Outline
 Principles of Ophthalmic Nursing Unit Outline Continuing Professional Education (CPE) Course: Application for credit worthiness in progress Prerequisites: Nil Incompatible Units: Nil Co ordinator: Amanda
Principles of Ophthalmic Nursing Unit Outline Continuing Professional Education (CPE) Course: Application for credit worthiness in progress Prerequisites: Nil Incompatible Units: Nil Co ordinator: Amanda
The Long-term Effects of Laser Photocoagulation Treatment in Patients with Diabetic Retinopathy
 The Long-term Effects of Laser Photocoagulation Treatment in Patients with Diabetic Retinopathy The Early Treatment Diabetic Retinopathy Follow-up Study Emily Y. Chew, MD, 1 Frederick L. Ferris III, MD,
The Long-term Effects of Laser Photocoagulation Treatment in Patients with Diabetic Retinopathy The Early Treatment Diabetic Retinopathy Follow-up Study Emily Y. Chew, MD, 1 Frederick L. Ferris III, MD,
Retinal Imaging Biomarkers for Early Diagnosis of Alzheimer s Disease
 Retinal Imaging Biomarkers for Early Diagnosis of Alzheimer s Disease Eleonora (Nora) Lad, MD, PhD Assistant Professor of Ophthalmology, Vitreoretinal diseases Duke Center for Macular Diseases Duke University
Retinal Imaging Biomarkers for Early Diagnosis of Alzheimer s Disease Eleonora (Nora) Lad, MD, PhD Assistant Professor of Ophthalmology, Vitreoretinal diseases Duke Center for Macular Diseases Duke University
Introduction Houston Retina Associates
 1 2 Introduction This book was written by Houston Retina Associates to provide our patients with basic knowledge about retinal anatomy, an introduction to some of the diagnostic tests and treatments used
1 2 Introduction This book was written by Houston Retina Associates to provide our patients with basic knowledge about retinal anatomy, an introduction to some of the diagnostic tests and treatments used
Re: LUMIGAN 0.03% (bimatoprost ophthalmic solution 0.03%) and LUMIGAN 0.01% (bimatoprost ophthalmic solution 0.01%)
 Allergan Ltd, 1st Floor, Marlow International, Parkway, Marlow, Bucks SL7 1YL Tel: (01628) 494444 Facsimile: (01628) 494449 Ref No: UK15-000677 Mandeep Allingham NW Surrey CCG February 18, 2015 Dear Ms
Allergan Ltd, 1st Floor, Marlow International, Parkway, Marlow, Bucks SL7 1YL Tel: (01628) 494444 Facsimile: (01628) 494449 Ref No: UK15-000677 Mandeep Allingham NW Surrey CCG February 18, 2015 Dear Ms
82 Gilbert Street, Adelaide, SA, 5000 82 Gilbert Street, Adelaide, SA, 5000 W +61 8 8104 5293 M +61 417 818 658 W +61 8 8104 5200 M +61 414 661 994
 ASX RELEASE Ellex Medical Lasers Limited (ASX:ELX) Adelaide, Australia Date: 14 September 2015 Release: Immediate Topic: Ellex releases first case study results for 2RT at major tradeshow Adelaide, Australia,
ASX RELEASE Ellex Medical Lasers Limited (ASX:ELX) Adelaide, Australia Date: 14 September 2015 Release: Immediate Topic: Ellex releases first case study results for 2RT at major tradeshow Adelaide, Australia,
EUROPEAN JOURNAL OF PHARMACEUTICAL AND MEDICAL RESEARCH www.ejpmr.com
 ejpmr, 2015,2(3), 436-440 EUROPEAN JOURNAL OF PHARMACEUTICAL AND MEDICAL RESEARCH www.ejpmr.com Tumram et al. SJIF Impact Factor 2.026 Research Article ISSN 3294-3211 EJPMR CLINICAL OUTCOME OF TORIC IOL
ejpmr, 2015,2(3), 436-440 EUROPEAN JOURNAL OF PHARMACEUTICAL AND MEDICAL RESEARCH www.ejpmr.com Tumram et al. SJIF Impact Factor 2.026 Research Article ISSN 3294-3211 EJPMR CLINICAL OUTCOME OF TORIC IOL
Nalin J. Mehta, MD ------------------------------------------------ Curriculum Vitae
 Nalin J. Mehta, MD ------------------------------------------------ Curriculum Vitae - -. Professional Mailing address: Nalin J. Mehta, MD PC. The Colorado Retina Center 274 Union Boulevard, Suite #120,
Nalin J. Mehta, MD ------------------------------------------------ Curriculum Vitae - -. Professional Mailing address: Nalin J. Mehta, MD PC. The Colorado Retina Center 274 Union Boulevard, Suite #120,
A Focus on Clinical Research
 SUPPLEMENT TO NOVEMBER 2014 www.reviewofoptometry.com A Focus on Clinical Research This year, our experts review and summarize the landmark trials that most directly influenced the course of retinal disease
SUPPLEMENT TO NOVEMBER 2014 www.reviewofoptometry.com A Focus on Clinical Research This year, our experts review and summarize the landmark trials that most directly influenced the course of retinal disease
VA high quality, complications low with phakic IOL
 Page 1 of 5 VA high quality, complications low with phakic IOL Use in keratoconus will continue, one surgeon predicts; another ponders long-term safety Nov 1, 2007 By:Nancy Groves Ophthalmology Times Several
Page 1 of 5 VA high quality, complications low with phakic IOL Use in keratoconus will continue, one surgeon predicts; another ponders long-term safety Nov 1, 2007 By:Nancy Groves Ophthalmology Times Several
2013 Diabetes and Eye Disease Fact Sheet
 The Georgia Department of Public Health 2013 Diabetes and Eye Disease Fact Sheet Diabetes is the leading cause of new cases of blindness among United States adults. 1-3 As Georgia s population ages and
The Georgia Department of Public Health 2013 Diabetes and Eye Disease Fact Sheet Diabetes is the leading cause of new cases of blindness among United States adults. 1-3 As Georgia s population ages and
Eye Department Baden-Baden. Prof. Dr. med. Frank Faude Dr. med. Susanne Faude Oxana Bräunlich. Ophthalmology specialists
 E y e D e p a r t m e n t Prof. Dr. med. Frank Faude Dr. med. Susanne Faude Oxana Bräunlich Ophthalmology specialists Prof. Dr. med. Frank Faude Inpatient and outpatient treatment at the Stadtklinik Baden-Baden
E y e D e p a r t m e n t Prof. Dr. med. Frank Faude Dr. med. Susanne Faude Oxana Bräunlich Ophthalmology specialists Prof. Dr. med. Frank Faude Inpatient and outpatient treatment at the Stadtklinik Baden-Baden
04/03/2015. EMR Documentation and Compliance: The Retina Point of View. Financial Disclosure. Financial Disclosure
 EMR Documentation and Compliance: The Retina Point of View William T. Koch, COA, COE, CPC The Retina Institute Kirk A. Mack, COMT, COE, CPC, CPMA Corcoran Consulting Group Financial Disclosure William
EMR Documentation and Compliance: The Retina Point of View William T. Koch, COA, COE, CPC The Retina Institute Kirk A. Mack, COMT, COE, CPC, CPMA Corcoran Consulting Group Financial Disclosure William
~Contributing to Eye Health~
 ~Contributing to Eye Health~ 26 Eye Care Market Trends FY2007 Retinal Camera market forecasts by eye disease FY 2010 Auto Keratorefractometer Retina 160 Refraction Retina 200 Refraction Auto Lens Edger
~Contributing to Eye Health~ 26 Eye Care Market Trends FY2007 Retinal Camera market forecasts by eye disease FY 2010 Auto Keratorefractometer Retina 160 Refraction Retina 200 Refraction Auto Lens Edger
Advocating for Improved Treatment and Outcomes for Diabetic Macular Edema
 Advocating for Improved Treatment and Outcomes for Diabetic Macular Edema A Report Based on an International Expert Summit Convened in Paris, June 2014 Acknowledgements This publication was supported by
Advocating for Improved Treatment and Outcomes for Diabetic Macular Edema A Report Based on an International Expert Summit Convened in Paris, June 2014 Acknowledgements This publication was supported by
Retina InSight Volume 2 Winter 2015/2016
 Retina InSight Volume 2 Winter 2015/2016 News from the Greater Houston Retina Research Foundation RCH Retina Rangers Give Back at Houston VisionWalk 2015 Hearts were pounding and pulses racing at the 2015
Retina InSight Volume 2 Winter 2015/2016 News from the Greater Houston Retina Research Foundation RCH Retina Rangers Give Back at Houston VisionWalk 2015 Hearts were pounding and pulses racing at the 2015
MEDICARE PAYMENTS FOR DRUGS USED TO TREAT WET AGE-RELATED MACULAR DEGENERATION
 Department of Health and Human Services OFFICE OF INSPECTOR GENERAL MEDICARE PAYMENTS FOR DRUGS USED TO TREAT WET AGE-RELATED MACULAR DEGENERATION Daniel R. Levinson Inspector General April 2012 OEI-03-10-00360
Department of Health and Human Services OFFICE OF INSPECTOR GENERAL MEDICARE PAYMENTS FOR DRUGS USED TO TREAT WET AGE-RELATED MACULAR DEGENERATION Daniel R. Levinson Inspector General April 2012 OEI-03-10-00360
Cecilia Lee, M.D. University of Washington Department of Ophthalmology 325 9 th Ave. Box 359608 Seattle WA 98104
 Cecilia Lee, M.D. University of Washington Department of Ophthalmology 325 9 th Ave. Box 359608 Seattle WA 98104 OPHTHALMOLOGY APPOINTMENTS 2014-Present University of Washington Seattle, WA Acting Instructor,
Cecilia Lee, M.D. University of Washington Department of Ophthalmology 325 9 th Ave. Box 359608 Seattle WA 98104 OPHTHALMOLOGY APPOINTMENTS 2014-Present University of Washington Seattle, WA Acting Instructor,
Long-Term Outcomes of Flap Amputation After LASIK
 Long-Term Outcomes of Flap Amputation After LASIK Priyanka Chhadva BS, Florence Cabot MD, Anat Galor MD, Sonia H. Yoo MD Bascom Palmer Eye Institute, University of Miami Miller School of Medicine Miami
Long-Term Outcomes of Flap Amputation After LASIK Priyanka Chhadva BS, Florence Cabot MD, Anat Galor MD, Sonia H. Yoo MD Bascom Palmer Eye Institute, University of Miami Miller School of Medicine Miami
RAJIV R. RATHOD, M.D., M.B.A. Fellow, American Academy of Ophthalmology
 CURRICULUM VITAE RAJIV R. RATHOD, M.D., M.B.A. Fellow, American Academy of Ophthalmology Office Address: Orange County Retina Medical Group 1200 North Tustin Avenue, Suite 140 Santa Ana, California 92705
CURRICULUM VITAE RAJIV R. RATHOD, M.D., M.B.A. Fellow, American Academy of Ophthalmology Office Address: Orange County Retina Medical Group 1200 North Tustin Avenue, Suite 140 Santa Ana, California 92705
A Modified Semiautomatic Method for Measurement of Hyperfluorescence Area in Fluorescein Angiography
 A Modified Semiautomatic Method for Measurement of Hyperfluorescence Area in Fluorescein Angiography Khalil Ghasemi Falavarjani, MD 1 Hanieh Mohammadreza, MSc 2 Sedigheh Marjaneh Hejazi, PhD 3 Masood Naseripour,
A Modified Semiautomatic Method for Measurement of Hyperfluorescence Area in Fluorescein Angiography Khalil Ghasemi Falavarjani, MD 1 Hanieh Mohammadreza, MSc 2 Sedigheh Marjaneh Hejazi, PhD 3 Masood Naseripour,
Eye and Vision Care in the Patient-Centered Medical Home
 1505 Prince Street, Alexandria, VA 22314 (703) 739-9200200 FAX: (703) 739-9497494 Eye and Vision Care in the Patient-Centered Medical Home The Patient Centered Medical Home (PCMH) is an approach to providing
1505 Prince Street, Alexandria, VA 22314 (703) 739-9200200 FAX: (703) 739-9497494 Eye and Vision Care in the Patient-Centered Medical Home The Patient Centered Medical Home (PCMH) is an approach to providing
September 2015 Corporate Presentation Ocata Therapeutics - NASDAQ OCAT
 September 2015 Corporate Presentation Ocata Therapeutics - NASDAQ OCAT Cautionary Statement Concerning Forward-Looking Statements These slides and the accompanying oral presentation contain statements
September 2015 Corporate Presentation Ocata Therapeutics - NASDAQ OCAT Cautionary Statement Concerning Forward-Looking Statements These slides and the accompanying oral presentation contain statements
Committee Approval Date: September 12, 2014 Next Review Date: September 2015
 Medication Policy Manual Policy No: dru361 Topic: Pradaxa, dabigatran Date of Origin: September 12, 2014 Committee Approval Date: September 12, 2014 Next Review Date: September 2015 Effective Date: November
Medication Policy Manual Policy No: dru361 Topic: Pradaxa, dabigatran Date of Origin: September 12, 2014 Committee Approval Date: September 12, 2014 Next Review Date: September 2015 Effective Date: November
Preliminary Results from a Phase 2 Study of ARQ 197 in Patients with Microphthalmia Transcription Factor Family (MiT) Associated Tumors
 Preliminary Results from a Phase 2 Study of ARQ 197 in Patients with Microphthalmia Transcription Factor Family (MiT) Associated Tumors John Goldberg 1 *, George Demetri 2, Edwin Choy 3, Lee Rosen 4, Alberto
Preliminary Results from a Phase 2 Study of ARQ 197 in Patients with Microphthalmia Transcription Factor Family (MiT) Associated Tumors John Goldberg 1 *, George Demetri 2, Edwin Choy 3, Lee Rosen 4, Alberto
Retinal Detachments Mark E. Hammer, M.D. Ivan J. Suñer, M.D. Marc C. Peden, M.D.
 Retinal Detachments Mark E. Hammer, M.D. Ivan J. Suñer, M.D. Marc C. Peden, M.D. What is a retinal detatchment? The retina is the light sensitive layer covering the inside of the back of the eye. It is
Retinal Detachments Mark E. Hammer, M.D. Ivan J. Suñer, M.D. Marc C. Peden, M.D. What is a retinal detatchment? The retina is the light sensitive layer covering the inside of the back of the eye. It is
Principles of Diagnosis MEDICAL DECISION-MAKING IN OPTOMETRIC PRACTICE. Medical Decision-Making. Medical Decision-Making.
 MEDICAL DECISION-MAKING IN OPTOMETRIC PRACTICE Craig Thomas, O.D. 3900 West Wheatland Road Dallas, Texas 75237 972-780-7199 thpckc@yahoo.com Principles of Diagnosis The study of the art and science of
MEDICAL DECISION-MAKING IN OPTOMETRIC PRACTICE Craig Thomas, O.D. 3900 West Wheatland Road Dallas, Texas 75237 972-780-7199 thpckc@yahoo.com Principles of Diagnosis The study of the art and science of
Incidence of Exudative Age-Related Macular Degeneration among Elderly Americans
 Incidence of Exudative Age-Related Macular Degeneration among Elderly Americans Jonathan C. Javitt, MD, MPH, 1 Zhiyuan Zhou, PhD, 2 Maureen G. Maguire, PhD, 2 Stuart L. Fine, MD, 3 Richard J. Willke, PhD
Incidence of Exudative Age-Related Macular Degeneration among Elderly Americans Jonathan C. Javitt, MD, MPH, 1 Zhiyuan Zhou, PhD, 2 Maureen G. Maguire, PhD, 2 Stuart L. Fine, MD, 3 Richard J. Willke, PhD
journal of medicine The new england Ranibizumab for Neovascular Age-Related Macular Degeneration ABSTRACT
 The new england journal of medicine established in 1812 october 5, 26 vol. 355 no. 14 Ranibizumab for Neovascular Age-Related Macular Degeneration Philip J. Rosenfeld, M.D., Ph.D., David M. Brown, M.D.,
The new england journal of medicine established in 1812 october 5, 26 vol. 355 no. 14 Ranibizumab for Neovascular Age-Related Macular Degeneration Philip J. Rosenfeld, M.D., Ph.D., David M. Brown, M.D.,
TABLE OF CONTENTS: LASER EYE SURGERY CONSENT FORM
 1 BoydVision TABLE OF CONTENTS: LASER EYE SURGERY CONSENT FORM Risks and Side Effects... 2 Risks Specific to PRK... 3 Risks Specific to LASIK... 4 Patient Statement of Consent... 5 Consent for Laser Eye
1 BoydVision TABLE OF CONTENTS: LASER EYE SURGERY CONSENT FORM Risks and Side Effects... 2 Risks Specific to PRK... 3 Risks Specific to LASIK... 4 Patient Statement of Consent... 5 Consent for Laser Eye
MASTER OCT LECTURE OUTLINE Ophthalmic Photographers Society November 16, 2013 New Orleans, LA
 MASTER OCT LECTURE OUTLINE Ophthalmic Photographers Society November 16, 2013 New Orleans, LA Authors: James B Soque, CRA COA and Pamela A Weber, MD Island Retina Shirley, New York OPS COURSE SA 3 B Financial
MASTER OCT LECTURE OUTLINE Ophthalmic Photographers Society November 16, 2013 New Orleans, LA Authors: James B Soque, CRA COA and Pamela A Weber, MD Island Retina Shirley, New York OPS COURSE SA 3 B Financial
Radiation Therapy for Small Choroidal Neovascularization in Age-related Macular Degeneration
 Radiation Therapy for Small Choroidal Neovascularization in Age-related Macular Degeneration Hideaki Matsuhashi,* Yasuko Noda,* Daisuke Takahashi* and Yasushi Mariya Departments of *Ophthalmology; Radiology,
Radiation Therapy for Small Choroidal Neovascularization in Age-related Macular Degeneration Hideaki Matsuhashi,* Yasuko Noda,* Daisuke Takahashi* and Yasushi Mariya Departments of *Ophthalmology; Radiology,
Avastin in breast cancer: Summary of clinical data
 Avastin in breast cancer: Summary of clinical data Worldwide, over one million people are diagnosed with breast cancer every year 1. It is the most frequently diagnosed cancer in women 1,2, and the leading
Avastin in breast cancer: Summary of clinical data Worldwide, over one million people are diagnosed with breast cancer every year 1. It is the most frequently diagnosed cancer in women 1,2, and the leading
Donna McCune, CCS-P, COE Vice President Corcoran Consulting Group
 Documentation and Coding for Ophthalmic Diagnostic Tests Financial Disclosure The instructor is a consultant for Corcoran Consulting Group and acknowledges a financial interest in the subject matter of
Documentation and Coding for Ophthalmic Diagnostic Tests Financial Disclosure The instructor is a consultant for Corcoran Consulting Group and acknowledges a financial interest in the subject matter of
Clinical Study Synopsis
 Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
Use of eye care services by people with diabetes: the Melbourne Visual Impairment Project
 410 Br J Ophthalmol 1998;82:410 414 Use of eye care services by people with diabetes: the Melbourne Visual Impairment Project Catherine A McCarty, Conrad W Lloyd-Smith, Sharon E Lee, Patricia M Livingston,
410 Br J Ophthalmol 1998;82:410 414 Use of eye care services by people with diabetes: the Melbourne Visual Impairment Project Catherine A McCarty, Conrad W Lloyd-Smith, Sharon E Lee, Patricia M Livingston,
Patient-Reported Outcomes with LASIK (PROWL-1) Results
 Patient-Reported Outcomes with LASIK (PROWL-1) Results Elizabeth M. Hofmeister, MD CAPT, MC, USN Naval Medical Center San Diego Refractive Surgery Advisor for Navy Ophthalmology Assistant Professor of
Patient-Reported Outcomes with LASIK (PROWL-1) Results Elizabeth M. Hofmeister, MD CAPT, MC, USN Naval Medical Center San Diego Refractive Surgery Advisor for Navy Ophthalmology Assistant Professor of
DISTANCE LEARNING COURSE. Anatomy of the Patient Exam. 2008 2012, BSM Consulting All rights reserved.
 DISTANCE LEARNING COURSE Anatomy of the Patient Exam 2008 2012, BSM Consulting All rights reserved. Table of Contents OVERVIEW... 1 PRELIMINARY WORK-UP... 1 OTHER DIAGNOSTIC TESTING... 3 DIAGNOSIS... 4
DISTANCE LEARNING COURSE Anatomy of the Patient Exam 2008 2012, BSM Consulting All rights reserved. Table of Contents OVERVIEW... 1 PRELIMINARY WORK-UP... 1 OTHER DIAGNOSTIC TESTING... 3 DIAGNOSIS... 4
Alexandria Fairfax Sterling Leesburg 703-931-9100 703-573-8080 703-430-4400 703-858-3170
 DIABETIC RETINOPATHY www.theeyecenter.com This pamphlet has been written to help people with diabetic retinopathy and their families and friends better understand the disease. It describes the cause, symptoms,
DIABETIC RETINOPATHY www.theeyecenter.com This pamphlet has been written to help people with diabetic retinopathy and their families and friends better understand the disease. It describes the cause, symptoms,
Fundus Photograph Reading Center
 Modified 7-Standard Field Digital Color Fundus Photography (7M-D) 8010 Excelsior Drive, Suite 100, Madison WI 53717 Telephone: (608) 410-0560 Fax: (608) 410-0566 Table of Contents 1. 7M-D Overview... 2
Modified 7-Standard Field Digital Color Fundus Photography (7M-D) 8010 Excelsior Drive, Suite 100, Madison WI 53717 Telephone: (608) 410-0560 Fax: (608) 410-0566 Table of Contents 1. 7M-D Overview... 2
Type 1 neovascularization with polypoidal lesions complicating dome shaped macula
 DOI 10.1186/s40942-015-0008-5 CASE REPORT Open Access Type 1 neovascularization with polypoidal lesions complicating dome shaped macula Jonathan Naysan 1,2,3,4, Kunal K Dansingani 1,2,5, Chandrakumar Balaratnasingam
DOI 10.1186/s40942-015-0008-5 CASE REPORT Open Access Type 1 neovascularization with polypoidal lesions complicating dome shaped macula Jonathan Naysan 1,2,3,4, Kunal K Dansingani 1,2,5, Chandrakumar Balaratnasingam
Benefits and Harms of Femtosecond Laser Assisted Cataract Surgery: A Systematic Review
 4 Department of Veterans Affairs Health Services Research & Development Service Benefits and Harms of Femtosecond Laser Assisted Cataract Surgery: A Systematic Review December 2013 Prepared for: Department
4 Department of Veterans Affairs Health Services Research & Development Service Benefits and Harms of Femtosecond Laser Assisted Cataract Surgery: A Systematic Review December 2013 Prepared for: Department
Literature Search on The Prevalence of Visual Disorders by Age Group in Older People
 Literature Search on The Prevalence of Visual Disorders by Age Group in Older People Aggressive Research Intelligence Facility West Midlands Health Technology Assessment Collaboration October 2006 For
Literature Search on The Prevalence of Visual Disorders by Age Group in Older People Aggressive Research Intelligence Facility West Midlands Health Technology Assessment Collaboration October 2006 For
Eye Diseases. 1995-2014, The Patient Education Institute, Inc. www.x-plain.com otf30101 Last reviewed: 05/21/2014 1
 Eye Diseases Introduction Some eye problems are minor and fleeting. But some lead to a permanent loss of vision. There are many diseases that can affect the eyes. The symptoms of eye diseases vary widely,
Eye Diseases Introduction Some eye problems are minor and fleeting. But some lead to a permanent loss of vision. There are many diseases that can affect the eyes. The symptoms of eye diseases vary widely,
Top Medicare Audit problems. Retinal Imaging Technology. Optometric Medical Coding. Unilateral codes. Modifiers
 Top Medicare Audit problems Evaluation, Diagnosis, Coding and Reimbursement Associated with Medical Vitreo-Retinal Conditions Kim Castleberry, OD Plano Eye Associates Patient/staff initiated billing complaints!
Top Medicare Audit problems Evaluation, Diagnosis, Coding and Reimbursement Associated with Medical Vitreo-Retinal Conditions Kim Castleberry, OD Plano Eye Associates Patient/staff initiated billing complaints!
INFORMED CONSENT TO HAVE LASIK
 A Division of Scott & Christie and Associates INFORMED CONSENT TO HAVE LASIK This information is to help you make an informed decision about having Laser Assisted Intrastromal Keratomileusis (LASIK), an
A Division of Scott & Christie and Associates INFORMED CONSENT TO HAVE LASIK This information is to help you make an informed decision about having Laser Assisted Intrastromal Keratomileusis (LASIK), an
Title:Clinical and Optic Coherence Tomography Findings of Focal Choroidal Excavation in Chinese Patients
 Author's response to reviews Title:Clinical and Optic Coherence Tomography Findings of Focal Choroidal Excavation in Chinese Patients Authors: Jie Guo (gjiexxx@163.com) Lu Zhong (luzhong_88@163.com) Chunhui
Author's response to reviews Title:Clinical and Optic Coherence Tomography Findings of Focal Choroidal Excavation in Chinese Patients Authors: Jie Guo (gjiexxx@163.com) Lu Zhong (luzhong_88@163.com) Chunhui
Cancer Treatments Subcommittee of PTAC Meeting held 18 September 2015. (minutes for web publishing)
 Cancer Treatments Subcommittee of PTAC Meeting held 18 September 2015 (minutes for web publishing) Cancer Treatments Subcommittee minutes are published in accordance with the Terms of Reference for the
Cancer Treatments Subcommittee of PTAC Meeting held 18 September 2015 (minutes for web publishing) Cancer Treatments Subcommittee minutes are published in accordance with the Terms of Reference for the
ClearVision. Pacific ClearVision Institute. Informed Consent For Cataract Surgery And/Or Implantation of an Intraocular Lens
 ClearVision Pacific ClearVision Institute Informed Consent For Cataract Surgery And/Or Implantation of an Intraocular Lens INTRODUCTION This information is given to you so that you can make an informed
ClearVision Pacific ClearVision Institute Informed Consent For Cataract Surgery And/Or Implantation of an Intraocular Lens INTRODUCTION This information is given to you so that you can make an informed
THOMAS IRA MARGOLIS, M. D. OFFICE ADDRESS: BIRTH DATE: May 11, 1962 1500 Tilton Road PROFESSIONAL EXPERIENCE
 THOMAS IRA MARGOLIS, M. D. OFFICE ADDRESS: BIRTH DATE: May 11, 1962 1500 Tilton Road Northfield, NJ 08225 PLACE OF BIRTH: Uniontown, PA 609-646-5200 FAX 609-646-9868 PROFESSIONAL EXPERIENCE RETINAL AND
THOMAS IRA MARGOLIS, M. D. OFFICE ADDRESS: BIRTH DATE: May 11, 1962 1500 Tilton Road Northfield, NJ 08225 PLACE OF BIRTH: Uniontown, PA 609-646-5200 FAX 609-646-9868 PROFESSIONAL EXPERIENCE RETINAL AND
