Molecular and genetic changes in asbestos-related lung cancer
|
|
|
- Cornelia Mitchell
- 8 years ago
- Views:
Transcription
1 Available online at Cancer Letters 265 (2008) 1 15 Mini-review Molecular and genetic changes in asbestos-related lung cancer Penny Nymark a, *, Harriet Wikman b, Tuija Hienonen-Kempas a, Sisko Anttila a,c a Finnish Institute of Occupational Health, Helsinki, Finland b Department of Tumor Biology, University Medical Center Hamburg-Eppendorf, Hamburg, Germany c Department of Pathology, Haartman Institute and HUSLAB, University of Helsinki and Helsinki University Central Hospital, Helsinki, Finland Received 14 December 2007; received in revised form 15 February 2008; accepted 19 February Abstract Asbestos-exposure is associated with an increased risk of lung cancer, one of the leading causes of cancer deaths worldwide. Asbestos is known to induce DNA and chromosomal damage as well as aberrations in signalling pathways, such as the MAPK and NF-jB cascades, crucial for cellular homeostasis. The alterations result from both indirect effects through e.g. reactive oxygen/nitrogen species and direct mechanical disturbances of cellular constituents. This review describes the current knowledge on genomic and pathway aberrations characterizing asbestos-related lung cancer. Specific asbestos-associated molecular signatures can assist the development of early biomarkers, molecular diagnosis, and molecular targeted treatments for asbestos-exposed lung cancer patients. Ó 2008 Elsevier Ireland Ltd. All rights reserved. Keywords: Asbestos; Copy number; Gene expression; Lung cancer; Molecular changes 1. Introduction Asbestos-exposure increases the risk of several malignancies, most of all the risk of malignant mesothelioma, followed by lung and laryngeal cancer. Lung cancer causes the most cancer-related deaths in the world and even if mesothelioma is commonly known as the primary asbestos-associated cancer type, it has been estimated that asbestos gives rise to an equal number or more lung cancers as compared to mesothelioma. The fraction of all * Corresponding author. Tel.: ; fax: addresses: penny.nymark@ttl.fi, penny.nymark@ helsinki.fi (P. Nymark). lung cancers attributable to asbestos-exposure to date is estimated at approximately 5 7% [1,2]. In developed countries the use of asbestos in manufacture or building has been banned or under strict control for longer than a decade, and asbestos demolition work tightly regulated by law. Nevertheless, asbestos will continue to burden public health also in developed countries due to the long latency period, generally years, between the initial exposure to asbestos and malignant disease. The manufacture and use of asbestos products peaked in the 1970s in Western Europe, North America, Japan and Australia. Today, a majority of the asbestos, produced worldwide is used in Eastern Europe, Latin America and Asia [1,3,4]. Asbestos-exposure and tobacco smoking have a synergistic, more than additive, effect on lung cancer /$ - see front matter Ó 2008 Elsevier Ireland Ltd. All rights reserved. doi: /j.canlet
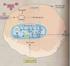
2 2 P. Nymark et al. / Cancer Letters 265 (2008) 1 15 risk, whereas asbestos-exposure is a sole risk factor for malignant mesothelioma [5,6]. It is not known, whether this difference reflects different carcinogenic mechanisms or different sensitivity of lung epithelial and mesothelial cell types to asbestos-induced carcinogenesis. The fact that most asbestos-exposed lung cancer patients are also tobacco smokers has made it very difficult to distinguish asbestos-related molecular changes from those related to tobacco carcinogens by molecular epidemiology [7]. Several genetic and molecular factors have been described to be involved in asbestos-induced carcinogenesis in vitro or in vivo, or identified in asbestosrelated human cancers. The emerging methodology of genomics, transcriptomics and proteomics has provided new screening tools for specific carcinogen-associated molecular profiles, especially when applied to well-designed experimental settings or carefully characterized patient materials. The increasing knowledge of the molecular changes in asbestos-related lung cancer is crucial for the discovery and development of early biomarkers, molecular diagnosis, and molecular targeted treatments. In the following we will review experimental and human data on the molecular mechanisms of asbestosinduced lung carcinogenesis. 2. Toxicity and carcinogenicity of asbestos fibres Asbestos is a generic term for industrially refined and produced fibrous silicate minerals. Asbestos is classified into six distinct mineralogical types, i.e., chrysotile, crocidolite, amosite, tremolite, anthophyllite, and actinolite. Chrysotile belongs to the serpentine group. It is a curly and thin fibre type. The other five types belong to the amphibole group of minerals, which are longer and needle-like. Chrysotile is the most commonly used and economically important asbestos type [1]. A number of animal experimental studies have shown that the longer the fibre, the more carcinogenic it is per se [8]. The genotoxicity depends also on the fibre s chemical composition and structure as well as the cell environment [9]. Amphibole fibres are chemically complex and contain variable amounts of associated mono-, di- and trivalent metals such as iron. It has been proposed that asbestos is not toxic by simply acting as a carrier of e.g. iron into the cell, but also by the particular way iron is bound to the fibre s surface enabling free radical generation [10,11]. Due to the metals, the fibre structure and their bio-persistence, the amphiboles are known to be more pathogenic in the human body compared to chrysotile. In contrast to chrysotile asbestos, which is fragmented and cleared from the lungs, amphiboles are considered insoluble in human lung [8,12]. In vitro studies have demonstrated that asbestos fibres are cytotoxic and clastogenic but surprisingly not mutagenic in Ames assay [13,14]. The main mechanisms behind these destructive effects are thought to be multiple, including generation of reactive oxygen (ROS) and nitrogen species (RNS), alteration in the mitochondrial function, physical disturbance of cell cycle progression, and activation of several signal transduction pathways [15,16]. Several mechanisms are likely to contribute to the synergistic carcinogenic effect of tobacco smoke and asbestos-exposure. It has been demonstrated that cigarette smoke augments the penetration of asbestos fibres in rat tracheal explants by an oxygen radical-mediated mechanism [17]. In addition, reactive oxygen species have been observed to alter the metabolism of a tobacco carcinogen, benzo[a]pyrene, by inhibiting the detoxification pathways [18]. On the other hand, tobacco carcinogens are known to be adsorbed on the surface of asbestos fibres increasing their uptake into the cells [19]. Furthermore, asbestos fibres induce cell proliferation, which may lead to clonal expansion of a cell with a heritable tobacco carcinogen-induced alteration in a critical gene [20]. 3. Molecular changes in asbestos-induced carcinogenesis In vitro studies have shown that all types of asbestos fibres are able to cause structural and/or numerical chromosomal aberrations, produce binucleated cells, and disturb cell division and homeostasis [21,22]. These alterations have been suggested to result from both an indirect effect of ROS and RNS on signalling cascades and a direct physiological interaction between the cell and the fibres [23] (Fig. 1). High-resolution time-lapse light microscopy studies on living cells have shown that asbestos fibres are actively transported along cytoplasmic microtubules to perinuclear regions where they can sterically block cytokinesis [21,24,25]. Recent results also show that asbestos fibres might directly bind to proteins that regulate the cell cycle, cytoskeleton, and mitotic processes contributing to significant spindle damage and chromosomal instability [26]. Furthermore, asbestos increases the frequency of
3 P. Nymark et al. / Cancer Letters 265 (2008) normal cell asbestos fibre signalling pathways EGFR/MAPK /ERK, NF-κB TNF-α ROS/RNS antioxidants and detoxifying enzymes (glutathione, MPO, EC-SOD) gene expression changes (TGFβ, oncogenes), release of cytokines (IL-1, -6, -8, -10) mitochondrial dysfunctions Mitochondria (apoptosis) DNA damage DNA repair (TP53, Ku70, PCNA) TP53 (cell cycle checkpoint) cancer Fig. 1. Asbestos-related carcinogenic pathways in the lung. homologous recombination (HR) suggested to be related with malignant transformation of cells carrying genetic alterations caused by other environmental mutagens and carcinogens [27]. To develop an in vitro model that would represent the actual in vivo progress of asbestos carcinogenesis has been challenging, since asbestos eventually causes apoptosis for many cell types in vitro. Nevertheless, a few malignant transformations by asbestos have been conducted on human bronchial epithelial cells, murine lung and hamster embryo cells [28 30]. Immortalized cell lines often show cytogenetic and molecular alterations and malignant transformation of such cells may be facilitated by these already existing changes. However, for example the HPV-18 immortalized human bronchial cell line, BEP2D, has been used in a variety of studies examining the transforming potential of various genotoxins, including asbestos [28,31,32]. The cell line has been shown to be karyotypically stable, although it does show some well documented abnormalities. The cells are also anchorage-dependent and do not form tumours in nude mice, not even in late passages [33]. Similar features apply to most cell lines used for this type of studies. An animal model for in vivo experiments has also been difficult to establish, since in humans asbestos fibres usually act in combination with other environmental carcinogens, such as tobacco smoke and silica, and asbestos-related changes accumulate during many decades. In vivo studies have, however, validated some in vitro experiments by identifying asbestos-related gene expression changes such as activation of the NF-jB pathway, p53 promoter activation, and cell proliferation induced by TNFa and -b as well as PDGF A and B [29,34 38]. Experimental and human data combined can give further insight into the central genes and pathways participating in the asbestos-induced malignant transformation of lung cells. In the following sections the current knowledge on asbestos-induced chromosomal aberrations, disturbances in key genes and pathways, as well as point mutations in the lungs will be described. The most important known chromosomal loci, genes and pathways associated with asbestos-related lung cancer are listed in Table Chromosomal aberrations The direct or indirect action of asbestos on DNA and proteins can cause many different types of DNA and chromosomal damage. Asbestos fibre-treated cells have shown increased amounts of
4 4 P. Nymark et al. / Cancer Letters 265 (2008) 1 15 Table 1 Changes in chromosomes, genes and pathways, related to asbestos-associated lung carcinogenesis Chromosome/gene/ pathway Aberration Asbestos-related carcinogenic association References a Chrom. 1 Break at the centromere [50,56,63] Copy number changes 1p36 and 1q21 Chrom. 3 LOH at 3p21 and 3p14 FHIT exon loss [62 65] Possible down-regulation of tumour suppressors Chrom. 5 Deletion or monosomy Possible down-regulation of tumour suppressors [33,63,64] Deletion at 5q35.2 q35.3 Chrom. 19 Monosomy Possible down-regulation of tumour suppressors [33,63,64,68] AI at 19p13 PPP (pentose phosphate pathway, e.g. G6PD) SOD Inhibition Depletion of glutathione followed by a decreased resistance against oxidative stress [73 75] Redistribution of extracellular Decreased resistance against ROS/RNS [72,77] SOD MPO Increased activity Increased production of RNS [97,141,143] G to A polymorphism in the 5 0 untranslated region of MPO Reduced lung cancer risk associated with MPO A-allele (G/A + A/A) genotype NF-jB pathway Up-regulation Tumour promotion through activation of [29,81,87,88,95] proto-oncogenes (e.g. c-myc) TNFa Up-regulation through NF-jB Enhances the interactions between cells and fibres by increasing the binding of asbestos to tracheal epithelial cells [80,81,149] Accumulative ROS generation through IL8 Interleukins (e.g. IL1, IL6, IL8 and IL10) Up-regulation Elevated in asbestos-exposed workers who develop lung cancer (IL6) Accumulative ROS generation (IL8) Apoptosis resistance (IL10) EGFR Phosphorylation/activation Cell proliferation through activation of the MAPK/ERK pathway MAPK/ERK pathway Activation through EGFR Tumour promotion through activation of AP-1 (e.g. ERK genes) dependent target genes Cell proliferation through activation by PKCd and adducin AP-1 dependent target Up-regulation by the NF-jB genes (e.g. c-fos) and MAPK/ERK pathways [81,82,84 86] [82,90,91] [64,90 92,95,100,101] Transcription factor for oncogenes [29,92 95] MMPs Activation Activation of EGFR [37,99] TGFb Up-regulation Promotes fibrosis [ ] Immune tolerance towards mesothelioma TP53 Up-regulation Cell proliferation through regulation of PCNA [38,114,123 Mutations Decreased tumour-suppressor activity 127,129] BCL2 and BCL2-like genes Activation/inhibition Apoptotic resistance [86, ] a Only asbestos-related aberrations mentioned in at least two studies have been included in this table. abnormalities, such as, lagging chromosomes, bridges, and sticky chromosomes in anaphase or telophase, formation of micronuclei, DNA singlestrand breaks and increased sister chromatid exchanges (SCE) [39 51]. The most typical asbestos-induced chromosomal aberrations in cell line experiments are deletions, breaks and fragments [14,52 57]. Most of these changes are found in a dose-dependent manner, but not surprisingly is the toxic dose highly dependent on the cell line and fibre type used [21,58,59]. In asbestos-exposed workers white blood cells, increased levels of SCE and DNA double-strand breaks as well as anti-doublestrand DNA antibodies have been reported [60,61]. Most studies describing chromosomal aberrations of human lung tumours have not reported the patients asbestos-exposure, and thus it is impossible to decipher the association between the aberrations and asbestos based on these reports. However, a few studies have described asbestos-specific
5 P. Nymark et al. / Cancer Letters 265 (2008) aberrations in e.g. chromosomes 1, 3p, 5, 8, 9, and 19p. Recently, two studies have shown that a common early aberration in lung cancer, namely loss of 3p, occurs more frequently in the tumours of asbestos-exposed than in non-exposed patients. Marsit et al. showed that allelic imbalance (AI) in 3p21.3 was associated with occupational asbestosexposure, TP53 mutations as well as better patient survival [62,63]. We also detected 3p21.3 as one of the main regions differentiating lung tumours of asbestos-exposed and non-exposed patients in a whole genome aberration and expression screening [63,64]. In lung tumours, loss of another region in the short arm of chromosome 3, namely 3p14 containing the FHIT gene, has also been associated with asbestos-exposure and tobacco smoking [65]. However, Pylkkänen et al. detected reduced FHIT expression in both asbestos-exposed and nonexposed patients lung tumours [66]. The region contains a fragile site, FRA3. In our array comparative genomic hybridization study we found that many of the regions with asbestos-related copy number changes were associated with fragile sites, demonstrating that asbestos may preferentially cause DNA damage at such sites [63]. Dopp et al. used fluorescence in situ hybridization (FISH) to investigate chromosomes 1 and 9 in human amniotic fibroblasts (AF) after asbestos treatment [50]. They reported the centromeric regions of these chromosomes to be affected by DNA breakage following asbestos-exposure. The same group treated the lymphocytes of smokers and non-smokers with asbestos fibres and again, damage in chromosome 1 was the main finding in non-smokers, whereas in smokers lymphocytes other chromosomes were damaged as well [56]. These findings support the hypothesis that, at least in part, the synergistic carcinogenic effect of tobacco smoke and asbestos fibres results in similar aberration patterns as compared to either exposure alone, but with more frequent aberrations found when both exposures are present. In a study by Suzuki et al., loss of one or two copies of chromosome 5, monosomy of chromosome 19, and trisomy of chromosome 8 were found to be common changes in five tumorigenic human bronchial epithelial cell lines transformed by chrysotile asbestos. The transformed cell lines showed especially high frequencies of the newly developed changes in chromosomes 5 and 19 which were not seen in the parental non-tumorigenic immortalized cell line BEP2D. Trisomy of chromosome 8 was not detected as a new change, but was seen more frequently in the tumourigenic cells. Furthermore, the non-tumorigenic parental BEP2D cell line generally showed hyperaneuploidy, with chromosomes, while all five tumorigenic cell lines showed hypoaneuploidy with chromosomes, demonstrating that asbestos causes loss of genomic material, consistent with other reports [33]. Interestingly, malignant transformation of the same cell line with radon-exposure did not cause the same type of aberrations, indicating that the changes could be asbestos-specific and not only cell line- or transformation-related [67]. Furthermore, in agreement with the results by Suzuki et al., we described loss of 5q35 and 19p combined with down-regulation of gene expression in asbestos-associated lung cancer [63,64,68] Genes and pathways associated with asbestosexposure The main cellular functions that are affected by asbestos fibres include oxidative stress response, inflammation, DNA damage response, mitochondrial activity and apoptosis (Fig. 1). Many genes and pathways involved in these functions have been identified and investigated, usually gene by gene. However, asbestos triggers a large amount of cellular responses and the difficulty has lied in recognizing the primary asbestos-related responses and especially the changes related to oncogenesis Oxidative stress and inflammation Asbestos-exposure causes oxidative stress through free radicals (ROS/RNS), which are released by macrophages upon unsuccessful phagocytosis of the fibres or generated directly through iron present on the surface of the fibres [69]. ROS cause DNA damage products, such as the mutagenic 8-hydroxy deoxyguanosine (8-OHdG) adducts, which have been found to be elevated in the white blood cells of asbestos-exposed workers [70]. Furthermore, asbestos induces gene expression changes through conversion of important signalling molecules, such as nitric oxide (NO), to free radicals (NO 2 ). NO is an important regulator of e.g. p53 and therefore affects a multitude of key cellular events [71] Antioxidant pathways. An imbalance in oxidant antioxidant levels has been proposed to underlie the pathogenesis of asbestos-related lung disease
6 6 P. Nymark et al. / Cancer Letters 265 (2008) 1 15 [72]. Disturbances in antioxidant pathways reduce the cells capacity to protect itself against ROS/ RNS. Studies have demonstrated that antioxidant enzymes, such as catalase (CAT) and superoxide dismutase (SOD) as well as antioxidant peptides including glutathione, can effectively protect cells in vitro against the mutagenic effects of asbestosinduced ROS/RNS production [55,73]. Indeed, lung carcinoma A549 cells, which contain a 3.5-fold glutathione content as compared to mesothelial Met- 5A cells, have been shown to be more resistant against oxidants and fibres than the Met-5A cells [73]. Endocytosis of asbestos fibres by both macrophages and lung epithelial cells has been shown to be followed by a decrease in intracellular and an increase in extracellular glutathione [74]. The depletion of glutathione is caused by asbestos-induced inhibition of the enzyme G6PD involved in an antioxidant pathway (pentose phosphate pathway). Synthetic fibres, containing almost no iron, do not exert the same effect, indicating that the surface features of asbestos fibres trigger the changes in the cells antioxidant activity [75,76]. In addition, another group showed that asbestos causes redistribution of extracellular SOD (EC-SOD) from the lung parenchyma to the air spaces in the lung, resulting in high levels of EC-SOD in bronchoalveolar lavage fluid in mice [72]. Increased sensitivity to asbestos-induced injury has been reported in mice lacking EC-SOD [77]. In contrast, increased manganese SOD (MnSOD) has been associated with higher sensitivity to oxidative stress in epithelial cells [78]. Janssen et al. found MnSOD to be increased both on mrna and protein level in the lungs of asbestosexposed rats [79]. Thus, the concept of oxidant antioxidant imbalance in relation to asbestos-induced injury seems to be more complex than initially thought TNFa/NF-jB pathway. The inflammatory cytokine, TNFa, has been shown to be activated in macrophages after asbestos-exposure in vitro [80]. TNFa induces IL8 expression in macrophages, which attracts neutrophils that in turn, release ROS and RNS. This leads to a feedback loop between ROS generation and increased TNFa expression, resulting in increased DNA damage [81]. Also, other interleukins are released by inflammatory cells [82] upon phagocytosis of fibres and e.g. IL6 has been shown to be up-regulated in airway epithelial cells by NF-jB in response to asbestos-exposure [83]. Increased IL6 correlates with increased serum levels of C-reactive protein (CRP). CRP has been found to be significantly more elevated in the serum of asbestos-exposed workers who developed cancer (lung cancer and mesothelioma) than in those that did not develop cancer in a follow up study [84]. In addition, IL1 and IL10 have been found to be upregulated by asbestos-induced oxidative stress in vitro [85,86]. NF-jB coordinates the inflammatory and cell proliferative responses to asbestos [29,87]. By exposing rat tracheal explants to asbestos, TNFa was found to increase the binding of fibres to the surface of tracheal epithelial cells via an NF-jB-dependent pathway, enhancing the interactions of the fibres with the tissue [81]. In mesothelial cells, treatment with TNFa activates NF-jB, which increases the percentage of cells that survive asbestos-exposure and thereby the amount of asbestos-damaged cells capable of undergoing malignant transformation [88]. Furthermore, NF-jB is a transcription factor for several oncogenes, and for example increased c-myc transcription has been detected upon asbestos-exposure in human embryonic lung cells [89] EGFR/MAPK/ERK pathway. Asbestosinduced oxidative stress causes activation of the epidermal growth factor receptor (EGFR) by phosphorylation [82,90,91]. EGFR activates the MAPK/ ERK pathway through phosphorylation of ERK1/2 and ERK5 [92]. Increased levels of phospho-erk1/ 2 and -ERK5 induce proliferation and activation of the proto-oncogenes c-fos, fra-1 and c-jun (AP-1 family members) [29,93 95]. It has been proposed that the synergistic properties of asbestos and tobacco smoke may be caused by separate activation of the ERK genes and JNK1/2, respectively, which both transactivate AP-1 [92]. Reactivation of the cell cycle in a critical DNA repair stage may lead to DNA damage bypass allowing cells with oncogenic changes to continue proliferating. Indeed, low levels of asbestos have been shown to cause cytoplasmic localization of phospho-erk1/ 2, which is followed by AP-1 dependent nuclear localization of cyclin D1 [96]. Cyclin D causes cell cycle reentry through progression from G 1 - to S-phase [97]. In addition, other growth factors such as the insulin-like growth factor (IGF) and platelet-derived growth factor (PDGF) are also known to promote S-phase after asbestos-exposure [98]. Activation of EGFR has also been shown to be caused by protein kinase C (PKC) activated matrix
7 P. Nymark et al. / Cancer Letters 265 (2008) metalloproteinases (MMP) [37], and e.g. MMP2 has been found to be up-regulated after combined exposure to chrysotile and cigarette smoke in vivo [99].In agreement, PKCd and its substrate, adducin, was shown to cause cell proliferation through activation of ERK1/2 in response to asbestos-exposure [100,101]. Noticeably, adducin (ADD1) has been found to be up-regulated in lung tumours of asbestos-exposed patients compared to those of nonexposed patients [64]. Finally, ERK1/2 has been shown to be activated by Src family kinase [102]. Growth promoting tyrosine kinases, such as Src are activated by the urokinase plasminogen activator (PLAU) pathway, involved in tissue reorganization events such as wound healing. The PLAU pathway has been shown to be activated by asbestos [103] TGFb pathway. The transforming growth factor b (TGFb) pathway has dual tumour suppressive and oncogenic effects depending on the stage of carcinogenesis [104]. High levels of the cytokine TGFb1 has been suggested to have an immunosuppressive role in mesothelioma, i.e., immune tolerance towards the tumour, allowing it to progress [105]. Asbestos causes over-expression of TGFb1 and it has been related to fibrogenesis in mice [106,107]. The TGFb-induced gene, bigh3, transcribes for an adhesion protein possibly involved in cell collagen interactions. The gene has been shown to be up-regulated in human lung carcinoma [108]. In contrast, bigh3 was found to be down-regulated in five asbestos-transformed human bronchial epithelial cell lines, derived from the BEP2D cells [109]. The transformed cell lines consistently formed tumours in nude mice. Reintroduction of the gene in a highly malignant asbestos-transformed cell line inhibited cell growth and tumour formation in mice [110]. Thus, it is intriguing to hypothesize that the bigh3 gene may specifically be involved in asbestos-induced carcinogenicity. It is not known what causes the down-regulation of bigh3, since TGFb1 is activated by asbestos [69,111,112]. However, it has been proposed that the bigh3 might be a target of asbestos-induced chromosome 5 losses [33,63]. The tumour suppressive role of bigh3 is still to be identified DNA damage repair Initially cells attempt to adapt and repair damage caused by asbestos-exposure and consequently, many DNA repair genes, such as TP53 and GADD153, are up-regulated in asbestos treated cells [113]. However, the accumulation of fibres in the lung and the continuous production of ROS/RNS causes repeated DNA damage, which may lead to increased genetic instability, a hallmark of neoplastic development. The DNA damage and instability may cause abnormal expression of DNA repair genes. In fact, Ku70 involved in nonhomologous DNA repair, has been found to be down-regulated in asbestos-transformed tumorigenic lung cell lines [109]. Over-expression of TP53 should assist the DNA repair processes. However, p53 has been shown to bind and regulate the proliferating cell nuclear antigen gene (PCNA), which is involved in DNA repair, but also in DNA damage bypass. Activated PCNA could together with growth factors, many of which are induced by asbestos, allow cells with DNA damage to continue proliferating. PCNA has been shown to be co-expressed with p53 in the lungs of asbestos-exposed rats [114], and it is also induced in lung epithelial cells after mechanical wounding of the cultured cell layer and after asbestos-exposure [100] Mitochondrial function and apoptosis Apoptosis protects against abnormal proliferation of cells with non-repairable DNA damage. Alterations in most of the previously mentioned asbestos-induced genes and pathways should eventually lead to apoptosis. However, the apoptotic pathways seem to be inhibited in asbestos-associated lung cancer as in many other cancers. Indeed, low doses of asbestos (<0,5 lg/cm 2 ) promote S- phase entry and thereby cell proliferation through an EGFR dependent pathway instead of apoptosis [96]. If apoptosis is bypassed, the asbestos-associated dysfunctions in the mitochondrial respiratory chain maintain the increased release of ROS. Genes in the apoptotic pathway could be potential target genes for therapy either through silencing or activation. For example, the expression and phosphorylation of camp responsive element binding protein (CREB) is thought to be an important regulator of apoptosis in asbestos-induced responses, and silencing of the gene dramatically increases asbestos-induced apoptosis in lung epithelial cells [115]. Similarly, over-expression of the 8-OHdG repair enzyme, OGG1 and its translocation to the mitochondria has been shown to reduce asbestos-induced apoptosis in HeLa cells [116].
8 8 P. Nymark et al. / Cancer Letters 265 (2008) 1 15 Furthermore, gene expression profiling of asbestostransformed tumorigenic lung cell lines has revealed down-regulation of an apoptosis-related putative tumour suppressor DCC (deleted in colorectal cancer) [109]. Miura et al. have produced an apoptosis resistant T-cell cell line through repeatedly exposing the cells to asbestos. By studying this cell line they proposed a model mechanism for acquiring of resistance to asbestos-induced apoptosis, involving activation of the genes Src family kinase, IL-10, STAT3 and BCL2. Interestingly, BCL2 was also found to be significantly up-regulated in the T cells of mesothelioma patients as compared to healthy volunteers and asbestosis patients, indicating a role in carcinogenesis [86]. Many other BCL2 related genes have been implicated in asbestos-induced apoptotic resistance or carcinogenesis, such as BNIP3L, Bax and Bcl-xl [ ] Point mutations Both crocidolite and chrysotile asbestos have been shown to increase the general mutation frequency in various hamster and rat cells [120,121]. However, the fibres are at most marginally positive for their ability to produce point mutations at specific loci in vitro. Nevertheless, some studies on human lung tumours have linked specific mutations to asbestos-exposure [48]. Some of these mutations could primarily be caused by tobacco specific carcinogens such as benzo[a]pyrene, which have been shown to have an enhanced mutagenic effect following co-exposure with amosite asbestos in rat lung [122]. TP53 is probably the most extensively studied gene in relation to asbestos-exposure due to its crucial role in DNA damage response. TP53 is not only up-regulated after asbestos-exposure but abnormal accumulation of the protein has been detected more frequently in tumours and serum from exposed patients compared with patients without asbestosexposure [114, ]. Increased levels of the protein could partially be caused by mutated p53. Indeed, mutations in p53 have been positively associated with asbestos-exposure in lung cancer patients in many [38,126,127] but not in all studies [62,128]. TP53 mutations have also been identified in mouse fibroblasts after crocidolite exposure [129]. Also p16/ink4a, a regulator of p53, has been associated with asbestos-exposure. Inactivation of p16/ink4a by either methylation [130] or by homozygous deletion has been found to correlate with asbestos-exposure in non-small cell lung cancer patients [131]. The biomarker for oxidative stress, 8-OHdG adduct, is mutagenic and may cause G:C to T:A transversions. These nucleotide transversions have been found to be more frequent in tumours of asbestos-exposed individuals [132]. High levels of G to T transversions in codons 12, 13 and 61 of the KRAS gene were found in a study population of lung cancer patients exposed to asbestos, but the association could not be confirmed in a study with added cases [128,133]. However, in a later study with 355 cases, mutations in codon 12 of the KRAS gene were significantly associated with asbestos-related lung adenocarcinoma [134]. In contrast, no mutations could be found in the KRAS gene in five asbestos-transformed malignant cell lines, which suggests that these mutations may be a result of the synergistic effects of asbestos and tobacco carcinogens [28]. 4. Genetic susceptibility to asbestos-related lung cancer Tobacco is clearly the most important single environmental factor for lung cancer. As not all of even the heaviest smokers contract lung cancer, individual differences in several host factors including differences in metabolism, DNA repair capacity, altered expression of proto-oncogenes and/or inactivation of tumour-suppressor genes, are anticipated to modify individual susceptibility to the disease. Variations in an individual s capacity to detoxify tobacco carcinogens as well as in DNA repair capacity, due to genetic polymorphisms, are well documented. These individual variations have indeed been linked to different risks in contracting tobacco-related lung cancer. Most results have, however, shown that lung cancer risk is only moderately increased by single polymorphic genes, but can be considerably enhanced by specific combinations of susceptibility genes [135]. Individual susceptibility to asbestos-related lung cancer is much less studied. However, a few research groups have investigated the relation between asbestos-exposure and polymorphisms in xenobiotic metabolizing enzymes (GSTM1, GSTT1, MPO, CYP1A1, and CYP2E1) and ROS defence (SOD2) [52, ]. Glutathione S transferases (GST) catalyse conjugation of reduced glutathione (GSH) to a large
Update of the scientific evidence on asbestos and cancer. Kurt Straif, MD MPH PhD. The IARC Monographs
 Update of the scientific evidence on asbestos and cancer Kurt Straif, MD MPH PhD International Agency for Research on Cancer Lyon, France World Health Organisation Asturias, 17 March 2011 The IARC Monographs
Update of the scientific evidence on asbestos and cancer Kurt Straif, MD MPH PhD International Agency for Research on Cancer Lyon, France World Health Organisation Asturias, 17 March 2011 The IARC Monographs
Mesothelioma. Mesothelioma and Asbestos 11/21/2009
 Mesothelioma Michele Carbone, M.D.,PH.D. Director Cancer Research Center of Hawaii Professor and Chairman, Dept. of Pathology J.A. Burns Medical School University of Hawaii Honolulu, HI 96813 Mesotheliomas
Mesothelioma Michele Carbone, M.D.,PH.D. Director Cancer Research Center of Hawaii Professor and Chairman, Dept. of Pathology J.A. Burns Medical School University of Hawaii Honolulu, HI 96813 Mesotheliomas
Kerosene soot genotoxicity: enhanced effect upon co-exposure with chrysotile asbestos in Syrian hamster embryo fibroblasts
 Toxicology Letters 114 (2000) 111 116 www.elsevier.com/locate/toxlet Kerosene soot genotoxicity: enhanced effect upon co-exposure with chrysotile asbestos in Syrian hamster embryo fibroblasts Mohtashim
Toxicology Letters 114 (2000) 111 116 www.elsevier.com/locate/toxlet Kerosene soot genotoxicity: enhanced effect upon co-exposure with chrysotile asbestos in Syrian hamster embryo fibroblasts Mohtashim
BE.104 Spring Evaluating Environmental Causes of Mesothelioma J. L. Sherley
 BE.104 Spring Evaluating Environmental Causes of Mesothelioma J. L. Sherley Outline: 1) Toxicological mechanisms and causation evaluations 2) An environetics case: Asbestos and Mesothelioma Toxicological
BE.104 Spring Evaluating Environmental Causes of Mesothelioma J. L. Sherley Outline: 1) Toxicological mechanisms and causation evaluations 2) An environetics case: Asbestos and Mesothelioma Toxicological
OCCUPATIONAL LUNG CANCER
 OCCUPATIONAL LUNG CANCER Anwar Jusuf, Agus Dwi Susanto Department of Pulmonology & Respiratory Medicine, Faculty of Medicine University Of Indonesia - Persahabatan Hospital-Jakarta INTRODUCTION Occupational
OCCUPATIONAL LUNG CANCER Anwar Jusuf, Agus Dwi Susanto Department of Pulmonology & Respiratory Medicine, Faculty of Medicine University Of Indonesia - Persahabatan Hospital-Jakarta INTRODUCTION Occupational
Lung cancer and asbestos
 Lung cancer and asbestos Bureau Veritas Training Bill Sanderson For the benefit of business and people To begin with.. There are known knowns, that is there are things we know that we know. There are known
Lung cancer and asbestos Bureau Veritas Training Bill Sanderson For the benefit of business and people To begin with.. There are known knowns, that is there are things we know that we know. There are known
D.M. Bernstein * WHO review Page 1 of 8
 A review of the WHO s document on the adverse health effects of exposure to asbestos and WHO's recommendations on the prevention of asbestos-related diseases D.M. Bernstein * The WHO s Programme on Occupational
A review of the WHO s document on the adverse health effects of exposure to asbestos and WHO's recommendations on the prevention of asbestos-related diseases D.M. Bernstein * The WHO s Programme on Occupational
Aberrations of chromosome 19 in asbestos-associated lung cancer and in asbestos-induced micronuclei of bronchial epithelial cells in vitro
 Carcinogenesis vol.29 no.5 pp.913 917, 2008 doi:10.1093/carcin/bgn068 Advance Access publication March 13, 2008 Aberrations of chromosome 19 in asbestos-associated lung cancer and in asbestos-induced micronuclei
Carcinogenesis vol.29 no.5 pp.913 917, 2008 doi:10.1093/carcin/bgn068 Advance Access publication March 13, 2008 Aberrations of chromosome 19 in asbestos-associated lung cancer and in asbestos-induced micronuclei
COPD, INFLAMMATION, AND LUNG CANCER. Jerome S Brody, M.D., Professor of Medicine and. Avrum Spira, M.D., Assistant Professor of Medicine
 COPD, INFLAMMATION, AND LUNG CANCER Jerome S Brody, M.D., Professor of Medicine and Avrum Spira, M.D., Assistant Professor of Medicine Pulmonary Center and Department of Medicine Boston University School
COPD, INFLAMMATION, AND LUNG CANCER Jerome S Brody, M.D., Professor of Medicine and Avrum Spira, M.D., Assistant Professor of Medicine Pulmonary Center and Department of Medicine Boston University School
Mechanism of short-term ERK activation by electromagnetic fields at mobile phone frequencies. Biochemistry Journal. August 1, 2007 405, pp.
 Mechanism of short-term ERK activation by electromagnetic fields at mobile phone frequencies 1 Biochemistry Journal August 1, 2007 405, pp. 559 568 Joseph Friedman, Sarah Kraus, Yirmi Hauptman, Yoni Schiff
Mechanism of short-term ERK activation by electromagnetic fields at mobile phone frequencies 1 Biochemistry Journal August 1, 2007 405, pp. 559 568 Joseph Friedman, Sarah Kraus, Yirmi Hauptman, Yoni Schiff
Asbestos-Induced Mesothelioma
 2 Asbestos-Induced Mesothelioma Maria E. Ramos-Nino, Marcella Martinelli, Luca Scapoli, and Brooke T. Mossman Asbestos, a group of chemically and physically distinct fibers, is one of the most notorious
2 Asbestos-Induced Mesothelioma Maria E. Ramos-Nino, Marcella Martinelli, Luca Scapoli, and Brooke T. Mossman Asbestos, a group of chemically and physically distinct fibers, is one of the most notorious
The Need for a PARP in vivo Pharmacodynamic Assay
 The Need for a PARP in vivo Pharmacodynamic Assay Jay George, Ph.D., Chief Scientific Officer, Trevigen, Inc., Gaithersburg, MD For further infomation, please contact: William Booth, Ph.D. Tel: +44 (0)1235
The Need for a PARP in vivo Pharmacodynamic Assay Jay George, Ph.D., Chief Scientific Officer, Trevigen, Inc., Gaithersburg, MD For further infomation, please contact: William Booth, Ph.D. Tel: +44 (0)1235
Cancer Risk Factors in Ontario. Dusts and Fibres
 Cancer Risk Factors in Ontario Dusts and Fibres dusts ANd fibres risk factor/exposure Cancer The context where high risks were reported Magnitude of risk* Strength of evidence a Asbestos Larynx Occupational
Cancer Risk Factors in Ontario Dusts and Fibres dusts ANd fibres risk factor/exposure Cancer The context where high risks were reported Magnitude of risk* Strength of evidence a Asbestos Larynx Occupational
Lack of Interaction between Asbestos Exposure and Glutathione S-Transferase M1 and T1 Genotypes in Lung Carcinogenesis
 Vol. 10, 1253 1258, December 2001 Cancer Epidemiology, Biomarkers & Prevention 1253 Lack of Interaction between Asbestos Exposure and Glutathione S-Transferase M1 and T1 Genotypes in Lung Carcinogenesis
Vol. 10, 1253 1258, December 2001 Cancer Epidemiology, Biomarkers & Prevention 1253 Lack of Interaction between Asbestos Exposure and Glutathione S-Transferase M1 and T1 Genotypes in Lung Carcinogenesis
Biological effects of naturally occurring and man-made fibres: in vitro cytotoxicity and mutagenesis in mammalian cells
 British Journal of Cancer (1999) 79(9/10), 1319 1324 1999 Cancer Research Campaign Article no. bjoc.1998.0213 Biological effects of naturally occurring and man-made fibres: in vitro cytotoxicity and mutagenesis
British Journal of Cancer (1999) 79(9/10), 1319 1324 1999 Cancer Research Campaign Article no. bjoc.1998.0213 Biological effects of naturally occurring and man-made fibres: in vitro cytotoxicity and mutagenesis
B Cell Generation, Activation & Differentiation. B cell maturation
 B Cell Generation, Activation & Differentiation Naïve B cells- have not encountered Ag. Have IgM and IgD on cell surface : have same binding VDJ regions but different constant region leaves bone marrow
B Cell Generation, Activation & Differentiation Naïve B cells- have not encountered Ag. Have IgM and IgD on cell surface : have same binding VDJ regions but different constant region leaves bone marrow
Chapter 8. Summary and Perspectives
 Chapter 8 Summary and Perspectives 131 Chapter 8 Summary Overexpression of the multidrug resistance protein MRP1 confer multidrug resistance (MDR) to cancer cells. The contents of this thesis describe
Chapter 8 Summary and Perspectives 131 Chapter 8 Summary Overexpression of the multidrug resistance protein MRP1 confer multidrug resistance (MDR) to cancer cells. The contents of this thesis describe
The Physiology of Hyperbaric Oxygen Therapy. Free Radicals and Reactive Oxygen Species. I. Introduction Definition, Source, function and Purpose
 The Physiology of Hyperbaric Oxygen Therapy Free Radicals and Reactive Oxygen Species I. Introduction Definition, Source, function and Purpose A. Definition of free radicals and reactive oxygen species
The Physiology of Hyperbaric Oxygen Therapy Free Radicals and Reactive Oxygen Species I. Introduction Definition, Source, function and Purpose A. Definition of free radicals and reactive oxygen species
MULTIPLE MYELOMA. Dr Malkit S Riyat. MBChB, FRCPath(UK) Consultant Haematologist
 MULTIPLE MYELOMA Dr Malkit S Riyat MBChB, FRCPath(UK) Consultant Haematologist Multiple myeloma is an incurable malignancy that arises from postgerminal centre, somatically hypermutated B cells.
MULTIPLE MYELOMA Dr Malkit S Riyat MBChB, FRCPath(UK) Consultant Haematologist Multiple myeloma is an incurable malignancy that arises from postgerminal centre, somatically hypermutated B cells.
What is Cancer? Cancer is a genetic disease: Cancer typically involves a change in gene expression/function:
 Cancer is a genetic disease: Inherited cancer Sporadic cancer What is Cancer? Cancer typically involves a change in gene expression/function: Qualitative change Quantitative change Any cancer causing genetic
Cancer is a genetic disease: Inherited cancer Sporadic cancer What is Cancer? Cancer typically involves a change in gene expression/function: Qualitative change Quantitative change Any cancer causing genetic
Multiple Mechanisms for the Carcinogenic Effects of Asbestos and Other Mineral Fibers by J. Carl Barrett,* Patricia W. Lamb,* and Roger W.
 Environmental Health Perspectives Vol. 81, pp. 81-89, 1989 Multiple Mechanisms for the Carcinogenic Effects of Asbestos and Other Mineral Fibers by J. Carl Barrett,* Patricia W. Lamb,* and Roger W. Wiseman*
Environmental Health Perspectives Vol. 81, pp. 81-89, 1989 Multiple Mechanisms for the Carcinogenic Effects of Asbestos and Other Mineral Fibers by J. Carl Barrett,* Patricia W. Lamb,* and Roger W. Wiseman*
Oxidant and antioxidant mechanisms of lung disease caused by asbestos fibres
 Eur Respir J 1999; 14: 706±716 Printed in UK ± all rights reserved Copyright #ERS Journals Ltd 1999 European Respiratory Journal ISSN 0903-1936 REVIEW Oxidant and antioxidant mechanisms of lung disease
Eur Respir J 1999; 14: 706±716 Printed in UK ± all rights reserved Copyright #ERS Journals Ltd 1999 European Respiratory Journal ISSN 0903-1936 REVIEW Oxidant and antioxidant mechanisms of lung disease
Pharmacology of the Respiratory Tract: COPD and Steroids
 Pharmacology of the Respiratory Tract: COPD and Steroids Dr. Tillie-Louise Hackett Department of Anesthesiology, Pharmacology and Therapeutics University of British Columbia Associate Head, Centre of Heart
Pharmacology of the Respiratory Tract: COPD and Steroids Dr. Tillie-Louise Hackett Department of Anesthesiology, Pharmacology and Therapeutics University of British Columbia Associate Head, Centre of Heart
Asbestos, Asbestosis, and Lung Cancer
 Asbestos, Asbestosis, and Lung Cancer David Weill, M.D. Stanford University Medical Center Stanford, CA David Weill, M.D., is a professor of medicine in the Division of Pulmonary and Critical Care Medicine
Asbestos, Asbestosis, and Lung Cancer David Weill, M.D. Stanford University Medical Center Stanford, CA David Weill, M.D., is a professor of medicine in the Division of Pulmonary and Critical Care Medicine
treatments) worked by killing cancerous cells using chemo or radiotherapy. While these techniques can
 Shristi Pandey Genomics and Medicine Winter 2011 Prof. Doug Brutlag Chronic Myeloid Leukemia: A look into how genomics is changing the way we treat Cancer. Until the late 1990s, nearly all treatment methods
Shristi Pandey Genomics and Medicine Winter 2011 Prof. Doug Brutlag Chronic Myeloid Leukemia: A look into how genomics is changing the way we treat Cancer. Until the late 1990s, nearly all treatment methods
Cell Division Mitosis and the Cell Cycle
 Cell Division Mitosis and the Cell Cycle A Chromosome and Sister Chromatids Key Points About Chromosome Structure A chromosome consists of DNA that is wrapped around proteins (histones) and condensed Each
Cell Division Mitosis and the Cell Cycle A Chromosome and Sister Chromatids Key Points About Chromosome Structure A chromosome consists of DNA that is wrapped around proteins (histones) and condensed Each
Chapter 12: The Cell Cycle
 Name Period Chapter 12: The Cell Cycle Overview: 1. What are the three key roles of cell division? State each role, and give an example. Key Role Reproduction Growth and development Tissue removal Example
Name Period Chapter 12: The Cell Cycle Overview: 1. What are the three key roles of cell division? State each role, and give an example. Key Role Reproduction Growth and development Tissue removal Example
Criteria for Attributing Lung Cancer to Asbestos Exposure
 Criteria for Attributing Lung Cancer to Asbestos Exposure Philip T. Cagle, MD The article by Mollo and coworkers 1 examines the criteria for attribution of lung cancers to asbestos exposure, suggesting
Criteria for Attributing Lung Cancer to Asbestos Exposure Philip T. Cagle, MD The article by Mollo and coworkers 1 examines the criteria for attribution of lung cancers to asbestos exposure, suggesting
June 20, 2002. 2002.06.20: Wagner Testimony on Workplace Exposure to Asbestos. This is an archive page. The links are no longer being updated.
 Page 1 of 6 skip navigational links This is an archive page. The links are no longer being updated. Statement by Gregory R. Wagner, M.D. Director, Division of Respiratory Disease Studies National Institute
Page 1 of 6 skip navigational links This is an archive page. The links are no longer being updated. Statement by Gregory R. Wagner, M.D. Director, Division of Respiratory Disease Studies National Institute
The molecular epidemiology of asbestos and tobacco in lung cancer
 (2002) 21, 7284 7288 ª 2002 Nature Publishing Group All rights reserved 0950 9232/02 $25.00 www.nature.com/onc The molecular epidemiology of asbestos and tobacco in lung cancer Heather H Nelson 1 and Karl
(2002) 21, 7284 7288 ª 2002 Nature Publishing Group All rights reserved 0950 9232/02 $25.00 www.nature.com/onc The molecular epidemiology of asbestos and tobacco in lung cancer Heather H Nelson 1 and Karl
A disease of populations of cells that live, divide, invade and spread without regard to normal limits
 1 Targeted Cancer Therapies Mark McKeage Medical Oncology Specialist Professor in Clinical Pharmacology 2 Cancer Definition- A disease of populations of cells that live, divide, invade and spread without
1 Targeted Cancer Therapies Mark McKeage Medical Oncology Specialist Professor in Clinical Pharmacology 2 Cancer Definition- A disease of populations of cells that live, divide, invade and spread without
How Cancer Begins???????? Chithra Manikandan Nov 2009
 Cancer Cancer is one of the most common diseases in the developed world: 1 in 4 deaths are due to cancer 1 in 17 deaths are due to lung cancer Lung cancer is the most common cancer in men Breast cancer
Cancer Cancer is one of the most common diseases in the developed world: 1 in 4 deaths are due to cancer 1 in 17 deaths are due to lung cancer Lung cancer is the most common cancer in men Breast cancer
Producing Analytical Data, Microscopy and Analytical Procedures
 1 The Role of Fiber Analysis in Asbestos Induced Lung Disease: TEM vs. SEM. Is There Controversy Elizabeth N. Pavlisko, M.D., Department of Pathology, Duke University Medical Center I. Introduction to
1 The Role of Fiber Analysis in Asbestos Induced Lung Disease: TEM vs. SEM. Is There Controversy Elizabeth N. Pavlisko, M.D., Department of Pathology, Duke University Medical Center I. Introduction to
Occupational respiratory diseases due to Asbestos. Dirk Dahmann, IGF, Bochum
 Occupational respiratory diseases due to Asbestos Dirk Dahmann, IGF, Bochum Contents Introduction Diseases Further Effects Preventive Strategies Conclusion Asbestos minerals Woitowitz, 2003 Imports (+
Occupational respiratory diseases due to Asbestos Dirk Dahmann, IGF, Bochum Contents Introduction Diseases Further Effects Preventive Strategies Conclusion Asbestos minerals Woitowitz, 2003 Imports (+
Pathologist s Discussion of Plaintiffs Latest Theories
 Pathologist s Discussion of Plaintiffs Latest Theories Mary Beth Beasley, MD Mt. Sinai Medical Center Annenberg Building 15th Floor Room 50 1468 Madison Avenue New York, NY 10029 (212) 241-5307 mbbeasleymd@yahoo.com
Pathologist s Discussion of Plaintiffs Latest Theories Mary Beth Beasley, MD Mt. Sinai Medical Center Annenberg Building 15th Floor Room 50 1468 Madison Avenue New York, NY 10029 (212) 241-5307 mbbeasleymd@yahoo.com
DNA Damage and Repair
 infoaging guides BIOLOGY OF AGING DNA Damage and Repair An introduction to aging science brought to you by the American Federation for Aging Research DNA BASICS DNA stands for deoxyribonucleic acid. The
infoaging guides BIOLOGY OF AGING DNA Damage and Repair An introduction to aging science brought to you by the American Federation for Aging Research DNA BASICS DNA stands for deoxyribonucleic acid. The
Molecular Alterations in Asbestos-Related Lung Cancer
 Penny Nymark Molecular Alterations in Asbestos-Related Lung Cancer People and Work Research Reports 90 Molecular Alterations in Asbestos-Related Lung Cancer Penny Nymark People and Work Research Reports
Penny Nymark Molecular Alterations in Asbestos-Related Lung Cancer People and Work Research Reports 90 Molecular Alterations in Asbestos-Related Lung Cancer Penny Nymark People and Work Research Reports
Publikationsliste Claudia Götz
 Publikationsliste Claudia Götz 1. Reinhard,B., Götz, C., and Faillard, H.: Synthesis of N-Acetyl-9-Oacetylneuraminic acid α-p-aminophenylthioketoside and its application as ligand in the affinity chromatography
Publikationsliste Claudia Götz 1. Reinhard,B., Götz, C., and Faillard, H.: Synthesis of N-Acetyl-9-Oacetylneuraminic acid α-p-aminophenylthioketoside and its application as ligand in the affinity chromatography
HANDLING LUNG CANCER CLAIMS
 HANDLING LUNG CANCER CLAIMS JENNIFER S. KILPATRICK SWANSON, MARTIN & BELL, LLP 330 North Wabash Avenue Suite 3300 Chicago, Illinois 60611-3604 (312) 321-3517 (312) 321-0990 jkilpatrick@smbtrials.com 1
HANDLING LUNG CANCER CLAIMS JENNIFER S. KILPATRICK SWANSON, MARTIN & BELL, LLP 330 North Wabash Avenue Suite 3300 Chicago, Illinois 60611-3604 (312) 321-3517 (312) 321-0990 jkilpatrick@smbtrials.com 1
Influence of Fiber Type, Size, and Number in Human Disease: Conclusions from Fiber Burden Analysis
 Influence of Fiber Type, Size, and Number in Human Disease: Conclusions from Fiber Burden Analysis Andrew Churg, MD Department of Pathology University of British Columbia Vancouver, BC, Canada Techniques,
Influence of Fiber Type, Size, and Number in Human Disease: Conclusions from Fiber Burden Analysis Andrew Churg, MD Department of Pathology University of British Columbia Vancouver, BC, Canada Techniques,
- Compensation issues
 Charité- Universitätsmedizin Berlin Institut für Arbeitsmedizin Prof. Dr. med. X. Baur Prevention, recognition and compensation of asbestosinduced diseases (AD) - Which diseases are asbestos-related? -
Charité- Universitätsmedizin Berlin Institut für Arbeitsmedizin Prof. Dr. med. X. Baur Prevention, recognition and compensation of asbestosinduced diseases (AD) - Which diseases are asbestos-related? -
Naturally Occurring Asbestos (NOA) General Overview
 Naturally Occurring Asbestos (NOA) General Overview Lee R. Shull PhD Health, Ecology and Risk Practice MWH Global Sacramento, CA CASH NOA Workshop January 24, 2006 Presentation Outline Brief background
Naturally Occurring Asbestos (NOA) General Overview Lee R. Shull PhD Health, Ecology and Risk Practice MWH Global Sacramento, CA CASH NOA Workshop January 24, 2006 Presentation Outline Brief background
Griffith University - Case for Support. Mesothelioma Research Program
 Griffith University - Case for Support Mesothelioma Research Program Professor Lyn Griffiths Director, Dean, Research and Director, Genomics Research Centre (GHI) Established in 2007. Integrated health
Griffith University - Case for Support Mesothelioma Research Program Professor Lyn Griffiths Director, Dean, Research and Director, Genomics Research Centre (GHI) Established in 2007. Integrated health
Cellular Reproduction
 9 Cellular Reproduction section 1 Cellular Growth Before You Read Think about the life cycle of a human. On the lines below, write some of the stages that occur in the life cycle of a human. In this section,
9 Cellular Reproduction section 1 Cellular Growth Before You Read Think about the life cycle of a human. On the lines below, write some of the stages that occur in the life cycle of a human. In this section,
Introduction. Pathogenesis of type 2 diabetes
 Introduction Type 2 diabetes mellitus (t2dm) is the most prevalent form of diabetes worldwide. It is characterised by high fasting and high postprandial blood glucose concentrations (hyperglycemia). Chronic
Introduction Type 2 diabetes mellitus (t2dm) is the most prevalent form of diabetes worldwide. It is characterised by high fasting and high postprandial blood glucose concentrations (hyperglycemia). Chronic
Francine Lortie-Monette, MD, MSc, CSPQ, MBA Department of Epidemiology and Biostatistics University of Western Ontario 2003
 ASBESTOS Francine Lortie-Monette, MD, MSc, CSPQ, MBA Department of Epidemiology and Biostatistics University of Western Ontario 2003 Asbestosis Asbestosis is a model for other dust diseases as well as
ASBESTOS Francine Lortie-Monette, MD, MSc, CSPQ, MBA Department of Epidemiology and Biostatistics University of Western Ontario 2003 Asbestosis Asbestosis is a model for other dust diseases as well as
CHAPTER 2: UNDERSTANDING CANCER
 CHAPTER 2: UNDERSTANDING CANCER INTRODUCTION We are witnessing an era of great discovery in the field of cancer research. New insights into the causes and development of cancer are emerging. These discoveries
CHAPTER 2: UNDERSTANDING CANCER INTRODUCTION We are witnessing an era of great discovery in the field of cancer research. New insights into the causes and development of cancer are emerging. These discoveries
Cancer - Dangers in the Process of High Butadiene and Network Marketing
 Remarks on the cancer risk values of 1,3-butadiene (draft Dutch Health Council 2012) Author: Wil ten Berge Date: 19 January 2013 1 Introduction The Dutch Health Council (DHC) has issued a proposal for
Remarks on the cancer risk values of 1,3-butadiene (draft Dutch Health Council 2012) Author: Wil ten Berge Date: 19 January 2013 1 Introduction The Dutch Health Council (DHC) has issued a proposal for
Autoimmunity and immunemediated. FOCiS. Lecture outline
 1 Autoimmunity and immunemediated inflammatory diseases Abul K. Abbas, MD UCSF FOCiS 2 Lecture outline Pathogenesis of autoimmunity: why selftolerance fails Genetics of autoimmune diseases Therapeutic
1 Autoimmunity and immunemediated inflammatory diseases Abul K. Abbas, MD UCSF FOCiS 2 Lecture outline Pathogenesis of autoimmunity: why selftolerance fails Genetics of autoimmune diseases Therapeutic
Mutations: 2 general ways to alter DNA. Mutations. What is a mutation? Mutations are rare. Changes in a single DNA base. Change a single DNA base
 Mutations Mutations: 2 general ways to alter DNA Change a single DNA base Or entire sections of DNA can move from one place to another What is a mutation? Any change in the nucleotide sequence of DNA Here
Mutations Mutations: 2 general ways to alter DNA Change a single DNA base Or entire sections of DNA can move from one place to another What is a mutation? Any change in the nucleotide sequence of DNA Here
Current Usage and Health Significance of the Modern Use of Chrysotile Products: Review of Recently Published Evidence
 Current Usage and Health Significance of the Modern Use of Chrysotile Products: Review of Recently Published Evidence John Hoskins Health & Safety Consultant, Haslemere, Surrey, UK ASBESTOS SERPENTINE
Current Usage and Health Significance of the Modern Use of Chrysotile Products: Review of Recently Published Evidence John Hoskins Health & Safety Consultant, Haslemere, Surrey, UK ASBESTOS SERPENTINE
Appendix C DNA Replication & Mitosis
 K.Muma Bio 6 Appendix C DNA Replication & Mitosis Study Objectives: Appendix C: DNA replication and Mitosis 1. Describe the structure of DNA and where it is found. 2. Explain complimentary base pairing:
K.Muma Bio 6 Appendix C DNA Replication & Mitosis Study Objectives: Appendix C: DNA replication and Mitosis 1. Describe the structure of DNA and where it is found. 2. Explain complimentary base pairing:
If and when cancer cells stop dividing, they do so at random points, not at the normal checkpoints in the cell cycle.
 Cancer cells have escaped from cell cycle controls Cancer cells divide excessively and invade other tissues because they are free of the body s control mechanisms. Cancer cells do not stop dividing when
Cancer cells have escaped from cell cycle controls Cancer cells divide excessively and invade other tissues because they are free of the body s control mechanisms. Cancer cells do not stop dividing when
Uses and Abuses of Pathology in Asbestos-exposed Populations
 Uses and Abuses of Pathology in Asbestos-exposed Populations Jerrold L. Abraham, MD Department of Pathology State University of New York Upstate Medical University Syracuse, NY, 13210 USA The term: Asbestosis,
Uses and Abuses of Pathology in Asbestos-exposed Populations Jerrold L. Abraham, MD Department of Pathology State University of New York Upstate Medical University Syracuse, NY, 13210 USA The term: Asbestosis,
Asbestos: health effects and risk. Peter Franklin Senior Scientific Officer, EHD Senior Research Fellow, UWA
 Asbestos: health effects and risk Peter Franklin Senior Scientific Officer, EHD Senior Research Fellow, UWA What is asbestos Naturally occurring mineral that has crystallised to form long thin fibres and
Asbestos: health effects and risk Peter Franklin Senior Scientific Officer, EHD Senior Research Fellow, UWA What is asbestos Naturally occurring mineral that has crystallised to form long thin fibres and
M. Ugolini 1,2, G. Babini 1,2, G. Baiocco 1,2, D. Cappelletti 1,2, L. Mariotti 1,2, J. Morini 1,2,3 and A. Ottolenghi 1,2
 M. Ugolini 1,2, G. Babini 1,2, G. Baiocco 1,2, D. Cappelletti 1,2, L. Mariotti 1,2, J. Morini 1,2,3 and A. Ottolenghi 1,2 1 Department of Physics, University of Pavia, Pavia, Italy 2 Istituto Nazionale
M. Ugolini 1,2, G. Babini 1,2, G. Baiocco 1,2, D. Cappelletti 1,2, L. Mariotti 1,2, J. Morini 1,2,3 and A. Ottolenghi 1,2 1 Department of Physics, University of Pavia, Pavia, Italy 2 Istituto Nazionale
Non Small Cell Lung Cancer: Scientific Discoveries and the Pursuit of Progress
 Non Small Cell Lung Cancer: Scientific Discoveries and the Pursuit of Progress Lung Cancer Accounts for 14% of All New Cancer Diagnoses in the United States 1 Lung cancer is the second most common malignancy
Non Small Cell Lung Cancer: Scientific Discoveries and the Pursuit of Progress Lung Cancer Accounts for 14% of All New Cancer Diagnoses in the United States 1 Lung cancer is the second most common malignancy
LESSON 3.5 WORKBOOK. How do cancer cells evolve? Workbook Lesson 3.5
 LESSON 3.5 WORKBOOK How do cancer cells evolve? In this unit we have learned how normal cells can be transformed so that they stop behaving as part of a tissue community and become unresponsive to regulation.
LESSON 3.5 WORKBOOK How do cancer cells evolve? In this unit we have learned how normal cells can be transformed so that they stop behaving as part of a tissue community and become unresponsive to regulation.
Unit 1 Higher Human Biology Summary Notes
 Unit 1 Higher Human Biology Summary Notes a. Cells tissues organs body systems Division of labour occurs in multicellular organisms (rather than each cell carrying out every function) Most cells become
Unit 1 Higher Human Biology Summary Notes a. Cells tissues organs body systems Division of labour occurs in multicellular organisms (rather than each cell carrying out every function) Most cells become
Antibody Function & Structure
 Antibody Function & Structure Specifically bind to antigens in both the recognition phase (cellular receptors) and during the effector phase (synthesis and secretion) of humoral immunity Serology: the
Antibody Function & Structure Specifically bind to antigens in both the recognition phase (cellular receptors) and during the effector phase (synthesis and secretion) of humoral immunity Serology: the
Summary and conclusion 2013
 The work presented in the thesis is focused on the problems related to the prostate gland. Benign prostatic hyperplasia (BPH) and prostate cancer (PCa) are the two major problems associated with prostate.
The work presented in the thesis is focused on the problems related to the prostate gland. Benign prostatic hyperplasia (BPH) and prostate cancer (PCa) are the two major problems associated with prostate.
Asbestos related cancers
 New cancer cases 1954-215 in Finland, Men Pukkala et al. 26 Asbestos related cancers Panu Oksa, MD, docent Course on asbestos-related diseases Tartu 4-5.12.26 Asbestos related cancer / FIOH / PO / 1.1.27
New cancer cases 1954-215 in Finland, Men Pukkala et al. 26 Asbestos related cancers Panu Oksa, MD, docent Course on asbestos-related diseases Tartu 4-5.12.26 Asbestos related cancer / FIOH / PO / 1.1.27
Asbestos. Toxicological overview
 Asbestos Toxicological overview Kinetics and metabolism Key Points Short asbestos fibres are deposited in the upper respiratory tract where they are cleared by mucociliary action Longer fibres are carried
Asbestos Toxicological overview Kinetics and metabolism Key Points Short asbestos fibres are deposited in the upper respiratory tract where they are cleared by mucociliary action Longer fibres are carried
Identification of Specific Gene Copy Number Changes in Asbestos-Related Lung Cancer
 Research Article Identification of Specific Gene Copy Number Changes in Asbestos-Related Lung Cancer Penny Nymark, 1,2 Harriet Wikman, 2,4 Salla Ruosaari, 3 Jaakko Hollmén, 3 Esa Vanhala, 2 Antti Karjalainen,
Research Article Identification of Specific Gene Copy Number Changes in Asbestos-Related Lung Cancer Penny Nymark, 1,2 Harriet Wikman, 2,4 Salla Ruosaari, 3 Jaakko Hollmén, 3 Esa Vanhala, 2 Antti Karjalainen,
Serial Review: Role of Reactive Oxygen and Nitrogen Species (ROS/RNS) in Lung Injury and Diseases Guest Editor: Brooke T. Mossman
 doi:10.1016/s0891-5849(03)00060-1 Free Radical Biology & Medicine, Vol. 34, No. 9, pp. 1117 1129, 2003 Copyright 2003 Elsevier Inc. Printed in the USA. All rights reserved 0891-5849/03/$ see front matter
doi:10.1016/s0891-5849(03)00060-1 Free Radical Biology & Medicine, Vol. 34, No. 9, pp. 1117 1129, 2003 Copyright 2003 Elsevier Inc. Printed in the USA. All rights reserved 0891-5849/03/$ see front matter
Department of BioScience Technology Chung Yuan Christian University 2015/08/13
 Department of BioScience Technology Chung Yuan Christian University 2015/08/13 Cancer Cells Cancer, the 1st leading cause of death, is an example of a disease that arises from abnormalities in cell function
Department of BioScience Technology Chung Yuan Christian University 2015/08/13 Cancer Cells Cancer, the 1st leading cause of death, is an example of a disease that arises from abnormalities in cell function
Chapter 5: Organization and Expression of Immunoglobulin Genes
 Chapter 5: Organization and Expression of Immunoglobulin Genes I. Genetic Model Compatible with Ig Structure A. Two models for Ab structure diversity 1. Germ-line theory: maintained that the genome contributed
Chapter 5: Organization and Expression of Immunoglobulin Genes I. Genetic Model Compatible with Ig Structure A. Two models for Ab structure diversity 1. Germ-line theory: maintained that the genome contributed
Induction of 8-hydroxydeoxyguanosine by man made vitreous fibres and crocidolite asbestos administered intraperitoneally in rats
 Mutation Research 553 (24) 59 65 Induction of 8-hydroxydeoxyguanosine by man made vitreous fibres and crocidolite asbestos administered intraperitoneally in rats Claudia Schürkes, Winfried Brock, Josef
Mutation Research 553 (24) 59 65 Induction of 8-hydroxydeoxyguanosine by man made vitreous fibres and crocidolite asbestos administered intraperitoneally in rats Claudia Schürkes, Winfried Brock, Josef
Asbestos. General information
 Asbestos General information Key Points Fire Non flammable and non combustible under normal conditions Chemically inert under normal conditions. Resistant to most solvents, acids and alkalis In the event
Asbestos General information Key Points Fire Non flammable and non combustible under normal conditions Chemically inert under normal conditions. Resistant to most solvents, acids and alkalis In the event
Possible mechanism of cell phone radiationinduced
 Possible mechanism of cell phone radiationinduced cancer Dariusz Leszczynski STUK Radiation and Nuclear Safety Authority, Helsinki Finland Swinburne University of Technology, Hawthorn/Melbourne, Australia
Possible mechanism of cell phone radiationinduced cancer Dariusz Leszczynski STUK Radiation and Nuclear Safety Authority, Helsinki Finland Swinburne University of Technology, Hawthorn/Melbourne, Australia
What is nanotoxicology?
 What is nanotoxicology? How to estimate the potential hazard related to nanoparticles? Dissemination report October 2008 DR-225 200810-5 Project ID NMP2-CT-2005-515843 An European Integrated Project Supported
What is nanotoxicology? How to estimate the potential hazard related to nanoparticles? Dissemination report October 2008 DR-225 200810-5 Project ID NMP2-CT-2005-515843 An European Integrated Project Supported
Introduction. Cancer Biology. Tumor-suppressor genes. Proto-oncogenes. DNA stability genes. Mechanisms of carcinogenesis.
 Cancer Biology Chapter 18 Eric J. Hall., Amato Giaccia, Radiobiology for the Radiologist Introduction Tissue homeostasis depends on the regulated cell division and self-elimination (programmed cell death)
Cancer Biology Chapter 18 Eric J. Hall., Amato Giaccia, Radiobiology for the Radiologist Introduction Tissue homeostasis depends on the regulated cell division and self-elimination (programmed cell death)
State of the Art. Mechanisms in the Pathogenesis of Asbestosis and Silicosis. BROOKE T. MOSSMAN and ANDREW CHURG CONTENTS
 State of the Art Mechanisms in the Pathogenesis of Asbestosis and Silicosis BROOKE T. MOSSMAN and ANDREW CHURG Departments of Pathology, University of Vermont College of Medicine, Burlington, Vermont;
State of the Art Mechanisms in the Pathogenesis of Asbestosis and Silicosis BROOKE T. MOSSMAN and ANDREW CHURG Departments of Pathology, University of Vermont College of Medicine, Burlington, Vermont;
Cancer SBL101. James Gomes School of Biological Sciences Indian Institute of Technology Delhi
 Cancer SBL101 James Gomes School of Biological Sciences Indian Institute of Technology Delhi All Figures in this Lecture are taken from 1. Molecular biology of the cell / Bruce Alberts et al., 5th ed.
Cancer SBL101 James Gomes School of Biological Sciences Indian Institute of Technology Delhi All Figures in this Lecture are taken from 1. Molecular biology of the cell / Bruce Alberts et al., 5th ed.
How To Determine The Risk Of Mesothelioma In Brake Workers
 1 Risk Anal. 2004 Jun;24(3):547-52. Related Articles, Links Mesothelioma among brake mechanics: an expanded analysis of a casecontrol study. Hessel PA, Teta MJ, Goodman M, Lau E. Exponent, Wood Dale, IL
1 Risk Anal. 2004 Jun;24(3):547-52. Related Articles, Links Mesothelioma among brake mechanics: an expanded analysis of a casecontrol study. Hessel PA, Teta MJ, Goodman M, Lau E. Exponent, Wood Dale, IL
Lecture 11 The Cell Cycle and Mitosis
 Lecture 11 The Cell Cycle and Mitosis In this lecture Cell division Chromosomes The cell cycle Mitosis PPMAT Apoptosis What is cell division? Cells divide in order to reproduce themselves The cell cycle
Lecture 11 The Cell Cycle and Mitosis In this lecture Cell division Chromosomes The cell cycle Mitosis PPMAT Apoptosis What is cell division? Cells divide in order to reproduce themselves The cell cycle
Gene mutation and molecular medicine Chapter 15
 Gene mutation and molecular medicine Chapter 15 Lecture Objectives What Are Mutations? How Are DNA Molecules and Mutations Analyzed? How Do Defective Proteins Lead to Diseases? What DNA Changes Lead to
Gene mutation and molecular medicine Chapter 15 Lecture Objectives What Are Mutations? How Are DNA Molecules and Mutations Analyzed? How Do Defective Proteins Lead to Diseases? What DNA Changes Lead to
The immune response Antibodies Antigens Epitopes (antigenic determinants) the part of a protein antigen recognized by an antibody Haptens small
 The immune response Antibodies Antigens Epitopes (antigenic determinants) the part of a protein antigen recognized by an antibody Haptens small molecules that can elicit an immune response when linked
The immune response Antibodies Antigens Epitopes (antigenic determinants) the part of a protein antigen recognized by an antibody Haptens small molecules that can elicit an immune response when linked
A Genetic Analysis of Rheumatoid Arthritis
 A Genetic Analysis of Rheumatoid Arthritis Introduction to Rheumatoid Arthritis: Classification and Diagnosis Rheumatoid arthritis is a chronic inflammatory disorder that affects mainly synovial joints.
A Genetic Analysis of Rheumatoid Arthritis Introduction to Rheumatoid Arthritis: Classification and Diagnosis Rheumatoid arthritis is a chronic inflammatory disorder that affects mainly synovial joints.
specific B cells Humoral immunity lymphocytes antibodies B cells bone marrow Cell-mediated immunity: T cells antibodies proteins
 Adaptive Immunity Chapter 17: Adaptive (specific) Immunity Bio 139 Dr. Amy Rogers Host defenses that are specific to a particular infectious agent Can be innate or genetic for humans as a group: most microbes
Adaptive Immunity Chapter 17: Adaptive (specific) Immunity Bio 139 Dr. Amy Rogers Host defenses that are specific to a particular infectious agent Can be innate or genetic for humans as a group: most microbes
PROTANDIM SEVEN PEER-REVIEWED SCIENTIFIC JOURNAL ABSTRACTS SINCE 2006. A Fundamentally New Approach to Antioxidant Therapy
 PROTANDIM SEVEN PEER-REVIEWED SCIENTIFIC JOURNAL ABSTRACTS SINCE 2006 A Fundamentally New Approach to Antioxidant Therapy Free Radical Biology & Medicine 40 (2006) 341 347 Original Contribution The induction
PROTANDIM SEVEN PEER-REVIEWED SCIENTIFIC JOURNAL ABSTRACTS SINCE 2006 A Fundamentally New Approach to Antioxidant Therapy Free Radical Biology & Medicine 40 (2006) 341 347 Original Contribution The induction
MUTATION, DNA REPAIR AND CANCER
 MUTATION, DNA REPAIR AND CANCER 1 Mutation A heritable change in the genetic material Essential to the continuity of life Source of variation for natural selection New mutations are more likely to be harmful
MUTATION, DNA REPAIR AND CANCER 1 Mutation A heritable change in the genetic material Essential to the continuity of life Source of variation for natural selection New mutations are more likely to be harmful
Scientific Update on Safe Use of Asbestos. Robert P. Nolan, PhD International Environmental Research Foundation New York, New York www.ierfinc.
 Scientific Update on Safe Use of Asbestos Robert P. Nolan, PhD International Environmental Research Foundation New York, New York www.ierfinc.org When We Talk about Asbestos What Do We Mean? Anthophyllite
Scientific Update on Safe Use of Asbestos Robert P. Nolan, PhD International Environmental Research Foundation New York, New York www.ierfinc.org When We Talk about Asbestos What Do We Mean? Anthophyllite
PART 3.3: MicroRNA and Cancer
 BIBM 2010 Tutorial: Epigenomics and Cancer PART 3.3: MicroRNA and Cancer Dec 18, 2010 Sun Kim at Indiana University Outline of Part 3.3 Background on microrna Role of microrna in cancer MicroRNA pathway
BIBM 2010 Tutorial: Epigenomics and Cancer PART 3.3: MicroRNA and Cancer Dec 18, 2010 Sun Kim at Indiana University Outline of Part 3.3 Background on microrna Role of microrna in cancer MicroRNA pathway
4.1 3T12 and 312 are immortalized cell lines with transforming potential:
 DISCUSSION CHAPTER 4 4.1 3T12 and 312 are immortalized cell lines with transforming potential: Transformation of a normal cell with finite number of divisions into a tumorigenic cell of potentially infinite
DISCUSSION CHAPTER 4 4.1 3T12 and 312 are immortalized cell lines with transforming potential: Transformation of a normal cell with finite number of divisions into a tumorigenic cell of potentially infinite
Translating DNA repair pathways into therapeutic targets: beyond the BRCA1/2 and PARP inhibitor saga. Jorge S Reis-Filho, MD PhD FRCPath
 Translating DNA repair pathways into therapeutic targets: beyond the BRCA1/2 and PARP inhibitor saga Jorge S Reis-Filho, MD PhD FRCPath Summary How do PARP inhibitors work? Synthetic lethality Potential
Translating DNA repair pathways into therapeutic targets: beyond the BRCA1/2 and PARP inhibitor saga Jorge S Reis-Filho, MD PhD FRCPath Summary How do PARP inhibitors work? Synthetic lethality Potential
ASBESTOS. Know what it is and how you can protect yourself. environmental affairs Department: Environmental Affairs REPUBLIC OF SOUTH AFRICA
 ASBESTOS Know what it is and how you can protect yourself environmental affairs Department: Environmental Affairs REPUBLIC OF SOUTH AFRICA 1 What is asbestos? The term asbestos designates a group of naturally
ASBESTOS Know what it is and how you can protect yourself environmental affairs Department: Environmental Affairs REPUBLIC OF SOUTH AFRICA 1 What is asbestos? The term asbestos designates a group of naturally
1. When new cells are formed through the process of mitosis, the number of chromosomes in the new cells
 Cell Growth and Reproduction 1. When new cells are formed through the process of mitosis, the number of chromosomes in the new cells A. is half of that of the parent cell. B. remains the same as in the
Cell Growth and Reproduction 1. When new cells are formed through the process of mitosis, the number of chromosomes in the new cells A. is half of that of the parent cell. B. remains the same as in the
Classify chromosomes in a karyotype according to size and centromere position. Identify metacentric, submetacentric and acrocentric chromosomes
 Mitosis, Meiosis and the Cell Cycle Prof. Alfred Cuschieri University of Malta Department of Anatomy Objectives By the end of the session the student shoud be able to: Define the meaning of chromosomes
Mitosis, Meiosis and the Cell Cycle Prof. Alfred Cuschieri University of Malta Department of Anatomy Objectives By the end of the session the student shoud be able to: Define the meaning of chromosomes
HEALTH CARE FOR EXPOSURE TO ASBESTOS. 2010 The SafetyNet Centre for Occupational Health and Safety Research Memorial University www.safetynet.mun.
 HEALTH CARE FOR PATIENTS WITH EXPOSURE TO ASBESTOS 2010 The SafetyNet Centre for Occupational Health and Safety Research Memorial University www.safetynet.mun.ca HEALTH CARE FOR PATIENTS WITH EXPOSURE
HEALTH CARE FOR PATIENTS WITH EXPOSURE TO ASBESTOS 2010 The SafetyNet Centre for Occupational Health and Safety Research Memorial University www.safetynet.mun.ca HEALTH CARE FOR PATIENTS WITH EXPOSURE
BIOMARKERS AND TOXICITY MECHANISMS 06 Mechanisms Metabolism & Detoxification. Luděk Bláha, PřF MU, RECETOX www.recetox.cz
 BIOMARKERS AND TOXICITY MECHANISMS 06 Mechanisms Metabolism & Detoxification Luděk Bláha, PřF MU, RECETOX www.recetox.cz Metabolism and detoxification Chemicals enter body... mostly via food Pass directly
BIOMARKERS AND TOXICITY MECHANISMS 06 Mechanisms Metabolism & Detoxification Luděk Bláha, PřF MU, RECETOX www.recetox.cz Metabolism and detoxification Chemicals enter body... mostly via food Pass directly
REPORT PERSPECTIVES IN LUNG CANCER 2010 AMSTERDAM
 REPORT PERSPECTIVES IN LUNG CANCER 2010 AMSTERDAM Valerie Van Damme, Isabelle Wauters, Johan Vansteenkiste Univ. Hospital Leuven and Leuven Lung Cancer Group Introduction Perspectives in Lung Cancer (PILC)
REPORT PERSPECTIVES IN LUNG CANCER 2010 AMSTERDAM Valerie Van Damme, Isabelle Wauters, Johan Vansteenkiste Univ. Hospital Leuven and Leuven Lung Cancer Group Introduction Perspectives in Lung Cancer (PILC)
by Lee S. Newman, M.D., and Cecile S. Rose, M.D., M.P.H.
 OCCUPATIONAL ASBESTOSIS AND RELATED DISEASES by Lee S. Newman, M.D., and Cecile S. Rose, M.D., M.P.H. A 63-year-old man consulted an internist complaining of dyspnea on exertion. He reported the following:
OCCUPATIONAL ASBESTOSIS AND RELATED DISEASES by Lee S. Newman, M.D., and Cecile S. Rose, M.D., M.P.H. A 63-year-old man consulted an internist complaining of dyspnea on exertion. He reported the following:
Asbestos and Mesothelioma a briefing document for the Metropolitan Police
 Asbestos and Mesothelioma a briefing document for the Metropolitan Police Prepared by Professor John Cherrie, Heriot Watt University, Edinburgh, UK. Introduction The purpose of this document is to provide
Asbestos and Mesothelioma a briefing document for the Metropolitan Police Prepared by Professor John Cherrie, Heriot Watt University, Edinburgh, UK. Introduction The purpose of this document is to provide
1 page Overview. CONCURRENT 1D, 1E, 1F Biology & Pathogenesis Multi-Modality Immunology 1
 1 page Overview 21 Oct Tuesday 1500 on REGISTRATION 1800 Welcome Reception & Cocktails at the Cape Town International Conference Centre (CTICC) 22 Oct Wednesday 0730 REGISTRATION 0830 OPENING 0900 PLENARY
1 page Overview 21 Oct Tuesday 1500 on REGISTRATION 1800 Welcome Reception & Cocktails at the Cape Town International Conference Centre (CTICC) 22 Oct Wednesday 0730 REGISTRATION 0830 OPENING 0900 PLENARY
B Cells and Antibodies
 B Cells and Antibodies Andrew Lichtman, MD PhD Brigham and Women's Hospital Harvard Medical School Lecture outline Functions of antibodies B cell activation; the role of helper T cells in antibody production
B Cells and Antibodies Andrew Lichtman, MD PhD Brigham and Women's Hospital Harvard Medical School Lecture outline Functions of antibodies B cell activation; the role of helper T cells in antibody production
Basic Overview of Preclinical Toxicology Animal Models
 Basic Overview of Preclinical Toxicology Animal Models Charles D. Hebert, Ph.D., D.A.B.T. December 5, 2013 Outline Background In Vitro Toxicology In Vivo Toxicology Animal Models What is Toxicology? Background
Basic Overview of Preclinical Toxicology Animal Models Charles D. Hebert, Ph.D., D.A.B.T. December 5, 2013 Outline Background In Vitro Toxicology In Vivo Toxicology Animal Models What is Toxicology? Background
Quantification of Non-Fibrous and Fibrous Particulates in Human Lungs: Twenty Year Update on Pneumoconiosis Database
 Ann. occup. Hyg., Vol. 46, Supplement 1, pp. 397 401, 2002 2002 British Occupational Hygiene Society Published by Oxford University Press DOI: 10.1093/annhyg/mef694 Quantification of Non-Fibrous and Fibrous
Ann. occup. Hyg., Vol. 46, Supplement 1, pp. 397 401, 2002 2002 British Occupational Hygiene Society Published by Oxford University Press DOI: 10.1093/annhyg/mef694 Quantification of Non-Fibrous and Fibrous
LEUKEMIA LYMPHOMA MYELOMA Advances in Clinical Trials
 LEUKEMIA LYMPHOMA MYELOMA Advances in Clinical Trials OUR FOCUS ABOUT emerging treatments Presentation for: Judith E. Karp, MD Advancements for Acute Myelogenous Leukemia Supported by an unrestricted educational
LEUKEMIA LYMPHOMA MYELOMA Advances in Clinical Trials OUR FOCUS ABOUT emerging treatments Presentation for: Judith E. Karp, MD Advancements for Acute Myelogenous Leukemia Supported by an unrestricted educational
Lesson 3 Reading Material: Oncogenes and Tumor Suppressor Genes
 Lesson 3 Reading Material: Oncogenes and Tumor Suppressor Genes Becoming a cancer cell isn t easy One of the fundamental molecular characteristics of cancer is that it does not develop all at once, but
Lesson 3 Reading Material: Oncogenes and Tumor Suppressor Genes Becoming a cancer cell isn t easy One of the fundamental molecular characteristics of cancer is that it does not develop all at once, but
