Direct Freeform Fabrication of Seeded Hydrogels in Arbitrary Geometries
|
|
|
- Bennett Fisher
- 8 years ago
- Views:
Transcription
1 TISSUE ENGINEERING Volume 12, Number 5, 2006 Mary Ann Liebert, Inc. Direct Freeform Fabrication of Seeded Hydrogels in Arbitrary Geometries DANIEL L. COHEN, Ph.D., 1 EVAN MALONE, Ph.D., 1 HOD LIPSON, Ph.D., 1,2 and LAWRENCE J. BONASSAR, Ph.D. 1,3 ABSTRACT A major challenge in tissue engineering is the generation of cell-seeded implants with structures that mimic native tissue, both in anatomic geometries and intratissue cell distributions. By combining the strengths of injection molding tissue engineering with those of solid freeform fabrication (SFF), three-dimensional (3-D) pre-seeded implants were fabricated without custom-tooling, enabling efficient production of patient-specific implants. The incorporation of SFF technology also enabled the fabrication of geometrically complex, multiple-material implants with spatially heterogeneous properties that would otherwise be challenging to produce. Utilizing a custom-built robotic SFF platform and gel deposition tools, alginate hydrogel was used with calcium sulfate as a crosslinking agent to produce pre-seeded living implants of arbitrary geometries. The process was determined to be sterile and viable at 94 5%. The GAG and hydroxyproline production was found to be similar to that of other implants fabricated using the same materials with different shaping methods. The geometric fidelity of the process was quantified by using the printing platform as a computerized measurement machine (CMM); the RMS surface roughness of printed samples in the z-dimension was found to be mm. INTRODUCTION THE GENERATION OF CELL-SEEDED IMPLANTS with structures that mimic native tissue, both in anatomic geometries and intratissue cell distributions, is a major challenge in tissue engineering. Indeed, the therapeutic promise of tissue engineering was most famously demonstrated in pioneering work by Charles Vacanti and colleagues in which tissue-engineered cartilage was shaped into the form of a human ear. 1 In additional work, this idea was extended to demonstrate the generation of cartilage of arbitrary shapes, 2 as well as many specific anatomic geometries including temporomandibular disc 3 and joint, 4 meniscus, 5 trachea, 6 intervertebral disc, 7 nasal tip, 8 and nasal septum. 9 These principles apply to other tissues as well, particularly bone, where the reproduction of anatomically shaped tissue has been demonstrated in the generation of femoral shaft 10 and mandibular condyle 4 in rats and culminating in the generation of a distal phalanx in a human patient. 11 Previous studies demonstrating the engineering of tissues in complex geometries used a variety of techniques to achieve control of shape. Tissues with complex twodimensional surface geometries have been generated by casting cell-seeded hydrogel onto substrate base layers with desired surface shapes. 12 Three-dimensional (3-D) tissues with complex geometries have been fabricated through seeding of cells onto molded scaffolds 13 or injection molding of cell-seeded hydrogels. 14 While these techniques were able to create constructs with complex shapes, they lacked the ability to easily create parts with spatial heterogeneities. Other studies have investigated methods to reproduce zonal spatial variations in articular cartilage constructs. 1 Department of Mechanical and Aerospace Engineering, 2 Department of Computer and Information Science, 3 Department of Biomedical Engineering, Cornell University, Ithaca, New York. 1325
2 1326 These approaches created spatially heterogeneous constructs by depositing multiple layers of chondrocytes 15 or chondrocyte-seeded hydrogels. 16,17 While these techniques produced zonal organization in engineered tissues, they were limited to spatially varying the construct properties along a single axis only. Furthermore, while these techniques were feasible in the case of in vitro construct fabrication, it is not inherently feasible to adapt them for in vivo, or in situ, fabrication. For example, it would be challenging to place pre-formed hydrogel sheets through a small orifice while achieving proper alignment and bonding, such as would be necessary for incorporation into a minimally invasive implantation scheme. Solid freeform fabrication (SFF) technology has been used both to aid in the fabrication of tissues with complex geometries, as well to enable the production of constructs with spatial variations along multiple axes. SFF, often referred to as rapid prototyping, is analogous to 3-D printing. In SFF, layers of material are deposited or fused subsequently until a complete freeform geometry has been built. SFF schemes have been used to engineer tissues with complex geometries by using rapid prototyping techniques and computer-aided manufacturing (CAM) to fabricate traditional porous scaffolds that are subsequently seeded with cells. 18 This approach was capable of creating scaffolds of high geometric complexity; however, problems inherent with traditional scaffolding techniques persisted, such as seeding-depth limitations. A second SFF-assisted approach to creating geometrically complex implants overcame seeding-depth limitations by rapid prototyping molds and injection of pre-seeded hydrogel. 19 A third SFF-based technique created pre-seeded geometrically complex implants without the need for a mold by photo-crosslinking PEO and PEGM layer-bylayer. 20 However, none of these three SFF-based techniques enabled the fabrication of implants with complex zonal variations in three dimensions (i.e., 3-D spatial variation in cell-type, cell density, etc.). Furthermore, these techniques are not ideal for minimally invasive implantation schemes. The SFF of traditional porous scaffolds 18 and molds 19 relied on in vitro fabrication of the implants and likely require invasive surgery to implant a full-sized construct. The photo-crosslinking technique 20 is not completely suitable for in situ fabrication because it imposes major constraints upon the environment in which constructs may be fabricated. For example, in the case of in vivo fabrication, it may not be feasible to separate the tissue of the patient widely and steadily enough for a pool of polymer to be maintained in a stable fashion. Also, it may be difficult to fit a light source into the fabrication cavity at an orientation that is nearly perpendicular to the direction of the filling of the polymer well. Additionally, orientation of the layers is important with SFF, due to the orientation s impact on mechanical properties; however, COHEN ET AL. the photo-crosslinking technique is restricted to creating layer orientation in a direction perpendicular to gravity because the orientation of the surface of the liquid polymer pool is governed by gravitational force. The disadvantageous constraints imposed upon the printing environment by the polymer pool are also shared by techniques that deposit hydrogel into baths of crosslinkers. 21 More recently, studies have investigated approaches using SFF technology that were capable of producing cell constructs with both geometric complexity and multi-axial zonal organization. Collagen I and pluronic F-127 were recently used in an SFF process to produce viable cell-gel constructs with complex geometries layer-bylayer. 22 While the technology was also capable of fabricating components with complex zonal organization, 22 constructs created with PF-127 and collagen I are generally not three dimensionally stable in culture. Furthermore, since the gelation was not initiated prior to the deposition of the material, but rather by means of heat flow, strict thermal constraints were placed upon the fabrication environment, which may adversely affect certain in situ or in vivo fabrication approaches. Thus, no SFF processes exist to create pre-seeded, arbitrarily shaped, spatially heterogeneous constructs that are three dimensionally stable in culture and minimally impose constraints upon the printing environment. The approach presented herein used a gantry robot to deposit pre-seeded alginate hydrogel layer-by-layer. This technology was capable of producing implants with both complex geometry and zonal organization that were three dimensionally stable in culture. Since the gel was pre-seeded and crosslinking was initiated prior to deposition, this technique relaxed the constraints imposed upon the printing environment because, unlike other techniques, it neither required seeding of the construct after deposition nor required added energy to the material during deposition, such as specific light or temperature conditions. The only energy needed during deposition was that required to extrude the gel; this power source can easily be located externally to the printing environment without interfering with the in situ printing objective, unlike light and thermal energy sources. Alginate serves as a favorable cell delivery material since it is: 1) three dimensionally stable in culture, 2) non-toxic, 3) extrusion compatible, 4) pre-seeding compatible, and 5) crosslinking can be initiated prior to deposition. The objectives of this study were to: 1) develop a process to print pre-cell-seeded alginate gels in arbitrary shapes; 2) document the sterility and viability of the process; 3) determine the mechanical properties and extracellular matrix production characteristics of printed samples; and 4) characterize the geometric fidelity of the printing technique.
3 3-D PRINTING OF LIVING IMPLANTS MATERIALS AND METHODS Robotic platform The solid freeform fabrication robotic platform was an open-architecture CAM system that consisted of a gantry robot and a set of interchangeable deposition tools (Fig. 1). 23 The deposition tools were attached to the X-Y axes of the gantry robot, and the hydrogel was deposited onto the Z stage, which served as the build surface. The gantry robot had a workspace of cm and was capable of carrying a deposition tool of up to 5 kg. The X-Y axes of the gantry had a maximum traverse speed of 0.05 m/s and a maximum acceleration of 2.1 g. High traverse acceleration was important in order to maintain constant velocity around corners with small radii of curvature. The gantry robot could position the tip of the deposition tool with 25 m precision. The deposition tool was a linear actuator-driven syringe with interchangeable syringe tips that served as nozzles of various diameters, ranging from 0.33 to 0.84 mm. The maximum volumetric flow rate was 1.4 ml/s. A disposable syringe served as the material bay of the deposition tool. The material bay simply slid out of the deposition tool to facilitate multiple-material print jobs in which the material cartridge was frequently interchanged in mid-print. Once the system was loaded with printing material, a CAD model was sent to the robot s control system in stereolithography (STL) format, a computer file format A commonly used in rapid prototyping applications. Custom-written software sliced the model and planned tool paths within each layer. The tool-path planner prescribed a contour boundary path followed by raster fill-paths. The trajectory was sent from the computer to the robot and printing commenced (Fig. 2). Cell isolation and culture Cartilage was harvested from the femeropatellar groove of 1- to 3-day-old calves. The tissue was digested for 18 h at 37 C and 5% CO 2 in Dulbecco s modified Eagle medium (DMEM) containing 0.3% collagenase. The digest solution was filtered with a 100 m cell strainer. The articular chondrocytes were isolated from the strained digest solution by centrifugation at 412 g for 7 min. Cells were washed twice with phosphate-buffered saline (PBS). 24 The initial viability was determined using trypan blue (Mediatech, Herndon, VA). Samples were cultured in DMEM with 10% fetal bovine serum. A 1% antibiotic-antimyotic (10,000 U/mL penicillin G sodium, 10,000 g/ml streptomycin sulfate, 25 g/ml amophoterocin B in 0.85% saline) was also added to the growth medium, except during the sterility test. The cells were cultured at 37 C and 5% CO Gel deposition 1327 During the printing experiments, a tip diameter of 0.84 mm was selected. The robot planned the tool paths B FIG. 1. Robotic printing platform. (A) CAD rendering of gantry robot. (B) Close-up view of deposition tool. (Color images are available online at
4 1328 COHEN ET AL. Viability test The viability of the deposition process was measured by testing viability immediately before and after the printing process. The process viability was defined as the ratio of cell viability after harvesting but before gel seeding to the cell viability immediately after deposition. Twenty-four disks, 6 mm in diameter 2 mm in height, were printed into a 24-well plate. Each sample was pulled from the plate and tested with the Live/Dead Viability Assay (Molecular Probes, Eugene, OR). The samples were exposed to 0.15 M calcein AM and 2 M ethidium homodimer-1 (EthD-1) for 60 min at room temperature. The stained samples were analyzed under a Nikon TE2000-S microscope equipped with an epiflourescence attachment and a Spot RT digital camera. The viability was calculated as the average of the ratios of live over total cells in a given field. FIG. 2. Screenshot of trajectory planner. (a) CAD model loaded into trajectory planning software before tool-path generation. (b) Planned tool-paths. (Color images are available online at for material streams with a width of 1.2 mm and a height of 0.8 mm. The gel flow rate was 0.6 ml/min. At the start of each new path, the deposition started 0.2 s before the motion of the gantry robot. Chemical formulation and cell encapsulation The chondrocytes were suspended in PBS and spundown into a pellet with a centrifuge. Low-viscosity, high G-content non-medical grade LF10/60 alginate (FMC Biopolymer, Drammen, Norway) was dissolved at a concentration of 20 mg/ml, passed through a 0.22 m filter, and added to the cell pellet to achieve a density of cells/ml. The alginate-cell suspension was vortexed and mixed in a 2:1 ratio with autoclaved 10 mg/ml CaSO 4 in PBS. After mixing, the resulting hydrogel had a cell density of cells/ml. Due to the time-dependent gelation process, the window for optimal printing was 15 min from the time that the crosslinker is mixed with the alginate. The hydrogel was loaded into a syringe and inserted into the deposition tool. Sterility An empty autoclave bag was sealed and autoclaved. The sealed bag was placed under a sterile hood, a slit was cut into the bottom end of the bag, and a sterile 24-well plate was inserted. The bag was resealed with tape and ready for printing. The autoclave bag served as a sterile envelope around the plate, which protected the printed samples from contamination. To test the effectiveness of the envelope concept, 24 disks, 6 mm in diameter 2 mm in height, were printed into an envelope-protected sterile plate and cultured for 8 days without antibiotics or antimyotics. After the incubation period, media from each well were pulled and tested for bacterial presence with 100 M BacLight Green Stain (Molecular Probes), according to the manufacturer s instructions. Bacterial counts were performed using a hemacytometer (Hausser Scientific, Horsham, PA) and a Nikon TE2000-S microscope equipped with an epiflourescence attachment and a digital camera. DNA content and ECM accumulation Disks, 6 mm 2 mm, were printed into a 24-well plate. The disks were cultured for a period of time ranging from 0 to 142 days. Disks were removed from culture, weighed, lyophilized, and weighed again. All of the dry samples were digested in 1 ml of papain digest buffer (0.1 mol/l sodium phosphate, 10 mmol sodium EDTA [BDH], 10 mmol cysteine hydrochloride [Sigma-Aldrich, St. Louis, MO], and 3.8 U/mL papain [Sigma]) at 65 C for 24 h. The digest was analyzed for DNA content, as a marker of cell quantity. The assay was carried out in 96-well plates (Nalge Nunc, Rochester, NY). In each well, 190 L of 0.1 g/ml Hoechst dye in TES buffer was added to 10 L of the digested samples. Calf thymus DNA was used as a standard and the absorbance was read
5 3-D PRINTING OF LIVING IMPLANTS using a Tecan microplate reader with the excitation wavelength set at 348 nm and the emission wavelength set at 456 nm. 25 The digest was also analyzed for glycosaminoglycans (GAG) as a marker of proteoglycans. The assay was carried out in 96-well plates. In each well, 50 L of digest was mixed with 250 L of dye containing 16 mg/l 1,9- dimethylmethylene blue (DMMB) and 3.04 g/l glycine (ph 1.5). The absorbance was read at 595 nm using a microplate reader. Chondroitin-6-sulfate from shark cartilage (Sigma) was used to construct the standard curve. 26 Additionally, the digest was analyzed for hydroxyproline as a marker of collagen. One hundred L of each sample s digest was hydrolyzed in 100 L of 2N NaOH at 110 C for 18 h. The following were combined into screw-cap microfuge tubes: 20 L 5N HCl, 100 L digested hydrolyzed sample, 100 L 0.01 M CuSO 4, 100 L 2.5 N NaOH, and 100 L 6% H 2 O 2. After the addition of H 2 O 2, the tubes were allowed to sit at room temperature for about 5 min while being shaken occasionally to remove gas bubbles. The tubes were then vortexed and placed in a heat block at 80 C for 5 min. The tubes were placed in an ice bath until cooled to room temperature. Four hundred L of 3N H 2 SO 4 and 200 L of DMAB were added to each tube. Each well of a 96-well plate was filled with 200 L of a treated sample and absorbance was read at 540 nm. 27 Mechanical characterization Disks less than 1-hour-old, 6 mm 2 mm, were printed and placed in an ELF 3200 (EnduraTec, Minnetonka, MN) mechanical test-frame in an unconfined compression chamber between two parallel plates. A 1000 g load cell (Sensotec, Columbus, OH) was attached to the bottom plate and a displacement sensor to the top plate. The bottom plate was filled with PBS in order to completely encompass the sample with fluid. The two plates started at a distance corresponding to 0% strain and stepped 0.1 mm towards each other every 100 s until 45% strain was achieved. Stress and strain data were acquired at a frequency of 5 Hz. 28 The stress-strain curve of each printed sample was analyzed by first finding the equilibrium stress corresponding to each imposed strain (0 45%). The linear region of the equilibrium stress-strain curve was fit linearly; the slope of this line was the Young s modulus of the sample printer and an electrical power source. Since the hydrogels being measured were electrically conductive, when the needle touched the alginate hydrogel, current flowed from the power source through the base plate, through the hydrogel sample, and back to the tip thus completing the electrical circuit. Positional data was logged when the robot sensed that the electrical circuit was completed. By stepping over the entire part in 0.35 mm X-Y coordinate increments, a map of the surface points of the hydrogel sample was constructed. Using the surface map, the average height of the X-Y plane boundary points was calculated, these points representing the height of the base plate. The Z coordinates of all points were shifted down by the mean boundary height in order to align the base plate with the X-Y plane. The list of points was truncated to exclude all points that represented the base plate itself (i.e., all points with zerovalued Z coordinates were removed). The mean X and Y coordinates were calculated and then the X and Y coordinates of the points were shifted by the mean values in order to center the samples in the X-Y plane. The overall average height of the sample was calculated by finding the mean z-value of the points. The average height, excluding the edge effect, was calculated by finding the mean z-value of the points that were in the inner X-Y region of the geometry; that is, all points whose X and Y coordinates fell within 90 90% of the total X-Y crosssection. The RMS surface roughness was determined to be the standard deviation of these inner points. In order to calculate the X-Y geometric fidelity, the point list was further truncated to exclude all points with z-values below half the total average height; doing so removes the effect of the sloping interface between the walls of the geometry and the base plate. The X-Y boundary points were identified using a standard convex hull Geometric fidelity The geometric fidelity of the printing process was determined by using the printing platform as a coordinate measuring machine (CMM). A metal needle was attached to the deposition tool with a wire soldered to the needle. Another wire was attached to the metal base plate of the FIG. 3. Printed crescent suspended with tool.
6 COHEN ET AL FIG. 4. CAD models and photographs of printed samples. (Color images are available online at algorithm, convhull, in Matlab (MathWorks, Natick, MA). These X-Y boundary points were subtracted by the intended X-Y coordinate corresponding to each point in order to generate an X-Y error map. The mean and standard deviation of this error were calculated to reflect the X-Y geometric fidelity. RESULTS Spatially heterogeneous gel constructs A set of gel constructs was created to showcase the ability of the system to fabricate complex, spatially het- erogeneous hydrogel geometries. A crescent-shaped part, 2.5 cm long 0.75 cm wide, was printed and elevated with a thin utensil to verify that the part maintained its shape while fully supporting its weight (Fig. 3). The crescent-shaped part was drawn in the SolidWorks CAD program (Fig. 4a1), input into the printer s control software, and printed (Fig. 4a2). To verify the system s ability to construct a multiplematerial, spatially heterogeneous construct, an IV discshaped CAD model was generated (Fig. 4b1), processed with the robot s control software, and printed (Fig. 4b2). The two-material component was fabricated from two different batches of alginate hydrogel, each stained with FIG. 5. Live and dead panels of the same view field from the viability test. (Color images are available online at online.com/ten).
7 3-D PRINTING OF LIVING IMPLANTS 1331 a dye of a different color. Utilizing the multiple-material capabilities of the robotic platform, the robot stopped at numerous points throughout the print and requested a change of materials, as prescribed by the robot s tool path planner. At each of these points, the material bay and syringe tip unit of the deposition tool were manually swapped with the designated material bay/syringe tip unit and printing commenced. In order to demonstrate the system s ability to create implants with complex geometries, a CAD model of an ovine meniscus was constructed from a CT scan (Fig. 4c1) and printed (Fig. 4c2). Viability and sterility Viability tests successfully detected both live (Fig. 5a) and dead (Fig. 5b) cells in printed gels. The viability of the printing process was determined to be 94 5% (n 15). The viability appeared to be spatially uniform. By inspection with phase contrast microscopy and fluorescence microscopy, the cell distribution was observed to be homogeneous. When inspected by phase contract microscopy, no abnormal morphology was observed. After 8 days of incubating a printed gel samples without any antibiotics or antimyotics, less than 1 bacterium per 0.9 L was detected (n 12). ECM accumulation GAG content increased in printed samples over time in incubation to g/ g DNA (n 4 17) by 3 weeks (Fig. 6). Printed disks were cultured beyond 3 weeks to 18 weeks, but GAG level did not change significantly. Hydroxyproline content increased in the printed samples over time in incubation as well. Hydroxyproline content reached g/ g DNA (n 4 17) by 14 weeks (Fig. 7). At 18 weeks, the disks did not appear to FIG. 7. Hydroxyproline content DNA normalized with time in culture. change shape grossly when compared to disks exposed to media immediately after printing (Fig. 8). Mechanical properties It was observed that the Young s modulus of samples increased with time after fabrication. The compressive elastic modulus of a typical printed disk that was less than 1 h old was determined to be kpa (n 6). Geometric fidelity When the point maps of the entire printed samples (Fig. 9) were considered, the average height of a single layer was determined to be mm and the RMS surface roughness was mm. When only the points with X-Y coordinates located in the inner region of the printed samples, the edge effect was ignored and the resulting average height was mm and the RMS surface roughness was mm. When the X-Y error was analyzed at the mid-height plane (Fig. 10), the average X-Y error was found to be mm. The standard deviation of the X-Y error was found to be mm. FIG. 6. GAG content DNA normalized with time in culture. DISCUSSION The objective of this research was to develop a technique for directly fabricating pre-seeded constructs in arbitrary geometries with multi-axial zonal organization. In order for the technique to be considered a feasible tissueengineering approach, we focused on a set of benchmarks including process viability, process sterility, ECM production by printed samples, and mechanical properties of printed samples.
8 1332 COHEN ET AL. FIG. 8. Printed disks after 1 day (A) and 142 (B) days in culture. (Color images are available online at Samples printed into simple geometries retained their shape for the duration of a 20 week culture. This result points to an advantage of using alginate as a material delivery system for 3-D printing. Other materials proposed for such applications are highly amenable to printing, 22 but do not retain their shape in culture, due to contraction, in the case of collagen, or swelling and dissolution, in the case of pluronic-f127. The results of the viability test suggest that the printing process, including the cell encapsulation and gel deposition stages, does not have a major harmful impact on the cell viability of printed cells. A 94 5% process viability is adequate for a new tissue-engineering technique. This viability value is markedly higher than other SFF approaches to pre-encapsulated construct fabrication, which are in the range of 60 70%. 20 The bacteria count of less than 1 bacterium per 0.9 L of media suggests that the samples were bacteria free and that the sterile envelope technique worked sufficiently well. The autoclave bag served as an inexpensive and disposable sterile envelope that can be fit to a printing workspace of any size. The use of an environmental envelope reduced the demands placed on the sterility of the production site. Furthermore, other than the syringe and syringe tip, the printer itself did not need to be sterilized. This technique would enable future applications in which the maintenance of environmental sterility would otherwise be challenging. The peak GAG content of 18.9 g/ g DNA and the peak hydroxyproline content of 0.92 g/ g DNA were similar to those observed in a number of studies in which bovine articular chondrocytes were seeded onto a variety of substrates (Table 1). This suggests that the printing process did not substaintially interfere with the ability of printed cells to assemble an extracellular matrix. The elastic modulus of the alginate hydrogel printed samples was higher than the modulus of samples created by some other SFF techniques that created pre-seeded Z Axis (Microns) Y Axis (Microns) X Axis (Microns) FIG. 9. Centered and truncated CMM data for an 8 8 mm printed geometry.
9 3-D PRINTING OF LIVING IMPLANTS FIG Cross-section of CMM map truncated at mid-height. (Color images are available online at implants. The modulus of alginate-printed samples was higher than those created with the photo-crosslinking SFF technique.20 Also, alginate hydrogels are much stiffer than those made from pluronic F-127, which has been investigated as a build material for SFF tissue-engineering approaches.22 Furthermore, the mechanical properties of printed alginate implants could be easily improved through the addition of calcium in or after the printing process. The tissue-engineering approach detailed herein combines the strengths of molding techniques with those of solid freeform fabrication technology. This technology successfully encapsulates bovine chondrocytes in alginate hydrogel and deposits them in arbitrary geometries while maintaining sterility and viability. Unlike injection molding of pre-seeded alginate TABLE 1. COMPARISON Alginate beads (weeks) GAG ( g/ g DNA) Hydroxyproline ( g/ g DNA) 2826 (3) 2826 (3) (6) OF ECM PROPERTIES Alginate injection molding (weeks) Alginate SFF (weeks) 1828 (10) (10) (3) (14) hydrogel, this technique does not require molds, nor does it require any custom tooling to fabricate an implant. Also, due to the inherent benefits of SFF, implants with complex geometries can be fabricated that could not otherwise be molded or easily fabricated using other methods such as layering sheets. This tissue-engineering approach combines the strength and biocompatibility of hydrogel injection molding with the geometric freedom and inherent patient-specificity capabilities of SFF. The method could allow for efficient fabrication of patient-specific implants without the cost and delay normally associated with creating custom tooling. Furthermore, this technique reduces the constraints imposed upon the fabrication environment, which is an important concern for in situ and minimally invasive in vivo implant fabrication. This technology could enable fabrication of geometrically complex, multiple-material implants that would otherwise not be producible. Also, this approach could enable the fabrication of large implants thatwould otherwise be difficult to seed with cells due to transport limitations. ACKNOWLEDGMENTS This work was funded by Cornell University. The authors would also like to thank Suzanne Maher at the Hospital for Special Surgery (New York, NY) for technical assistance with CT scans of ovine meniscus.
10 1334 REFERENCES 1. Cao, Y., Vacanti, J.P., Paige, K.T., Upton, J., and Vacanti, C.A. Transplantation of chondrocytes utilizing a polymercell construct to produce tissue-engineered cartilage in the shape of a human ear. Plast. Reconstr. Surg. 100, 297, Kim, W.S., Vacanti, J.P., Cima, L., Mooney, D., Upton, J., Puelacher, W.C., and Vacanti, C.A. Cartilage engineered in predetermined shapes employing cell transplantation on synthetic biodegradable polymers. Plast. Reconstr. Surg. 94, 233, Puelacher, W.C., Wisser, J., Vacanti, C.A., Ferraro, N.F., Jaramillo, D., and Vacanti, J.P. Temporomandibular joint disc replacement made by tissue-engineered growth of cartilage. J. Oral Maxillofac. Surg. 52, 1172, Weng, Y., Cao, Y., Silva, C.A., Vacanti, M.P., and Vacanti, C.A. Tissue-engineered composites of bone and cartilage for mandible condylar reconstruction. J. Oral Maxillofac. Surg. 59, 185, Ibarra, C., Jannetta, C., Vacanti, C.A., Cao, Y., Kim, T.H., Upton, J., and Vacanti, J.P. Tissue engineered meniscus: a potential new alternative to allogeneic meniscus transplantation. Transplant. Proc. 29, 986, Kojima, K., Bonassar, L.J., Ignotz, R.A., Syed, K., Cortiella, J., and Vacanti, C.A. Comparison of tracheal and nasal chondrocytes for tissue engineering of trachea. Ann. Thorac. 76, 1884, Mizuno, H., Roy, A.K., Vacanti, C.A., Kojima, K., Ueda, M., and Bonassar, L.J. Tissue-engineered composites of annulus fibrosus and nucleus pulposus for intervertebral disc replacement. Spine 29, 1290, Kamil, S.H., Kojima, K., Vacanti, M.P., Bonassar, L.J., Vacanti, C.A., and Eavey, R.D. In vitro tissue engineering to generate a human-sized auricle and nasal tip. Laryngoscope 113, 90, Puelacher, W.C., Mooney, D., Langer, R., Upton, J., Vacanti, J.P., and Vacanti, C.A. Design of nasoseptal cartilage replacements synthesized from biodegradable polymers and chondrocytes. Biomaterials 15, 774, Puelacher, W.C., Vacanti, J.P., Ferraro, N.F., Schloo, B., and Vacanti, C.A. Femoral shaft reconstruction using tissue-engineered growth of bone. Int. J. Oral Maxillofac. Surg. 25, 223, Vacanti, C.A., Bonassar, L.J., Vacanti, M.P., and Shufflebarger, J. Replacement of an avulsed phalanx with tissueengineered bone. N. Engl. J. Med. 244, 1511, Hung, C.T., Lima, E.G., Mauch, R.L., Taki, E., LeRoux, M.A., Lu, H.H., Stark, R.G., Guo, X.E., and Ateshian, G.A. Anatomically shaped osteochondral constructs for articular cartilage repair. J. Biomech. 36, 1853, Cao, Y., Rodriguez, A., Vacanti, M., Ibarra, C., Arevalo, C., and Vacanti, C.A. Comparative study of the use of poly(glycolic acid), calcium alginate and pluronics in the engineering of autologous porcine cartilage. J. Biomater.Sci. 9, 475, Chang, S.C., Rowley, J.A., Tobias, G., Genes, N.G., Roy, A.K., Mooney, D.J., Vacanti, C.A., and Bonassar, L.J. Injection molding of chondrocyte/alginate constructs in the shape of facial implants. J. Biomed. Mat. Res. 55, 503, COHEN ET AL. 15. Klein, T.J., Schumacher, B.L., Schmidt, T.A., Li, K.W., Voegtline, M.S., Masuda, K., Thonar, E.J., and Sah, R.L. Tissue engineering of stratified articular cartilage from chondrocyte subpopulations. Osteoarth. Cart. 11, 595, Kim, T.K., Sharma, B., Williams, C.G., Ruffner, M.A., Malik, A., McFarland, E.G., and Elisseeff, J.H. Experimental model for cartilage tissue engineering to regenerate the zonal organization of articular cartilage. Osteoarth. Cart. 11, 653, Ng, K.W., Wang, C.C., Mauck, R.L., Kelly, T.A., Chahine, N.O., Costa, K.D., Ateshian, G.A., and Hung, C.T. A layered agarose approach to fabricate depth-dependent inhomogeneity in chondrocyte-seeded constructs. J. Orthop. Res. 23, 134, Sun, W., Starly, B., Darling, A., and Gomez, C. Computeraided tissue engineering: application to biomimetic modeling and design of tissue scaffolds. Biotechnol. Appl. Biochem. 39, 49, Ballyns, J., Gleghorn, J., Niebrzydowski, V., Rawlinson, J., Maher, S., and Bonassar, L.J. Tissue engineering of meniscus using microct imaging, CAD and injection molding. Abstract presented at the 51 st Annual Meeting of the Orthopaedic Research Society, Washington D.C., Abstract no Busaina, D., Hunt, E., and Boland, T. Rapid prototyping of tissue-engineering constructs, using photopolymerizable hydrogels and stereolithography. Tissue Eng. 10, 1316, Khalil, S., Nam, J., and Sun, W. Multi-nozzle deposition for construction of 3D biopolymer tissue scaffolds. Rapid Prototyp. J. 11, 9, Smith, C.M., Stone, A.L., Parkhill, R.L., Stewart, R.L., Simpkins, M.W., Kachurin, A.M., Warren, W.L., and Williams, S.K. Three-dimensional BioAssembly tool for generating viable tissue-engineered constructs. Tissue Eng. 10, 1566, Malone E., Rasa K., Cohen D. L., Isaacson T., Lashley H., and Lipson H. Freeform fabrication of 3D zinc-air batteries and functional electro-mechanical assemblies. Rapid Prototyp. J. 10, 58, Genes, N.G., Rowley, J.A., Mooney, D.J., and Bonassar, L.J. Effect of substrate mechanics on chondrocyte adhesion to modified alginate surfaces. Arch. Biochem. Biophys. 422, 161, Kim, Y.J., Sah, R.L., Doong, J.Y., and Grodzinsky A.J. Fluorometric assay of DNA in cartilage explants using Hoechst Anal. Biochem. 174, 168, Enobakhare, B.O., Bader D.L., and Lee D.A. Quantification of sulfated glycosaminoglycans in chondrocyte/alginate cultures, by use of 1,9-dimethylmethylene blue. Anal. Biochem. 243, 189, Neuman, R.E., and Logan, M.A. The determination of hydroxyproline. J. Biol. Chem. 184, 299, Kim, Y.J., Bonassar, L.J., and Grodzinsky, A.J. The role of cartilage streaming potential, fluid flow and pressure in the stimulation of chondrocyte biosynthesis during dynamic compression. J. Biomech. 28, 1055, Beekman, B., Verzijl, N., Bank, R.A., von der Mark, K., and TeKoppelle, J.M. Synthesis of collagen by bovine
11 3-D PRINTING OF LIVING IMPLANTS chondrocytes cultured in alginate; posttranslational modifications and cell-matrix interaction. Exp. Cell Res. 237, 135, Hott, M.E., Megerian, C.A., Beane, R., and Bonassar, L.J. Fabrication of tissue engineered tympanic membrane patches using computer-aided design and injection molding. Laryngoscope 114, 1290, Ragan, P.M., Chin, V.I., Hung, H.H., Masuda, K., Thonar, E.J., Arner, E.C., Grodzinsky, A.J., and Sandy, J.D. Chondrocyte extracellular matrix synthesis and turnover are influenced by static compression in a new algi nate disk culture system. Arch. Biochem. Biophys. 383, 256, Address reprint requests to: Lawrence J. Bonassar, Ph.D. Department of Biomedical Engineering Cornell University 218 Upson Hall Ithaca, NY lb244@cornell.edu
12
protocol handbook 3D cell culture mimsys G hydrogel
 handbook 3D cell culture mimsys G hydrogel supporting real discovery handbook Index 01 Cell encapsulation in hydrogels 02 Cell viability by MTS assay 03 Live/Dead assay to assess cell viability 04 Fluorescent
handbook 3D cell culture mimsys G hydrogel supporting real discovery handbook Index 01 Cell encapsulation in hydrogels 02 Cell viability by MTS assay 03 Live/Dead assay to assess cell viability 04 Fluorescent
Z-Stacking and Z-Projection using a Scaffold-based 3D Cell Culture Model
 A p p l i c a t i o n N o t e Z-Stacking and Z-Projection using a Scaffold-based 3D Cell Culture Model Brad Larson and Peter Banks, BioTek Instruments, Inc., Winooski, VT Grant Cameron, TAP Biosystems
A p p l i c a t i o n N o t e Z-Stacking and Z-Projection using a Scaffold-based 3D Cell Culture Model Brad Larson and Peter Banks, BioTek Instruments, Inc., Winooski, VT Grant Cameron, TAP Biosystems
ArC Amine Reactive Compensation Bead Kit
 ArC Amine Reactive Compensation Bead Kit Catalog no. A1346 Table 1. Contents and storage information. Material Amount Composition Storage Stability ArC reactive beads (Component A) ArC negative beads (Component
ArC Amine Reactive Compensation Bead Kit Catalog no. A1346 Table 1. Contents and storage information. Material Amount Composition Storage Stability ArC reactive beads (Component A) ArC negative beads (Component
BD PuraMatrix Peptide Hydrogel
 BD PuraMatrix Peptide Hydrogel Catalog No. 354250 Guidelines for Use FOR RESEARCH USE ONLY NOT FOR CLINICAL, DIAGNOSTIC OR THERAPEUTIC USE SPC-354250-G rev 2.0 1 TABLE OF CONTENTS Intended Use... 2 Materials
BD PuraMatrix Peptide Hydrogel Catalog No. 354250 Guidelines for Use FOR RESEARCH USE ONLY NOT FOR CLINICAL, DIAGNOSTIC OR THERAPEUTIC USE SPC-354250-G rev 2.0 1 TABLE OF CONTENTS Intended Use... 2 Materials
Transformation Protocol
 To make Glycerol Stocks of Plasmids ** To be done in the hood and use RNase/DNase free tips** 1. In a 10 ml sterile tube add 3 ml autoclaved LB broth and 1.5 ul antibiotic (@ 100 ug/ul) or 3 ul antibiotic
To make Glycerol Stocks of Plasmids ** To be done in the hood and use RNase/DNase free tips** 1. In a 10 ml sterile tube add 3 ml autoclaved LB broth and 1.5 ul antibiotic (@ 100 ug/ul) or 3 ul antibiotic
Radius 24-Well Cell Migration Assay (Laminin Coated)
 Product Manual Radius 24-Well Cell Migration Assay (Laminin Coated) Catalog Number CBA-125-LN 24 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Cell migration is a highly
Product Manual Radius 24-Well Cell Migration Assay (Laminin Coated) Catalog Number CBA-125-LN 24 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Cell migration is a highly
Evaporation-based Microfluidic Production of. Oil-free Cell-Containing Hydrogel Particles
 Supporting Information Evaporation-based Microfluidic Production of Oil-free Cell-Containing Hydrogel Particles Rong Fan, a Kubra Naqvi, b Krishna Patel, c Jun Sun d and Jiandi Wan *a a Microsystems Engineering
Supporting Information Evaporation-based Microfluidic Production of Oil-free Cell-Containing Hydrogel Particles Rong Fan, a Kubra Naqvi, b Krishna Patel, c Jun Sun d and Jiandi Wan *a a Microsystems Engineering
Measuring Cell Viability/Cytotoxicity: Cell Counting Kit-F
 Introduction The Cell Counting Kit-F is a fluorometic assay for the determination of viable cell numbers. Calcein-AM in this kit passes through the cell membrane and is hydrolized by the esterase in the
Introduction The Cell Counting Kit-F is a fluorometic assay for the determination of viable cell numbers. Calcein-AM in this kit passes through the cell membrane and is hydrolized by the esterase in the
3D Cell Culture mimsys G
 3D Cell Culture mimsys G xeno-free & nutrient permeable hydrogel for 3D cell culture mimsys G is a xeno-free and non-immunogenic, easy to handle hydrogel for cell encapsulation in 3D experiments in vitro
3D Cell Culture mimsys G xeno-free & nutrient permeable hydrogel for 3D cell culture mimsys G is a xeno-free and non-immunogenic, easy to handle hydrogel for cell encapsulation in 3D experiments in vitro
How To Build A 3D Model From Scratch
 SERVICES AND CAPABILITIES 1. Rapid prototyping What is rapid prototyping? Rapid prototyping (RP) or more recently Free Form Fabrication refers to the fabrication of a physical, three-dimensional part of
SERVICES AND CAPABILITIES 1. Rapid prototyping What is rapid prototyping? Rapid prototyping (RP) or more recently Free Form Fabrication refers to the fabrication of a physical, three-dimensional part of
RAPID PROTOTYPING. Learning Objectives: By the end of the lecture the student should be able to: Explain the fundamentals of Rapid Prototyping
 RAPID PROTOTYPING Learning Objectives: By the end of the lecture the student should be able to: Explain the fundamentals of Rapid Prototyping Outline and explain differences of Rapid Prototyping Technologies
RAPID PROTOTYPING Learning Objectives: By the end of the lecture the student should be able to: Explain the fundamentals of Rapid Prototyping Outline and explain differences of Rapid Prototyping Technologies
PRELIMINARY COMPONENT INTEGRATION USING RAPID PROTOTYPING TECHNIQUES
 J! PRELIMINARY COMPONENT INTEGRATION USING RAPID PROTOTYPING TECHNIQUES by Ken Cooper National Aeronautics and Space Administration Building 4707, Marshall Space Flight Center George C. Marshall Space
J! PRELIMINARY COMPONENT INTEGRATION USING RAPID PROTOTYPING TECHNIQUES by Ken Cooper National Aeronautics and Space Administration Building 4707, Marshall Space Flight Center George C. Marshall Space
Additive Manufacturing: Processes and Standard Terminology
 Additive Manufacturing: Processes and Standard Terminology Gary Coykendall Copyright Edmonds Community College 2012; Permission granted for use and reproduction for educational purposes only. Abstract
Additive Manufacturing: Processes and Standard Terminology Gary Coykendall Copyright Edmonds Community College 2012; Permission granted for use and reproduction for educational purposes only. Abstract
Making Better Medical Devices with Multisensor Metrology
 Making Better Medical Devices with Multisensor Metrology by Nate J. Rose, Chief Applications Engineer, Optical Gaging Products (OGP) Multisensor metrology is becoming a preferred quality control technology
Making Better Medical Devices with Multisensor Metrology by Nate J. Rose, Chief Applications Engineer, Optical Gaging Products (OGP) Multisensor metrology is becoming a preferred quality control technology
PROTOCOL. Immunocytochemistry (ICC) MATERIALS AND EQUIPMENT REQUIRED
 PROTOCOL Immunocytochemistry (ICC) 1850 Millrace Drive, Suite 3A Eugene, Oregon 97403 11-07 MATERIALS AND EQUIPMENT REQUIRED Materials: MitoSciences primary monoclonal antibody/antibodies Fluorophore-conjugated
PROTOCOL Immunocytochemistry (ICC) 1850 Millrace Drive, Suite 3A Eugene, Oregon 97403 11-07 MATERIALS AND EQUIPMENT REQUIRED Materials: MitoSciences primary monoclonal antibody/antibodies Fluorophore-conjugated
As published in PIM International
 As published in PIM International www.pim-international.com 64 Powder Injection Moulding International September 2012 Rapid prototyping of highperformance ceramics opens new opportunities for the CIM industry
As published in PIM International www.pim-international.com 64 Powder Injection Moulding International September 2012 Rapid prototyping of highperformance ceramics opens new opportunities for the CIM industry
BioResearch. RAFT 3D Cell Culture Kit Protocol
 BioResearch RAFT 3D Cell Culture Kit Protocol RAFT 3D Cell Culture Kit This protocol describes in detail how to produce RAFT 3D Cell Cultures in 96-well plates, 24-well plates and 24-well inserts. It is
BioResearch RAFT 3D Cell Culture Kit Protocol RAFT 3D Cell Culture Kit This protocol describes in detail how to produce RAFT 3D Cell Cultures in 96-well plates, 24-well plates and 24-well inserts. It is
SmartFlare RNA Detection Probes: Principles, protocols and troubleshooting
 Technical Guide SmartFlare RNA Detection Probes: Principles, protocols and troubleshooting Principles of SmartFlare technology RNA detection traditionally requires transfection, laborious sample prep,
Technical Guide SmartFlare RNA Detection Probes: Principles, protocols and troubleshooting Principles of SmartFlare technology RNA detection traditionally requires transfection, laborious sample prep,
Slicing Issues in CAD Translation to STL in Rapid Prototyping
 Paper 021, ENG 103 Slicing Issues in CAD Translation to STL in Rapid Prototyping Divesh R. Sahatoo In-Corr-Tech Ltd., San Fernando, Trinidad drsahatoo@yahoo.com Boppana V. Chowdary University of The West
Paper 021, ENG 103 Slicing Issues in CAD Translation to STL in Rapid Prototyping Divesh R. Sahatoo In-Corr-Tech Ltd., San Fernando, Trinidad drsahatoo@yahoo.com Boppana V. Chowdary University of The West
* This work is an official contribution of the National Institute of Standards and Technology and
 Variability in the Geometric Accuracy of Additively Manufactured Test Parts A.L. Cooke and J.A. Soons National Institute of Standards and Technology * Gaithersburg, MD, USA Abstract This paper describes
Variability in the Geometric Accuracy of Additively Manufactured Test Parts A.L. Cooke and J.A. Soons National Institute of Standards and Technology * Gaithersburg, MD, USA Abstract This paper describes
Southern Blot Analysis (from Baker lab, university of Florida)
 Southern Blot Analysis (from Baker lab, university of Florida) DNA Prep Prepare DNA via your favorite method. You may find a protocol under Mini Yeast Genomic Prep. Restriction Digest 1.Digest DNA with
Southern Blot Analysis (from Baker lab, university of Florida) DNA Prep Prepare DNA via your favorite method. You may find a protocol under Mini Yeast Genomic Prep. Restriction Digest 1.Digest DNA with
CytoSelect Cell Viability and Cytotoxicity Assay Kit
 Product Manual CytoSelect Cell Viability and Cytotoxicity Assay Kit Catalog Number CBA-240 96 assays (96-well plate) FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction The measurement
Product Manual CytoSelect Cell Viability and Cytotoxicity Assay Kit Catalog Number CBA-240 96 assays (96-well plate) FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction The measurement
3D Printing Design Guidelines Sierra College CACT
 3D Printing Design Guidelines Sierra College CACT Prepared by ARW Consulting June 30, 2009 Table of Contents Scope of this Document.. 1 General Overview... 2 File Format.. 3 SST Support Material.. 4 Venting
3D Printing Design Guidelines Sierra College CACT Prepared by ARW Consulting June 30, 2009 Table of Contents Scope of this Document.. 1 General Overview... 2 File Format.. 3 SST Support Material.. 4 Venting
Western Blot Analysis with Cell Samples Grown in Channel-µ-Slides
 Western Blot Analysis with Cell Samples Grown in Channel-µ-Slides Polyacrylamide gel electrophoresis (PAGE) and subsequent analyses are common tools in biochemistry and molecular biology. This Application
Western Blot Analysis with Cell Samples Grown in Channel-µ-Slides Polyacrylamide gel electrophoresis (PAGE) and subsequent analyses are common tools in biochemistry and molecular biology. This Application
Chromatin Immunoprecipitation
 Chromatin Immunoprecipitation A) Prepare a yeast culture (see the Galactose Induction Protocol for details). 1) Start a small culture (e.g. 2 ml) in YEPD or selective media from a single colony. 2) Spin
Chromatin Immunoprecipitation A) Prepare a yeast culture (see the Galactose Induction Protocol for details). 1) Start a small culture (e.g. 2 ml) in YEPD or selective media from a single colony. 2) Spin
HyStem. Hydrogels CELLULAR MATRICES FOR TRANSLATIONAL RESEARCH. esibio.com
 HyStem Hydrogels CELLULAR MATRICES FOR TRANSLATIONAL RESEARCH HyStem Hydrogel Extracellular Matrices Chemically-defined For use in 2D and 3D formats in vitro and in vivo applications Customizable for many
HyStem Hydrogels CELLULAR MATRICES FOR TRANSLATIONAL RESEARCH HyStem Hydrogel Extracellular Matrices Chemically-defined For use in 2D and 3D formats in vitro and in vivo applications Customizable for many
Computer Aided Engineering (CAE) Techniques Applied To Hip Implant
 International Journal Of Computational Engineering Research (ijceronline.com) Vol. 3 Issue. 3 Computer Aided Engineering (CAE) Techniques Applied To Hip Implant 1, M. S. Abo_Elkhair, 2, M. E. Abo-Elnor,
International Journal Of Computational Engineering Research (ijceronline.com) Vol. 3 Issue. 3 Computer Aided Engineering (CAE) Techniques Applied To Hip Implant 1, M. S. Abo_Elkhair, 2, M. E. Abo-Elnor,
GLOBAL MANUFACTURING. ARAUJO, Anna Carla AUG, 2015 Mechanical Engineering Department POLI/COPPE/UFRJ
 GLOBAL MANUFACTURING ARAUJO, Anna Carla AUG, 2015 Mechanical Engineering Department POLI/COPPE/UFRJ Workpiece Presentation Powder Metallurgy and Additive Manufacturing [#7] Powder Metallurgy PM parts can
GLOBAL MANUFACTURING ARAUJO, Anna Carla AUG, 2015 Mechanical Engineering Department POLI/COPPE/UFRJ Workpiece Presentation Powder Metallurgy and Additive Manufacturing [#7] Powder Metallurgy PM parts can
Chapter 5 POWDER-BASED RAPID PROTOTYPING SYSTEMS
 Chapter 5 POWDER-BASED RAPID PROTOTYPING SYSTEMS 5.1 3D SYSTEMS SELECTIVE LASER SINTERING (SLS) 5.1.1 Company 3D Systems Corporation was founded by Charles W. Hull and Raymond S. Freed in 1986. The founding
Chapter 5 POWDER-BASED RAPID PROTOTYPING SYSTEMS 5.1 3D SYSTEMS SELECTIVE LASER SINTERING (SLS) 5.1.1 Company 3D Systems Corporation was founded by Charles W. Hull and Raymond S. Freed in 1986. The founding
Protease Peptide Microarrays Ready-to-use microarrays for protease profiling
 Protocol Protease Peptide Microarrays Ready-to-use microarrays for protease profiling Contact us: InfoLine: +49-30-97893-117 Order per fax: +49-30-97893-299 Or e-mail: peptide@jpt.com www: www.jpt.com
Protocol Protease Peptide Microarrays Ready-to-use microarrays for protease profiling Contact us: InfoLine: +49-30-97893-117 Order per fax: +49-30-97893-299 Or e-mail: peptide@jpt.com www: www.jpt.com
Human Free Testosterone(F-TESTO) ELISA Kit
 Human Free Testosterone(F-TESTO) ELISA Kit Catalog Number. MBS700040 For the quantitative determination of human free testosterone(f-testo) concentrations in serum, plasma. This package insert must be
Human Free Testosterone(F-TESTO) ELISA Kit Catalog Number. MBS700040 For the quantitative determination of human free testosterone(f-testo) concentrations in serum, plasma. This package insert must be
Lead Testing and On Site Calibration for Water Testing Detection Range: 2 100ppb
 Document: AND Lead 100 7 2013 Lead Testing and On Site Calibration for Water Testing Detection Range: 2 100ppb July, 2013 Edition 1 ANDalyze, Inc., 2012. All rights reserved. Printed in USA. Table of Contents
Document: AND Lead 100 7 2013 Lead Testing and On Site Calibration for Water Testing Detection Range: 2 100ppb July, 2013 Edition 1 ANDalyze, Inc., 2012. All rights reserved. Printed in USA. Table of Contents
Human Adult Mesothelial Cell Manual
 Human Adult Mesothelial Cell Manual INSTRUCTION MANUAL ZBM0025.01 SHIPPING CONDITIONS Human Adult Mesothelial Cells Orders are delivered via Federal Express courier. All US and Canada orders are shipped
Human Adult Mesothelial Cell Manual INSTRUCTION MANUAL ZBM0025.01 SHIPPING CONDITIONS Human Adult Mesothelial Cells Orders are delivered via Federal Express courier. All US and Canada orders are shipped
5-Axis Test-Piece Influence of Machining Position
 5-Axis Test-Piece Influence of Machining Position Michael Gebhardt, Wolfgang Knapp, Konrad Wegener Institute of Machine Tools and Manufacturing (IWF), Swiss Federal Institute of Technology (ETH), Zurich,
5-Axis Test-Piece Influence of Machining Position Michael Gebhardt, Wolfgang Knapp, Konrad Wegener Institute of Machine Tools and Manufacturing (IWF), Swiss Federal Institute of Technology (ETH), Zurich,
INSTRUCTIONS 56-1190-98. Edition AC
 Sephacryl S-100 High Resolution Sephacryl S-200 High Resolution Sephacryl S-300 High Resolution Sephacryl S-400 High Resolution Sephacryl S-500 High Resolution INSTRUCTIONS Sephacryl High Resolution chromatography
Sephacryl S-100 High Resolution Sephacryl S-200 High Resolution Sephacryl S-300 High Resolution Sephacryl S-400 High Resolution Sephacryl S-500 High Resolution INSTRUCTIONS Sephacryl High Resolution chromatography
Chondrogenical Differentiation of Umbilical Cord Lining. Stem Cells
 Chondrogenical Differentiation of Umbilical Cord Lining Stem Cells He Xi, Masilamani Jeyakumar, Phan Toan Thang Bioengineering, National University of Bioengineering Abstract Cartilage defects, such as
Chondrogenical Differentiation of Umbilical Cord Lining Stem Cells He Xi, Masilamani Jeyakumar, Phan Toan Thang Bioengineering, National University of Bioengineering Abstract Cartilage defects, such as
Running protein gels and detection of proteins
 Running protein gels and detection of proteins 1. Protein concentration determination using the BIO RAD reagent This assay uses a colour change reaction to give a direct measurement of protein concentration.
Running protein gels and detection of proteins 1. Protein concentration determination using the BIO RAD reagent This assay uses a colour change reaction to give a direct measurement of protein concentration.
MEF Starter Nucleofector Kit
 page 1 of 7 MEF Starter Nucleofector Kit for Mouse Embryonic Fibroblasts (MEF) MEF display significant phenotypic variations which depend on the strain, the genetic background of the mice they are isolated
page 1 of 7 MEF Starter Nucleofector Kit for Mouse Embryonic Fibroblasts (MEF) MEF display significant phenotypic variations which depend on the strain, the genetic background of the mice they are isolated
Formation of Oriented Fibers Using Injection of PEO Solutions inside Electric Fields Defined by Two Parallel Suspended Electrodes
 06 (43)-AF:Modelo-AF 8/20/11 6:41 AM Page 122 Formation of Oriented Fibers Using Injection of PEO Solutions inside Electric Fields Defined by Two Parallel Suspended R. Furlan 1, J. A. M. Rosado 2, A. N.
06 (43)-AF:Modelo-AF 8/20/11 6:41 AM Page 122 Formation of Oriented Fibers Using Injection of PEO Solutions inside Electric Fields Defined by Two Parallel Suspended R. Furlan 1, J. A. M. Rosado 2, A. N.
Measurement of Soil Parameters by Using Penetrometer Needle Apparatus
 Vol.3, Issue.1, Jan-Feb. 2013 pp-284-290 ISSN: 2249-6645 Measurement of Soil Parameters by Using Penetrometer Needle Apparatus Mahmoud M. Abu zeid, 1 Amr M. Radwan, 2 Emad A. Osman, 3 Ahmed M.Abu-bakr,
Vol.3, Issue.1, Jan-Feb. 2013 pp-284-290 ISSN: 2249-6645 Measurement of Soil Parameters by Using Penetrometer Needle Apparatus Mahmoud M. Abu zeid, 1 Amr M. Radwan, 2 Emad A. Osman, 3 Ahmed M.Abu-bakr,
Rat Creatine Kinase MB isoenzyme,ck-mb ELISA Kit
 Rat Creatine Kinase MB isoenzyme,ck-mb ELISA Kit Catalog No: E0479r 96 Tests Operating instructions www.eiaab.com FOR RESEARCH USE ONLY; NOT FOR THERAPEUTIC OR DIAGNOSTIC APPLICATIONS! PLEASE READ THROUGH
Rat Creatine Kinase MB isoenzyme,ck-mb ELISA Kit Catalog No: E0479r 96 Tests Operating instructions www.eiaab.com FOR RESEARCH USE ONLY; NOT FOR THERAPEUTIC OR DIAGNOSTIC APPLICATIONS! PLEASE READ THROUGH
BNG 331 Cell-Tissue Material Interactions. Biomaterial Surfaces
 BNG 331 Cell-Tissue Material Interactions Biomaterial Surfaces Course update Updated syllabus Homework 4 due today LBL 5 Friday Schedule for today: Chapter 8 Biomaterial surface characterization Surface
BNG 331 Cell-Tissue Material Interactions Biomaterial Surfaces Course update Updated syllabus Homework 4 due today LBL 5 Friday Schedule for today: Chapter 8 Biomaterial surface characterization Surface
Overview. Creation of 3D printed phantoms for clinical radiation therapy 7/7/2015. Eric Ehler, PhD Assistant Professor University of Minnesota
 Creation of 3D printed phantoms for clinical radiation therapy Eric Ehler, PhD Assistant Professor University of Minnesota ehler 046@umn.edu Overview Background of 3D Printing Practical Information Current
Creation of 3D printed phantoms for clinical radiation therapy Eric Ehler, PhD Assistant Professor University of Minnesota ehler 046@umn.edu Overview Background of 3D Printing Practical Information Current
When the fluid velocity is zero, called the hydrostatic condition, the pressure variation is due only to the weight of the fluid.
 Fluid Statics When the fluid velocity is zero, called the hydrostatic condition, the pressure variation is due only to the weight of the fluid. Consider a small wedge of fluid at rest of size Δx, Δz, Δs
Fluid Statics When the fluid velocity is zero, called the hydrostatic condition, the pressure variation is due only to the weight of the fluid. Consider a small wedge of fluid at rest of size Δx, Δz, Δs
INVESTIGATION OF VISCOELASTICITY AND CURE SHRINKAGE IN AN EPOXY RESIN DURING PROCESSING
 TH INTERNATIONAL CONFERENCE ON COMPOSITE MATERIALS INVESTIGATION OF VISCOELASTICITY AND CURE SHRINKAGE IN AN EPOXY RESIN DURING PROCESSING T. Shimizu *, H. Koinuma, K. Nagai Mitsubishi Heavy Industries,
TH INTERNATIONAL CONFERENCE ON COMPOSITE MATERIALS INVESTIGATION OF VISCOELASTICITY AND CURE SHRINKAGE IN AN EPOXY RESIN DURING PROCESSING T. Shimizu *, H. Koinuma, K. Nagai Mitsubishi Heavy Industries,
Inc. Wuhan. Quantity Pre-coated, ready to use 96-well strip plate 1 Plate sealer for 96 wells 4 Standard (liquid) 2
 Uscn Life Science Inc. Wuhan Website: www.uscnk.com Phone: +86 27 84259552 Fax: +86 27 84259551 E-mail: uscnk@uscnk.com ELISA Kit for Human Prostaglandin E1(PG-E1) Instruction manual Cat. No.: E0904Hu
Uscn Life Science Inc. Wuhan Website: www.uscnk.com Phone: +86 27 84259552 Fax: +86 27 84259551 E-mail: uscnk@uscnk.com ELISA Kit for Human Prostaglandin E1(PG-E1) Instruction manual Cat. No.: E0904Hu
Related topics: Application Note 27 Data Analysis of Tube Formation Assays.
 Tube Formation Assays in µ-slide Angiogenesis Related topics: Application Note 27 Data Analysis of Tube Formation Assays. Contents 1. General Information... 1 2. Material... 2 3. Work Flow Overview...
Tube Formation Assays in µ-slide Angiogenesis Related topics: Application Note 27 Data Analysis of Tube Formation Assays. Contents 1. General Information... 1 2. Material... 2 3. Work Flow Overview...
Manual for simulation of EB processing. Software ModeRTL
 1 Manual for simulation of EB processing Software ModeRTL How to get results. Software ModeRTL. Software ModeRTL consists of five thematic modules and service blocks. (See Fig.1). Analytic module is intended
1 Manual for simulation of EB processing Software ModeRTL How to get results. Software ModeRTL. Software ModeRTL consists of five thematic modules and service blocks. (See Fig.1). Analytic module is intended
Induction of Enzyme Activity in Bacteria:The Lac Operon. Preparation for Laboratory: Web Tutorial - Lac Operon - submit questions
 Induction of Enzyme Activity in Bacteria:The Lac Operon Preparation for Laboratory: Web Tutorial - Lac Operon - submit questions I. Background: For the last week you explored the functioning of the enzyme
Induction of Enzyme Activity in Bacteria:The Lac Operon Preparation for Laboratory: Web Tutorial - Lac Operon - submit questions I. Background: For the last week you explored the functioning of the enzyme
Canine creatine kinase MB isoenzyme (CK-MB)ELISA Kit
 Canine creatine kinase MB isoenzyme (CK-MB)ELISA Kit Catalog No. CSB-E15852c (96T) This immunoassay kit allows for the in vitro quantitative determination of canine CK-MB concentrations in serum and plasma.
Canine creatine kinase MB isoenzyme (CK-MB)ELISA Kit Catalog No. CSB-E15852c (96T) This immunoassay kit allows for the in vitro quantitative determination of canine CK-MB concentrations in serum and plasma.
ISOLATION AND PROPERTIES OF SECRETORY GRANULES FROM RAT ISLETS OF LANGERHANS. II. Ultrastructure of the Beta Granule
 ISOLATION AND PROPERTIES OF SECRETORY GRANULES FROM RAT ISLETS OF LANGERHANS II Ultrastructure of the Beta Granule MARIE H GREIDER, S L HOWELL, and P E LACY From the Department of Pathology, Washington
ISOLATION AND PROPERTIES OF SECRETORY GRANULES FROM RAT ISLETS OF LANGERHANS II Ultrastructure of the Beta Granule MARIE H GREIDER, S L HOWELL, and P E LACY From the Department of Pathology, Washington
Structural Integrity Analysis
 Structural Integrity Analysis 1. STRESS CONCENTRATION Igor Kokcharov 1.1 STRESSES AND CONCENTRATORS 1.1.1 Stress An applied external force F causes inner forces in the carrying structure. Inner forces
Structural Integrity Analysis 1. STRESS CONCENTRATION Igor Kokcharov 1.1 STRESSES AND CONCENTRATORS 1.1.1 Stress An applied external force F causes inner forces in the carrying structure. Inner forces
Zimmer DeNovo NT Natural Tissue Graft
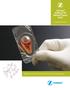 Zimmer DeNovo NT Natural Tissue Graft Surgical Technique Biologic treatment for early intervention and cartilage repair. Overview DeNovo NT Natural Tissue Graft (Fig. 1) is an off-the-shelf human tissue,
Zimmer DeNovo NT Natural Tissue Graft Surgical Technique Biologic treatment for early intervention and cartilage repair. Overview DeNovo NT Natural Tissue Graft (Fig. 1) is an off-the-shelf human tissue,
Aerobic Count. Interpretation Guide. 3M Food Safety 3M Petrifilm Aerobic Count Plate
 3M Food Safety 3M Petrifilm Aerobic Count Plate Aerobic Count Interpretation Guide The 3M Petrifilm Aerobic Count (AC) Plate is a ready-made culture medium system that contains Standard Methods nutrients,
3M Food Safety 3M Petrifilm Aerobic Count Plate Aerobic Count Interpretation Guide The 3M Petrifilm Aerobic Count (AC) Plate is a ready-made culture medium system that contains Standard Methods nutrients,
ID@GT prepared by Gabe Landes for T. Purdy 2009
 Rapid prototyping is the automatic construction of physical objects using solid freeform fabrication. The first techniques for rapid prototyping became available in the late 1980s and were used to produce
Rapid prototyping is the automatic construction of physical objects using solid freeform fabrication. The first techniques for rapid prototyping became available in the late 1980s and were used to produce
Western Blotting. Prepare samples:
 Western Blotting Sive Lab Protocol March 2007 Prepare samples: For zebrafish embryos: Option 1: Take live embryos and put into 1.5 ml tube with E3. Centrifuge gently for 1-2 minutes -yolk lipids will rise
Western Blotting Sive Lab Protocol March 2007 Prepare samples: For zebrafish embryos: Option 1: Take live embryos and put into 1.5 ml tube with E3. Centrifuge gently for 1-2 minutes -yolk lipids will rise
Preparation and Characterization of Gelatin-Poly(vinyl alcohol) Hydrogels for Three-Dimensional Cell Culture
 J. Ind. Eng. Chem., Vol. 13, No. 1, (2007) 116-120 Preparation and Characterization of Gelatin-Poly(vinyl alcohol) Hydrogels for Three-Dimensional Cell Culture Su Jung You*, Woong Schick Ahn*, Hong Seok
J. Ind. Eng. Chem., Vol. 13, No. 1, (2007) 116-120 Preparation and Characterization of Gelatin-Poly(vinyl alcohol) Hydrogels for Three-Dimensional Cell Culture Su Jung You*, Woong Schick Ahn*, Hong Seok
INTERNATIONAL HOCKEY FEDERATION PERFORMANCE REQUIREMENTS AND TEST PROCEDURES FOR HOCKEY BALLS. Published: April 1999
 INTERNATIONAL HOCKEY FEDERATION PERFORMANCE REQUIREMENTS AND TEST PROCEDURES FOR HOCKEY BALLS Published: April 1999 CONTENTS 1 Introduction 2 Test Procedures General 3 Standards and Specific Test Procedures
INTERNATIONAL HOCKEY FEDERATION PERFORMANCE REQUIREMENTS AND TEST PROCEDURES FOR HOCKEY BALLS Published: April 1999 CONTENTS 1 Introduction 2 Test Procedures General 3 Standards and Specific Test Procedures
Rat Creatine Kinase MB Isoenzyme (CKMB) ELISA
 Rat Creatine Kinase MB Isoenzyme (CKMB) ELISA For the quantitative determination of rat CKMB in serum, plasma, tissue homogenates and other biological fluids. Cat. No. KT-12247 For Research Use Only. Not
Rat Creatine Kinase MB Isoenzyme (CKMB) ELISA For the quantitative determination of rat CKMB in serum, plasma, tissue homogenates and other biological fluids. Cat. No. KT-12247 For Research Use Only. Not
Transfection reagent for visualizing lipofection with DNA. For ordering information, MSDS, publications and application notes see www.biontex.
 METAFECTENE FluoR Transfection reagent for visualizing lipofection with DNA For ordering information, MSDS, publications and application notes see www.biontex.com Description Cat. No. Size METAFECTENE
METAFECTENE FluoR Transfection reagent for visualizing lipofection with DNA For ordering information, MSDS, publications and application notes see www.biontex.com Description Cat. No. Size METAFECTENE
Module 1 : Conduction. Lecture 5 : 1D conduction example problems. 2D conduction
 Module 1 : Conduction Lecture 5 : 1D conduction example problems. 2D conduction Objectives In this class: An example of optimization for insulation thickness is solved. The 1D conduction is considered
Module 1 : Conduction Lecture 5 : 1D conduction example problems. 2D conduction Objectives In this class: An example of optimization for insulation thickness is solved. The 1D conduction is considered
TransformAid Bacterial Transformation Kit
 Home Contacts Order Catalog Support Search Alphabetical Index Numerical Index Restriction Endonucleases Modifying Enzymes PCR Kits Markers Nucleic Acids Nucleotides & Oligonucleotides Media Transfection
Home Contacts Order Catalog Support Search Alphabetical Index Numerical Index Restriction Endonucleases Modifying Enzymes PCR Kits Markers Nucleic Acids Nucleotides & Oligonucleotides Media Transfection
HARVESTING AND CRYOPRESERVATION OF HUMAN EMBRYONIC STEM CELLS (hescs)
 HARVESTING AND CRYOPRESERVATION OF HUMAN EMBRYONIC STEM CELLS (hescs) OBJECTIVE: can be cryopreserved in a liquid nitrogen (LN 2 ) freezer for long-term storage. This Standard Operating Procedure (SOP)
HARVESTING AND CRYOPRESERVATION OF HUMAN EMBRYONIC STEM CELLS (hescs) OBJECTIVE: can be cryopreserved in a liquid nitrogen (LN 2 ) freezer for long-term storage. This Standard Operating Procedure (SOP)
Table of Content. Enzymes and Their Functions Teacher Version 1
 Enzymes and Their Functions Jeisa Pelet, Cornell University Carolyn Wilczynski, Binghamton High School Cornell Learning Initiative in Medicine and Bioengineering (CLIMB) Table of Content Title Page Abstract..
Enzymes and Their Functions Jeisa Pelet, Cornell University Carolyn Wilczynski, Binghamton High School Cornell Learning Initiative in Medicine and Bioengineering (CLIMB) Table of Content Title Page Abstract..
NCL Method ITA-6. Leukocyte Proliferation Assay
 NCL Method ITA-6 Leukocyte Proliferation Assay Nanotechnology Characterization Laboratory National Cancer Institute-Frederick SAIC-Frederick Frederick, MD 21702 (301) 846-6939 ncl@mail.nih.gov December
NCL Method ITA-6 Leukocyte Proliferation Assay Nanotechnology Characterization Laboratory National Cancer Institute-Frederick SAIC-Frederick Frederick, MD 21702 (301) 846-6939 ncl@mail.nih.gov December
GASTRIC ORGANOID CULTURE PROTOCOL
 GASTRIC ORGANOID CULTURE PROTOCOL THIS PROTOCOL PROVIDES THE PROCEDURE FOR SUBCULTURING NORMAL HUMAN GASTRIC ORGANOIDS WHICH WAS DERIVED FROM THE SUBMERGED METHOD AS DESCRIBED IN BARKER N, ET AL. LGR5+VE
GASTRIC ORGANOID CULTURE PROTOCOL THIS PROTOCOL PROVIDES THE PROCEDURE FOR SUBCULTURING NORMAL HUMAN GASTRIC ORGANOIDS WHICH WAS DERIVED FROM THE SUBMERGED METHOD AS DESCRIBED IN BARKER N, ET AL. LGR5+VE
JBS FUNDAMENT Thermofluor Screen
 Cat. No. CS-330 Amount 1 Kit For in vitro use only. Quality guaranteed for 3 months. Store at 4 C. Application Screen for thermal stability of proteins as a function of the FUNDAMENTAL variables ph and
Cat. No. CS-330 Amount 1 Kit For in vitro use only. Quality guaranteed for 3 months. Store at 4 C. Application Screen for thermal stability of proteins as a function of the FUNDAMENTAL variables ph and
Tissue Train Culture System
 TECH REPORT 100: Tissue Train Culture System A method for culture and mechanical loading of cells in a 3D matrix Authors: Albert J. Banes, Ph.D., Jie Qi, Ph.D., David S. Anderson B.S., P.E., Melissa Maloney
TECH REPORT 100: Tissue Train Culture System A method for culture and mechanical loading of cells in a 3D matrix Authors: Albert J. Banes, Ph.D., Jie Qi, Ph.D., David S. Anderson B.S., P.E., Melissa Maloney
RAGE. Plugs for RAGE/PFGE
 1 RAGE Rotating Field Gel Electrophoresis (RAGE) is a variation on Pulsed Field Gel Electrophoresis (PFGE) and gives similar results. We use equipment that was only briefly marketed by Stratagene, at far
1 RAGE Rotating Field Gel Electrophoresis (RAGE) is a variation on Pulsed Field Gel Electrophoresis (PFGE) and gives similar results. We use equipment that was only briefly marketed by Stratagene, at far
P. Lu, Sh. Huang and K. Jiang
 416 Rev. Adv. Mater. Sci. 33 (2013) 416-422 P. Lu, Sh. Huang and K. Jiang NUMERICAL ANALYSIS FOR THREE-DIMENSIONAL BULK METAL FORMING PROCESSES WITH ARBITRARILY SHAPED DIES USING THE RIGID/VISCO-PLASTIC
416 Rev. Adv. Mater. Sci. 33 (2013) 416-422 P. Lu, Sh. Huang and K. Jiang NUMERICAL ANALYSIS FOR THREE-DIMENSIONAL BULK METAL FORMING PROCESSES WITH ARBITRARILY SHAPED DIES USING THE RIGID/VISCO-PLASTIC
TECHNICAL BULLETIN. HIS-Select Nickel Affinity Gel. Catalog Number P6611 Storage Temperature 2 8 C
 HIS-Select Nickel Affinity Gel Catalog Number P6611 Storage Temperature 2 8 C TECHNICAL BULLETIN Product Description HIS-Select Nickel Affinity Gel is an immobilized metalion affinity chromatography (IMAC)
HIS-Select Nickel Affinity Gel Catalog Number P6611 Storage Temperature 2 8 C TECHNICAL BULLETIN Product Description HIS-Select Nickel Affinity Gel is an immobilized metalion affinity chromatography (IMAC)
MTT Cell Proliferation Assay
 ATCC 30-1010K Store at 4 C This product is intended for laboratory research purposes only. It is not intended for use in humans, animals or for diagnostics. Introduction Measurement of cell viability and
ATCC 30-1010K Store at 4 C This product is intended for laboratory research purposes only. It is not intended for use in humans, animals or for diagnostics. Introduction Measurement of cell viability and
Workshop 14-16 February 2006
 Theoretical and practical approaches of Hepatocyte primary culture Workshop 14-16 February 2006 Lecture (2) Disaggregation & purification of target cells Coarse organizer Dr. Abo bakr Mohamed Eltayeb General
Theoretical and practical approaches of Hepatocyte primary culture Workshop 14-16 February 2006 Lecture (2) Disaggregation & purification of target cells Coarse organizer Dr. Abo bakr Mohamed Eltayeb General
Notes on Polymer Rheology Outline
 1 Why is rheology important? Examples of its importance Summary of important variables Description of the flow equations Flow regimes - laminar vs. turbulent - Reynolds number - definition of viscosity
1 Why is rheology important? Examples of its importance Summary of important variables Description of the flow equations Flow regimes - laminar vs. turbulent - Reynolds number - definition of viscosity
Characterization and Quantification of Lyophilized Product Appearance and Structure
 Characterization and Quantification of Lyophilized Product Appearance and Structure Dr Kevin R. Ward B.Sc. Ph.D. MRSC Director of Research & Development Biopharma Technology Ltd. Winchester SO23 0LD, UK
Characterization and Quantification of Lyophilized Product Appearance and Structure Dr Kevin R. Ward B.Sc. Ph.D. MRSC Director of Research & Development Biopharma Technology Ltd. Winchester SO23 0LD, UK
Getting rid of the wires: Curved Layer Fused Deposition Modeling in Conductive Polymer Additive Manufacturing
 Getting rid of the wires: Curved Layer Fused Deposition Modeling in Conductive Polymer Additive Manufacturing DIEGEL, Olaf 1, a, SINGAMNENI, Sarat 1, b, HUANG, Ben 1, c, GIBSON, Ian 2, d 1 Centre for Rapid
Getting rid of the wires: Curved Layer Fused Deposition Modeling in Conductive Polymer Additive Manufacturing DIEGEL, Olaf 1, a, SINGAMNENI, Sarat 1, b, HUANG, Ben 1, c, GIBSON, Ian 2, d 1 Centre for Rapid
Economic Mass Producible Mirror Panels for Solar Concentrators
 Abstract G, G. Burgess, K. Lovegrove and A. Luzzi Centre for Sustainable Energy Systems Australian National University, Canberra 2 Australia E-mail: Glen@faceng.anu.edu.au The Centre for Sustainable Energy
Abstract G, G. Burgess, K. Lovegrove and A. Luzzi Centre for Sustainable Energy Systems Australian National University, Canberra 2 Australia E-mail: Glen@faceng.anu.edu.au The Centre for Sustainable Energy
α-cyclodextrin SYNONYMS α-schardinger dextrin, α-dextrin, cyclohexaamylose, cyclomaltohexaose, α- cycloamylase
 α-cyclodextrin New specifications prepared at the 57th JECFA (2001) and published in FNP 52 Add 9 (2001). An ADI not specified was established at the 57th JECFA (2001). SYNONYMS α-schardinger dextrin,
α-cyclodextrin New specifications prepared at the 57th JECFA (2001) and published in FNP 52 Add 9 (2001). An ADI not specified was established at the 57th JECFA (2001). SYNONYMS α-schardinger dextrin,
Biological Stability and Activity of sirna in Ionic Liquids**
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Supporting Information Biological Stability and Activity of sirna in Ionic Liquids** Romiza R.
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Supporting Information Biological Stability and Activity of sirna in Ionic Liquids** Romiza R.
MEF Nucleofector Kit 1 and 2
 page 1 of 7 MEF Nucleofector Kit 1 and 2 for Mouse Embryonic Fibroblasts (MEF) MEF display significant phenotypic variations which depend on the strain, the genetic background of the they are isolated
page 1 of 7 MEF Nucleofector Kit 1 and 2 for Mouse Embryonic Fibroblasts (MEF) MEF display significant phenotypic variations which depend on the strain, the genetic background of the they are isolated
Cellartis Protocol Culturing of hes Cells
 Cellartis Protocol Culturing of hes Cells This material was cultured and frozen using Cellartis protocols. WiCell recommends that stem cells should be thawed and established in the conditions in which
Cellartis Protocol Culturing of hes Cells This material was cultured and frozen using Cellartis protocols. WiCell recommends that stem cells should be thawed and established in the conditions in which
The EpiOcular Model. Protocol, Performance and Experience
 The EpiOcular Model Protocol, Performance and Experience Rodger Curren, Ph.D., John Harbell, Ph.D. and Kevin Trouba, Ph.D. Institute for In Vitro Sciences, Inc. EpiOcular Outline Background Protocol Performance
The EpiOcular Model Protocol, Performance and Experience Rodger Curren, Ph.D., John Harbell, Ph.D. and Kevin Trouba, Ph.D. Institute for In Vitro Sciences, Inc. EpiOcular Outline Background Protocol Performance
Atomic Force Microscope and Magnetic Force Microscope Background Information
 Atomic Force Microscope and Magnetic Force Microscope Background Information Lego Building Instructions There are several places to find the building instructions for building the Lego models of atomic
Atomic Force Microscope and Magnetic Force Microscope Background Information Lego Building Instructions There are several places to find the building instructions for building the Lego models of atomic
Trace Gas Exchange Measurements with Standard Infrared Analyzers
 Practical Environmental Measurement Methods Trace Gas Exchange Measurements with Standard Infrared Analyzers Last change of document: February 23, 2007 Supervisor: Charles Robert Room no: S 4381 ph: 4352
Practical Environmental Measurement Methods Trace Gas Exchange Measurements with Standard Infrared Analyzers Last change of document: February 23, 2007 Supervisor: Charles Robert Room no: S 4381 ph: 4352
PROCESS PARTICLE COUNTER (PPC) SENSOR/CONTROLLER FOR OPTIMIZING POWER RECOVERY EXPANDER AND GAS TURBINE PERFORMANCE
 PROCESS PARTICLE COUNTER (PPC) SENSOR/CONTROLLER FOR OPTIMIZING POWER RECOVERY EXPANDER AND GAS TURBINE PERFORMANCE APPLICATIONS NOTE FOR MEASUREMENTS AT THE ENTRANCE AND EXIT OF A THIRD STAGE SEPARATOR
PROCESS PARTICLE COUNTER (PPC) SENSOR/CONTROLLER FOR OPTIMIZING POWER RECOVERY EXPANDER AND GAS TURBINE PERFORMANCE APPLICATIONS NOTE FOR MEASUREMENTS AT THE ENTRANCE AND EXIT OF A THIRD STAGE SEPARATOR
Custom Design of Knee Joint Prosthesis By Using Computerized Tomography (CT) Images and 3D Modelling
 Custom Design of Knee Joint Prosthesis By Using Computerized Tomography (CT) Images and 3D Modelling Qasem Alyazji Near East University, Department of Biomedical Engineering Nicosia, North Cyprus, Turkey
Custom Design of Knee Joint Prosthesis By Using Computerized Tomography (CT) Images and 3D Modelling Qasem Alyazji Near East University, Department of Biomedical Engineering Nicosia, North Cyprus, Turkey
BRIEFING 661.2 Plastic Packaging Systems for Pharmaceutical Use.
 BRIEFING 661.2 Plastic Packaging Systems for Pharmaceutical Use. USP proposes the revision and development of a suite of plastic packaging system standards in the current issue of PF. General test chapter
BRIEFING 661.2 Plastic Packaging Systems for Pharmaceutical Use. USP proposes the revision and development of a suite of plastic packaging system standards in the current issue of PF. General test chapter
Precision Miniature Load Cell. Models 8431, 8432 with Overload Protection
 w Technical Product Information Precision Miniature Load Cell with Overload Protection 1. Introduction The load cells in the model 8431 and 8432 series are primarily designed for the measurement of force
w Technical Product Information Precision Miniature Load Cell with Overload Protection 1. Introduction The load cells in the model 8431 and 8432 series are primarily designed for the measurement of force
AGAROSE GEL ELECTROPHORESIS:
 AGAROSE GEL ELECTROPHORESIS: BEST PRACTICES (BACK TO THE BASICS) Unit of Tropical Laboratory Medicine April 2009 Marcella Mori WORKFLOW OF AGAROSE GEL ELECTROPHORESIS: THREE STEPS Agarose gel electrophoresis
AGAROSE GEL ELECTROPHORESIS: BEST PRACTICES (BACK TO THE BASICS) Unit of Tropical Laboratory Medicine April 2009 Marcella Mori WORKFLOW OF AGAROSE GEL ELECTROPHORESIS: THREE STEPS Agarose gel electrophoresis
Cell Phone Radiation Effects on Cancer. Tyler Barkich Central Catholic High School Grade 11
 Cell Phone Radiation Effects on Cancer Tyler Barkich Central Catholic High School Grade 11 Cancer Overview Cancer cells are cells that grow and divide at an irregular, unregulated pace. Apoptosis does
Cell Phone Radiation Effects on Cancer Tyler Barkich Central Catholic High School Grade 11 Cancer Overview Cancer cells are cells that grow and divide at an irregular, unregulated pace. Apoptosis does
INJECTION BLOW MOLDING WITH FDM
 INJECTION BLOW MOLDING WITH FDM 3D PRODUCTION SYSTEMS Time Required Cost Skill Level By Susan Sciortino, Stratasys Inc. OVERVIEW Blow molding is a manufacturing process in which air pressure inflates heated
INJECTION BLOW MOLDING WITH FDM 3D PRODUCTION SYSTEMS Time Required Cost Skill Level By Susan Sciortino, Stratasys Inc. OVERVIEW Blow molding is a manufacturing process in which air pressure inflates heated
Fluid Mechanics: Static s Kinematics Dynamics Fluid
 Fluid Mechanics: Fluid mechanics may be defined as that branch of engineering science that deals with the behavior of fluid under the condition of rest and motion Fluid mechanics may be divided into three
Fluid Mechanics: Fluid mechanics may be defined as that branch of engineering science that deals with the behavior of fluid under the condition of rest and motion Fluid mechanics may be divided into three
ab185915 Protein Sumoylation Assay Ultra Kit
 ab185915 Protein Sumoylation Assay Ultra Kit Instructions for Use For the measuring in vivo protein sumoylation in various samples This product is for research use only and is not intended for diagnostic
ab185915 Protein Sumoylation Assay Ultra Kit Instructions for Use For the measuring in vivo protein sumoylation in various samples This product is for research use only and is not intended for diagnostic
Rigid-Flex Technology: Mainstream Use but More Complex Designs by John Isaac October 1, 2007
 Rigid-Flex Technology: Mainstream Use but More Complex Designs by John Isaac October 1, 2007 In the past, flex and rigid-flex technology was typically used in applications that could tolerate long design
Rigid-Flex Technology: Mainstream Use but More Complex Designs by John Isaac October 1, 2007 In the past, flex and rigid-flex technology was typically used in applications that could tolerate long design
DURABILITY OF MORTAR LININGS IN DUCTILE IRON PIPES Durability of mortar linings
 DURABILITY OF MORTAR LININGS IN DUCTILE IRON PIPES Durability of mortar linings I. S. MELAND SINTEF Civil and Environmental Engineering, Cement and Concrete, Trondheim, Norway Durability of Building Materials
DURABILITY OF MORTAR LININGS IN DUCTILE IRON PIPES Durability of mortar linings I. S. MELAND SINTEF Civil and Environmental Engineering, Cement and Concrete, Trondheim, Norway Durability of Building Materials
THE THERMAL FLOW METER, A GAS METER FOR ENERGY MEASUREMENT
 THE THERMAL FLOW METER, A GAS METER FOR ENERGY MEASUREMENT Kazuto Otakane, Tokyo Gas Co., Ltd Katsuhito Sakai, Tokyo Gas Co., Ltd Minoru Seto, Tokyo Gas Co., Ltd 1. INTRODUCTION Tokyo Gas s new gas meter,
THE THERMAL FLOW METER, A GAS METER FOR ENERGY MEASUREMENT Kazuto Otakane, Tokyo Gas Co., Ltd Katsuhito Sakai, Tokyo Gas Co., Ltd Minoru Seto, Tokyo Gas Co., Ltd 1. INTRODUCTION Tokyo Gas s new gas meter,
STANDARD OPERATING PROCEDURE
 STANDARD OPERATING PROCEDURE Title: Antibody Production at Strategic Diagnostics Inc. SOP#: M-119 Version #: 1 Date Approved: August 6, 2009 Author: Strategic Diagnostic Inc. Date Modified: 1. PURPOSE
STANDARD OPERATING PROCEDURE Title: Antibody Production at Strategic Diagnostics Inc. SOP#: M-119 Version #: 1 Date Approved: August 6, 2009 Author: Strategic Diagnostic Inc. Date Modified: 1. PURPOSE
THE PSEUDO SINGLE ROW RADIATOR DESIGN
 International Journal of Mechanical Engineering and Technology (IJMET) Volume 7, Issue 1, Jan-Feb 2016, pp. 146-153, Article ID: IJMET_07_01_015 Available online at http://www.iaeme.com/ijmet/issues.asp?jtype=ijmet&vtype=7&itype=1
International Journal of Mechanical Engineering and Technology (IJMET) Volume 7, Issue 1, Jan-Feb 2016, pp. 146-153, Article ID: IJMET_07_01_015 Available online at http://www.iaeme.com/ijmet/issues.asp?jtype=ijmet&vtype=7&itype=1
National Food Safety Standard Food microbiological examination: Aerobic plate count
 National Food Safety Standard of the People s Republic of China GB4789.2-2010 National Food Safety Standard Food microbiological examination: Aerobic plate count Issued by 2010-03-26 Implemented by 2010-06-01
National Food Safety Standard of the People s Republic of China GB4789.2-2010 National Food Safety Standard Food microbiological examination: Aerobic plate count Issued by 2010-03-26 Implemented by 2010-06-01
